Abstract
It has been reported that geographical variation influences bone mineral density (BMD), obesity, and sarcopenia related traits in other countries. However, there is lack of similar studies in the US population. In this study, we compared data from three US study cohorts to evaluate geographical variations of BMD and body composition. BMD, fat mass and lean mass were collected from Dual-energy X-ray absorptiometry machine. ANCOVA and Chi-square tests were used to compare the differences between BMDs, obesity and sarcopenia related traits from different regional sites (Omaha, Kansas City and Baton Rouge/New Orleans). Eta-squared was used to measure the effect sizes of these differences. A total of 11,315 Caucasians from our previous three study cohorts were compared. There was no significant geographical difference in BMD for males or females under the criteria of p-values < 0.05 and effect size η2 > 0.01. There were significant geographical differences with medium effect size (p-value < 0.001, 0.01 < η2 < 0.14) for whole body fat mass percentage and index of low muscle mass. For Caucasians in the United States, there is no significant geographical effect found on BMD. The obesity and sarcopenia related traits are significantly different between the three study cohorts.
Similar content being viewed by others
Introduction
Osteoporosis, obesity, and sarcopenia (age-related decrease in skeletal muscle mass) are three of the most common health issues in the world. They are influenced by multiple factors, such as age, genetics, gender, and race, but few studies have focused on the potential contributions of geographical location. Geographical variation is indeed associated with a number of important factors for human health, including differences in latitude, sun exposure time, and diet. All these factors are found to be associated with vitamin D level1,2,3 and vitamin D deficiency will result in osteoporosis and sarcopenia4,5,6. Meanwhile, gut bacteria that are known associated with obesity was found to vary by geographic latitude7.
Osteoporosis is a skeletal disease characterized by a reduction of bone mineral dentistry (BMD). Dual x-ray absorptiometry (DXA) is widely used to determine BMD in osteoporosis-related studies8. Several studies in Europe9,10,11,12, Canada13 and Mexico14 have consistently found geographic variation in BMD; however, evidence is scarce in the United States. Only one study15 two decades ago showed that there was a small difference (1–4%) in femur BMD between various United States regions. As years passed and the recent rapid development of the study diseases with aging populations, an up-to-date survey is required in the United States.
Obesity is a condition of excessive body fat that causes or exacerbates many public health problems. Studies on geographical variation of obesity are based on body mass index (BMI). For example, using BMI as obesity index, DNPPAO (Division of Nutrition, Physical Activity, and Obesity)16 found that there were geographical variations in obesity prevalence in United States. BMI is calculated as total body weight divided by the square of the body height. Body weight is a heterogeneous phenotype, consisting of fat mass, muscle mass, and bone mass. There are several alternative techniques to BMI to define obesity17, such as waist circumference, skinfold thickness, whole body fat mass percentage (WBFP) and etc. Rather than BMI, WBFP would better identify metabolic risk18 and by DXA, WBFP could be accurately determined comparing to other alternative techniques17. To the best of our knowledge, no studies have evaluated geographical variations of obesity using WBFP in United States.
Sarcopenia is a condition characterized by age-related loss of skeletal muscle mass and strength19. Individuals with sarcopenia exhibited impaired balance, increased physical disability, poor quality of life, and death20. Despite there is no universally accepted operational definition of sarcopenia at this time21,22, the recent announcement of an ICD-10 code (M62.84) dedicated to sarcopenia is an important step towards recognizing sarcopenia as an independent disease entity and increasing clinical awareness of the condition23. Most of the proposed operational definitions for sarcopenia24,25,26,27,28 included appendicular lean mass (ALM), which is the sum of the lean mass of both arms and legs measured by DXA, as an approximation of muscle mass, and recommended using the ratio of ALM over height squared (ALM/ht2) as an index of low muscle mass (ALMI). A project conducted by multiple centers in Europe and US showed that there were differences in mean ALM and ALMI among the subjects from the studies performed in different regions28. However, the races of subjects in this project were various. To the best of our knowledge, there are no comprehensive studies on geographical variations in ALMI in American Caucasians.
The objective of our study was to determine whether there are geographical variations of BMD and body composition in Caucasians. We compared BMD, WBFP, and ALMI for Caucasian subjects from three different regional studies in United States.
Results
Basic characteristics
Table 1 shows the basic characteristics and lifestyle information of the study subjects, which was divided into six subgroups by geographical location and gender. There was a total of 11,315 subjects (Age: 52.1 ± 14.9 years, 7,076 females, and 4,239 males). For both genders, significant differences in age, height and weight were observed among the three study cohorts (p-value < 0.001). Meanwhile, lifestyle was significantly different among the subjects from the three geographical regions. For example, compared with LOS and OOS, the subjects from KCOS had largest percentage of subjects with regular physical exercise and smallest percentage for alcohol use, but the percentage of smokers for men and women was largest and second largest, respectively.
Geographical differences of BMD
Table 2 shows the geographical differences of BMD among LOS, KCOS, and OOS cohorts. HIP-BMD, FNK-BMD, and SPN-BMD were compared by the ANCOVA model, controlling for age, height, weight, smoking, alcohol use, and regular physical activity. FNK-BMD and SPN-BMD were not significantly different between genders among the three studies under the criteria of p < 0.05, whereas there were geographical differences for HIP-BMD (p = 0.004 for females and p = 0.003 for males). However, due to the limitations of a null hypothesis test, when a large sample size is used, even a trivial effect could be exaggerated with small p-values29,30. In the study, the effect sizes (eta-squared) were reported to determine the regional differences. When HIP-BMD values were compared among the three cohorts, the effect size was smaller than 0.01 (Table 2), which is considered to be the cutoff value of small effect size31. The results indicate no significant geographical difference in BMD for males or females under the criteria of η2 > 0.01.
Geographical differences in obesity and sarcopenia traits
For all three body composition traits (BMI, WBFP, and ALMI), we identified significant geographical differences (p-values < 0.001) among the three datasets (LOS, KCOS and OOS), even when adjusted for age, height, weight, smoking, alcohol use, and regular physical activity (Table 3). The geographical patterns for BMI were the same for both female and male subjects (KCOS < LOS < OOS). Although BMI showed significant differences among three groups (p-values < 0.001), the effect size was relatively small (η2 = 0.004 for female and η2 = 0.003 for male). WBFP showed the same ranking pattern in all three study regions (KCOS < LOS < OOS) as BMI. For sarcopenia, subjects from KCOS had the highest ALMI value and subjects from LOS had the lowest ALMI value. The effect size of geographical differences was large for WBFP and ALMI (η2 > 0.01).
Figure 1 shows the prevalence of sarcopenia and obesity in the three datasets. The sex-specific cutoff values for sarcopenia defined by ALMI were 7.26 kg/m2 (male) and 5.45 kg/m2 (female). The cutoff values for obesity defined by WBFP were 30% (male) and 40% (female). For subjects from KCOS, both males and females had the lowest rates of sarcopenia (4.6% for females and 10.94% for males) and obesity defined by WBFP threshold (16.61% for females and 7.73% for males). Subjects from OOS had the highest percentage of obesity in both male and female subjects, regardless of whether it is defined by WBFP or BMI. When obesity was defined by WBFP, significant differences can be found in each pair of datasets; however, when obesity was defined as BMI ≥ 30 kg/m2, there were no significant differences in prevalence of obesity for subjects between KCOS and LOS (female: 24.37% vs 24.51% and male: 23.13% vs 24.81%). This indicates that it is easier to identify dataset differences using WBFP than using BMI.
Prevalence of sarcopenia and obesity in subjects from LOS, KCOS, and OOS. The sex-specific cutoff values for sarcopenia are 7.26 kg/m2 (male) and 5.45 kg/m2 (female). Using BMI value, obesity was defined as BMI ≥ 30 kg/m2. Using WBFP, the cutoff points of 30% and 40% were proposed for males and females, respectively.
Discussion
The purpose of this study was to determine whether there were geographical differences in BMD and body composition of non-Hispanic whites in the United States. After adjusting for age, weight, height, smoking, alcohol usage, and regular physical activity, we found no significant differences among the studies from three geographical regions for HIP-BMD, FNK-BMD, and SPN-BMD, if we use stringent criteria p-values < 0.05 and η2 > 0.01. Significant geographical differences were found for body composition phenotypes, such as WBFP and ALMI.
To the best of our knowledge, in the past decade, this is the only study to compare the geographical differences of both BMD and body composition of non-Hispanic whites in the United States. Our data showed that there was no regional effect on BMD variation in three selected geographical regions in the United States. This is different from studies in other countries. In previous studies, European between-center differences in BMD were observed in the European Vertebral Osteoporosis (EVO)9, the Network in Europe on Male Osteoporosis (NEMO)10 and the Osteoporosis and Ultrasound study (OPUS)12. In a study published in 19979 of 16 European populations, both males and females aged between 50 and 80 were recruited randomly from local population registers. BMD values were determined by DXA from Hologic, Lunar, or Norland9. The study demonstrated that there are major differences between BMD values in European population samples from different geographical sites9. In another study conducted in 200810, normal hip and spine BMD data of 1,163 males and 329 females aged 19–29 were collected from 17 centers across Europe with strong evidence of heterogeneity among countries, but not among centers of the same country. In 200911, 10,504 hip BMD scans conducted during 1997–2002 from two population-based studies across Norway were used to validate the geographical difference. The authors found that women ≥60 years in Tromsø had higher age-adjusted BMD than women in Bergen, whereas BMD among women <60 years was similar in Tromsø and Bergen. For men, age-adjusted total hip BMD was lower in Bergen compared with Tromsø. In 201112, proximal femur BMD of females aged 20–39 (n = 258) and males aged 55–79 (n = 1,426) from three European centers were compared. A clear geographical difference in hip BMD did exist; however, there was less evidence for femoral neck BMD12. Similar research was also conducted in Canada13 and Mexico14 and found geographical variation in BMD. Older survey results15 (earlier than 2000) showed that although there were differences among regions in the United States (BMD levels of whites tended to be lower in the southern United States), the magnitude of this difference was small. They found that mean total femur BMD levels in the North, Central and West regions were only approximately 0.02–0.03 g/cm2, or 1–4%, higher than in the South. In our study, we did not find significant differences in FNK-BMD or SPN-BMD among the three groups. For HIP-BMD, although the p-value was less than 0.05, the effect size was smaller than 0.01. This indicates that the geographic differences had minimal effects on HIP-BMD variation. These controversial results can be explained by the adjustment of lifestyle factors in our study, which include smoking, exercise, and alcohol use. These factors were significantly different among the cohorts and may affect BMD variation32,33,34.
In this study, we found that geographical locations were associated with WBFP and ALMI, which is used to investigate obesity and sarcopenia, respectively. Our WBFP results were consistent with a previous meta-analysis of literature data from global studies35. It showed that European Caucasians had higher (P < 0.05) body fat percentage (28.4%) compared to the American Caucasians (23.4%)35. For sarcopenia, a large project using pooled sample (n = 26,625 participants) showed that, for both genders, there were differences in mean ALM and ALMI among the subjects from the studies performed in different regions28, including multiple regions in USA and Europe. However, this project did not conduct a detailed analysis of geographical variations in ALM or ALMI, such as dividing the subjects into various geographical areas for further comparison, and also contained a small number of non-white subjects28. In our study, only Caucasian subjects were recruited and ALM and ALMI were also found to vary by different geographic locations.
Different definitions of obesity may lead to different results. If we use BMI as a phenotype to define obesity in the study, there was no significant difference in the prevalence of obesity for subjects from LOS and KCOS (Fig. 1). If we use WBFP as criteria for obesity classification, the obesity rate of KCOS was dramatically reduced from 23.13% (by BMI) to 7.73% (by WBFP) for males and reduced from 24.37% (by BMI) to 16.61% (by WBFP) for females. For LOS, the percentage of obesity among female subjects increased from 24.51% (by BMI) to 33.83% (by WBFP) and the obesity rate of male subjects decreased slightly, from 24.81% (by BMI) to 22.24% (by WBFP). These results indicate that BMI should not be the only standard for obesity, especially when study geographical difference which involve subjects from different regions. A systematic review and meta-analysis36 found that commonly used BMI cutoff values have high specificity but low sensitivity to identify excessive body adiposity. Thus, WBFP may be a good additional measurement to identify geographical difference for traits related to obesity.
Our study has several advantages compared to previous studies. First, in this study, lifestyle factors (exercise, smoking and alcohol use) were controlled to evaluate the effect of geographical variation on BMD and body composition. Second, the three cohorts were recruited by the same research group using the same inclusion and exclusion criteria. This may reduce the selection bias. Third, we used the same standard bone osteodensitometers machine, QDR 4500, the gold standard for bone mass measurement in humans37, which may further reduce systematic errors. Fourth, in this study, effect size was considered in order to show the degree of difference. In previous studies, p-value was the only way that data were compared. A proper inference requires full reporting as suggested by SAS in 201638. P-value does not measure the size of an effect or the importance of a result.
Our study had some limitations. First, our sample size was relatively small. Only three regions were included in the study. We did not recruit subjects from western or eastern parts of the United States. Second, we could not investigate the factors that may cause the regional variation of BMD and body composition, like diet or sun exposure. Since we found WBFP variation among the three locations in different latitudes, for the next step, we would like to conduct further studies and try to define if the WBFP variation was caused by the potential mechanisms, like different sun exposure time via vitamin D metabolism, temperature variation in different latitudes or diet differences.
In summary, we found geographical differences of obesity and sarcopenia when defined by WBFP and ALMI respectively. No significant geographical effects on BMD variation were identified. The regional effects on obesity and sarcopenia were still significant after being adjusted for age, weight, height, smoking, alcohol use, and regular physical activity. This indicates that unknown region-related factors may contribute to variations in body composition. We also found that the geographical differences in obesity were easier to identify using WBFP, compared with BMI.
Materials and Methods
All the methods were conducted in accordance with the rules and guidelines of the Institutional Review Boards of University of Missouri Kansas City, Creighton University and Tulane University. The Institutional Review Boards of University of Missouri Kansas City, Creighton University and Tulane University approved the study. Written informed consent was obtained from all participants before inclusion in the study.
Study participants
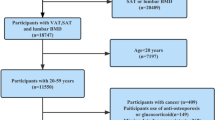
This study used 11,315 randomly recruited Caucasian subjects, from three cross-sectional study cohorts: 5,939 from the Louisiana Osteoporosis Study (LOS, New Orleans and Baton Rouge area, Coordinates: ~30oN~90–91oW)39, 3,497 from the Kansas City Osteoporosis Study (KCOS, Kansas City area, Coordinates: ~39oN~94oW)40, and 1,879 from the Omaha Osteoporosis Study (OOS, Omaha area, Coordinates: ~41oN~96oW)41. All samples were approved by the respective institutional ethics review boards and all participants signed informed-consent documents before entering the studies. Only healthy people (defined by the exclusion criteria) were included in the analyses. All of the study subjects were Caucasians with age > 18 years. The exclusion criteria are:
-
(1)
Female subjects who are or could be pregnant;
-
(2)
Female subjects who have had bilateral oophorectomy;
-
(3)
Serious residuals from cerebral vascular disease;
-
(4)
Diabetes mellitus, except for those controlled under medication;
-
(5)
Chronic renal failure;
-
(6)
Chronic liver failure;
-
(7)
Significant chronic lung disease;
-
(8)
Alcohol abuse as defined by those who cannot limit drinking, get drunk regularly, and cannot fulfill major responsibilities at work, school, or home.
-
(9)
Chronic obstructive pulmonary disease (COPD);
-
(10)
Corticosteroid therapy at pharmacologic levels for more than 6 months duration;
-
(11)
Treatment with anticonvulsant therapy for more than 6 months duration;
-
(12)
Evidence of other metabolic or inherited bone disease such as hyper- or hypoparathyroidism, Paget’s disease, osteomalacia, osteogenisis imperfecta or others;
-
(13)
Rheumatoid arthritis (except for minor cases that involve only hand joint and wrist);
-
(14)
Collagen disease (i.e., osteogenesis imperfecta and hypochondrogenesis);
-
(15)
Chronic gastrointestinal diseases including celiac disease, postgastrectomy, Crohn’s disease, ulcerative colitis, liver transplant, cirrhosis;
-
(16)
Weight over 300 pounds.
A medical history questionnaire was given to each subject to obtain self-reported information on age, sex, family history, physical activity, drinking, and smoking history.
Measurement of BMD and body composition
All BMDs at regional sites, including hip (HIP-BMD), spine (SPN-BMD), femoral neck (FNK-BMD) were measured, as were body composition including fat mass and lean mass at all regional sites, using Hologic QDR-4500 DXA scanners. DXA instruments were operated by trained technicians and calibrated daily by measuring spine phantoms. Long-term precision in our hands, the coefficient of variation (CV), was less than 1.6% for BMD measurement.
WBFP was calculated as whole body fat mass divided by whole body mass. ALM was determined by the sum of lean mass of both arms and legs (in unit of kilograms). The ALMI was calculated as ALM/height2 (in unit of kilograms/meter2). BMI was calculated as weight in kilograms divided by the square of height in meters.
Definition of sarcopenia and obesity
Sarcopenia was defined here using ALMI25. If a subject’s ALMI fell into lower than 2 SD below mean of young adults, the subject would be considered as sarcopenia in the study24,25. In this definition, sarcopenia was significantly associated with physical disability in both genders, and was independent of ethnicity, age, morbidity, obesity, income and health behaviors24. The cutoff values for definition of sarcopenia were ALMI ≤ 7.26 kg/m2 for males and ≤ 5.45 kg/m2 for females. These cutoff values were broadly accepted in a number of sarcopenia studies (e.g.24,25,42).
We defined obesity using BMI or WBFP. The WHO defines obesity as BMI ≥ 30 kg/m2. When we used WBFP to define obesity, cutoff values of 30% and 40% were proposed to define obesity for males and females, respectively. These cutoff values were adopted in a wide range of multiple studies (e.g.43,44).
Data and statistical analyses
We performed a Grubbs’ test to remove outliers. We applied a one-tailed test to detect outliers, which have values statistically different from other values (p-value < 0.05). We removed outliers and reanalyzed data as needed until no outliers were detected. ANCOVA was performed to detect geographical differences of BMD and body composition traits. In this study, we choose age, weight, height, smoking32,45,46, alcohol use47, and regular physical exercise33,48 as covariates in the ANCOVA model. The comparison of percentage of regular physical exercise, alcohol use, and smoking were performed by Chi-square test. All other basic characteristics in this study were compared using ANOVA. Eta-squared based on ANOVA was also reported to reflect the effect size of differences due to its importance to empirical studies34. Given a sufficiently large sample size, a non-null statistical comparison will always show a statistically significant result. Therefore, in this study, we used both p-value and effect size as criteria to define statistical significance. The effect sizes were defined as small (η2 = 0.01), medium (η2 = 0.06), and large (η2 = 0.14) based on benchmarks suggested by Cohen31. A test is significant if η2 > 0.01 and p-values < 0.05.
Brief summary
There is lack of studies on geographical variation influencing bone mineral density (BMD), obesity, and sarcopenia related traits in the US population. By comparing three study cohorts of various latitudes, we found no geographical effects on BMD. Obesity and sarcopenia related traits were significantly different among the three study cohorts.
References
Leary, P. F., Zamfirova, I., Au, J. & McCracken, W. H. Effect of Latitude on Vitamin D Levels. J Am Osteopath Assoc. 117(7):433–9, https://doi.org/10.7556/jaoa.2017.089. PubMed PMID: 28662556 (2017).
Wacker, M. & Holick, M. F. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol. 5(1):51–108., https://doi.org/10.4161/derm.24494, PubMed PMID: 24494042; PubMed Central PMCID: PMCPMC3897598 (2013).
Institute of Medicine FaNB. Dietary Reference Intakes for Calcium and Vitamin D. (Washington, DC: National Academy Press 2010).
Lips, P. & van Schoor, N. M. The effect of vitamin D on bone and osteoporosis. Best Pract Res Clin Endocrinol Metab. 25(4):585–91, https://doi.org/10.1016/j.beem.2011.05.002, PubMed PMID: 21872800 (2011).
Girgis, C. M., Clifton-Bligh, R. J., Hamrick, M. W., Holick, M. F. & Gunton, J. E. The roles of vitamin D in skeletal muscle: form, function, and metabolism. Endocr Rev. 34(1):33–83, https://doi.org/10.1210/er.2012-1012, PubMed PMID: 23169676 (2013).
Gunton, J. E. & Girgis, C. M. Vitamin D and muscle. Bone Rep. 8:163–7, https://doi.org/10.1016/j.bonr.2018.04.004. PubMed PMID: 29963601; PubMed Central PMCID: PMCPMC6021354 (2018).
Suzuki, T. A. & Worobey, M. Geographical variation of human gut microbial composition. Biol Lett., 10(2):20131037, https://doi.org/10.1098/rsbl.2013.1037, PubMed PMID: 24522631; PubMed Central PMCID: PMCPMC3949373 (2014).
Blake, G. M. & Fogelman, I. The role of DXA bone density scans in the diagnosis and treatment of osteoporosis. Postgrad Med J., 83(982):509–17, https://doi.org/10.1136/pgmj.2007.057505. PubMed PMID: 17675543; PubMed Central PMCID: PMCPMC2600106 (2007).
Lunt, M et al. Population-based geographic variations in DXA bone density in Europe: the EVOS Study. European Vertebral Osteoporosis. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 7(3):175-89 PubMed PMID: 9205628 (1997).
Kaptoge, S. et al. Geographical variation in DXA bone mineral density in young European men and women. Results from the Network in Europe on Male Osteoporosis (NEMO) study. Bone. 43(2):332–9, https://doi.org/10.1016/j.bone.2008.04.001, PubMed PMID: 18519175. (2008)
Omsland, T. K., Gjesdal, C. G., Emaus, N., Tell, G. S. & Meyer, H. E. Regional differences in hip bone mineral density levels in Norway: the NOREPOS study. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 20(4):631–8, https://doi.org/10.1007/s00198-008-0699-7, PubMed PMID: 18633663 (2009).
Paggiosi, M. A. et al. International variation in proximal femur bone mineral density. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 22(2):721-9, https://doi.org/10.1007/s00198-010-1336-9, PubMed PMID: 20632159 (2011).
Langsetmo, L. et al. Geographic variation of bone mineral density and selected risk factors for prediction of incident fracture among Canadians 50 and older. Bone. 43(4), 672–8, https://doi.org/10.1016/j.bone.2008.06.009, PubMed PMID: 18640295; PubMed Central PMCID: PMCPMC5101048 (2008).
Deleze, M. et al. Geographic differences in bone mineral density of Mexican women. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 11(7):562–9, https://doi.org/10.1007/s001980070076, PubMed PMID: 11069189 (2000).
Looker, A. C. et al. Updated data on proximal femur bone mineral levels of US adults. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 8(5):468–89 PubMed PMID: 9850356 (1998).
Services USDoHH. Division of Nutrition, Physical Activity, and Obesity 2015. Available from, https://www.cdc.gov/obesity/data/prevalence-maps.html [updated September 23, 2015].
Hu, F. Measurements of Adiposity and Body Composition. Obesity Epidemiology. New York City: Oxford University Press. p. 53–83 (2008).
De Lorenzo, A. et al. Adiposity rather than BMI determines metabolic risk. Int J Cardiol. 166(1):111–7, https://doi.org/10.1016/j.ijcard.2011.10.006, PubMed PMID: 22088224 (2013).
Dodds, R. M., Roberts, H. C., Cooper, C & Sayer, A. A. The Epidemiology of Sarcopenia. J Clin Densitom. 18(4):461–6, https://doi.org/10.1016/j.jocd.2015.04.012, PubMed PMID: 26073423; PubMed Central PMCID: PMCPMC4629409 (2015).
Santilli, V., Bernetti, A., Mangone, M. & Paoloni, M. Clinical definition of sarcopenia. Clinical cases in mineral and bone metabolism: the official journal of the Italian Society of Osteoporosis, Mineral Metabolism, and Skeletal Diseases 11(3), 177–80. PubMed PMID: 25568649; PubMed Central PMCID: PMCPMC4269139 (2014).
Dam, T. T. et al. An evidence-based comparison of operational criteria for the presence of sarcopenia. J Gerontol A Biol Sci Med Sci. 69(5), 584–90, https://doi.org/10.1093/gerona/glu013, PubMed PMID: 24737561; PubMed Central PMCID: PMCPMC3991139 (2014).
McLean, R. R. & Kiel, D. P. Developing consensus criteria for sarcopenia: an update. J Bone Miner Res., 0(4), 588–92, https://doi.org/10.1002/jbmr.2492, PubMed PMID: 25735999 (2015).
Cao, L. & Morley, J. E. Sarcopenia Is Recognized as an Independent Condition by an International Classification of Disease, Tenth Revision, Clinical Modification (ICD-10-CM) Code. J Am Med Dir Assoc., 17(8), 675–7, https://doi.org/10.1016/j.jamda.2016.06.001, PubMed PMID: 27470918 (2016).
Baumgartner, R. N. et al. Epidemiology of sarcopenia among the elderly in New Mexico. American journal of epidemiology., 147(8), 755–63. Epub 1998/04/29. PubMed PMID: 9554417 (1998).
Fielding, R. A. et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc., 12(4), 249–56, https://doi.org/10.1016/j.jamda.2011.01.003 PubMed PMID: 21527165; PubMed Central PMCID: PMCPMC3377163 (2011).
Newman, A. B. et al. Sarcopenia: alternative definitions and associations with lower extremity function. Journal of the American Geriatrics Society., 51(11), 1602–9. PubMed PMID: 14687390 (2003).
Kim, K. M., Jang, H. C. & Lim, S. Differences among skeletal muscle mass indices derived from height-, weight-, and body mass index-adjusted models in assessing sarcopenia. Korean J Intern Med., 31(4), 643–50, https://doi.org/10.3904/kjim.2016.015, PubMed PMID: 27334763; PubMed Central PMCID: PMCPMC4939509 (2016).
Studenski, S. A. et al. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci., 69(5), 547–58, https://doi.org/10.1093/gerona/glu010. PubMed PMID: 24737557; PubMed Central PMCID: PMCPMC3991146 (2014).
Sullivan, G. M. & Feinn, R. Using Effect Size-or Why the P Value Is Not Enough. Journal of graduate medical education., 4(3), 279–82, https://doi.org/10.4300/JGME-D-12-00156.1 PubMed PMID: 23997866; PubMed Central PMCID: PMCPMC3444174 (2012).
Levine, T. R. & Hullett, C. R. Eta Squared, Partial Eta Squared, and Misreporting of Effect Size in Communication Research. Human Communication Research., 28(4), 612–25, https://doi.org/10.1111/j.1468-2958.2002.tb00828.x (2002).
J. C. Statistical Power Analysis for the Behavioral Sciences (New York, NY: Routledge Academic 1988).
Ward, K. D. & Klesges, R. C. A meta-analysis of the effects of cigarette smoking on bone mineral density. Calcified tissue international 68(5), 259–70 (2001). PubMed PMID: 11683532.
Paccou, J. et al. Relationships between bone geometry, volumetric bone mineral density and bone microarchitecture of the distal radius and tibia with alcohol consumption. Bone. 78, 122–9, https://doi.org/10.1016/j.bone.2015.05.002, PubMed PMID: 25959415 (2015).
Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Frontiers in psychology. 4, 863, https://doi.org/10.3389/fpsyg.2013.00863, PubMed PMID: 24324449; PubMed Central PMCID: PMCPMC3840331 (2013).
Deurenberg, P., Yap, M. & van Staveren, W. A. Body mass index and percent body fat: a meta analysis among different ethnic groups. Int J Obes Relat Metab Disord. 22(12), 1164–71. PubMed PMID: 9877251 (1998).
Okorodudu, D. O. et al. Diagnostic performance of body mass index to identify obesity as defined by body adiposity: a systematic review and meta-analysis. Int J Obes (Lond). 34(5), 791–9, https://doi.org/10.1038/ijo.2010.5, PubMed PMID: 20125098 (2010).
Mas, I. D., Biscardi, A. & Ripamonti, U. Comparison of two Hologic DXA systems (QDR 1000 and QDR 4500/A) for in vivo bone mass measurement in the baboon (Papio ursinus). J Clin Densitom. 5(3):313–8. PubMed PMID: 12357069 (2002).
Wasserstein, R. L. & Lazar, N. A. The ASA’s Statement onp-Values: Context, Process, and Purpose. The American Statistician. 70(2), 129–33 Epub Feb. 2016, https://doi.org/10.1080/00031305.2016.1154108 (2016).
He, H. et al. Relationship of sarcopenia and body composition with osteoporosis. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 27(2), 473–82. Epub 2015/08/06, https://doi.org/10.1007/s00198-015-3241-8 PubMed PMID: 26243357 (2016).
Yang, T. L. et al. Genetic variants in the SOX6 gene are associated with bone mineral density in both Caucasian and Chinese populations. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 23(2), 781–7, https://doi.org/10.1007/s00198-011-1626-x PubMed PMID: 21625884; PubMed Central PMCID: PMCPMC4171834 (2012).
Guo, Y. et al. Genome-wide association study identifies ALDH7A1 as a novel susceptibility gene for osteoporosis. PLoS genetics. 6(1), e1000806, https://doi.org/10.1371/journal.pgen.1000806 PubMed PMID: 20072603; PubMed Central PMCID: PMC2794362 (2010).
Iannuzzi-Sucich, M., Prestwood, K. M. & Kenny, A. M. Prevalence of sarcopenia and predictors of skeletal muscle mass in healthy, older men and women. J Gerontol A Biol Sci Med Sci. 57(12), M772–7, PubMed PMID: 12456735 (2002).
Waters, D. L., Hale, L., Grant, A. M., Herbison, P. & Goulding, A. Osteoporosis and gait and balance disturbances in older sarcopenic obese New Zealanders. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 21(2), 351–7, https://doi.org/10.1007/s00198-009-0947-5 PubMed PMID: 19436938 (2010).
Ilich, J. Z. et al. Interrelationship among muscle, fat, and bone: connecting the dots on cellular, hormonal, and whole body levels. Ageing Res Rev. 15, 51–60, https://doi.org/10.1016/j.arr.2014.02.007 PubMed PMID: 24632496 (2014).
Yoon, V., Maalouf, N. M. & Sakhaee, K. The effects of smoking on bone metabolism. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 23(8), 2081–92, https://doi.org/10.1007/s00198-012-1940-y PubMed PMID: 22349964 (2012).
Kanis, J. A. et al. Smoking and fracture risk: a meta-analysis. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 16(2), 155–62, https://doi.org/10.1007/s00198-004-1640-3, PubMed PMID: 15175845 (2005).
Sioka, C., Bougias, C., Al-Bokharhli, J. & Fotopoulos, A. Smoking and alcohol use as risk factors for low bone mineral density. Rheumatology international. 27(2), 207–8, https://doi.org/10.1007/s00296-006-0189-1 PubMed PMID: 16932957 (2006).
Sritara, C. et al. Work- and travel-related physical activity and alcohol consumption: relationship with bone mineral density and calcaneal quantitative ultrasonometry. J Clin Densitom. 18(1), 37–43, https://doi.org/10.1016/j.jocd.2014.04.117. PubMed PMID: 24878331 (2015).
Acknowledgements
We thank all study participants for volunteering to provide phenotype information. We would like to thank all staff who provided the clinical expertise, collected, and managed the data. The investigators of this work were partially supported by grants from the NIH (AR069055, U19 AG055373, R01 MH104680, R01AR059781 and P20GM109036), and the Edward G. Schlieder Endowment as well as the Drs. W.C. Tsai and P.T. Kung Professorship in Biostatistics from Tulane University.
Author information
Authors and Affiliations
Contributions
Y.Z., H.W.D. and L.J.Z. conceived and designed research; Y.Z., K.H.W. and J.G.Z. performed research and analyzed data; Y.Z., K.H.W., H.S., H.W.D. and L.J.Z. wrote/revised the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhou, Y., Wu, K., Shen, H. et al. Geographical differences in osteoporosis, obesity, and sarcopenia related traits in white American cohorts. Sci Rep 9, 12311 (2019). https://doi.org/10.1038/s41598-019-48734-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48734-9
This article is cited by
-
Geographic variation in bone mineral density and prevalent fractures in the Canadian longitudinal study on aging
Osteoporosis International (2024)
-
A spatial obesity risk score for describing the obesogenic environment using kernel density estimation: development and parameter variation
BMC Medical Research Methodology (2023)
-
Associations of physical activity with sarcopenia and sarcopenic obesity in middle-aged and older adults: the Louisiana osteoporosis study
BMC Public Health (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.