Abstract
Available epidemiological data on primary biliary cholangitis (PBC) in Sweden originate from regional studies in the 1980s and may not reflect modern day PBC. We aimed to estimate incidence and prevalence, survival and death causes, and gender differences in PBC. We used international classification of disease (ICD) codes to identify patients with PBC in inpatient and outpatient registries 1987–2014 who were then linked to the Swedish cause of death, cancer and prescribed drug registries. Each PBC patient was matched with 10 reference individuals from the general population. In sensitivity analyses, we examined PBC patients identified through clinical patient records from Karolinska, Sahlgrenska and Örebro University Hospitals. We identified 5,350 adults with PBC. Prevalence of PBC increased steadily from 5.0 (1987) to 34.6 (2014) per 100,000 inhabitants whereas the yearly incidence rate was relatively constant with a median of 2.6 per 100,000 person-years, with a female:male gender ratio of 4:1. Compared to reference individuals, PBC individuals aged 15–39 years at diagnosis had a substantially higher risk of death (Hazard Ratio [HR] 12.7, 95% Confidence Interval [CI] 8.3–19.5) than those diagnosed between 40–59 (HR 4.1, 95% CI 3.7–4.5) and >60 (HR 3.7, 95% CI 3.5–3.9) years of age. Relative risks of mortality were highest in men. In conclusion, we found that recorded prevalence of PBC in Sweden has increased substantially during the last 30 years although incidence has been stable. Patients diagnosed in young adulthood were at a 12.7-fold increased risk of death, and male PBC patients had worse prognosis.
Similar content being viewed by others
Introduction
Primary Biliary Cholangitis (PBC) is a chronic immune-mediated liver disease characterized by progressive cholestasis, biliary fibrosis and eventually cirrhosis1. The disease was formerly known as Primary Biliary Cirrhosis, but this nomenclature was changed as only a minority of patients develop cirrhosis. About two out of three patients treated with ursodeoxycholic acid (UDCA) have an expected survival similar to the general population2. The pathogenesis of PBC remains unclear but the disease seems to be initiated by a combination of susceptible genetic background and exposure to environmental triggers, and as a result the prevalence varies between regions, with the highest incidence rates in northern Europe and Northern America1. The disease predominantly affects women and is typically diagnosed between 50 and 60 years of age1.
The majority of patients are asymptomatic at diagnosis and often PBC is suspected because of abnormal laboratory tests, particularly a raised serum alkaline phosphatase (AP) level. The diagnosis requires that at least 2/3 objective criteria are fulfilled; elevation of AP of liver origin for ≥6 months, elevation of serum anti-mitochondrial antibodies (AMA; titer ≥40) and characteristic histological features. Performing a liver biopsy is not necessary for the diagnosis if the two other criteria are present but is sometimes done for histological staging and/or to diagnose PBC with features of autoimmune hepatitis (AIH)3.
For almost twenty years, UDCA has been the standard and only approved treatment of PBC. It has been shown to prevent histologic progression and to improve survival without transplantation3. However, the response to UDCA varies, and unresponsive patients have a markedly worse prognosis. Regardless of biochemical response, UDCA does not seem to alleviate the cardinal symptoms of PBC: fatigue and pruritus4.
In recent years, much focus has been on finding optimal criteria to define UDCA responders and non-responders. A number of scoring systems have been established, in particular the dichotomized Barcelona5, Paris-16, Rotterdam7, Toronto8, and Paris-29 criteria. In 2015, the non-dichotomized GLOBE score was introduced, which is based on five variables: age at start of UDCA therapy, bilirubin, albumin, AP and platelet count at one year of follow-up10. In 2016, a similar scoring system was presented by UK-PBC that is based on baseline platelet count and serum albumin in combination with 1-year follow-up values of serum bilirubin, transaminases and AP11. The UK-PBC consortium and the Italian PBC group recently also developed a model that based on pretreatment variables is able to accurately predict UDCA response12.
All scoring systems aim to identify those patients who could benefit from therapies other than or in addition to UDCA but also those with a low risk of developing cirrhosis/end-stage liver disease and thus in no need of additional second-line therapy. Recently, obeticholic acid (OCA), a bile acid derivative that act on the nuclear farnesoid X receptor (FXR) was approved as second line therapy for patients not responding or being intolerant to UDCA13. Other agents such as peroxisome proliferator-activated receptor (PPAR) agonists may evolve as approved second line treatments3 and favorable results with bezafibrate have already been published14.
To underscore the need of novel treatment options, updated and robust data on the epidemiology and current management of PBC are needed also for Sweden as available information dates from regional studies in the 1980s15,16,17. This paper aims to provide these data by two complimentary approaches. First, by linking the Swedish inpatient and outpatient registries with the causes of death, cancer and prescribed drug registries, we estimate the contemporary (1) incidence and prevalence of PBC, (2) survival and death causes, (3) gender differences, (4) comorbidities, and (5) adherence to current treatment recommendations and potential second-lines treatments. Secondly, we validate these data from 2005, when the drug prescription registry was established, and provide information about UDCA response rates in different scoring systems, by comprehensively analyzing PBC patient files at Karolinska, Sahlgrenska and Örebro University Hospitals.
Material and Methods
Registry cohorts
A search was performed for adults (15 years of age and older) who had been assigned an International Classification of Diseases (ICD) code for PBC (ICD-9 (571G) until 1996 and/or ICD-10 (K743) from 1997) in the Swedish inpatient (1987–2014) or outpatient (2001–2014) registries. Extracted data were linked to the Swedish causes of death (1987–2014), cancer (1987–2014) and prescribed drug (July 2005–2014) registries (this latter register started in July 2005). We performed a search for liver diseases, liver-related complications and interventions, autoimmune and cardiovascular diseases, and cancer using relevant ICD-9 and ICD-10 codes, and PBC-specific medication by using Anatomic Therapeutic Chemical (ATC) classification codes as listed in Suppl. Table 1.
Unexposed reference cohort
Each individual with PBC was matched for age, sex, calendar period and county with 10 reference individuals without a PBC diagnosis, by the government agency Statistics Sweden. Since this matching was done unintentially at first occurrence of any diagnosis, not necessarily PBC, some reference individuals were deceased at the time when the index individual was first diagnosed with PBC. Deceased reference individuals (n = 3365; 6.2% of the total reference group) were removed from further analyses.
Verification cohort
All information about PBC patients (ICD-10 code K74.3) at Karolinska University Hospital Stockholm, Sahlgrenska University Hospital Gothenburg, and Örebro University Hospital was retrieved from the electronic clinical record file systems, electronic laboratory resources, and the electronic archives. The PBC diagnosis, i.e., fulfilment of at least 2/3 criteria (elevation of serum AP of liver origin for at least 6 months, elevation of serum anti-mitochondrial antibodies (AMA; titre ≥40) or characteristic histological features) was verified and information was gathered about time of diagnosis, PBC with features of AIH, other autoimmune disease, hepatocellular carcinoma, and time of liver transplantation or death. All patients with PBC diagnosed in 2005–2014 were selected for further assessment of medication and to evaluate the first year of UDCA treatment.
For every patient, UDCA treatment and all relevant variables for the seven scoring systems were recorded, such as sex, age (both current and at start of UDCA treatment), height, weight and BMI (calculated) at diagnosis, laboratory parameters at start of UDCA and at 1-year follow-up including bilirubin, ALT/AST, AP, albumin, platelet count, creatinine and PK-INR (using the spread sheet shown as Suppl. Table 2). When relevant data from the main hospital were missing, referring hospitals or caregivers were contacted by mail. In a few cases, where sufficient data were still not available, patients were excluded from all analyses.
All scores were calculated according to their original publications. Five of the scoring systems (Paris I, Paris II, Barcelona, Toronto, Rotterdam5,6,7,8,9) yield a dichotomized result, UDCA responder or non-responder, whereas the other two (UK-PBC risk score and GLOBE Score10,11) yield numeric scores and calculate risk or transplant-free survival at 3, 5, 10 and 15 years, respectively. For the Globe Score we also used a cut off value of 0.3 to define whether a patient is at high-risk for significantly shorter times of transplant-free survival or not10.
Ethical approval
This study was approved, and individual informed consent was waived as this study was observational in nature18, by the regional ethics committees in Gothenburg (diary numbers, Dnr: 135–12, 870–15) and Uppsala (Dnr: 2016–262).
Statistics
Incidence and prevalence were calculated using Swedish total population data from Statistics Sweden as a reference for each calendar year. Cox proportional hazards models were used to analyze differences in survival between the register cohort and the matched reference cohort. The models were stratified where each stratum contained one patient from the register cohort and the related matched reference individuals. Differences in comorbilities were tested using a 2-sample proportion test.
Chi square test was used to calculate differences between the register cohort and the verification cohort regarding sex and numbers treated. Since the verification cohort consists of patients from two large University Hospitals (Karolinska and Sahlgrenska) and one minor only recently approved University Hospital (Örebro), we compared these small cohorts internally using Chi square and Chi square for trends when appropriate. To calculate treatment response of individual biochemical variables, we used paired t-test. A p-value < 0.05 was considered statistically significant.
Results
Registry cohort
Incidence and prevalence
We identified 5,350 adults (15 years of age and older) with a PBC diagnosis and 50,145 reference individuals (Suppl. Table 3). The PBC incidence rate was around 2.6 per 100,000 person-years (Fig. 1) with some variability that mainly can be explained by the start of different registers (a likely overestimation 1987–1988, as well as 2001–2002), change in ICD-coding system and changes in care of patients. There was a steady increase in the registered prevalence of PBC from 5.0 (1987) to 34.6 (2014) per 100,000 inhabitants (Fig. 1 and Suppl. Table 4), also with some variability due to the difference in available registers. From 2004 to 2014 the data is however likely to cover the full prevalent cohort of PBC diagnosed patients, where the incidence is very stable, and the increase in prevalence is close to linear.
Gender relation
The median age of the whole study population was 64 years at diagnosis (range 15–94), and 9 out of 10 patients were born in Sweden (Suppl. Table 3). The female:male ratio in the whole study population was 4.0:1, with a higher proportion of women (5.4:1, 84.5%) in the outpatient population 2001–2014 (Suppl. Table 3).
Survival and liver events
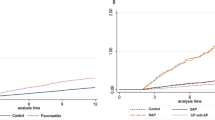
Patients with PBC were at increased risk of death (Suppl. Table 6A). This was most evident for men of whom only 37.4%, in contrast to 58.9% of women, were alive 10 years after the first diagnosis of PBC, despite almost identical mean age at diagnosis (61.2 for men and 61.9 for women), as shown in the Kaplan-Meier plot (Fig. 2). The highest risk of death was observed in the first year after PBC diagnosis (Hazard Ratio, HR 9.0). The risk declined to a HR of 2.9 after more than 5 years (Suppl. Table 6A). Stratified by period of diagnosis, the risk constantly declined, from HR 5.6 during 1984–1989 to 3.0 to 2002–2015 (Suppl. Table 6B). Young age at diagnosis of PBC substantially increased the risk of death, with HR 12.7 at 15–39 years as compared to HR 4.1 between 40–59, and HR 3.7 from 60 years of age on (Suppl. Table 6C).
Significantly more men than women with PBC were diagnosed with liver complications such as esophageal varices and/or gastric varices, liver failure or ascites within ten years (32.9% vs. 17.1%, Fig. 3 and Suppl. Table 6D). Of note, survival analyses could not be stratified for whether or not the patients were treated with and responders to UDCA.
Causes of death
Patients with PBC had a significantly (p < 0.0001) increased risk of death from diagnoses related to digestive (HR, 22.85), cardiovascular (HR 1.43), cancer (HR 1.77), respiratory (HR 1.76) and other disease (HR 1.98) groups (Table 1). The major causes of death in PBC patients were digestive diseases related (22.9% vs. 2.5% in reference individuals), most of them liver related (13.8% vs. 0.2%, Table 2). Although, when hepatic complications such as liver cirrhosis, liver failure, esophageal or gastric varices, and ascites were removed from all digestive disease-related death diagnoses, relative mortality was still high (HR 8.8, Table 1). Since digestive and liver related death causes predominated in PBC patients, other common death causes were reported in relatively lower proportion, such as circulatory diseases (22.9% vs. 43.6%) and all cancers (18.7% vs. 25.4%), but hepatocellular cancer (HCC) was about 20 times more often cause of death as in reference individuals (4.1% vs. 0.2%, Table 2).
Co-morbidities
At the time of diagnosis, patients with PBC compared to reference individuals had significantly (p < 0.001) more often a comorbidity diagnosis within all disease groups (Suppl. Table 7): infectious diseases (1.8% vs. 0.3%, in particular viral hepatitis), sarcoidosis (0.4% vs. 0.1%), endocrine and metabolic diseases (15.2% vs. 6.4%, in particular thyroid disease and diabetes mellitus, both type 1 and 2), circulatory diseases (28.0% vs. 14.4%, in particular essential hypertension) and among other than liver–related digestive diseases, Crohn’s disease (1.0% vs. 0.3%), ulcerative colitis (2.6% vs. 0.4%) and celiac disease (1.1% vs. 0.1%), There was also a higher prevalence of discoid lupus erythematosus (0.3% vs. 0.1%), psoriasis (2.2% vs. 0.9%) and a four-fold higher prevalence of musculoskeletal diseases (10.1% vs. 2.5%, in particular systemic lupus erythematosus and sicca/Sjögren syndrome).
Significant differences (p ≤ 0.001) for additional co-morbidities between PBC and reference individuals emerged throughout life-time follow-up (Suppl. Table 7), in particular for cancer: whereas liver cancer was more often found in patients with PBC than in reference individuals (4.4% vs. 0.3%), interestingly, it was the opposite for female cancer forms, breast cancer (3.8% vs. 5.1%), corpus uteri cancer (0.8% vs. 1.3%) and ovarian cancer (0.4% vs. 0.9%). Of note, there were no differences in any form of ischemic heart disease and actually a lower occurrence of cerebral infarction (5.8% vs. 7.1%). The diagnosis of primary sclerosing cholangitis (PSC) was not given to any reference individual but increased from 1.1% at baseline to 2.7% at any time in PBC patients, as did AIH, from 1.9% to 4.9%, during the course of PBC.
PBC related drug treatment and liver transplantation
Data for drug treatment were available from July 1, 2005. UDCA was administered as the recommended first line treatment to 79.9% of PBC patients (Suppl. Table 8), and usually within about a month after diagnosis, whereas second line treatments such as fibrates (0.5%) or budesonide (5.5%), including patients having PBC with features of AIH, were rare. Immunomodulators such as azathioprine, budesonide, 5-mercaptopurine and prednisolone were administered to at least 37.2% of PBC patients (Suppl. Table 8).
Liver transplantations for PBC were in Sweden performed from 1997 on, at a median number of 9.5 per year after PBC diagnosis (range, 4–14) (Suppl. Table 9). Related to the prevalence of PBC in Sweden, some 0.5% of the patients were transplanted annually, with no significant trend over time.
Verification cohort characteristics and UDCA response rates
At Karolinska, Sahlgrenska and Örebro University Hospitals, 259 patients were diagnosed with incident PBC between 2004 and 2015 of which 231 patients with a mean age at diagnosis of 58.1 ± 1.6 years were suitable for UDCA response evaluation. Mean follow up was 6.4 ± 0.8 years, 200 (85.8%) were women (for additional characteristics see Table 3). Patients who did not fulfill the European Association for the Study of Liver (EASL) criteria for PBC3 (n = 8), who had insufficient data (n = 17) or had a follow up shorter than 12 months (n = 3) were excluded.
UDCA was prescribed to 194 (84.0%) of all 231 patients, with no differences between women and men. Younger patients tended to be prescribed UDCA more often than elder ones: about 95.0% of those diagnosed with PBC between 18–39 years of age compared to 85.7 of those diagnosed between 40–59 and 80.2% of those over 60 years, p = 0.086 (adjusted for sex and age at diagnosis).
The verification cohort demonstrated higher AP levels (5.1 x upper limit normal (ULN), 3.0–7.5; median, interquartile range)) than the large cohorts of the GLOBE score10 (2.11 x ULN; 1.37–3.79) and UK-PBC Risk Score11 (1.9 x ULN; 1.2–3.5). AP but not transaminase levels changed significantly after one year of UDCA treatment. The median AP levels decreased from 5.1 x ULN (3.0–7.5) to 2.9 x ULN (1.7–5.6) (p < 0.0001), which is a reduction by 43.1%.
Overall treatment response rates varied between 73.3 ± 7.7% % for Paris 1 and 59.2 ± 2.7% % for Paris 2 criteria, with the other criteria in between, with no significant differences between Hospitals (Table 3). By summarizing all five dichotomized systems, 82 (42.3%) of all 194 UDCA-treated patients were unanimously identified as responders, and only 9 (4.6%) as non-responders. Additional 36 patients were UDCA-responders according to 4, and 10 according to 3 scoring systems.
For the GLOBE Score, separate cut off values were defined for different age groups to define High Risk patients regardless of age. In our material 26 (13.4%) patients were defined as High Risk patients. The UK-PBC risk score was calculated for 158 of the 194 treated patients (the data set was incomplete for 38 patients). Median hazard ratios and IQRs of a liver event (liver death or liver transplantation) were: after 5 years; 0.71 (0.41–1.18), after 10 years, 2.34 (1.36–3.90), and after 15 years, 4.32 (2.51–7.14).
Discussion
This nationwide population-based study found a prevalence rate of PBC in Sweden of about 35 per 100,000 inhabitants at the end of the observation period. This is substantially higher than the 9.2–15.1 per 100,000 reported >30 years ago and based on only 162 patients15,16,17. Our study included 5,350 adult PBC cases and the prevalence is among the highest reported worldwide19, higher than in Danmark but close to Iceland (19.0 and 38.3/100,000 inhabitants, respectively, in 2009/201020,21). We found a female to male ratio of 4.0:1, which is almost identical to that in Denmark (4.2:1)21. As we did not observe a significant trend in the incidence rates between 1987 and 2014, the linearly increasing prevalence rates argue against influence of practice changes regarding input of data into the registries during the time period.
Our data do not show significantly increased overall survival since the introduction of UDCA in the Swedish health care in 1998. UDCA is now considered as recommended first line treatment for PBC patients. However, 20.1% of Swedish PBC patients were not treated with UDCA, at least during the last 10 years of follow-up when prescribed drug data is available, which is in line with a recently reported rate of UDCA intolerance13. We were not able to stratify overall mortality or clinical disease progression according to UDCA response, since these data were unavailable in our registry cohort. Second line treatment of PBC according to previous EASL guidelines, i.e., fibrates or budesonide, was rare22. Immunomodulators, such as azathiopurine, budesonide, 5-mercaptopurine and prednis(ol)one were most likely administered for the treatment of other immune-mediated endocrine, gastrointestinal, musculoskeletal and skin diseases rather than true AIH-PBC overlap.
Indeed, our registry study confirms a strong association of PBC with other immune-mediated endocrine, musculoskeletal and skin diseases such as thyroid disease, sicca syndrome, discoid and systemic lupus erythematosus, but also with inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis. Of note, our registry study found that 1.1% of PBC patients also had a diagnosis of celiac disease. This percentage is lower than previously reported (3.1%23). The finding of 2.7% of PSC among the PBC-patients in the registry cohort, is somewhat troublesome, especially since overlap PBC-PSC is rare, and all earlier research is based solely on case-reports. Most probably this reflects misdiagnosis.
Consistent with UK data24, Swedish PBC patients are at increased risk of death, with an excess mortality especially in men. Men were in the early years also over-represented among the inpatients. As expected, we found an excess mortality due to liver-related deaths. PBC patients had overall lower cancer mortality despite a substantially increased risk of HCC. Our finding of a 19% overall cancer mortality is higher than old data from Sweden (12%)25 but in perfect agreement with a recent meta-analysis that found both an increased overall risk for cancer (pooled rate ratio, RR, 1.55) and for HCC (pooled RR 18.8) in PBC but not for other cancers26.
Our verification cohort shows similar characteristics to PBC cohorts around the world5,6,7,8,9,10,11, and confirmed registry findings for 2005–2014 regarding gender ratio and adherence to treatment with UDCA. Between 15% and 20% of PBC patients are male and only about 80% of PBC patients are treated with UDCA. A bias might be introduced by the low UDCA prescription rate of 61.3% for PBC patients at Örebro University Hospital but still, treatment rates of 87.5% at Karolinska and Sahlgrenska University Hospitals were less than expected.
We developed a spread sheet for clinical and biochemical data that was easy to use and immediately reported UDCA response in all major PBC scoring systems (Suppl. Table 2). Our verification cohort demonstrated higher AP levels than the large cohorts of the GLOBE10 and UK-PBC Risk Scores11, but so did also the cohorts used for the Paris 16, Paris 29 and Barcelona5 criteria. We observed a decrease of AP levels after one year of treatment with UDCA of 43.1%, which is about the same as the decreases of 45% and 49%, respectively, in the Paris-1 and Paris-2 cohorts, respectively, and more than the decreases of 36% and 37%, respectively, in the GLOBE and UK-PBC cohorts. Treatment response rates in our verification cohort were between 59.2% (Paris 2) and 73.3% (Paris 1). This is similar to earlier findings that up to 30% show inadequate biochemical response to UDCA (rising to >50% in patients presenting under the age of 40 years)24. In the UK cohort, 80% of patients responded to treatment according to the Paris-1 criteria24, a figure similar to the 73.3% in our verification cohort, further supporting the use of at least this easy-to-use criterion in clinical practice, in particular outside specialized centers. Of note, Paris 2 criteria have been designed specifically to better fit early-stage patients9 but disease stage information was not available from our verification cohort. Otherwise Paris 1 is recommended, as this score evaluates the two most important biochemical parameters, bilirubin and AP. Sereval studies have found that this score best discriminates between low-risk and high-risk PBC patients22.
Smartphone applications may in the near future allow easy application of more complex algorithms to estimate outcome10,11 and potentially even without awaiting UDCA response12 and thus ensure timely treatment27. This has important consequences both for the individual patient, in choosing candidates for second line treatment, for future research and for health economic purposes as patients with a good prognosis could be managed at a primary care level.
Strengths and Limitations
In this nationwide population-based study spanning almost 30 years of follow-up we evaluated more than 5000 individuals with PBC. Through the unique personal identity number28 we were able to link study participants to age- and sex-matched reference individuals for comparisions but also to major healthcare registers. The personal identity number of all participants also guarantees a virtually complete follow-up. We used the Swedish Patient Register to identify PBC. While we are unaware of any validation of PBC, the positive predictive value for most chronic diagnoses in this register is 85–95%29. The current study was not designed to examine sensitivity of the in-patient vs. out-patient-registries per se, but this has been looked at before30. Swedish healthcare is tax-funded and this should rule ut major selection bias due to socioeconomic status30. Finally, we were able to verify our data using an extensive verification cohort from three different university clinics. Among the weaknesses is the fact that the Patient Register only include outpatient care since 2001 and this may explain the initial increase in prevalence and incidence after 2001. Also, we were only able to examine UDCA treatment since 2005, because the Prescribed Drug Register started in this year.
Conclusion
In conclusion, this nationwide population-based cohort found a stable incidence but rising prevalence of PBC in Sweden, with a much higher proportion of men than reported earlier. PBC has a substantial morbidity and mortality, especially from liver related causes. First-line therapy, and where UDCA fails second-line treatment, are crucial for patient management.
References
Carey, E. J., Ali, A. H. & Lindor, K. D. Primary biliary cirrhosis. Lancet 386, 1565–1575, https://doi.org/10.1016/S0140-6736(15)00154-3 (2015).
Beuers, U. et al. Changing nomenclature for PBC: From ‘cirrhosis’ to ‘cholangitis’. J. Hepatol. 63, 1285–1287, https://doi.org/10.1016/j.jhep.2015.06.031 (2015).
Liver, E. A. F. T. S. O. T. EASL Clinical Practice Guidelines: The diagnosis and management of patients with primary biliary cholangitis. J. Hepatol. 67, 145–172, https://doi.org/10.1016/j.jhep.2017.03.022 (2017).
Dyson, J. K. et al. Novel therapeutic targets in primary biliary cirrhosis. Nat. Rev. Gastroenterol. Hepatol. 12, 147–158, https://doi.org/10.1038/nrgastro.2015.12 (2015).
Pares, A., Caballeria, L. & Rodes, J. Excellent long-term survival in patients with primary biliary cirrhosis and biochemical response to ursodeoxycholic Acid. Gastroenterology 130, 715–720, https://doi.org/10.1053/j.gastro.2005.12.029 (2006).
Corpechot, C. et al. Biochemical response to ursodeoxycholic acid and long-term prognosis in primary biliary cirrhosis. Hepatology 48, 871–877, https://doi.org/10.1002/hep.22428 (2008).
Kuiper, E. M. et al. Improved prognosis of patients with primary biliary cirrhosis that have a biochemical response to ursodeoxycholic acid. Gastroenterology 136, 1281–1287, https://doi.org/10.1053/j.gastro.2009.01.003 (2009).
Kumagi, T. et al. Baseline ductopenia and treatment response predict long-term histological progression in primary biliary cirrhosis. Am. J. Gastroenterol. 105, 2186–2194, https://doi.org/10.1038/ajg.2010.216 (2010).
Corpechot, C., Chazouilleres, O. & Poupon, R. Early primary biliary cirrhosis: biochemical response to treatment and prediction of long-term outcome. J. Hepatol. 55, 1361–1367, https://doi.org/10.1016/j.jhep.2011.02.031 (2011).
Lammers, W. J. et al. Development and Validation of a Scoring System to Predict Outcomes of Patients With Primary Biliary Cirrhosis Receiving Ursodeoxycholic Acid Therapy. Gastroenterology 149, 1804–1812 e1804, https://doi.org/10.1053/j.gastro.2015.07.061 (2015).
Carbone, M. et al. The UK-PBC risk scores: Derivation and validation of a scoring system for long-term prediction of end-stage liver disease in primary biliary cholangitis. Hepatology 63, 930–950, https://doi.org/10.1002/hep.28017 (2016).
Carbone, M. et al. Pretreatment prediction of response to ursodeoxycholic acid in primary biliary cholangitis: development and validation of the UDCA Response Score. Lancet Gastroenterol Hepatol, https://doi.org/10.1016/S2468-1253(18)30163-8 (2018).
Nevens, F. et al. A Placebo-Controlled Trial of Obeticholic Acid in Primary Biliary Cholangitis. N. Engl. J. Med. 375, 631–643, https://doi.org/10.1056/NEJMoa1509840 (2016).
Corpechot, C. et al. A Placebo-Controlled Trial of Bezafibrate in Primary Biliary Cholangitis. N. Engl. J. Med. 378, 2171–2181, https://doi.org/10.1056/NEJMoa1714519 (2018).
Eriksson, S. & Lindgren, S. The prevalence and clinical spectrum of primary biliary cirrhosis in a defined population. Scand. J. Gastroenterol. 19, 971–976 (1984).
Lofgren, J., Jarnerot, G., Danielsson, D. & Hemdal, I. Incidence and prevalence of primary biliary cirrhosis in a defined population in Sweden. Scand. J. Gastroenterol. 20, 647–650 (1985).
Danielsson, A., Boqvist, L. & Uddenfeldt, P. Epidemiology of primary biliary cirrhosis in a defined rural population in the northern part of Sweden. Hepatology 11, 458–464 (1990).
Ludvigsson, J. F. et al. Registers of the Swedish total population and their use in medical research. Eur. J. Epidemiol. 31, 125–136, https://doi.org/10.1007/s10654-016-0117-y (2016).
Boonstra, K., Beuers, U. & Ponsioen, C. Y. Epidemiology of primary sclerosing cholangitis and primary biliary cirrhosis: a systematic review. J. Hepatol. 56, 1181–1188, https://doi.org/10.1016/j.jhep.2011.10.025 (2012).
Baldursdottir, T. R. et al. The epidemiology and natural history of primary biliary cirrhosis: a nationwide population-based study. Eur. J. Gastroenterol. Hepatol. 24, 824–830, https://doi.org/10.1097/MEG.0b013e328353753d (2012).
Lleo, A. et al. Evolving Trends in Female to Male Incidence and Male Mortality of Primary Biliary Cholangitis. Sci. Rep. 6, 25906, https://doi.org/10.1038/srep25906 (2016).
EASL Clinical Practice Guidelines: management of cholestatic liver diseases. J. Hepatol. 51, 237–267, S0168-8278(09)00309-2 [pii], https://doi.org/10.1016/j.jhep.2009.04.009 (2009).
Volta, U., Caio, G., Tovoli, F. & De Giorgio, R. Gut-liver axis: an immune link between celiac disease and primary biliary cirrhosis. Expert Rev. Gastroenterol. Hepatol. 7, 253–261, https://doi.org/10.1586/egh.13.5 (2013).
Carbone, M. et al. Sex and age are determinants of the clinical phenotype of primary biliary cirrhosis and response to ursodeoxycholic acid. Gastroenterology 144, 560–569 e567; quiz e513-564, https://doi.org/10.1053/j.gastro.2012.12.005 (2013).
Loof, L. et al. Cancer risk in primary biliary cirrhosis: a population-based study from Sweden. Hepatology 20, 101–104 (1994).
Liang, Y., Yang, Z. & Zhong, R. Primary biliary cirrhosis and cancer risk: a systematic review and meta-analysis. Hepatology 56, 1409–1417, https://doi.org/10.1002/hep.25788 (2012).
Marschall, H. U. Ensuring timely treatment of patients with primary biliary cholangitis. Lancet Gastroenterol Hepatol, https://doi.org/10.1016/S2468-1253(18)30177-8 (2018).
Ludvigsson, J. F., Otterblad-Olausson, P., Pettersson, B. U. & Ekbom, A. The Swedish personal identity number: possibilities and pitfalls in healthcare and medical research. Eur. J. Epidemiol. 24, 659–667, https://doi.org/10.1007/s10654-009-9350-y (2009).
Ludvigsson, J. F. et al. External review and validation of the Swedish national inpatient register. BMC Public Health 11, 450, https://doi.org/10.1186/1471-2458-11-450 (2011).
Anell, A. The public-private pendulum–patient choice and equity in Sweden. N. Engl. J. Med. 372, 1–4, https://doi.org/10.1056/NEJMp1411430 (2015).
Acknowledgements
This study was financed an unrestricted grant by Intercept Pharma (16003).
Author information
Authors and Affiliations
Contributions
H.U.M., S.W. and J.L. designed the study and drafted the manuscript. H.U.M., I.H., S.L. and S.W. analyzed the patients’ records, F.S., M.T. and J.L. analysed the registry data All authors provided critical input to the final version of manuscript.
Corresponding author
Ethics declarations
Competing Interests
H.U.M. received a grant (for this study) by Intercept, and consultant fees and material support, by Albireo and Intercept. All other authors have nothing to declare.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marschall, HU., Henriksson, I., Lindberg, S. et al. Incidence, prevalence, and outcome of primary biliary cholangitis in a nationwide Swedish population-based cohort. Sci Rep 9, 11525 (2019). https://doi.org/10.1038/s41598-019-47890-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47890-2
This article is cited by
-
Epidemiology and clinical course of primary biliary cholangitis in Eastern Slovakia
International Journal of Public Health (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.