Abstract
Bariatric surgery in morbid obesity, either through sleeve gastrectomy (SG) or Roux-Y gastric bypass (RYGB), leads to sustainable weight loss, improvement of metabolic disorders and changes in intestinal microbiota. Yet, the relationship between changes in gut microbiota, weight loss and surgical procedure remains incompletely understood. We determined temporal changes in microbiota composition in 45 obese patients undergoing crash diet followed by SG (n = 22) or RYGB (n = 23). Intestinal microbiota composition was determined before intervention (baseline, S1), 2 weeks after crash diet (S2), and 1 week (S3), 3 months (S4) and 6 months (S5) after surgery. Relative to S1, the microbial diversity index declined at S2 and S3 (p < 0.05), and gradually returned to baseline levels at S5. Rikenellaceae relative abundance increased and Ruminococcaceae and Streptococcaceae abundance decreased at S2 (p < 0.05). At S3, Bifidobacteriaceae abundance decreased, whereas those of Streptococcaceae and Enterobacteriaceae increased (p < 0.05). Increased weight loss between S3-S5 was not associated with major changes in microbiota composition. No significant differences appeared between both surgical procedures. In conclusion, undergoing a crash diet and bariatric surgery were associated with an immediate but temporary decline in microbial diversity, with immediate and permanent changes in microbiota composition, independent of surgery type.
Similar content being viewed by others
Introduction
Bariatric surgery is the only sustainable effective treatment for obesity1. Surgical procedures such as Roux-Y Gastric bypass (RYGB) and sleeve gastrectomy (SG) facilitate a 50–70% decrease in excess body weight and fat mass1. In addition, surgery leads to decreased caloric intake or malabsorption and to metabolic changes, such as an improved glucose metabolism, and is associated with a changed intestinal microbiota2,3,4. The role of altered host-microbial interactions in this process is incompletely understood1. Studies on the composition of the distal gut microbiota in obesity and after RYGB in humans and rodents yielded long lasting changes in types and relative proportions of enteric bacteria3,5,6,7,8. Furthermore, transfer of the gut microbiota from RYGB-treated mice to non-operated, germ-free mice resulted in weight loss and decreased fat mass in the recipient animals5. These findings support a direct effect of the microbiota on weight and adiposity. Recently Liu et al. demonstrate using metagenomic shotgun sequencing that the abundance of glutamate-fermenting Bacteriodes thetaiotaomincron is decreased in obese Chinese individuals and glutamate levels are increased9. Weight loss induced by SG partially reversed metabolic and microbial alterations, including reduced abundance of B. thetaiotaomicron and increased serum glutamate9.
To further elucidate the results of the entire bariatric surgery procedure on the intestinal microbiota composition we investigated sequentially collected stool samples from 45 morbid obese patients undergoing either RYGB or SG at five different time points before and after surgery.
Results
RYGB and SG resulted in significant decrease of BMI in all patients
In this study, 45 Caucasian Dutch patients were included with an average age of 43 years, 36 (84%) being female, 11 (24%) using proton pump inhibitors and 4 (9%) having type 2 Diabetes Mellitus at baseline (Table 1). After a crash diet, 22 patients underwent SG and 23 underwent RYGB. At baseline the mean BMI was 42.9 (+/−6.56) and 43 (+/−4.13) for patients undergoing RYGB and SG, respectively. At 6 months after the procedure BMI declined to 30.81 (+/−5.35) and 31.52 (+/−3.86), respectively, with no significant difference based on surgery type (Table 1).
Crash diet reduces microbial alpha diversity, which is restored to baseline levels 6 months after surgery, irrespective of surgery type
In total 221 fecal samples were collected, with 4 samples missing from 4 unique times points from 4 different patients. Using a pre-defined cut-off value of 20001 reads, 220 samples could be analyzed.
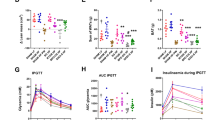
The initial crash diet had a strong effect on total microbiota diversity as the Shannon diversity index declined from 4.5 at baseline (S1) to a minimum value of 4.0 after the crash diet (p < 0.05) (S2) and then gradually returned to 4.5 at 3 (S4) and 6 months (S5) after surgery (Fig. 1A). Differences in diversity are reflected by an initial decrease and subsequent rise in numbers of distinct microbial OTUs. At baseline, 3 months and 6 months after surgery more than 500 OTUs were identified, whereas after the crash diet and at 1 week after surgery this number was reduced to below 400 OTUs (Fig. 1B).
Observed and estimated richness of gut microbiota at different time points during the bariatric surgery procedure. (A) Shannon diversity index estimated a decrease in bacterial richness at S2 and S3. (B) Rarefaction curves showed a reduction in bacterial richness at S2 and S3. (C) Principal component analysis (PCA) plot of similarity between the samples; each dot represents 1 sample, each color a different time point. S1. before surgery (red); S2. after 2 weeks of crash diet (orange); S3. 1 week after surgery (yellow); S4. 3 months after surgery (light blue); S5. 6 months after surgery (dark blue).
Patients at baseline (S1) and after crash diet (S2) are more similar to each other when compared to the time points after surgery (S3, S4, S5), which cluster together, as demonstrated by Principle Component Analysis (PCA) (Fig. 1C).
Distinct microbial changes appear directly after crash diet, but are replaced by persistent distinct changes shortly after surgery
Significant changes in total relative abundance of specific families in the different time points were observed (Fig. 2H). After the crash diet (S2) there was a significant reduction in relative abundance of 2 microbial families, Streptococcaceae and Ruminococcaceae (Fig. 2A,D), and a significant increase in 1 family, Rikenellaceae (Fig. 2E). Subsequent comparison of the microbial composition pre-surgery (S2) and 1-week post-surgery (S3) revealed a significant increase in the relative abundance of Streptococcaceae and Enterobacteriaceae families (Fig. 2A,B) and a decrease in Bifidobacteriaceae, which persisted until 6 months post-surgery (S5) (Fig. 2C). In these 6 months (at S5) microbiota complexity was restored (Fig. 1A,B), which when compared with 1 week after surgery (S3) coincided with increased relative abundance of low abundance families Veillonellaceae and the Clostridiales order with no further family classification (Fig. 2F,G).
Relative abundance of bacterial families in the gut microbiota at the five time points analyzed. (A–G) Boxplots show the average relative abundance of 7 families that significantly changed between 2 different time points. (A) Streptococcaceae. (B) Enterobacteriaceae. (C) Bifidobacteriaceae. (D) Ruminococcaceae. (E) Rikenellaceae. (F) Veillonellaceae. (G) O_Clostridiales_f_others. H. Relative abundance of all families identified at the different time points. Significant families are represented in the same color. Asterisk (in red) indicates significant fold change differences (p < 0,05) analyzed by ANCOM.
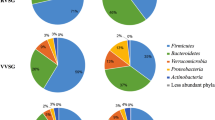
When RYGB and SG surgery was analyzed separately, no significant differences in microbiota composition based on beta diversity and relative abundance was observed at baseline (S1) (Fig. 3A), 1 week (S3) (Fig. 3B) or 6 months after surgery (S5) (Fig. 3C) between patients that underwent either SG or RYGB (Fig. 3D).
(A–C). Principal coordinate analysis (PCoA) plots comparing beta diversity of Sleeve Gastrectomy (SG) versus Roux-Y Gastric bypass (RYGB) surgery at baseline (S1) (A) 1 week after surgery (S3) (B) and 6 months after surgery (S5). (C) SG is indicated in red, RYGB is indicated in blue. (D) Relative abundance of bacterial families in the gut microbiota at the five time points analyzed in SG versus RYGB surgery.
Significant associations between microbiota changes and clinical markers
Clinical parameters in patients were analyzed at baseline and 6 months after surgery. Besides weight loss, serum levels of vitamin D, B6, cholesterol, bilirubin, HbA1c, iron, ferritin and folate improved 6 months after surgery when compared to baseline (FDR-adjusted, p < 0.05, Table 1).
These changes were associated to overall differences in microbial abundance in relation to the changes in clinical parameters at S5 versus S1, which are highlighted in Fig. 4. Significant associations were only found in low abundance families. Increased bilirubin levels was associated with decreased abundance of Prevotellaceae, Bacteroidales and Peptococcaceae taxa; and increased iron level was associated with increased abundance of Pasteurellaceae. In addition, a decreased HbA1c was associated with a decrease in Coriobacteriaceae and an increase in Clostridiales taxa. The most pronounced measured effect in the dataset was a negative association between Prevotellaceae, Veillonellaceae, Streptococcaceae, Bifidobacteriaceae and Enterobacteriaceae taxa in relation to decreased serum cholesterol level, whereas the most pronounced positive association was found between Lachnospiraceae and Coriobacteriaceae taxa in relation to decreased cholesterol level (Fig. 4). Yet, these associations were not statistically significant after FDR adjustment (Fig. 4).
Association between clinical parameters and family taxa calculated based on the difference between 6 months after surgery (S6) and baseline (S1). Significant associations (false discovery rate (FDR) adjusted p-value < 0.05) are indicated with an “x”. The red color indicates a positive association and the blue color a negative association. HbA1c, glycated hemoglobin; VitD, vitamin D; VitB6, vitamin B6.
Discussion
The novel aspect of this study resides in microbiota profiles before and very shortly after bariatric surgery with subsequent follow-up profiles. We describe the sequential impact of a crash diet followed by either RYGB or SG surgery, resulting in progressive weight loss and changes in the gut microbiota composition. Several other studies report sequential sampling of patients after bariatric surgery, but none of the studies define timing of the baseline sample in relation to a crash diet3,9,10,11,12,13, and, therefore, renders the relative impact of the different measures in the bariatric procedure difficult to dissect. In addition, this study is unique in that a very early postsurgical sampling time point is included. Apart from substantial weight loss and improvements in clinical parameters, as reported by others, bariatric surgery induces long-lasting changes in microbiota composition in most patients. The most apparent immediate change in microbiota composition occurred after the crash diet, with a concurrent reduction in alpha diversity, whereas surgery was associated with early and sustained replacement of distinct bacterial taxa and restoration of the diversity.
Although significant microbial changes are identified in the gut microbiota of bariatric surgery patients, at 6 months after surgery the total microbial diversity was similar to microbial diversity measured at baseline. This sudden decline in alpha diversity probably reflects a severe stress on the human microbiota by a crash diet, with a significant change in catabolic state14. Persisting post-surgery microbiota changes suggest adaptation to anatomic and physiologic changes, such as reduced acid production, increased oxygen content, altered bile acid concentrations delivered to the colon, induced by the surgery. Previous reported effects of bariatric surgery on microbiota diversity have ranged from an increase in total diversity15,16 to absence of change and even a decrease in alpha diversity17. We suspect that baseline sampling in relation to crash diet may vary between studies, and might contribute to the reported differences between studies. In addition, follow-up longer than 6 months after surgery, might result in further, yet relatively smaller, increase in alpha diversity.
Besides a stable bacterial alpha diversity after surgery, we observed profound differences after each consecutive intervention on bacterial taxa composition.
The crash diet immediately resulted in an increase in the relative abundance of Bifidobacteriaceae and decrease in the relative abundance of Streptococcaceae, whereas the opposite effect was observed after surgery; an increase in abundance of Streptococcaceae and decline in Bifidobacteriaceae that persisted for at least 6 months was observed. Increased abundance of Veillonellaceae, like increased abundance of Streptococcaceae, may reflect survival of oral microbiota into the intestine. In addition, persistent increase in Enterobacteriaceae after surgery, confirms previous sustained changes reported in humans and animal models (rats), associated with increased pH7,14,18,19. Other main differences, exposure to undigested nutrients and biliopancreatic enzymes, may play important roles in the microbial composition, intestinal permeability and intestinal adaptation20. Since increased intestinal permeability is associated with inflammation and reduction in alpha diversity, which is also associated with obesity, it remains questionable whether restoration of alpha diversity to baseline level, instead of further increase, may also reflect persistent inflammation in the post-surgery state at 6 months, which has been previously related to increase in Enterobacteriaceae10. This corresponds to the observed higher alpha diversity of fecal samples from a healthy normal weight cohort compared to the slightly lower diversity of postoperative patients10.
Although others observed microbiota changes only after RYGB5, here we observed this in both surgery types. This suggests that despite the 2 procedures result in distinct anatomic differences, this did not seem to influence the post-surgery changes in relative abundance of Bifidobacteriaceae, Streptococcaceae and Enterobacteriaceae observed amongst both patient groups and which were similar for both types of surgery. Interestingly, unlike Liu et al.9 and Ilhan et al.21 both patients groups here after surgery develop comparable weight loss irrespective of surgery type, and this may explain why we find similar changes in gut microbiota composition. Also baseline characteristics did not differ significantly. Moreover, we suggest that bariatric surgery in itself, unlike crash diet, results in an altered long-lasting composition of the microbiota.
Although a significant association with changed clinical parameters between baseline and 6 months after surgery was lacking, the relative abundance of Bifidobacteriaceae, Streptococcaceae and Enterobacteriaceae taxa changed significantly shortly after surgery. This sudden adjustment further confirms that the altered postoperative microbiota more likely reflects surgery induced effects, rather than improved clinical parameters10,13,21. We observed a significant association between increased serum bilirubin level and decreased relative abundance of Bacteroidales, Peptococcaceae and Prevotellaceae taxa in this dataset. The abundance of Bacteroidales in the gut microbiota could contribute to the increase in bilirubin level, since Bacteroides fragilis, which is part of Bacteroidales taxa, is one of the bacterial species described to be able to metabolize bilirubin in the gut22,23. In addition, a decreased HbA1c was found significantly associated with decreased Coriobacteriaceae and increased Clostridiales taxa. Nevertheless, the exact meaning of changes of these low abundance taxa is unknown.
This study failed to confirm the suggested relationship between increased abundance of Firmicutes and Bacteroidetes and obesity24,25, as the relative abundance of the family members of these phyla remained stable before and after surgery, despite significant weight loss. In addition, other studies described that Faecalibacterium (F. prausnitzii) was assumed to play a role in inflammation and glucose homeostasis in obesity with a reduced relative abundance after RYGB surgery3,8,26,27. In our study, a decreased abundance of the Ruminococcaceae family, to which F. prausnitzii belongs, was observed after the crash diet, yet this change did not sustain after surgery.
In conclusion, here we illustrate that temporal sampling of bariatric surgery patients with subsequent microbiota analysis can lead to increased insights into the relative contribution of interventions on stability and composition of the microbiota. We show that a crash diet invoked profound temporary changes in total microbiota diversity and composition, yet surgery precluded early fixed changes of microbial composition and restoration of the microbial diversity that likely contribute to weight loss.
Subjects and Methods
Ethics statement
The study protocol was approved and in accordance with the regulations of the Ethics Committee of Catharina Hospital Eindhoven (study number 2014–3). Written informed consent was not required according to the regulations regarding non-invasive sampling (Dutch law on Medical Research in Humans). All 45 patients were informed they could withdraw samples from on-going studies at any time without need for clarification.
Study design
In this observational study patients with morbid obesity were recruited from the Catharina Hospital Eindhoven out-patient obesity clinic between September 2014 and November 2014. All 45 patients fulfilled the criteria for bariatric surgery and were screened before surgery for eligibility by a team including a surgeon, dietician and a psychologist. Two weeks before the planned surgery, all patients were subjected to a crash diet (commercially available Modifast intensive diet) consisting of 500 calories a day for 2 weeks. Type of surgery was determined based on clinical criteria and shared decision making between surgeon and patient. Patients with reflux disease (GERD) or diabetes were offered a RYGB. Patients that were dependent on medication that needed a specific therapeutic concentration (for example psychiatric medication) or patients with previous abdominal surgery making a gastric bypass more difficult were offered a sleeve. In patients in which both options were possible, patient preference was also weighed. During surgery, patients received 1 g cefazolin antibiotic prophylaxis intravenously. All included patients did not receive antibiotics in the 6 months before the start of the study other than preoperative cefazolin. After hospitalization, general practitioners managed adjustments of insulin, oral diabetics and other medication in the home setting. Patients visited the out-patient clinic at 3, 6 and 12 months for evaluation and will remain in follow up for 5 years.
Sample collection and DNA extraction
Stool samples (Sterilin specimen container, Thermo-Fisher) were gathered at the out-patient clinic or at patient homes. Samples were always stored in the freezer and collected at the homes of the patients using dry-ice and stored at the hospital at −80 °C. Sample were collected at 5 different time points; before the start of the crash diet (S1), 2 weeks after the crash diet (S2), and 1 week (S3), 3 months (S4) and 5 to 6 months after surgery (S5).
Total bacterial DNA from feces samples was isolated according to Godon et al.28. When isolated DNA contained PCR inhibitors (20% of the samples random distributed over the time points), samples were submitted to an extra step of isopropanol precipitation and column purification with QiAamp stool mini kit (Qiagen). DNA was stored at −20 °C prior to further analysis.
16S rRNA gene sequencing strategy and analysis
A 469 bp encompassing the V3 and V4 hypervariable regions of the 16S rRNA gene was amplified and sequenced using the Illumina MiSeq Reagent Kit v3 (600-cycle) on an Illumina MiSeq instrument according to Fadrosh et al.29. Negative controls, buffer controls were included in the DNA extraction, amplification and sequencing protocol to monitor for potential contamination. A total of 3 amplicon pools were sequenced, generating 8.9, 7.8 and 14.4 (mean of 10.3) million total reads. These 2 × 300 bp paired-end reads were pre-processed as follows. The first 12 bp of each paired-end containing the index sequences were extracted and afterwards concatenated to dual-index barcodes of 24 bp specific for each read-pair and sample. Paired reads were merged, as an overlap of about 90 bp was expected, using FLASH (version 1.2.11)30. Subsequently, these merged reads were de-multiplexed using the split_libraries_fastq.py script rom and analyzed by the QIIME microbial community analysis pipeline (version 1.9.1)31. Quality filtering was also performed during this step, truncating reads with an average PHRED quality score of 20 or less. After removal of the barcodes, heterogeneity spacers, and primer sequences about 19.8 million sequences were left with a mean length of 410 bp (median length of 405). The obtained sequences with a minimum of 97% similarity were assigned to operational taxonomic units (OTUs) using QIIME’s open-reference OTU picking workflow (pick_open_reference_otus.py). This workflow was carried out using USEARCH (version 6.1.544)32 for OTU picking, in addition to detection and removal of chimeric sequences. The obtained OTU sequences were aligned to the Greengenes 16 S rRNA gene database (gg_13_8_otus), followed by removal of OTUs represented by less than 0.005% of the total number of sequences. The generated OTU table and phylogenetic tree were used for assessing alpha- and beta-diversity using QIIME’s core_diversity_analyses.py workflow with a rarefaction depth of 20001 sequences. The weighted UniFrac distance was used to calculate beta-diversity of the samples, while the Shannon index was used for the alpha-diversity. For Principal Component Analysis (PCA) R 3.5.0 in an environment of RStudio 1.1.383 (RStudio Team, Boston, MA)33 was employed, using zCompositions, clr transformation and ggpplot R packages10,34,35.
Statistical analysis
Microbiota changes between time points and operation types were investigated using ANCOM36 in R 3.3.333 with Benjamini-Hochberg37 correction for multiple testing using an alpha of 0.05 as a threshold for significance. Changes in the clinical parameters (BMI, vitamin D, vitamin B6, cholesterol, bilirubin, glycated hemoglobin (HbA1c), iron, ferritin and folate) between baseline and 6 months after surgery was analyzed by applying t-test in Prism GraphPad (version 7.0). Associations between changes in total read counts at family level (at baseline versus 6 months after surgery) and changes in patient characteristics (at baseline versus 6 months after surgery) were investigated using a linear regression model. To eliminate possible confounding effects, age and sex were included as covariates. For these analyses, changes in total read counts were used as outcome, whereas changes in patient characteristics were used as predictor (model: change_in_read_counts ∼ β1·age + β2·sex + β3·change_in_patient_characteristic). For association analysis R 3.5.0 in an environment of RStudio 1.1.383 (RStudio Team, Boston, MA) was employed33. Results are presented using pheatmap package (https://CRAN.R-project.org/package=pheatmap). To correct for the multiple testing, we have used adjusted q-values by an optimized false discovery rates (FDR) approach as in Strimmer et al.38.
References
Tilg, H. Obesity, metabolic syndrome, and microbiota: multiple interactions. J Clin Gastroenterol 44(Suppl 1), S16–18, https://doi.org/10.1097/MCG.0b013e3181dd8b64 (2010).
Kobyliak, N. et al. Probiotics in prevention and treatment of obesity: a critical view. Nutr Metab (Lond) 13, 14, https://doi.org/10.1186/s12986-016-0067-0 (2016).
Furet, J. P. et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: links with metabolic and low-grade inflammation markers. Diabetes 59, 3049–3057, https://doi.org/10.2337/db10-0253 (2010).
Kong, L. C. et al. Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am J Clin Nutr 98, 16–24, https://doi.org/10.3945/ajcn.113.058743 (2013).
Liou, A. P. et al. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med 5, 178ra141, https://doi.org/10.1126/scitranslmed.3005687 (2013).
Sweeney, T. E. & Morton, J. M. The human gut microbiome: a review of the effect of obesity and surgically induced weight loss. JAMA Surg 148, 563–569, https://doi.org/10.1001/jamasurg.2013.5 (2013).
Zhang, H. et al. Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci USA 106, 2365–2370, https://doi.org/10.1073/pnas.0812600106 (2009).
Graessler, J. et al. Metagenomic sequencing of the human gut microbiome before and after bariatric surgery in obese patients with type 2 diabetes: correlation with inflammatory and metabolic parameters. Pharmacogenomics J 13, 514–522, https://doi.org/10.1038/tpj.2012.43 (2013).
Liu, R. et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med 23, 859–868, https://doi.org/10.1038/nm.4358 (2017).
Palleja, A. et al. Roux-en-Y gastric bypass surgery of morbidly obese patients induces swift and persistent changes of the individual gut microbiota. Genome Med 8, 67, https://doi.org/10.1186/s13073-016-0312-1 (2016).
Murphy, R. et al. Differential Changes in Gut Microbiota After Gastric Bypass and Sleeve Gastrectomy Bariatric Surgery Vary According to Diabetes Remission. Obes Surg 27, 917–925, https://doi.org/10.1007/s11695-016-2399-2 (2017).
Tremaroli, V. et al. Roux-en-Y Gastric Bypass and Vertical Banded Gastroplasty Induce Long-Term Changes on the Human Gut Microbiome Contributing to Fat Mass Regulation. Cell Metab 22, 228–238, https://doi.org/10.1016/j.cmet.2015.07.009 (2015).
Aron-Wisnewsky, J. et al. Major microbiota dysbiosis in severe obesity: fate after bariatric surgery. Gut, https://doi.org/10.1136/gutjnl-2018-316103 (2018).
Li, J. V. et al. Metabolic surgery profoundly influences gut microbial-host metabolic cross-talk. Gut 60, 1214–1223, https://doi.org/10.1136/gut.2010.234708 (2011).
Guo, Y. et al. Gut microbiota after Roux-en-Y gastric bypass and sleeve gastrectomy in a diabetic rat model: Increased diversity and associations of discriminant genera with metabolic changes. Diabetes Metab Res Rev 33, https://doi.org/10.1002/dmrr.2857 (2017).
Basso, N. et al. Insulin Resistance, Microbiota, and Fat Distribution Changes by a New Model of Vertical Sleeve Gastrectomy in Obese Rats. Diabetes 65, 2990–3001, https://doi.org/10.2337/db16-0039 (2016).
Ward, E. K. et al. The effect of PPI use on human gut microbiota and weight loss in patients undergoing laparoscopic Roux-en-Y gastric bypass. Obes Surg 24, 1567–1571, https://doi.org/10.1007/s11695-014-1275-1 (2014).
Patrone, V. et al. Postoperative Changes in Fecal Bacterial Communities and Fermentation Products in Obese Patients Undergoing Bilio-Intestinal Bypass. Front Microbiol 7, 200, https://doi.org/10.3389/fmicb.2016.00200 (2016).
Shao, Y. et al. Alterations of Gut Microbiota After Roux-en-Y Gastric Bypass and Sleeve Gastrectomy in Sprague-Dawley Rats. Obes Surg 27, 295–302, https://doi.org/10.1007/s11695-016-2297-7 (2017).
Guo, Y. et al. Modulation of the gut microbiome: a systematic review of the effect of bariatric surgery. Eur J Endocrinol 178, 43–56, https://doi.org/10.1530/EJE-17-0403 (2018).
Ilhan, Z. E. et al. Distinctive microbiomes and metabolites linked with weight loss after gastric bypass, but not gastric banding. ISME J 11, 2047–2058, https://doi.org/10.1038/ismej.2017.71 (2017).
Fahmy, K., Gray, C. H. & Nicholson, D. C. The reduction of bile pigments by faecal and intestinal bacteria. Biochimica et Biophysica Acta (BBA) - General Subjects 264, https://doi.org/10.1016/0304-4165(72)90119-5 (1972).
Hamoud, A. R, Weaver, L., Stec, D. E. & Hinds, T. D Jr. Bilirubin in the Liver–Gut Signaling Axis. Trends in Endocrinology & Metabolism 29, https://doi.org/10.1016/j.tem.2018.01.002 (2018).
Turnbaugh, P. J., Backhed, F., Fulton, L. & Gordon, J. I. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 3, 213–223, https://doi.org/10.1016/j.chom.2008.02.015 (2008).
le Roux, C. W. et al. Gastric bypass reduces fat intake and preference. Am J Physiol Regul Integr Comp Physiol 301, R1057–1066, https://doi.org/10.1152/ajpregu.00139.2011 (2011).
Damms-Machado, A. et al. Effects of surgical and dietary weight loss therapy for obesity on gut microbiota composition and nutrient absorption. Biomed Res Int 2015, 806248, https://doi.org/10.1155/2015/806248 (2015).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci USA 105, 16731–16736, https://doi.org/10.1073/pnas.0804812105 (2008).
Godon, J. J., Zumstein, E., Dabert, P., Habouzit, F. & Moletta, R. Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl Environ Microbiol 63, 2802–2813 (1997).
Fadrosh, D. W. et al. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2, 6, https://doi.org/10.1186/2049-2618-2-6 (2014).
Magoc, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957–2963, https://doi.org/10.1093/bioinformatics/btr507 (2011).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7, 335–336, https://doi.org/10.1038/nmeth.f.303 (2010).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461, https://doi.org/10.1093/bioinformatics/btq461 (2010).
R: A language and environment for statistical computing (2018).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York (2016).
Gregory, B. & Gloor, G. R. Compositional analysis: a valid approach to analyze microbiome high-throughput sequencing data. Canadian Journal of Microbiology 62, https://doi.org/10.1139/cjm-2015-0821 (2016).
Mandal, S. et al. Analysis of composition of microbiomes: a novel method for studying microbial composition. Microb Ecol Health Dis 26, 27663, https://doi.org/10.3402/mehd.v26.27663 (2015).
Benjamini, Y. H., Yose. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society 57 (1995).
Strimmer, K. A unified approach to false discovery rate estimation. BMC Bioinformatics 9, 303, https://doi.org/10.1186/1471-2105-9-303 (2008).
Author information
Authors and Affiliations
Contributions
Authors contribution: conceived and designed the experiments: M.L., D.A., M.J.M.B., A.C.F., H.L.L. Performed the experiments: M.L., D.A., A.P.A.H., J.G., M.C.V. Analyzed the data: F.L.P., M.R.C.R., C.M.H., H.W.U., R.M.B. Wrote the paper: F.L.P., M.L., C.M.H., H.W.U., M.R.C.R., D.A., R.M.B., A.P.A.H., M.C.V., J.G., M.J.M.B., A.C.F., R.J.L.W., H.L.L. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Paganelli, F.L., Luyer, M., Hazelbag, C.M. et al. Roux-Y Gastric Bypass and Sleeve Gastrectomy directly change gut microbiota composition independent of surgery type. Sci Rep 9, 10979 (2019). https://doi.org/10.1038/s41598-019-47332-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47332-z
This article is cited by
-
Hypoabsorptive surgeries cause limb-dependent changes in the gut endocannabinoidome and microbiome in association with beneficial metabolic effects
International Journal of Obesity (2023)
-
Roles of the gut microbiome in weight management
Nature Reviews Microbiology (2023)
-
Implications of Bariatric Surgery on the Pharmacokinetics of Antiretrovirals in People Living with HIV
Clinical Pharmacokinetics (2022)
-
Renal Function Following Bariatric Surgery: a Literature Review of Potential Mechanisms
Obesity Surgery (2022)
-
A review on gut microbiota: a central factor in the pathophysiology of obesity
Lipids in Health and Disease (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.