Abstract
Physical fractionation methods used in soil organic matter (SOM) research commonly include density-based procedures with heavy liquids to separate SOM pools with varying turnover rates and functions. Once separated, the heavy SOM pools are often thoroughly rinsed with water to wash off any residues of the heavy liquids. Using four soils with contrasting properties, we investigated the effects of using either sodium polytungstate (SPT) or sodium iodide (NaI), two of the most commonly used heavy liquids, on the distribution of organic carbon (C) and total nitrogen (N) in free light, intra-aggregate light, and mineral-associated heavy SOM pools isolated by a common fractionation scheme. We also determined the effects of washing the mineral-associated heavy SOM fractions on the recovery of organic C and total N after separation. Because of its smaller viscosity compared to that of NaI, SPT consistently yielded greater intra-aggregate and smaller mineral-associated soil organic C contents. We also confirm that some commercial SPT products, such as the one used here, can contaminate organo-mineral heavy pools with N during density-based fractionation procedures. We do not recommend the repeated washing of heavy fractions separated with Na-based heavy liquids, as this can mobilize SOM.
Similar content being viewed by others
Introduction
Soil organic matter (SOM) is key to terrestrial ecosystems, contributing to the support of natural vegetation and agricultural production, filtering and holding water, and storing carbon (C)1,2. The preservation of SOM is of paramount importance because of the need to maintain these ecosystem functions and services to face major global issues, such as food security, desertification, and climate change3,4. Key mechanisms controlling the preservation of SOM include occlusion within soil aggregates and sorption onto mineral surfaces, which limit the accessibility of SOM to decomposers and enzymes3,5,6,7.
The acknowledge of the importance of these preservation mechanisms has led to the increased use of physical fractionation methods to isolate SOM pools of distinct location within the soil mineral matrix5,8,9. Among the large number of physical fractionation methods available in the literature (see Poeplau et al.9 for a recent comparative description of many of them), the density-based separation scheme developed by Golchin et al.10 is one of the most commonly used and has constituted the basis for the development of other fractionation methods6,11,12,13,14,15,16,17. This scheme is intended to separate three fractions: a free light SOM fraction not physically disconnected from microorganisms and enzymes; a occluded light SOM fraction located within aggregates, which forms a physical barrier that limits O2 diffusion and the accessibility of microorganisms and enzymes; and a heavy fraction consisting of SOM intimately associated with minerals, which decreases microbial and enzymatic capacity to decompose organic substrates10. Briefly, the free SOM fraction is isolated by an initial density separation, and the intra-aggregate SOM is then separated from the mineral-associated pool by a second density separation after ultrasonic disruption of stable aggregates10.
Sodium polytungstate (SPT, Na6[H2W12O40]) and sodium iodide (NaI) are the chemical reagents most commonly used to prepare the heavy liquids for density-based fractionation of SOM6,9,10,11,12,13,14,15,16,17. After separating the light SOM fractions, the mineral-associated heavy SOM fractions are commonly washed with deionized water to eliminate the remaining SPT and NaI. Having different properties, these heavy liquids may interact differently with the soil and therefore yield different fractionation results. Similarly, the washing intended to eliminate SPT and NaI may arguably cause solubilization and loss of mineral-associated SOM because of hydrolysis reactions and increased pH18,19, thus affecting the recovery of SOM after fractionation. In spite of the potential importance of these effects, there is a lack of studies in the literature specifically and systematically addressing these issues, which may seriously compromise meaningful interpretations of SOM dynamics.
Here we (a) investigated comparatively the effects of two heavy liquids, SPT versus NaI, on the distribution of organic C and total N in free, intra-aggregate, and mineral-associated pools separated by a common density-based procedure similar to the one described by Golchin et al.10; (b) determined the effects of washing the mineral-associated heavy SOM fraction isolated using either SPT or NaI on its organic C and total N contents; and (c) evaluated the influence of soil properties on the potential effects of the heavy liquid and washing. To do this, we used four samples collected from the surface layer of four contrasting soils (SOIL 1 to 4), with the aim of covering a wide range of textures (Table 1)17,20,21. The final purpose of this study was to provide general recommendations to prevent misinterpretation of fractionation results in SOM research.
Results
The SPT used here to prepare the heavy liquid for density fractionations contained no detectable amounts of C but 0.646 g kg−1 of total N. The NaI powder had no detectable amounts of total C and N. The SPT solution had a viscosity of 1.1 mPa s, a pH of 3.3, and an electrical conductivity of 61 dS m−1 whereas the NaI liquid had a viscosity of 1.7 mPa s, a pH of 8.0, and an electrical conductivity of 154 dS m−1.
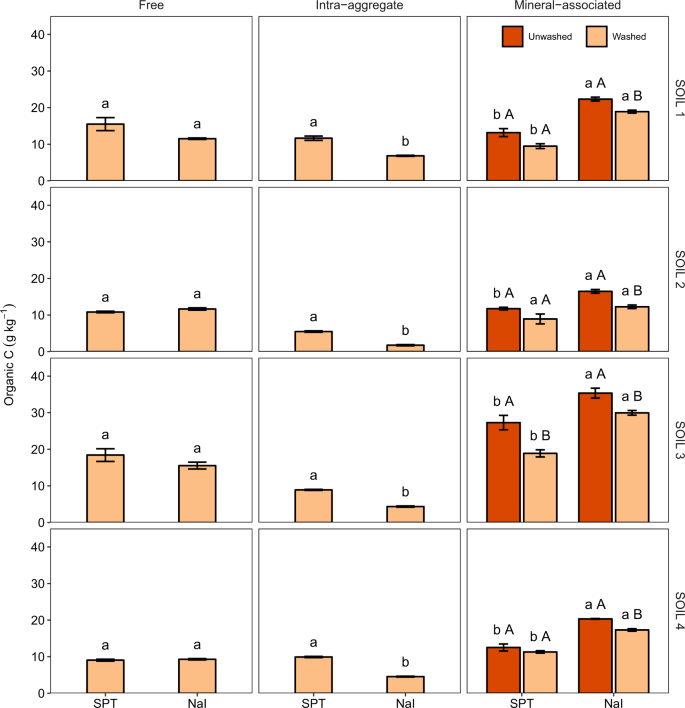
Analysis of variance tests revealed significant main and interaction effects (P < 0.001) of the heavy liquid and soil on free organic C and total N contents (Table S1). Post hoc pairwise tests, however, being more conservative, failed to detect significant differences between the effects of SPT and NaI on free organic C and total N contents for any of the soils examined (Figs 1 and 2). Nonetheless, compared to SPT, NaI tended to yield slightly smaller free organic C and total N contents for the soils with the largest total organic C and total N contents, SOIL 1 and SOIL 3 (Table 1).
Free, intra-aggregate, and mineral-associated organic C content (mean ± standard error) of soils (SOIL 1 to 4) as affected by the heavy liquid used for separation (sodium polytungstate, SPT, vs. sodium iodide, NaI) and washing. Within the same soil, fraction, and washing treatment, different lowercase letters indicate statistically significant differences at the 0.05 level. Within the same soil and heavy liquid, different uppercase letters indicate statistically significant differences at the 0.05 level.
Free, intra-aggregate, and mineral-associated N content (mean ± standard error) of soils (SOIL 1 to 4) as affected by the heavy liquid used for separation (sodium polytungstate, SPT, vs. sodium iodide, NaI) and washing. Within the same soil, fraction, and washing treatment, different lowercase letters indicate statistically significant differences at the 0.05 level. Within the same soil and heavy liquid, different uppercase letters indicate statistically significant differences at the 0.05 level.
Intra-aggregate organic C and N contents and C/N ratio were significantly affected (P < 0.001) by the heavy liquid and soil factors and their interaction (Tables S1). Compared to SPT, NaI resulted in significantly smaller intra-aggregate organic C and total N contents for the four soils examined, with the magnitude of the differences slightly depending on the soil (Figs 1 and 2).
We also found significant two-way interaction effects of the heavy liquid and soil (P < 0.001) and the washing and soil (P = 0.004) on mineral-associated organic C content, and significant three-way interaction effects of the heavy liquid, washing, and soil on mineral-associated N content (P = 0.007) (Table S1). Specifically, with respect to SPT, NaI resulted in significantly larger mineral-associated organic C content for the four soils, especially for SOIL 1 and SOIL 3, regardless of the washing treatment (Fig. 1). Independently of the heavy liquid used, washing decreased mineral-associated organic C content, especially for SOIL 3. Compared to NaI, SPT yielded larger mineral-associated N content (Fig. 2). The size and significance of these differences depended on the soil. For SPT treated soils, washing decreased mineral-associated N content, but had a negligible effect for NaI treated soils (Fig. 2).
Total organic C recovery after fractionation was significantly affected by the interaction of the washing and soil (P = 0.026) (Table S1). In particular, washing decreased organic C recovery by 0.2% for SOIL 1 to 21% for SOIL 3 (Table 2). We also found significant interaction effects of the washing and soil (P = 0.022) and the heavy liquid and washing (P < 0.001) on total N recovery (Table S1). Specifically, N recovery after SOM separation with SPT and without washing the mineral-associated fraction ranged from 106 (SOIL 1) to 143% (SOIL 4) (Table 2). With washing, N recovery after SOM separation with SPT decreased (especially for SOIL 3 and SOIL 4), ranging from 88 to 110%. Total N recovery with NaI was markedly smaller and not significantly affected by the washing treatment (Table 2).
Discussion
Our research highlights substantial differences between the heavy liquids in SOM fractionation results. These differences may be mainly related to the significantly different physicochemical and chemical properties of the heavy liquids, while only secondarily depend on the specific properties of the soil under study. Despite the observed interaction effects of the heavy liquid and soil, our results do not point to clear relationships between such interactions and soil texture or any other specific soil properties. The effects of the heavy liquid tend to dominate and be consistent across soils.
The no significant differences found between the effects of SPT and NaI on free organic C and total N contents for any of the soils examined is consistent with previous observations13. The greater intra-aggregate and smaller mineral-associated organic C contents obtained after fractionation with SPT, compared to NaI, which is in general agreement with previous works14,15, may be attributed to the smaller viscosity of the SPT solution. Smaller viscosities favor the propagation of the ultrasonic pulses and the cavitation process22. This may increase the effectiveness of the ultrasonic disruption treatment to break up aggregates and the release of intra-aggregate SOM, which in turn decreases the amount of SOM recovered in the mineral-associated pool.
Because of the lower viscosity and higher efficacy of sonication, SPT also results in greater intra-aggregate N compared to NaI, but not in smaller mineral-associated N. This is because, unlike NaI, the SPT powder used to prepared the heavy solution is contaminated with N as supplied. Some of the N in SPT inevitably ends up in the heavy fraction remaining after the last density separation, thus adding up to the mineral-associated N pool. The concentration of N in the SPT used here is within the range detected in commercially available standard SPT for the preparation of heavy liquid in a previous study23. Conversely, low N content SPT, which can be purchased from some manufacturers, has been shown not to adversely contaminate soils after density fractionation23.
To remove SPT and NaI residues, the organo-mineral heavy fractions remaining after density-based separations are often thoroughly rinsed with water. Our results show that this procedure may result in significant losses of organic C from the mineral-associated SOM fraction, thus hampering the interpretation of fractionation results. These C losses, which tend to become more evident with increasing the number of washes and soil organic C content (Supplementary Fig. S1), can be attributed to soil dispersion and organic matter solubilization induced by the high amounts of Na added with the SPT and NaI solutions. Initially, the high electrolyte concentration in the heavy solutions helps maintain soil aggregated during density fractionation19. With washing, this coagulation effect of a high electrolyte concentration disappears (Fig. 3a), and soil particles disperse. Washing Na-saturated soils causes hydrolysis reactions and increased pH (Fig. 3b). This rise in pH may also cause the disruption of bonds between organic matter and minerals and the conversion of acidic organic components to their soluble salt forms18,19. This may help explain why total organic C recovery from SOIL 1, which has the lowest pH, decreases with washing to a lesser extent than does the total organic C recovery from the other soils examined.
As a whole, our results reveal substantial effects of the heavy liquid used for density fractionation on the distribution of organic C and total N in intra-aggregate and mineral associated pools. These effects are largely independent of the soil and have several implications that need to be accounted for when applying SOM fractionation methods and interpreting SOM fractionation data. First, to facilitate the comparison of results across studies, ultrasonic energy has to be properly calibrated not only for the soil under study but also for the heavy liquid and applied to an extent to completely disrupt aggregates. Our results also highlight the need of determining C and N levels in the heavy liquids used for density-based SOM separations, particularly N in SPT, to prevent or account for potential contamination effects. Finally, we do not recommend repeated washing of the organo-mineral heavy soil fractions separated with Na-based heavy liquids, as this can lead to mobilization of SOM.
Consistently with methods commonly used in previous studies, here we used heavy liquids at a density of 1.85 g mL−1 11,24,25,26 and soil samples passed through a 2-mm sieve9,10. Future research should test other densities, and consider the use of soil samples sieved through larger mesh sizes, to further improve our understanding about the impact of heavy liquids and washing on the distribution of organic C and total N in SOM fractions. Here we only focused on the effects of washing the mineral associated heavy SOM fraction. The effects of washing the free and intra-aggregate light SOM fractions also need to be investigated in future studies.
Methods
Soil samples
We collected four samples from the surface layer of four contrasting soils (SOIL 1 to 4), with the aim of covering a wide range of textures17,20,21. The samples were air dried, gently crushed, and sieved to 2 mm.
Soil organic matter fractionation
The prepared soil samples (dried and 2-mm sieved) were subjected to the physical fractionation scheme developed by Golchin et al.10 to isolate free, intra-aggregate, and mineral-associated SOM. The fractionation was conducted using a solution of either SPT or NaI as heavy liquid and either not washing or washing the isolated mineral-associated SOM fraction. Specifically, 80 mL of either SPT (purum p.a., for the preparation of heavy liquid, for sink-float analysis, ≥85% WO3 basis, Fluka, Sigma-Aldrich, St. Luois, MO) or NaI (ACS reagent, ≥99.5%, Sigma-Aldrich, St. Luois, MO) at a density of 1.85 g mL−1 was added to 10 g of soil in a 100-mL centrifuge tube. The centrifuge tube was rotated at 1 revolution s−1 for 30 s in an overhead shaker to allow free SOM outside aggregates to float. After centrifugation at 2500 g for 30 min, the floating light fraction (free SOM) was separated from the heavy fraction by suction and filtration through a glass fiber filter (GF/A, Whatman, UK) and washed thoroughly with deionized water. The heavy fraction in the centrifuge tube was resuspended and dispersed in the SPT or NaI solution by sonication at an energy input of 1500 J g−1. The floating light fraction (intra-aggregate SOM) was separated from the heavy fraction (mineral-associated SOM) by centrifugation at 2500 g for 60 min, suction, and filtration through a glass fiber filter, and washed thoroughly with deionized water. The fractionation procedure was repeated six times for each soil and heavy liquid. Three of the six replicates of the isolated mineral-associated SOM were not washed, and the other three replicates were thoroughly washed three times by adding 80 mL of deionized water, shaking for 10 min, and centrifuging at 2500 g for 30 min.
Chemical and physicochemical analysis
The soils and SOM fractions were analyzed for organic C and total N content by dry combustion using a Thermo Flash 2000 NC Soil Analyzer. Prior to organic C analysis, the soils and mineral-associated SOM fractions were subjected to acid fumigation to remove carbonates27.
The SPT and NaI reagents (powders) used to prepare the solutions for density fractionations were analyzed for total C and N by dry combustion, as described above for the soils and SOM fractions. The prepared heavy liquids were analyzed for viscosity, pH, and electrical conductivity at 20 °C using a capillary viscometer, a pH meter, and a conductivity meter, respectively.
Data analysis
We used two-way analysis of variance (ANOVA) tests to evaluate the main and interaction effects of the heavy liquid and soil on free and intra-aggregate organic C and total N, and three-way ANOVA tests to evaluate the effects of the heavy liquid, washing, and soil on mineral-associated organic C and total N and on the recovery of each element after fractionation. When assumptions of normality and homoscedasticity were not met, we used nonparametric ANOVA on aligned rank transformed data. Post hoc pairwise comparisons within each soil were conducted using t or Wilcoxon rank-sum tests at the 0.05 level. All data analyses were performed using R statistical software version 3.5.128.
Data Availability
The data and R scripts related to this study are available from the corresponding author upon request.
References
Lal, R. Challenges and opportunities in soil organic matter research. Eur. J. Soil Sci. 60, 158–169 (2009).
Paul, E. A. The nature and dynamics of soil organic matter: Plant inputs, microbial transformations, and organic matter stabilization. Soil Biol. Biochem. 98, 109–126 (2016).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Plaza, C. et al. Soil resources and element stocks in drylands to face global issues. Sci. Rep. 8, 13788 (2018).
Lützow, M. V. et al. Stabilization of organic matter in temperate soils: mechanisms and their relevance under different soil conditions – a review. Eur. J. Soil Sci. 57, 426–445 (2006).
Plaza, C., Courtier-Murias, D., Fernández, J. M., Polo, A. & Simpson, A. J. Physical, chemical, and biochemical mechanisms of soil organic matter stabilization under conservation tillage systems: A central role for microbes and microbial by-products in C sequestration. Soil Biol. Biochem. 57, 124–134 (2013).
Lehmann, J. & Kleber, M. The contentious nature of soil organic matter. Nature 528, 60–68 (2015).
Crow, S. E., Swanston, C. W., Lajtha, K., Brooks, J. R. & Keirstead, H. Density fractionation of forest soils: methodological questions and interpretation of incubation results and turnover time in an ecosystem context. Biogeochemistry 85, 69–90 (2007).
Poeplau, C. et al. Isolating organic carbon fractions with varying turnover rates in temperate agricultural soils – A comprehensive method comparison. Soil Biol. Biochem. 125, 10–26 (2018).
Golchin, A., Oades, J. M., Skjemstad, J. O. & Clarke, P. Soil structure and carbon cycling. Aust. J. Soil Res. 32, 1043–1068 (1994).
Sohi, S. P. et al. A procedure for isolating soil organic matter fractions suitable for modeling. Soil Sci. Soc. Am. J. 65, 1121–1128 (2001).
Plaza, C., Fernández, J. M., Pereira, E. I. P. & Polo, A. A comprehensive method for fractionating soil organic matter not protected and protected from decomposition by physical and chemical mechanisms. Clean - Soil Air Water 40, 134–139 (2012).
Sequeira, C. H., Alley, M. M. & Jones, B. P. Evaluation of potentially labile soil organic carbon and nitrogen fractionation procedures. Soil Biol. Biochem. 43, 438–444 (2011).
Conceição, P. C. et al. Eficiência de soluções densas no fracionamento físico da matéria orgânica do solo. Rev. Bras. Cienc. do Solo 39, 490–497 (2015).
Guareschi, R. F., Pereira, M. G. & Perin, A. Densimetric fractionation of organic matter in an agricultural chronosequence in no-till areas in the Cerrado region, Brazil. Semin. Agrar. 37, 595–610 (2016).
Courtier-Murias, D. et al. Unraveling the long-term stabilization mechanisms of organic materials in soils by physical fractionation and NMR spectroscopy. Agric. Ecosyst. Environ. 171, 9–18 (2013).
Giannetta, B., Plaza, C., Vischetti, C., Cotrufo, M. F. & Zaccone, C. Distribution and thermal stability of physically and chemically protected organic matter fractions in soils across different ecosystems. Biol. Fertil. Soils 54, 671–681 (2018).
Stevenson, F. J. Humus Chemistry: Genesis, Composition, Reactions. (John Wiley & Sons, 1994).
Sparks, D. L. Environmental Soil Chemistry. (Academic Press, 2003).
Jiménez-González, M. A., Álvarez, A. M., Carral, P., González-Vila, F. J. & Almendros, G. The diversity of methoxyphenols released by pyrolysis-gas chromatography as predictor of soil carbon storage. J. Chromatogr. A 1508, 130–137 (2017).
Jiménez-González, M. A., Álvarez, A. M., Hernández, Z. & Almendros, G. Soil carbon storage predicted from the diversity of pyrolytic alkanes. Biol. Fertil. Soils 54, 617–629 (2018).
Santos, H. M., Lodeiro, C. & Capello-Martínez, J. L. The power of ultrasound. in Ultrasound in Chemistry: Analytical Applications (ed. Capello-Martínez, J. L.) (Wiley-Vch, Verlag GmbH & Co. KGaA, 2009).
Kramer, M. G., Lajtha, K., Thomas, G. & Sollins, P. Contamination effects on soil density fractions from high N or C content sodium polytungstate. Biogeochemistry 92, 177–181 (2009).
Six, J., Elliott, E. T., Paustian, K. & Doran, J. W. Aggregation and Soil Organic Matter Accumulation in Cultivated and Native Grassland Soils. Soil Sci. Soc. Am. J. 62, 1367 (1998).
Six, J., Elliott, E. & Paustian, K. Aggregate and soil organic matter dynamics under conventional and no-tillage systems. Soil Sci. Soc. Am. J. 63, 1350–1358 (1999).
Six, J., Paustian, K., Elliott, E. & Combrink, C. Soil Structure and OM: I. Aggregate-Size Classes and Aggregate-Associated Carbon. Soil Sci. Soc. Am. J. 64, 681–689 (2000).
Harris, D., Horwáth, W. R. & van Kessel, C. Acid fumigation of soils to remove carbonates prior to total organic carbon or carbon-13 isotopic analysis. Soil Sci. Soc. Am. J. 65, 1853–1856 (2001).
R Core Team. R: A language and environment for statistical computing. (2018).
Sparks, D. L.et al. (eds) Methods of Soil Analysis Part 3—Chemical Methods, Soil Science Society of America Book Series 5.3. (Soil Science Society of America, American Society of Agronomy, 1996).
Acknowledgements
This project has received funding from the Spanish Ministry of Economy and Competitiveness (AGL2016-75762-R). The authors are grateful to M.A. Jiménez-González, for providing one of the soil samples used in the study.
Author information
Authors and Affiliations
Contributions
C.P. and C.Z. conceived the study and designed the research. B.G., I.B. and C.V performed lab and field research. C.P., C.Z. and B.G. analyzed the data. All the authors discussed the results. C.P. and C.Z. wrote the manuscript, with contributions from all authors.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Plaza, C., Giannetta, B., Benavente, I. et al. Density-based fractionation of soil organic matter: effects of heavy liquid and heavy fraction washing. Sci Rep 9, 10146 (2019). https://doi.org/10.1038/s41598-019-46577-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-46577-y
This article is cited by
-
Stabilization mechanisms of organic matter in Andosols under long-term fertilization as revealed from structural, molecular, and stable isotopic signatures
Journal of Soils and Sediments (2024)
-
Ecosystem productivity has a stronger influence than soil age on surface soil carbon storage across global biomes
Communications Earth & Environment (2022)
-
Exogenous fulvic acid enhances stability of mineral-associated soil organic matter better than manure
Environmental Science and Pollution Research (2022)
-
The role of clay content and mineral surface area for soil organic carbon storage in an arable toposequence
Biogeochemistry (2021)
-
The C/N ratio and phenolic groups of exogenous dissolved organic matter together as an indicator for evaluating the stability of mineral-organic associations in red soil
Journal of Soils and Sediments (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.