Abstract
Strong evidence suggests that genetic variations in DNA methyltransferases (DNMTs) may alter the downstream expression and DNA methylation patterns of neuronal genes and influence cognition. This study investigates the association between a DNMT1 polymorphism, rs2162560, and chemotherapy-associated cognitive impairment (CACI) in a cohort of breast cancer patients. This is a prospective, longitudinal cohort study. From 2011 to 2017, 351 early-stage breast cancer patients receiving chemotherapy were assessed at baseline, the midpoint, and the end of chemotherapy. DNA was extracted from whole blood, and genotyping was performed using Sanger sequencing. Patients’ self-perceived cognitive function and cognitive performance were assessed at three different time points using FACT-Cog (v.3) and a neuropsychological battery, respectively. The association between DNMT1 rs2162560 and cognitive function was evaluated using logistic regression analyses. Overall, 33.3% of the patients reported impairment relative to baseline in one or more cognitive domains. Cognitive impairment was observed in various objective cognitive domains, with incidences ranging from 7.2% to 36.9%. The DNMT1 rs2162560 A allele was observed in 21.8% of patients and this was associated with lower odds of self-reported cognitive decline in the concentration (OR = 0.45, 95% CI: 0.25–0.82, P = 0.01) and functional interference (OR = 0.48, 95% CI: 0.24–0.95, P = 0.03) domains. No significant association was observed between DNMT1 rs2162560 and objective cognitive impairment. This is the first study to show a significant association between the DNMT1 rs2162560 polymorphism and CACI. Our data suggest that epigenetic processes could contribute to CACI, and further studies are needed to validate these findings.
Similar content being viewed by others
Introduction
Chemotherapy-associated cognitive impairment (CACI) is highly prevalent among cancer patients receiving chemotherapy1,2,3. Reports have shown that CACI negatively affects patients’ social functioning and quality of life3,4. The exact mechanism underlying CACI has yet to be elucidated. However, genetic factors are known to contribute to CACI5,6,7,8.
Epigenetic modifications such as DNA methylation can modify intermediate neuronal gene expression, leading to changes in cognitive performance9. DNA methylation is catalyzed by DNA methyltransferase (DNMT) enzymes and involves the addition of a methyl group to the 5′-position of cytosine bases, primarily at cytosine-phosphate-guanine (CpG) dinucleoside sites, which results in gene silencing10. In mammalian cells, DNMT1 is the most abundant form of DNA methyltransferases. It is primarily responsible for maintaining methylation and has a higher affinity for hemimethylated DNA11. DNMT-mediated DNA methylation regulates multiple aspects of neuronal development and function, with important roles in learning and memory12,13,14. Clinically, the dysregulated expression or aberrant function of DNMT1 could affect neurocognitive function by altering the methylation patterns of cognitive gene targets15,16. Recently, a prospective study reported cognitive decline is associated with DNA methylation of leukocytes in breast cancer patients receiving chemotherapy, providing evidence of epigenetic links to CACI17.
The extent of methylation is controlled by the expression and function of DNMT1, which can be altered by single nucleotide polymorphisms (SNPs) situated within its genetic code18. DNMT SNPs have been studied in Alzheimer’s disease and schizophrenia19,20. However, DNMT SNPs have not been investigated in CACI. Using a candidate gene approach, we identified the DNMT1 SNP rs2162560 from the literature19,20 and evaluated its association with CACI in a cohort of breast cancer patients. We hypothesize that carriers of the A allele of the rs2162560 polymorphism have increased DNA methylation activity that protects them from CACI.
Methods
Study design
This multicenter, prospective, longitudinal cohort study was conducted in Singapore between 2011 and 2017. This study was approved by the SingHealth Centralized Institutional Review Board. Written informed consent was obtained from all participants. All experiments were performed in accordance with relevant guidelines and regulations.
Participants
Patients were eligible to participate in this study if they fulfilled the following inclusion criteria: (i) at least 21 years old, (ii) diagnosed with stage I-IIIA breast cancer, (iii) scheduled for four cycles of anthracycline- or taxane-based chemotherapy, (iv) no history of chemotherapy or radiotherapy, and (v) read and understand English or Mandarin. Patients who were (i) symptomatically ill, (ii) diagnosed with brain metastasis or any neuropsychiatric illness that may cause cognitive impairment, or (iii) physically or mentally incapable of giving written informed consent were excluded.
Study procedure
The first time point (T1) occurred at baseline before the initiation of chemotherapy. The second time point (T2) was timed at 6 weeks after T1 and coincided with the first day of the third cycle of chemotherapy. The third time point (T3) was 12 weeks after T1 when the primary chemotherapy was completed. Overall, the approximate duration between each time point was 6 weeks. Patients completed both objective and subjective neuropsychological assessments and self-reported questionnaires to assess their health-related quality of life, fatigue, and anxiety. All tools were available in English and Chinese and were administered by trained bilingual interviewers.
Assessment tools
Subjective cognitive functioning was assessed using the Functional Assessment of Cancer Therapy – Cognitive Function (FACT-Cog) version 3, which is a patient-reported questionnaire that measures self-perceived cognitive impairment21. It evaluates the cognitive domains of mental acuity, concentration, memory, functional interference, verbal fluency, and multitasking. The domain scores are calculated by summing the individual domain items, and the FACT-Cog summation score is obtained by adding all item scores together. The English and Chinese versions of the FACT-Cog used in this study were previously validated and have demonstrated equivalence and reliability22.
Objective cognitive function was initially assessed using Headminder. As Headminder was commercially discontinued in 2014, the Cambridge Neuropsychological Test Automated Battery (CANTAB, Cambridge Cognition Ltd., UK) was used for objective cognitive assessment for the remainder of the study. Both neuropsychological tests are language-independent and computer-based. For both batteries, four cognitive domains were assessed: processing speed, response speed, memory, and attention. The specific measures used by Headminder and CANTAB as neuropsychological assessments are described in Supplementary Table 1. These tests have been validated and show sensitivity in capturing alterations in neuropsychological performance in the four cognitive domains23,24.
Anxiety, cancer-related fatigue, and insomnia are known confounders of cognition and were assessed by validated patient-reported questionnaires – the Beck Anxiety Inventory (BAI)25, the Brief Fatigue Inventory (BFI)26, and the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire C-30 (EORTC QLQ-C30)27, respectively.
Defining cognitive impairment
The overall impairment in self-perceived cognitive function was defined as a reduction in the FACT-Cog summation score by ≥10.6 points during (T2) or after chemotherapy (T3) compared with the baseline value. This reduction is based on the minimal clinically important difference (MCID) that we previously established as clinically significant28. The FACT-Cog MCID range is in line with a change of 5–10% in the EORTC-QLQ-C30 scales, which is proposed to be the minimal clinically significant change. For individual cognitive domains, patients were considered to be experiencing clinically significant cognitive impairment if their domain scores at T2 or T3 were ≥15% lower than their baseline score8.
Reliable change index (RCI) scores were calculated to assess changes in objective cognition. RCI was computed by calculating the difference of Headminder or CANTAB scores between baseline and T2 or T3, subtracting the mean practice effect extrapolated from a normative group and dividing by the standard error of difference. The RCI for both Headminder and CANTAB were calculated using two separate reference groups (Supplementary Table 2). Patients were classified as having impairment in each of the cognitive domains if the RCI score was ≤−1.529.
Genotyping
Upon recruitment, 10 mL of whole blood was drawn from each participant into an ethylenediaminetetraacetic acid (EDTA) tube. The samples were centrifuged at 2500 rpm for 10 minutes within 30 minutes of collection. The buffy coat was drawn and stored at −80 °C until analysis. Genomic DNA was extracted from the buffy coat using the QIAamp DNA Blood Mini Kit (Qiagen) according to the manufacturer’s protocol. The regions containing polymorphisms were amplified by polymerase chain reaction (PCR) using the following optimized primers: 5′-AAGCACAAAGGCAGGTTCGC-3′ (forward) and 5′-GTGCCCAGCTGCAAAGTGTT-3′ (reverse). Genotyping was performed without knowledge of clinical outcomes by AITbiotech Pte Ltd. (Singapore).
Statistical analysis
Descriptive statistics were used to summarize the demographic and clinical characteristics of the patients. Differences in the demographic data and questionnaire scores between patients with and without cognitive impairment were compared using the independent-sample t-test if normally distributed. The Mann-Whitney U-test was used to compare non-normally distributed continuous and ordinal data. The chi-squared test was used to identify differences in categorical demographic data between the two groups. The Friedman test was used to evaluate changes in questionnaire and neuropsychological test scores across the three time points.
Genotype and allele frequencies were checked for deviation from the Hardy-Weinberg equilibrium using the chi-squared test with one degree of freedom. Binary logistic regression analysis assuming a dominant model was carried out to evaluate associations between the SNP of interest and cognitive function while adjusting for ethnicity and documented confounders of CACI (age, fatigue, menopausal status, chemotherapy regimen, and education level)30,31. In addition, anxiety, insomnia and brain-derived neurotrophic factor (BDNF) rs6265 polymorphism status were incorporated as confounders in the analysis model for subjective cognitive impairment as they are associated with self-reported cognitive impairment in breast cancer patients3,8. Univariate analysis was conducted and any variables that achieved P ≤ 0.1 were included in the logistic regression model. Anxiety (total BAI score), fatigue (total BFI score), and insomnia (EORTC QLQ-C30 insomnia score) scores corresponding to the time point at which cognitive impairment occurred were used in the logistic regression model. Using the median age as a cut-off, a subgroup analysis was conducted to evaluate whether similar genetic associations were observed in younger patients (below the median age of the cohort).
Results were reported as odds ratios (ORs) and 95% confidence intervals (CIs). Only cases with complete genetic and clinical information were included in the analysis. Statistical analyses were carried out using IBM’s Statistical Package for the Social Sciences (SPSS version 20) with P ≤ 0.05 considered as statistically significant.
Results
Patient demographics
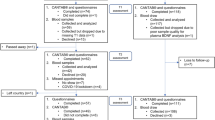
A total of 425 patients were recruited, of which 351 patients were included in the final analysis (Fig. 1). The mean age (±SD) was 51.2 ± 9.1 years. Patients were predominantly Chinese (81.2%), and 85.2% had received at least high school education. Over half (59.8%) were diagnosed with stage II breast cancer and almost all (95.6%) had a baseline Eastern Cooperative Oncology Group (ECOG) score of 0, indicating an ambulatory status without restrictions on daily activities. Two-thirds (64.7%) of the patients received an anthracycline-based chemotherapy regimen. Among patients who completed objective neuropsychological tests, 125 (50.6%) were assessed using Headminder, while 122 (49.4%) were assessed using CANTAB. The demographic and clinical characteristics of the participants receiving Headminder and CANTAB were similar (Table 1). There was a statistically significant reduction in the FACT-Cog summation score as well as the domain scores over time (Supplementary Table S3) with approximately one-third of the patients experiencing clinically significant decline in self-perceived cognitive function (Table 2). Cognitive impairment was also observed in various objective cognitive domains, with incidences ranging from 7.2% to 36.9% (Table 2). Patients assessed with Headminder showed stable to improved cognitive function over time in 3 out of 4 cognitive domains. In contrast, improvement of cognitive function over time was only observed in 1 out of 4 cognitive domains among patients assessed with CANTAB (Supplementary Table S3).
Genotyping and allele frequencies
All patients included in the final analysis were successfully genotyped for DNMT1 SNPs. A total of 215 (61.3%) patients were homozygous for the major allele (GG) of rs2162560 and 17 patients (4.8%) were homozygous for the minor allele (Table 3). A total of 153 patients (21.8%) were carriers of the A allele. There was no significant difference between the genotype frequencies between patients assessed by Headminder and CANTAB (P = 0.55).
Association of DNMT1 SNP rs2162560 with CACI
Analysis of self-perceived cognitive impairment revealed that patients with the A allele of DNMT1 rs2162560 had significantly lower odds of cognitive impairment in the concentration ability domain (OR = 0.45, 95% CI: 0.25–0.82, P = 0.01). Similarly, the presence of the A allele was also associated with lower odds of impairment in the functional interference domain (OR = 0.48, 95% CI: 0.24–0.95, P = 0.03) (Table 4). There was no statistically significant association between the DNMT1 rs2162560 genotype and other cognitive domains. An analysis performed using a general genetic model yielded similar results (Supplementary Table S4).
No association between the DNMT1 rs2162560 genotype and objective cognitive impairment assessed was observed (Table 5).
Subgroup analysis
A subgroup analysis was carried out with patients below the median age of the cohort, which was ≤51 years of age (N = 177). The results revealed that the DNMT1 rs2162560 A allele was protective against deteriorations in the memory domain (OR = 0.26, 95% CI: 0.09–0.71, P = 0.01), concentration domain (OR = 0.30, 95% CI: 0.12–0.74, P = 0.01), and mental acuity domain (OR = 0.42, 95% CI = 0.18–0.96, P = 0.04) of the FACT-Cog (Supplementary Table S5).
Discussion
In this study, we found an association between the DNMT1 rs2162560 SNP and self-perceived cognitive impairment in breast cancer patients; carriers of the A allele experienced lower odds of self-reported cognitive decline in two cognitive domains: concentration and functional interference. These findings are relevant and novel as no known associations between SNPs in DNMT1 and CACI have been described in the literature. A study involving 210 Brazilian Caucasian participants found no association between DNMT1 rs2162560 and Alzheimer’s disease19. A study on 632 South Indians reported a significant association between schizophrenia and DNMT1 rs2114724 and rs2228611, but no association was found with DNMT1 rs216256020. These results are not necessarily conflicting because cognitive disorders are complex and the mechanisms of disease manifestation in AD, schizophrenia, and CACI are very different. Both AD and CACI are associated with oxidative stress but they are propagated by different mechanistic pathways32. For example, subjective CACI has been characterized to be exacerbated by psychosocial or behavioral conditions including fatigue and anxiety3. In fact, the cognitive symptoms reported by patients are likely to be an extension of these conditions. The established association between DNMT1 SNPs and behavioral disorders could explain why we observed an association between DNMT1 rs2162560 and self-perceived cognitive impairment but not other domains of objective cognitive impairment. Furthermore, the populations analyzed in these studies may differ in genetic background and response to environmental factors, including chemotherapy treatment. DNA methylation is an epigenetic alteration that enables interaction with internal and external cues to create long-lasting changes in gene expression and possibly alter their homeostatic function and subjective experience33. Hence, the effects of DNMT1 SNPs may vary according to disease state or population.
The rs2162560 polymorphism is located in the intronic region of the DNMT1 gene on chromosome 19. Although introns are in the non-coding region, they may be involved in functions such as regulating alternative splicing or enhancing gene expression34. The rs2162560 mutant A allele is suspected to confer an intronic enhancer effect, similar to the DNMT1 rs2114724 T allele, which is associated with schizophrenia20. Global methylation status, as measured by the methylation of surrogate marker LINE-1, is higher in men with at least one mutant rs2114724 allele35. Since rs2162560 and rs2114724 were highly correlated in a linkage disequilibrium study (coefficient = 0.85, P < 0.001)15, carriers of the rs2162560 polymorphism will likely display increased DNA methylation activity. This supports our hypothesis that the A allele of rs2162560 confers a protective effect in cognitive domains such as concentration and memory potentially by increasing methylation activity.
In cancer patients undergoing chemotherapy, both cancer and its treatment have been speculated to trigger reprogramming of the genome, resulting in changes to gene expression and neuronal transmission33. A study using the TumorGraft mouse model found that global DNA methylation and DNMT1 levels decreased in the pre-frontal cortex of mice with triple-negative or progesterone-positive breast cancer TumorGrafts36. The A allele polymorphism may change DNMT1 activity to mitigate these adverse effects.
Genome-wide DNA methylation and expression levels of DNMTs in the pre-frontal cortex and hippocampus decrease with age and affect learning and memory37. We found that the DNMT1 rs2162560 A allele conferred a protective effect against declines in the memory, concentration, and mental acuity domains of the FACT-Cog in a younger subgroup of patients under 51 years of age. One possible explanation may be that, while rs2162560 exerts a protective effect through enhancing DNMT1 function, the decreased amount of DNMT1 in older patients diminishes any positive effect. Further studies are needed to elucidate how aging confounds the association between DNMT1 polymorphisms and CACI.
This study has multiple strengths, including the pre-treatment and longitudinal assessment of cognitive function and behavioral symptoms across three time points, adjustment for known clinical confounders of cognition, and use of validated tools to evaluate cognition. We have also included both subjective and objective cognitive assessments, as recommended by the International Cognition and Cancer Task Force (ICCTF)29. Cognitive profiles observed in this study are highly consistent with our previous study where we have presented the existence of heterogeneous cognitive trajectories among cancer patients receiving chemotherapy3. As CACI is a multifactorial, complex phenotype, the precision of the results could be improved by adjusting for additional clinical, behavioral, and environmental factors, along with assessing different aspects of cognitive function. We adjusted for different covariates in the regression models (subjective and objective assessments) to ensure that the most clinically relevant variables were represented in the models. However, it remains unknown whether these results are applicable to delayed-onset CACI. Another limitation is changing the objective cognition assessment tool mid-study to CANTAB since the Headminder system was no longer commercially available. Due to this switch in assessment tools and other logistical issues, cognitive assessments were not completed for all participants. It is challenging to compare data generated from two different assessment tools, as they may have different sensitivities or subtle differences in the cognition functions assessed. This also explains why we observed fewer patients classified as impaired by Headminder than CANTAB. Hence, we reported the data obtained from both tools separately and did not pool the data. However, we ensured that all neuropsychological tests used were validated for the same cognitive domains23,24. We also calibrated the RCI to standardize the measurement of cognitive changes indicated by each tool.
Conclusions
In conclusion, to the best of our knowledge, this is the first study to evaluate the genetic association between a DNMT1 polymorphism and CACI. The A allele of rs2162560 showed a neuroprotective effect in the concentration and functional interference domains. These findings suggest that subjective CACI in cancer patients, which is often exacerbated by behavioral symptoms, is associated with epigenetic processes. Further validation of the current findings is required. These future studies should include measurement of plasma methylation levels at various time points to correlate the effect of DNMT1 polymorphisms with cognitive function, which will provide further insight into the underlying epigenetic process. We could use this knowledge to identify patients who are at higher risk of developing CACI post-chemotherapy and provide targeted preventive strategies. Lastly, this study also provides the impetus to explore the use of pharmacological agents that target methylation in managing CACI.
References
Cheung, Y. T. et al. Association of proinflammatory cytokines and chemotherapy-associated cognitive impairment in breast cancer patients: a multi-centered, prospective, cohort study. Annals of oncology: official journal of the European Society for Medical Oncology 26, 1446–1451, https://doi.org/10.1093/annonc/mdv206 (2015).
Janelsins, M. C. et al. An update on cancer- and chemotherapy-related cognitive dysfunction: current status. Seminars in oncology 38, 431–438, https://doi.org/10.1053/j.seminoncol.2011.03.014 (2011).
Ng, T. et al. Distinct and heterogeneous trajectories of self-perceived cognitive impairment among Asian breast cancer survivors. Psycho-oncology 27, 1185–1192, https://doi.org/10.1002/pon.4635 (2018).
Koppelmans, V. et al. Neuropsychological performance in survivors of breast cancer more than 20 years after adjuvant chemotherapy. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 30, 1080–1086, https://doi.org/10.1200/jco.2011.37.0189 (2012).
Ahles, T. A. & Saykin, A. J. Candidate mechanisms for chemotherapy-induced cognitive changes. Nature reviews. Cancer 7, 192–201, https://doi.org/10.1038/nrc2073 (2007).
Krull, K. R. et al. Genetic mediators of neurocognitive outcomes in survivors of childhood acute lymphoblastic leukemia. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 31, 2182–2188, https://doi.org/10.1200/jco.2012.46.7944 (2013).
Ng, T., Chan, M., Khor, C. C., Ho, H. K. & Chan, A. The genetic variants underlying breast cancer treatment-induced chronic and late toxicities: a systematic review. Cancer treatment reviews 40, 1199–1214, https://doi.org/10.1016/j.ctrv.2014.10.001 (2014).
Ng, T. et al. Brain-derived neurotrophic factor genetic polymorphism (rs6265) is protective against chemotherapy-associated cognitive impairment in patients with early-stage breast cancer. Neuro-oncology 18, 244–251, https://doi.org/10.1093/neuonc/nov162 (2016).
Graff, J. & Mansuy, I. M. Epigenetic codes in cognition and behaviour. Behavioural brain research 192, 70–87, https://doi.org/10.1016/j.bbr.2008.01.021 (2008).
Jones, P. L. et al. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nature genetics 19, 187–191, https://doi.org/10.1038/561 (1998).
Dhe-Paganon, S., Syeda, F. & Park, L. DNA methyl transferase 1: regulatory mechanisms and implications in health and disease. International journal of biochemistry and molecular biology 2, 58–66 (2011).
Day, J. J. & Sweatt, J. D. DNA methylation and memory formation. Nature neuroscience 13, 1319–1323, https://doi.org/10.1038/nn.2666 (2010).
Feng, J. et al. Dnmt1 and Dnmt3a maintain DNA methylation and regulate synaptic function in adult forebrain neurons. Nature neuroscience 13, 423–430, https://doi.org/10.1038/nn.2514 (2010).
Miller, C. A. & Sweatt, J. D. Covalent modification of DNA regulates memory formation. Neuron 53, 857–869, https://doi.org/10.1016/j.neuron.2007.02.022 (2007).
Haggarty, P. et al. Human intelligence and polymorphisms in the DNA methyltransferase genes involved in epigenetic marking. PloS one 5, e11329, https://doi.org/10.1371/journal.pone.0011329 (2010).
Wilkinson, L. S., Davies, W. & Isles, A. R. Genomic imprinting effects on brain development and function. Nature reviews. Neuroscience 8, 832–843, https://doi.org/10.1038/nrn2235 (2007).
Yao, S. et al. Impact of chemotherapy for breast cancer on leukocyte DNA methylation landscape and cognitive function: a prospective study. Clin Epigenetics 11, 45, https://doi.org/10.1186/s13148-019-0641-1 (2019).
Mostowska, A., Sajdak, S., Pawlik, P., Lianeri, M. & Jagodzinski, P. P. DNMT1, DNMT3A and DNMT3B gene variants in relation to ovarian cancer risk in the Polish population. Molecular biology reports 40, 4893–4899, https://doi.org/10.1007/s11033-013-2589-0 (2013).
Pezzi, J. C. et al. DNA methyltransferase haplotype is associated with Alzheimer’s disease. Neuroscience letters 579, 70–74, https://doi.org/10.1016/j.neulet.2014.07.013 (2014).
Saradalekshmi, K. R. et al. DNA methyl transferase (DNMT) gene polymorphisms could be a primary event in epigenetic susceptibility to schizophrenia. PloS one 9, e98182, https://doi.org/10.1371/journal.pone.0098182 (2014).
Wagner, L. I., Sweet, J., Butt, Z., Lai, J. S. & Cella, D. Measuring patient self-reported cognitive function: development of the Functional Assessment of Cancer Therapy-Cognitive Function instrument. Journal of Supportive Oncology 7, W32–W39 (2009).
Cheung, Y. T., Lim, S. R., Shwe, M., Tan, Y. P. & Chan, A. Psychometric properties and measurement equivalence of the English and Chinese versions of the functional assessment of cancer therapy-cognitive in Asian patients with breast cancer. Value in health: the journal of the International Society for Pharmacoeconomics and Outcomes Research 16, 1001–1013, https://doi.org/10.1016/j.jval.2013.06.017 (2013).
Erlanger, D. M. et al. Development and validation of a web-based screening tool for monitoring cognitive status. The Journal of head trauma rehabilitation 17, 458–476 (2002).
Robbins, T. W. et al. Cambridge Neuropsychological Test Automated Battery (CANTAB): a factor analytic study of a large sample of normal elderly volunteers. Dementia (Basel, Switzerland) 5, 266–281 (1994).
Beck, A. T., Epstein, N., Brown, G. & Steer, R. A. An inventory for measuring clinical anxiety: psychometric properties. Journal of consulting and clinical psychology 56, 893–897 (1988).
Mendoza, T. R. et al. The rapid assessment of fatigue severity in cancer patients: use of the Brief Fatigue Inventory. Cancer 85, 1186–1196 (1999).
Aaronson, N. K. et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute 85, 365–376 (1993).
Cheung, Y. T. et al. Minimal clinically important difference (MCID) for the functional assessment of cancer therapy: cognitive function (FACT-Cog) in breast cancer patients. Journal of clinical epidemiology 67, 811–820, https://doi.org/10.1016/j.jclinepi.2013.12.011 (2014).
Wefel, J. S., Vardy, J., Ahles, T. & Schagen, S. B. International Cognition and Cancer Task Force recommendations to harmonise studies of cognitive function in patients with cancer. The Lancet. Oncology 12, 703–708, https://doi.org/10.1016/s1470-2045(10)70294-1 (2011).
Moore, H. C. An overview of chemotherapy-related cognitive dysfunction, or ‘chemobrain’. Oncology (Williston Park, N.Y.) 28, 797–804 (2014).
Vardy, J. Cognitive function in breast cancer survivors. Cancer treatment and research 151, 387–419, https://doi.org/10.1007/978-0-387-75115-3_24 (2009).
Butterfield, D. A. The 2013 SFRBM discovery award: selected discoveries from the butterfield laboratory of oxidative stress and its sequela in brain in cognitive disorders exemplified by Alzheimer disease and chemotherapy induced cognitive impairment. Free radical biology & medicine 74, 157–174, https://doi.org/10.1016/j.freeradbiomed.2014.06.006 (2014).
Wang, X. M. et al. Chemobrain: a critical review and causal hypothesis of link between cytokines and epigenetic reprogramming associated with chemotherapy. Cytokine 72, 86–96, https://doi.org/10.1016/j.cyto.2014.12.006 (2015).
Jo, B. S. & Choi, S. S. Introns: The Functional Benefits of Introns in Genomes. Genomics & informatics 13, 112–118, https://doi.org/10.5808/gi.2015.13.4.112 (2015).
Inoue-Choi, M. et al. Plasma S-adenosylmethionine, DNMT polymorphisms, and peripheral blood LINE-1 methylation among healthy Chinese adults in Singapore. BMC cancer 13, 389, https://doi.org/10.1186/1471-2407-13-389 (2013).
Kovalchuk, A. et al. Chemo brain or tumor brain - that is the question: the presence of extracranial tumors profoundly affects molecular processes in the prefrontal cortex of TumorGraft mice. Aging 9, 1660–1676, https://doi.org/10.18632/aging.101243 (2017).
Cui, D. & Xu, X. DNA Methyltransferases, DNA Methylation, and Age-Associated Cognitive Function. International Journal of Molecular Sciences 19, https://doi.org/10.3390/ijms19051315 (2018).
Acknowledgements
The authors acknowledge the contributions from Dr Kiley W.J. Loh, Dr Si Lin Koo, Dr Amit Jain, Dr Guek Eng Lee, Dr Mabel Wong, Dr Wen Yee Chay, Dr Rebecca Dent, Dr Yoon Sim Yap, Dr Raymond Ng and Dr Sok Yuen Beh, Mr. Ming Jong Chia and all the study participants. We thank the Department of Pharmacy of the National University of Singapore and the Department of Pharmacy of the National Cancer Centre Singapore for providing support for this project.
Author information
Authors and Affiliations
Contributions
A.C. was involved in conceptualization, methodology, supervision, funding acquisition and writing the original draft. A.Y. was involved in investigation, formal analysis, visualization and writing the original draft. M.S. was involved in investigation. C.J.T. was involved in investigation, formal analysis and writing the original draft. K.M.F. and P.C. were involved in investigation. C.C.K. was involved in the methodology. H.H.K. was involved in the methodology and contributed resources. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chan, A., Yeo, A., Shwe, M. et al. An Evaluation of DNA Methyltransferase 1 (DNMT1) Single Nucleotide Polymorphisms and Chemotherapy-Associated Cognitive Impairment: A Prospective, Longitudinal Study. Sci Rep 9, 14570 (2019). https://doi.org/10.1038/s41598-019-51203-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-51203-y
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.