Abstract
Glaucoma is a major cause of visual impairment, and secondary glaucoma manifested as neovascular glaucoma has long been known to occur following high-dose fractionated radiation therapy. In contrast, little is known as to whether ionizing radiation exposure causes primary glaucoma, except that a single study in Japanese atomic bomb survivors has reported a significantly increase risk. Therefore, the effect of lower dose and lower dose rate remains unclear. Here we report that in Russian Mayak Production Association workers occupationally exposed to chronic radiation for prolonged periods, incidence of total primary glaucoma and primary open-angle glaucoma is significantly associated with various non-radiation factors (sex, attained age, and cataract diagnosed prior to glaucoma), but neither with cumulative dose from external γ-rays nor with cumulative neutron dose nor with the unweighted sum of cumulative γ and neutron doses. The present results suggest for the first time that chronic radiation exposure does not cause primary glaucoma, although the analyses need to be made in other cohorts exposed at various dose and dose rate.
Similar content being viewed by others
Introduction
Glaucoma is a significant medical and social issue in many countries including Russia and Japan. Glaucoma reduces the quality of life of a patient, and represent one of the major causes of blindness worldwide1. Glaucoma covers a large group of ocular diseases sharing similar clinical, pathogenic and pathomorphological features. When the eyes suffer from glaucoma, the intraocular pressure (IOP) permanently or occasionally exceeds a level of tolerance, distinctive optic disk (OD) changes and ganglion cell degeneration (glaucomatous optic neuropathy) develop, leading to associated ocular dysfunction. Glaucoma may develop at different ages starting from birth, but its incidence increases with age1,2. Glaucoma is classified into specific types: congenital, primary glaucoma, and secondary glaucoma. Congenital glaucoma is related to fetus disorders and birth injuries. Primary glaucoma has a multifactorial nature and is associated with age-related (involutional) changes in the eyes. Primary glaucoma is classified into primary open-angle glaucoma (POAG) and primary angle-closure glaucoma (PACG), the former being further classified into normal-tension glaucoma (NTG) and high-tension glaucoma (HTG). Secondary glaucoma (e.g., neovascular glaucoma) is caused by another ocular or somatic pathology. Among risk factors for glaucoma are elderly age, heredity (excess disease incidence associated with familial group), vascular factors (increased or decreased blood pressure, atherosclerosis), and endocrine disorders (diabetes mellitus, abnormal glucocorticoid metabolism)3,4.
Local ocular or head and neck therapeutic irradiation at high doses (>about 40–50 Gy) is known to increase risk of secondary glaucoma (mostly, neovascular)5,6,7,8,9,10,11. Studies of atomic bomb survivors acutely exposed to radiation at high dose rates demonstrated increased risk only for primary open angular normal tension glaucoma only12,13. A recently published study of US radiologic technologists (USRT) showed no significant association of glaucoma with occupational radiation at low doses14. Therefore, a question of whether ionizing radiation induces glaucoma development remains open and other radiation exposed cohort studies are needed to replicate the observed results. This study aims to estimate primary glaucoma incidence risk in Mayak PA workers occupationally exposed to chronic radiation at low dose rates for prolonged periods.
Methods
Study population and follow-up
The Mayak Production Association (PA) is the first large-scale Russian nuclear enterprise. It is located in the Southern Urals close to the city of Ozyorsk and started its operation in 1948.
The study cohort included all workers of the enterprise first employed at one of the main facilities (reactors, radiochemical and plutonium production plants) in 1948–1982 (the mean calendar year of employment was 1959.4 ± 9.8 years), who were followed up until the end of 2008: there were a total of 22,377 individuals (of whom 25.4% were females). As for start of employment, the mean and standard deviations of age was 24.9 ± 7.5 years (min–max: 17–65 years). Duration of employment at the Mayak PA was 17.9 ± 14.1 years and ranged from 1 month up to 60 years. Only 4.70% of workers were employed at the Mayak PA less than 1 year.
The cohort follow-up started from the date of first employment at one of the main facilities and ended at the earliest of the following dates: disease diagnosis, date of death, 31 December 2008 for alive workers still residing in Ozyorsk (‘residents’), date of ‘the last medical information’ for workers-residents with an unknown vital status and for those who had left Ozyorsk (‘migrants’). The main characteristics of the study cohort are presented in Tables 1 and S1. By the end of the follow-up period, a vital status was available for 95.0% of the study cohort members with 53.5% deceased and 46.5% alive.
All glaucoma cases were retrospectively coded according to the International Classification of Diseases, ninth revision (ICD-9)15. All (total 634) cases of glaucoma (ICD-9: 365.0–365.9) diagnosed in members of the study cohort were identified using the medical and dosimetry database ‘Clinic’16 regardless of its type. Glaucoma was diagnosed by a qualified ophthalmologist based on generally accepted criteria (complaints, measurements of IOP, eye fundus examination, visual field tests, and gonioscopy). Of 634 cases, 476 cases (75.1%) were primary glaucoma, comprising 461 cases (96.8%) of POAG and 15 cases (3.2%) of PACG. The study outcomes included these 476 cases of primary glaucoma, because secondary glaucoma (158 cases) occurs as complications accompanying an ocular or somatic pathology.
Dosimetry
For analyses, absorbed doses from external γ-rays and neutrons were used based on the Mayak Worker Dosimetry System 2008 (MWDS–2008)17. MWDS–2008 provides absorbed doses in 18 organs and tissues (lungs, red bone marrow, breast, bladder, kidney, skin, stomach, gonads (ovaries/testicles), thyroid, uterus, esophagus, bone surface, skeleton, liver, small intestine, large intestine, spleen), but eye dose is not available. This study therefore used individual brain absorbed doses from external γ-rays and neutrons18. It should be noted that there is no considerable difference among types of organ absorbed doses from external radiation. Mean cumulative brain absorbed dose from external γ-rays was 0.46 ± 0.67 Gy in males and 0.36 ± 0.56 Gy in females. Only 4083 Mayak PA workers (18.2%) were exposed to neutrons. For these workers, the mean cumulative brain absorbed external radiation dose from neutrons was 0.0016 ± 0.0043 Gy in males and 0.0016 ± 0.0050 Gy in females. For those workers who had not been exposed to neutrons, doses from neutrons were not estimated and in the present study they were considered as ‘unmeasured 0.00’. The mean unweighted sum of cumulative γ and neutron brain absorbed doses was 0.46 ± 0.67 Gy in males and 0.37 ± 0.56 Gy in females. Distribution of the study cohort workers by radiation dose is presented in Table S1.
Statistical analysis
To perform analyses of the data on the study cohort, the statistical techniques and methods were consistent with previous studies19,20,21. The dataset for analyses was restricted to a period of residence in Ozyorsk, because information on diseases, the results of annual eye examinations and non-radiation factors was unavailable for migrants after they had left the city. This study excluded 43 workers with acute radiation sickness due to acute single high dose-rate γ + neutron exposure, and also excluded 614 workers with missing medical information due to lost medical charts and 77 workers with glaucoma diagnosed prior to employment at the Mayak PA.
As in the previous studies19,20,21, the analysis provided estimates of relative risk (RR) for categories designated for one or more variables while adjusted for other variables. RR was computed based on maximum likelihood using the AMFIT module of the EPICURE software22. In order to test a statistical significance, 95% confidence intervals (CIs) for RRs, and p values were computed using likelihood-based techniques integrated in the AMFIT module.
In consistence with the previous studies19,20,21, categorical analyses and dose-response analyses were run using Poisson regression in the AMFIT module of EPICURE software19. Excess relative risk per unit dose (ERR/Gy) was modeled by a linear trend with dose from external γ-rays, neutron dose and unweighted γ + neutron dose lagged for 5 years including adjustment (via stratification) for non-radiation factors (sex, attained age (<20, 20–25, …, 80–85, ≥85 years), birth cohort (<1910, 1910–1919, 1920–1929, 1930–1939, 1940–1949, ≥1950), hypertension (hypertension-free, hypertension, unknown), body mass index (BMI) (below normal, normal, above normal, obese, unknown), diabetes mellitus (DM) (free from DM, diagnosed with DM) and neutron dose for γ-ray exposure association analysis and vice versa.
Analysis of glaucoma incidence associated with dose from external γ radiation adjusted for neutron dose was set as the default analysis. It considered the whole cohort. The neutron dose adjustment was included using stratification, and the neutron dose was treated as a categorical variable. In consistence with the previous studies19,20,21, workers who were assumed not to have been exposed to neutrons were not excluded from the analyses; they were included in the ‘unmeasured 0.00’ category. Analyses of the linear trend with neutron dose and unweighted γ + neutron dose were conducted as sensitivity analyses. The analysis of the linear trend with neutron dose was restricted to workers with measured (or known, or estimated) doses from neutrons (4083 workers). The analysis of the linear trend with unweighted γ + neutron dose considered the whole cohort. To estimate the unweighted γ + neutron dose, the unmeasured neutron dose was regarded as equal to 0.00.
In addition, sensitivity analyses were performed to investigate effects of dose lagging (0, 10, 15, 20-year lag periods) and additional non-radiation factors, such as diagnosed cataract or cataract removal surgery prior to a report of glaucoma, alcohol consumption and smoking status, smoking index (<10, 10–20, >20 pack-years) rather than smoking status parameter on the observed risk estimates in relation to external radiation exposure.
Using log-linear modifications of excess relative risk per unit dose we tested whether such factors as sex, age at first employment and attained age of workers could modify the radiation risk of glaucoma incidence. All p-values were for two-sided test. Differences were judged to be significant at p < 0.05.
Since hypertension, BMI and DM are standard risk factor for glaucoma, all these factors were considered in the default analysis (with 5-year lag) via stratification. We excluded these variables as well as the variable of neutron dose, one by one thus investigating the effect of removing these variables from the model.
Definitions and categorizations of risk factors considered in this study are provided in details in Supplementary Section.
Information about whether a worker had a diagnosis of DM and cataract and whether he/she underwent cataract surgery was available for every worker of the study cohort. Cases with cataract and cataract removal surgery were included in the analyses if they were registered before glaucoma was diagnosed in a worker.
An additional analysis was performed to provide odds ratios of various risk factors for primary glaucoma and POAG. The analysis was based on a logit regression and is described in details in Supplementary Section.
Ethical statement
The study was reviewed and approved by the SUBI Institutional Review Board who confirmed that no signed consents were needed from members of the study cohort, because the present record-based epidemiological study did not require any contact with cohort members.
Results
By the end of the follow-up period, 476 cases of primary glaucoma were registered over 544,236 person-years of follow-up. Of these, RR of glaucoma incidence was calculated each for primary glaucoma and POAG, but not for PACG due to a small number of cases (15 cases).
Risk analysis started with assessment of its associations with known non-radiation factors promoting glaucoma (sex, attained age, smoking, alcohol consumption, cataract and cataract removal surgery, birth cohort, calendar period of diagnosis date, age at first employment, hypertension, BMI, and DM). Tables 2 and S2 summarize the results of the analysis of glaucoma incidence in relation to non-radiation factors.
RR for incidence of both primary glaucoma and POAG was significantly lower in females than in males. The incidence risks of primary glaucoma and POAG increased significantly with attained age of workers of both sexes. Incidence of primary glaucoma and POAG was not significantly associated with a calendar period of first employment except for the significantly lower incidence among females hired in 1969–1972 (RR = 0.30 (95% CIs: 0.05, 0.97) and 0.31 (95% CIs: 0.05, 0.99), respectively), which could likely be explained by a small number of cases falling into this group (2 cases). Incidence of primary glaucoma and POAG was significantly associated with certain calendar periods for workers of both sexes. RR for incidence of primary glaucoma and POAG was insignificant in groups of workers by age of first employment at the Mayak PA except for decreased risk in males hired at age 25–30 years compared to those hired before age 20 years. None of smoking status, alcohol consumption status, smoking index and BMI affected incidence of primary glaucoma and POAG in both males and females. Incidence of primary glaucoma and POAG was notably higher in workers diagnosed with cataracts (preceding glaucoma) than those without cataracts: RR in males was 12.09 (95% CIs: 9.08, 16.23) and 12.56 (95% CIs: 9.40, 16.95), respectively; RR in females was 8.43 (95% CIs: 5.54, 13.19) and 9.45 (95% CIs: 6.09, 15.16), respectively. Increased albeit insignificant risk estimates were demonstrated for incidence of primary glaucoma and POAG among workers after cataract removal surgery as well as those with diagnosed DM.
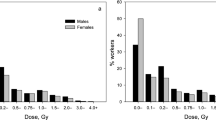
Next, RRs were analyzed for incidence of primary glaucoma and POAG in relation to cumulative dose from external γ-rays, neutrons and γ + neutron exposure. RRs for incidence of primary glaucoma and POAG resulting from all these analyses were insignificant in all dose groups (Table 3). Using a linear model, the primary glaucoma incidence risk, in particular, risk of POAG, was analyzed in relation to external γ-dose (Table 4, Fig. 1). Incidence of primary glaucoma and POAG was not significantly associated with cumulative dose from external γ-rays, regardless of adjustment for neutron dose with ERR/Gy of 0.01 (95% CIs: −0.13, 0.19) and 0.0003 (95% CIs: −0.13, 0.19) when adjusted and 0.01 (95% CIs: −0.12, 0.20) and 0.01 (95% CIs: −0.13, 0.19) when unadjusted, respectively. Moreover, no significant associations were found with cumulative neutron dose and with cumulative γ + neutron dose for either primary glaucoma or POAG (Table 4, Fig. 1). Dose lagging (0, 10, 15, 20-year lag periods), inclusion of additional adjustments for non-radiation factors (smoking index, cataract and cataract surgery, smoking status and alcohol consumption) did not significantly modify the observed findings. Exclusion of adjustments for hypertension, BMI and DM did not considerably affect the risk estimate either. Sex, attained age and age at first employment at the enterprise did not significantly modify risk for primary glaucoma (p = 0.27, p = 0.38 and p = 0.27, respectively) and for POAG (p = 0.28, p > 0.5 and p = 0.31, respectively).
Results of the additional analysis providing odds ratios of various risk factors for primary glaucoma and POAG using the logit regression are summarized in Table S3. The analysis demonstrated that its results are in good agreement with the results of the analyses based on the Poisson regression described above.
Discussion
This is a retrospective cohort study with a number of advantages, such as the large size of the study cohort, information on both sexes (with 25.4% of females), the extensive follow-up period (up to 70 years), availability of medical information and data on non-radiation risk factors over the entire follow-up period. The main advantage of the present study are the mandatory annual health examinations over the entire follow-up. The annual health examinations were performed using a standard protocol and included a mandatory check by an ophthalmologist and an ophthalmological examination using conventional techniques regardless of workers’ sex, age, working site, occupation, radiation dose, etc.
As expected, the present study found significant associations between primary glaucoma and some known non-radiation risk factors such as sex, attained age, cataract diagnosed before glaucoma manifestation, and cataract removal surgery. These results are in line with those observed in other studies3,4,23,24,25.
The present study did not reveal any significant associations between primary glaucoma incidence and doses from external γ + neutron radiation exposure and its separate components (γ-rays, neutrons) whether adjusted for each other or not. Further one-by-one exclusion of known risk factors (hypertension, BMI, DM) in the model did not modify the ERR/Gy estimate either for primary glaucoma or for POAG (Table 4). While investigating effects following fractionated exposures at dose above tens Gy (each fraction delivered at high dose rate), a number of studies have demonstrated neovascular glaucoma as one of the normal tissue complications following ocular or head and neck radiotherapy, which have been mediated by plaque formation in microcirculation and further neovascularization5,6,7,8,9,10,11. As shown in Table 5, following single, acute exposure at several Gy, glaucoma risk has been reported in Japanese atomic bomb survivors: Yamada et al. first reported that incidence of glaucoma in aggregate (i.e., without distinguishing any type of glaucoma) significantly decreases with increasing radiation dose26. In a subsequent study, and as shown in Table 5, Kiuchi et al. reported an Odds ratio at 1 Gy of 1.31 (95% CIs: 1.11, 1.53, p = 0.001) for normal-tension glaucoma, albeit with no significant associations between radiation doses and other types of glaucoma12, and the authors in their recent paper13 confirmed the significant odds ratio at 1 Gy for NTG for both right and left eyes (Table 5). However, Kiuchi et al.12 note that it was a screening study and the cohort member participation rate in that study was relatively low, underlining a need to interpret these findings with caution. Following occupational protracted exposures at low dose, there was no literature available until Little et al. very recently reported an insignificantly decreased risk for self-reported glaucoma in aggregate in the US radiologic technologists (USRT) cohort14 (and see also Table 5), which was based solely on questionnaire information obtained from medically literate workers, without any ophthalmological validation. Accordingly, it remains unclear whether members of the USRT cohort and Mayak workers exhibit a significantly increased risk for normal-tension glaucoma as reported in atomic bomb survivors. As such, our future studies include evaluation of the POAG risk separately for normal-tension type and high-tension type based on the measured IOP data.
Biological mechanisms underlying radiation-induced glaucoma remain unclear27. The most likely mechanisms that have been suggested thus far involve impaired retinal circulation since some data have demonstrated the impaired circulation at 30–45 years after radiation exposure28 as well as retinal vessel atherosclerosis29. In addition, other possible mechanisms of radiation glaucoma may include neural excitotoxicity30, inflammation31, genetic predisposition32,33,34,35, autoimmune reactions36 and retinal circulation disorders37,38. Some investigators believe that the most likely of them might be retinal circulation disorders12, since there is evidence of radiation-induced blood flow decrease in the retina 30 or 45 years after the exposure28, as well as of the significant association of retinal arteriosclerosis with radiation exposure in atomic bomb survivors 55 years after the exposure29.
In conclusion, significant association was found neither between cumulative dose from external γ-rays nor cumulative neutron dose nor the unweighted sum of cumulative γ and neutron doses and incidence of primary glaucoma and POAG in the cohort of Mayak PA workers. Glaucoma incidence was significantly associated with non-radiation risk factors (sex, attained age, and cataract diagnosed prior to glaucoma). Further analyses to distinguish normal-tension type and high-tension type are needed.
Data Availability
The dataset is the intellectual property of the Southern Urals Biophysics Institute, Ozyorsk, Chelyabinsk Region, 456780, Russia. For privacy reasons it is not publicly available. Any access to the Mayak Workers Cohort must be approved by SUBI’s Institutional Review Board. These restrictions on data availability are imposed by Federal Act No. 323 of 21 November 2011 on the basics of health care for Russian citizens and Federal Act No. 152 of 27 July 2014 on personal data. To request the data used in the presented analyses, please, contact Dr. Tamara Azizova, the head of the clinical department of the Southern Urals Biophysics Institute. Any access to the Mayak Workers Cohort must be approved by SUBI’s Institutional Review Board. Please, contact Dr. Valentina Rybkina, MD, leading researcher of SUBI, IAB member (rybkina@subi.su; +73513029953).
References
Thylefors, B. & Negrel, A. D. The global impact of glaucoma. Bull World Health Organ. 72, 323–326 (1994).
Quigley, H. A. & Broman, A. T. Number of people with glaucoma worldwide in 2010 and 2020. Br J Ohthalmol. 90, 262–267 (2006).
Le, A., Mukesh, B. N., McCarty, C. A. & Taylor, H. R. Risk factors associated with the incidence of open-angle glaucoma: the visual impairment project. Invest Ophthalmol Vis Sci. 44(9), 3783–9 (2003).
Leske, M. C., Wu, S. Y., Hennis, A., Honkanen, R. & Nemesure, B. BESs Study Group. Risk factors for incident open-angle glaucoma: the Barbados Eye Studies. Ophthalmology. 115(1), 85–93 (2008).
Shields, C. L. et al. Plaque radiotherapy for retinoblastoma: long-term tumor control and treatment complications in 208 tumors. Ophthalmology. 108(11), 2116–21 (2001).
Shields, C. L. et al. Combined plaque radiotherapy and transpupillary thermotherapy for choroidal melanoma: tumor control and treatment complications in 270 consecutive patients. Arch Ophthalmol. 120(7), 933–40 (2002).
Dieckmann, K. et al. LINAC based stereotactic radiotherapy of uveal melanoma: 4 years clinical experience. Radiother Oncol. 67(2), 199–206 (2003).
Takeda, A. et al. Late retinal complications of radiation therapy for nasal and paranasal malignancies: relationship between irradiated-dose area and severity. Int J Radiat Oncol Biol Phys. 44(3), 599–605 (1999).
Bechrakis, N. et al. 125I Brachytherapy vs. trans-scleral tumor resection. Ophthalmology. 109, 1855–1861 (2002).
Desjardins, L. et al. Current concepts in uveal melanoma. Dev. Ophthalmol. 49, 41–57 (2012).
Hayashi, K. et al. Efficacy and safety of carbon-ion radiotherapy for lacrimal gland carcinomas with extraorbital extension: a retrospective cohort study. Oncotarget. 9(16), 12932–12940 (2018).
Kuichi, Y. et al. Glaucoma in atomic bomb survivors. Radiat. Res. 180, 422–430 (2013).
Kuichi, Y. et al. Association between radiation, glaucoma subtype, and retinal vessel diameter in atomic bomb survivors. Sci. Rep. 9, 8642 (2019).
Little, M. P. et al. Occupational radiation exposure and glaucoma and macular degeneration in the US radiologic technologists. Sci Rep. 8, 10481, https://doi.org/10.1038/s41598-018-28620-6 (2018).
ICD-9 guidelines for coding diseases, injuries and causes of death/revision 1975. (Geneva, Switzerland: WHO, 1980).
Azizova, T. V. et al. The “Clinic” medical-dosimetric database of Mayak production association workers: structure, characteristics and prospects of utilization. Health Phys. 94, 449–458 (2008).
Khokhryakov, V. V. et al. Mayak Worker Dosimetry System 2008 (MWDS-2008): Assessment of internal alpha-dose from measurement results of plutonium activity in urine. Health Phys. 104, 366–378 (2013).
ICRP. 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann. ICRP 37, 2–4 (2007).
Azizova, T. V., Bragin, E. V., Hamada, N. & Bannikova, M. V. Risk of Cataract Incidence in a Cohort of Mayak PA Workers following Chronic Occupational Radiation exposure. PLoS ONE 11(10), e0164357 (2016).
Azizova, T. V., Hamada, N., Grigoryeva, E. S. & Bragin, E. V. Risk of Various Types of Cataracts in a Cohort of Mayak Workers following Chronic Occupational Exposure to Ionizing Radiation. Eur J Epidemiol 33(12), 1193–1204 (2018).
Azizova, T. V., Hamada, N., Bragib, E. V., Bannikova, M. V. & Grigoryeva, E. S. Risk of Cataract Removal Surgery in Mayak PA Workers Occupationally Exposed to Ionizing Radiation over Prolonged Periods. Radiat Environ Biophys 58(2), 139–149 (2019).
Preston, D., Lubin, J., Pierce, D. & McConney, M. Epicure Users Guide. (Hirosoft, Seattle, W. A., 1993).
Prokofyeva, E. & Zrenner, E. Epidemiology of Eye Diseases Leading to Blindness. Ophthalmic Res. 47, 171–188, https://doi.org/10.1159/000329603 (2012).
Lee, J. Y. et al. Relationship between anthropometric parameters and open angle glaucoma: The Korea National Health and Nutrition Examination Survey. Plos One. 12, e0176894, https://doi.org/10.1371/jornal.pone/0176894 (2017).
Zhao, D., Cho, J., Kim, M. H., Friedman, D. S. & Guallar, E. Diabetes, fasting glucose, and the risk of glaucoma: a meta-analysis. Ophthalmology 122, 72–78, https://doi.org/10.1016/j.ophtha.2014.07.051 (2015).
Yamada, M., Wong, F. L., Fujiwara, S., Akahoshi, M. & Suzuki, G. Noncancer disease incidence in atomic bomb survivors, 1958–1998. Radiat. Res. 161(6), 622–632 (2004).
Hamada, N. et al. Glaucomagenesis following ionizing radiation exposure. Mutat Res. 779, 36–44 (2019).
Peiretti, E., Slakter, J. S., Wu, S., Iranmanesh, R. & Yannuzzi, L. A. Late effect of external eye irradiation on choroidal circulation. Eur J Ophthalmol. 16(4), 637–40 (2006).
Minamoto, A. et al. Cataract in atomic bomb survivors. Int J Radiat Biol. 80, 339–345 (2004).
Caprioli, J. Glaucoma is a neuronal disease. Eye. 21, s6–10 (2007).
Leibovitch, I. et al. C-reactive protein levels in normal tension glaucoma. J Glaucoma. 14(5), 384–6 (2005).
Shibuya, E. et al. Association of Toll-like receptor 4 gene polymorphisms with normal tension glaucoma. Invest Ophthalmol Vis Sci. 49(10), 4453–7 (2008).
Wang, C. Y. et al. Investigation of the association between interleukin-1beta polymorphism and normal tension glaucoma. Mol Vis. 14, 719–23 (2007).
Jeoung, J. W. et al. Investigation of the association between normal-tension glaucoma and single nucleotide polymorphisms in natriuretic peptide gene. Korean J Ophthalmol 21(1), 33–8 (2007).
Mabuchi, F. et al. The OPA1 gene polymorphism is associated with normal tension and high tension glaucoma. Am J Ophthalmol. 143(1), 125–130 (2007).
Hammam, T., Montgomery, D., Morris, D. & Imrie, F. Prevalence of serum autoantibodies and paraproteins in patients with glaucoma. Eye. 22(3), 349–53 (2008).
Flammer, J. et al. The impact of ocular blood flow in glaucoma. Prog Retin Eye Res. 21(4), 359–93 (2002).
Amerasinghe, N. et al. Evidence of retinal vascular narrowing in glaucomatous eyes in an Asian population. Invest Ophthalmol Vis Sci. 49(12), 5397–402 (2008).
Author information
Authors and Affiliations
Contributions
T.V.A. and N.H. conceived the study. T.V.A. designed the study. E.V.B. was responsible for acquisition of data. T.V.A., E.S.G. produced an analytical plan. M.V.B. was responsible for data analysis. T.V.A. and N.H. interpreted the results and produce the initial draft of the manuscript. All authors provided intellectual input and approved the manuscript. T.V.A. is a guarantor.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bragin, E.V., Azizova, T.V., Bannikova, M.V. et al. Glaucoma incidence risk in a cohort of Mayak PA workers occupationally exposed to ionizing radiation. Sci Rep 9, 12469 (2019). https://doi.org/10.1038/s41598-019-48915-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48915-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.