Abstract
Ammonia oxidation is the first and rate-limiting step of nitrification, driven by ammonia-oxidizing bacteria (AOB) and ammonia-oxidizing archaea (AOA). Straw and straw biochar retention are the popular ways to utilize the agricultural by-products in China, but their long-term effects on AOB and AOA still remain poorly understood. Based on a 7-year plot experiment, which had 4 fertilization regimes: no fertilizer (CK), regular fertilization (RT), straw retention (SR) and straw biochar retention (SB), the abundance and the composition of AOB and AOA was investigated before both the harvest of rice and wheat season by quantitative PCR and 454 high-throughput pyrosequencing, respectively. (1) Compared to RT, straw and straw biochar increased AOB abundance and diversity significantly only in wheat season (P < 0.05), and they both ranked as SB > SR > RT. Among fertilized treatments, a significant difference between SR and RT was found in AOB community composition of the winter season (R value = 0.58, P value = 0.02); (2) In contrast, AOA was almost not responsive to organic addition, except the significant enhancement of abundance by biochar in wheat season; (3) After straw and straw biochar addition, soil potential nitrification rates (PNR) was positive correlated to AOB abundance in both rice and wheat season (P < 0.01), not to AOA abundance (P = 0.211 and 0.068, respectively). This study provides scientific support for the potential of straw utilization to improve nitrification in rice-wheat rotation system with respect to soil ammonia oxidation microorganism.
Similar content being viewed by others
Introduction
The crucial and rate-limiting step of nitrification, ammonia oxidation, converting ammonia to nitrite, is controlled by both ammonia-oxidizing bacteria (AOB) and ammonia oxidizing archaea (AOA)1,2. AOA abundance (amoA gene copy numbers) usually outnumbers AOB abundance in terrestrial systems, especially in agricultural ecosystems3,4. However, the contributions of AOB and AOA in nitrification vary depending on environmental conditions5,6. Therefore, studies of AOB and AOA community abundance and diversity, and their links with the ecological function are of great importance.
Soil pH was commonly considered as a key driving environmental factor influencing AOB and AOA community. AOA appeared to be more active in acidic environment, while AOB contributed more to nitrification in alkaline and neutral environment5,7. Soil nutrient level was another direct factor to affect AOB and AOA community. AOB seemed to be predominant under high ammonia concentration but had little response to organic matter input8,9, while AOA may play an important role under limited available nutrient levels10. In addition to soil properties, both short-terms and long-terms chemical fertilization can increase AOB abundance and alter its community composition, but almost have no impact on AOA11,12. Organic inputs such as organic fertilizer, straw, biochar affect AOB community significantly, but maybe because of the mixtrophic character of AOA, have no clear effect pattern with AOA community9,13,14. Besides, soil types and texture, vegetation type, temperature and precipitation can affect the composition of AOB and AOA community15,16. Although extensive researches had explored how the multiple environmental factors affect AOB and AOA communities, the effects of organic inputs on AOB and AOA community diversity and their relative contributions to nitrification are still not clear.
Crop straw is the largest renewable resource on earth. However, it is often burned after harvest, resulting in loss of nutrients and environmental pollution. With current advocate for ecological agriculture in China, straw and straw biochar returning to fields have become important modes of reutilization of agricultural by-products. Straw and straw biochar could impact the soil microbial community directly by their own and indirectly through changing soil physico-chemical properties. Hai17 reported that fertilizer combined with straw could increase AOB number, and AOA abundance was higher than AOB abundance, but independent of fertilizer regimes. However, the incubation experiment of Wua18 showed that adding wheat straw did not change the AOB and AOA abundance significantly. Similarly, the effect of biochar addition on ammonia oxidation microbial community was also not consistent. The results of Bi19 in vegetable soil demonstrated N fertilizer+ biochar increased abundance of AOB rather than which of AOA. He20 found that the biochar raised gene copies of AOB and AOA significantly in oxisols but had little effect in cambosols. Therefore, more studies should be employed for the establishment of more efficient use of straw to conserve the abundance and diversity of AOB and AOA community.
Rice-wheat rotation was applied widely in eastern China, and it is a representative wet-dry alternative system, resulting in an oxic/anoxic soil environment, which can also impact AOB and AOA community abundance and diversity significantly21,22. However, most of the researches determining the composition of ammonia oxidation microbial community were based on sampling in one season. Sampling in both rice and wheat seasons may give us a better understanding of the changes in the composition of ammonia oxidation microbial community in the whole rotation systems.
In this study, soil samples were collected before the harvest of rice and wheat season. Based on long-term plot experiment of straw and straw biochar, 454 high-throughput pyrosequencing and real-time quantitative PCR (qPCR) approaches were employed to investigate the abundances and community composition of AOB and AOA in the rice-wheat rotation ecosystems. The aims of this research were under the effects of straw and straw biochar (1) to identify the abundance and community composition shifts of AOA and AOB in rice and wheat season; (2) to detect the relative contribution of AOA and AOB to nitrification seasonally; (3) to provide scientific support for the establishment of a straw utilization technique to efficiently conserve the diversities of AOB and AOA communities.
Materials and Methods
Site description and experimental design
The six years experiment is located at Shanghai engineering research center of low-carbon agriculture, Shanghai, China (31°53′N, 121°23′E). The annual average temperature of this area is 15.7 degrees and the annual precipitation is 1162.0 mm; there are 225 d of frost free period in a year. The soil type is sandy loam.
The experiment started from rice season of 2011, and ended in wheat season of 2017. There were 4 treatments: regular treatment (RT), straw retention treatment (SR), straw biochar treatment (SB) and no fertilizer control (CK). Each treatment had 3 replicates, and all the replicates were arranged in random block design. Every replicate field was 60 m2, and separated by cement ridge and Impermeable membrane. All 4 treatments were treated with the same amount of nitrogen (N), phosphorus (P) and potassium (K) annually (225, 112.5 and 255 kg·ha−1 for rice season; 180, 90 and 204 kg·ha−1 for wheat season, respectively). The amount of applied N, P, K input was set according to the average application of rice-wheat rotation systems in Shanghai. The fertilizers used in the experiment were urea, Ca(H2PO4)2 and K2SO4. In SR treatment, the half amount of straw were all directly returned to the field; in SB treatment, the half amount of straw (rice straw applied in the wheat season and wheat straw applied in the rice season) was processed into biochar and returned to the field. Preparation of biochar was anaerobically performed at 500 °C for 6 h in a vertical charcoal furnace (ECO-5000, Wuneng Environment Co., Ltd., Zhejiang, China). N, P, K of straw and straw biochar was included in the total amount of applied fertilizer. The average N, P, K content of rice and wheat straw were 0.33 ± 0.02%, 0.19 ± 0.01%, 1.35 ± 0.08% and 0.38 ± 0.03%, 0.24 ± 0.01%, 1.84 ± 0.10%, respectively. The average N, P, K content of straw biochar in rice and wheat season were 0.84 ± 0.05%, 0.52 ± 0.04%, 5.30 ± 0.32% and 1.71 ± 0.09%, 0.64 ± 0.04%, 4.72 ± 0.21%, respectively.
Soil sampling and analysis
Soil samples were collected in Nov. 4, 2016 and May 15, 2017, before the harvest of rice and wheat, respectively. Soil samples (0–20 cm) taken randomly from 5 replicate points of each block and mixed into one, resulting in 24 samples totally. The soil samples were stored in polyethylene bags and taken back to lab immediately. One portion of the samples was freeze-dried for soil properties determination, and the others were stored at −20 °C for DNA extraction and microbial analysis.
Soil total N was determined by Kjeldahl method; exchangeable NH4+-N and NO3−-N were extracted with 2M KCl (soil/water ratio was 1:5) and measured by SmartChem Intelligent automatic chemical analyzer (Alliance Instruments, France); soil pH was determined by potentiometry (soil -water ratio was 2.5:1); soil organic C was determined by dichromate oxidation.
Potential nitrification rate (PNR) was determined according to Kurola et al.23. Five g of soil sample were put into a 50 mL tube, mixed with 20 mL of phosphate buffered saline solution with 1 mM (NH4)2SO4. The tubes were shaken at the speed of 180 rpm in the dark under 25 °C for 24 h, 10 mg·L−1of potassium chlorate was added to inhibit nitrite oxidation. Five mL of 2M KCl were added to extract NO2-N after shaking (170 rpm for 24 h). The NO2-N concentrations in the supernatant were determined by Griess reagent colorimetric method (measured absorption at 545 nm). PNR was expressed by the unit as μg (NO2-N)·g−1·h−1 dry soil.
DNA extraction and quantitative PCR analysis
Soil DNA was extracted using MoBio PowerSoil® DNA Isolation Kit (12888) according to the manufacturer’s instruction. The extracted DNA concentration and quality were determined by spectrophotometer (RS232G, Eppendorf, Germany).
The AOB and AOA gene copy numbers were determined using Applied Biosystems ABI StepOnePlus Real-Time PCR instrument. The primer pairs for AOB and AOA were amoA1F (5′-GGGGTTTCTACTGGTGGT-3′)/amoA2R (5′-CCCCTCKGSAAAGCCTTCTTC-3′)24 and ArchamoAF (5′-STAATGGTCTGGCTTAGACG-3′)/Arch-amoAR (5′-GCGGCCATCCATCTGTATGT-3′)25, respectively. The qPCR amplifications were carried out in a total volume of 20 μL, containing 10 μl of SYBR real-time PCR premix (Takara Biotechnology, Dalian, China), 4 μM of respective primer and 1 μL (approximately 20 ng) DNA template. Amplifications were conducted with the following thermal conditions: 6 min initial denaturation at 95 °C, followed by 45 cycles of 95 °C for 15 s, and 60 °C for 45 s. The qPCR was run in duplicates, and the specificity and efficiency of amplification was confirmed by melting curve analysis after every run. Standard curves were generated by a serial dilution of purified plasmid DNA harboring amoA genes (Bacteria and Archaea).
PCR amplification and Pyrosequencing
AOB and AOA amoA genes were amplified with the primer pairs of amoA-1F/amoA-2R and Arch-amoAF/Arch-amoAR, respectively. The PCR amplifications were carried out in a total volume of 25 μL, containing 40 ng of DNA template, 1.0 μL each primer (10 μM), 0.25 μL Q5 high-fidelity DNA polymerase, 5.0 μL 5* High GC Buffer, 5.0 μL 5*Reaction Buffer, 0.5 μL dNTP (10 mM). Amplifications were conducted with the following thermal conditions: 30 s initial denaturation at 98 °C, followed by 27 cycles of denaturation at 98 °C for 15 s, annealing at 58 °C/55 °C (AOB/AOA) for 30 s, extension at 72 °C for 30 or 45 s (for amoA genes of AOB and AOA, respectively), and a final extension at 72 °C for 7 min. The quantity and quality of the PCR products was detected using NanoDrop ND-1000 UV-Vis spectrophotometer. After successful amplifications, the PCR products were pooled in equimolar concentrations of 10 ng·μl−1 for pyrosequencing.
The pyrosequencing was performed on a Roche massively parallel 454 GS-FLX Titanium sequencer (Roche 454 Life Sciences, Branford, CT, USA) at Shanghai Personal Biotechnology Co., Ltd. (Shanghai, China). Raw sequences were going through quality control and assigned on unique 10-bp barcodes using QIIME software (version 1.7.0)26. Then the remaining sequences were clustered into operational taxonomic units (OTUs) based on the level of 97% sequence similarity.
Statistical analysis
One-way analyses of variance (ANOVA) with Turkey’s HSD tests were performed to discriminate significant differences between treatments using SPSS 19.0. The AOB and AOA richness and diversity indices (Chao1, Abundance-based and Coverage Estimator ACE, Shannon and Simpson) were estimated using Mothur (version v.1.30.1). Venn diagrams were generated to show the shared and unique genera among 4 treatments by Venn Diagram package of R software. Principal components analysis (PCA) based on the OTU composition was carried out to investigate the differences in the composition of microbial community using R software (Version 3.0.2). Analysis of similarity (ANOSIM) was applied to examine the effects’ significance of fertilization regimes on the composition of AOA and AOB using QIIME. Pearson correlation was performed to test the associations between the amoA gene abundances (AOA, AOB) and the environmental variables.
Results
Soil properties characteristics and crop yield
The soil properties were affected significantly by fertilization regimes both in rice and wheat seasons (Table 1). In the harvest stage of rice season, compared to RT, SR and SB increased organic C (SOC), total N (TN) and exchangeable NH4+-N, and deceased pH and bulk density of soil significantly. There was no significant difference in NO3−-N among 4 soils. SOC, pH and exchangeable NH4+-N under SB were higher than under SR (P < 0.05). SR had the lowest C/N (8.5) among 4 treatments. In wheat season, there was no significant difference in all measured properties between SR and SB. Compared to RT and CK, SR and SB enhanced SOC, TN, C/N, and reduced pH, soil bulk density significantly. There was no significant difference in exchangeable NH4+-N and NO3−-N among RT, SR and SB. From rice season to wheat season, NO3−-N rocketed, while exchangeable NH4+-N declined significantly in all treatments. Fertilization increased the crop yield of rice and wheat significantly (Fig. 1). There was no significant difference among 3 fertilization treatments in rice season. However, the wheat yield of SR and SB were higher than RT by 7.3% and 8.1%, respectively.
Abundance of AOA and AOB
Both AOA and AOB amoA gene copy numbers were higher in fertilized soil than CK in rice and wheat seasons (P < 0.01) (Fig. 2). AOA gene copy numbers were not significantly different among RT, SR and SB in the rice season, but they were higher in SB than RT and SR in the wheat season (P < 0.01). There was also no significant difference of AOB gene copy numbers among RT, SR and SB in the rice season, but there were significant differences among RT, SR and SB (P < 0.01) in the wheat season, and they ranked as SB > SR > RT. AOA gene copy numbers in the whole year ranged from 3.93 × 106 gene copy•g−1 dry soil to 1.84 × 107 gene copy•g−1 dry soil, and AOB gene copy numbers ranged from 1.81 × 105 gene copy•g−1 dry soil to 2.08 × 106 gene copy•g−1 dry soil. AOA gene copy numbers were always higher than AOB gene copy numbers. AOA/AOB ratio varied from 8.1 to 21.7 and 8.8 to 27.3 in rice and wheat season, respectively. SB and CK had the lowest and highest AOA/AOB ratio in both rice and wheat seasons. AOA/AOB ratio under SR was higher than under RT in the rice season but there was no significant difference in the wheat season (P < 0.01).
Alpha-diversity of AOA and AOB
Richness and diversity of AOA and AOB under different fertilization regimes were reported in Table 2. Chao1 and abundance based coverage estimation (ACE) were employed as richness indices. Simpson and Shannon indices, estimated by richness and species abundances, were used as diversity indices. For all the indices estimated, CK had the lowest richness and diversity for AOA and AOB in the whole year. The three different fertilization regimes had not affected on α-diversity of AOA community both in rice and wheat season. According to richness and diversity indices of AOB, there were significant differences among SB, SR and RT treatments in the wheat season, and these treatments ranked as SB > SR > RT (P < 0.05); while there was no significant differences in the rice season.
Community composition of AOA and AOB
Shared and unique genera among 4 fertilization regimes were determined via Venn diagram (Fig. 3). A total of 1314 AOA-related OTUs and 1530 AOB-related OTUs under 4 treatments were observed in the rice season, respectively. The numbers of shared AOA and AOB related OTUs were 316 (24.0% of total) and 619 (40.5% of total), respectively. A total of 826 AOA-related OTUs and 724 AOB-related OTUs were observed in the wheat season, respectively. The numbers of shared AOA and AOB related OTUs were 196 (23.7% of total) and 111 (15.3% of total), respectively.
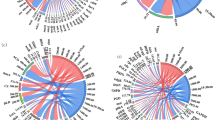
PCA analysis showed that there were no group effects on the composition of AOA communities among 4 treatments in both rice and wheat season (Fig. 4). However, the composition of AOB communities under CK and other 3 fertilization treatments were separated by the first component (PC1) in rice and wheat season, which explained 98.23% and 98.08% of variation, respectively. In the wheat season, the AOB community composition under SR and RT were separated by the second component (PC2), explaining 1.92% of variation. It was also confirmed by ANOSIM that the R values of AOB composition (R value = 0.52 and 0.67, P value = 0.02 and 0.01 in rice and wheat season, respectively) were the higher than which of AOA composition. The results showed that fertilization regimes exerted higher influence on the composition of AOB community than on that of AOA. Among the treatments of RT, SR and SB, there was only one significant difference between SR and RT in AOB community composition of winter season (R value = 0.58, P value = 0.02).
Correlation between soil properties and abundances of AOA and AOB
Pearson’s correlation was applied to determine the relationships between soil properties and abundances of AOA and AOB (Table 3). In the rice season, the AOA abundance was significantly and negatively correlated to pH (P < 0.01) and C/N (P < 0.05). The AOB abundance was significantly and negatively correlated to pH (P < 0.01) and positively correlated to exchangeable NH4+-N (P < 0.01). In the wheat season, the AOA abundance was significantly and negatively correlated to pH (P < 0.01), while positively correlated to SOC (P < 0.01). The AOB abundance was significantly and negatively correlated to pH (P < 0.01) and also positively correlated to SOC (P < 0.01).
Soil potential nitrification rate (PNR) and relative contributions of AOA and AOB to nitrification
Soil PNR under 4 treatments ranged from 1.30–2.97 and 1.10–4.27 μg N·g−1·h−1 dry soil in rice and wheat season, respectively (Fig. 5). PNR of treatments with fertilization were greater than that of CK in the whole year. There was no significant difference of PNR among RT, SR and SB in the rice season. On the contrary, PNR in the wheat season among RT, SR and SB were significantly different. The sequence was SB > SR > RT (P < 0.05). PNR was not significantly correlated with AOA abundance in rice (R2 = 0.068, P = 0.209) and wheat season (R2 = 0.211, P = 0.075) (Fig. 6). However, PNR was positively correlated with AOB abundances both in rice (R2 = 0.567, P = 0.003) and wheat season (R2 = 0.735, P = 0.002).
Discussion
AOB and AOA abundance and community diversity response to straw and straw biochar
In both acid and alkaline soil, AOA outnumbered AOB in most cases irrespective of crop types, fertilization and irrigation managements, and AOA/AOB ratio varied from 1.2 to 2383 in the bulk and rhizosphere soil21,27,28,29. Only a few exceptions found that AOB abundance was higher than AOA (AOA/AOB ratio ranged from 0.39 to 12.3)30,31,32. The result of present study confirmed that amoA gene numbers of AOA were greater than those of AOB in the rice-wheat rotation system under different fertilization regimes.
Our study showed that compared to RT and SR, SB stimulated the growth of AOA and AOB in the wheat season, but there was no significant difference among the fertilization treatments in the rice season. Song13 found that the addition of biochar derived from cotton stalk increased the abundance of AOA and AOB in coastal alkaline soil and the same increase by wood biochar was reported by Prommer33. Bi19 reported that the combination of rice straw biochar with N fertilizer could increase AOB abundance significantly in greenhouse vegetable soil, but had no effect on AOA abundance. He20 suggested that biochar increased AOA and AOB abundances in oxisols (acid soil), but not in cambosols (alkaline soil). The inconsistency indicated the effects of biochar on the abundance of ammonia oxidizers may be influenced by soil pH (acid soil or alkaline soil), crop type (grain fields or vegetable fields), oxygen level (flooded soil or dry soil), etc, and it is difficult to draw a general conclusion.
Compared to RT, straw + N fertilizer increased AOB abundance significantly in the wheat season, while had no influences on AOB abundance in the rice season and AOA abundance in the whole year. Similar results were reported by Hai17 and Lin33. However, Wessén34 observed that there was no change of AOA and AOB amoA gene numbers under the treatment of maize straw + N fertilizer in an acido-neutral soil condition (pH ranged from 5.6–6.6). AOB was widely considered to be favorable in alkaline and neutral soil35,36. The reason for AOB abundance increase in our study may be because of the higher soil pH (pH ranged from 8.0–8.6 in our study, and 5.6–6.6 in reference), which could generate more NH3 than NH4+ as the substrate, and was more beneficial for the growing of AOB37.
The abundances of ammonia oxidizers were also highly dependent on soil properties, which were strongly influenced by fertilization regimes. Soil pH was considered as a vital environmental factor to influence ammonia-oxidizing bacterial and archaeal community. Extensive studies demonstrated that soil pH (ranged from 3.5–8.7, including acidic, acido-neutral, and alkalinophilic conditions) was strongly correlated with AOB and AOA abundances37,38,39. Our results were similar to these previous studies that Pearson’s correlation analysis showed that AOB and AOA abundances increased significantly with the decrease of soil pH (P < 0.05) in both rice and wheat season (Table 3). Compared to RT, SR and SB decreased soil pH significantly (Table 1). Therefore, the additions of straw and straw biochar had the potential to enhance AOB and AOA abundances. In our study, the growth AOB was highly stimulated when pH decreased in wheat season, while the response of AOA abundance was not significant. The reason for little increase of AOA abundance (Fig. 2) may also attribute to soil pH. AOB prefer to grow in neutral and alkaline soil, but the growth of AOA was considered more suitable in acid environment, and the response of AOA to environmental factors was weak in alkaline soil5,27,28.
Exchangeable NH4+-N was a direct substrate resource for ammonia oxidizers, and the increase of exchangeable NH4+-N concentration often led to a significant impact on AOB abundance31,40. Correlation analysis in our study had similar results that NH4+-N concentration was positive to AOB abundance in rice (P < 0.01) and wheat season (P < 0.05), while there was no clear correlation to the AOA abundance. After 7 years cultivation, the addition of straw and straw biochar increased exchangeable NH4+-N concentration in the rice season, showing the potential to stimulate AOB growing.
SOC was another environmental factor that influenced ammonia oxidizers abundances significantly. AOA and AOB abundances were both positive to SOC in the wheat season (P < 0.01). These findings were agreed with several previous studies who reported AOA and AOB abundances increased with SOC in dry land, such as cotton, maize31,41. Besides, the AOA abundance was negatively correlated with C/N in the rice season (P < 0.05). Extensive researches suggested that AOA could withstand some extreme environment, like low oxygen level as in rice season, and also had the ability to be alternative between chemoautotrophy and heterotrophic growth according to different carbon supplying conditions42,43,44. Therefore, the ability to metabolize other energy sources like rice root exudates under anaerobic condition may lead to the negative correlation between AOA abundance and C/N.
According to the PCA analysis (Fig. 4), 3 fertilized treatments were distinctly separated from unfertilized CK in rice and wheat season, suggesting long-term N input influenced AOB community in the rice-wheat rotation system. This was agreed to some previous studies in both dry (cotton field) and wet agricultural soil (paddy field), found distinct differences in AOB community composition between fertilized and unfertilized soil21,31,45. Among three fertilized treatments, there was only one significant difference between SR and RT in AOB community composition of winter season (R value = 0.58, P value = 0.02), indicating the higher effect of straw addition than straw biochar on AOB community composition. Conversely, there was no difference about AOA community between 4 treatments in a whole year. However, there was no consensus about the effects of fertilization regimes on AOA community. Some researchers reported AOA composition was separated between fertilized and unfertilized treatments4,34; while some revealed AOA abundance and structure remained constant after fertilization46,47. The effects were varied from different agricultural management, climate and geographic conditions. This inconsistency of the effects on AOB and AOA may due to the soil nutrient level. Several previous studies found AOB was more favorable in nitrogen rich soil while AOA prefer low nitrogen level soil48,49.
Based on the alpha-diversity analysis (Table 2), our findings evidenced that for both AOA and AOB, fertilized treatments were all higher than CK in the whole year. For all the indices estimated, the sequence of AOB diversity was SB > SR > RT (P < 0.05) in the wheat season. There were generally three reasons causing the effects of biochar and straw on the diversity of AOB. The first was the input of organic C and N through the addition of biochar and straw, which could directly provide substantial nutrient for AOB36,37; the second was the addition of organic improved soil properties (Table 1), such as balancing pH, reducing bulk density etc, enhancing AOB community diversity; the last was the porous structure of biochar, which was beneficial to soil water and nutrient retention, strengthening the sustainability of nutrient supply50.
Venn diagram (Fig. 3) demonstrated that in rice and wheat season, the shared AOA-related OTUs were 24.0% and 23.7% of total, while the shared AOB-related OTUs were 40.5% and 15.3% of total, respectively. These findings suggested fertilization regimes affected AOA-related OTUs almost equally in rice and wheat season, while the influence of fertilization on AOB-related OTUs in the wheat season was much more than in rice season. AOA and AOB are normally aerobic microbes. The activity of AOB might be depressed in the rice season due to the lower oxygen level caused by flooded soil environment21,33. However, AOA could keep active in the rice season because of the characters to be adapted to a wider range of oxygen level and the ability to switch between autotrophy and heterotrophy42,44.
Relative contributions of AOA and AOB to nitrification
Fertilization could increase agricultural soil PNR significantly. In the present study, no significant difference of PNR was observed among different fertilization regimes in the rice season, while the situation was opposite in the wheat season. PNR of organic addition treatments were higher than SR, and SB was the highest treatment (P < 0.05). This was partially supporting some recent reports, which evidenced that synthetic fertilizer combined with biochar, cattle manure or organic fertilizer enhanced soil PNR in dry agricultural soil9,19,31. Organic addition might have the ability to stimulate soil nitrification and enhance the available nutrient supply for crops.
Although AOA outnumbered AOB in the whole year, the results of regression analysis demonstrated that PNR was highly correlated with AOB abundance in both rice and wheat season (P < 0.01), not AOA abundance (Fig. 6). Wang9 tested in 8 periods in one rice-wheat rotation, and found AOB was more important than AOA in ammonia oxidation process. Our results also were similar to Jia51 and Di35, who reported nitrification was driven by AOB rather than AOA. It’s worth noting that these patterns were all observed in alkaline or neutral and nitrogen-rich agricultural soil. Lower correlation with PNR should not indicate the less importance of AOA in soil nitrification. Even in the AOB dominant situation, 10–20% of nitrification still was attributed to AOA52. AOA was also believed to play an important role in nitrification of acid soil or uncultivated land53,54. DNA Stable-Isotope Probing (DNA-SIP) should be applied in future experiment to further describe the specific contribution of AOA and AOB to nitrification.
Due to the characteristics of nutrients slow release of straw and straw biochar, after 7 years application, exchangeable NH4+-N under SR and SB were significantly higher than RT in the harvest stage of rice season (Table 1), which became an important stimulation of AOB growth in wheat season, leading to the higher abundance and diversity of AOB community52,55. This could also be the reason for the rise of wheat yield (Fig. 1). Therefore, under the alkaline soil condition, controlling the activity of AOB in rice-wheat rotation system through agricultural management or NH4+-N addition would be an effective potential strategy to adjust soil nitrification intensity and therefore to improve the crop yield.
In this study, qPCR and high throughout pyrosequencing were employed to evaluate the responses of ammonia oxidizers to successive straw and straw biochar addition in alkaline rice-wheat rotation soil for 7 years. Our results demonstrated straw and straw biochar addition affected AOB more than AOA, especially in the wheat season. Compared to regular fertilization, straw and straw biochar addition increased AOB abundance and diversity significantly only in wheat season (P < 0.05), and they both ranked as SB > SR > RT. Among fertilized treatments, a significant difference between SR and RT was found in AOB community composition of the winter season (R value = 0.58, P value = 0.02). However, AOA was less responsive to organic addition, except the enhancement of abundance by biochar in the wheat season. The abundance of AOB and AOA was negatively correlated with soil pH both in rice and wheat season (P < 0.01), and positively correlated with SOC only in wheat season (P < 0.01). Under our experimental alkaline and nitrogen-rich soil conditions, straw and straw biochar increased soil PNR significantly. AOB abundance was highly positive to soil PNR in both rice and wheat season (P < 0.01), not to AOA abundance, indicating that with the addition of straw and straw biochar, the soil ammonia oxidation process was primarily driven by AOB rather than AOA. AOB had the potential to be regulated for better nitrogen uptake and crop yield in the rice-wheat rotation system.
References
Falkowski, P. G., Fenchel, T. & Delong, E. F. The microbial engines that drive Earth’s biogeochemical cycles. Science 320, 1034–1039 (2008).
Wuchter, C. et al. Archaeal nitrification in the ocean. Proceedings of the National Academy of Sciences USA 103, 12317–12322 (2006).
Leininger, S. et al. Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 442, 806–809 (2006).
He, J. Z. et al. Quantitative analyses of the abundance and composition of ammonia-oxidizing bacteria and ammonia-oxidizing archaea of a Chinese upland red soil under long-term fertilization practices. Environmental microbiology 9, 2364–2374 (2007).
Zhang, L. M., Hu, H. W., Shen, J. P. & He, J. Z. Ammonia-oxidizing archaea have more important role than ammonia-oxidizing bacteria in ammonia oxidation of strongly acidic soils. The ISME Journal 6, 1032–1045 (2012).
Lu, L. et al. Nitrification of archaeal ammonia oxidizers in acid soils is supported by hydrolysis of urea. The ISME Journal 6, 1978–1984 (2012).
Zheng, L. et al. Occurrence and abundance of ammonia-oxidizing archaea and bacteria from the surface to below the water table, in deep soil, and their contributions to nitrification. Microbiology Open 6, e488 (2017).
Ke, X., Angel, R., Lu, Y. & Conrad, R. Niche differentiation of ammonia oxidizers and nitrite oxidizers in rice paddy soil. Environmental microbiology 15, 2275–2292 (2013).
Wang, J. et al. Dynamic Response of Ammonia-Oxidizers to Four Fertilization Regimes across a Wheat-Rice Rotation System. Frontiers in Microbiology 8, 630 (2017).
Tourna, M. et al. Nitrososphaera viennensis, an ammonia oxidizing archaeon from soil. Proceedings of the National Academy of Sciences USA 108, 8420–8425 (2011).
Ai, C. et al. Different roles of rhizosphere effect and long-term fertilization in the activity and community structure of ammonia oxidizers in a calcareous fluvo-aquic soil. Soil Biology and Biochemistry 57, 30–42 (2013).
Xia, W. W. et al. Autotrophic growth of nitrifying community in an agricultural soil. The ISME Journal 5, 1226–1236 (2011).
Song, Y., Zhang, X., Ma, B., Chang, S. X. & Gong, J. Biochar addition affected the dynamics of ammonia oxidizers and nitrification in microcosms of a coastal alkaline soil. Biology and Fertility of Soils 50, 321–332 (2014).
Prosser, J. I. & Nicol, G. W. Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends in Microbiology 20, 523–531 (2012).
Jiang, H. C. et al. Latitudinal Distribution of Ammonia-Oxidizing Bacteria and Archaea in the Agricultural Soils of Eastern China. Applied and Environmental Microbiology 80, 5593–5602 (2014).
de Vries, F. T. et al. Abiotic drivers and plant traits explain landscapescale patterns in soil microbial communities. Ecology Letters 15, 1230–1239 (2012).
Hai, B. et al. Quantification of Key Genes Steering the Microbial Nitrogen Cycle in the Rhizosphere of Sorghum Cultivars in Tropical Agroecosystems. Applied and Environmental Microbiology 75, 4993–5000 (2009).
Wua, D. et al. Nitrification inhibitors mitigate N2O emissions more effectively under straw-induced conditions favoring denitrification. Soil Biology and Biochemistry 104, 197–207 (2017).
Bi, Q. F. et al. Effects of combined application of nitrogen fertilizer and biochar on the nitrification and ammonia oxidizers in an intensive vegetable soil. AMB Express 7, 198 (2017).
He, L. L. et al. Comparison of straw-biochar-mediated changes in nitrification and ammonia oxidizers in agricultural oxisols and cambosols. Biology and Fertility of Soils 52, 137–149 (2016).
Chen, X., Zhang, L. M., Shen, J. P., Wei, W. X. & He, J. Z. Abundance and community structure of ammonia-oxidizing archaea and bacteria in an acid paddy soil. Biology and Fertility of Soils 47, 323–331 (2011).
Chen, X., Zhang, L. M., Shen, J. P., Xu, Z. H. & He, J. Z. Soil type determines the abundance and community structure of ammonia-oxidizing bacteria and archaea in flooded paddy soils. Journal of Soils and Sediments 10, 1510–1516 (2010).
Kurola, J., Salkinoja-Salonen, M., Aarnio, T., Hultman, J. & Romantschuk, M. Activity, diversity and population size of ammonia-oxidising bacteria in oilcontaminated land farming soil. FEMS Microbiology Letters 250, 33–38 (2005).
Rotthauw, J. H., Witzel, K. P. & Liesack, W. The ammonia monooxygenase structural gene amoA as a functional marker: molecular fine-scale analysis of natural ammonia-oxidizing populations. Applied and Environmental Microbiology 63, 4704–4712 (1997).
Francis, C. A., Roberts, K. J., Beman, J. M., Santoro, A. E. & Oakley, B. B. Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proceedings of the National Academy of Sciences USA 102, 14683–14688 (2005).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336 (2010).
Chen, X., Zhu, Y., Xia, Y., Shen, J. P. & He, J. Z. Ammonia-oxidizing archaea: Important players in paddy rhizosphere soil? Environmental microbiology 10, 1978–1987 (2008).
Wang, J., Wang, W. & Gu, J. D. Community structure and abundance of ammonia-oxidizing archaea and bacteria after conversion from soybean to rice paddy in albic soils of Northeast China. Applied Microbiology and Biotechnology 98, 2765–2778 (2014).
Yang, X. R. et al. Potential contribution of anammox to nitrogen loss from paddy soils in Southern China. Applied and Environmental Microbiology 81, 938–947 (2015).
Wang, J. et al. Ammonia oxidizer abundance in paddy soil profile with different fertilizer regimes. Applied Soil Ecology 84, 38–44 (2014).
Tao, R., Steven, A. W., Liang, Y. C. & Chu, G. X. Response of ammonia-oxidizing archaea and bacteria in calcareous soil to mineral and organic fertilizer application and their relative contribution to nitrification. Soil Biology and Biochemistry 114, 20–30 (2017).
Prommer, J. et al. Biochar decelerates soil organic nitrogen cycling but stimulates soil nitrification in a temperate arable field trial. PLoS One 9, e86388 (2014).
Lin, Y. et al. Wheat straw-derived biochar amendment stimulated N2O emissions from rice paddy soils by regulating the amoA genes of ammonia-oxidizing bacteria. Soil Biology and Biochemistry 113, 89–98 (2017).
Wessén, E., Nyberg, K., Jansson, J. K. & Hallin, S. Responses of bacterial and archaeal ammonia oxidizers to soil organic and fertilizer amendments under long-term management. Applied Soil Ecology 45, 193–200 (2010).
Di, H. J. et al. Nitrification driven by bacteria and not archaea in nitrogen rich grassland soils. Nature Geoscience 2, 621–624 (2009).
Di, H. J. et al. Ammonia-oxidizing bacteria and archaea grow under contrasting soil nitrogen conditions. FEMS Microbiology Ecology 72, 386–394 (2010).
Gubry-Rangin, C. et al. Niche specialization of terrestrial archaeal ammonia oxidizers. Proceedings of the National Academy of Sciences USA 108, 21206–21211 (2011).
Stempfhuber, B. et al. pH as a driver for ammonia-oxidizing archaea in forest soils. Microbial Ecology 69, 879–883 (2015).
Li, H., Weng, B. S., Huang, F. Y., Su, J. Q. & Yang, X. R. pH regulates ammonia-oxidizing bacteria and archaea in paddy soils in Southern China. Applied Microbiology and Biotechnology 99, 6113–6123 (2015).
Offre, P., Prosser, J. I. & Nicol, G. W. Growth of ammonia-oxidizing archaea in soil microcosms is inhibited by acetylene. FEMS Microbiology Ecology 70, 99–108 (2009).
Muema, E. K., Cadisch, G., Röhl, C., Vanlauwe, B. & Rasche, F. Response of ammonia-oxidizing bacteria and archaea to biochemical quality of organic inputs combined with mineral nitrogen fertilizer in an arable soil. Applied Soil Ecology 95, 128–139 (2015).
Erguder, T. H., Boon, N., Wittebolle, L., Marzorati, M. & Verstraete, W. Environmental factors shaping the ecological niches of ammonia-oxidizing archaea. FEMS Microbiology Reviews 33, 855–869 (2009).
Pester, M., Schleper, C. & Wagner, M. The Thaumarchaeota: an emerging view of their phylogeny and ecophysiology. Current opinion in microbiology 14, 300–306 (2011).
Walker, C. B. et al. Nitrosopumilus maritimus genome reveals unique mechanisms for nitrification and autotrophy in globally distributed marine crenarchaea. Proceedings of the National Academy of Sciences USA 107, 8818–8823 (2010).
Bannert, A. et al. Changes in diversity and functional gene abundances of microbial communities involved in nitrogen fixation, nitrification, and denitrification in a tidal wetland versus paddy soils cultivated for different time periods. Applied and Environmental Microbiology 77, 6109–6116 (2011).
Wang, Y., Zhu, G., Song, L., Wang, S. & Yin, C. Manure fertilization alters the population of ammonia-oxidizing bacteria rather than ammonia-oxidizing archaea in a paddy soil. Journal of basic microbiology 54, 190–197 (2014).
Ke, X. & Lu, Y. H. Adaptation of ammonia-oxidizing microorganism to environment shift of paddy field soil. FEMS Microbiology Ecology 80, 87–97 (2012).
Shen, J. P., Zhang, L. M., Di, H. J. & He, J. Z. A review of ammonia-oxidizing bacteria and archaea in Chinese soils. Frontiers In Microbiology 3, 296 (2012).
Wu, Y. et al. Long-term field fertilization significantly alters community structure of ammonia-oxidizing bacteria rather than archaea in a paddy soil. Soil Science Society of America 75, 1431–1439 (2011).
Steinbeiss, S., Gleixner, G. & Antonietti, M. Effect of biochar amendment on soil carbon balance and soil microbial activity. Soil Biology and Biochemistry 41, 1301–1310 (2009).
Jia, Z. J. & Conrad, R. Bacteria rather than archaea dominate microbial ammonia oxidation in an agricultural soil. Environmental Microbiology 11, 1658–1671 (2009).
Ouyang, Y., Norton, J. M., Stark, J. M., Reevea, J. R. & Habteselassiec, M. Y. Ammonia-oxidizing bacteria are more responsive than archaea to nitrogen source in an agricultural soil. Soil Biology and Biochemistry 96, 4–15 (2016).
Huang, Q. et al. Archaeal and bacterial diversity in hot springs on the Tibetan Plateau, China. Extremophiles 15, 549–563 (2011).
Isobe, K. et al. High abundance of ammonia-oxidizing archaea in acidified subtropical forest soils in southern China after long-term N deposition. FEMS Microbiology Ecology 80, 193–203 (2012).
Taylor, A. E., Zeglin, L. H., Wanzek, T. A., Myrold, D. D. & Bottomley, P. J. Dynamics of ammonia-oxidizing archaea and bacteria populations and contributions to soil nitrification potentials. ISME Journal 6, 2024–2032 (2012).
Acknowledgements
This work was financially supported by The National Natural Science Foundation of China (No. 41501259) and The National Key Research and Development Program of China (No. 2016YFD0200804) the Outstanding Team Program of Shanghai academy of agricultural sciences [nong ke chuang 2017(A-03)].
Author information
Authors and Affiliations
Contributions
H. Zhang, S. Zhou and W. Lv designed the study; H. Zhang, H. Sun and X. Zheng performed the experiments; H. Zhang, N. Bai, S. Li, J. Zhang and W. Lv analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, H., Sun, H., Zhou, S. et al. Effect of Straw and Straw Biochar on the Community Structure and Diversity of Ammonia-oxidizing Bacteria and Archaea in Rice-wheat Rotation Ecosystems. Sci Rep 9, 9367 (2019). https://doi.org/10.1038/s41598-019-45877-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45877-7
This article is cited by
-
Influence of conditioner and straw on the herbaceous plant-based phytoremediation copper tailings: a field trial at Liujiagou tailings pond, China
Environmental Science and Pollution Research (2024)
-
Warming and altered precipitation rarely alter N addition effects on soil greenhouse gas fluxes: a meta-analysis
Ecological Processes (2023)
-
Fertilization altered co-occurrence patterns and microbial assembly process of ammonia-oxidizing microorganisms
Scientific Reports (2023)
-
Biochar amendment reassembles microbial community in a long-term phosphorus fertilization paddy soil
Applied Microbiology and Biotechnology (2023)
-
Corn Straw Neutralizes Negative Effect of Nitrification Inhibitor on Spinach Biomass at High Nitrogen Levels
Journal of Soil Science and Plant Nutrition (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.