Abstract
Recently, Goji berry (Lycium barbarum L.) has been extensively cultivated to improve the fragile ecological environment and increase the income of residents in Qinghai Province, northwestern China. However, few studies have focused on the physiological responses of Goji berry under salt stress and alkali stress. Gas exchange, photosynthetic pigments, and chlorophyll fluorescence were evaluated in response to neutral (NaCl) and alkali (NaHCO3) salt stresses. Nine irrigation treatments were applied over 30 days and included 0(Control group), 50, 100, 200, and 300 mM NaCl and NaHCO3. The results showed that salt and alkali stress reduced all the indicators and that alkali stress was more harmful to Goji berry than salt stress under the same solution concentrations. The salt tolerance and alkali resistance thresholds were identified when the index value exceeded the 50% standard of the control group, and threshold values of 246.3 ± 2.9 mM and 108.4.7 ± 2.1 mM, respectively, were determined by regression analysis. These results were used to identify the optimal water content for Goji berry. The minimum soil water content to cultivate Goji berry should be 16.22% and 23.37% under mild and moderate salt stress soils, respectively, and 29.10% and 42.68% under mild and moderate alkali stress soil, respectively.
Similar content being viewed by others
Introduction
Salt-alkalization is becoming an increasing environmental and socioeconomic problem because it leads to the loss of agricultural land at a rate of 0.25 to 0.5 Mha annually around the world1. Alkaline soils are primarily caused by the accumulation of NaHCO3, whereas saline soils are mainly due to NaCl accumulation2,3. In China, especially northwestern China, overgrazing, evaporation, overirrigation, and deforestation are crucial factors that result in severe secondary salinization, which can significantly reduce crop productivity4,5,6. Conventional engineering salt washing has many deficiencies, such as high costs, water waste, and side effects. Therefore, phytoremediation with native plants which has a high salt and alkali tolerance is a practical solution from an economic and environmental perspective7,8. Generally, salt stress in the form of NaCl is associated with imbalances in ion levels and homeostasis7,9, leading to oxidative stress and ion overload10,11,12. In contrast, alkali stress is mainly caused by excess NaHCO3 in the soil solution that results in a high pH. Recent studies have shown that salt stress can cause damage to physiological processes, including the photosynthetic apparatus, chlorophyll fluorescence, osmotic adjusting materials and antioxidant defence mechanisms13. In recent years, studies have primarily focused on plant responses to salt stress, while little attention has been paid to the effect of alkalinity, especially the severe effects of alkalinity compared to salinity14. Hence, understanding the physiological response of plants to alkalinity is of great significance for selecting adaptive crop plants with increased resistance to alkalinity.
Photosynthesis is of paramount importance and represents a physiological process that can be inhibited by salt or alkali stress15,16. Researchers have indicated that stomatal closure is the main factor that decreases the assimilation ability of CO2 under salt stress and results in a decline in photosynthetic capacity17. Salt stress also has an influence on photosynthetic components and chloroplast ultrastructure18,19, which are believed to be nonstomatal factors that affect the decline in photosynthetic capacity20,21. Chlorophyll (Chl) fluorescence represents a sensitive and noninvasive probe and has been used to study the function and performance of the photosynthetic machinery of various plants, and several recent works have shown that this tool can be useful for identifying stress factors that affect or/and limit plant growth22. However, the effects of alkali stress on photosynthesis, especially on Chl fluorescence, are poorly understood. Malondialdehyde (MDA), superoxide dismutase (SOD), peroxidase (POD) and catalase (CAT) play paramount roles in osmotic adjustment and may also be of great importance in eliminating reactive oxygen species (ROS) when plants are under salt stress23. However, the relationship between antioxidant enzymes and alkali stress has not been clarified.
Qinghai Province in China is located in an extremely cold region—the transition zone between the Tibetan Plateau and the Loess Plateau. Qinghai suffers from serious soil salt-alkalization and has a salt-alkali area of 15,720 km2, which accounts for 2.3% of the total area of China according to the work of Cai24. The most severe salinization in Qinhai Province is centralized in Qaidam basin. The main ion types of this area are Na+, Cl−, and HCO3−, and the ion density of each ion is approximately 2.71, 7.67 and 0.28 g kg−1, respectively. Saline or alkali soils seriously affect the vegetation cover and ecological environment, as well as affecting the land use status, thus influencing the economic development of the entire Qinghai Province. Based on the geo-ecological uniformity, an effective method for ameliorating the fragile ecological environment in Qinghai Province is to improve soil salt-alkalization by cultivating abundant native plants that are highly capable of acclimating to and improving the adverse soil conditions25.
Goji berry (Lycium barbarum L.), which is also named Wolfberry, is a perennial plant in arid areas that is used in traditional herbal medicine. The Goji berry is usually consumed for dietetic and medicinal purposes, as its leaves are rich in flavonoids, polysaccharides, and amino acids26. Recently, interest in Goji berry has greatly increased, and the role of Goji berry in local economies has become increasingly important in recent years. Goji berry has also been proposed as a potential pioneer plant to reclaim salinized and alkalinized soils because Goji berry growth and photosynthesis have been shown to be negatively affected by high levels of salt stress27. However, previous studies on the effects of salt stress on Goji berry have primarily focused on physiological changes under salt stress, and the response to alkali stress has not been sufficiently studied.
Considering the abovementioned issues, the specific objectives of this study were to investigate the variations in leaf gas exchange, Chl content, Chl fluorescence and antioxidant enzymes in Goji berry under salt and alkali stress. In addition, we performed a regression analysis to determine the salt tolerance and alkali tolerance threshold of Goji berry, which was used to establish the correlation between the saline-alkali content and soil moisture content. Our results regarding the parameters mentioned above will advance our understanding of the mechanisms of salt and alkali tolerance of Goji berry and provide a scientific basis for the field production and water management of Goji berry under different saline-alkali conditions in Qinghai Province.
Results
Effects of salt stress and alkali stress on gas exchange
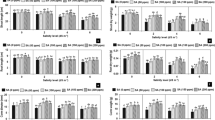
As shown in Fig. 1, 50 mM NaCl did not have a significant effect on the net photosynthetic rate (PN), transpiration rate (E), stomatal conductance (gs) or intercellular CO2 concentration (Ci), whereas 50 mM NaHCO3 had a significant effect on the values of these parameters (P < 0.05, Fig. 1). With increasing concentrations of salt or alkali (greater than 50 mM), the PN, E and gs values apparently decreased, and the extent of the reduction under alkali stress was greater than that under salt stress (P < 0.05, Fig. 1). However, the Ci value exhibited a constant decreasing trend at salinity levels ranging from 0~200 mM, and the value increased at a salinity level of 300 mM (P < 0.05, Fig. 1D). The trend exhibited by Ci under alkali stress was similar to that exhibited under salt stress, although the value started to increase at 200 mM (P < 0.05, Fig. 1D). The water use efficiency (WUE) of Goji berry under both salt stress and alkali stress showed an increasing trend with an increasing concentration of salt or alkali (P < 0.05, Fig. 1E). Interestingly, the WUE of Goji berry under 300 mM alkali stress was extremely lower than other alkali treatments.
Effects of NaCl and NaHCO3 concentrations on the (A) net photosynthetic rate (PN), (B) transpiration rate (E), (C) stomatal conductance (gs), (D) intercellular CO2 concentration (Ci) and (E) water use efficiency (WUE) in the leaves of Goji berry. Values are represented as the mean ± SE (n = 5). Different letters indicate a significant difference at P < 0.05 by the least significant difference (LSD) test.
Effects of salt stress and alkali stress on the chlorophyll pigment content
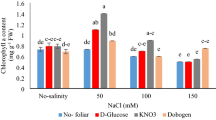
Differences were not observed in the Chl pigment content of Goji berry between the low-concentration (50 mM) salt treatment and the control. However, salt stress markedly decreased the Chl a, Chl b, carotenoids (Cars) and Chl (a + b) values in the leaves at salinity levels higher than 100 mM (P < 0.05, Fig. 2). Plants treated with NaHCO3 exhibited a constant decrease in Chl pigment content, including the Chl a, b, Cars and (a + b) values (P < 0.05, Fig. 2), and the rate of decrease was greater than that observed under salt stress treatment at the corresponding concentrations. Maximum reductions in Chl a, b, Cars and (a + b) values (67.74, 65.65, 73.35 and 66.70%, respectively) were observed in the 300 mM salt stress treatment, whereas these values were 89.18, 94.22, 87.72 and 90.99% in the 300 mM alkali stress treatment. In general, the Chl a and b values were more sensitive (or less resistant) to alkali stress than salt stress at the same concentrations, which caused a distinct decrease in the Chl (a + b) value (P < 0.05, Fig. 2).
Effects of salt stress (SS) and alkali stress (AS) on the contents of (A) chlorophyll a, (B) chlorophyll b, (C) carotenoids and (D) chlorophyll (a + b). Means values indicated by different letters in the same curve are significantly different at P < 0.05 according to the least significant difference (LSD) test.
Effects of salt stress and alkali stress on the chlorophyll fluorescence
As shown in Fig. 3, salt stress decreased the maximum quantum yield of PSII (Fv/Fm) value by 3.16, 11.58 and 30.76% compared to the value of the control group in Goji berry subjected to 100, 200 and 300 mM, respectively (P < 0.05). On the other hand, salt stress decreased the Fv/Fo values by 5.82, 15.96, 24.91 and 34.48% compared to the value of the control group when the seedlings were subjected to 50, 100, 200 and 300 mM salinity, respectively (P < 0.05, Fig. 3A). Furthermore, a magnitude of Fv/Fm greater than 0.78 was observed in only the group treated with the 50 mM salt solution. Under alkali stress, the Fv/Fm value was found to decrease by 44.35 and 55.15%, (P < 0.05, Fig. 3B) when treated with 200 and 300 mM, while the Fv/Fo value was found to decrease by 20.79 and 38.35% in Goji berry treated with 200 and 300 mM alkali solution, respectively (P < 0.05, Fig. 3B).
Effects of (A) NaCl and (B) NaHCO3 on the Fv/Fm and Fv/Fo values of Goji berry after 20 days of treatment with different concentrations. Values are represented as the mean ± SE (n = 5). Abbreviations: Fv/Fm: maximum quantum yield of electron transfer of PSII, Fv/Fo: activity of the water-splitting complex.
In terms of the parameters associated with Chl fluorescence, salt stress increased the nonvariable Chl fluorescemce level (Fo), relative variable fluorescence at the J step (Vj), reductions of QA (Sm), absorption energy flux (ABS/RC), energy dissipation in the unit reaction center (DIo/CSo) and energy capture in the unit reaction center (TRo/CSo) values by 29.7, 35.53, 17.72, 12.28, 80.09 and 19.89%, respectively (P < 0.05, Fig. 4A). However, salt stress decreased the variable fluorescence (Fv), ETo/RC, TRo/CSm, ETo/CSm and performance index of intersystem electron acceptors (PI) values by 12.77, 22.20, 12.78, 32.74 and 56.20%, respectively (P < 0.05, Fig. 4A). In addition, salt stress had no significant effect on the time to reach maximum fluorescence intensity (tFm), maximum fluorescence induction (Fm), TRo/RC, ETo/CSo and ABS/CSm values (P > 0.05, Fig. 4A).
When Goji berry was subjected to 300 mM alkali solution, the alkali stress increased the Fm, Fv, Vj, ABS/CSm, TRo/CSm, and ETo/CSm values by 38.88, 74.94, 50.77, 37.75, 49.16 and 22.15%, respectively (P < 0.05, Fig. 4B). However, alkali stress decreased the tFm, Fo, Sm, ABS/RC, ETo/RC, and DIo/CSo values by 43.70, 16.93, 47.19, 22.60, 24.02 and 62.69%, respectively (P < 0.05, Fig. 4B). In addition, no significant difference was observed in the TRo/RC, TRo/CSo, ETo/CSo and PI values (P > 0.05, Fig. 4B).
Effects of salt stress and alkali stress on antioxidant enzyme activities
Significant differences were not observed in the MDA content at a 50 mM salinity level (P < 0.05, Fig. 5A); however, the MDA content increased significantly with increasing salinity. The subsequent assays suggested that the MDA content in the leaves was higher under alkali stress than under salt stress. In addition, seedlings under alkali stress exhibited the same increasing tendency as those under salt stress (P < 0.05, Fig. 5A). Under both types of stress, the MDA levels in the seedlings plateaued in the 300 mM treatment (Fig. 5A).
Effects of NaCl and NaHCO3 concentrations on (A) malondialdehyde (MDA) levels, (B) catalase (CAT) activity, (C) superoxide dismutase (SOD) activity, and (D) peroxidase (POD) activity in the leaves of Goji berry. Values are represented as the mean ± SE (n = 5). Different letters indicate a significant difference at P < 0.05 by the least significant difference (LSD) test.
The results of the CAT activity assay showed that CAT increased in a linear manner under salt stress at salinity levels ranging from 50 to 100 mM (P < 0.05, Fig. 5B). Under alkali stress, the CAT activity increased until the alkali concentration reached 200 mM, and then, the CAT activity decreased (P < 0.05, Fig. 5B). Compared with the control, the CAT activity under alkali stress was higher than that under salt stress (P < 0.05, Fig. 5B).
The protective enzyme activity determinations revealed that the SOD and POD activities in the leaves were similar under both types of stress. In general, the variation trend of POD activity was similar to that of SOD activity in response to salt or alkali stress. Compared with the control, salt stress significantly increased the SOD and POD activities, whereas alkali stress increased the SOD and POD activities at alkali concentrations ranging from 50 to 200 mM and then decreased these activities at the concentration of 300 mM (P < 0.05, Fig. 5C,D).
Salt tolerance and alkali tolerance evaluation of Goji berry
Table 1 shows the correlations between the concentration, photosynthetic traits, and antioxidant attributes. PN is significantly and positively correlated with gS, whereas PN is significantly and negatively correlated with Ci. As Table 1 shows, PN and SOD have the highest correlation coefficient with concentration. Therefore, PN and SOD were selected to characterize the salt and alkali tolerance of Goji berry. PN and SOD were separately fitted to the saline-alkali concentration, and the salt concentration of each index exceeding 50% of the control group was used as the salt tolerance and alkali threshold. The calculated thresholds of salt stress on PN and SOD were 246.3 ± 2.9 and 254.3 ± 2.1 mM, respectively (P < 0.05, Table 2), and the lower value of 246.3 ± 2.1 mM was used as the salt tolerance threshold. On the other hand, the thresholds of alkali stress on PN and SOD were 183.4 ± 3.4 and 108.4 ± 2.1 mM, respectively (P < 0.05, Table 2), and the lower value of 108.4 ± 2.1 mM was used as the alkali tolerance threshold.
Linear regression of soli salt content and soil water content
Based on the analysis of PN and SOD activity of Goji berry under salt and alkali stress, the salt and alkali tolerance threshold range of Goji berry was determined to be 246.3 ± 2.9 mM and 108.4 ± 2.1 mM, respectively (P < 0.05, Table 2). The relationship between the salt content and water content of the local soil is shown in Fig. 6, and it is calculated based on the threshold of salinity (NaCl) and alkalinity (NaHCO3) of Goji berry. According to the Chinese criteria for the classification of soil salt-alkalization, a salt content of 2–4% indicates mild stress, 4–6% indicates moderate stress, and over 6% indicates severe stress28. As shown in Fig. 6, the minimum soil water content to cultivate Goji berry should be 16.22% and 23.37% in mild and moderate stress soils, respectively, and 29.10% and 42.68% in mild and moderate alkali stress soils, respectively.
Discussion
The photosynthetic process is one of the major factors that controls plant growth under adverse conditions29. In the present experiment, PN, gS, and E decreased significantly in Goji berry under salt stress and alkali stress, which is consistent with previous reports of stress-induced limitations of photosynthetic capacity26,30,31. PN was positively correlated with gS under all stress levels (Table 1.), which could explain why the reduction in gS caused a simultaneous decrease in both PN and E. Such an inhibitory effect on photosynthesis has also been reported in another plant32. The reduction in PN under salt and alkali stress is generally considered to be stomatal restriction caused by partial closure of stomata and decreased cell viability33,34,35. Moreover, the decrease in Ci and gS were also the main reasons for the reduction of PN36. Yan et al.37 hypothesized that stomatal factors that inhibit photosynthetic activity are observed under conditions of intermediate salinity and alkalinity while nonstomatal factors function at high salinity and alkalinity. Moreover, the decrease in PN, gS, and E was even greater under alkali stress than under salt stress (Fig. 1A–C). Although salt stress and alkali stress significantly reduce the photosynthesis and transpiration rates of Goji berry, the water use efficiency in this species was enhanced, which is in accordance with the findings in Kosteletzfkya virginica L. Presl and Quercus aliena by Yue et al. and Farissi et al.38,39. It’s interesting that the water use efficiency of Goji berry under 300 mM alkali treatment was extremely lower than the other alkali treatment, which means that the alkali-tolerant threshold of Goji berry may be under 300 mM. This phenomenon showed that high pH induced by alkaline conditions might stimulate Goji berry roots to generate physical or chemical signals to affect stomatal opening and closing and gas exchange. Thus, the values of these photosynthetic parameters likely indicate the adaptability of Goji berry, and the values are far lower under alkali stress than under salt stress, which suggests that Goji berry seedlings are less adaptable to alkali stress than salt stress.
The Chl content is a crucial indicator of the status of chloroplasts. Chloroplast is the most important photosynthesizing organelles in plant, play an important role in the absorption and transformation of light energy and are extremely sensitive to adverse conditions. The concentration of Chl was decreased under both stresses. Under salt stress, the Chl concentration may have decreased because the capacity to compartmentalize adverse ion away from the chloroplasts was exceeded and the structures of the grana lamellae and thylakoids in the chloroplasts were damaged, which ultimately resulted in the reduction in the Chl concentration40,41. Both treatments added the same amount of Na+, although alkali stressed seedlings were affected to a greater degree, which may have been related to the precipitation of metal ions and phosphorus together with the disruption of ionic balance and pH homeostasis in plant tissues under alkali stress42,43.
We found that salt stress induced a decline in the Fv/Fo and Fv/Fm values by 34.48 and 30.76%, respectively, in Goji berry treated with 300 mM salt solution, whereas alkali stress induced a reduction in the Fv/Fo and Fv/Fm values by 38.35 and 55.15%, respectively, in Goji berry treated with 300 mM alkali solution. These results are in accordance with the report by Oukarroum et al.44 and are caused by the disruption of electron transport function in photosynthesis and an increase in the number of inactive RCs which results in the inhibition of the QA reduction process and can also lead to the same result under severe stress45. Furthermore, the values of Fv/Fm greater than 0.78 were observed in only the group treated with 50 mM salt solution. This result indicated that the plants in the other treatments were all subjected to stress since the Fv/Fm values were less than 0.78 only when plants grow under stress46. In our experiment, the values of Fv/Fo and Fv/Fm under salt stress were lower than under alkali stress. We also found that salt and alkali stress both induced a decrease in PI values, indicating that plant vitality and PS II function were somewhat suppressed under severe stress47. Comparing the rate of change in Fv/Fm and PI in each treatment, we found that PI declined more than Fv/Fm which is in agreement with the report that PI is much more sensitive than Fv/Fm by PDR van Heerden48. It is generally believed that higher values of Vj indicate decreased plastoquinone levels49. In our results, Vj was highest in Goji berry treated with 300 mM salt and alkali solution. In addition, the value of Vj under alkali stress was higher than that under salt stress. The increase in Vj indicated that electron transfer activity to PSII is inhibited due to the decrease in PQ in the chloroplasts49. The high pH of the alkali stress might damage the photosynthetic machinery and primary electron acceptors, inhibiting the photochemical reaction and the activity of photosystem II50, which results in photoinhibition in the plant and the activation of photoprotection mechanisms that involve blocking electron transport. These changes cause the values of Fo and Fm to simultaneously increase and decrease, respectively, which may be inhibited by the electron transfer process. This can subsequently disrupt the photophosphorylation reaction resulting in the reduction of ATP synthesis and lead to a lower Vj under salt stress than under alkali stress. The augments in Sm, ABS/RC, ETo/RC and DIO/CSo under alkali stress were verified by the prompt reaction of photosynthetic machinery to stress51. The reasonable interpretation of this phenomenon may be the decrease in the efficiency of light energy conversion leading to a self-regulating photosynthetic mechanism that absorbs increased amounts of light energy and is used in the RC activities, thus reducing stress-induced damage.
The results of this experiment indicated that both stresses harmed the cell membrane, which was manifested by the increase in MDA, a product of lipid peroxidation. In addition, alkali stress induced a greater increase in the MDA content than salt stress (Fig. 5A), which indicated that alkali stress caused more damage to Goji berry than salt stress. The salt treatment also increased the CAT, SOD, and POD activity. SOD is the first line of defence against ROS in plants. SOD catalyses the reaction of 2O2− + 2 H + → H2O2 + O2, after which H2O2 is broken down by POD and CAT52. Therefore, it is reasonable to assume that the increase in MDA content under both stresses results in ROS accumulation. Compared with the variations in the MDA content, the CAT, SOD, and POD activities under the salt treatment present a parallel trend with MDA, and the increase in CAT, SOD, and POD activities likely improves the resistance to salt stress by eliminating ROS, which can lead to lipid membrane peroxidation. We found that the critical alkalinity level for Goji berry is 200 mM because at this point the CAT, SOD, and POD activities started to decrease under alkali stress. The alkali-induced increase in CAT, SOD, and POD activities suggested a reduced ability to eliminate ROS. ROS might initiate membrane lipid peroxidation, thereby resulting in reduced membrane lipid unsaturation and membrane protein polymerization and leading to increased membrane permeability. Thus, the accumulation of ROS may have negative effects on cellular tissues and antioxidant system mechanisms, thereby leading to increased mortality in Goji berry. The above results indicate that alkali stress induces greater damage to the antioxidant system than salt stress.
According to the quantitative analysis of the tolerance of Goji berry under salt stress and alkali stress, we found that under the conditions of this experiment, the optimal salt concentration for Goji berry growth ranged from 0 to 246.3 ± 2.1 mM, whereas the optimum alkali concentration ranged from 0 to 126.7 ± 2.7 mM. The relationship between the soil salt content and soil water content was fitted based on the salinity (NaCl) and alkalinity (NaHCO3) thresholds of Goji berry, and the soil water content required to maintain normal growth was determined according to the regression curve. The minimum soil water content to cultivate Goji berry should be 16.22% and 23.37% under mild and moderate salt stress in soils, respectively, and 29.10% and 42.68% under mild and moderate alkali stress, respectively (Fig. 6). In areas with mild and moderate salinity, whether the Goji berry needs to be irrigated or not can be roughly determined according to this method. In addition, whether Goji berry is suitable for planting in the area can be estimated based on the actual conditions, such as soil salinity and long-term achievable soil moisture content. However, because of the limitations of this experiment, the drought stress caused by the soil water content and the complex species of salt or alkali were not considered, and the method is not suitable for the soil with extremely low or high contents of salt or alkali. An extremely low content of salt or alkali corresponds to an extremely low soil water content, which will cause drought stress on plants and inhibit the growth of plants. An extremely high salt or alkali content corresponds to an extremely high soil water content, which might damage plants; moreover, soil water content is difficult to maintain in actual production.
Conclusion
In summary, salt and alkali stress had negative effects on the photosynthesis, Chl fluorescence, and physiology of Goji berry. Both salt and alkali stress reduced all the indicators, and alkali stress was more harmful to Goji berry than salt stress under the same solution concentration. Based on the regression analysis, the thresholds of salt tolerance and alkali tolerance of Goji berry were determined. These thresholds were then used to determine the optimal water content of Goji berry to maintain growth. This research provides insights into the damaging effects of salt stress and alkali stress in general and in Goji berry in particular, and the results of this study can be used as a basic guide for the production and irrigation requirements of Goji berry in Qinghai. Further studies are needed to understand the coupling of multiple salt complex stresses and drought stress as well as the comprehensive relationship between soil salt content and soil water content.
Methods
Plant material, growth conditions and stress conditions
Goji berry seedlings were collected from the Academy of Agriculture and Forestry Sciences, Qinghai University, China. The seedlings were used in a pot experiment that was conducted under rain-protected and ambient conditions in the greenhouse of Qinghai Nuomhon Farm in July 2016. Prior to the planting of seedlings, a pre-test was conducted to determine the optimum moisture content of Goji berry. Twelve well-grown plants were divided into four groups and the water contents of the four groups were set at the following four levels: 10.7%, 12.9%, 15.1%, and 17.2%. The soil water content of the pots was determined by the weighing method, in which the pots were sealed with plastic wrap, weighed and replenished with water at 8:00 every day. The optimal moisture content was determined by monitoring the photosynthetic indices of Goji berry in the four treatments after the pre-test. The optimal moisture content value in Nuomhon area was at the field capacity of 16.9%.
Three weeks after cultivation, uniform one-year-old seedlings (70 cm in height) were individually transplanted into polyethylene pots (36 cm × 34.5 cm × 40 cm), with each containing 10 kg of non-saline sandy soil (0~60 cm) collected from a Goji berry field. This soil was air-dried and passed through a 2-mm sieve to remove large stones, litter, and plant fragments. This soil has a gravimetric water content at the field capacity of 7.5%, a field capacity of 22.7%, a bulk density of 1.6 g cm−3, a pH of 7.1 and a total humus percentage of 2%. The ion concentrations were as follows (mg g−1): CO32−, 0.02; HCO3−, 0.07; Cl−, 0.13; SO42−, 0.36; Na+, 0.15; Ca2+, 0.03; K+, 0.05 and Mg+, 0.02.
The seedlings were divided into 9 groups, among which one group was used as a control, 4 groups were used for the salt stress experiments and 4 groups were used for alkali stress experiments. The plants for the different stress experiments were irrigated with half-strength Hoagland and Arnon’s solution supplemented with 0 (control), 50, 100, 200 or 300 mM NaCl (SS) and the same four NaHCO3 (AS) treatments. Each treatment included six replicates. The stress concentration was treated by adding 50 mM per day until the predetermined concentration was reached to prevent exciting the reaction. All pots were placed in a greenhouse with plastic trays underneath to prevent salt and alkali stress caused by water supplementation. To obtain the response of different stresses in the natural environment, the pots were moved outdoors every morning for natural light irradiation and were covered with a moveable shelter to avoid the impact of rainfall. Additionally, the average day and night temperatures of the greenhouse were 25.5 ± 1.5 °C and 18.5 ± 1.5 °C, respectively, and the photoperiod ranged from 10 to 12 h. The water deficit was calculated by weight, the soil was replenished with deionized water every day, and the moisture was maintained at 16.9% during the course of the experiment.
Chlorophyll pigment content, photosynthetic performance and chlorophyll fluorescence
Chlorophyll a, chlorophyll b and carotenoidss were extracted from fresh fully expanded leaves (1.0 g) with a mixture of acetone, ethanol, and water in a volumetric ratio of 4.5:4.5:1. The absorbance values of the extract were measured using a spectrophotometer (UV-7504; Shanghai Precision Instrument Co., Ltd., Shanghai, China) at wavelengths of 440, 663 and 645 nm. The concentrations of Chl a, Chl b and Cars were determined by the following formulas53:
D440, D663 and D645 are absorbance at 440, 663 and 645 nm, respectively. V is the extraction volume (ml), W is the leaf weight (g) and Chl (a + b) is the sum of the Chl a content and Chl b content.
The net photosynthetic rate (PN), stomatal conductance (gs), transpiration rate (E) and intercellular CO2 concentration (Ci) of leaves were estimated at 8:00, 10:00, 12:00, 14:00, 16:00 and 18:00 on mature leaves using a LI-6400 portable open-flow gas exchange system (LI-COR Biosciences, Lincoln, NE, USA) at 20 days after stress treatment. The values obtained for PN, gS, E and Ci were expressed as μmol m−2 s−1, mol m−2 s−1, mol m−2 s−1and μmol mol−1, respectively. The photosynthetically active radiation was 1 mmol m−2 s−1 (saturation light). The ambient CO2 concentration was 335 ± 15 μmol mol−1, the air temperature was 20 ± 1.5 °C, and the air humidity was approximately 40 ± 5%. Measurements were repeated five times for each of five blades per pot, and the average values were recorded. The water use efficiency (WUE) was calculated according to the following formula:
where WUE is the water use efficiency and PN (μmol m−2 s−1) and E (mmol m−2 s−1) are the net photosynthetic rate and transpiration rate, respectively.
Chl fluorescence was measured using a LI-6400 system with a 6400-40 leaf chamber fluorometer (LI, Washington, DC, USA). Seedlings were preserved in the dark for approximately 30 min, before recording the fluorescence of the blades, which were the same blades for which the photosynthetic parameters were determined. The basal nonvariable Chl fluorescence level (Fo) was determined by a modulated light, which was maintained at a sufficiently low intensity (<1 μmol m−2 s−1) so that notable variations in fluorescence did not occur. The maximum fluorescence induction (Fm) was estimated by a 0.8-s saturation pulse at 4200 μmol m−2 s−1 on dark-adapted leaves. The variable fluorescence (Fv = Fm − Fo) was then calculated. Measurements of the maximum quantum yield of PSII (Fv/Fm) were obtained by application of a saturation light pulse. A number of variables were measured, including the absorption energy flux (ABS), the performance index of intersystem electron acceptors (PI), the electron transport (ETo), the relative variable fluorescence at the J step (Vj), the trapping (TRo), and other Chl fluorescence parameters.
Antioxidant enzyme assays
The MDA content was measured by following the method described by Xu et al.54 SOD activity was assayed by monitoring the capability of the enzyme to inhibit the photochemical reduction of nitro blue tetrazolium (NBT)55. POD activity was analysed by measuring the increase in absorbance at 470 nm recorded 40 s after adding H2O256. CAT activity was determined based on the amount of H2O2 consumed during the reaction57.
Statistical analysis
All data were subjected to a one-way analysis of variance (ANOVA), and the mean differences were compared by the least significant difference (LSD) test at the P ≤ 0.05 level with Duncan’s multiple range test (DMRT). Pearson’s correlation coefficients were also analysed between the parameters. Data are expressed as the mean ± standard error (SE) in the figures and tables. Statistical analyses were performed with SPSS v. 19.0 software (SPSS Inc., Chicago, IL, USA). All data were plotted using OriginPro 2018 (Originlab Corporation Northampton, MA, USA).
References
Peng, Y. L. et al. Eco-physiological characteristics of alfalfa seedlings in response to various mixed salt-alkaline stresses. J Integr Plant Biol 50, 29–39 (2008).
Yang, C. W. et al. Comparative effects of salt-stress and alkali-stress on the growth, photosynthesis, solute accumulation, and ion balance of barley plants. Photosynthetica 47, 79–86 (2009).
Shi, D. C. & Sheng, Y. M. Effect of various salt-alkaline mixed stress conditions on sunflower seedlings and analysis of their stress factors. Environ Exp Bot 54, 8–21 (2005).
van Straten, G., de Vos, A. C., Rozema, J., Bruning, B. & van Bodegom, P. M. An improved methodology to evaluate crop salt tolerance from field trials. Agr Water Manage 213, 375–387 (2019).
Shi, D. C. & Wang, D. L. Effects of various salt-alkaline mixed stresses on Aneurolepidium chinense (Trin.) Kitag. Plant Soil 271, 15–26 (2005).
Anderson, E. K., Voigt, T. B., Kim, S. & Lee, D. K. Determining effects of sodicity and salinity on switchgrass and prairie cordgrass germination and plant growth. Ind Crop Prod 64, 79–87 (2015).
Mokhamed, A. M., Raldugina, G. N., Kholodova, V. P. & Kuznetsov, V. V. Osmolyte accumulation in different rape genotypes under sodium chloride salinity. Russ J Plant Physl 53, 649–655 (2006).
Maimaiti, A. et al. Effects of salinity on growth, photosynthesis, inorganic and organic osmolyte accumulation in Elaeagnus oxycarpa seedlings. Acta Physiol Plant 36, 881–892 (2014).
Huh, G. H. E. A. Salt causes ion disequilibrium-induced programmed cell death in yeast and plants. Plant J 29, 649–659 (2002).
Summart, J., Thanonkeo, P., Panichajakul, S., Prathepha, P. & McManus, M. T. Effect of salt stress on growth, inorganic ion and proline accumulation in Thai aromatic rice, Khao Dawk Mali 105, callus culture. Afr J Biotechnol 9, 145–152 (2010).
de Lacerda, C. F., Cambraia, J., Oliva, M. A., Ruiz, H. A. & Prisco, J. T. Solute accumulation and distribution during shoot and leaf development in two sorghum genotypes under salt stress. Environ Exp Bot 49, 107–120 (2003).
Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ 25, 239–250 (2002).
Cheng, Y. W. E. A. New changes in the plasmamembrane-associated proteome of rice roots under salt stress. Proteomics 9, 3100–3114 (2009).
Hartung, W. et al. Abscisic acid concentration, root pH and anatomy do not explain growth differences of chickpea (Cicer arietinum L.) and lupin (Lupinus angustifolius L.) on acid and alkaline soils. 240, 191–199, https://doi.org/10.1023/a:1015831610452 (2002).
Javid, M., Ford, R. & Nicolas, M. E. Tolerance responses of Brassica juncea to salinity, alkalinity and alkaline salinity. Funct Plant Biol 39, 699–707 (2012).
Qiu, Z. Y., Wang, L. H. & Zhou, Q. Effects of bisphenol A on growth, photosynthesis and chlorophyll fluorescence in above-ground organs of soybean seedlings. Chemosphere 90, 1274–1280 (2013).
Flexas, J. et al. Rapid variations of mesophyll conductance in response to changes in CO2 concentration around leaves. Plant Cell and Environment 30, 1284–1298 (2007).
Qiu, N. W., Lu, Q. T. & Lu, C. M. Photosynthesis, photosystem II efficiency and the xanthophyll cycle in the salt-adapted halophyte Atriplex centralasiatica. New Phytol 159, 479–486 (2003).
Fidalgo, F., Santos, A., Santos, I. & Salema, R. Effects of long-term salt stress on antioxidant defence systems, leaf water relations and chloroplast ultrastructure of potato plants. Ann Appl Biol 145, 185–192 (2004).
Gratani, L., Crescente, M. F., Fabrini, G. & Varone, L. Growth pattern of Bidens cernua L.: relationships between relative growth rate and its physiological and morphological components. Photosynthetica 46, 179–184 (2008).
Bergmann, I., Geiss-Brunschweiger, U., Hagemann, M. & Schoor, A. Salinity tolerance of the chlorophyll b-synthesizing cyanobacterium Prochlorothrix hollandica strain SAG 10.89. Microb Ecol 55, 685–696 (2008).
Kalaji, H. M. et al. Can chlorophyll-a fluorescence parameters be used as bio-indicators to distinguish between drought and salinity stress in Tilia cordata Mill? Environ Exp Bot 152, 149–157 (2018).
Campestre, M. P. et al. Polyamine catabolism is involved in response to salt stress in soybean hypocotyls. J Plant Physiol 168, 1234–1240 (2011).
D, C. The present condition cause of dysfunction and control measures of the alpine grassland ecosystem in Qinghai Province. Pratacultural (in Chinese), 7–11 (2006).
Cao, S. X., Chen, L. & Yu, X. X. Impact of China’s Grain for Green Project on the landscape of vulnerable arid and semi-arid agricultural regions: a case study in northern Shaanxi Province. J Appl Ecol 46, 536–543 (2009).
Wang, Y. et al. In Lycium Barbarum and Human Health (eds Raymond Chuen-Chung Chang & Kwok-Fai So) 1–26 (Springer Netherlands, 2015).
Wei, Y., Xu, X., Tao, H. & Wang, P. Growth performance and physiological response in the halophyte Lycium barbarum grown at salt-affected soil. Ann Appl Biol 149, 263–269 (2006).
Z. Q., W. Salinized Soil of China. Vol. 1–3, 130–136 (in Chinese) (Science Press, 1993).
Sudhir, P. & Murthy, S. D. S. Effects of salt stress on basic processes of photosynthesis. Photosynthetica 42, 481–486 (2004).
Sultana, N., Ikeda, T. & Itoh, R. Effect of NaCl salinity on photosynthesis and dry matter accumulation in developing rice grains. Environ Exp Bot 42, 211–220, https://doi.org/10.1016/S0098-8472(99)00035-0 (1999).
Koyro, H. W. Effect of salinity on growth, photosynthesis, water relations and solute composition of the potential cash crop halophyte Plantago coronopus (L.). Environ Exp Bot 56, 136–146, https://doi.org/10.1016/j.envexpbot.2005.02.001 (2006).
Qin, J. et al. Physiological responses to salinity in Silver buffaloberry (Shepherdia argentea) introduced to Qinghai high-cold and saline area, China. Photosynthetica 48, 51–58 (2010).
Krishnamurthy, S. L. et al. Identification of mega-environments and rice genotypes for general and specific adaptation to saline and alkaline stresses in India. Sci Rep-Uk 7 (2017).
Mercedes-Martin, R. et al. Growing spherulitic calcite grains in saline, hyperalkaline lakes: experimental evaluation of the effects of Mg-clays and organic acids. Sediment Geol 335, 93–102 (2016).
Andriani, A., Tachibana, S. & Itoh, K. Effects of saline-alkaline stress on benzo[a]pyrene biotransformation and ligninolytic enzyme expression by Bjerkandera adusta SM46. World J Microb Biot 32 (2016).
Sorokin, D. Y., Kolganova, T. V., Khijniak, T. V., Jones, B. E. & Kublanov, I. V. Diversity of cultivated aerobic poly-hydrolytic bacteria in saline alkaline soils. Peerj 5 (2017).
Yan, H., Hu, X. T. & Li, F. S. Leaf photosynthesis, chlorophyll fluorescence, ion content and free amino acids in Caragana korshinskii Kom exposed to NaCl stress. Acta Physiol Plant 34, 2285–2295 (2012).
Jiao, Y. et al. Metabolomics and its physiological regulation process reveal the salt tolerant mechanism in Glycine soja seedling roots. Plant Physiol Bioch 126, 187–196 (2018).
Farissi, M. et al. Growth, Nutrients Concentrations, and Enzymes Involved in Plants Nutrition of Alfalfa Populations under Saline Conditions. J Agr Sci Tech-Iran 16, 301–314 (2014).
Santos, C. V. Regulation of chlorophyll biosynthesis and degradation by salt stress in sunflower leaves. Sci Hortic-Amsterdam 103, 93–99 (2004).
Roy, S. J., Negrao, S. & Tester, M. Salt resistant crop plants. Curr Opin Biotech 26, 115–124 (2014).
Lucena, J. Effects of bicarbonate, nitrate and other environmental factors on iron deficiency chlorosis. A review. Vol. 23 (2000).
Yang, C. W., Shi, D. C. & Wang, D. L. Comparative effects of salt and alkali stresses on growth, osmotic adjustment and ionic balance of an alkali-resistant halophyte Suaeda glauca (Bge.). Plant Growth Regul 56, 179–190 (2008).
Oukarroum, A., Bussotti, F., Goltsev, V. & Kalaji, H. M. Correlation between reactive oxygen species production and photochemistry of photosystems I and II in Lemna gibba L. plants under salt stress. Environ Exp Bot 109, 80–88 (2015).
Lu, C. M., Zhang, L. Q. T. & Kuang, J. H. T.Y. Characterization of photosynthetic pigment composition, photosystem II photochemistry and thermal energy dissipation during leaf senescence of wheat plants grown in the field. Journal of Experimental Botany 52, 1805–1810, https://doi.org/10.1093/jexbot/52.362.1805 (2001).
Murchie, E. H. & Lawson, T. Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. Journal of Experimental Botany 64, 3983–3998 (2013).
Schansker, G., Toth, S. Z. & Strasser, R. J. Dark recovery of the Chl a fluorescence transient (OJIP) after light adaptation: The qT-component of non-photochemical quenching is related to an activated photosystem I acceptor side. Bba-Bioenergetics 1757, 787–797 (2006).
van Heerden, P. D. R., Swanepoel, J. W. & Kruer, G. H. J. Modulation of photosynthesis by drought in two desert scrub species exhibiting C-3-mode CO2 assimilation. Environ Exp Bot 61, 124–136 (2007).
Mehta, P., Jajoo, A., Mathur, S. & Bharti, S. Chlorophyll a fluorescence study revealing effects of high salt stress on Photosystem II in wheat leaves. Plant Physiol Bioch 48, 16–20 (2010).
Liu, J. & Shi, D. C. Photosynthesis, chlorophyll fluorescence, inorganic ion and organic acid accumulations of sunflower in responses to salt and salt-alkaline mixed stress. Photosynthetica 48, 127–134 (2010).
Sun, C. X. et al. Unintended effects of genetic transformation on photosynthetic gas exchange, leaf reflectance and plant growth properties in barley (Hordeum vulgare L.). Photosynthetica 51, 22–32 (2013).
Qin, Y. et al. Comparative effects of salt and alkali stress on photosynthesis and root physiology of oat at anthesis. Arch Biol Sci 70, 329–338 (2018).
Lu W. J. & L. Y. S. Plant Physiology Experiment Course. (China Forestry Publishing House, 2012).
Xu, P. L., Guo, Y. K., Bai, J. G., Shang, L. & Wang, X. J. Effects of long-term chilling on ultrastructure and antioxidant activity in leaves of two cucumber cultivars under low light. Physiol Plantarum 132, 467–478 (2008).
Giannopolitis, C. N. & Ries, S. K. Superoxide dismutases: I. Occurrence in higher plants. Plant physiology 59, 309–314 (1977).
Tan, W. et al. Alterations in photosynthesis and antioxidant enzyme activity in winter wheat subjected to post-anthesis water-logging. Photosynthetica 46, 21–27 (2008).
Nakano, Y. & Asada, K. Hydrogen Peroxide is Scavenged by Ascorbate-specific Peroxidase in Spinach Chloroplasts. Plant and cell physiology 22, 867–880, https://doi.org/10.1093/oxfordjournals.pcp.a076232 (1981).
Acknowledgements
This research was supported by the Key Laboratory of State Forestry Administration on Soil and Water Conservation in Beijing Forestry University, Qinghai Academy of Agricultural and Forestry Sciences and Institute of Water Resources and Hydropower of Qinghai Province which also provided the experimental conditions.
Author information
Authors and Affiliations
Contributions
Conceptualization: Zhenzhong Zhang and Kangning He. Experimental instruments and experimental sites supply: Runjie Li. Experiment performance: Zhenzhong Zhang, Tan Zhang and Da Tang. Data curation: Tan Zhang and Da Tang. Data analysis: Zhenzhong Zhang, Tan Zhang and Da Tang. Writing-original draft: Zhenzhong Zhang. Writing-review & editing: Zhenzhong Zhang, Kangning He and Shaofeng Jia.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Z., He, K., Zhang, T. et al. Physiological responses of Goji berry (Lycium barbarum L.) to saline-alkaline soil from Qinghai region, China. Sci Rep 9, 12057 (2019). https://doi.org/10.1038/s41598-019-48514-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48514-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.