Abstract
Monitoring of tuberculosis (TB) treatment outcome is essential to ensure an effective TB control program. In this nationwide retrospective cohort study from Denmark we present TB treatment outcome rates and risk factors associated with an unfavourable outcome. All patients notified with pulmonary TB from 2009 through 2014 were included. Logistic regression analyses were used to identify risk factors for unfavourable outcome. In total, 1681 pulmonary TB cases were included. TB treatment success rates increased during the study period. In 2014, the treatment success rate reached 85% for new culture positive cases whereas 7% cases interrupted treatment. The mortality decreased during the study period from 12.3% to 4.1%. Several risk factors associated with unfavourable outcome were identified in a multivariable model: male (OR: 2.56), Greenlandic origin (OR: 1.80), abuse of alcohol (OR: 2.90), history of mental disorder (OR: 2.46), and anaemia at time of treatment initiation (OR: 1.92). In a TB low incidence setting such as the Danish, it is important to maintain focus on preventing an unfavourable TB outcome. Patient management and treatment can be optimized by taking into consideration risk factors such as those identified in the present study.
Similar content being viewed by others
Introduction
Timely and effective tuberculosis (TB) treatment is a cornerstone in TB control in order to cure the patient, limit the transmission and prevent drug resistance1,2,3. Thus, TB treatment outcome monitoring is essential when evaluating TB control strategies. Furthermore, treatment outcome monitoring allows comparison between European countries and geographic areas. Denmark has a low incidence of TB. In 2017, 275 TB cases were reported to the Danish national TB surveillance system resulting in an incidence of 4.8/100.000 population. The incidence trend has been decreasing since 2001 (9.6/100.000 population).
In Denmark, reporting of TB treatment outcome is voluntary. As a result, the outcome data are incomplete and often reported with delay. TB treatment outcome data from 2008 through 2010 was published in 2013 and revealed a success rate for new culture positive pulmonary TB (PTB) cases of 79%4. In the most recent Surveillance Report from the European Centre for Disease Prevention and Control (ECDC), Denmark reported a treatment success rate 37.5% in 2017, however 45.7% of the cases had not been evaluated5. This suggests the true treatment success rate was higher.
For countries within the European Union and European Economic Area, the World Health Organization (WHO) and ECDC recommend a treatment success rate of 85% at 12 months for all new culture positive PTB cases and a treatment success rate of 90% for all new smear positive PTB cases6,7. This indicates that Denmark has not been able to meet WHO/ECDC targets for TB treatment success.
Identifying and targeting risk factors for an unfavourable outcome is potentially beneficial in order to improve treatment success rates. Ideally, the probability of an unfavourable outcome should be estimated from information available at the time of TB diagnosis and taken into account.
This study was conducted in order to provide updated and complete data on treatment outcome of PTB in Denmark. Furthermore, to explore predictors for an unfavourable treatment outcome in order to ensure better treatment success rates in the future.
Methods
We conducted a nationwide retrospective cohort study including all patients notified with PTB in Denmark from January 1st, 2009 to December 31st, 2014.
Data source
Notification data
TB notification has been mandatory in Denmark since 1905 and centralized since 19208. Today, TB is notifiable to the Regional Unit for Supervision and Guidance under the Danish Patient Safety Authority and to the Danish national TB surveillance system hosted by the Department of Infectious Disease Epidemiology & Prevention (DIDEP) at Statens Serum Institut (SSI). The duty of notification applies to the physician responsible for the treatment. TB cases are notified according to WHO definitions9. Standardized voluntary reporting of TB treatment outcome was initiated in 2000 in Denmark. The DIDEP provided the following regarding all TB cases notified during the study period: identification codes (Civil Registration numbers – CRN), demographics, immigrant status and date of notification. Status as immigrant was defined as patients born abroad or those born in Denmark for whom one or both parents had been born abroad.
Microbiological data
In Denmark, all culture-based analyses are performed at the International Reference Laboratory of Mycobacteriology (IRLM) at SSI. Results of microscopy, Polymerase Chain Reaction (PCR), culture, and drug susceptibility test were obtained from IRLM.
Danish national patient registry
The Danish National Patient Registry (DNPR) contains data on all admissions to Danish public hospitals since 1977 and data on outpatient contacts since 199410. Data was obtained on all patients, who were notified with TB in Denmark during the study period. Data from DNPR was used to determine history of previous illnesses, including previous mental disorder (ICD10: DF00-DF99). and to calculate Charlson Comorbidity Index (CCI)11.
Data linkage
In order to cross-link the registers, we used the unique Danish CRN, which is assigned to all residents of Denmark at birth or after residing legally in Denmark for 3 months. Patients who do not meet the criteria for obtaining a CRN are assigned a temporary CRN at first point of contact with the healthcare system. To accommodate modification in temporary CRN, probabilistic linkage was done. All CRN obtained from the notification system were linked to the DNPR data and to the data from IRLM.
Hospital records
For all patients identified by the notification system, medical records were reviewed for socio-demographics, clinical characteristics, TB treatment, and treatment outcome (Tables 1–3).
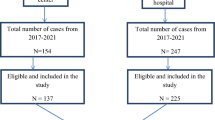
The study population is described in Fig. 1.
Patients were followed from time of first contact with the hospital due to TB until 1 year after TB treatment was completed.
Statistical analysis
All PTB cases were included in the data analysis describing patient characteristics. Categorical data was described by total and percentages, the denominator for calculated percentages was the number of cases with known information. Data comparisons were made using the chi-square test or Fisher’s exact test if 20% of expected cell values were ≤5. Continuous variables were described as medians and interquartile ranges and compared using the Wilcoxon rank sum test. A p-value of less than 0.05 (5%) was considered statistically significant.
Extensively drug-resistant (XDR), multidrug-resistant (MDR), isoniazid resistant PTB treated with second-line drugs, and patients who died or transferred out were excluded in the data analysis describing predictors for unfavourable TOs. Cases were excluded if the patient died or transferred from Denmark during the observation period to ensure that all patients in the study population had a chance to complete treatment successfully. Treatment outcome was categorized into two categories as summarized in the last two rows in Table 1.
Characteristics of cases with an unfavourable outcome were compared with cases with the outcome “treatment success”, using univariable logistic regression.
Variables for multivariable analysis were partly selected a priori (country of origin, previous TB). Other variables were selected if they showed a univariable association with defaulting (p-value < 0.05). Wald tests were used to select statistically significant variables for unsuccessful treatment outcome, and the goodness of fit of the model was tested with the Hosmer-Lemeshow method.
Data from hospital records were entered into a Microsoft Excel 2010 (Microsoft, USA) workbook. All statistical analysis was performed using Stata, version 15.2 (StataCorp Inc., USA).
Ethics
The study was approved by the Danish Data Protection Agency (Jnr. 15/34961) and the Danish Health Authority (Jnr 3-3013-1213/1).
In accordance with Danish law, observational studies performed in Denmark do not need approval from the Medical Ethics Committee or written consent from participants. All analyses are presented anonymously.
Results
Population
During the study period, a total of 2,150 TB cases were notified of these 2,131 cases were available for treatment outcome analysis of which PTB accounted for 1681 (78.9%) cases (Fig. 1).
Among the pulmonary cases; previously treated cases represented 16.4% (n = 265). A total of 892 (54.6%) cases were smear positive and 1,424 (89.1%) cases were culture positive. Drug susceptibility testing increased during the study period from 97.6% to 99.1%. Drug resistance was detected in 81 cases (6.0%), of which 1.2% (n = 1) and 9.9% (n = 8) were MDR and XDR TB cases, respectively. The remaining were mainly isoniazid monoresistant.
Treatment
In total, 1,658 PTB cases started TB treatment, the remaining 23 patients (1.4%) died before treatment was initiated. The median treatment duration was 6 months (IQR: 5.7–6.5).
A total of 819 (50.3%) of drug susceptible PTB cases received the standard treatment of 2 (3) months (the national guideline was modified in 2010: the intensive phase was decreased from 3 to 2 months) intensive phase including 4 drugs (rifampicin, isoniazid, pyrazinamide, ethambutol) and 4 months continuation phase including 2 drugs (rifampicin, isoniazid).
Socio-demographic and clinical characteristics
The population was subdivided into 3 categories according to country of origin (Table 2). The majority of immigrants originated from Asia (40.0%), with patients from Afghanistan, Pakistan and Philippines accounting for 38.1% cases. Patients from other European countries comprised 29.5%, whereas 28.9% were from Africa among whom the majority originated from Somalia (54.2% of the African cases).
Men accounted for 64.4% and the median age of the study population was 44 years (IQR: 31–54). Abuse of alcohol, tobacco, cannabis, illegal drugs, being homeless, and a history of incarceration was significantly more frequent among men. Individuals who abused alcohol more frequently had one or more of the following risk factors: use of cannabis or other illegal drugs, homelessness (75.6%). While individuals diagnosed with a mental health disorder more frequently had an abuse of illegal drugs or alcohol or were homeless (66.4% vs 49.8%, p < 0.01). A total of 779 (53.7%) had anaemia at time of diagnosis, which was significantly more frequent among women (57.6% vs 51.6%, p = 0.03). The highest proportion of anaemia was among individual from Greenland (64.0%), followed by individuals originating from Africa (59.0) while Danes (47.4%) had the lowest prevalence of anaemia. In cases with anemia there were a significantly greater proportion of smear positive (63.5% vs 45.5%, p < 0.01) and nucleic amplification acid positive (81.3% vs 72.0%, p. < 0.01) sputum samples. In addition, the proportion of sputum samples quantified as “3+ acid fast bacilli” were significantly greater among cases with anemia (42.3% vs 24.1%, p < 0.01).
A concomitant site of TB was significantly more frequent among immigrants (11.0%) with lymphatic TB (29.2%) being the most common site.
A total of 53 (4.6%) was co-infected with HIV with the highest number among immigrants (7.9%). During the study period, the number of cases tested for HIV increased significantly from 49.0% in 2009 to 79.3% in 2014.
Treatment outcome
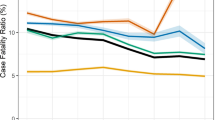
Overall, the proportion classified as treatment success was 80.5% (n = 1353), this increased during the study period from 76.8% to 84.1% with the highest treatment success rate in 2014 (Fig. 2).
The proportion of Greenlanders achieving treatment success was significantly lower (p-value < 0.01) but increased during the study period from 71.1% to 77.8%.
In further analyses the following cases are excluded: XDR-TB (n = 1), MDR-TB (n = 8) and isoniazid resistance cases treated with second-line drugs (n = 41).
Treatment outcome in new culture confirmed and new smear positive cases are presented in Fig. 2. In the univariate analysis male gender, Greenlandic origin, abuse of alcohol, tobacco, cannabis, illegal drugs, homelessness, a history of imprisonment, anaemia, previous TB and mental disorder were associated with an unfavourable outcome (Table 4).
The three most frequent symptoms: cough, fever and weight loss were combined with a symptom duration of more than 3 weeks, however there was no association with outcome.
Table 4 shows the final multivariable model fitted on 1,188 cases. A significantly increased risk of an unfavourable outcome was found among males (OR: 2.56; CI: 1.19–3.63), with cases originating from Greenland (OR: 1.80; CI: 1.06–3.07), had alcohol abuse (OR: 2.90; CI: 1.67–5.04), a history of mental disorder (OR: 2.46; CI: 1.28–4.72) or who had anaemia at time of treatment initiation (OR: 1.92; CI: 1.22–3.03).
Discussion
In this nationwide study, Denmark met the WHO/ECDC recommendations regarding treatment success rate of at least 85% for new culture confirmed PTB cases in 2013 and 2014. An unfavourable outcome was associated with male gender, Greenlandic origin, abuse of alcohol, history of mental disorder, and anaemia at time of diagnosis. The majority of unfavourable outcome was lost to follow up.
The overall treatment success rate for PTB in Denmark was 84% in 2014. In new culture-positive cases, 86.6% were classified as treatment success, of these 51.7% achieved the outcome “cured”. For the cases classified as “treatment completed” (34.9%), a second sputum sample was not obtained during the last month of treatment, as observed in another resent European study12. In the End TB Strategy, WHO recommends that the treatment success rate should be at least 90% for drug-susceptible and drug-resistant TB combined by 202513. This requires further improvement in TB surveillance, treatment and control programs in Denmark.
The present voluntary reporting of TB treatment outcome in Denmark resulted in a response rate of only 40% in 2000. In order to improve the response rate, a new procedure was implemented: DIDEP systematically contacted departments with TB patients urging them to report the treatment outcome result. This led to an improved response rate of more than 80% the subsequent years. This indicates that reporting of TB treatment outcome needs to be mandatory in order to achieve completeness of the surveillance system and avoid systematic errors, e.g. not reporting unfavourable outcomes.
Immigrants from TB high burden countries constitute a considerable proportion of TB cases in many European countries14,15,16,17,18,19. In our study, Greenlanders represented about one third of immigrants, thus we subdivided the population into Danes, Greenlanders and Immigrants. We found great disparities between the three groups in terms of age, predisposing factors and adherence to the treatment regime. The population of Greenlanders in Denmark represents 1% of the population but represented 20.3% of the pulmonary TB cases. An earlier study has identified an ongoing spreading of the outbreak strain C2/1112-15 among Greenlanders living in Denmark, this is also in accordance with 20% of the cases were identified by contact tracing20. Cases from Greenland had a higher risk of unfavourable outcome which might be attributed to the vast majority (89%) had abuse of alcohol or illegal drugs or were homeless. Immigrants from other countries did not differ significantly from Danes in terms of outcome. This is in discrepancy with previous studies and could be explained by foreign origin being related to migration and thereby to the treatment outcome: “transfer”, which has been included in unfavourable treatment outcome in other studies14,15,16,17,21. Additionally, only 26% of immigrants had the following risk factors: alcohol or illegal drug use or homelessness.
We identified male gender to be independently associated with an unfavourable outcome. The poorer treatment outcome in men can be attributed to some behavioural components such as alcohol and drug abuse, which we found to be significantly more common among men in this study in line with other EU countries14,15,16,17,18,19,20,21,22.
Increasing age and comorbidity in terms of CCI were not associated with unfavourable outcome. This may be explained as the outcome “died” was excluded in the final analysis. It is well known that mortality from TB increases with increasing age, which will contribute to an unfavourable outcome when the outcome “died” is included15,16,19,21,23. However, a history of mental disorder was associated with an unfavourable outcome. It is recognized that mental disorder is associated with poor adherence to medical treatment and earlier studies have also found that mental disorder was associated with non-adherence to TB treatment24,25.
Anaemia has been associated with delayed sputum smear conversion among sputum-positive TB patients, advanced disease and mortality26,27,28,29. Anaemia was associated with an unfavourable outcome in this study. This suggests that anaemia can be a proxy for poor health status and advanced disease, which is also supported by the fact that the bacilli load was greater in this group.
In our study we have identified additional risk factors associated with an unfavorable outcome. In addition, the vast majority of patients categorized with an unfavorable outcome were lost to follow-up (95.4%). These patients could benefit from increased support, monitoring and potentially directly observed therapy (DOT). An increased focus on optimizing the living conditions among homeless, provide support to overcome alcohol/illegal drug use and focus on involving family or other support persons in TB treatment could help to reduce the risk of being lost to follow up.
The limitations of this study are: the population was identified by notification data, hence patients who are not notified were not included. However, a recent study from Denmark has assessed the underreporting of TB to 7.5%30. The non-notified cases were all culture-negative and did not differ significantly in treatment outcome and risk factors from the notified cases30. All clinical information was from hospital records, hence no direct patient contact. In 24.2% of cases, information regarding symptom duration was missing, potentially underestimating the effect of symptom duration on treatment outcome. CCI could not be assessed for patients with temporary CRN, because they are not registered in the DNPR, these patients accounted for 3.9% (n = 65) of the entire population.
Denmark has the best prospects of obtaining high treatment success rates with state-of-the-art TB diagnostics and free tax-supported medical care. The overall trend of TB incidence has been decreasing since 2001 and the treatment success rates increased during our study period. The decline in incidence has been observed in all three subgroups, with the highest decline among Danish born men. These results are encouraging in terms of the National TB control program. However, Finland and Sweden have observed a more rapid decline in TB incidence among their native populations31. More than 6% of cases in Denmark interrupted TB treatment, this group of patients potentially pose a risk of disease progression at the individual level leading to continuous transmission in the society. The Danish public health legislation does not allow the use of mandatory treatment and hospitalization for infectious patients. Whereas there is a provision for DOT, administered at the patient’s home/shelter or at the hospital.
We conclude that the TB treatment success rate has been increasing in Denmark during the study period, reaching the WHO target for new culture confirmed PTB in the last part of the period.
To further reduce unfavourable outcome, risk factors identified by this study should be utilized and preventive approaches should be initiated in order to optimize management of patients, when TB treatment is initiated.
References
Pablos-Mendez, A., Knirsch, C. A., Barr, R. G., Lerner, B. H. & Frieden, T. R. Nonadherence in tuberculosis treatment: predictors and consequences in New York City. Am J Med 102, 164–170 (1997).
Bradford, W. Z. et al. The changing epidemiology of acquired drug-resistant tuberculosis in San Francisco, USA. Lancet 348, 928–931, https://doi.org/10.1016/S0140-6736(96)03027-9 (1996).
Cegielski, J. P. et al. Extensive Drug Resistance Acquired During Treatment of Multidrug-Resistant Tuberculosis. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America, https://doi.org/10.1093/cid/ciu572 (2014).
Statens Serum Institut. Tuberkulosebehandling 2008–2010, www.ssi.dk/aktuelt/nyhedsbreve/epi-nyt/2013/Uge27-2013. (Date last updated 2013. Date last accesed: Jan 1 2019).
WHO Regional Office for Europe/European Centre for Disease Prevention and Control. Tuberculosis surveillance and monitoring in Europe 2019–2017 data. Copenhagen: WHO Regional Office for Europe; (2019).
ECDC. Special Report: Progressing towards TB elimination. A follow-up to the Framework Action Plan to Fight Tuberculosis in the EU (2010).
WHO. Global Tuberculosis Report (2018).
Holm, J. Tuberculosis Control in Denmark. Public Health Reports 61, 1426–1443 (1946).
World Health Organization. Definitions and reporting framework for tuberculosis – 2013 revision (updated December 2014) (2014).
Schmidt, M. et al. The Danish National Patient Registry: a review of content, data quality, and research potential. Clin Epidemiol 7, 449–490, https://doi.org/10.2147/CLEP.S91125 (2015).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40, 373–383 (1987).
Martin, J. et al. Tuberculosis Treatment Outcomes in Europe: Based on Treatment Completion, Not Cure. AJRCCM 196, 3 (2017).
World Health Organization. The End TB Strategy. (Geneva, Switzerland, 2015).
Antoine, D. & Che, D. Treatment outcome monitoring of pulmonary tuberculosis cases notified in France in 2009. Euro surveillance: bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin 18 (2013).
Farah, M. G. et al. Treatment outcome of new culture positive pulmonary tuberculosis in Norway. BMC Public Health 5, 14, https://doi.org/10.1186/1471-2458-5-14 (2005).
Baussano, I., Pivetta, E., Vizzini, L., Abbona, F. & Bugiani, M. Predicting tuberculosis treatment outcome in a low-incidence area. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 12, 1441–1448 (2008).
Zellweger, J. P. & Coulon, P. Outcome of patients treated for tuberculosis in Vaud County, Switzerland. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 2, 372–377 (1998).
Diel, R. & Niemann, S. Outcome of tuberculosis treatment in Hamburg: a survey, 1997–2001. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 7, 124–131 (2003).
Borgdorff, M. W., Veen, J., Kalisvaart, N. A., Broekmans, J. F. & Nagelkerke, N. J. Defaulting from tuberculosis treatment in The Netherlands: rates, risk factors and trend in the period 1993–1997. The European respiratory journal 16, 209–213 (2000).
Lillebaek, T. et al. Mycobacterium tuberculosis outbreak strain of Danish origin spreading at worrying rates among greenland-born persons in Denmark and Greenland. Journal of clinical microbiology 51, 4040–4044, https://doi.org/10.1128/JCM.01916-13 (2013).
Karo, B. et al. Tuberculosis treatment outcome in the European Union and European Economic Area: an analysis of surveillance data from 2002–2011. Euro surveillance: bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin 20, https://doi.org/10.2807/1560-7917.ES.2015.20.49.30087 (2015).
Falzon, D., Le Strat, Y., Belghiti, F., Infuso, A. & Euro, T. B. C. Exploring the determinants of treatment success for tuberculosis cases in. Europe. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 9, 1224–1229 (2005).
Chin, D. P. et al. Progress and problems in achieving the United States national target for completion of antituberculosis treatment. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 4, 744–751 (2000).
Story, A. et al. Tuberculosis in London: the importance of homelessness, problem drug use and prison. Thorax 62, 667–671, https://doi.org/10.1136/thx.2006.065409 (2007).
Valin, N. et al. Management and treatment outcomes of tuberculous patients, eastern Paris, France, 2004. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 13, 881–887 (2009).
Isanaka, S. et al. Iron deficiency and anemia predict mortality in patients with tuberculosis. J Nutr 142, 350–357, https://doi.org/10.3945/jn.111.144287 (2012).
Kourbatova, E. V. et al. Risk factors for mortality among adult patients with newly diagnosed tuberculosis in Samara, Russia. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 10, 1224–1230 (2006).
Agrawal, Y., Goyal, V., Singh, A. & Lal, S. Role of Anaemia and Magnesium Levels at the Initiation of Tuberculosis Therapy with Sputum Conversion among Pulmonary Tuberculosis Patients. J Clin Diagn Res 11, BC01–BC04, https://doi.org/10.7860/JCDR/2017/23734.9975 (2017).
Hussain, S. F. et al. Clinical characteristics of 110 miliary tuberculosis patients from a low HIV prevalence country. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 8, 493–499 (2004).
Thrane, F. D., Andersen, P. H., Johansen, I. S. & Holden, I. K. Underreporting of patients diagnosed with tuberculosis in the Region of Southern Denmark. Under review (2019).
Pedersen, M. K. et al. Trends and differences in tuberculosis incidences and clustering among natives in Denmark, Sweden and Finland: comparison of native incidences and molecular epidemiology among three low-incidence countries. Clin Microbiol Infect 24, 717–723, https://doi.org/10.1016/j.cmi.2017.10.005 (2018).
Author information
Authors and Affiliations
Contributions
The concept of the paper was generated by I.J., I.H., T.L. and C.W. with a first draft written by I.H. and I.H., I.J., T.L., C.W., P.A., N.S. contributing to the final manuscript. Data collection and statistical analysis was undertaken by I.H.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Holden, I.K., Lillebaek, T., Seersholm, N. et al. Predictors for Pulmonary Tuberculosis Treatment Outcome in Denmark 2009–2014. Sci Rep 9, 12995 (2019). https://doi.org/10.1038/s41598-019-49439-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-49439-9
This article is cited by
-
Review of tuberculosis treatment outcome reporting system in Denmark, a retrospective study cohort study from 2009 through 2014
BMC Health Services Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.