Abstract
This was a single-center, observational, prospective study designed to compare the effectiveness of a real-time, ultrasound- with landmark-guided technique for subclavian vein cannulation. Two groups of 74 consecutive patients each underwent subclavian vein catheterization. One group included patients from intensive care unit, studied by using an ultrasound-guided technique. The other group included patients from surgery or emergency units, studied by using a landmark technique. The primary outcome for comparison between techniques was the success rate of catheterization. Secondary outcomes were the number of attempts, cannulation failure, and mechanical complications. Although there was no difference in total success rate between ultrasound-guided and landmark groups (71 vs. 68, p = 0.464), the ultrasound-guided technique was more frequently successful at first attempt (64 vs. 30, p < 0.001) and required less attempts (1 to 2 vs. 1 to 6, p < 0.001) than landmark technique. Moreover, the ultrasound-guided technique was associated with less complications (2 vs. 13, p < 0.001), interruptions of mechanical ventilation (1 vs. 57, p < 0.001), and post-procedure chest X-ray (43 vs. 62, p = 0.001). In comparison with landmark-guided technique, the use of an ultrasound-guided technique for subclavian catheterization offers advantages in terms of reduced number of attempts and complications.
Similar content being viewed by others
Introduction
The subclavian vein (SCV) is a common site of percutaneous access for central vein cannulation in intensive care. This site offers several advantages, including a lower incidence of thrombosis and central venous catheter (CVC)-related sepsis, with better patient comfort and easier nursing care1,2,3. SCV is an alternative to internal jugular vein (IJV) when this is difficult to locate, as in hypovolemic or obese patients4,5. SCV cannulation has the advantage of fixed landmarks but may be associated with potentially severe complications, e.g., pneumothorax or hemothorax, likely related to limited operator experience6.
The role of ultrasound (US) for IJV catheterization has been accepted as the standard of care after the recommendations by National Institute for Health and Clinical Excellence (NICE) in 2002, but the same guidelines stated that there was insufficient evidence to support the use of US for SCV catheterization. A Cochrane systematic review published in 2015 concluded that “two-dimensional US offers small advantages in terms of safety and quality in comparison with an anatomical landmark technique for either subclavian or femoral vein cannulation”8. On the other hand, in recent years, several trials have shown a reduction in complications and an improvement in first-pass success when US were used9,10,11,12.
In our center, after long-standing experience with US-guided IJV and femoral vein cannulation, we have extended this technique to SCV cannulation with encouraging results. The goals of present observational study were to compare, in two groups of adult patients, the effectiveness and safety of SCV cannulation with US- or landmark-guided technique.
Methods
After approval by the Local Research Ethics Committee (number 23185, May 2018), a single-center, observational, prospective study took place at Pisa University Hospital from May 1 to July 30, 2018. Clinical Trials registration number was NCT03207932. Informed consent was obtained from all conscious patients. Unconscious patients were included after assessment of the benefit-to-risk ratio with the attending physician and consent was obtained when the patient regained consciousness. If a patient had not regained consciousness or died, consent was waived in accordance with the directives of our ethics committee. The study was done in accordance with the Declaration of Helsinki13 and the applicable STROBE guidelines.
Study population and protocol
We recruited 148 critically ill adults requiring CVC positioning (Table 1). One group included 74 consecutive patients admitted to intensive care unit and studied by using a US-guided technique, another group included 74 consecutive patients admitted to emergency department and studied by using a landmark-guided technique. Indications for CVC insertion were the following: hemodynamic monitoring, treatments with vasopressors or any drug likely to induce phlebitis, temporary cardiac pacemaker and hemodialysis. Life-threatening conditions requiring an emergency CVC positioning were exclusion criteria.
All catheterizations were done by anesthesiologists with 3-to-6 years’ experience of more than 50 SCV cannulations per year, approximately half landmark and half US guided
Data collection
The following variables were recorded: site of cannulation (right or left SCV), first placement success rate, number of attempts, mechanical ventilation withdrawal, cannulation failure, reason for site change, pre- and post-procedural check (ultrasound, bubble test, chest X-ray) and type of complications.
CVC technique
The lateral subclavian vein, which is the extension of the axillary vein beyond the junction with the cephalic vein, was highlighted by the acoustic shadow of the first rib14,15,16. In the landmark group, an infra-clavicular approach was used17, with landmark points being the clavicle (the “break” or transition point, which is the junction between the medial one-third and lateral two-thirds of the clavicle) and the sternal notch. The needle was passed below the clavicle and above the first rib, with the appropriate point for cutaneous puncture being 1–2 cm below and laterally to the clavicular transition point. The needle was advanced parallel to the floor, through the subclavian muscle, until it entered the subclavian vein (Fig. 1).
Landmark infra-clavicular approach to cannulate subclavian vein. Infra-clavicular approach was used in landmark approach, with landmark points being the clavicle (the “break” or transition point, which is the junction of the medial one-third and lateral two-thirds of the clavicle) and the sternal notch. (a) The needle should pass below the clavicle and above the first rib, with appropriate point for cutaneous puncture being 1–2 cm below and laterally to the clavicular transition point. (b) The needle is advanced parallel to the floor, through the subclavian muscle, until it enter the subclavian vein.
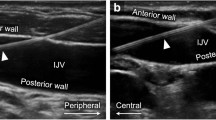
In the US-guided groups, we used a longitudinal “in-plane” approach6,18. Subclavian and axillary veins were visualized by placing a linear transducer in the infra-clavicular fossa, to obtain a short axis view of vein and artery; the transducer was then rotated until a longitudinal view was obtained and tilted until it disappeared below the clavicle in order to visualize the vessel (Fig. 2). This view enabled the visualization of the transition from medial axillary to lateral subclavian vein and the pleural line, thus enabling to visualize lung sliding and identify pre- and post-procedural pneumothorax, if any. In the longitudinal orientation, the needle was inserted at transducer midpoint providing an in-plane orientation. The needle was advanced slowly and its tip visualized throughout the procedure while maintaining a view of the vessel that was finally entered at its lateral border, just before the acoustic shadow of the clavicle and far from the cephalic vein confluence. After the needle-puncture of the vessel, the guidewire was inserted and visualized in real time (Fig. 3).
Ultrasound- guided subclavian cannulation using in-plane approach. Subclavian and axillary veins were visualized by placing a linear transducer in the infra-clavicular fossa, to obtain a short axis view of vein and artery; then, the transducer was rotated until a longitudinal view was obtained (a) and tilted in order to visualize the vessel until its disappearance below the clavicle. In the longitudinal orientation, the needle was inserted at transducer midpoint providing an in-plane orientation (b).
Probe positioning and ultrasound visualization of axillary vein, subclavian vein and pleural line before puncture (a,b). Real-time visualization of needle inserted in subclavian vein (c,d). In (a) linear probe is positioned in infra-clavicular fossa in-line with subclavian vein to obtain longitudinal view. Yellow asterisk marks transition from axillary to subclavian vein in (a) and (b). In (c,d) the needle was advanced slowly and its tip visualized throughout the procedure while maintaining a view of the vessel that was finally entered at its lateral border, just before the acoustic shadow of the clavicle, marks with yellow hash in (b), and far from the cephalic vein confluence. Pleural line is visualized throughout the procedure, it is marked with white dotted line in (b). Yellow dotted line marks the acoustic shadow of first rib (b).
Cannulation failure was defined as a change in cannulation site due to ensuing complication or unsuccessful attempts. Subsequent attempts were made at site different from subclavian vein (jugular or femoral) by using the same cannulation technique, i.e., landmark- or US-guided.
Correct catheters placements were checked by post-procedural chest X-ray or US bubble test, the choice between these being based on clinical judgment.
US bubble test was performed after CVC insertion as originally described19 and currently recommended20. Both, B-mode ultrasound and contrast enhanced US (CEUS) were performed. First, conventional B-mode ultrasound was used to examine both SV and IJV. Then, the heart was visualized through the epigastric and subcostal acoustic windows along the short heart axis, in order to see both cava veins and the right atrium at the same time confirming catheter placement. Catheter misplacement was defined as the CVC tip in the right atrium or in a vein other than superior vena cava (SVC) or SVC-to-right atrium junction. The catheter tip identification in the right atrium was confirmed by CEUS, using an air-blood-saline mixture containing 8 mL of saline, 1 mL of blood and 1 mL of air. The homogeneous solution thus obtained, was rapidly injected as a bolus. The test was deemed positive for correct CVC placement when real-time CEUS recorded a laminar jet flow of multiple microbubbles flowing from the SVC within 1 to 2 seconds after the start of injection. The test was deemed negative when the catheter tip was seen in the atrium or inferior vena cava or when the real-time CEUS recorded a turbulent flow coming from the atrium or inferior vena cava.
Statistical analysis
To calculate the sample size for SCV cannulation, we followed the recommendations of Kim and Seo21, assuming a statistical power of 0.90 (alpha = 0.05) with success rates of 100% using US 87.5% using landmark technique22. The precise sample size estimation was 74 per group and 148 in total.
Data were expressed as mean ± SD or as count and percentage (%) when appropriate. Success rate and 95% confidence interval (CI) were calculated for the cumulative cannulation attempts. Unpaired Student’s t test, chi-square or Fisher’s exact test were used to identify significance of differences between groups. A two-sided p value < 0.05 was considered significant. SPSS software (version 11.0; SPSS Inc, Chicago, IL) was used for all statistical analyses.
Ethics approval and consent to participate
Local Research Ethics Committee of Pisa University Hospital approved the present study (approval number 23185, May 2018). Written informed consent was obtained from all the participants in the study. Procedure was carried out in accordance with the Declaration of Helsinki13.
Results
Anthropometric characteristics were not different between groups (Table 1). Admissions for medical reasons were more frequent in US than landmark-guided group (66 vs. 35%, p < 0.001) and for trauma more frequent in the latter (31 vs. 3%, p < 0.001).
In both groups, there was a preference for right-site cannulation, which was chosen in 69% and 77% of US-guided and landmark-guided procedures, respectively.
Despite similar success rates between groups, i.e., 96% in US-guided and 92% in landmark-guided group (Table 2), the success at first attempt was more frequent in the former (86.5 vs 40%, p < 0.001). Furthermore, we found that in ultrasound-guided group the mean number of attempts was significantly (p < 0.001) lower than in landmark group, i.e., 1.14 (0.40) vs 2.08 (1.29).
Mechanical complications were significantly less in US- than landmark-guided group (3/74 vs 13/74, p < 0.001). Arterial punctures of subclavian artery occurred in five patients of landmark-guided but none of US-guided group (p = 0.018), whereas pneumothorax occurred with similar frequency in both groups (p = 0.591).
Concerning post-procedure controls (Table 3), more chest X-rays were necessary in landmark- than US-guided group (83 vs 58% p < 0.001). US scanning was necessary in 20% of landmark group, 10% before and 20% after cannulation. As expected, in US-guided group there were more US scans than in landmark-guided group, either before (94% vs 10%, p < 0.001) or after cannulation (55% vs 20%, p < 0.001). Correct cannulation was proved with bubble test in more patients of US- than landmark-guided group (44 vs 17%, p < 0.001). Mechanical ventilation was interrupted in 77% of patients in landmark- vs 1.3% of those of US-guided group (p < 0.001).
Discussion
Our analysis demonstrated that US guidance significantly reduced the adverse events of SCV cannulation, though the overall success rate was not significantly different from landmark technique. This result is in line with a recent systematic review showing clear advantages for US-guided versus anatomic landmark-guided techniques at cannulation site, including fewer complications and higher success rates11. Real-time US guidance for vein cannulation is supported by increasing evidence7 and has been recommended to be used routinely for CVC insertion at any site20. Nevertheless, its systematical use still faces barriers and resistance especially in particular clinical settings, like emergency departments where the US availability is still a limiting factor23.
As expected, the need of chest X-ray to confirm correct catheter placement was less in US-guided group, thus with less radiation exposure and costs, though not quantified in the present study. In our previous study, we demonstrated a reduction of chest X-ray after introduction of pulmonary US in intensive care routine, a change that reduced costs by 57% without affecting outcomes24.
The catheter placement was confirmed by US-based bubble test more frequently in the US-guided group, thus avoiding the use of chest X-ray. On the contrary, chest X-ray was the preferred confirmatory method in the landmark-guided group.
Another advantage of using US is the possibility to verify vein patency, anatomic variations, artery and pleura locations before cannulation in a systematical approach, which allows making correct choice of cannulation site and reducing of adverse events25.
Invasive mechanical ventilation in patients during the procedures was stopped in < 2% of cases in the US-guided but in 77% of those in the landmark-guided group, because the visualization of pleura made it unnecessary. There was no evidence of efficacy in the interruption of mechanical ventilation in terms of lower incidence of pneumothorax, but it has been shown that it may cause hypoxemia in critically ill patients25.
This study has several limitations. First, the study was not randomized, so it was impossible to standardize the procedures. Second, patients’ characteristics were different, with the landmark-guided procedure used more frequently in emergency department where clinical conditions, technical and human resources are more demanding and with a significant higher number of trauma patients. Third, we did not obtain the measurement of time to complete the procedures.
Conclusions
In this observational study, representative of the every-day clinical practice in a large tertiary Italian hospital, the subclavian vein cannulation with US-guided technique has been confirmed to provide better results than landmark-guided technique in critical care patients, with a lower incidence of complications and higher first-pass success rate. In the light of the growing evidence of efficacy and safety of the subclavian vein cannulation with US guidance, we hope that its use may be extended in the future even to emergency settings.
Data Availability
Please contact author for data requests
References
O’Grady, N. P. et al. Guidelines for the prevention of intravascular catheter-related infections. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 52, e162–193, https://doi.org/10.1093/cid/cir257 (2011).
Arvaniti, K. et al. Cumulative Evidence of Randomized Controlled and Observational Studies on Catheter-Related Infection Risk of Central Venous Catheter Insertion Site in ICU Patients: A Pairwise and Network Meta-Analysis. Critical care medicine 45, e437–e448, https://doi.org/10.1097/ccm.0000000000002092 (2017).
Ge, X. et al. Central venous access sites for the prevention of venous thrombosis, stenosis and infection. The Cochrane database of systematic reviews, Cd004084, https://doi.org/10.1002/14651858.CD004084.pub3 (2012).
Brusasco, C. et al. Ultrasound-guided central venous cannulation in bariatric patients. Obesity surgery 19, 1365–1370, https://doi.org/10.1007/s11695-009-9902-y (2009).
Shah, A., Smith, A. & Panchatsharam, S. Ultrasound-guided subclavian venous catheterisation - is this the way forward? A narrative review. International journal of clinical practice 67, 726–732, https://doi.org/10.1111/ijcp.12146 (2013).
Rezayat, T. et al. Ultrasound-Guided Cannulation: Time to Bring Subclavian Central Lines Back. The western journal of emergency medicine 17, 216–221, https://doi.org/10.5811/westjem.2016.1.29462 (2016).
National Institute for Health and Clinical Excellence. Technology Appraisal 49: Central Venous Catheters – Ultrasound locating devices: guidance, https://www.nice.org.uk/guidance/ta49 (accessed November 2018).
Brass, P., Hellmich, M., Kolodziej, L., Schick, G. & Smith, A. F. Ultrasound guidance versus anatomical landmarks for subclavian or femoral vein catheterization. The Cochrane database of systematic reviews 1, Cd011447, https://doi.org/10.1002/14651858.cd011447 (2015).
Sharma, A., Bodenham, A. R. & Mallick, A. Ultrasound-guided infraclavicular axillary vein cannulation for central venous access. British journal of anaesthesia 93, 188–192, https://doi.org/10.1093/bja/aeh187 (2004).
Vezzani, A. et al. A randomized clinical trial of ultrasound-guided infra-clavicular cannulation of the subclavian vein in cardiac surgical patients: short-axis versus long-axis approach. Intensive care medicine 43, 1594–1601, https://doi.org/10.1007/s00134-017-4756-6 (2017).
Fragou, M. et al. Real-time ultrasound-guided subclavian vein cannulation versus the landmark method in critical care patients: a prospective randomized study. Critical care medicine 39, 1607–1612, https://doi.org/10.1097/CCM.0b013e318218a1ae (2011).
Lalu, M. M. et al. Ultrasound-Guided Subclavian Vein Catheterization: A Systematic Review and Meta-Analysis. Critical care medicine 43, 1498–1507, https://doi.org/10.1097/ccm.0000000000000973 (2015).
World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama 310, 2191–2194, https://doi.org/10.1001/jama.2013.281053 (2013).
Lee, H. Y. et al. The axillary vein and its tributaries are not in the mirror image of the axillary artery and its branches. PLoS One 14(1), e0210464, https://doi.org/10.1371/journal.pone.0210464 (2019).
Richard, H. M. III, Selby, J. B. Jr., Gay, S. B. & Tegtmeyer, C. J. Normal venous anatomy and collateral pathways in upper extremity venous thrombosis. Radiographics: a review publication of the Radiological Society of North America, Inc 12, 527–534, https://doi.org/10.1148/radiographics.12.3.1609143 (1992).
Shiloh, A. L. et al. Ultrasound-guided subclavian and axillary vein cannulation via an infraclavicular approach: In the tradition of Robert Aubaniac. Critical care medicine 40, 2922–2923, https://doi.org/10.1097/CCM.0b013e31825cea64 (2012).
Bannon, M. P., Heller, S. F. & Rivera, M. Anatomic considerations for central venous cannulation. Risk management and healthcare policy 4, 27–39, https://doi.org/10.2147/rmhp.s10383 (2011).
Troianos, C. A. et al. Guidelines for performing ultrasound guided vascular cannulation: recommendations of the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. Journal of the American Society of Echocardiography: official publication of the American Society of Echocardiography 24, 1291–1318, https://doi.org/10.1016/j.echo.2011.09.021 (2011).
Vezzani, A. et al. Ultrasound localization of central vein catheter and detection of postprocedural pneumothorax: an alternative to chest radiography. Critical care medicine 38, 533–538, https://doi.org/10.1097/CCM.0b013e3181c0328f (2010).
Schmidt, G. A. et al. Ultrasound-guided vascular access in critical illness. Intensive care medicine, https://doi.org/10.1007/s00134-019-05564-7 (2019).
Kim, J. & Seo, B. S. How to calculate sample size and why. Clinics in orthopedic surgery 5, 235–242, https://doi.org/10.4055/cios.2013.5.3.235 (2013).
Saugel, B., Scheeren, T. W. L. & Teboul, J. L. Ultrasound-guided central venous catheter placement: a structured review and recommendations for clinical practice. Critical care (London, England) 21, 225, https://doi.org/10.1186/s13054-017-1814-y (2017).
Maizel, J. et al. Practice of ultrasound-guided central venous catheter technique by the French intensivists: a survey from the BoReal study group. Annals of intensive care 6, 76, https://doi.org/10.1186/s13613-016-0177-x (2016).
Brogi, E. et al. Could the use of bedside lung ultrasound reduce the number of chest x-rays in the intensive care unit? Cardiovascular ultrasound 15, 23, https://doi.org/10.1186/s12947-017-0113-8 (2017).
Kim, E., Kim, H. J., Hong, D. M., Park, H. P. & Bahk, J. H. Influence of Mechanical Ventilation on the Incidence of Pneumothorax During Infraclavicular Subclavian Vein Catheterization: A Prospective Randomized Noninferiority Trial. Anesthesia and analgesia 123, 636–640, https://doi.org/10.1213/ane.0000000000001431 (2016).
Author information
Authors and Affiliations
Contributions
A.S. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. E.B. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. G.B. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. S.C. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. F.G. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. P.M. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. L.T. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. M.B. concept and design, acquisition of data, analysis and interpretation of data, drafting/critical revision the manuscript, control and guarantee that all aspects of the work were investigated and resolved. G.S. study concept and design, analysis or interpretation of data, drafting/revising the manuscript, critical revision of the manuscript for important intellectual content, study supervision. F.C. study concept and design, acquisition of data, analysis or interpretation of data, drafting/revising the manuscript, control and guarantee that all aspects of the work were investigated and resolved, critical revision of the manuscript for important intellectual content, study supervision. F.F. study concept and design, acquisition of data, analysis or interpretation of data, drafting/revising the manuscript, control and guarantee that all aspects of the work were investigated and resolved, critical revision of the manuscript for important intellectual content, study supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sidoti, A., Brogi, E., Biancofiore, G. et al. Ultrasound- versus landmark-guided subclavian vein catheterization: a prospective observational study from a tertiary referral hospital. Sci Rep 9, 12248 (2019). https://doi.org/10.1038/s41598-019-48766-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48766-1
This article is cited by
-
Operator gender differences in major mechanical complications after central line insertions: a subgroup analysis of a prospective multicentre cohort study
BMC Anesthesiology (2024)
-
Effects of ipsilateral tilt position on the cross-sectional area of the subclavian vein and the clinical performance of subclavian vein catheterization: a prospective randomized trial
BMC Anesthesiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.