Abstract
Pulmonary hypertension is a fatal disease, however reliable prognostic tools are lacking. Heart rhythm complexity analysis is derived from non-linear heart rate variability (HRV) analysis and has shown excellent performance in predicting clinical outcomes in several cardiovascular diseases. However, heart rhythm complexity has not previously been studied in pulmonary hypertension patients. We prospectively analyzed 57 patients with pulmonary hypertension (31 with pulmonary arterial hypertension and 26 with chronic thromboembolic pulmonary hypertension) and compared them to 57 age- and sex-matched control subjects. Heart rhythm complexity including detrended fluctuation analysis (DFA) and multiscale entropy (MSE) and linear HRV parameters were analyzed. The patients with pulmonary hypertension had significantly lower mean RR, SDRR, pNN20, VLF, LF, LF/HF ratio, DFAα1, MSE slope 5, scale 5, area 1–5 and area 6–20 compared to the controls. Receiver operating characteristic curve analysis showed that heart rhythm complexity parameters were better than traditional HRV parameters to predict pulmonary hypertension. Among all parameters, scale 5 had the greatest power to differentiate the pulmonary hypertension patients from controls (AUC: 0.845, P < 0.001). Furthermore, adding heart rhythm complexity parameters significantly improved the discriminatory power of the traditional HRV parameters in both net reclassification improvement and integrated discrimination improvement models. In conclusion, the patients with pulmonary hypertension had worse heart rhythm complexity. MSE parameters, especially scale 5, had excellent single discriminatory power to predict whether or not patients had pulmonary hypertension.
Similar content being viewed by others
Introduction
Pulmonary hypertension is a progressive and debilitating diseases caused by complex and heterogeneous pathogeneses1,2. Pulmonary artery hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) are important subgroups of pulmonary hypertension which share similar hemodynamic physiology3,4. Both diseases are characterized by progressive precapillary vessel arteriopathy with progressive elevated pulmonary vascular resistance5,6, and patients with these diseases have a poor prognosis if untreated7,8. The major causes of death include right heart failure, sudden cardiac death and respiratory failure9,10.
Changes in linear heart rate variability (HRV) have been reported to be associated with pulmonary hypertension and its cardiovascular outcomes11,12,13. In addition, changes in linear HRV have been shown to be partially reversible after treatment with subcutaneous treprostinil14. This suggests that the deterioration in linear HRV in patients with pulmonary hypertension may be associated with hemodynamic abnormalities.
In addition to traditional linear HRV parameters, new methods using non-linear HRV analysis have been developed in recent years which have focused on measuring complexity instead of variability beneath heart rate dynamics15,16. Two of the most frequently used methods to estimate heart rhythm complexity are detrended fluctuation analysis (DFA) and multiscale entropy (MSE). In previous studies, both DFA and MSE have shown better predictive ability for clinical outcomes in many diseases compared with traditional HRV analysis17,18,19,20. However, studies of heart rhythm complexity in patients with pulmonary hypertension are lacking. In this study, we aimed to evaluate changes in heart rhythm complexity in patients with pulmonary hypertension and the potential clinical applications.
Results
Patient characteristics
The clinical, echocardiographic and hemodynamic data and information on pulmonary hypertension-specific medications are presented in Table 1. The patients with pulmonary hypertension had a significantly lower body mass index (BMI), lower prevalence of hypertension and higher tricuspid regurgitation peak gradient (TRPG) than the controls. In medication, the control group had significantly higher rates of beta blocker, calcium channel blocker (CCB) and angiotensin II receptor blocker (ARB) or angiotensin-converting enzyme inhibitors (ACEI) use. Other parameters were compatible between the two groups except for data on hemodynamics and medications for pulmonary hypertension which were only available in the patients with pulmonary hypertension. The mean pulmonary arterial pressure, pulmonary capillary wedge pressure, cardiac output and pulmonary vascular resistance in the patients with pulmonary hypertension were 46 ± 15 mmHg, 13 ± 4 mmHg, 4.1 ± 1.5 L/min and 723 ± 419 dyn·s·cm−5, respectively.
Holter data
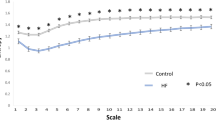
In linear analysis, the patients with pulmonary hypertension had significantly lower mean RR, standard deviation of R-R intervals (SDRR), percentage of absolute differences in normal RR intervals greater than 20 ms (pNN20), very low frequency (VLF), low frequency (LF) and low frequency/high frequency (LF/HF) ratio compared to the controls (Table 2). In heart rhythm complexity parameters including MSE and DFA, DFAα1, MSE slope 5, scale 5, area under the MSE curve for scale 1–5 (area 1–5) and 6–20 (area 6–20) were significantly lower in the pulmonary hypertension group compared to the control group. The value of DFAα2 was comparable between the two groups (Table 2). The entropies of different time scales of MSE curves were significantly different between the patients with and without pulmonary hypertension (Fig. 1).
Logistic regression analysis to predict pulmonary hypertension
In univariate logistic regression, lower linear HRV parameters including mean RR, VLF and LF/HF ratio, and lower heart rhythm complexity including DFAα1, MSE slope 5, scale 5, area 1–5 and area 6–20 were significantly associated with the presence of pulmonary hypertension. These parameters were further analyzed using multivariate logistic regression, which showed that lower mean RR, DFAα1 and scale 5 were significantly associated with pulmonary hypertension (Table 3). Then, these 3 parameters including mean RR, DFAα1 and MSE scale 5 were adjusted by age, sex, BMI, HTN, DM, beta blocker, CCB and ARB or ACEI use in different models. In the five models with different adjustments, only DFAα1 and MSE scale 5 remained as independent predictors of pulmonary hypertension (Table 4).
Comparisons of all linear HRV and heart rhythm complexity parameters to differentiate the patients with and without pulmonary hypertension
Receiver operating characteristic (ROC) curve analysis showed that MSE scale 5 had the greatest discriminatory power to predict the presence of pulmonary hypertension among all linear and heart rhythm complexity parameters (area under the curve [AUC] 0.845).
The AUCs of other linear and non-linear HRV parameters including mean RR, SDRR, pNN20, pNN50, VLF, LF, HF, LF/HF ratio, DFAα1, DFAα2, slope 5, area 1–5, and area 6–20 were 0.660, 0.610, 0.630, 0.493, 0.681, 0.609, 0.432, 0.748, 0.745, 0.545, 0.644, 0.777 and 0.794, respectively (Fig. 2). In addition, the AUCs of clinical parameters including BMI, coronary artery disease, diabetes mellitus and hypertension were 0.355, 0.526, 0.489 and 0.341, respectively.
Analysis of the discrimination power of the two group by receiver operating characteristic curve analysis. The areas under the curve of mean RR, SDRR, VLF, LF, LF/HF ratio, DFAα1, MSE slope 5, scale 5, area 1–5 and area 6–20 were 0.660, 0.610, 0.681, 0.609, 0.748, 0.745, 0.644, 0.845, 0.777 and 0.794, respectively.
The advantage of adding DFA or MSE parameters to the linear parameters to discriminate the presence of pulmonary hypertension
MSE parameters including scale 5, area 1–5 and area 6–20 significantly improved the discriminatory power of mean RR, pNN20, VLF, LF and LF/HF ratio in both net reclassification improvement (NRI) and integrated discrimination improvement (IDI) models. DFAα1 significantly improved the discriminatory power of mean RR, pNN20, VLF and LF in both NRI and IDI models and LF/HF ratio in IDI model (Table 5).
Discussion
The three major findings of this study are: (1) the patients with pulmonary hypertension had both worse HRV and heart rhythm complexity compared to those without pulmonary hypertension; (2) MSE scale 5 had the greatest single discriminatory power to detect the presence of pulmonary hypertension among all HRV and clinical parameters; (3) the combination of linear HRV and heart rhythm complexity parameters improved the discriminatory power to predict pulmonary hypertension.
Patients with pulmonary hypertension have a poor prognosis, even after using pulmonary hypertension-specific drugs21. The major causes of death are right heart failure and sudden death, which account for about 60% of all cases of mortality22,23. Unlike in left heart failure, ventricular tachycardia or fibrillation is relatively rare in patients with pulmonary hypertension. Instead, severe bradycardia and pulseless electrical activity are the most common causes of sudden cardiac death in PAH22. A possible predisposing factor for arrhythmia in PAH is modulation of autonomic activity9,22,24. Elevated levels of serum norepinephrine and its association with pulmonary vascular resistance25 support the hypothesis of increased sympathetic activity in patients with PAH.
Heart rate variability is a validated and non-invasive tool to evaluate cardiac autonomic function26. Folino et al. reported decreased HRV and increased ventricular ectopy in patients with PAH27, and Bienias and Witte et al. also reported worse linear HRV in patients with pulmonary hypertension23,24. In the current study, we demonstrated similar results which highlight the prominent autonomic dysregulation in patients with pulmonary hypertension. In addition to linear HRV analysis, heart rhythm complexity analysis derived from non-linear HRV analysis has been studied as a better predictor of outcomes in many diseases compared with linear analysis. Data obtained from the DIAMOND-CHF trial showed that heart rhythm complexity impairment was the strongest electrocardiographic risk predictor, exceeding the value of traditional linear HRV analysis28. However, non-linear HRV analysis has never been reported in patients with pulmonary hypertension. To the best of our knowledge, non-linear HRV analysis has only been reported in one animal model experiment, in which Gonçalves et al. demonstrated decreases in both linear and non-linear HRV parameters in a rat model of monocrotaline-induced pulmonary hypertension29.
Heart rhythm complexity derived from non-linear analysis including DFA and MSE based on fractal and chaos theories, respectively, focuses on measuring the complexity beneath seemingly stationary biological signals15,16. A normal healthy subject is capable of making adjustments to deal with a dynamic environment through highly complex multisystemic cooperation. In a diseased subject, the balance in the systems breaks down and the complexity decreases. Heart rhythm complexity analysis can quantify this complexity, and it has been studied in many different diseases with excellent results. It has been associated with the prognosis of heart failure20, outcomes of acute stroke17, primary aldosteronism30, severity of abdominal aorta calcification19, critical illnesses requiring extracorporeal life support18 and post-myocardial infarction heart function31. In the current study, heart rhythm complexity, and especially MSE scale 5, had a better discriminatory power for pulmonary hypertension compared to linear HRV analysis. The DFAα1 and MSE scale 5 remained as independent predictors of pulmonary hypertension after clinical parameters adjustments. Furthermore, a combination of heart rhythm complexity and linear HRV analysis further significantly improved the predictive power of linear HRV parameters to differentiate between the patients with and without pulmonary hypertension. Our results provide valuable evidence supporting an altered autonomic system and decreased heart rhythm complexity in patients with pulmonary hypertension.
There are several limitations to this study. First, this is a small pilot study and the data were only derived from both PAH and CTEPH patients. Patients with other pulmonary hypertension groups such as group 2 (left heart disease related) or group 3 (pulmonary disease/hypoxia related) were not included in this study. The results of this study should be confirmed in larger clinical studies. Second, the baseline characteristics including BMI, prevalence of HTN and medication were different in control and pulmonary hypertension groups which may still be confounders in this study. Third, the baseline physical activity difference between these two group may influence the HRV parameters and may also be a confounder in this study. Fourth, this is a cross-sectional study without long-term follow-up data. Further studies are needed to evaluate the prognostic value of heart rhythm complexity in patients with pulmonary hypertension.
In conclusion, heart rhythm complexity could predict the presence of pulmonary hypertension in this study, and MSE scale 5 had the greatest single discriminatory power. In addition, heart rhythm complexity parameters including DFA and MSE significantly improved the discriminatory power of linear HRV parameters, which supports the advantage of combining linear and heart rhythm complexity parameters.
Methods
Patients
We prospectively enrolled 57 Taiwanese patients with pulmonary hypertension, including 31 patients with PAH (World Health Organization, WHO group 1) and 26 patients with CTEPH (WHO group 4). Patients with left heart disease (WHO group 2) and COPD (WHO group 3) were not enrolled in this study to prevent bias deprived from complexed and heterogenous disease mechanisms among different pulmonary hypertension groups. The diagnosed and categorized of pulmonary hypertension were based on ESC guideline. For the control group, we enrolled 57 age- and sex-matched participants who admitted to our hospital and received coronary angiogram examination which revealed patent coronary artery. Patients with chronic pulmonary disease, chronic atrial fibrillation, prior myocardial infarction, left heart failure, cerebrovascular events, or peripheral artery disease were excluded.
All subjects in this study received echocardiography and 24-h ambulatory ECG Holter recording. All patients with pulmonary hypertension received right heart catheterization to confirm the diagnosis. The baseline characteristics, medical history and biochemistry data were recorded at enrollment. Holter recordings were performed one month before or after (mostly one week before) right heart catheterization in the patients with pulmonary hypertension. In the control group, Holter recordings were performed within one week after coronary angiogram.
This study was approved by the Institutional Review Board of National Taiwan University Hospital, and all subjects provided written informed consent. All research was performed in accordance with relevant guidelines and regulations.
Echocardiography
All subjects received standard transthoracic echocardiography (iE33 xMATRIX Echocardiography System, Philips, Amsterdam, Netherlands). The TRPG was determined from the peak flow velocity of tricuspid regurgitation (TRV) using a simplified Bernoulli equation: TRPG = 4 × TRV2, and LVEF (M-mode) was measured via a parasternal long axis view in accordance with the recommendations of the American Society of Echocardiography32.
24-hour Holter recording and data pre-processing
24-h ambulatory ECG Holter (Zymed DigiTrak Plus 24-Hour Holter Monitor Recorder and Digitrak XT Holter Recorder 24 Hour, Philips, Amsterdam, Netherlands) recordings were conducted in all subjects. All subjects maintained their daily activity during the examination. A stable 4-h segment of daytime RR intervals (between 9 AM and 5 PM) was selected. The selected data were automatically annotated using an algorithm, and then examined by two experienced technicians. MATLAB program with self-writing code was used to derive HRV parameters for signal processing.
Linear analysis
Traditional linear HRV including time and frequency domain analysis was conducted according to the recommendations of the North American Society of Pacing Electrophysiology and the European Society of Cardiology33. Time domain HRV parameters including mean RR, SDRR, pNN20 and pNN50 were calculated to represent the sympathetic and parasympathetic modulation of heart beats. The frequency domain parameters, high frequency (HF; 0.15–0.4 Hz), low frequency (LF; 0.04–0.15 Hz), and very low frequency (VLF; 0.003–0.04 Hz) were analyzed after Fourier transformation.
Non-linear analysis
Non-linear HRV analysis focuses on the complexity of heart rate dynamics. Two non-linear methods, MSE and DFA, were conducted in this study based on fractal and chaos theories, respectively.
Detrended fluctuation analysis
DFA provides a mathematical algorithm to uncover the fractal behavior beneath seemingly nonstationary RR dynamics by removing these trends from the integrated time series and quantifying the degree of self-affinity based on fractal theory15. DFA was performed by summing the detrended integrated time series in each scale. The log-log plots of fluctuations against time scales were constructed, and the slope (α exponent) of the plot represented the fractal correlation property of the time series. A crossover phenomenon of α exponents of RR dynamics was observed in normal and diseased subjects. Therefore, short (α1; 4–11 beats) and long (α2; 11–64 beats) time scales were calculated to better understand the fractal property of the heart rate dynamics.
Multiscale entropy analysis
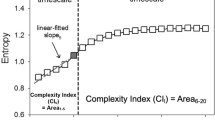
MSE analysis can be used to estimate the entropies of physiological signals in different time scales, and it can also be used to predict sequential changes over different time scales16. Since traditional entropy analysis can only evaluate the single entropy of a biological signal, MSE uses a coarse-graining process (i.e. averaging consecutive beats to form a new time series) to construct many different time scales. After this process, the estimated entropies over different time scales can be calculated, which represent the complexity of the physiological signals34. In this study, the entropy values of scale 5 (scale 5), the linear-fitted slope of scale 1–5 (slope 5), the summation of entropy values of scales 1–5 (area 1–5) and 6–20 (area 6–20) were calculated as MSE parameters to quantify the complexity of the RR dynamics exhibited in short and long time scales.
Statistical analysis
Data were expressed as mean ± standard deviation and median (25th and 75th percentiles) for normally distributed and non-normally distributed data, respectively. Comparisons of data between patients with and without pulmonary hypertension were made using the independent t-test and the Mann-Whitney U test. Differences between proportions were assessed using the chi-square test or Fisher’s exact test. Logistic regression analysis was used to validate associations between parameters and the presence of pulmonary hypertension. Significant determinants in univariate logistic regression analysis (P < 0.05) including mean RR, VLF, LF/HF ratio, DFAα1, slope 5, scale 5, area 1–5 and area 6–20 were then tested in multivariate logistic regression analysis with stepwise subset selection to identify independent factors to predict the presence of pulmonary hypertension. Then the independent HRV predictors of pulmonary hypertension in the multivariate regression model including mean RR, DFAα1 and MSE scale 5 were adjusted by clinical parameters including age, sex, BMI, HTN, DM, beta blocker, CCB and ARB or ACEI use in 5 logistic regression models. The AUC was used to assess the discriminatory power of the model to predict pulmonary hypertension. Furthermore, NRI and IDI were used to evaluate improvements in the predictive power after adding a single heart rhythm complexity parameter into a logistic regression model using only linear parameters35. The significance of NRI and IDI statistics was based on approximate normal distributions. All statistical analyses were performed using R software (http://www.r-project.org/) and SPSS version 25 for Windows (SPSS Inc., IL, USA). The significance level of the statistical analysis was set at 0.05.
References
Galie, N. et al. 2015 ESC/ERS Guidelines for the Diagnosis and Treatment of Pulmonary Hypertension. Rev Esp Cardiol (Engl Ed) 69, 177, https://doi.org/10.1016/j.rec.2016.01.002 (2016).
Hoeper, M. M. et al. Definitions and diagnosis of pulmonary hypertension. J Am Coll Cardiol 62, D42–50, https://doi.org/10.1016/j.jacc.2013.10.032 (2013).
Tuder, R. M. et al. Development and pathology of pulmonary hypertension. J Am Coll Cardiol 54, S3–9, https://doi.org/10.1016/j.jacc.2009.04.009 (2009).
Humbert, M. Pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension: pathophysiology. Eur Respir Rev 19, 59–63, https://doi.org/10.1183/09059180.00007309 (2010).
Hoeper, M. M. et al. Chronic thromboembolic pulmonary hypertension. Lancet Respir Med 2, 573–582, https://doi.org/10.1016/S2213-2600(14)70089-X (2014).
Thenappan, T., Ormiston, M. L., Ryan, J. J. & Archer, S. L. Pulmonary arterial hypertension: pathogenesis and clinical management. BMJ 360, j5492, https://doi.org/10.1136/bmj.j5492 (2018).
D’Alonzo, G. E. et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med 115, 343–349 (1991).
Delcroix, M. et al. Long-Term Outcome of Patients With Chronic Thromboembolic Pulmonary Hypertension: Results From an International Prospective Registry. Circulation 133, 859–871, https://doi.org/10.1161/CIRCULATIONAHA.115.016522 (2016).
Demerouti, E. A., Manginas, A. N., Athanassopoulos, G. D. & Karatasakis, G. T. Complications leading to sudden cardiac death in pulmonary arterial hypertension. Respir Care 58, 1246–1254, https://doi.org/10.4187/respcare.02252 (2013).
Mandel, J. & Poch, D. In the clinic. Pulmonary hypertension. Ann Intern Med 158, ITC5-1–16, https://doi.org/10.7326/0003-4819-158-9-201305070-01005 (2013).
Tsuji, H. et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 94, 2850–2855 (1996).
Wensel, R. et al. Impaired cardiac autonomic control relates to disease severity in pulmonary hypertension. Eur Respir J 34, 895–901, https://doi.org/10.1183/09031936.00145708 (2009).
Bienias, P. et al. Functional class and type of pulmonary hypertension determinate severity of cardiac autonomic dysfunction assessed by heart rate variability and turbulence. Acta Cardiol 70, 286–296, https://doi.org/10.2143/AC.70.3.3080633 (2015).
Rosas-Peralta, M. et al. Clinical implications and prognostic significance of the study on the circadian variation of heart rate variability in patients with severe pulmonary hypertension. Gac Med Mex 142, 19–28 (2006).
Peng, C. K., Havlin, S., Stanley, H. E. & Goldberger, A. L. Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series. Chaos 5, 82–87, https://doi.org/10.1063/1.166141 (1995).
Costa, M., Goldberger, A. L. & Peng, C. K. Multiscale entropy analysis of biological signals. Phys Rev E Stat Nonlin Soft Matter Phys 71, 021906, https://doi.org/10.1103/PhysRevE.71.021906 (2005).
Tang, S. C. et al. Complexity of heart rate variability predicts outcome in intensive care unit admitted patients with acute stroke. J Neurol Neurosurg Psychiatry 86, 95–100, https://doi.org/10.1136/jnnp-2014-308389 (2015).
Lin, Y. H. et al. Multi-scale symbolic entropy analysis provides prognostic prediction in patients receiving extracorporeal life support. Crit Care 18, 548, https://doi.org/10.1186/s13054-014-0548-3 (2014).
Tsai, C. H. et al. The association between heart rhythm complexity and the severity of abdominal aorta calcification in peritoneal dialysis patients. Sci Rep 8, 15627, https://doi.org/10.1038/s41598-018-33789-x (2018).
Ho, Y. L., Lin, C., Lin, Y. H. & Lo, M. T. The prognostic value of non-linear analysis of heart rate variability in patients with congestive heart failure–a pilot study of multiscale entropy. PLoS One 6, e18699, https://doi.org/10.1371/journal.pone.0018699 (2011).
Thenappan, T., Shah, S. J., Rich, S. & Gomberg-Maitland, M. A USA-based registry for pulmonary arterial hypertension: 1982–2006. Eur Respir J 30, 1103–1110, https://doi.org/10.1183/09031936.00042107 (2007).
Rajdev, A., Garan, H. & Biviano, A. Arrhythmias in pulmonary arterial hypertension. Prog Cardiovasc Dis 55, 180–186, https://doi.org/10.1016/j.pcad.2012.06.002 (2012).
Bienias, P. et al. Severity of arterial and chronic thromboembolic pulmonary hypertension is associated with impairment of heart rate turbulence. Ann Noninvasive Electrocardiol 20, 69–78, https://doi.org/10.1111/anec.12169 (2015).
Witte, C. et al. Heart Rate Variability and Arrhythmic Burden in Pulmonary Hypertension. Adv Exp Med Biol 934, 9–22, https://doi.org/10.1007/5584_2016_18 (2016).
Nootens, M. et al. Neurohormonal activation in patients with right ventricular failure from pulmonary hypertension: relation to hemodynamic variables and endothelin levels. J Am Coll Cardiol 26, 1581–1585, https://doi.org/10.1016/0735-1097(95)00399-1 (1995).
La Rovere, M. T. et al. Short-term heart rate variability strongly predicts sudden cardiac death in chronic heart failure patients. Circulation 107, 565–570 (2003).
Folino, A. F. et al. Ventricular arrhythmias and autonomic profile in patients with primary pulmonary hypertension. Lung 181, 321–328, https://doi.org/10.1007/s00408-003-1034-x (2003).
Makikallio, T. H. et al. Fractal analysis and time- and frequency-domain measures of heart rate variability as predictors of mortality in patients with heart failure. Am J Cardiol 87, 178–182 (2001).
Goncalves, H. et al. Analysis of heart rate variability in a rat model of induced pulmonary hypertension. Med Eng Phys 32, 746–752, https://doi.org/10.1016/j.medengphy.2010.04.018 (2010).
Lin, Y. H. et al. Reversible heart rhythm complexity impairment in patients with primary aldosteronism. Sci Rep 5, 11249, https://doi.org/10.1038/srep11249 (2015).
Chiu, H. C. et al. Serial heart rhythm complexity changes in patients with anterior wall ST segment elevation myocardial infarction. Sci Rep 7, 43507, https://doi.org/10.1038/srep43507 (2017).
Mitchell, C. et al. Guidelines for Performing a Comprehensive Transthoracic Echocardiographic Examination in Adults: Recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr, https://doi.org/10.1016/j.echo.2018.06.004 (2018).
Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93, 1043–1065 (1996).
Richman, J. S. & Moorman, J. R. Physiological time-series analysis using approximate entropy and sample entropy. American Journal of Physiology-Heart and Circulatory Physiology 278, H2039–H2049 (2000).
Steyerberg, E. W. et al. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology 21, 128–138, https://doi.org/10.1097/EDE.0b013e3181c30fb2 (2010).
Acknowledgements
This study was supported by grants from National Taiwan University Hospital (NTUH 104-S2696, UN103-065) and the Ministry of Science and Technology (MOST 103-2221-E-008-006-MY3). M.T.L. and C.L. gratefully acknowledge support from the Ministry of Science and Technology (Grant No. 104-3115-E-008 -001 and 104-2745-B-008 -001) and the joint foundations (Grant No. 103CGH-NCU-A1, VGHUST103-G1-3-3). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.H.L. conceived and designed the experiments. H.P.M., Y.T.L., C.S.H., M.C.H., T.Y.C., P.H.K., C.L., M.T.L. and H.H.H. analyzed the data. C.H.T. and Y.H.L. wrote the paper. C.K.P. made scientific comments on the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tsai, CH., Ma, HP., Lin, YT. et al. Heart Rhythm Complexity Impairment in Patients with Pulmonary Hypertension. Sci Rep 9, 10710 (2019). https://doi.org/10.1038/s41598-019-47144-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47144-1
This article is cited by
-
Heart rhythm complexity analysis in patients with inferior ST-elevation myocardial infarction
Scientific Reports (2023)
-
Real-time machine learning model to predict in-hospital cardiac arrest using heart rate variability in ICU
npj Digital Medicine (2023)
-
Usefulness of heart rhythm complexity in heart failure detection and diagnosis
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.