Abstract
Tropical forests are known for their high diversity. Yet, forest patches do occur in the tropics where a single tree species is dominant. Such “monodominant” forests are known from all of the main tropical regions. For Amazonia, we sampled the occurrence of monodominance in a massive, basin-wide database of forest-inventory plots from the Amazon Tree Diversity Network (ATDN). Utilizing a simple defining metric of at least half of the trees ≥ 10 cm diameter belonging to one species, we found only a few occurrences of monodominance in Amazonia, and the phenomenon was not significantly linked to previously hypothesized life history traits such wood density, seed mass, ectomycorrhizal associations, or Rhizobium nodulation. In our analysis, coppicing (the formation of sprouts at the base of the tree or on roots) was the only trait significantly linked to monodominance. While at specific locales coppicing or ectomycorrhizal associations may confer a considerable advantage to a tree species and lead to its monodominance, very few species have these traits. Mining of the ATDN dataset suggests that monodominance is quite rare in Amazonia, and may be linked primarily to edaphic factors.
Similar content being viewed by others
Introduction
Tropical forests contain Earth’s highest levels of biodiversity. Over 250 tree species ≥10 cm diameter can be found in a 1-ha plot of the continental lowland tropics, whereas a similar area in the most diverse temperate broadleaf forest may hold 20–30 species1,2. Within such hyperdiverse tropical forests, however, patches occur that are dominated by a single tree species (hereafter “monodominant”). The earliest reports of tropical monodominant forests in the Amazon Basin were given by the explorers Spruce and Wallace3,4,5, who noted forests highly dominated by Eperua purpurea Benth. and E. leucantha Benth. on the white sands of the Upper Rio Negro Basin. Later Hamilton-Rice6 discovered large stands dominated by Peltogyne gracilipes Ducke (Fabaceae) during a 1924–25 expedition to northern Brazil. These perplexing single-dominant forests did not fit the traditional perception of uniformly tree-diverse tropical forests6. Similarly, the botanical explorer J. G. Myers, on his trek through the Pakaraima Mountains of then British Guiana, observed forests heavily dominated by either Peltogyne sp., Micrandra glabra Schultes (Euphorbiaceae), or multi-stemmed Dicymbe corymbosa Spruce ex. Benth. (Fabaceae)7. Monodominance has since been documented in all the tropical regions8,9,10,11,12,13. A stand has traditionally been considered monodominant when the number of canopy-level trees belonging to the same species is ≥60%9,14. Monodominant forests are “persistently dominant” when the dominant species dominates all strata/age classes in the stand, and will remain monodominant through time (i.e. late-successional, and not being a dominant, but transient, earlier successional stage).
Several mechanisms have been suggested to explain monodominance but a full understanding has yet to be achieved. Monodominance remains a topic of intensive research with controversial findings e.g.15,16. The term ‘classical monodominance’ was introduced by Peh17 and is defined as the occurrence of monodominant forests with environmental conditions similar to those of adjacent mixed-forests. Several studies, however, have revealed environmental differences between these forests, previously undetected. For example, soil nutrient or moisture availability may vary between monodominant and mixed forests10,11,13,18,19,20,21. Conversely, other studies have indicated that soil characteristics cannot alone explain monodominance13,22,23,24,25,26.
Peh et al.15 summarized several of these contrasting studies on different continents and constructed a conceptual mechanistic framework that could explain monodominance in tropical forests. They suggested that monodominance is likely to emerge under a combination of mechanisms. Furthermore, the combination of traits and mechanisms leading to monodominance can differ between tree species and tropical areas11,16,21,24. Peh et al.15 hypothesized potential pathways to monodominance as based on two well-studied monodominant systems. The first pathway was based on the Afro-tropical, ectomycorrhizal (EM), monodominant canopy tree Gilbertiodendron dewevrei (De Wild.) J. Léonard. The most important mechanisms described for this species were based on seedling shade-tolerance and slow decomposition of leaves, resulting in deep leaf litter. Slow decomposition and deep leaf litter affect soil nutrient cycling and could negatively influence the survival of individuals of many species. These conditions could be advantageous for large seeded trees because they have more reserves for germination11. Trees with large seeds also tend to have shade-tolerant seedlings; seedlings of G. dewevrei are well adapted to the heavily shaded understory, resulting in a competitive advantage over non-shade tolerant pioneer species11,14. While not fully considered by Peh, et al.15, it is well-established that G. dewevrei adults are heavily EM throughout their trans-Congo range and that seedlings of the species share many EM fungal symbionts with their parents27. Gilbertiodendron dewevrei, under a minimal disturbance regime, could attain monodominance, as is described by the mechanisms of Peh, et al.15. This pathway was further examined by Kazmierczak, et al.16, who constructed a model demonstrating that species can obtain monodominance by possessing the intrinsic traits of seeds with large mass and low dispersibility.
The second example pathway was based on Dicymbe corymbosa, a Neotropical species. This species is a mast fruiting tree, which shows coppicing (the formation of sprouts at the base of the tree or on the roots) of shoots and roots28. It has been suggested that there is a link between mast seeding and EM associations that would lead to satiation of seed predators and increased seedling densities29,30,31. Henkel23 indicated that EM associations might also promote coppicing of shoots and roots by enhancing host plant nutrient supplies. Peh et al. pointed out that such positive feedbacks could, over time, result in the dominance of a tree species via competitive exclusion15. While documented examples of monodominant forests exist for the South American tropics, almost all cases are currently known from the Guiana Shield of the region’s northeast. Given the immensity of greater Amazonia, it begs the questions of how widespread monodominant forests might be, what tree species are involved, where they occur, and what environmental drivers are involved.
Here we examine the occurrence of monodominance within the context of a large plot network in Amazonia, the single largest, and arguably the richest, tropical forest on earth (Fig. S1). In line with the earlier concept of hyperdominance32, which was defined by the most common species that make up half of all trees across a region, here we call a site monodominant when a single species of tree constitutes more than half of the individual trees ≥10 cm diameter at breast height (dbh) in a stand of ~1 ha. Questions addressed were: (1) How common or rare is monodominance in Amazonia? (2) Which tree species can become monodominant? (3) Does monodominance occur more frequently in certain families? (4) Which traits characterize monodominant species? (5) In which regions do monodominant trees occur? and, 6) What environmental factors may drive monodominance? Given the paucity of published records from Amazonia, we tested not only for monodominance but also for lower dominance levels. As possible causes of monodominance, we investigated four main mechanisms:
-
1)
Competitive exclusion. Monodominant forest in the tropics may develop when the forest does not experience large-scale disturbance over a long time period9. This mechanism is based on a study of Eggeling33, who compared tropical rainforest in Uganda in different successional stages. Eggeling showed that over the years, when no disturbance occurred, colonizing stands developed into climax stands with low species diversity, dominated by a few shade-tolerant species. This study led to the development of the intermediate-disturbance hypothesis (IDH)34. The IDH posits that a lack of (internal or external) disturbance leads to unconstrained succession and finally competitive exclusion, where the species that is best adapted to the environmental conditions will out-compete all other species and attain dominance34,35. Under such a mechanism, lowland tropical rainforests typically would not reach this endpoint due to frequent but spatiotemporally stochastic canopy-disturbing tree falls that allow influx of early-successional species into local gap areas, overall promoting persistence of high tree alpha-diversity. The IDH has been supported by evidence in some tropical rain forests36,37 but may have little effect on actual tree diversity38. If a lack of disturbance leads to competitive exclusion, we expect highest dominance in mature forests with the lowest numbers of pioneer species.
-
2)
Traits linked to above ground competition for light and space. Functional traits may indicate a species-specific ecological strategy. For example, seed size and wood density give an indication of a species’ mode of establishment, growth rate, and survival, and have been used to characterize pioneer versus climax species37,39. If multiple functional traits of a monodominant species differ from those of non-dominant species, they could indicate a distinctive strategy leading to monodominance. However, if the functional traits differ between monodominant species, this would suggest that different mechanisms drive the trajectory to dominance9. Other hypotheses for monodominance are associated with specific competitive traits, such as seed size, where large seeds have low dispersal ability and seedlings establish near parent trees, leading to conspecific replacement over time11,16. Furthermore, the deep litter layers that have been found in monodominant forests could act as a physical barrier for seedling establishment, where large seeds have an advantage over small-seeded species, as they have ample reserves to germinate and establish root systems11. However, small-seeded monodominant species have also been documented, and studies have shown that deep leaf litter does not always affect seedling establishment of non-dominant species. This suggests that large seeds could be a contributing, but not the sole, trait for monodominance15,40. The formation of coppices has been linked to monodominance23,28,41,42. Coppicing involves the formation of multiple shoots at the base of the tree’s stem or from the root system in the absence of major crown injury. The phenomenon allows an individual to persist indefinitely in one location, as one or more shoots may take over when the original stem dies. However, coppicing has so far been found only in a handful of species of a few Amazonian tree genera. If competitive traits lead to monodominance we expect highest dominance by species with traits that are linked with competitive ability.
-
3)
Competitive traits linked to root-soil interactions. A prominent hypothesis for how monodominance can emerge involves EM symbiosis creating a nutritional advantage for an EM-monodominant tree species with regard to establishment and survival9,43,44,45,46. The EM association consists of a mutualistic symbiosis between plant roots and fungi in which soil nutrients are provided by the fungus to the plant. It is striking that although most tropical trees are arbuscular mycorrhizal (AM), many monodominant tree species have EM associations47,48. The mechanisms behind this relationship are still not fully understood, but most likely involve plant-soil feedback mechanisms in which the local soil-litter conditions are altered in ways favouring the dominant EM tree species9,18,44,49. While both EM and AM fungi are dependent on their host plant for carbohydrate nutrition, and must obtain soil mineral nutrients for transfer to the plant, EM fungi have the enzymatic capacity to access organic forms of mineral nutrients directly from litter while avoiding major cellulolysis50. In systems dominated by EM plants, this mechanism would leave little for AM fungi, which are dependent on mineralized forms of nutrients for uptake. This implies direct competition between these fungi for mineral nutrients50,51. As a result, EM fungi may lead to slower overall decomposition (by mining of organic minerals and thus reducing the rate of saprotrophic cellulolysis) and reduced mineral nutrient availability for AM trees, this giving EM trees a competitive advantage9,52.
Reliance on the “EM mechanism” to explain tropical monodominance is, however, fraught with difficulties, as (1) EM is not exclusively found in monodominant species; (2) monodominant species also occur without EM; (3) some monodominant species possess a combination of EM and AM; and (4) EM may not necessarily slow decomposition rates in tropical forests18,44,53,54. Therefore, we also tested other root-soil interactions including nitrogen (N-) fixation and aluminium accumulation that have yet to be linked to monodominance but could confer competitive advantages on nutrient poor or toxic soils. In the tropics, N-fixation occurs primarily in Fabaceae. While fixation leads to higher nitrogen in leaves of N-fixing species, especially in the wet tropics55, N-fixing Fabaceae do not dominate the most oligotrophic Amazonian ecosystems56, instead appear to have the greatest advantage in tropical dry successional forests57. Aluminium accumulation is found predominantly in a select number of families (e.g. Rubiaceae, Melastomataceae, Myrtaceae and Vochysiaceae [for Amazonian families])58,59,60,61, including a relation with monodominance (Vochysia divergens) in wet areas in the Brazilian Pantanal62. Large numbers of non-monodominant aluminium accumulators are found in the dry Cerrado areas e.g.61. If root-soil interactions are important drivers of competition, we expect EM, nodulating or aluminium accumulating species to be monodominant more frequently than expected by chance.
-
4)
Area. All Amazon soil types present one or more stress factors to trees. For example, white sand soils are often dry and always low in nutrients, igapó and várzea both experience a single pulse of short to long-term flooding (up to 300 days), igapó soils are nutrient poor, várzea soils are nutrient rich, swamp soils are nearly permanently flooded or waterlogged with low oxygen tension, and terra firme soils are high in potentially toxic iron and aluminium. Because of several trade-offs, a tree species cannot be a good competitor on all of these soils e.g.63,64. Consequently, most common Amazonian tree species have a demonstrable preference for one of these soil types32. These tests often fail for rare species, either because they are non-preferential or are too infrequent to allow for a quantitative test32. With an assumption that all species in Amazonia have a near perfect habitat preference, we should expect that the total area of the soil types (and their level of fragmentation) has an effect on their tree species richness, with larger areas having more species65,66,67. Thus, a fourth possible mechanism for monodominance could be related to species-area relationships, where area is a controlling factor for species richness and dominance66,68. If the area of a distinct ‘edaphic forest type’ controls species richness, we expect monodominance to be more often found in plots in forests types that are small in total areal extent.
Because domestication has previously been linked to dominance in Amazonia69, we also investigated whether domesticated species are linked to monodominance.
Results
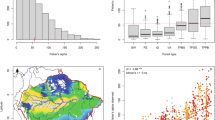
Only 50 plots (2.6% of all plots) had levels of dominance over 50% of individuals >10 cm dbh of a single tree species (Fig. 1A) - for classical monodominance [>60%] these numbers were 19 plots (0.98%). In fact only 350 plots (18% of all plots) had dominance levels over 20%. Only 26 species (0.50% of all species) attained levels of dominance of ≥50%: Eschweilera tenuifolia, Micrandra glabra, Ruizterania retusa, Pachira nitida, Machaerium hirtum, Spirotropis longifolia, Tabebuia aurea, Mauritia flexuosa, Brosimum rubescens, Lueheopsis hoehnei, Micrandra sprucei, Dicymbe corymbosa, Eperua falcata, Triplaris weigeltiana, Phyllanthus elsiae, Digomphia densicoma, Mora excelsa, Vitex cymosa, Euterpe oleracea, Oxandra polyantha, Macrolobium multijugum, Tachigali vaupesiana, Pachira brevipes, Astrocaryum macrocalyx, Attalea speciosa, Astrocaryum murumuru (for species authorities see ter Steege, et al.70). The great majority of species (4863, 97%) did not attain 20% dominance or more. Stand-level dominant species, thus, account for a tiny minority of the tree species in Amazonia (Fig. 1B). Data by species and plot are given in Appendix S1.
(A) Dominance at plot level (= relative abundance of the most abundant species of each plot) of 1946 inventory plots in Amazonia. Plots are ranked in order from high to low dominance. (B) Maximum relative abundance for each species (5029 species) found in 1946 inventory plots in Amazonia. Species are ranked from high to low maximum abundance. In each graph the red lines indicate 20 and 50% dominance.
Twelve out of 117 tree families had species which showed monodominance: Annonaceae, Arecaceae, Bignoniaceae, Euphorbiaceae, Fabaceae, Lamiaceae, Lecythidaceae, Malvaceae, Moraceae, Phyllanthaceae, Polygonaceae, Vochysiaceae (Appendix S2). Although Fabaceae species are a very prominent component of Amazonian forests32, the family had only seven monodominant species in the plots (Machaerium hirtum, Spirotropis longifolia, Eperua falcata, Dicymbe corymbosa, Mora excelsa, Macrolobium multijugum, Tachigali vaupesiana). Although this was the highest number of monodominant species by family, the number was not higher (nor lower) than expected by chance based on the number of species of Fabaceae in all plots (780). Arecaceae species are among the most hyperdominant in Amazonia32 and had five monodominant species in the plots (Mauritia flexuosa, Euterpe oleracea, Astrocaryum macrocalyx, Attalea speciosa, Astrocaryum murumuru). The randomization tests suggested that the number of families found with monodominant species did not deviate from a random expectation, except in the case of dominance over 20% and over 80%. Subsequent tests with Bonferroni correction suggested that only Arecaceae and Bignoniaceae have more species with dominance higher than 20% and only Vochysiaceae higher than 80%. Thus with monodominance defined at 50% or higher no family has more monodominant species than expected by chance. Based on the tests with Bonferroni correction alone Arecaceae showed more often dominance from 20–50%. There was no consistent family pattern in the dominance classes of 60% and higher.
Dominance by plot appeared affected by the percentage of pioneer species (Fig. 2). While there was a weak (but significant) linear relationship between the two variables (p ≪ 0.001), maximum dominance appeared constrained more by a larger number of pioneers, as exemplified by a quantile regression for the upper 10% of the data (Tau = 0.9, p = 0.035), than did the average dominance (Fig. 2). Monodominance was found only on plots with less than 0.8% pioneers. However, this result was influenced by the low number of observations that had a high abundance of pioneers. We resampled the data 10,000 times taking 40 plots randomly from the ranges 0; 0–0.2; 0.2–0.4; 0.4–0.6; 0.6–0.8 and over 0.8% of pioneers. The average slope of the upper 10% quantile was -9.2, still showing a negative relationship but the 95% c.i. of the mean included also zero slope. Hence we could not detect a significant relationship between percentage of pioneers and maximum dominance.
Dominance (= relative abundance of the most abundant species of each plot) by plot as a function of the percentage of pioneer trees in the plot. Lower red: Quantile regression line that separates the lower 10% from the upper 90% of the data (tau = 0.1, p = 0.34, i.e. slope not different from 0); middle red: quantile regression (tau = 0.5, p ≪ 0.001); upper red: quantile regression that separates the upper 10% of the data from the lower 90% (tau = 0.9, p = 0.035); black horizontal line: line of 50% dominance.
Community weighted wood density and community weighted seed mass class had little effect but the average maximum dominance was highest with the lowest and highest values of each, consistent with the traits being part of the pioneer-climax continuum (Fig. 3). Monodominance was found in 14 genera, EM in ten, nodulation in 66, aluminium accumulation in 35, and coppicing in five (Dimorphandra, Dicymbe, Euterpe, Pentaclethra, and Spirotropis). The combination of monodominance and EM was found in Dicymbe (p = 0.16); monodominance and nodulation in the three genera Machaerium, Spirotropis, and Tachigali (p = 0.46); monodominance and aluminium accumulation in Ruizterania (p = 0.35), and monodominance and coppicing in two genera (Dicymbe and Spirotropis, p = 0.007). Thus, in our data, the only ecological trait significantly linked to monodominance was coppicing. On 201 plots one of the 85 recognized Amazonian domesticated species69 was the most abundant species (Appendix S3). In almost all cases (173 plots) and in all cases with a dominance over 30% this most dominant species was an Arecaceae species (Appendix S3). Theobroma cacao was the most dominant species on 10 plots.
The highest percentages (outliers) of trees belonging to potential EM genera were found in white sand forest (PZ) and/or the Guiana Shield (Fig. 4). This forest type and region had both the highest median values as well as most of the high values for percentage EM. However, forest type explained only 2.6% (p ≪ 0.001) of the variation in EM percentage, and region 1.1% (p ≪ 0.001).
Maximum dominance was highest on those soil types with the smallest area in Amazonia (Fig. 5A). Each of the smaller forest types had higher median maximum dominance than terra firme. Forest type explained 28% (p ≪ 0.001) of the maximum dominance by plot (ANOVA). Median dominance was strongly related (power function) to area (Fig. S5). Region had only a very small effect (3% explained variation, p ≪ 0.001, Fig. 5B).
Discussion
Monodominance (defined here as ≥50% of individuals ≥10 cm dbh in a stand belong to a single tree species) appears to be quite rare over the greater Amazonian region. In the ATDN analysis presented here, a very small percentage of all plots (2.6%) and species (0.5%) exhibited monodominance by the above definition, and even less, at 1.0% and 0.3%, respectively, under the definition of classical monodominance sensu Peh, et al.15. Even dominance between 20% and 50% was not common. The overall scarcity of monodominance at plot and species level may partially be a reflection of lack of specific sampling throughout the ATDN dataset. If tree plots were not set up to capture stands with clearly dominant tree species but rather set up to capture tree-diverse forest types, the dominant stands would be “missed”. Some ATDN plots, however, were set up to study monodominance, such as those dominated by Brosimum rubescens13, Dicymbe corymbosa23, Spirotropis longifolia42 and studies of plant communities in white sand systems of Guyana and Suriname71. In general we believe most plots were not selected on the basis of selecting or avoiding monodominance. Records do exist for some dominant tree species that were not confirmed as monodominant or were not captured in the ATDN plot data. For example, Dicymbe altsonii and Dicymbe jenmanii are each dominant to monodominant in parts of Guyana46,72,73, as well as Pakaraimaea dipterocarpacea in W. Guyana/E. Venezuela72,74, Aldina spp. in N. Brazil/S. Venezuela/W. Guyana75,76,77,78, and Pseudomonotes tropenbosii in E. Colombia (Aida Vasco-Palacios pers. comm.). All of these genera are confirmed EM46,75. Several ATDN plots have been established in peat swamps, the habitat type that probably accounts for the largest area of monodominant forests in Amazonia, within which only Mauritia flexuosa and Pachira nitida were able to attain monodominance79. Micrandra spp. (Euphorbiaceae) are also known to strongly dominate poorly-drained soils in W. Guyana80,81, T.W. Henkel pers. obs. and adjacent Venezuela82. All told, the overall ranking of dominance suggests a rather smooth transition across all dominance levels (Fig. 1).
Only two families had more species at dominance classes over 20% than expected by chance. The most consistent family with significant dominance (based on Bonferroni correction alone) from 20% to 50% is Arecaceae, including 20/74 species in our data. This is consistent with Arecaceae also having a five times higher than expected number of hyperdominant species32, reflecting their regularly high local dominance. Palms may reach high dominance because they are competitive in large wet areas but they must also be fairly resistant to frequency dependent mortality, as should other hyperdominant and monodominant species. For all other families the monodominance level is rather unpredictable, so we have no reason to suggest that certain families have a predisposition for monodominance in Amazonian forests.
Disturbance, as measured by its proxy pioneer abundance, and traits related to the pioneer-climax continuum had no significant effect on dominance or diversity, contrary to findings in an earlier Afro-tropical study in Ghana38 and a study of the effects of gap-scale disturbance in Amazonian forest that found a very small effect of disturbance on diversity or dominance83. Two French Guiana studies that used pioneer species as surrogates for disturbance regime found a stronger relationship36,84. Thus as in our data monodominance was only observed in plots with very low abundance of pioneers (Fig. 2), this was not a significant pattern.
In our analysis coppicing was the only trait significantly linked to monodominance. Coppicing occurs in many species after logging or clear felling but many coppices eventually die (HtS pers. obs.). Coppicing is not common as a natural means of regeneration and has been observed mainly in species of Fabaceae genera (Dimorphandra, Dicymbe, Pentaclethra, Spirotropis), one palm (Euterpe oleracea), Humiria and Theobroma cacao85. In Dimorphandra and Humiria coppicing is a rapid response to fire damage and species of each can become dominant in Guyana and Suriname in fire-prone savannah-forest ecotones86. In closed-canopy forest they are also found as non-coppicing tall trees. Dicymbe species exhibit both EM and very pronounced coppicing in the absence of mechanical disturbance, especially in D. corymbosa, and to a lesser extent D. altsonii87. Woolley, et al.28 hypothesised that the coppicing in D. corymbosa was an evolved response to persistent infections with heart-rot fungi, the adaptive significance being that the coppicing insures persistence of the individual beyond that which would occur with a heart-rotted, single-bole tree. Thus, while coppicing was observed as an important reproduction strategy for some Amazonian tree species or as a major regeneration process in secondary growth forests, none of the above can answer the obvious question as to why more species do not spontaneously coppice in mature forest.
Similar questions can be asked for the EM habit. Seedlings of species with access to an EM network may have higher survivorship, growth and reduced density-dependent mortality relative to AM trees45. Ectomycorrhizal associations may also provide a competitive edge by directly accessing organic forms of nutrients in litter, leaving little for saprotrophic fungi or AM mycorrhizae88. However, Mayor & Henkel (2006) used reciprocal litter transplants in Dicymbe monodominant forest and mixed AM-dominated forests and found no differences in litter decomposition rate between the forest types, or within the Dicymbe forest between trenched (EM-absent) and non-trenched (EM-present) plots. Conversely, McGuire, et al.89 found slower litter decomposition in monodominant Dicymbe forest, and lower richness of saprotrophic fungi than in adjacent mixed forest. Although EM has often been linked mechanistically to monodominance9,43,44,45,46, we did not find a significant relationship between monodominance and EM in the ATDN analysis. This contrasts with the review of Corrales, et al.48 in which both monodominance and confirmed mycorrhizal type were linked in both the Paleo- and Neotropics, and the majority of fully documented monodominant tree species were EM.
Neither nodulation nor aluminium accumulation were significantly related to monodominance. While N-fixing arguably should confer a large benefit on nitrogen-limited soils, none of the monodominant Fabaceae fix nitrogen, as is the case in the Fabaceae in the wet Afrotropics, where in contrast to Amazonia most dominant Fabaceae are EM e.g.90. At an Amazonia-wide scale Fabaceae dominance and N-fixing appear negatively correlated, and N-fixing Fabaceae do not dominate the most oligotrophic Amazonian ecosystems56. N-fixing is more prominent in forests richer in species56 and appears to have the greatest advantage in tropical dry successional forests57. Aluminium accumulation is found predominantly in a select number of families (e.g. Rubiaceae, Melastomataceae, Myrtaceae and Vochysiaceae; see references in Introduction) and one species (Ruizteranea retusa) was found as monodominant in our plots in southern Amazonia, while another monodominant species, Vochysia divergens, has been observed in the Brazilian Pantanal62. While aluminium accumulators are found abundantly in the Cerrado south of Amazonia e.g.61, they appear rare in wet Amazonian forests.
Although domesticated species were the most dominant species on 201 of the 1946 plots, in almost all cases these were Arecaceae, which tend to dominate large stretches of swamp forest in Amazonia (Oenocarpus bataua, Euterpe oleracea, Mauritia flexuosa) and in the case of monodominance only Euterpe oleracea and Mauritia flexuosa. With regard to Mauritia flexuosa this species had already attained high prominence in the Amazonian landscape prior to the arrival of humans91,92.
Area had a strong effect on dominance. The ‘forest type’ with the smallest areal coverage had by far the highest mean dominance (Fig. S5). It has been argued before that smaller ecosystems in Amazonia would have lower overall tree diversity and more dominant species66,93. This would be in line with ecological theories where equilibria of immigration and extinction maintain diversity94,95. Connell and Lowman9 noted that “Single-species dominance is of less interest in regions that have smaller species pools” and did “not consider tropical forests at high altitudes, on small islands, or with low or very seasonal rainfall and/or extreme soil conditions, for example, frequently flooded freshwater swamps or mangrove forests, all habitats with few species”. In the Amazon, however, this may be the most common road to monodominance. Nascimento, et al.19 also argued that drainage and other edaphic factors drive monodominance of Peltogyne gracilipes in one Amazonian forest. Similarly, Draper, et al.79 argued that the extreme environmental conditions of Amazonian peatland forests (waterlogging and low fertility), contributed to monodominance of Mauritia flexuosa and Pachira nitida. In the case of classical monodominance of the Congolian G. dewevrei, the discussion is ongoing. Kearsley, et al.21 suggested that “environmental filtering prevailed in the monodominant G. dewevrei forest, leading to lower functional diversity in this forest type, with the dominant species showing beneficial traits related to its common riverine locations and with reduced soil N and P availability found in this environment, both co-regulating the tree community assembly”. Others, however, found no edaphic differences between the monodominant G. dewevrei forest and adjacent mixed forest25,26, a result also found with monodominant D. corymbosa in Guyana23,46. Environmental filtering would also not explain the extreme monodominance of G. dewevrei over hundreds of km2 of upland area in the Congo region11,96.
We were unable to test for basal area monodominance here as the majority of plots in the ATDN lack stem diameter measurements. Instead we used the number of individuals as our metric to determine monodominance. While this is an easily available measure, others have taken basal area and estimates of above-ground biomass as metrics. Monodominant species can differ at the plot level, e.g. D. corymbosa can have less than 60% of all individuals ≥ 10 cm dbh in some plots, but exhibit 80–90% basal area dominance, due to its complete dominance of individuals in the very large size classes23. Such a stand is still most definitely monodominant, in terms of a single species commanding the majority of site resources, and in these same stands conspecifics will always be dominant in the seedling and sapling classes. Connell and Lowman9 pointed out that monodominance can be defined as a single species comprising >60% of individuals >10 cm dbh, or >60% of stand basal area, or both, and be considered “monodominant”.
While seed mass, shade tolerance, and longevity may theoretically lead to monodominance15,16, we find little support for these traits as being causal to monodominance. While monodominance can be mechanistically related to EM and coppicing, very few tree species have used these traits to dominate Amazonian forests. Large stands dominated by single species appear linked primarily to edaphic factors, such as swamps (e.g. many palm species), nutrient poor floodplains (Eschweilera tenuifolia, Macrolobium, Triplaris, Symphonia) and soils with poor drainage (Micrandra spp.)80, white sands (Dicymbe, Eperua, Aldina), soil chemical constraints (Peltogyne, Brosimum), or may be related to fire history (Dimorphandra).
In summary, we found that monodominance, as defined by stem abundance, is extremely rare in Amazonian tropical forests, at least within the extensive ATDN dataset, and found little support for a single mechanism for monodominance. The occurrence of monodominance was most strongly linked to metacommunity dynamics of small rare ecosystems, such as white sands, peats and flooded areas. Because the edaphic differences of the forest types with smaller fragmented areas (white sand forests, Várzea, Igapó and swamp forests) with the major forest type in the Amazon (terra firme), the “forest types” in Amazonia have their to a large extent a distinct tree flora. Within these areas dominance may be in part attributed to chance – the smaller and more fragmented the forest type area, the higher the chance for local dominance.
Material and Methods
All tree data were derived from the Amazon Tree Diversity Network (ATDN, http://atdn.myspecies.info/), comprised of a long-term data set now containing >2000 tree inventory plots across Amazonia. Our analyses were based on 1946 plots, comprised of 127 families, 798 genera, and 5027 identified tree species. All analyses were performed using the R programming language97.
Firstly, dominance was calculated by plot. Dominance was defined as the relative abundance of the most abundant tree species within the community and was calculated as:
where Nd is the number of individuals of the most abundant species and Ntot the total number of individuals in the tree plot (Dominance calculated this way is also known as the Berger-Parker index). We calculated rank dominance curves for dominance based on plots and mapped dominance across Amazonia. To study which families have more dominant species than expected by chance we listed all species with dominance over eight dominance classes (20–90%) by family. Then, with a Monte Carlo randomization test (1000 randomizations) we determined which tree families have more dominant/competitive species in each dominance class than expected by chance (based on the total number of tree species in the family). Maximum relative abundance of each species was also calculated, thus including species that were never the most dominant species in a plot. As we carried out as many tests as there are families at an error level of 5%, we can expect that at least 5% of the families may become false positives. We tested this by calculating for each of the 1000 randomizations how many families met this criterion and calculated mean and standard deviation. If the number of families found was significantly higher than this mean we applied Bonferroni correction (adjusting p as (p/number of families)), to find those families that were most likely to be the true positives of this test.
To test for competitive exclusion as a mechanism for dominance we used the percentage of pioneer species (log transformed to normalize the data) at plot level, as a proxy for disturbance36,38,84,98,99. We identified pioneers by combining low wood density and low seed mass under the condition (WD < 0.7 Λ SMC < 4, Fig. S2) sensu99. We used a loess regression to test for a relationship between the disturbance proxy and dominance at plot level.
To test if particular traits are linked to monodominance we examined two traits, wood density and seed mass, that are generally linked to longevity and dominance39,100. We calculated the community weighted average for both wood density and seed size as follows:
where CWA is the community weighted average, ∑Ni is the sum of the number of individuals with trait data, trait is the corresponding trait value on genus level for either wood density or seed mass class and ∑N is the total number of individuals in the tree community. We then carried out a loess regression to assess the relationship between the CWA of the functional traits and dominance.
To test if an EM association may lead to dominance we checked the most recent literature for confirmed EM tree species101. We tested if EM is more abundant on monodominant plots and if EM species are more likely to be monodominant. For a similar test for nodulation we used Sprent102 and Soltis, et al.103. For aluminium accumulation we used Jansen et al. (2002, 2003) and references therein. There is no single source for intrinsic coppicing, a means of persistence once an individual is established, in tropical trees - this information was collected from observations on our plots. We tested the association with Monte Carlo randomizations (n = 10,000).
To test if an area effect may lead to (mono-)dominance we used ANOVA to test if monodominance is more common in the forest types that have a smaller extent in Amazonia: white sand forest (4.6%)104, igapó and várzea (10%)104,105 and swamps (1.7%)106, compared to terra firme which covers most of the remaining area.
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Gentry, A. H. Tropical forest biodiversity: distributional patterns and their conservational significance. Oikos, 19–28 (1992).
Condit, R. et al. Species-area and species-individual relationships for tropical trees: A comparison of three 50-ha plots. Journal of Ecology 84, 549–562 (1996).
Wallace, A. R. A narrative of travels on the Amazon and Río Negro, with an account of the native tribes, and observations on the climate, geology, and natural history of the Amazon Valley (pp. 363. Reeve and Co, London, 1853).
Spruce, R. Letter to Joseph Hooker dated 19 March 1854. Hooker’s Journal of Botany and Kew Garden Miscellany 6, 333–337 (1854).
Spruce, R. Notes on some insect and other migration observed in Equatorial America. Journal of the Linnean Society (Zoology) 9, 346–267 (1868).
Hamilton-Rice, A. The Rio Branco, Uraricuera and Parima. The Geographical Journal 71, 113–143, 209–223, 345–357 (1928).
Myers, J. G. Savanna and forest vegetation of the Interior Guiana Plateau. Journal of Ecology 24, 162–184 (1936).
Richards, P. W. The Tropical Rain Forest: An Ecological Study. 2nd edition. (Cambridge University Press, 1996).
Connell, J. H. & Lowman, M. D. Low-diversity tropical rain forests: some possible mechanisms for their existence. The American Naturalist 134, 88–119 (1989).
Nascimento, M. T. & Proctor, J. Soil and plant changes across a monodominant rain forest boundary on Maracá Island, Roraima, Brazil. Global Ecology and Biogeography Letters 6, 387, https://doi.org/10.2307/2997339 (1997).
Torti, S. D., Coley, P. D. & Kursar, T. A. Causes and consequences of monodominance in tropical lowland forests. Am Nat 157, 141–153, https://doi.org/10.1086/318629 (2001).
Davis, T. W. & Richards, P. W. The vegetation of Moraballi Creek, British Guiana: an ecological study of a limited area of tropical rain forest. Part II. Journal of Ecology 22, 106–155 (1934).
Marimon, B. S., Felfili, J. M. & Haridasan, M. Studies in monodominant forests in eastern Mato Grosso, Brazil: I. A forest of Brosimum rubescens Taub. Edinburgh Journal of Botany 58, 123–137 (2001).
Hart, T. B., Hart, J. A. & Murphy, P. G. Monodominant and species-rich forests of the humid tropics: causes for their co-occurrence. The American Naturalist 133, 613–633 (1989).
Peh, K. S.-H., Lewis, S. L. & Lloyd, J. Mechanisms of monodominance in diverse tropical tree‐dominated systems. Journal of Ecology 99, 891–898 (2011).
Kazmierczak, M. et al. Monodominance in tropical forests: modelling reveals emerging clusters and phase transitions. Journal of the Royal Society, Interface 13, 20160123, https://doi.org/10.1098/rsif.2016.0123 (2016).
Peh, K. S.-H. The relationship between species diversity and ecosystem function in low-and high-diversity tropical African forests PhD thesis, University of Leeds, (2009).
Corrales, A., Mangan, S. A., Turner, B. L. & Dalling, J. W. An ectomycorrhizal nitrogen economy facilitates monodominance in a neotropical forest. Ecology Letters 19, 383–392, https://doi.org/10.1111/ele.12570 (2016).
Nascimento, M. T. et al. Is the Peltogyne gracilipes monodominant forest characterised by distinct soils? Acta Oecologica 85, 104–107, https://doi.org/10.1016/j.actao.2017.10.001 (2017).
Fayolle, A. et al. A new insight in the structure, composition and functioning of central African moist forests. Forest Ecology and Management 329, 195–205 (2014).
Kearsley, E. et al. Functional community structure of African monodominant Gilbertiodendron dewevrei forest influenced by local environmental filtering. Ecology and Evolution 7, 295–304, https://doi.org/10.1002/ece3.2589 (2017).
Newbery, D., Alexander, I. J. & Rother, J. A. Phosphorus dynamics in a lowland African rainforest: The influence of ectomycorrhizal trees. Ecological Monographs 67, 367–409 (1997).
Henkel, T. W. Monodominance in the ectomycorrhizal Dicymbe corymbosa (Caesalpiniaceae) from Guyana. Journal of Tropical Ecology 19, 417–437 (2003).
Peh, K. S.-H., Sonké, B., Lloyd, J., Quesada, C. A. & Lewis, S. L. Soil does not explain monodominance in a Central African tropical forest. PLoS One 6, e16996 (2011).
Conway, D. & Alexander, I. J. Soil conditions under monodominant Gilbertiodendron dewevrei and mixed forest Ituri forest reserve, Zaire. Tropical Biology Newsletter, unpaginated (1992).
Lokonda, M., Freycon, V., Gourlet-Fleury, S. & Kombele, F. Are soils under monodominant Gilbertiodendron dewevrei and under adjacent mixed forests similar? A case study in the Democratic Republic of Congo. Journal of Tropical Ecology 34, 176–185, https://doi.org/10.1017/s0266467418000135 (2018).
Ebenye, M. H. C. et al. Ectomycorrhizal fungi are shared between seedlings and adults in a monodominant Gilbertiodendron dewevrei rain forest in Cameroon. Biotropica 49, 256–267, https://doi.org/10.1111/btp.12415 (2017).
Woolley, L. P., Henkel, T. W. & Sillett, S. C. Reiteration in the monodominant tropical tree Dicymbe corymbosa (Caesalpiniaceae) and its potential adaptive significance. Biotropica 40, 32–43 (2008).
Henkel, T. W., Mayor, J. R. & Woolley, L. P. Mast fruiting and seedling survival of the ectomycorrhizal, monodominant Dicymbe corymbosa (Caesalpiniaceae) in Guyana. New Phytologist 167, 543–556, https://doi.org/10.1111/j.1469-8137.2005.01431.x (2005).
Newbery, D. M. Ectomycorrhizas and mast fruiting in trees: Linked by climate‐driven tree resources? New Phytologist 167, 324–326 (2005).
Henkel, T. W. & Mayor, J. R. Implications of a long-term mast seeding cycle for climatic entrainment, seedling establishment, and persistent monodominance in a Neotropical, ectomycorrhizal canopy tree. Ecological Research 34, 472–484 (2019).
ter Steege, H. et al. Hyperdominance in the Amazonian tree flora. Science 342, 1243092, https://doi.org/10.1126/science.1243092 (2013).
Eggeling, W. J. Observations on the ecology of the Budongo rain forest, Uganda. Journal of Ecology 34, 20–87 (1947).
Connell, J. H. Diversity in tropical rain forests and coral reefs. Science 199, 1302–1310, https://doi.org/10.1126/science.199.4335.1302 (1978).
Sheil, D. & Burslem, D. F. R. P. Disturbing hypotheses in tropical forests. Trends in Ecology & Evolution 18, 18–26 (2003).
Molino, J. F. & Sabatier, D. Tree diversity in tropical rain forests: a validation of the intermediate disturbance hypothesis. Science 294, 1702–1704, https://doi.org/10.1126/science.1060284 (2001).
ter Steege, H. & Hammond, D. S. Character convergence, diversity, and disturbance in tropical rain forest in Guyana. Ecology 82, 3197–3212 (2001).
Bongers, F., Poorter, L., Hawthorne, W. D. & Sheil, D. The intermediate disturbance hypothesis applies to tropical forests, but disturbance contributes little to tree diversity. Ecology Letters 12, 798–805, https://doi.org/10.1111/j.1461-0248.2009.01329.x (2009).
Swaine, M. D. & Whitmore, T. C. On the definition of ecological species groups in tropical forests. Vegetatio 75, 81–86 (1988).
Martijena, N. E. Soil properties and seedling establishment in soils from monodominant and high‐diversity stands of the tropical deciduous forests of México. Journal of Biogeography 25, 707–719 (1998).
Gaddis, K. D., Zukin, H. L., Dieterich, I. A., Braker, E. & Sork, V. L. Effect of clonal reproduction on genetic structure in Pentaclethra macroloba (Fabaceae: Mimosoideae). Revista de Biología Tropical 62, 443–454 (2014).
Fonty, E., Molino, J. F., Prévost, M. F. & Sabatier, D. A new case of neotropical monodominant forest: Spirotropis longifolia (Leguminosae-Papilionoideae) in French Guiana. Journal of Tropical Ecology 27, 641–644 (2011).
Singer, R. & Araujo, I. J. S. Litter decomposition and ectomycorrhiza in Amazonian forests. 1. A comparison of litter decomposing and ectomycorrhizal basidiomycetes in latosol-terra-firme rain forest and white podzol campinarana. Acta Amazonica 9, 25–42 (1979).
Torti, S. D. & Coley, P. D. Tropical monodominance: A preliminary test of the ectomycorrhizal hypothesis. Biotropica 31, 220–228 (1999).
McGuire, K. L. Common ectomycorrhizal networks may maintain monodominance in a tropical rain forest. Ecology 88, 567–574 (2007).
Henkel, T. W., Terborgh, J. & Vilgalys, R. J. Ectomycorrhizal fungi and their leguminous hosts in the Pakaraima Mountains of Guyana. Mycological Research 106, 515–531 (2002).
McGuire, K. L., Allison, S. D., Fierer, N. & Treseder, K. K. Ectomycorrhizal-dominated boreal and tropical forests have distinct fungal communities, but analogous spatial patterns across soil horizons. PLoS One 8, e68278 (2013).
Corrales, A., Henkel, T. W. & Smith, M. E. Ectomycorrhizal associations in the tropics - biogeography, diversity patterns and ecosystem roles. New phytologist 220, 1076–1091, https://doi.org/10.1111/nph.15151 (2018).
Fukami, T. et al. Geographical variation in community divergence: Insights from tropical forest monodominance by ectomycorrhizal trees. American Naturalist 190, S105–S122, https://doi.org/10.1086/692439 (2017).
Hobbie, E. A. & Hogberg, P. Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New phytologist 196, 367–382, https://doi.org/10.1111/j.1469-8137.2012.04300.x (2012).
Averill, C., Turner, B. L. & Finzi, A. C. Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nature 505, 543–545 (2014).
Phillips, R. P., Brzostek, E. & Midgley, M. G. The mycorrhizal‐associated nutrient economy: a new framework for predicting carbon–nutrient couplings in temperate forests. New Phytologist 199, 41–51 (2013).
Torti, S. D., Coley, P. & Janos, D. P. Vesicular-arbuscular mycorrhizae in two tropical monodominant trees. Journal of Tropical Ecology 13, 623–629 (1997).
Mayor, J. R. & Henkel, T. W. Do ectomycorrhizas alter leaf-litter decomposition in monodominant tropical forests of Guyana? New Phytologist 169, 579–588 (2006).
Adams, M. A., Turnbull, T. L., Sprent, J. I. & Buchmann, N. Legumes are different: Leaf nitrogen, photosynthesis, and water use efficiency. Proceedings of the National Academy of Sciences 113, 4098–4103, https://doi.org/10.1073/pnas.1523936113 (2016).
ter Steege, H. et al. Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 443, 444–447, https://doi.org/10.1038/nature05134 (2006).
Gei, M. et al. Legume abundance along successional and rainfall gradients in Neotropical forests. Nat Ecol Evol 2, 1104–1111, https://doi.org/10.1038/s41559-018-0559-6 (2018).
Chenery, E. M. & Sporne, K. R. A note on the evolutionary status of aluminium‐accumulators among Dicotyledons. New Phytologist 76, 551–554 (1976).
Jansen, S., Watanabe, T., Dessein, S., Smets, E. & Robbrecht, E. A comparative study of metal levels in leaves of some Al‐accumulating Rubiaceae. Annals of Botany 91, 657–663 (2003).
Jansen, S., Watanabe, T. & Smets, E. Aluminium accumulation in leaves of 127 species in Melastomataceae, with comments on the order Myrtales. Ann Bot 90, 53–64 (2002).
Haridasan, M. & De Araújo, G. M. Aluminium-accumulating species in two forest communities in the cerrado region of central Brazil. Forest Ecology and Management 24, 15–26 (1988).
Nascimento, M. T. & Cunha, C. N. Estrutura e composição florística de um cambarazal no pantanal de Poconé-MT. Acta Botanica Brasilica 3, 03–23 (1989).
Fine, P. V., Mesones, I. & Coley, P. D. Herbivores promote habitat specialization by trees in Amazonian forests. Science 305, 663–665, https://doi.org/10.1126/science.1098982 (2004).
Herrera, R., Jordan, C. F., Klinge, H. & Medina, E. Amazon ecosystems. Their structure and functioning with particular emphasis on nutrients. Interciencia 3, 223–231 (1978).
Rosenzweig, M. L. Species Diversity in Space and Time. (Cambridge University Press, 1995).
ter Steege, H. et al. An analysis of the floristic composition and diversity of Amazonian forests including those of the Guiana Shield. Journal of Tropical Ecology 16, 801–828 (2000).
Fine, P. V. A. Ecological and evolutionary drivers of geographic variation in species diversity. Annual Review of Ecology, Evolution, and Systematics 46, 369–392 (2015).
Stropp, J., Sleen, P. V., Assunção, P. A., Silva, A. L. & Steege, H. T. Tree communities of white-sand and terra-firme forests of the upper Rio Negro. Acta Amazonica 41, 521–544 (2011).
Levis, C. et al. Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 355, 925–931, https://doi.org/10.1126/science.aal0157 (2017).
ter Steege, H. et al. Towards a dynamic list of Amazonian tree species. Sci Rep 9, 3501, https://doi.org/10.1038/s41598-019-40101-y (2019).
Bánki, O. S. Does neutral theory explain community composition in the Guiana Shield forests?, Dept. Biology, Utrecht University, (2010).
Smith, M. E. et al. The ectomycorrhizal fungal community in a neotropical forest dominated by the endemic dipterocarp Pakaraimaea dipterocarpacea. PLoS One 8, e55160, https://doi.org/10.1371/journal.pone.0055160 (2013).
Zagt, R. J. Pre-dispersal and early post-dispersal demography, and reproductive litter production, in the tropical tree Dicymbe altsonii in Guyana. Journal of Tropical Ecology 13, 511–526 (1997).
Moyersoen, B. Pakaraimaea dipterocarpacea is ectomycorrhizal, indicating an ancient Gondwanaland origin for the ectomycorrhizal habit in Dipterocarpaceae. New Phytologist 172, 753–762 (2006).
McGuire, K. L. et al. Dual mycorrhizal colonization of forest-dominating tropical trees and the mycorrhizal status of non-dominant tree and liana species. Mycorrhiza 18, 217–222, https://doi.org/10.1007/s00572-008-0170-9 (2008).
Smith, M. E., Henkel, T. W., Catherine Aime, M., Fremier, A. K. & Vilgalys, R. Ectomycorrhizal fungal diversity and community structure on three co-occurring leguminous canopy tree species in a Neotropical rainforest. The New phytologist 192, 699–712, https://doi.org/10.1111/j.1469-8137.2011.03844.x (2011).
Moyersoen, B. Ectomicorrizas y micorrizas vesículo-arbusculares en Caatinga Amazónica del Sur de Venezuela. Scientia Guaianae 3, 1–82 (1993).
Mardegan, S. F., Nardoto, G. B., Higuchi, N., Moreira, M. Z. & Martinelli, L. A. Nitrogen availability patterns in white-sand vegetations of Central Brazilian Amazon. Trees Structure and Function 23, 479–488 (2009).
Draper, F. C. et al. Peatland forests are the least diverse tree communities documented in Amazonia, but contribute to high regional beta-diversity. Ecography 41, 1256–1269, https://doi.org/10.1111/ecog.03126 (2018).
Degagne, R. S., Henkel, T. W., Steinberg, S. J. & Fox Iii, L. Identifying Dicymbe corymbosa monodominant forests in Guyana using satellite imagery. Biotropica 41, 7–15, https://doi.org/10.1111/j.1744-7429.2008.00446.x (2009).
ter Steege, H. Mapping Forest vegetation in Guyana at Regional and National Level. (Guyana Forestry Commission, Georgetown, 2001).
Huber, M. O. In Flora of the Venezulean Guyana. Volume 1. Introduction Vol. 1 (eds Steyermark, J. A., Berry, P. E. & Holst, B. K.) 97–160 (Timber Press Inc., 1995).
Baker, T. R. et al. Consistent, small effects of treefall disturbances on the composition and diversity of four Amazonian forests. Journal of Ecology 104, 497–506, https://doi.org/10.1111/1365-2745.12529 (2016).
Guitet, S. et al. Disturbance regimes drive the diversity of regional floristic pools across Guianan rainforest landscapes. Sci Rep 8, 3872, https://doi.org/10.1038/s41598-018-22209-9 (2018).
Sabatier, D. & Prévost, M. F. Une forêt a cacaoyers sauvages sur le haut-camopi, en Guyane Française. (1987).
ter Steege, H. & Zondervan, G. In Plant diversity in Guyana. with recommendations for a National Protected Area Strategy (ed. ter Steege, H.) 35–54 (Tropenbos International, 2000).
Zagt, R. J. Tree demography in the tropical rainforest of Guyana PhD thesis, Utrecht University, (1997).
Smith, S. E. & Read, D. J. Mycorrhizal Symbiosis. (Academic Press, Elsevier, 2010).
McGuire, K. L., Zak, D. R., Edwards, I. P., Blackwood, C. B. & Upchurch, R. Slowed decomposition is biotically mediated in an ectomycorrhizal, tropical rain forest. Oecologia 164, 785–795, https://doi.org/10.1007/s00442-010-1686-1 (2010).
Newbery, D. M., Alexander, I. J., Thomas, D. W. & Gartlan, J. S. Ectomycorrhizal rain-forest legumes and soil phosphorus in Korup National Park, Cameroon. New Phytologist 109, 433–450, https://doi.org/10.1111/j.1469-8137.1988.tb03719.x (1988).
de Lima, N. E., Lima-Ribeiro, M. S., Tinoco, C. F., Terribile, L. C. & Collevatti, R. G. Phylogeography and ecological niche modelling, coupled with the fossil pollen record, unravel the demographic history of a Neotropical swamp palm through the Quaternary. Journal of Biogeography 41, 673–686, https://doi.org/10.1111/jbi.12269 (2014).
Rull, V. Biogeographical and evolutionary considerations of Mauritia (Arecaceae), based on palynological evidence. Review of Palaeobotany and Palynology 100, 109–122 (1998).
Stropp, J., ter Steege, H. & Malhi, Y. ATDN & RAINFOR. Disentangling regional and local tree diversity in the Amazon. Ecography 32, 46–54 (2009).
MacArthur, R. R. & Wilson, E. O. The theory of Island Biogeography. (Princeton University Press, 1967).
Hubbell, S. P. The Unified Neutral Theory of Biodiversity and Biogeography. (Princeton University Press, 2001).
Hart, T. B. Seed, seedling and sub-canopy survival in monodominant and mixed forests of the Ituri Forest, Africa. Journal of Tropical Ecology 11, 443–459, https://doi.org/10.1017/s0266467400008919 (2009).
R: A language and environment for statistical computing (R Foundation for Statistical Computing, Vienna, Austria, 2017).
Rödig, E., Cuntz, M., Heinke, J., Rammig, A. & Huth, A. The importance of forest structure for carbon flux estimates in the Amazon rainforest. Environmental Research Letters 26, 1292–1302 (2018).
ter Steege, H., Welch, I. & Zagt, R. J. Long-term effect of timber harvesting in the Bartica Triangle, Central Guyana. Forest Ecology and Management 170, 127–144 (2002).
Keddy, P. A. Assembly and response rules: two goals for predictive community ecology. Journal of Vegetation Science 3, 157–164 (1992).
Tedersoo, L. & Brundrett, M. C. In Biogeography of Mycorrhizal Symbiosis (ed. Tedersoo, L.) 407–467 (Springer International Publishing, 2017).
Sprent, J. I. Nodulation in Legumes. (Royal Botanic Gardens, 2001).
Soltis, P. S., Soltis, D. E. & Chase, M. W. Angiosperm phylogeny inferred from multiple genes as a tool for comparative biology. Nature 402, 402–404 (1999).
Quesada, C. A. et al. Soils of Amazonia with particular reference to the RAINFOR sites. Biogeosciences 8, 1415–1440, https://doi.org/10.5194/bg-8-1415-2011 (2011).
Junk, W. J. et al. A classification of major naturally-occurring Amazonian lowland wetlands. Wetlands 31, 623–640 (2011).
Page, S. E., Rieley, J. O. & Banks, C. J. Global and regional importance of the tropical peatland carbon pool. Global Change Biology 17, 798–818 (2011).
Acknowledgements
This paper is the result of the work of hundreds of different scientists and research institutions in the Amazon over the past 80 years. Without their hard work this analysis would have been impossible. We thank Charles Zartman for the use of plots from Jutai. HtS and RS were supported by grant 407232/2013-3 - PVE - MEC/MCTI/CAPES/CNPq/FAPs; CB was supported by grant FAPESP 95/3058-0 - CRS 068/96 WWF Brasil - The Body Shop; DS, JFM, JE, PP and JC benefited from an “Investissement d’Avenir” grant managed by the Agence Nationale de la Recherche (CEBA: ANR-10-LABX-25-01); Floristic identification in plots in the RAINFOR forest monitoring network have been supported by the Natural Environment Research Council (grants NE/B503384/1, NE/ D01025X/1, NE/I02982X/1, NE/F005806/1, NE/D005590/1 and NE/I028122/1) and the Gordon and Betty Moore Foundation; B.M.F. is funded by FAPESP grant 2016/25086-3. The 25-ha Long-Term Ecological Research Project of Amacayacu is a collaborative project of the Instituto Amazónico de Investigaciones Científicas Sinchi and the Universidad Nacional de Colombia Sede Medellín, in parternship with the Unidad de Manejo Especial de Parques Naturales Nacionales and the Center for Tropical Forest Science of the Smithsonian Tropical Research Institute (CTFS). The Amacayacu Forest Dynamics Plot is part of the Center for Tropical Forest Science, a global network of large-scale demographic tree plots. We acknowledge the Director and staff of the Amacayacu National Park for supporting and maintaining the project in this National Park. We also thank two anonymous reviewers for their helpful, constructive comments.
Author information
Authors and Affiliations
Contributions
D.S., B.H.M., B.M. and H.t.S. initiated the study; H.t.S. and N.H. carried out the analyses, H.t.S., T.H. and N.H. wrote the manuscript, D.S., B.H.M., B.M., A.H. and J.G. gave continuous input to the writing team and comments on the analyses. All members of ATDN provided tree inventory data. All authors reviewed and added comments and additions on/to the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
ter Steege, H., Henkel, T.W., Helal, N. et al. Rarity of monodominance in hyperdiverse Amazonian forests. Sci Rep 9, 13822 (2019). https://doi.org/10.1038/s41598-019-50323-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-50323-9
This article is cited by
-
Periodic flooding and edaphic factors shape Erythrina fusca dominance in riparian forests in the Pantanal wetland
Tropical Ecology (2024)
-
Mapping density, diversity and species-richness of the Amazon tree flora
Communications Biology (2023)
-
Climate defined but not soil-restricted: the distribution of a Neotropical tree through space and time
Plant and Soil (2022)
-
Tetrathylacium vraem (Salicaceae), a new remarkable tree from the highly threatened forest in the southern Peruvian Amazon
Kew Bulletin (2022)
-
Relationships between species richness and ecosystem services in Amazonian forests strongly influenced by biogeographical strata and forest types
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.