Abstract
In eastern North America, including Canada, Lyme disease is caused by Borrelia burgdorferi sensu stricto and transmitted to humans by the blacklegged tick, Ixodes scapularis. The last decade has seen a growing incidence of Lyme disease in Canada, following the northward range expansion of I. scapularis tick populations from endemic areas in eastern United States. This may be attributable to movement of the many hosts that they parasitize, including songbirds, deer and small mammals. In this study, we wanted to test the effect of spatial, temporal and ecological variables, on blacklegged tick density and infection rates, near the northern limit of their distribution in Ontario and Quebec, Canada. We found an effect of both proportion of forested areas and distance to roads, on density of I. scapularis ticks and prevalence of infection by B. burgdorferi. We also found an effect of both sampling year and ordinal sampling data on prevalence of infection by B. burgdorferi. In six adjacent sites showing evidence of reproducing I. scapularis populations, we found that forest composition and structure influenced density of I. scapularis ticks. Our results suggest that blacklegged tick density and infection rate in Canada may be influenced by a variety of factors.
Similar content being viewed by others
Introduction
Lyme disease is a tick-borne illness caused by several species of Borrelia bacteria that are present in parts of Europe, Asia and North America, and transmitted to humans through the bite of a range of Ixodes tick species1. The disease is highly prevalent in eastern North America, where blacklegged ticks (Ixodes scapularis) infected by Borrelia burgdorferi sensu stricto may efficiently transmit the infection to humans and domestic animals1,2,3. It is estimated that Lyme disease is responsible for 300,000 cases in the United States each year, accounting for underreporting of the disease4. In Canada, Lyme disease incidence has risen dramatically, with the reported number of cases increasing from 144 to 2025 between 2009 and 20175.
Blacklegged tick populations have been increasingly observed in eastern Canada in the past several years, particularly in regions bordering endemic areas in the United States to the south. Their establishment in new areas is attributed to multiple factors, including spatial diffusion from established areas, alongside changes in climate and the environment that have impacted tick survival and the range of blacklegged tick hosts6,7,8,9. Based on surveillance at 36 sites in eastern Ontario, there is evidence that I. scapularis has spread northward at a rate of approximately 50 km/year, in line with previous predictions based on climate change10,11. An association between Lyme disease emergence and the advance of blacklegged ticks has been observed in the same region from 2010 to 201612. Although less likely, Lyme disease emergence may also be associated with range expansion of the white-footed mouse, the main reservoir host of B. burgdorferi in eastern Canada13. The bordering cities of Ottawa in Ontario and Gatineau in Quebec represent a major urban and suburban region situated at the northern range front of blacklegged ticks13,14, making this an important region for understanding factors related to tick and tick-borne pathogen emergence.
Northward dispersal of blacklegged ticks may be attributable to movement of the many hosts they parasitize, in particular songbirds, but also deer and other small mammals1. Long-distance transport of ticks by the means of migratory songbirds is a major contributing factor for the dispersal of ticks outside of their current range distribution. It has been associated with dispersal of I. scapularis and B. burgdorferi from the northeastern United States into eastern Canada15, and likely also from southern parts of the eastern Canadian provinces, where blacklegged tick populations are now well-established, into the Ottawa-Gatineau region in Canada. Migratory songbirds usually follow flyways that are situated along wetlands and major rivers, through systems known as riparian corridors, during their spring and fall migrations16,17,18,19. Water bodies of at least 5 hectares are thought to be the most important in these regards19. Thus, areas surrounding these landscape elements may be prone to colonization by adventitious populations of blacklegged ticks and B. burgdorferi. This may translate to local increases in environmental risk for Lyme disease if suitable hosts exist to support tick populations (i.e. deer) or provide infection to immature ticks (i.e. rodents, other small vertebrates). In contrast, tick movement by the means of mammals, such as deer and small mammals, has been associated with forest fragmentation20,21,22. In Europe, road and freeway edges may facilitate the dispersal of blacklegged ticks as they hitchhike on deer and small mammals in these forest-fringe areas23,24. Proportion of canopy cover was found to influence tick density in the United States and Canada8,25. Several competing or interacting landscape characteristics may therefore contribute to elevate Lyme disease risk in human populations26, through their impacts on local tick abundance8,23,25 and B. burgdorferi prevalence in ticks24.
The identification of landscape features associated with elevated tick abundance and infection rates can help to characterize areas of environmental risk for human Lyme disease infection. This in turn can help to target public health interventions and reduce Lyme disease risk to human populations. This is particularly important in regions of recent and ongoing disease emergence, where risk areas are rapidly changing6. In this study, situated at the northernmost limit of the distribution of blacklegged ticks in eastern Ontario and southwestern Quebec, Canada, we assessed variations in the distribution and abundance of I. scapularis, and their prevalence of infection with B. burgdorferi. We wanted to test the effect of proximity to land cover elements that may be conducive to movement of tick hosts, such as water bodies that are important for bird migration, and roads which fragment the landscape and act as dispersal corridors for mammals, on blacklegged tick density and infection rate across the study area. We considered water bodies of at least 5 hectares because they are the most important for bird migration, and we considered roads of one or two lanes because they are spread out around the study area and are one of the main drivers of landscape fragmentation. We strengthened these analyses with inclusion of time of sampling and proportion of forest cover to account for other processes that may affect tick density and infection rates. Lastly, we investigated the effects of ecological variables related to forest composition and structure on abundance of ticks, at six adjacent sites showing evidence of reproducing tick populations.
Results
Study site characteristics and tick density
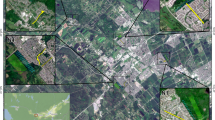
We collected a total of 576 I. scapularis ticks in 33 sites in two rounds of sampling, in late Spring and in early Fall, each year from 2017 to 2018, including 471 adults, 83 nymphs and 22 larvae (Table 1; Supplementary Table S1). A total of 572 were collected from 30 sites in Ottawa (Site O1 to O30; Table 1), while only four ticks were collected from three sites in Gatineau (G1 to G3; Table 1). The majority of ticks were collected from sites in western and south-western areas of Ottawa. Sampling effort varied between 1.5 and 19.2 hours per site (Table 1), depending on meteorological conditions, cluttered understory in certain sites which reduced efficiency of drag sampling, and seasonal closure of certain sites for maintenance.
Straight-line distance to water bodies from study sites’ centroid varied between 6 and 9327 meters (average of 1728 meters), straight-line distance to roads varied between 0 and 386 meters (average of 79 meters), and proportion of forested areas varied between 0 and 85% (average of 29%). Within the six tick-endemic sites in western Ottawa (O7, O9, O11, O16, O20 and O23; Fig. 1), we sampled a total of 241 transects of 100 meters (total of 24.1 km). Nearly three quarters of transects were in areas dominated by maples, cedars or pines; nearly half were in areas with medium abundance of the dominant tree type; more than half were in forested habitat; and nearly three quarters were in areas with flat terrain (Table 2). Most sites were characterized by sparse understory and deep litter layer, although these variables varied considerably among sites (Table 2).
Map of blacklegged tick dragging sites around the cities of Ottawa and Gatineau, Canada. Format, size and shading of points represent number of collected ticks, along with number of ticks infected by Borrelia burgdorferi, per hour of sampling, as in Legend. Sites where non-zero tick abundance was observed are labelled by site ID, as listed in Table 1. Built-up land is shown in light grey shading and water bodies in lighter blue shading. Map was created using ArcGIS 10.5 (ESRI, Redlands, CA, United States).
Tick infection rates
Following qPCR screening assays targeting B. burgdorferi DNA in the 554 adult and nymphal I. scapularis ticks sampled across the two-year study period, we identified 184 (33%) infected with B. burgdorferi, 169 of which were adults, and 15 were nymphs (Table 1). All individuals that tested positive for the 23S rRNA gene also tested positive for the OspA gene. The majority of B. burgdorferi-infected ticks were collected from sites from the western and south-western areas of Ottawa, similar to our observed patterns in tick density, with the highest density in O20, a large marshland situated on the southern shore of the Ottawa River (Fig. 1). Almost all infected nymphs were found in O13 (12 out of 15 infected nymphs), a provincial park situated in the southwest of the study area outside of the city of Ottawa (Fig. 1). The overall B. burgdorferi infection rate for sites within the city of Ottawa (n = 494; Table 1) was 32%, compared to 41% at the provincial park southwest of Ottawa (site O13, n = 56), and 25% for sites in Gatineau (sites G1, G2 and G3; n = 4), although the latter infection rate should be interpreted with caution owing to limited sample size.
Factors associated with tick density and infection rates across the study area
Our area-wide analysis, which considered all 33 sites in the study area, attempted to test our hypotheses about proximity to land cover elements potentially conducive to tick host movement, namely water bodies of 5 hectares or more, and roads with one or two lanes, on tick density of all life stages together and separately, and on prevalence of infection by B. burgdorferi. Across a total of 22 models constructed for each of the response variables, which included a variety of combinations of predictors, the best-fit model (i.e. model with the lowest AICc) included different combinations of fixed-effects variables, depending on the response variable under scrutiny.
While the best-fit model explaining total number of ticks included both distance to roads and proportion of forested areas, the best-fit model explaining number of adults only included proportion of forested areas. Both models had a high fit (Conditional pseudo-R2 = 0.87 and 0.80, respectively; Table 3). The best-fit model explaining number of nymphs and the best-fit model explaining number of larvae both had a relatively low fit (Conditional pseudo-R2 = 0.36 and 0.12, respectively; Table 3). They both included proportion of forested areas, although the former also included ordinal sampling date and the latter also included sampling year.
Relative variable importance for proportion for distance to roads for predicting total number of ticks was moderately high, at 0.78, and relationship was negative (Table 4), signifying that sites which are farther away from roads were associated with lower density of ticks. Relative variable importance of sampling year for predicting number of larvae was high, at 0.92 and the relationship was negative (Table 4), which means the year 2018 was associated with lower density of larvae than 2017. Relative variable importance of ordinal sampling date for predicting number of nymphs was high, at 1.00, and the relationship was negative (Table 4), signifying that spring season was associated with higher numbers of nymphs.
The best-fit model explaining prevalence of infection by B. burgdorferi included sampling year, ordinal sampling date, proportion of forested areas, and distance to roads, with a relatively high model fit (Conditional pseudo-R2 = 0.64; Table 3). Relative variable importance of sampling year, ordinal sampling date and distance to roads were high, at 1.00 for all three variables (Table 4). Relationship was negative for year of sampling, signifying year 2018 held lower infection prevalence than 2017. Relationship was positive for ordinal sampling date and distance to roads, signifying that ticks collected at a later sampling date or in a site that is situated farther away from roads yielded higher infection prevalence. Relative variable importance of proportion of forested areas was moderately high, at 0.77, and relationship was positive, meaning sites that are surrounded by larger forest cover are associated with higher infection prevalence (Table 4).
Proportion of forested areas and distance to water bodies showed a correlation coefficient of r = 0.4. However, all other correlation coefficients between pairs of predictors were low among all numerical predictors (r < ±0.1), and Moran’s I calculated with the full model incorporating all predictors was non-significant (P > 0.2). We kept all predictors in our analyses due to low probability of any major collinearity or autocorrelation effect that could affect the outcomes of our analyses.
Factors associated with tick abundance at specific sites
Our site-specific analysis, which considered six sites in the western end of the city of Ottawa with established tick populations (O7, O9, O10, O11, O16 and O23; Table 1; Fig. 1), tested the effect of ecological variables on tick abundance of all life stages and of the whole tick population.
We found a significant effect of dominant tree type on total number of ticks and on number of adults (Table 5). In the analysis of total number of ticks, the difference of means was significantly higher in locations characterized by cedar and maple trees, but without ash trees, than vice versa (Fig. 2). This effect was also present in the analysis of abundance of adult ticks, except for maple trees, which did not have any effect on the difference of means. These results suggest that transects with cedars or maples as dominant tree types are associated with higher numbers of ticks, while transects dominated by ash trees are associated with lower numbers of ticks.
Histogram of difference of means for each dominant tree type on number of blacklegged ticks. Data was collected through tick dragging of 100-meter transects in six sites in Ottawa in 2018 (O7, O9, O10, O11, O16 and O23; Table 1; Fig. 1). Letters next to bars represent significantly different (P < 0.05) groups of means of dominant tree types.
We also found a significant effect of habitat type on number of adult ticks (Table 5). The difference of means was higher in locations characterized by forest and woodland (canopy cover of 50% and higher), and without shrubland, than vice versa (Fig. 3). This suggests that tick abundance in environments with high canopy cover is significantly higher than tick abundance in shrubland environments, in our study area.
Histogram of difference of means for each habitat type on number of adult blacklegged ticks. Data was collected through tick dragging of 100-meter transects in six sites in Ottawa in 2018 (O7, O9, O10, O11, O16 and O23; Table 1; Fig. 1). Letters next to bars represent significantly different (P < 0.05) groups of means of habitat types.
Generalized variance inflation factors calculated for each predictor in a full generalized linear model showed values below 2 for all predictors. We kept all predictors in our analyses due to low probability of any major collinearity effect that could affect the outcomes of our analyses.
Discussion
Previous studies suggest that migratory songbirds may be an important means of long-distance dispersal for ticks15, and that songbirds usually follow riparian corridors to reach their wintering or reproduction grounds through major flyways used by multiple species18,19. Adult ticks may colonize new areas outside of their native range, and possibly establish founding populations if climate conditions, host density and habitat are appropriate for tick survival and reproduction11,13,14. Our results do not support that proximity to water bodies of at least 5 hectares may be associated with higher likelihood of tick establishment and environmental risk for Lyme disease in the Ottawa-Gatineau region. However, additional data on abundance at migratory stopover points and most likely migration routes of various species of migratory birds are needed to clarify the influence of water bodies on tick population establishment. Our results nonetheless suggest that establishment of adventitious tick populations may not necessarily be happening in the vicinity of migratory dispersal corridors, and potentially may require additional landscape features to be successful.
Landscape fragmentation has been linked with increased blacklegged tick density and higher Lyme disease incidence20,26. Among others, roads may facilitate movement of ticks through hitchhiking on their mammalian hosts, as seen in two studies in Europe23,24. As per our predictions, we found a negative effect of distance to roads of one or two lanes on the density of the whole blacklegged tick population. Therefore, our results support that proximity to roads of one or two lanes may be associated with higher likelihood of tick establishment. However, opposite to our predictions, we detected a positive relationship between distance to roads and infection prevalence by B. burgdorferi in blacklegged ticks. A complex interplay between forest fragmentation and diversity of blacklegged tick hosts, with variable competency for B. burgdorferi, may be influencing pathogen transmission rates20,26, and may not be fully captured by our analysis.
Our study detected inter-annual differences in the density of larval ticks and infection rates of adult and nymphal ticks in our study area, after considering differences in sampling effort between years. We found a higher densityof larvae and nymphal and adult infection rates in 2017 than in 2018. With no other information, it is impossible to ascertain the exact cause of these observed differences. Cumulative growing degree days, or total degrees above 10 °C since the beginning of the year, calculated using daily mean temperatures, through week 20 of the year, was shown to be the best predictor of the start week of the Lyme disease season in northeastern United States27. Climatic and weather patterns, including drought, may also affect development, survival and host-seeking behavior of blacklegged ticks28. While this investigation was outside the scope of the current study, it is possible that variation in meteorological patterns across years may affect the start of the reproductive activity in blacklegged ticks and may affect abundance and density of host-seeking blacklegged ticks throughout the warm season. Analysis of climatic and weather patterns and tick abundance data over multiple years may help to better understand the drivers of inter-annual fluctuations in this region.
Our results suggest seasonal variation in tick abundance and the abundance of ticks infected with B. burgdorferi. We found a higher abundance of nymphs early in the year, as expected based on tick phenology29,30. Interestingly, we also found a higher infection prevalence by B. burgdorferi late in the year, which is similar to findings from another study in Europe31, although this trend is not consistently observed in all regions or all years32. Our results support a general pattern seen throughout the range of I. scapularis, with the Spring and Fall seasons posing different levels of Lyme disease risk owing to changes in environmental risk and human exposure. The late Spring season brings increased human exposure during outdoor activities in addition to larger numbers of nymphal I. scapularis, which are harder to detect and promptly remove than adult ticks, and therefore pose the highest risk of Lyme disease transmission to humans29,33. On the other hand, the Fall season may be characterized by more B. burgdorferi-infected ticks in the environment, but lower human-tick exposure and the predominance of adult ticks during this season reduces the overall risk of Lyme disease transmission to humans. Further data collection over multiple sampling periods and sites is needed to better understand the factors that may contribute to the observed differences in infection prevalence in the Spring and Fall seasons.
We found a positive relationship between proportion of forested areas and several indices of tick establishment or Lyme disease environmental risk across the study area: total abundance of ticks, number of adult ticks, number of nymphs and infection prevalence by B. burgdorferi. Additionally, in our site-specific analysis, areas with tree/shrub cover >50% were associated with higher abundance of adult ticks than shrublands, with tree cover around 0%. This is in contrast to a study performed at a large spatial scale across all of southern and eastern Ontario, which found no such association with tick abundance11. On the other hand, a study at a relatively small spatial scale in eastern Ontario found an effect of canopy cover on nymphal blacklegged tick abundance25. Another study at a relatively small spatial scale in Virginia found forest cover to be the most important driver of I. scapularis density8. Altogether, spatial scale appears to be important in determining the effect of ecological variables on tick abundance, and forest cover could be an important determinant of tick abundance within a specific site.
Finally, our results show that transects dominated by cedars and maples held the highest abundance of I. scapularis ticks, while locations dominated by ash trees held significantly lower tick numbers. These results may be explained, at least in part, by the behavior of the preferred reproductive hosts of blacklegged ticks, namely white-tailed deer1. A previous study in eastern Ontario found that the abundance of white-tailed deer was positively correlated with nymphal blacklegged tick abundance25. Similarly, forest stands used by roe deer in Europe was associated with Ixodes ricinus abundance34. Studies in the Midwest of the United States have shown deer herbivory to be strongest on northern white cedars, eastern hemlocks and white pines35,36. Deer are known to follow forest edges of certain conifer species, including balsam firs in Anticosti Island in eastern Quebec37 and northern white cedars in northern Michigan38, during winter to minimize locomotion costs and increasing access to forage during the cold season. Therefore, if deer aggregate around certain conifer species in the Spring and Fall seasons around Ottawa, these trees would be associated with higher tick abundance in the following warm season. Additionally, maple forests have frequently been associated with higher rates of I. scapularis survival in southern Ontario39. Another possible explanation for these results is about the difficulty of sampling ticks across forests dominated by certain tree species, compared to other tree assemblages, due to clutter at the understory level, which restricts sampler movement. One limitation of our study is the variation in sampling effort across sites, which can lead to significant differences in tick densities at the local scale40.
In conclusion, our study presents a contemporary picture of the distribution of I. scapularis ticks and their prevalence of infection with B. burgdorferi in a zone of recent and ongoing Lyme disease emergence in eastern Canada12,41. Using data from active tick surveillance over two consecutive years, we identified factors that are associated with higher tick density. We also identified spatial and temporal variation in tick infection rates, with an estimated 33% of blacklegged ticks infected with B. burgdorferi over this time period. These results can help to characterize variation in environmental risk for Lyme disease in this densely populated region, and identify potential entry points for intervention to reduce human-tick exposure. These effects should be investigated in other emerging blacklegged tick-prone areas across Canada. Among others, the effects of distance to roads on infection prevalence, the increase in infection prevalence late in the year, the overall inter-annual variation in density and infection rates of ticks, and the effects of forest composition on tick density are surprising, and will need further investigations. Finally, more research is needed to better understand how landscape features interact with wildlife host and tick populations through time in this region to facilitate tick establishment and Lyme disease transmission, in order to better predict areas that may be at highest risk of Lyme disease establishment and spread.
Methods
Study location
Our study area encompassed 30 sites within and bordering the city of Ottawa (ON, Canada) and three sites within and bordering the city of Gatineau (QC, Canada), which are separated by the Ottawa River. The Ottawa-Gatineau region is comprised of a highly built-up downtown core, surrounded by large suburban and rural areas, as well as actively managed forests and conservation areas. To assess the abundance of blacklegged ticks in a variety of environments across the study area with potential risk of human exposure to ticks, we selected parks, recreational trails and conservation areas that were spatially representative of the study region as our study sites (Fig. 1). We did not select sampling sites based on prior knowledge about local blacklegged tick densities, to avoid sampling bias which could affect the outcomes of our analyses.
Field data
A team of two to three trained field collectors visited each site in the Spring (May-July) and Fall (September-November) seasons of 2017 and 2018. In each site, ticks were collected for a minimum of 1.5 person-hours (i.e. combined number of sampling hours for all collectors present on any given day), using the drag sampling method, which consists of dragging a white flannel sheet along the ground to collect questing ticks42. Our sampling effort varied based on meteorological conditions, cluttered understory in certain sites which reduced efficiency of drag sampling, and seasonal closure of certain sites for maintenance. Collectors stopped roughly every 50 meters to remove ticks from the flannel and collect GPS coordinates. We preserved all live-collected ticks in small plastic tubes and stored them at −20 °C upon arrival at the laboratory.
During the second year of sampling (i.e. 2018), we selected a subset of sites in the western end of the city of Ottawa, where tick abundance and infection rates where highest, to collect additional site-specific data, which were derived from several studies25,40,43, for an additional, complementary analysis. These sites included those where we consistently collected ticks of multiple life stages across sampling seasons in 2017, thus reflecting sites with potential reproducing populations of I. scapularis44. Selecting sites with reproducing tick populations was likely to capture signals from local ecological drivers of tick density. In a total of six sites (O7, O9, O10, O11, O16 and O23; Table 1; Fig. 1) covering an area of about 150 square kilometers, we visually estimated field data for seven variables. We identified the dominant tree type in the area (maple, elm, poplar, oak, beech, birch, ash, pine, spruce, fir, cedar, or no tree; using the taxonomic key from Bertrand et al.45), the relative abundance of the dominant tree (high: >75%, moderate: 25–75%, low: <25%, or negligible: ~0%), the habitat type (bare: tree/shrub cover ~0%, shrubland: shrub cover >25% and tree cover ~0%, savannah: tree/shrub cover between 25–50%, woodland: tree/shrub cover between 50–75%, or forest: tree/shrub cover >75%), understory density (open: <25%, sparse: 25–50%, full: 50–75%, dense: >75%), litter layer depth (shallow: 0–2 cm, moderate: 3–5 cm, or deep: >5 cm), soil moisture (dry, fresh, moist or wet) and site aspect (flat, depression, slope or hilltop). We collected site-specific field data midway between every two 50-meter sampling points in the course of tick dragging, and linked these data to the number of collected I. scapularis ticks of each life stage totaled for the corresponding two locations. This resulted in 100-meter transects as the measurement unit for each variable. This was done over two rounds of sampling per site, one in late Spring and one in early Fall, and over a total of 6 person-hours per site, i.e. 3 person-hours per round of sampling per site.
Spatial data
We derived the centroid coordinates for each site using ArcGIS v10.5 (Redlands, CA, United States). We obtained spatial land cover data of water bodies (rivers, lakes and ponds), roads (local and arterial roads) and forest patches, from the CanVec Series topographic datasets produced by Natural Resources Canada46. We calculated the shortest straight-line metric distance from each site’s centroid to the nearest water body and to the nearest road, using ‘Spatial Analyst’ in ArcGIS v10.5. We drew concentric buffer zones of an area of 1 km2 at each study site’s centroid, and calculated the intersecting area between each site’s buffer zone and the forest patch layer, using ‘Spatial Analyst’ in ArcGIS v10.5. For mapping purposes, we calculated tick density as the total number of ticks divided by the person-hours of sampling effort.
Laboratory analyses
For all collected ticks, we identified the species and stage using microscopy and taxonomic keys47. We tested I. scapularis adult and nymphal ticks for B. burgdorferi using quantitative polymerase chain reaction (qPCR) assays according to previously published protocols48,49,50. Briefly, we extracted total genomic DNA using the QIAamp DNA mini kit (QIAGEN Inc., Mississauga, ON, Canada) and we used qPCR screening assays targeting 23S rRNA to identify presence of Borrelia DNA. For positive samples, we conducted a second qPCR assay targeting the OspA gene to confirm presence of B. burgdorferi DNA.
Statistical analyses
We conducted two sets of analyses to assess (1) factors associated with tick density and infection rates across the study area at all 33 sites, and (2) factors associated with tick abundance at six tick-endemic sites.
For the area-wide analysis, we used a mixed-effects modeling approach to determine the effect of year of sampling (2017 and 2018), ordinal date of sampling, proportion of forested lands, distance to nearest water body of 5 hectares or more and distance to nearest road of one or two lanes on five response variables: total number of ticks, number of adults, number of nymphs, number of larvae, and prevalence of B. burgdorferi infection in adults and nymphs (in percentage, without decimals). We used the ‘glmer’ function from the ‘lme4’ package51 in R 3.5.1. We performed the analysis on every possible combination of fixed-effects variables, including a null model excluding all fixed-effects variables. We included site identifier (i.e. to account for repeated sampling in a number of sites) and sampling effort (i.e. to account for varying number of person-hours of sampling across sites) as random-effects variables. We assessed variable importance using model averaging52. We used the Poisson modeling family, due to the zero-inflated positive nature of the response variables. We calculated a marginal and conditional pseudo-R2 for each model, following the method of Johnson53, using the ‘r.squaredGLMM’ function from the ‘MuMIn’ package54 in R 3.5.1 to measure the strength of the effect of predictors in our models.
Prior to these analyses, we tested for correlation among pairs of predictors, to detect potential collinearity effects on the results. We also tested for spatial autocorrelation of data points, with the ‘Moran.I’ function in the ‘ape’ package55 and a matrix of inverse spatial distances as ‘weights’, in R 3.5.1. Finally, we standardized all numeric variables, by subtracting the mean and dividing by the standard deviation, for more meaningful comparisons among predictors and to remove dispersion issues in the models.
For the site-specific analysis, we conducted an analysis of variance with repeated measures to determine the effect of dominant tree type, abundance of dominant tree type, habitat type, understory density, litter layer depth, soil moisture and site aspect on four response variables: total number of ticks, number of adults, number of nymphs, and number of larvae. To this end, we used the ‘aov’ function from the ‘stats’ package in R 3.5.1. We used the site identifier as error term to correct for repeated sampling at each site, which could bias our analysis due to pseudo-replication. For variables displaying significant effect (P < 0.05), we conducted a post hoc test to determine which levels in the variables display a significant difference in means. To this end, we used the ‘TukeyC’ function from the ‘TukeyC’ package56 in R 3.5.1.
Prior to these analyses, we calculated generalized variance inflation factors adjusted for the number of degrees of freedom for all predictors in a generalized linear model, to detect potential collinearity effects on the results. We used the ‘glm’ function from the ‘lme4’ package51 and the ‘vif’ function from the ‘car’ package57 in R 3.5.1.
Data availability
All area-wide data can be found in the Supplementary Table S1.
References
Steere, A. C., Coburn, J. & Glickstein, L. The emergence of Lyme disease. J. Clin. Invest. 113, 1093–1101 (2004).
Steere, A. C. et al. The spirochetal etiology of Lyme disease. N. Engl. J. Med. 308, 733–740 (1983).
Thompson, C., Spielman, A. & Krause, P. J. Coinfecting deer‐associated zoonoses: Lyme disease, babesiosis, and ehrlichiosis. Clin. Infect. Dis. 33, 676–685 (2001).
Kuehn, B. M. CDC estimates 300 000 US cases of Lyme disease annually. JAMA 310, 1110 (2013).
Public Health Agency of Canada. Surveillance of Lyme Disease. Available at: https://www.canada.ca/en/public-health/services/diseases/lyme-disease/surveillance-lyme-disease.html (2018).
Ogden, N. H. et al. Risk maps for range expansion of the Lyme disease vector, Ixodes scapularis, in Canada now and with climate change. Int. J. Health Geogr. 7, 24 (2008).
Simon, J. A. et al. Climate change and habitat fragmentation drive the occurrence of Borrelia burgdorferi, the agent of Lyme disease, at the northeastern limit of its distribution. Evol. Appl. 7, 750–764 (2014).
Ferrell, A. & Brinkerhoff, R. Using landscape analysis to test hypotheses about drivers of tick abundance and infection prevalence with Borrelia burgdorferi. Int. J. Environ. Res. Public. Health 15, 737 (2018).
Halsey, S. J., Allan, B. F. & Miller, J. R. The role of Ixodes scapularis, Borrelia burgdorferi and wildlife hosts in Lyme disease prevalence: a quantitative review. Ticks Tick-Borne Dis. 9, 1103–1114 (2018).
Leighton, P. A., Koffi, J. K., Pelcat, Y., Lindsay, L. R. & Ogden, N. H. Predicting the speed of tick invasion: an empirical model of range expansion for the Lyme disease vector Ixodes scapularis in Canada: Predicting I. scapularis invasion. J. Appl. Ecol. 49, 457–464 (2012).
Clow, K. M. et al. Northward range expansion of Ixodes scapularis evident over a short timescale in Ontario, Canada. PLoS One 12, e0189393 (2017).
Kulkarni, M. A., Narula, I., Slatculescu, A. M. & Russell, C. Lyme disease emergence after invasion of the blacklegged tick, Ixodes scapularis, Ontario, Canada, 2010–2016. Emerg. Infect. Dis. 25, 328–332 (2019).
Roy-Dufresne, E., Logan, T., Simon, J. A., Chmura, G. L. & Millien, V. Poleward expansion of the white-footed mouse (Peromyscus leucopus) under climate change: implications for the spread of Lyme disease. PLoS One 8, e80724 (2013).
Soucy, J.-P. R. et al. High-resolution ecological niche modeling of Ixodes scapularis ticks based on passive surveillance data at the northern frontier of Lyme disease emergence in North America. Vector-Borne Zoonotic Dis. 18, 235–242 (2018).
Ogden, N. H. et al. Role of migratory birds in introduction and range expansion of Ixodes scapularis ticks and of Borrelia burgdorferi and Anaplasma phagocytophilum in Canada. Appl. Environ. Microbiol. 74, 1780–1790 (2008).
Bingman, V. P. Inland morning flight behavior of nocturnal passerine migrants in Eastern New York. The Auk 97, 465–472 (1980).
DeLong, J. P., Cox, S. W. & Cox, N. S. A comparison of avian use of high- and low-elevation sites during autumn migration in central New Mexico. J. Field Ornithol. 76, 326–333 (2005).
Kirsch, E. M., Heglund, P. J., Gray, B. R. & McKann, P. Songbird use of floodplain and upland forests along the Upper Mississippi River corridor during spring migration. The Condor 115, 115–130 (2013).
Pocewicz, A. et al. Modeling the distribution of migratory bird stopovers to inform landscape-scale siting of wind development. PLoS One 8, e75363 (2013).
Allan, B. F., Keesing, F. & Ostfeld, R. S. Effect of forest fragmentation on Lyme disease risk. Conserv. Biol. 17, 267–272 (2003).
Trout Fryxell, R. T. et al. Habitat and vegetation variables are not enough when predicting tick populations in the Southeastern United States. PLoS One 10, e0144092 (2015).
Ehrmann, S. et al. Habitat properties are key drivers of Borrelia burgdorferi (s.l.) prevalence in Ixodes ricinus populations of deciduous forest fragments. Parasit. Vectors 11 (2018).
Delgado, J. D., Abreu-Yanes, E., Abreu-Acosta, N., Flor, M. D. & Foronda, P. Vertebrate ticks distribution and their role as vectors in relation to road edges and underpasses. Vector-Borne Zoonotic Dis. 17, 376–383 (2017).
Hornok, S. et al. Impact of a freeway on the dispersal of ticks and Ixodes ricinus-borne pathogens: forested resting areas may become Lyme disease hotspots. Acta Vet. Hung. 65, 242–252 (2017).
Werden, L. et al. Geography, deer, and host biodiversity shape the pattern of Lyme disease emergence in the Thousand Islands archipelago of Ontario, Canada. PLoS One 9, e85640 (2014).
McClure, M. & Diuk-Wasser, M. Reconciling the entomological hazard and disease risk in the Lyme disease system. Int. J. Environ. Res. Public. Health 15, 1048 (2018).
Eisen, R. J., Monaghan, A., Mead, P. & Moore, S. M. Meteorological influences on the seasonality of Lyme disease in the United States. Am. J. Trop. Med. Hyg. 90, 486–496 (2014).
Eisen, R. J., Eisen, L., Ogden, N. H. & Beard, C. B. Linkages of weather and climate with Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae), enzootic transmission of Borrelia burgdorferi, and Lyme disease in North America. J. Med. Entomol. 53, 250–261 (2016).
Diuk-Wasser, M. A. et al. Spatiotemporal patterns of host-seeking Ixodes scapularis nymphs (Acari: Ixodidae) in the United States. J. Med. Entomol. 43, 166–176 (2006).
Needham, G. R., Stewart, R. L. & Curran, K. L. Ecology and environmental management of Lyme disease. J. Med. Entomol. 32, 209–210 (1995).
Kybicová, K., Baštová, K. & Malý, M. Detection of Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in questing ticks Ixodes ricinus from the Czech Republic. Ticks Tick-Borne Dis. 8, 483–487 (2017).
Roome, A., Spathis, R., Hill, L., Darcy, J. & Garruto, R. Lyme disease transmission risk: seasonal variation in the built environment. Healthcare 6, 84 (2018).
Rodgers, S. E., Miller, N. J. & Mather, T. N. Seasonal variation in nymphal blacklegged tick abundance in southern New England forests. J. Med. Entomol. 44, 898–900 (2007).
Tack, W., Madder, M., Baeten, L., De Frenne, P. & Verheyen, K. The abundance of Ixodes ricinus ticks depends on tree species composition and shrub cover. Parasitology 139, 1273–1281 (2012).
Rooney, T. P. Deer impacts on forest ecosystems: a North American perspective. Forestry 74, 201–208 (2001).
Bradshaw, L. & Waller, D. M. Impacts of white-tailed deer on regional patterns of forest tree recruitment. For. Ecol. Manag. 375, 1–11 (2016).
Massé, A. & Côté, S. D. Linking alternative food sources to winter habitat selection of herbivores in overbrowsed landscapes. J. Wildl. Manag. 76, 544–556 (2012).
Van Deelen, T. R. Deer–cedar interactions during a period of mild winters: implications for conservation of conifer swamp deeryards in the Great Lakes region. Nat. Areas J. 19, 263–274 (1999).
Lindsay, L. R. et al. Survival and development of the different life stages of Ixodes scapularis (Acari: Ixodidae) held within four habitats on Long Point, Ontario, Canada. J. Med. Entomol. 35, 189–199 (1998).
Ripoche, M. et al. Multi-scale clustering of Lyme disease risk at the expanding leading edge of the range of Ixodes scapularis in Canada. Int. J. Environ. Res. Public. Health 15, 603 (2018).
Kulkarni, M. et al. Ixodes scapularis tick distribution and infection rates in Ottawa, Ontario, 2017. Can. Commun. Dis. Rep. 44, 237–242 (2018).
Public Health Ontario. Active tick dragging: standard operating procedure. (Queen’s Printer for Ontario, 2015).
Clow, K. M. et al. The influence of abiotic and biotic factors on the invasion of Ixodes scapularis in Ontario, Canada. Ticks Tick-Borne Dis. 8, 554–563 (2017).
Clow, K. M. et al. A field-based indicator for determining the likelihood of Ixodes scapularis establishment at sites in Ontario, Canada. PLoS One 13, e0193524 (2018).
Bertrand, N., Carpentier, B., Thibault, J.-P. Québec (Province) & Ministère des ressources naturelles. Les arbres du Québec. (Québec, Ministère des ressources naturelles, 2006).
Natural Resources Canada. Topographic data of Canada - CanVec Series. Available at: https://open.canada.ca/data/en/dataset/8ba2aa2a-7bb9-4448-b4d7-f164409fe056 (2019).
Lindquist, E. et al. A handbook to the ticks (Ixodida: Ixodidae, Argasidae) of Canada. (Biological Survey of Canada, 2016).
Courtney, J. W., Kostelnik, L. M., Zeidner, N. S. & Massung, R. F. Multiplex real-time PCR for detection of Anaplasma phagocytophilum and Borrelia burgdorferi. J. Clin. Microbiol. 42, 3164–3168 (2004).
Nelder, M. P. et al. Population-based passive tick surveillance and detection of expanding foci of blacklegged ticks Ixodes scapularis and the Lyme disease agent Borrelia burgdorferi in Ontario, Canada. PLoS One 9, e105358 (2014).
Werden, L. et al. Prevalence of Anaplasma phagocytophilum and Babesia microti in Ixodes scapularis from a newly established Lyme disease endemic area, the Thousand Islands region of Ontario, Canada. Vector-Borne Zoonotic Dis. 15, 627–629 (2015).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67 (2015).
Symonds, M. R. E. & Moussalli, A. A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaike’s Information Criterion. Behav. Ecol. Sociobiol. 65, 13–21 (2011).
Johnson, P. C. D. Extension of Nakagawa & Schielzeth’s R 2 GLMM to random slopes models. Methods Ecol. Evol. 5, 944–946 (2014).
Barton, K. MuMIn: multi-model inference. (Institute of Nature Conservation, 2013).
Paradis, E. & Schliep, K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35, 526–528 (2019).
Faria, J. C., Jelihovschi, E. G. & Allaman, I. B. Conventional Tukey Test. (Universidade Estadual de Santa Cruz, 2018).
Fox, J. & Weisberg, S. An R companion to applied regression. (Sage, 2011).
Acknowledgements
We thank all members from the INSIGHT lab for their suggestions and comments on the analyses and the initial draft of the manuscript. This project was supported by grants from the Canadian Institutes of Health Research and Public Health Agency of Canada to M.A.K.
Author information
Authors and Affiliations
Contributions
B.T. and M.A.K. conceived the study and wrote the manuscript. B.T., A.S., C.R.T. and R.M. performed field collection of samples and laboratory work. B.T., J.K.K., P.A.L. and M.A.K. interpreted results of statistical analyses and revised the writing. All authors read, reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Talbot, B., Slatculescu, A., Thickstun, C.R. et al. Landscape determinants of density of blacklegged ticks, vectors of Lyme disease, at the northern edge of their distribution in Canada. Sci Rep 9, 16652 (2019). https://doi.org/10.1038/s41598-019-50858-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-50858-x
This article is cited by
-
Ixodes scapularis density and Borrelia burgdorferi prevalence along a residential-woodland gradient in a region of emerging Lyme disease risk
Scientific Reports (2024)
-
Environmental Drivers of Immature Ixodes scapularis in Minnesota’s Metro Area
EcoHealth (2023)
-
Spatiotemporal trends and socioecological factors associated with Lyme disease in eastern Ontario, Canada from 2010–2017
BMC Public Health (2022)
-
Phylogeographic dynamics of the arthropod vector, the blacklegged tick (Ixodes scapularis)
Parasites & Vectors (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.