Abstract
Potamodromous fish are considered important indicators of habitat connectivity in freshwater ecosystems, but they are globally threatened by anthropogenic impacts. Hence, non-invasive techniques are necessary for monitoring during spawning migrations. The use of environmental DNA (eDNA) potentially facilitates these efforts, albeit quantitative examinations of spawning migrations remain so far mostly uncharted. Here, we investigated spawning migrations of Danube bleak, Alburnus mento, and Vimba bream, Vimba vimba, and found a strong correlation between daily visual fish counts and downstream eDNA signals obtained from filtered water samples analysed with digital PCR and end-point PCR coupled with capillary electrophoresis. By accounting for daily discharge fluctuations, it was possible to predict eDNA signal strength from the number of migrating fish: first, the whole spawning reach was taken into account. Second, the model was validated using eDNA signals and fish counts obtained from the upper half of the examined river stretch. Consequently, fish counts and their day-to-day changes could be described via an eDNA-based time series model for the whole migration period. Our findings highlight the capability of eDNA beyond delivering simple presence/absence data towards efficient and informative monitoring of highly dynamic aquatic processes such as spawning migrations of potamodromous fish species.
Similar content being viewed by others
Introduction
Generally, three main types of fish migration patterns are distinguished according to the type of water body inhabited: while diadromous and oceanodromous migrations commonly receive more attention due to the economic relevance of taxa such as salmon, tuna, and eel, the importance of potamodromous fish migrating solely within freshwaters has been frequently overlooked1. Nevertheless, these species can be key for the functioning of river ecosystems by influencing nutrient cycling and energy transfer between ecosystems2. Main drivers for potamodromous migrations are feeding, refuge-seeking (e.g. overwintering), and spawning1. The continuity of a river, associated with the possibility to reach suitable habitats, is crucial for maintaining sustainable populations of these mostly iteroparous species, characterised by multiple non-lethal spawning migrations during their lifetime3,4. In regions such as Europe, where 74% of river stretches are classified as “strongly affected” by channel fragmentation and water flow regulation5, tracing and quantifying the remaining spawning migrations of potamodromous fish species is highly efficient for monitoring aquatic ecosystem connectivity and the status of these often-threatened species.
As it is of particular importance to minimise disturbance at this sensitive stage of the life cycle1, numerous conventional methods such as electrofishing, mark-recapture, tags (incl. telemetry), trapping, netting via fish-counting fences or determining stable isotope ratios in fish tissues are not suitable due to their invasiveness6. Thus, visual observations from outside the river or by snorkelling are often considered the best approach to monitor fish spawning migrations in rivers1. With the ongoing increasing use of DNA-based methods in the field of ecology, a new powerful approach has emerged for studies of aquatic ecosystems7. The investigation of aquatic species via environmental DNA (eDNA) – DNA fragments deposited into the water through faeces, mucus, or spawning products and extracted via precipitation or filtration from water samples – can be advantageous in many aspects8. eDNA analysis can be carried out with minimal disturbance to detect individual species via diagnostic end-point PCR or real-time qPCR9,10, or to assess whole fish communities via metabarcoding11. Furthermore, rare or invasive fish species are more likely to be detected via eDNA compared to conventional approaches12,13 and economic advantages such as lower costs14 and lower sampling efforts15,16 have been previously highlighted.
When eDNA is captured and analysed in riverine systems, downstream transport coupled with discharge and other environmental variables must be taken into account8,17. Transport is positively correlated with river size17 with maximum detection distances below 1 km in small streams9,18 and between 3 and 60 km in large rivers (discharge >3 m³/s)17,19. The transport distance is also influenced by the riverbed through the DNA retention properties of different substrate types20,21 and finally, increased discharge results in higher dilutions of eDNA22. Nevertheless, the eDNA signal was found to vanish within one day after the removal of source organisms from lotic systems18,23. Apart from monitoring fish communities in rivers, the potential of eDNA for evaluating fish migrations in these systems has been recently recognised and used to confirm the proper functioning of fish ladders16 and upstream migration of endangered sturgeons24. Besides the detection of fish species, there have also been attempts to relate eDNA concentrations to species abundance or biomass showing that the molecular approach does in general have the potential to reflect these parameters25,26,27. In rivers, eDNA copy number has been found to positively correlate with visual fish observations28,29, fish biomass in cages18, and electrofishing surveys30,31.
Despite these encouraging findings, eDNA is rarely used to monitor fish spawning migrations: for example, upstream migrating European anadromous shads were recently detected via eDNA32, and spawning activity of Australian Macquarie perch was estimated by comparing ratios of nuclear and mitochondrial eDNA33. Regarding the quantitative assessment of spawning migrations, case studies on Bigheaded carps, sockeye salmon and the sea lamprey find a positive correlation between adult fish numbers and eDNA signal strength22,34,35. However, the direct correlation between fish numbers, daily eDNA concentrations, and discharge has only been established recently for salmon migrations at an artificial weir36. Prior to this, semelparity, i.e. dead salmon releasing approximately ten times more eDNA compared to living individuals, partly obstructed the direct predictions of adult fish numbers35.
The aim of the present study was to evaluate the potential of eDNA for quantitative monitoring of the spawning migration patterns of the protected potamodromous and iteropoarous fish species Alburnus mento (Danube bleak), and the co-migrating species Vimba vimba (Vimba bream)37 in the Natura 2000 protected region “Mondsee-Attersee” (Austria, see Fig. 1). Artificially modified parts of the river bottom and banks (Supplementary Information 1) could hinder protected A. mento from reaching suitable spawning areas. Therefore, regular quantitative monitoring of migrating individuals is key to evaluate future population development. The two species were jointly counted every day throughout their spawning season and water samples for eDNA analyses were collected at the downstream end and in the middle of the examined river stretch. First, we evaluated the general relationship between fish counts and eDNA signals taking into account the influence of fish located upstream of the water sampling points and the mean daily discharge. Second, we tested if the two datasets follow the same pattern over time, i.e. show the same day-to-day fluctuations in the course of a spawning season. Finally, an eDNA-based time series model was tested for its potential to describe fish counts over time in order to assess the possibility of solely eDNA-based quantitative monitoring of spawning migrations in the future.
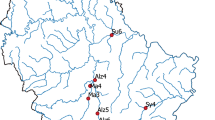
Overview of the Natura 2000 region “Mondsee-Attersee” (Austria) with the river Zeller Ache on the north-eastern tip of lake Mondsee; investigated river sections of the Zeller Ache: I: outflow-section, II: section with paved riverbed and bank, III: preferred spawning ground in previous years, IV and VI: restructured river sections, VII: most distant section from outflow with impassable dam on upstream end, V: non-accessible section (ca. 100 m) (see Supplementary Information 1 for detailed description of the river sections). Arrows indicate locations of eDNA water sampling and physicochemical measurements (point 1 and 2); the dot marks the location of the automated discharge measurement station. The asterisk shows the location of the Research Department for Limnology, of the University of Innsbruck (ILIM). This map was modified from Google Maps (Google Maps. Mondsee, Austria. Retrieved October 8, 2018, from https://goo.gl/maps/817sNygimuThE58t7 (n.d.)) using Adobe Photoshop C2 Version 9.0.
Results
eDNA signals, spawning migration, and fish counts
Weak eDNA signals (1.3–5.3 copies/µl extract) of both A. mento and V. vimba were detected via digital PCR between the 19th April and 12th May 2017, with first detections via endpoint PCR coupled with capillary electrophoresis (further on “CE-PCR”) on the latter day followed by the first visual observations and a steep increase in eDNA signals on 13th May. The maximum number of A. mento and V. vimba individuals (further on “fish counts”) in the whole river was estimated as 8,625 on 17th and 18th May (see Materials and Methods for details on fish counts). At the upper end of the examined reach (section VII), less than 10 individuals were observed on three sampling days. Most spawning activity occurred at the river’s downstream end plus in the middle (sections I and III; Fig. 1). Generally, target eDNA concentrations were approx. twice as strong for A. mento (mean: 2,464 copies/µl extract ±5,747 SD; max: 39,473 copies/µl extract) compared to V. vimba (mean: 1,031 copies/µl extract ±2,176 SD; max: 15,525 copies/µl extract). This difference was not detectable in CE-PCR (A. mento: mean: 1.48 relative fluorescence units (RFU) ± 1.13 SD; max: 3.20 RFU; V. vimba: mean: 1.36 RFU ± 1.13 SD; max: 3.49 RFU).
Relationship between eDNA signals and fish counts
During the actual spawning migration (13th May to 21st July), eDNA signals (digital PCR and CE-PCR) from samples taken at the downstream end of the Zeller Ache were best described by a model including fish counts obtained from the whole examined river stretch (sections I to VII; 1.1 km) and the effect of mean daily discharge (model DD3: R² = 0.64; model EP3: R² = 0.71; Tables 1 and 2, and Fig. 2). Fish counts were positively related to target eDNA signals obtained via the two molecular approaches (digital PCR: parameter estimate = 3.66; P < 0.001; CE-PCR: parameter estimate = 0.72; P < 0.001; from DD3 and EP3 in Table 3). The optimum model derived from fish counts in section III and upstream and water samples taken at point 2, i.e. the downstream end of section III (Table 3: EP4; R² = 0.58; based on CE-PCR and the discharge corrected sum of mean fish counts), did not differ significantly from the best performing model derived for the whole river stretch (EP3), which was fitted onto this dataset (R² = 0.58; Fig. 2).
Relationship between eDNA concentrations/signals and discharge corrected fish counts. The left panel displays eDNA concentrations (copies/µl extract) obtained at sampling point 1 and discharge corrected fish counts of the whole river stretch plus model DD3. The middle panel shows RFUs obtained at sampling point 1 and discharge corrected fish counts of the whole river stretch combined with model EP3; 95% confidence areas of the models are depicted in grey. The right panel displays eDNA signals (RFU) obtained from sampling point 2 plotted against the sum of mean fish counts of section III and upstream (discharge corrected). The black line depicts EP3 fitted to the displayed data (R² = 0.58). The red, dashed line shows the optimum model obtained from this data (sub)set (EP4), which remains inside the confidence limits of model EP3 for the entire data range. X-axes of the middle and right panels are scaled logarithmically.
Characteristics of target eDNA concentration and fish counts over time
The analysis of both target eDNA concentrations (digital PCR) and fish counts over time (see Materials and Methods for data transformations and included discharge effect) showed that the two time series were highly correlated (rs = 0.91) across the whole investigation period (Fig. 3). Cross-correlation between eDNA signals and fish counts was highest when the two time series were not lagged (cross-correlation = 0.82; but see Supplementary Fig. 1 for cross-correlations at different lags), meaning that simultaneous day-to-day changes showed the highest similarity between the two datasets. The observed cross-correlation was significant, as it lay outside the 95% confidence interval generated from 1,000 cross-correlation values from random sorting of the two time series. A closer investigation of daily trends showed that eDNA concentrations and fish counts were changing along the same direction on 62 out of 70 days (both eDNA and fish counts >0), which was significantly different from a 1:1 ratio (Chi² = 22.69; P < 0.001). Furthermore, both time series showed significant auto-correlation i.e. influence of previous values (eDNA: Chi² = 39.26, P < 0.001; fish counts: Chi² = 50.67, P < 0.001).
Time series of target eDNA concentration obtained via digital PCR (upper panel) and fish counts (lower panel). To obtain comparable amplitudes between the two time series, fish counts were summed up for the whole river stretch and divided by discharge; the resulting values were thereafter individually multiplied by five. Gaps denote days without measurements, which were filled by averaging neighbouring values for time series analysis. The dotted lines indicate the start of the actual spawning season on 13th May characterised by first visual observations and a steep increase in eDNA signals.
Time series modelling
To evaluate the potential of solely eDNA-based monitoring for spawning migrations, the relationship between target eDNA concentration (digital PCR) and fish counts was evaluated via time series modelling in three steps: first, we determined the optimum ARIMA model for describing eDNA concentrations. Second, we tested whether the same ARIMA model structure (order, differencing, and order of the moving-average model) was suitable to describe the fish count data. Third, we evaluated whether actual daily fish counts could be described by the optimum eDNA-based ARIMA model. eDNA concentrations were best described via an ARIMA model ((2, 0, 0); AIC = 1,885.45) taking into account two lagged values, i.e. eDNA signals from the two previous days (order). This model was further characterised by a mean average error of 2,930.86 and a first-order auto-correlation coefficient of 0.0004 (Table 4). It presented a good fit (based on standardised residuals and non-significant Ljung-Box statistics at lags 1 to 10) to the measured eDNA concentrations. These lay outside the 95% confidence area only on six days; five were characterised by large signal amplitudes and one by rapid change in eDNA concentration (Fig. 4a). The ARIMA model structure was subsequently applied to the fish count time series to test whether target eDNA concentration and fish counts could be described in the same way. Generally, a sub-optimal fit of the ARIMA model structure to fish count data was indicated by two significant Ljung-Box statistics (lags 4 and 6), a higher first-order auto-correlation coefficient of 0.14, and a higher AIC of 1,890.64. Nevertheless, visual inspection (Fig. 4b) showed a good fit between modelled and measured fish counts: fish count values lay outside the 95% confidence area only on seven days (six at large signal amplitudes; one at a drastic day-to-day change; Fig. 4b). Finally, the eDNA-based ARIMA model was compared directly to the obtained fish counts: the latter lay inside the 95% confidence area of the model except for seven days, which were all characterised by high fish counts (Fig. 4c).
Time series models in comparison to measured target eDNA concentration and fish counts. (a) The ARIMA (2, 0, 0)-modelled eDNA concentration (grey line) and 95% confidence area (blue shading) in comparison to measured eDNA concentration (blue line). (b) Modelled fish counts using the same ARIMA model structure (grey line; red shading for 95% confidence area) in comparison to the actual fish counts (red line). (c) ARIMA-modelled eDNA concentrations (grey line and blue shading) in comparison to actual fish counts (red line). Values outside the 95% confidence area are marked as black dots, eDNA concentration is based on the sum of Alburnus mento and Vimba vimba (copies/µl extract) from point 1 samples, and fish counts are the sum of mean counts from all river sections divided by discharge; to enable direct comparison between time series, these values were additionally multiplied by five.
Discussion
Our study substantiates the applicability of eDNA for the investigation of potamodromous fish spawning migrations not only in terms of presence/absence data, but also concerning quantitative approximations. As expected, there was a positive correlation between eDNA concentrations and visual fish counts including parallel changes in both parameters on a day-to-day basis. Besides fish individuals present in close proximity to the water sampling points, fish from further upstream (up to 1.1 km) significantly contributed to the detected eDNA signals. Increased mean daily discharge negatively influenced eDNA concentration and model fit was substantially improved when it was taken into account. The relationship between discharge corrected fish counts of the whole river stretch and eDNA signals (CE-PCR-based) from its downstream end could also describe the situation in the upper half of the river. Concerning the changes of both eDNA concentrations and fish counts during the spawning season, the same time series model structure was capable to depict both eDNA concentrations and fish counts within the 95% confidence area for 94% of the examination period. Additionally, the eDNA-based time series model was suitable to describe fish counts directly during the whole field study, except for days with highest fish numbers, on which the model slightly underestimated the height of the fish count spikes. This demonstrates the high potential of quantitative eDNA analysis to monitor spawning migrations of potamodromous fish species under known discharge conditions.
The general positive correlation between eDNA signals and the number of fish commencing spawning migrations confirmed results of previous studies on Bigheaded carps, sockeye salmon, and sea lampreys22,34,35. As the present study used visual fish counts, it was possible to relate eDNA signals to the whole adult A. mento and V. vimba population present in the examined river stretch similar to salmon counts at an artificial weir36. Additionally, spawning has no lethal effect on the examined iteroparous species A. mento and V. vimba37,38 omitting the effect of dead individuals, which can significantly influence eDNA levels35. Albeit a positive correlation between visual observations and eDNA signals has been previously observed28,39, factors such as discharge and the distribution of target organisms in the river need to be considered to correctly describe this relationship35,36,40. Our model comparison indicated a substantial contribution of fish individuals from further upstream (up to 1.1 km) to target eDNA concentrations at the downstream end of the examined reach. The role of eDNA deposition could not be directly assessed in the Zeller Ache based on the obtained measurements and during preliminary data analysis only a very small sedimentation rate of 1% per 100 m (compared to 50% within 500–1,000 m based on previous studies9,17) showed a favourable effect on model fit (Supplementary Method 1). Hence, it was deemed unjustifiable to include eDNA sedimentation in the modelling process at all. Explanations for this are provided by the structure of the riverbed and the distribution of migrating fish individuals: rows of large boulders in sections IV and VI induced high turbulence and partially coarse substrate in sections I, III, and VII hindered eDNA deposition21. Additionally, the riverbed in section II (~250 m), connecting the two sections with highest fish counts, was fully paved, which potentially blocks the entire eDNA deposition process (Supplementary Information 1).
Apart from influencing downstream transport distance, higher discharge potentially leads to lower eDNA signals via dilution18,36 and was found to be critical for a meaningful quantitative interpretation of eDNA signals emitted from migrating salmon36. Potamodromous fish/cyprinids usually migrate only a few kilometres to suitable spawning grounds in medium sized rivers, which are characterised by high discharge fluctuations during spring and summer3. In our study, the mean daily discharge of the Zeller Ache varied tenfold during the spawning period from 0.25 to 2.5 m³/s. Therefore, the necessary inclusion of discharge in the models best describing the relation between fish counts and eDNA signals was not surprising. To evaluate the applicability of a model best describing the situation in the Zeller Ache to similar spawning migrations, its performance was tested on a subsection of the river (section III and upstream) based on independent eDNA samples and ~60% of the total fish counts. The model obtained from the whole river stretch did not differ significantly from the optimum model estimated for this “subset” (Fig. 2). Albeit fish counts in the comparison were not independent, this indicates that the model is applicable in similar situations concerning fish counts, discharge, and size of a spawning reach.
Other factors influencing eDNA decomposition and leading to reduced detectability include higher water temperatures, UV-B radiation, acidic pH, and water sample storage prior to filtration41,42,43. Fortunately, fast downstream transport and the proximity between migrating fish and water sampling points in our study should have minimised the influence of the above mentioned factors, which exhibit negative affects after hours or days23,42. Water sample storage was previously found to negatively affect eDNA persistence43,44, but the close proximity between sampling points and the laboratory used for filtration (Fig. 1) enabled sample processing within ~1.5 h. Additionally, water samples were always taken in the same order and the time until filtration remained constant throughout the study. Hence, eventual DNA decomposition prior to filtering can be considered similar for all samples.
In samples taken during the two weeks prior to the start of the spawning migration, very low levels of A. mento and V. vimba eDNA were detected via digital PCR (1.3–5.3 copies/µl extract). These could stem either from individual adult fish probing the lowest section of the Zeller Ache before the onset of the actual spawning migration or from populations with unknown spawning patterns in Lake Irrsee (Fig. 1) on the upper end of the Zeller Ache45. eDNA signals from upstream are, however, unlikely to have a general influence on the obtained data as the upper end of the examined spawning reach and Irrsee are separated by ~6.3 km and 16 impassable transverse structures. Additionally, low flow velocities at the outflow of Irrsee render this short part of the river inexpedient for spawning of A. mento and V. Vimba45.
Time series analysis of eDNA concentrations obtained via digital PCR and fish counts showed that both datasets follow the same pattern during the whole examination period including similar day-to-day changes and influence of values measured on two previous days. The eDNA-based ARIMA model onlystruggled to incorporate the highest fish counts within the 95% confidence area (Fig. 4c). This discrepancy could be caused by the categorical estimates of fish numbers (see Materials and Methods). To minimise subjective perception, two skilled experts did the estimations simultaneously and classifying fish abundance in six categories was easily possible. However, category size increased with increasing fish numbers and estimations were less precise at higher numbers (Fig. 2 left panel, Fig. 4c). Despite this limitation, we considered visual observations most appropriate for fish counts in the examined situation1, especially since the size of the Zeller Ache (wetted width: ~5 m; depth: 0.2–1.5 m) coupled with low turbidity throughout the spawning season enabled reliable fish counts. On the four days when the water was not completely clear, turbidity was still low enough to estimate fish numbers. Nevertheless, A. mento and V. vimba were hardly distinguishable from outside of the water, resulting in joint fish counts and the potential for bias in case the two species release eDNA at different rates. As both are cyprinids of the same average size (~25 cm), inhabit Alpine foreland lakes, and display the same activity level during spawning38, species-specific eDNA emission rates are an unlikely bias in the present study. Furthermore, A. mento and V. vimba constitute over 90% of all migrating fish during spring and early summer in the Zeller Ache37, making other species, such as the rheophile Salmo trutta, negligible in the examined situation. Additionally, the third and fourth most abundant species (Rutilus meidingeri and Squalius cephalus) could be excluded due to their much larger size and earlier spawning of the former37. Finally, fish present in the unobservable section V could have biased fish counts, but due to its short length (100 m) and comparably low fish counts in section IV below (16% of total counts) the influence should be minute.
Throughout the whole investigation period, it was not possible to quantify actual spawning activity. Hence, milt, eggs, and hatched larvae potentially affect the correlation between eDNA concentrations and fish counts. The influence of milt and eggs should be minute in the examined situation as spermatozoa primarily contain nuclear DNA33 and milt degrades within hours in mesocosms46. Furthermore, the eggs of A. mento and V. vimba stick to the substrate4 and the number of drifting eggs could so far not be correlated with eDNA signal strength34. Hatched fish larvae are readily detectable via eDNA as demonstrated for burrowing lampreys47 and potentially influence species-specific eDNA signals. However, the larvae of A. mento are known to migrate downstream soon after hatching38 and high flow velocities prevailing in the river should lead to a fast downstream transport. When comparing eDNA concentrations to fish counts, additional peaks or inflated peak heights induced by milt, eggs, and larvae were not detected. Only the height of two small peaks in the second half of the examination period points to the influence of hatched larvae, as fish counts did not follow this pattern or were less high on these two occasions.
We applied two molecular detection systems based on species-specific primers and digital PCR or CE-PCR, the first permitting absolute quantification of target DNA copy number48, the second enabling semi-quantitative estimations (Supplementary Method 2). The applied primers amplify short fragments (≤200 bp) and were subjected to extensive specificity testing to exclude the possibility of non-target DNA amplification in central Europe at the reported PCR conditions49 (Supplementary Method 2). A direct comparison between digital PCR and CE-PCR confirmed the higher sensitivity of digital PCR and showed reliable positive CE-PCR detections from more than 25 (V. vimba) and 42 (A. mento) copies per µl extract, which was sufficient for the amplification of eDNA stemming from ~11 fish individuals present in and above section III (600 m) at 2.44 m³/s discharge. In practice, this sensitivity difference only played a role for samples taken before the onset of the actual spawning migration, some of which tested positive in digital PCR but not in CE-PCR. The high eDNA levels detected during the actual spawning migration also resulted in a saturation effect in CE-PCR50 and similar signal strengths for A. mento and V. vimba (Supplementary Method 2), whilst absolute eDNA concentrations were higher for A. mento, corresponding to fish trap data collected in the Zeller Ache in 201037. Concerning the general relation between fish counts and eDNA signals, the saturated CE-PCR in this special case coincided with less precise fish counts in higher categories and led to smaller dispersion in CE-PCR-based models (Fig. 2).
Two factors warranting additional discussion of water sample processing and molecular analysis are DNA cross-contamination and PCR inhibition, both of which frequently affect eDNA-based studies9,44. We down-corrected eDNA signals of samples affected by cross-contamination via filtering equipment by the respective RFUs or copy numbers of the six filtration controls, all of which showed weak signals (<0.23 RFU; <1.32 copies/µl) in comparison to affected field samples (>2.1 RFU; >1,100 copies/µl). Thus, the influence of DNA contamination was likely removed from the data set without completely dropping affected samples and avoiding data gaps in time series analysis. The DNA contaminations most likely stem from the water filtration process, as negative controls added during DNA extraction and PCRs all resulted negative. These slight cross-contaminations occurred despite rigorous cleaning procedures (see Materials and Methods). However, only sodium hypochlorite concentrations of 3.6% were available contrasting to the 6% commonly used in US laboratories44. Future eDNA studies would definitely benefit from the use of encapsulated filters and higher concentrations of sodium hypochlorite and/or longer incubation times44,51. With regard to PCR inhibition, additional clean up steps were not applied during sample processing. Nevertheless, BSA, known to reduce PCR inhibition52, was included in all CE-PCR assays and the applied extraction method performs well with problematic samples such as plants or tissues with a high lipid content53. Finally, plant litter and associated humic substances, previously found to hinder successful amplification of eDNA18,52, were not present throughout the study period.
Our study demonstrates the positive correlation between eDNA concentration and fish counts on a day-to-day basis during spawning migrations of potamodromous fish. In the medium-sized Zeller Ache and its 1.1 km long spawning reach, downstream eDNA signals were defined by fish individuals present in this whole stretch and changes in the daily discharge significantly affected the relationship between eDNA levels and fish counts, indicating a strong dilution effect associated with increased water run-off. Finally, the eDNA-based time series model allowed approximating fish counts for almost the entire examination period. As such, our findings highlight the capability of eDNA beyond simple presence/absence investigations towards efficient and informative monitoring of dynamic aquatic processes and support future conservation efforts within the scope of the water framework directive and habitats directive of the EU.
Materials and Methods
Study site
The river Zeller Ache is the main spawning ground of A. mento and V. vimba, two spring- and early summer-spawning cyprinid species migrating from May until the end of July out of the Upper-Austrian lake Mondsee (N47°48′56.8″ E13°22′54.8″) upstream into the river Zeller Ache for reproduction until their path is blocked by an impassable dam after around 1.1 km (Fig. 1)37. The Zeller Ache (38.3 km² catchment area) has a mean discharge of 1.98 m³/s and flows ~7.4 km from lake Irrsee into Mondsee with a height drop of 73 m54. The wetted width in the examined reach was ~5 m and depth ranged from 0.2 to 1.5 m. Due to the proximity to villages and streets, it is heavily modified with stone built river beds or banks and numerous transverse structures45 leading to higher flow velocities and fewer opportunities to rest (Supplementary Information 1). All field work along the Zeller Ache was carried out between 19th April and 21st July 2017 (94 days) between the inflow into Mondsee and an impassable dam around 1.1 km upstream. This reach was separated into seven sections based on structural differences of the riverbed (e.g. natural or artificially paved river bottom, known or unknown use for spawning in former seasons; Fig. 1, Supplementary Information 1). River section V (100 m) could not be accessed as it is completely bounded by private properties and buildings.
Visual fish counts and environmental parameters
In each section, upstream migrating A. mento and V. vimba were visually counted on a daily basis from the riverside (if accessible) and/or from bridges (except days 5, 71, and 79). Observations were carried out by two skilled experts moving upstream within approx. one hour and mostly in the morning between 8 am and 12 noon. Per section, only five to 10 min were used for fish estimations to exclude double counts by upstream migrating individuals. Fish were counted categorically in each section (Cat. 1 = 1–5 individuals, Cat. 2 = 5–10 ind., Cat. 3 = 10–50 ind., Cat. 4 = 50–100 ind., Cat. 5 = 100–500 ind., Cat. 6 ≥ 500 ind.) whereby A. mento and V. vimba were grouped together as these two species are hard to distinguish from outside of the waterbody. Due to the high numbers and movement of the migrating fish, it was not possible to obtain exact fish counts from visual observations. To account for larger estimate errors associated with increasing individual numbers, it was furthermore necessary to increase category sizes with increasing numbers.
eDNA sampling and processing
Water for eDNA analysis was taken daily (except days 5, 12, 28, 36, 40, 66, 67, 71, 72, and 79) in wide neck bottles with a volume of 2 L and an opening diameter of 50 mm. These were treated with chlorine bleach (3.6 g sodium hypochlorite per 100 g liquid) overnight and thoroughly washed with fish-DNA-free tap water prior to use. One water sample was taken at point 1 (downstream end of section I) and point 2 (downstream end of section III; Fig. 1) in the middle of the river at 5–10 cm depth and bottles were plunged at arm length against the current. Fresh, DNA-free gloves were used for handling of each water sample, which were stored and transported within two hours to the Research Department for Limnology (Mondsee, Austria; Fig. 1) for further processing. After water sampling, the water temperature, pH, oxygen saturation and electric conductivity were measured with a portable multi meter HQ40d (HACH) at point 1 and 2. Water temperature ranged from 6.8 to 24 °C with 13 °C at the beginning of migration activity; pH ranged between 8.23 and 8.81. Due to time constraints, turbidity was only visually classified into “low”, “medium”, and “high”, and found to be low to medium with clear visibility of the river bottom except for days 20, 21, and 88 (before the beginning and at the end of the spawning migration). Data on the daily mean discharge (m³/s) during the investigation period were collected at section VII (Fig. 1) at a fixed measurement station and provided by the department of surface water management Upper Austria. Discharge ranged from 0.25 to 4.79 m³/s (thunderstorm induced peak on the 9th May; day 22) and never exceeded 0.5 m³/s between 18th June and 12th July. Above the measurement point, the Zeller Ache has no inflow until the upper end of section VII; below the measurement point, the river has two small inflows with approximate widths of 0.5 and 1.5 m; their influence was neglected for all further calculations.
On arrival at the institute, samples were vacuum-filtered through 47 mm glass fibre filters (1.2 µm mesh width; Whatman GF/C) using a water-jet pump (BRAND). Between samples, the filter equipment was decontaminated for about 15 min in chlorine bleach (0.36 g sodium hypochlorite per 100 g liquid) and thoroughly washed with fish-DNA-free tap water. On every second to third day, a negative control (2 L MilliQ water) was filtered before and after the field samples of the respective day. Filters were folded with DNA-free, flamed forceps and stored separately in 1.5 ml reaction tubes at −20 °C until DNA extraction. Every day, lab surfaces were cleaned with bleach and ethanol (70%) prior to filtering and DNA-free gloves were worn at all times. For further processing, filters were transferred in a cooling box to a clean room laboratory at the University of Innsbruck (Austria) within three hours. Lysis, DNA extraction, amplification, visualisation, and quantification using digital PCR and CE-PCR were carried out there (Supplementary Method 2). This was accompanied by a test with dilution series confirming the suitability of the latter approach for semi-quantitative estimations of target DNA in PCR. Additionally, absolute target DNA concentrations (digital PCR) and eDNA signal strength (CE-PCR) were directly compared for field samples obtained from point 1 (Supplementary Method 2).
Analysis
All statistical analysis and calculations were carried out with R (R Development Core Team 2017) using packages and functions described in Supplementary Method 3. To make fish counts comparable with the obtained eDNA signals, the mean number of fish of the respective categories were used (e.g. mean of Cat 1 (1–5 individuals) = 2.5). For Cat 6 (≥500 individuals) a maximum of 5,000 fish was estimated resulting in a mean value of 2,750 individuals. Fish counts of A. mento and V. vimba could not be carried out separately, hence, the eDNA concentrations (digital PCR) and signal strengths (obtained via CE-PCR and measured in RFUs) of A. mento and V. vimba were added up.
The absolute quantification (target DNA copies/µl extract) resulting from digital PCR permits the direct comparison of eDNA concentration to fish counts via linear models. To enable this comparison for eDNA signal strength obtained from CE-PCR, logarithmic models were necessary. For these, both independent (mean fish counts) and dependent (eDNA signal) variables were elevated by 1. Only data obtained between the 13th May and 21st July during the actual spawning migration of A. mento and V. vimba were used for model comparison to remove the bias of zero values and extremely low values obtained before the actual onset of the spawning migration. We compared different models (Table 1) for their descriptive performance based on ordinal ranking of the AICc, ∆AICc, and the weights of the AICc values55. For both molecular methods (digital PCR and CE-PCR), three models were compared using the same combinations of mean fish counts and discharge to explain eDNA signals at sampling point 1 (Table 1). In models DD1 and EP1, only fish counts from the most downstream section of the Zeller Ache (closest to point 1) were used. Models DD2 and EP2 included the sum of mean fish counts from all observed river sections. In models DD3 and EP3, mean fish counts from all river sections were additionally divided by the mean daily discharge. We refrained from the inclusion of a distance effect, after initial data analysis (Supplementary Method 1). In a next step, an additional logarithmic model (EP4) was calculated based on CE-PCR derived eDNA signals at sampling point 2 and the sum of mean fish counts from sections III and upstream (60% of total fish counted) divided by the daily discharge. The fit to this data “subset” was compared between model EP4 and the best performing model from the ordinal ranking.
To compare fish counts and target eDNA concentrations (digital PCR) in time series analysis, it was necessary to transform fish counts in a way to obtain similar fluctuation range and scaling for both data sets. Therefore, the value obtained by dividing mean fish counts by mean daily discharge was additionally multiplied by five. For missing values in both series, approximations were calculated using the mean of the previous and following value. At first, correlation and cross-correlation between the two time series was examined by calculating the Spearman correlation rs and the normalised cross correlation. To test whether the obtained cross-correlation was significant, the fish counts and associated target eDNA concentrations were 1,000 times randomly sorted, the maximum correlation stored for each of the calculations, and a 95% confidence interval calculated (lower bound: 0.24; upper bound: 0.25). Each series was also separately tested for auto-correlation using the “Ljung-Box”-test. In a next step, we explored if fish counts and eDNA concentration change along the same direction from day to day: days without fish counts and eDNA signals were excluded from this analysis. To model eDNA concentration over time, an ARIMA model was calculated. Model fit was examined based on standardised residuals, auto-correlation, Ljung-Box statistic and the confidence intervals of coefficients. Finally, the obtained ARIMA-model was used to model fish counts; then, fit between modelled and measured eDNA signals and fish counts was compared by determining whether the actual values were located within the confidence area of the modelled time series.
Data availability
All data on fish counts, eDNA analysis, and abiotic parameters are available on Figshare; https://doi.org/10.6084/m9.figshare.9868193.v1.
References
Lucas, M. C. & Baras, E. Migration of Freshwater Fishes. xiii–xiv, 66–84, and 230–270 (Blackwell Science Ltd, 2001).
Pennuto, C. M., Cudney, K. A. & Janik, C. E. Fish invasion alters ecosystem function in a small heterotrophic stream. Biol. Invasions 20, 1033–1047 (2018).
Jungwirth, M., Schmutz, S. & Weiss, S. Fish Migration and Fish Bypasses. 438 (Blackwell Science Ltd, 1998).
Wootton, R. J. Ecology of Teleost Fishes . Fish and Fisheries Series 24. 2 Ed., 87–106, 141–143, and 259–283 (Kluwer Academic Publishers, 1998).
Nilsson, C., Reidy, C. A., Dynesius, M. & Revenga, C. Fragmentation and flow regulation of the world’s large river systems. Science 308, 405–408, https://doi.org/10.1126/science.1107887 (2005).
Hill, D., Fasham, M., Tucker, G., Shewry, M. & Shaw, P. Handbook of Biodiversity Methods: Survey, Evaluation and Monitoring. 368–386 (Cambridge University Press, 2005).
Blanchet, S. The use of molecular tools in invasion biology: an emphasis on freshwater ecosystems. Fish Manag Ecol 19, 120–132, https://doi.org/10.1111/j.1365-2400.2011.00832.x (2012).
Rees, H. C., Maddison, B. C., Middleditch, D. J., Patmore, J. R. M. & Gough, K. C. The detection of aquatic animal species using environmental DNA - a review of eDNA as a survey tool in ecology. J Appl Ecol 51, 1450–1459, https://doi.org/10.1111/1365-2664.12306 (2014).
Wilcox, T. M. et al. Understanding environmental DNA detection probabilities: a case study using a stream-dwelling char Salvelinus fontinalis. Biol Cons 194, 209–216, https://doi.org/10.1016/j.biocon.2015.12.023 (2016).
Davison, P. I. et al. Laboratory and field validation of a simple method for detecting four species of non-native freshwater fish using eDNA. J Fish Biol 89, 1782–1793, https://doi.org/10.1111/jfb.13086 (2016).
Hänfling, B. et al. Environmental DNA metabarcoding of lake fish communities reflects long-term data from established survey methods. Mol Ecol 25, 3101–3119, https://doi.org/10.1111/mec.13660 (2016).
Balasingham, K. D., Walter, R. P., Mandrak, N. E. & Heath, D. D. Environmental DNA detection of rare and invasive fish species in two Great Lakes tributaries. Mol Ecol 27, 112–127, https://doi.org/10.1111/mec.14395 (2018).
Jerde, C. L., Mahon, A. R., Chadderton, W. L. & Lodge, D. M. “Sight-unseen” detection of rare aquatic species using environmental DNA. Conserv Lett 4, 150–157, https://doi.org/10.1111/j.1755-263X.2010.00158.x (2011).
Lacoursiere-Roussel, A., Cote, G., Leclerc, V. & Bernatchez, L. Quantifying relative fish abundance with eDNA: a promising tool for fisheries management. J Appl Ecol 53, 1148–1157, https://doi.org/10.1111/1365-2664.12598 (2016).
Sigsgaard, E. E., Carl, H., Moller, P. R. & Thomsen, P. F. Monitoring the near-extinct European weather loach in Denmark based on environmental DNA from water samples. Biol Cons 183, 46–52, https://doi.org/10.1016/j.biocon.2014.11.023 (2015).
Yamanaka, H. & Minamoto, T. The use of environmental DNA of fishes as an efficient method of determining habitat connectivity. Ecol Indic 62, 147–153, https://doi.org/10.1016/j.ecolind.2015.11.022 (2016).
Pont, D. et al. Environmental DNA reveals quantitative patterns of fish biodiversity in large rivers despite its downstream transportation. Sci Rep 8, 10361, https://doi.org/10.1038/s41598-018-28424-8 (2018).
Jane, S. F. et al. Distance, flow and PCR inhibition: eDNA dynamics in two headwater streams. Mol Ecol Res 15, 216–227, https://doi.org/10.1111/1755-0998.12285 (2015).
Deiner, K. & Altermatt, F. Transport distance of invertebrate environmental DNA in a natural river. PLoS One 9, e88786, https://doi.org/10.1371/journal.pone.0088786 (2014).
Jerde, C. L. et al. Influence of stream bottom substrate on retention and transport of vertebrate environmental DNA. Environ Sci Technol 50, 8770–8779, https://doi.org/10.1021/acs.est.6b01761 (2016).
Shogren, A. J. et al. Controls on eDNA movement in streams: Transport, retention, and resuspension. Sci Rep 7, 5065, https://doi.org/10.1038/s41598-017-05223-1 (2017).
Bracken, F. S. A., Rooney, S. M., Kelly-Quinn, M., King, J. J. & Carlsson, J. Identifying spawning sites and other critical habitat in lotic systems using eDNA “snapshots”: A case study using the sea lamprey Petromyzon marinus L. Ecol Evol 9, 553–567, https://doi.org/10.1002/ece3.4777 (2019).
Pilliod, D. S., Goldberg, C. S., Arkle, R. S. & Waits, L. P. Factors influencing detection of eDNA from a stream-dwelling amphibian. Mol Ecol Res 14, 109–116, https://doi.org/10.1111/1755-0998.12159 (2014).
Pfleger, M. O., Rider, S. J., Johnston, C. E. & Janosik, A. M. Saving the doomed: Using eDNA to aid in detection of rare sturgeon for conservation (Acipenseridae). Glob Ecol Conserv 8, 99–107, https://doi.org/10.1016/j.gecco.2016.08.008 (2016).
Klobucar, S. L., Rodgers, T. W. & Budy, P. At the forefront: evidence of the applicability of using environmental DNA to quantify the abundance of fish populations in natural lentic waters with additional sampling considerations. Can J Fish Aquat Sci 74, 2030–2034, https://doi.org/10.1139/cjfas-2017-0114 (2017).
Takahara, T., Minamoto, T., Yamanaka, H., Doi, H. & Kawabata, Z. Estimation of fish biomass using environmental DNA. PLoS One 7, e35868, https://doi.org/10.1371/journal.pone.0035868 (2012).
Thomsen, P. F. et al. Monitoring endangered freshwater biodiversity using environmental DNA. Mol Ecol 21, 2565–2573, https://doi.org/10.1111/j.1365-294X.2011.05418.x (2012).
Mahon, A. R. et al. Validation of eDNA surveillance sensitivity for detection of Asian carps in controlled and field experiments. PLoS One 8, e58316, https://doi.org/10.1371/journal.pone.0058316 (2013).
Uchii, K., Doi, H. & Minamoto, T. A novel environmental DNA approach to quantify the cryptic invasion of non-native genotypes. Mol Ecol Res 16, 415–422, https://doi.org/10.1111/1755-0998.12460 (2016).
Baldigo, B. P., Sporn, L. A., George, S. D. & Ball, J. A. Efficacy of environmental DNA to detect and quantify brook trout populations in headwater streams of the Adirondack Mountains, New York. T Am Fish Soc 146, 99–111, https://doi.org/10.1080/00028487.2016.1243578 (2017).
Thalinger, B. et al. Quantifizierung von Fischbeständen mittels eDNA in alpinen Fließgewässern. Wasserwirtschaft 2–3, 30–34, https://doi.org/10.1007/s35147-018-0009-z (2018).
Antognazza, C. M. et al. Environmental DNA as a non‐invasive sampling tool to detect the spawning distribution of European anadromous shads (Alosa spp.). Aquat Conserv 29, 148–152, https://doi.org/10.1002/aqc.3010 (2019).
Bylemans, J. et al. An environmental DNA-based method for monitoring spawning activity: a case study, using the endangered Macquarie perch (Macquaria australasica). Methods Ecol Evol 8, 646–655, https://doi.org/10.1111/2041-210x.12709 (2017).
Erickson, R. A. et al. Detecting the movement and spawning activity of bigheaded carps with environmental DNA. Mol Ecol Res 16, 957–965, https://doi.org/10.1111/1755-0998.12533 (2016).
Tillotson, M. D. et al. Concentrations of environmental DNA (eDNA) reflect spawning salmon abundance at fine spatial and temporal scales. Biol Cons 220, 1–11, https://doi.org/10.1016/j.biocon.2018.01.030 (2018).
Levi, T. et al. Environmental DNA for the enumeration and management of Pacific salmon. Mol. Ecol Res 19, 597–608, https://doi.org/10.1111/1755-0998.12987 (2019).
Csar, D. & Gumpinger, C. Die Migration der Fischfauna im Unterlauf von Seeache und Zeller Ache: unter besonderer Berücksichtigung der Natura 2000 Schutzgüter Perlfisch (Rutilus meidingeri) und Seelaube (Alburnus mento). (Technisches Büro für Gewässerökologie DI Clemens Gumpinger, Wels, Austria, 2010).
Kottelat, M. & Freyhof, J. Handbook of European Freshwater Fishes. 171 and 291–292 (Publ. Kottelat, 2007).
Doi, H. et al. Environmental DNA analysis for estimating the abundance and biomass of stream fish. Freshw Biol 62, 30–39, https://doi.org/10.1111/fwb.12846 (2017).
Wilcox, T. M. et al. Capture enrichment of aquatic environmental DNA: a first proof of concept. Mol Ecol Res 18, 1392–1401, https://doi.org/10.1111/1755-0998.12928 (2018).
Eichmiller, J. J., Best, S. E. & Sorensen, P. W. Effects of temperature and trophic state on degradation of environmental DNA in lake water. Environ Sci Tech 50, 1859–1867, https://doi.org/10.1021/acs.est.5b05672 (2016).
Strickler, K. M., Fremier, A. K. & Goldberg, C. S. Quantifying effects of UV-B, temperature, and pH on eDNA degradation in aquatic microcosms. Biol Cons 183, 85–92, https://doi.org/10.1016/j.biocon.2014.11.038 (2015).
Yamanaka, H. et al. On-site filtration of water samples for environmental DNA analysis to avoid DNA degradation during transportation. Ecol Res 31, 963–967, https://doi.org/10.1007/s11284-016-1400-9 (2016).
Goldberg, C. S. et al. Critical considerations for the application of environmental DNA methods to detect aquatic species. Methods Ecol Evol 7, 1299–1307, https://doi.org/10.1111/2041-210x.12595 (2016).
Gumpinger, C. Gewässerschutzbericht 44: Potentialstudie Salzkammergut: Ökologischer Zustand und Verbesserungsmöglichkeiten an den Zuflüssen von Mondsee und Irrsee (Technisches Büro für Gewässerökologie, Linz, Austria, 2011).
Lance, R. et al. Experimental observations on the decay of environmental DNA from bighead and silver carps. Manag Biol Invasion 8, 343–359, https://doi.org/10.3391/mbi.2017.8.3.08 (2017).
Gingera, T. D. et al. Detection and identification of lampreys in Great Lakes streams using environmental DNA. J Great Lakes Res 42, 649–659, https://doi.org/10.1016/j.jglr.2016.02.017 (2016).
Tsuji, S., Takahara, T., Doi, H., Shibata, N. & Yamanaka, H. The detection of aquatic macroorganisms using environmental DNA analysis-A review of methods for collection, extraction, and detection. Environmental DNA 1, 99–108, https://doi.org/10.1002/edn3.21 (2019).
Thalinger, B. et al. Molecular prey identification in Central European piscivores. Mol. Ecol Res 16, 123–137, https://doi.org/10.1111/1755-0998.12436 (2016).
Hunter, M. E. et al. Detection limits of quantitative and digital PCR assays and their influence in presence-absence surveys of environmental DNA. Mol Ecol Res 17, 221–229, https://doi.org/10.1111/1755-0998.12619 (2017).
Spens, J. et al. Comparison of capture and storage methods for aqueous macrobial eDNA using an optimized extraction protocol: advantage of enclosed filter. Methods Ecol Evol 8, 635–645, https://doi.org/10.1111/2041-210x.12683 (2017).
McKee, A. M., Spear, S. F. & Pierson, T. W. The effect of dilution and the use of a post-extraction nucleic acid purification column on the accuracy, precision, and inhibition of environmental DNA samples. Biol Cons 183, 70–76, https://doi.org/10.1016/j.biocon.2014.11.031 (2015).
Wallinger, C. et al. Evaluation of an automated protocol for efficient and reliable DNA extraction of dietary samples. Ecol Evol 7, 6382–6389, https://doi.org/10.1002/ece3.3197 (2017).
BMLFUW Abteilung IV/4 - Wasserhaushalt. Hydrografisches Jahrbuch von Österreich 2013: 121. Band - Daten und Auswertungen. 976 (Vienna, Austria, 2013).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach . 2 Ed. (Springer-Verlag New York, 2002).
Acknowledgements
This research was funded by a Swarovski research grant provided by the University of Innsbruck and awarded to JW, the Austria Research Promotion Agency (FFG; project number 853219), and the publishing fund of the University of Innsbruck . We are very grateful to Bernhard Teufl for supporting us during visual fish counts, Martina Nindl and Yannick Pütz for their extraordinary commitment, organisation skills, and support in the laboratory, and the department of surface water management Upper Austria, which provided us with discharge data of the Zeller Ache. Finally, we thank four anonymous reviewers for their constructive feedback on earlier versions of this manuscript.
Author information
Authors and Affiliations
Contributions
J.W. conceived the study; E.W. and J.W. were responsible for study design and execution regarding visual fish counts. B.T., E.W., M.T. and J.W. together set up eDNA sampling, which was carried out by E.W., B.T. and E.W. carried out the subsequent molecular work. All data were compiled by E.W. and analysed by B.T. who both drafted first versions of this manuscript, which was revised by J.W. and M.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thalinger, B., Wolf, E., Traugott, M. et al. Monitoring spawning migrations of potamodromous fish species via eDNA. Sci Rep 9, 15388 (2019). https://doi.org/10.1038/s41598-019-51398-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-51398-0
This article is cited by
-
Exploring uncharted territory: new frontiers in environmental DNA for tropical fisheries management
Environmental Monitoring and Assessment (2024)
-
Comparative study on nuclear and mitochondrial DNA of Ayu Plecoglossus altivelis for environmental DNA-based spawning evaluation
Landscape and Ecological Engineering (2023)
-
Detection of brook trout in spatiotemporally separate locations using validated eDNA technology
Journal of Environmental Studies and Sciences (2023)
-
On the relevance of animal behavior to the management and conservation of fishes and fisheries
Environmental Biology of Fishes (2023)
-
Highly sensitive environmental DNA detection of topmouth gudgeon, Pseudorasbora parva: a comparison of qPCR and microfluidic qdPCR
Biological Invasions (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.