Abstract
Cholangiocarcinoma (CCA) is a malignant neoplasm of the biliary tract. Thailand reports the highest incidence of CCA in the world. The aim of this study was to map the distribution of CCA and identify spatial disease clusters in Northeast Thailand. Individual-level data of patients with histopathologically confirmed CCA, aggregated at the sub-district level, were obtained from the Cholangiocarcinoma Screening and Care Program (CASCAP) between February 2013 and December 2017. For analysis a multivariate Zero-inflated, Poisson (ZIP) regression model was developed. This model incorporated a conditional autoregressive (CAR) prior structure, with posterior parameters estimated using Bayesian Markov chain Monte Carlo (MCMC) simulation with Gibbs sampling. Covariates included in the models were age, sex, normalized vegetation index (NDVI), and distance to water body. There was a total of 1,299 cases out of 358,981 participants. CCA incidence increased 2.94 fold (95% credible interval [CrI] 2.62–3.31) in patients >60 years as compared to ≤60 years. Males were 2.53 fold (95% CrI: 2.24–2.85) more likely to have CCA when compared to females. CCA decreased with a 1 unit increase of NDVI (Relative Risk =0.06; 95% CrI: 0.01–0.63). When posterior means were mapped spatial clustering was evident after accounting for the model covariates. Age, sex and environmental variables were associated with an increase in the incidence of CCA. When these covariates were included in models the maps of the posterior means of the spatially structured random effects demonstrated evidence of spatial clustering.
Similar content being viewed by others
Introduction
Cholangiocarcinoma (CCA) is a malignant neoplasm of the biliary tract and globally is the second most common primary liver cancer1. CCA is particularly challenging to address due to it being generally diagnosed late, giving poor survival rates. According to the International Statistical Classification of Diseases and Related Health Problems, 10th revision (ICD-10), CCA comprises: intrahepatic bile duct carcinoma (C22.1), extrahepatic bile duct carcinoma (C24.0), and ampulla of Vater carcinoma (C24.1). Incidence rates for CCA vary widely from around 0.2 per 100,000 population in Australia to 96 per 100,000 in men in Northeast Thailand2. Overall the largest burdens for this cancer are found in and Southeast Asia3. Within Northeast Thailand itself, which has the world’s highest rates of CCA, age-standardized incidence varies. For example, rates ranging between 93.8 to 317.6 per 100,000 person-years between villages in Khon Kaen Province from 1990 to 20014. This variation in incidence rates is most likely due to differences in environmental and genetic factors that affect the disease and its aetiology5.
There are several recognized aetiological agents for CCA including infection with Opisthorchis viverrini, a hepatobiliary fluke. This infection has been identified as the leading cause of CCA in Southeast Asia, including Northeast Thailand6. The International Agency for Research on Cancer within the World Health Organization (WHO), classifies O. viverrini as a Group 1 human carcinogen7. This liver fluke is widely distributed in Thailand, Lao People’s Democratic Republic (PDR), Cambodia, Vietnam and Myanmar. In Lao PDR and Thailand an estimated 10 million people are infected8,9. Infection with O. viverrini is acquired by eating raw or insufficiently cooked cyprinid fish containing O. viverrini metacercariae. Once ingestion occurs, O. viverrini commonly invade the bile ducts and cause pathological changes to the bile ducts, liver and gall bladder6. Infection with O. viverrini has been recognized as leading to several hepatobiliary diseases. These include cholangitis, obstructive jaundice, hepatomegaly, fibrosis of the periportal system, cholecystitis, and cholelithiasis and is a major aetiological agent of CCA10,11. Chronic O.viverrini infections lead to bile duct inflammation, inducing oxidative and nitrative damages of tissues and DNA, which may finally progress to CCA and death6,12,13,14. The intermediate host of the O.viverrini liver fluke is the Bithynia snail and environmental suitability for this snail host is important for the ability of the O. viverrini life cycle to continue.
To gain a more comprehensive understanding of the distribution of CCA and its determinants it is important to assess the geographic and environmental factors that may help explain the distribution. Geographical information system (GIS), remote sensing (RS) and spatial Bayesian statistical methods are tools that have been used for mapping cancers15,16,17 and diseases with an infectious origin18,19,20 that are strongly influenced by environmental characteristics including climate. The outcomes of such mapping exercises can help inform the design of large-scale disease control programmes such as is required to address CCA in Thailand. In this study, we aimed to map the distribution and identify spatial clusters of CCA in Northeast Thailand.
Results
Descriptive statistics
Table 1 and Fig. 1 describe the study participants. There were 358,981 subjects enrolled in the CASCAP project between 2013 and 2017. Of these, approximately two-third (61.2%, 219,666) were female and around one-quarter (26.2%, 93,478) were aged >60 years, with a mean age of 54.49 (SD = 9.47) years. Overall, 1,299 participants or 0.36% of the cohort were diagnosed as CCA cases. The CCA cases were more common among males than females (0.6% and 0.21%, respectively). Around 0.73% (683) of the CCA cases were >60 years old. In stratified analysis, CCA cases were most common among males aged more than 60 years old, at 1.06% (451/42,635) (Table 2).
The overall crude incidence rate of CCA for the five-year period was 2.64 cases per 100,000 population, with sub-district rates ranging from 0 to 38 cases per 100,000 population. Strong spatial variations were observed in the CCA incidence rate across sub-districts. Figure 2 shows the spatial distribution of the SMRs for CCA in Northeast Thailand at the sub-district level.
Spatial poisson regression analysis
The Bayesian spatial and non-spatial models for CCA are presented in Table 3. Using the DIC, the CCA model III with both unstructured and structured random effects was the best-fitting model. In this best-fit model (Model III), age, sex, and NDVI were significantly associated with incidence of CCA. Individuals aged >60 years were found to be 2.94 times (95% CrI: 2.62–3.31) more likely to have CCA than those aged ≤60 years, and males were 2.53 times (95% CrI: 2.24–2.85) more likely to have CCA when compared to females. NDVI was negatively associated with CCA incidence. There was an estimated decrease of 94% (95% CrI: 37%-99%) of CCA cases for 1-unit increase in NDVI. The map of the posterior means of the spatially structured random effects demonstrated evidence of spatial clustering after accounting for the model covariates (Fig. 3). A large high-risk area cluster of CCA was found in Maha Sarakham Province, which is located in the Chi and Mun River Basin. More circumscribed areas of high risk of CCA were observed in Nong Bua Lamphu, Khon Kaen and Chiyaphum Provinces. There were more isolated high risk sub-districts found in Nakhon Phanom, Sakon Nakhon and Udon Thani Provinces, which are located in the Mekong River Basin (Fig. 3A) (see Supplementary file). The unstructured random effects showed a random spatial pattern as expected (Fig. 3B).
Discussion
The current study represents a spatial analysis of the largest CCA screening program carried out to date, and which is part of the planning activities of a nation-wide opisthorchiasis and CCA control programme. We found that CCA was geographically clustered in Maha Sarakham Province. Sub-District characteristics associated with higher rates of CCA were identified including the demographic profile and NDVI.
We used data on CCA cases identified by the CASCAP program between 2013 and 2017. This data source is ideal for a number of reasons. Firstly, the CASCAP program adopts an active recruitment and screening process. This means CASCAP is able to identify CCA cases at an early stage of progression. Secondly, the CASCAP project uses the complete range of confirmatory diagnosis methods (ultrasound, MRI, CT and histopathology). This means the CASCAP data have high reliability and validity. Finally, the CASCAP project is designed as a cohort study. Those identified as at risk of developing CCA were actively followed and received regular screening giving us a more reliable source of information on CCA progression42.
CCA cases had positive associations with demographic variables in terms of increasing age and being male. Many previous research studies conducted in Thailand have also reported that CCA is common among older age groups and males6,21,22,23. Recently, Chaiteerakij et al. (2017) utilized the National Hospital Admission Data registry to identify the characteristics of CCA patients between 5 different regions of Thailand in the period 2008 and 2013. In this study, similar to our results, CCA patients in Thailand had a mean age of 64.0 ± 11.7 years and 61.2% were males. The greatest number of CCA cases (24,239; 61.5%) was found in the Northeast region23. This finding is also similar to previous research reported by Haswell-Elkins et al., (1994) which identified older aged participants (>50 years old) as having 9.21 times the odds of having CCA when compared with younger age groups (confidence interval [CI] (1.10–77.11)21,24. The exact reasons for the increased CCA risk in older age in this region are not yet fully understood. Many factors were reported such as chronic inflammation, accumulated genetic alterations, immune response and cumulative past exposure to O. viverrini infection6,25,26,27. The patterns of infection with O. viverrini in Northeast Thailand assessed by egg count, parasite-specific antibody levels and worm burden increased significantly with age28. This finding shows that the infection began at an early age. In endemic areas where there is continuous infection, age is a quantitative representation of the length of time over which liver fluke infections can occur. Males were also 3.00 times (OR 3.00; 95% CI 0.80–11.25) more likely to develop CCA than females21,22,24. Males appear more likely to incur O. viverrini infection than females, due to their behavior related to eating raw cyprinid fish, smoking behavior, alcohol consumption as well as genetics27,29, and this then leads to higher CCA rates.
We also found that risk of CCA was negatively associated with NDVI, suggesting that NDVI can explain some of the spatial distribution of CCA. The impact of NDVI on CCA is likely to relate to the environmental factors that affect snail habitats30,31. The first intermediate host for O. viverrini are Bithynia sp. snails. These snails release cercariae into water sources facilitating transmission to cyprinid fish, which harbor the infective stage to humans (metacercaria). Infection in humans then is influenced by a range of factors including the density and distribution of snail populations. These factors include the likelihood of infected human and reservoir species faeces reaching the water body, and the likely survival and success of the free swimming cercariae that exit the snail, attach and penetrate into the tissue of cyprinid fish, determine the potential infective dose to humans8,32.
Snail survival appears to be negatively associated with NDVI; as the NDVI increases, snail survival probability decreases33. Recent studies by Pratumchart and colleagues (2019) used Maximum Entropy (MaxEnt) algorithm software to generate predictive risk map of distribution pattern for B. s. goniomphalos in relation to climatic and environmental factors in Northeast Thailand. The study found that the distribution of B. s. goniomphalos was negatively correlated with NDVI34. The dominant habitat type of B. s. goniomphalos is rice paddy fields35. In Northeast Thailand, paddy fields are the most common type of land cover with patches of remnant forests, which can either evergreen and deciduous forests. The paddy fields represent low NDVI as opposed to dry evergreen forests (highest score)36. Smaller snail populations would reduce the risk of O. viverrini infection and resulting CCA, these results help to explain the relationships found in our study. In addition, the transmission dymanic of O. viverrini infection mostly occurred in rice paddy fields where the two intermediate hosts of O. viverrini are abundant and local inhabitants, and where people get infections37,38,39,40,41.
Significant clusters of CCA were located in Maha Sarakham, Nong Bua Lamphu Khon Kaen, Chiyaphum, Nakhon Phanom, Sakon Nakhon and Udon Thani Provinces. These areas lie in the basins of the Chi and Mekong Rivers, where the high prevalence of opisthorchiasis and cholangiocarcinoma have been documented6,42. Our study was undertaken using co-variates that mainly focused on environmental factors including NDVI, NDWI, LST and altitude. However, future studies should include other important factors such as soil type, soil texture and land cover that could further explain spatial variation in CCA risk.
Conclusions
This work demonstrates that older (>60 years), male individuals were at a higher risk of CCA than younger individuals and females. NDVI was associated with a reduced risk of CCA, likely related to vegetation type (with low-NDVI rice paddy areas having the highest risk). The maps of the posterior means of the spatially structured random effects of CCA demonstrated evidence of spatial clustering after accounting for the model covariates. These results can help inform the current Thai government CCA control program by allowing appropriate targeting of resources to surveillance, CCA treatment and preventive interventions targeting the O. viverrini parasite.
Methods
Study area
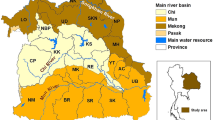
The study was undertaken in Northeast Thailand, which is located between latitudes 14.50°N and 17.50°N, and between longitudes 102.12°E and 104.90°E and which covers an area of approximately 168,854 km2(Fig. 4). This study investigates CCA distribution in all 2,678 sub-districts in all 20 provinces of Northeast Thailand, with a population of 22.24 million in 2018.
Study participants
The Cholangiocarcinoma Screening and Care Program (CASCAP) is a prospective cohort study undertaken by the Faculty of Medicine, Khon Kaen University, in cooperation with the Cholangiocarcinoma Foundation, Thailand. CASCAP program activities comprise of a prevention and patient care program among at-risk populations in high-risk areas for CCA. The three stage program includes: (1) opisthorchiasis screening, (2) CCA and periductal fibrosis screening, and (3) confirmatory diagnosis of suspected CCA patients, plus treatment and care43. Detailed recruitment procedures have been published elsewhere, but in summary, participants are enrolled actively through primary health care units if they fit the inclusion criteria, or are self-enrolled when they perceive risk of CCA and attend a health facility44.
CASCAP participants were enrolled in the CCA and periductal fibrosis screening stage of the program if they met the following criteria: (1) reside in Northeast Thailand, (2) aged ≥40 years, (3) past history of O. viverrini infection, (4) past history of treatment for O. viverrini infection with praziquantel, and (5) past history of consumption of raw or undercooked cyprinid fish44,45. The individuals at risk for O. viverrini-induced CCA underwent a liver ultrasound examination to determine their bile duct or liver pathology. Those suspected of CCA by ultrasound or clinical symptoms were further subjected to a confirmatory test/diagnosis using magnetic resonance imaging (MRI) or computerized tomography (CT) scanning and histopathological diagnosis to confirm CCA44. The patients with CCA were offered treatment and follow up care.
The study population then for this study was all CASCAP participants who received histopathological confirmation of CCA diagnosis between February 2013 and December 2017. Individual-level data for these patients were aggregated at the sub-district level, based on the CASCAP database. Sub-district populations for the study period (2013–2017) were obtained from the Official Statistics Registration Systems of Thailand website (http://stat.bora.dopa.go.th/new_stat/webPage/statByYear.php). For this analysis, CCA cases were stratified into two age groups reflecting CCA risk: ≤60 years and >60 years.
Environmental data
MODIS data products including land surface temperature (LST), normalized difference vegetation index (NDVI) and normalized difference water index (NDWI) were downloaded from the United States Geological Survey (USGS) EROS Data Center. Environmental data were collected for 1 January 2001 through 31 December 2017. Resolution was set at 0.25 km2 and data were processed to produce a mosaic covering the study area in Thailand using ArcGIS 10.5.1 (ESRI Inc., Redlands, CA, USA). Altitude data were obtained from the WorldClim database (www.worldclim.org). Polyline shapefiles for streams and rivers and a polygon shapefile for administrative boundaries at the sub-district level of Northeast Thailand were obtained from the DIVA-GIS website (www.diva-gis.org). The administrative boundary map included 2,678 sub-district level areas.
Spatial data including CCA cases, demographic and environmental data, were imported into the ArcGIS 10.5.1 and projected to the Universal Transverse Mercator (UTM) coordinate system zone 48 N. The datasets were linked according to the location of the administrative boundary map of Northeast Thailand to summarize and extract the data by sub-district area and define parameters for subsequent statistical analyses.
Data Analysis
CCA incidence rate
Firstly, a descriptive analysis of CCA cases was performed. The crude incidence rate of CCA was calculated by dividing the number of CCA cases recorded in the five-year study period (2013–2017) by the total population during the same time period. The Standardized Morbidity Ratios (SMR) for CCA were calculated for each sub-district in Northeast Thailand for 2013–2017 using the formula:
In this formula where Y represents the SMR in sub-district i, O the reported number of CCA cases in the sub-district and E the expected number of CCA cases occurring in that sub-district during the study period. To estimate the expected number of CCA cases for each sub-district the population of each sub-district was multiplied by the overall crude CCA incidence rate for the study area and period46.
Variable selection
A preliminary univariate Poisson regression of CCA case numbers was undertaken with age, sex, NDVI, NDWI, LST, altitude, and distance to water body to examine associations with CCA incidence. Age, sex, NDVI and distance from the water body were the only significant variables (p < 0.05) in the univariate models and were selected for the final models based on them having the lowest Bayesian information criterion (BIC)19,47. Finally, Pearson correlation analyses were conducted to assess collinearity among all the included variables. All the preliminary statistical analyses were performed using the STATA software version 14.0 (Stata Corporation, College Station, TX, USA).
Spatial poisson regression analysis
Zero-inflated Poisson (ZIP) regression models were built using a Bayesian framework in the WinBUGS software, version 1.4.3 (Medical Research Council, Cambridge, UK). Explanatory variables included in the first model (Model I) were age (≤60 years and >60 years), sex, NDVI and distance to water body. An unstructured random effect at the sub-district level was also included. The same explanatory variables were included in the second model (Model II) as well as a spatially structured random effect. The final model (Model III), a convolution model, included all of the variables of the preceding two models.
The convoluted model, with an outcome of observed cases of CCA (numbers), Y, for ith sub-district in Northeast Thailand (i = 1 to 2678), for the kth age group and lth sex group was structured as follows:
θikl = α + β1 × agek + β2 × sexl + β3 × NDVIi + β4 × distance to water bodyi + ui + si where Eikl is the expected number of CCA cases (offsetting population size) in sub-district i, age-group k, sex group l and θikl is the mean log relative risk (RR); α is the intercept, and β1, β2, β3, and β4 the coefficients for age (≤60 years as the reference category), sex (female as the reference category), NDVI, and distance to water body; ui is the unstructured random effect (assumed to have a mean of zero and variance σu2) and si is the spatially structured random effect (assumed to have a mean of zero and variance σs2).
In modelling the spatially structured random effects, a conditional autoregressive (CAR) prior structure was used. An adjacency weights matrix was then used to assess spatial relationships between sub-districts. Where two sub-districts shared a border, a weight of 1 was assigned, otherwise, the weight was 0. The intercept was defined utilizing a flat prior distribution. For the coefficients a normal prior distribution was used. A mean of zero was set, and precision, the inverse of variance, was set at 0.0001. Non-informative gamma distribution equal to 0.001 was used as The priors for the precision of unstructured and spatially structured random effects.
The model was run for an initial burn-in of 10,000 iterations and discarded. Then subsequent sets of 15,000 iterations were run with Monte Carlo chains until convergence was achieved. A total of three sets of 15,000 iterations were run (and discarded) before convergence was achieved as assessed by visual examination of posterior density plots and confirmed by Gelman-Rubin statistics. The best fit model was selected based on the lowest deviance information criterion (DIC), more parsimonious model. Statistical significance at an α-level of 0.05 (calcualted by 95% credible intervals (95% CrI) for relative risks (RR) that excluded 1) was used for this study47. ArcGIS 10.5.1 software was used to generate maps of the spatial distribution of posterior means of the unstructured and structured random effects. The significance level of posterior random means were calculated using the using 5% and 95% CrI of posterior random effects and a significance map was created (Fig. 5).
Ethics statement
The Khon Kaen University Ethics Committee for Human Research approved this study (HE611035). The CASCAP was conducted according to the principles of Good Clinical Practice, the Declaration of Helsinki, and national laws and regulations about clinical studies, and was approved by the Khon Kaen University Ethics Committee for Human Research under the reference number HE551404. All patients gave written informed consent for the CASCAP study.
Data Availability
The datasets generated during and/or analyzed for the current study will be made available from the corresponding author on reasonable request.
References
Banales, J. M. et al. Expert consensus document: Cholangiocarcinoma: current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat Rev Gastroenterol Hepatol 13, 261–280 (2016).
Charbel, H. & Al-Kawas, F. H. Cholangiocarcinoma: Epidemiology, Risk Factors, Pathogenesis, and Diagnosis. Current Gastroenterology Reports 13, 182–187 (2011).
Hughes, T. et al. Opisthorchiasis and cholangiocarcinoma in Southeast Asia: an unresolved problem. Int J Gen Med 10, 227–237 (2017).
Sriamporn, S. et al. Prevalence of Opisthorchis viverrini infection and incidence of cholangiocarcinoma in Khon Kaen, Northeast Thailand. Trop Med Int Health 9, 588–594 (2004).
Kirstein, M. M. & Vogel, A. Epidemiology and Risk Factors of Cholangiocarcinoma. Visc Med 32, 395–400 (2016).
Sripa, B. et al. Opisthorchiasis and Opisthorchis-associated cholangiocarcinoma in Thailand and Laos. Acta Trop 120(Suppl 1), S158–168 (2011).
IARC. IARC monographs on the evaluation of carcinogenic risks to humans Vol. 100B 341-370 (World Health Organization, International Agency for Research on Cancer, 2012).
Suwannatrai, A., Saichua, P. & Haswell, M. Epidemiology of Opisthorchis viverrini Infection. Adv Parasitol 101, 41–67 (2018).
Sithithaworn, P. et al. The current status of opisthorchiasis and clonorchiasis in the Mekong Basin. Parasitol Int 61, 10–16 (2012).
Sripa, B. et al. Liver fluke induces cholangiocarcinoma. PLoS Med 4, e201 (2007).
Mairiang, E. & Mairiang, P. Clinical manifestation of opisthorchiasis and treatment. Acta Trop 88, 221–227 (2003).
Mairiang, E. et al. Relationship between intensity of Opisthorchis viverrini infection and hepatobiliary disease detected by ultrasonography. J. Gastroenterol. Hepatol 7, 17–21 (1992).
Harinasuta, T., Riganti, M. & Bunnag, D. Opisthorchis viverrini infection: pathogenesis and clinical features. Arzneimittelforschung 34, 1167–1169 (1984).
Yongvanit, P., Pinlaor, S. & Loilome, W. Risk biomarkers for assessment and chemoprevention of liver fluke-associated cholangiocarcinoma. J Hepatobiliary Pancreat Sci 21, 309–315 (2014).
Roquette, R., Painho, M. & Nunes, B. Spatial epidemiology of cancer: a review of data sources, methods and risk factors. Geospat Health 12, 504 (2017).
Kirby, R. S., Delmelle, E. & Eberth, J. M. Advances in spatial epidemiology and geographic information systems. Ann Epidemiol 27, 1–9 (2017).
Cramb, S. M., Mengersen, K. L. & Baade, P. D. Spatio-temporal relative survival of breast and colorectal cancer in Queensland, Australia 2001–2011. Spat Spatiotemporal Epidemiol 19, 103–114 (2016).
Clements, A. C. et al. Bayesian spatial analysis and disease mapping: tools to enhance planning and implementation of a schistosomiasis control programme in Tanzania. Trop. Med. Int. Health 11, 490–503 (2006).
Cadavid Restrepo, A. M. et al. Spatiotemporal patterns and environmental drivers of human echinococcoses over a twenty-year period in Ningxia Hui Autonomous Region, China. Parasit Vectors 11, 108 (2018).
Wangdi, K., Clements, A. C. A., Du, T. & Nery, S. V. Spatial and temporal patterns of dengue infections in Timor-Leste, 2005-2013. Parasit Vectors 11, 9 (2018).
Haswell-Elkins, M. R. et al. Cross-sectional study of Opisthorchis viverrini infection and cholangiocarcinoma in communities within a high-risk area in northeast Thailand. Int J Cancer 59, 505–509 (1994).
Parkin, D. M. et al. Liver cancer in Thailand. I. A case-control study of cholangiocarcinoma. Int J Cancer 48, 323–328 (1991).
Chaiteerakij, R. et al. Characteristics and outcomes of cholangiocarcinoma by region in Thailand: A nationwide study. World J Gastroenterol 23, 7160–7167 (2017).
Kamsa-ard, S. et al. Risk Factors for Cholangiocarcinoma in Thailand: A Systematic Review and Meta-Analysis. Asian Pac J Cancer Prev 19, 605–614 (2018).
Sithithaworn, P., Yongvanit, P., Duenngai, K., Kiatsopit, N. & Pairojkul, C. Roles of liver fluke infection as risk factor for cholangiocarcinoma. J Hepatobiliary Pancreat Sci 21, 301–308 (2014).
Haswell-Elkins, M. R. et al. Immune responsiveness and parasite-specific antibody levels in human hepatobiliary disease associated with Opisthorchis viverrini infection. Clin Exp Immunol 84, 213–218 (1991).
Honjo, S. et al. Genetic and environmental determinants of risk for cholangiocarcinoma via Opisthorchis viverrini in a densely infested area in Nakhon Phanom, northeast Thailand. Int J Cancer 117, 854–860 (2005).
Haswell-Elkins, M. R., Elkins, D. B., Sithithaworn, P., Treesarawat, P. & Kaewkes, S. Distribution patterns of Opisthorchis viverrini within a human community. Parasitology 103(Pt 1), 97–101 (1991).
Steele, J. A. et al. Thinking beyond Opisthorchis viverrini for risk of cholangiocarcinoma in the lower Mekong region: a systematic review and meta-analysis. Infect Dis Poverty 7, 44 (2018).
Yang, G. J., Vounatsou, P., Tanner, M., Zhou, X. N. & Utzinger, J. Remote sensing for predicting potential habitats of Oncomelania hupensis in Hongze, Baima and Gaoyou lakes in Jiangsu province, China. Geospat Health 1, 85–92 (2006).
Zhu, H. R., Liu, L., Zhou, X. N. & Yang, G. J. Ecological Model to Predict Potential Habitats of Oncomelania hupensis, the Intermediate Host of Schistosoma japonicum in the Mountainous Regions, China. PLoS Negl Trop Dis 9, e0004028 (2015).
Upatham, E. S. A review of experimental and field research on the human liver fluke, Opisthorchis viverrini. J.Sci.Soc.Thailand 14, 245–262 (1988).
Shi, Y., Qiu, J., Li, R., Shen, Q. & Huang, D. Identification of Potential High-Risk Habitats within the Transmission Reach of Oncomelania hupensis after Floods Based on SAR Techniques in a Plane Region in China. Int J Environ Res Public Health 14 (2017).
Pratumchart, K. et al. Ecological Niche Model based on Maximum Entropy for mapping distribution of Bithynia siamensis goniomphalos, first intermediate host snail of Opisthorchis viverrini in Thailand. Acta Trop 193, 183–191 (2019).
Suwannatrai, A. et al. Effect of soil surface salt on the density and distribution of the snail Bithynia siamensis goniomphalos in northeast Thailand. Geospat Health 5, 183–190 (2011).
Thavorntam, W. & Tantemsapya, N. Vegetation greenness modeling in response to climate change for Northeast Thailand. J Geogr Sci 23, 1052–1068 (2013).
Vonghachack, Y. et al. Transmission of Opisthorchis viverrini, Schistosoma mekongi and soil-transmitted helminthes on the Mekong Islands, Southern Lao PDR. Infect Dis Poverty 6, 131 (2017).
Sato, M. et al. Patterns of trematode infections of Opisthorchis viverrini (Opisthorchiidae) and Haplorchis taichui (Heterophyidae) in human populations from two villages in Savannakhet Province, Lao PDR. J Helminthol 89, 439–445 (2015).
Madsen, H. et al. The role of rice fields, fish ponds and water canals for transmission of fish-borne zoonotic trematodes in aquaculture ponds in Nam Dinh Province, Vietnam. Parasit Vectors 8, 625 (2015).
Kim, C. S. et al. Seasonal and Spatial Environmental Influence on Opisthorchis viverrini Intermediate Hosts, Abundance, and Distribution: Insights on Transmission Dynamics and Sustainable Control. PLoS Negl Trop Dis 10, e0005121 (2016).
Phongluxa, K. et al. Helminth infection in southern Laos: high prevalence and low awareness. Parasit Vectors 6, 328 (2013).
Sripa, B. & Pairojkul, C. Cholangiocarcinoma: lessons from Thailand. Curr Opin Gastroenterol 24, 349–356 (2008).
Khuntikeo, N. et al. Current Perspectives on Opisthorchiasis Control and Cholangiocarcinoma Detection in Southeast Asia. Front Med (Lausanne) 5, 117 (2018).
Khuntikeo, N. et al. Cohort profile: cholangiocarcinoma screening and care program (CASCAP). BMC Cancer 15, 459 (2015).
Songthamwat, M. et al. Evaluating a preoperative protocol that includes magnetic resonance imaging for lymph node metastasis in the Cholangiocarcinoma Screening and Care Program (CASCAP) in Thailand. World J Surg Oncol 15, 176 (2017).
Alene, K. A., Viney, K., McBryde, E. S. & Clements, A. C. Spatial patterns of multidrug resistant tuberculosis and relationships to socio-economic, demographic and household factors in northwest Ethiopia. PLoS One 12, e0171800 (2017).
Wangdi, K. & Clements, A. C. Spatial and temporal patterns of diarrhoea in Bhutan 2003-2013. BMC Infect Dis 17, 507 (2017).
Acknowledgements
This work was supported by Khon Kaen University though the CASCAP project, the National Research Council of Thailand though the Thailand Grand Challenges: Fluke Free Thailand Project, Cholangiocarcinoma Research Institute and Cholangiocarcinoma Foundation of Thailand. The study was also supported by the Data Management and Statistical Analysis Center (DAMASAC), Faculty of Public Health, KKU, Thailand.
Author information
Authors and Affiliations
Contributions
A.T.S., A.C.A.C., B.T., N.K. and K.W. conceived and designed this study. A.T.S., K.T., K.S. and K.W. performed the analysis. A.T.S., K.T., K.S., A.C.A.C., M.K., D.J.G. and K.W. wrote the manuscript. A.T.S., K.T., N.K. and B.T. collected the data and generated the clinical database. All authors have been involved in revising the manuscript, and all authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suwannatrai, A.T., Thinkhamrop, K., Clements, A.C.A. et al. Bayesian spatial analysis of cholangiocarcinoma in Northeast Thailand. Sci Rep 9, 14263 (2019). https://doi.org/10.1038/s41598-019-50476-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-50476-7
This article is cited by
-
Human Vγ9Vδ2 T cell expansion and their cytotoxic responses against cholangiocarcinoma
Scientific Reports (2024)
-
Spatial analysis of cholangiocarcinoma in relation to diabetes mellitus and Opisthorchis viverrini infection in Northeast Thailand
Scientific Reports (2024)
-
Current prevalence and geographic distribution of helminth infections in the parasitic endemic areas of rural Northeastern Thailand
BMC Public Health (2023)
-
Associations between ultrasound screening findings and cholangiocarcinoma diagnosis in an at-risk population
Scientific Reports (2022)
-
Spatial analysis of hepatobiliary abnormalities in a population at high-risk of cholangiocarcinoma in Thailand
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.