Abstract
Vector-borne diseases are a heavy burden to human-kind. Global warming and urbanization have a significant impact on vector-borne disease transmission, resulting in more severe outbreaks, and outbreaks in formerly non-endemic areas. Miami-Dade County, Florida was the most affected area in the continental United States during the 2016 Zika virus outbreak. Miami is an important gateway and has suitable conditions for mosquitoes year-round. Therefore, it was critical to establish and validate a surveillance system to guide and improve mosquito control operations. Here we assess two years of mosquito surveillance in Miami established after the 2016 Zika virus outbreak. Our results show that the most abundant mosquito species are either well adapted to urban environments or are adapting to it. The five most abundant species comprised 85% of all specimens collected, with four of them being primary vectors of arboviruses. Aedes aegypti and Culex quinquefasciatus were found year-round throughout Miami regardless of urbanization level, vegetation, or socioeconomic variations. This study serves as a foundation for future efforts to improve mosquito surveillance and control operations.
Similar content being viewed by others
Introduction
Vector-borne diseases (VBDs) affect more than half of all human populations living in endemic areas of the globe1. Current estimates show that dengue virus (DENV) infects around 390 million people every year2. The Pan American Health Organization (PAHO) officially confirmed 1,003,509 cases of Zika virus (ZIKV) between 2015 and 2018 in the Americas3, and subsequent studies have also shown the increase in fetus malformation with ZIKV infection during pregnancy4,5.
Considerable efforts have been allocated to fight vector mosquitoes. However, the efforts to control mosquito populations have only achieved limited success, and the global incidence of VBDs is currently on the rise6,7,8,9. Not only have more severe VBD outbreaks been reported but outbreaks have occurred in formerly non-endemic countries such as Italy, France and Croatia10,11,12,13. Furthermore, several arboviruses are circulating in tropical regions of the world, and many more are circulating under the radar14,15,16,17,18,19.
There are only limited options for the treatment of arbovirus infections and their prevention by vaccination. Therefore, controlling vector mosquito populations is widely accepted as the most effective way to prevent the transmission of VBDs20. Controlling vector mosquitoes rely on many steps that logically build on each other. Effective surveillance is fundamental as mosquitoes are often locally concentrated, abundant, and harder to control primarily in those specific, definable habitats at the neighborhood level. These favorable habitats provide optimal conditions and environmental resources needed for mosquito survival, a key determinant of their vectorial capacity. It is critical for the development of any mosquito control strategy to know through effective surveillance the geographic distribution, community composition and relative abundance of vector mosquitoes as well as the risk of introduction of arboviruses21,22.
Miami-Dade County, Florida was the most affected area in the continental United States during the 2016 Zika virus outbreak23. Miami is not only one of the most important gateways to the U.S. with an increased flow of people coming and going from endemic areas, but its proximity to the Caribbean region and Latin America substantially increases the risk of introduction of arboviruses to the U.S. Moreover, Miami also has the appropriate conditions for mosquitoes, its climate is defined as tropical monsoon24, and is conducive for mosquitoes even during the winter. Miami is also undergoing an intense increase in urbanization25 that impacts the population dynamics of vector mosquitoes and patterns of VBD transmission26.
Historically, Miami-Dade County and, in a broader perspective, South Florida have been afflicted by arbovirus outbreaks for decades, including DENV, West Nile virus (WNV) and YFV27,28,29,30,31,32. However, as seen during the most recent ZIKV outbreak, the virus was introduced in Miami multiple times on different occasions33, exposing the real vulnerability of Miami to the introduction of arboviruses and subsequent VBD outbreaks.
Therefore, it was critical to establish a state-of-the-art surveillance system to determine the community composition and abundance of mosquitoes in Miami-Dade County, Florida, to help inform, guide and improve mosquito control operations. Here our objective was to assess the last two years of mosquito surveillance data in Miami-Dade County, Florida.
Results
Mosquito composition and abundance
A total of 2,711,983 mosquitoes were collected in Miami-Dade County from August 2016 to November 2018 by the 157 BG-Sentinel and 34 CDC traps. The collected mosquitoes comprised 41 species from 9 genera. The most abundant species was Culex nigripalpus comprising 1,057,485 (38%) specimens collected, followed by Aedes taeniorhynchus 626,163 (23%), Culex quinquefasciatus 373,571 (13%), Aedes aegypti 150,588 (5%) and Anopheles crucians 132,741 (4%). These 5 species comprised 85% of all collected specimens.
BG-Sentinel traps collected a total of 568,565 mosquitoes, from which 355,381 were Cx. quinquefasciatus (62%) and 134,652 Ae. aegypti (23%), comprising 85% of all collected mosquitoes. CDC traps collected a total of 2,143,418 mosquitoes, from which 1,034,119 were Cx. nigripalpus (48%), 610,547 Ae. taeniorhynchus (28%), 131,077 An. crucians (6%) and 95,193 Aedes tortilis (4%), comprising 85% of all collected mosquitoes. BG-Sentinel traps did not collect Aedes fulvuspallens, Aedes scapularis, Anopheles walker, Culex bahamensis, Culex cedecei, Psorophora howardii and Psorophora johnstonii. CDC traps failed to collect Culex biscaynensis (Table 1).
Biodiversity indices
Mosquito counts obtained by both BG-Sentinel and CDC traps displayed higher levels of variation for the Shannon index and log evenness. However, the log abundance remained stable (Fig. 1A). The analysis of the mosquito counts obtained by the BG-Sentinel traps pointed out many oscillations on the index values (represented by the dots in the lines), indicating higher levels of variation between samples when compared to the data obtained by the CDC traps. On the other hand, the results from the CDC traps indicated a much more heterogeneous scenario, one with considerably more variation than from the mosquitoes collected with BG-Sentinel traps (Fig. 1B).
The biodiversity indices for the CDC trap data had substantially more variation, especially with fewer samples. The log abundance yielded similar values for both BG-Sentinel and CDC traps. However, BG-Sentinel traps reached equilibrium more rapidly than CDC traps, indicating that more specimens were needed for the CDC traps to reach sampling sufficiency. Similar results were also found for the Shannon index, in which CDC traps needed more specimens to reach sampling sufficiency. However, after reaching sampling sufficiency the values remained stable, contrasting with the ones for the BG-Sentinel traps that showed a subtle but steady decline. These results may be due to the fewer number of species collected by BG-Sentinel traps. The results for the log evenness indicate that the mosquito assembly is not even for both traps, with the data from the CDC traps reaching sampling sufficiency and stabilizing, contrasting with the results from the data collected by BG-Sentinel traps, which displayed a constant decreasing trend in value (Fig. 1C).
The individual rarefaction curves analysis considering all mosquitoes collected by both BG-Sentinel and CDC traps indicated that Ae. aegypti, Ae. taeniorhynchus, Cx. nigripalpus and Cx. quinquefasciatus yielded highly asymptotic curves (Fig. 2A). When the data from mosquitoes that were collected by BG-Sentinel traps were analyzed, only Ae. aegypti and Cx. quinquefasciatus reached the asymptote (Fig. 2B). The data obtained by CDC traps also indicated that only two species reached the asymptote, Ae. taeniorhynchus and Cx. nigripalpus (Fig. 2C). Results for the remaining species did not reach the asymptote indicating that sampling sufficiency was not achieved for these species.
Geographical distribution and abundance of species
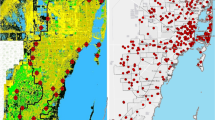
The overall presence and abundance of mosquitoes were found to be higher near the Everglades and the coast than in the core of the city where urbanization is more intense (Fig. 3A). However, when species were analyzed individually a broader spectrum of variation was found. Culex quinquefasciatus was distributed in high numbers throughout the urbanized areas regardless of their location indicating that this species is well adapted to the urban environment of Miami-Dade County (Fig. 3B). Culex nigripalpus, on the other hand, was shown to be more limited to the border of the urban areas, not commonly found in urbanized areas (Fig. 3C). A similar result was found for An. crucians, in which this species was more abundant in areas bordering the limits of the urbanized areas of the Miami-Dade County (Fig. 3D). Aedes taeniorhynchus was collected in high numbers in areas near its natural habitats, including mangrove remnants in urbanized areas such as in the Virginia Keys and Miami Beach (Fig. 3E). Culex coronator was collected in relatively high numbers throughout the whole Miami-Dade County, including urbanized areas (Fig. 3F). As expected, Ae. albopictus was more abundantly found in areas at the border of the urban areas, not being commonly found in highly urbanized areas (Fig. 3G). Aedes aegypti, on the other hand, was the most prevalent species collected in Miami-Dade County, being abundantly collected throughout the urban areas of Miami-Dade County (Fig. 3H).
Seasonal variation of species
The seasonal variation varied greatly accordingly to species. However, from a broader perspective, there were essentially two main patterns, species that peaked in abundance during the warmer months of the year and others during the colder. Unexpectedly, most species had their peak abundance in November when temperatures are considerably cooler than during the peak of the summer in August. On the other hand, Ae. aegypti, Ae. taeniorhynchus and Cx. coronator had their peak abundance during the summer. Culex quinquefasciatus showed a unique pattern, being more abundant during the winter and decreasing in numbers during the summer. Aedes aegypti and Cx. quinquefasciatus were collected in high numbers in all months of the year, with the lowest number of specimens collected being 585 Ae. aegypti in January 2017, and 2,515 Cx. quinquefasciatus in August 2016 (Fig. 4).
Discussion
Urbanization processes are often responsible for decreasing the richness of species, followed by the increase in abundance of the few species that are able to endure and thrive in urban environments34,35,36. This process may lead not only to local adaptation of vector mosquitoes26,37,38,39,40,41, but also can profoundly affect the ecology and behavior of species that are not fully adapted to urban environments42.
Our results revealed that the mosquito community in Miami-Dade County, Florida is comprised of five highly dominant species. Of the five most abundant species, Cx. nigripalpus, Cx. quinquefasciatus, Ae. aegypti and An. crucians are primary vectors of arboviruses. Aedes aegypti and Cx. quinquefasciatus were abundantly found throughout Miami-Dade County regardless of urbanization level, vegetation or socioeconomic variations. To a lesser extent. An. crucians, Cx. coronator and Cx. nigripalpus were also widely distributed,
Furthermore, Ae. aegypti and Cx. quinquefasciatus were abundant year-round, indicating that these species are well adapted to thrive in the urban environments of Miami-Dade County. Culex coronator also had a relatively high abundance and is increasingly becoming a public health concern. Growing evidence shows that Cx. coronator is becoming more adapted to thrive in urban environments and, as a consequence, it is increasing in presence and abundance in urban areas43. Culex coronator was reported in the U.S. in 2004 in Louisiana44. Since then, it has spread and has been commonly found in most of the Southeastern states including Mississippi, Alabama, Florida and Georgia. In 2017, Cx. coronator was detected in the state of Tennessee, where it is currently considered an established species45,46.
SHE profiles reached equilibrium after a rapid initial variation. This indicates that the cumulative species log abundance, Shannon index and log evenness are representative of the community composition and abundance of mosquitoes, and moreover that the sampling effort was adequate. On the other hand, despite the high number of traps and more than two years of weekly collections, the individual rarefaction curves analysis indicated that only Ae. aegypti, Ae. taeniorhynchus, Cx. nigripalpus and Cx. quinquefasciatus reached sampling sufficiency with substantial degrees of confidence for predicting the expected presence of those species for smaller sample sizes. The remaining species did not reach the asymptote and therefore were not a representative measure of the completeness of sampling indicating that their presence and abundance may have been underestimated. These species were found in relatively low densities and were locally concentrated in specific habitats, leading to an increased variance in their sampling. The underestimation of vector species should be of concern since many of the species found in Miami-Dade County were previously implicated in the transmission of arboviruses in the state of Florida28,29,30,43,47.
Our results imply that an increase in the surveillance efforts, such as the inclusion of immature surveys should be considered in addition to the current surveillance system. The combined immature and adult mosquito surveillance methods are complementary and would help detect mosquito species that are not always successfully detected solely by the collection of adult mosquitoes.
Overall, our results suggest that the most abundant mosquito species present in Miami-Dade County are well adapted to thrive in urban environments, or are becoming more adapted to it. Furthermore, the intensification in urbanization processes have resulted in the increased proximity of residential areas to natural areas, therefore, increasing the contact between humans and naturally occurring native sylvatic mosquitoes, such as the aggressive Ae. taeniorhynchus, as well as creating more suitable habitats for mosquito species adapted to thrive in urban environments.
Controlling mosquito populations is becoming more problematic due to, among other things, the increase in the levels of insecticide resistance and lack of effective new control tools22,48. In this context, Miami-Dade County is exceptionally vulnerable to the introduction of arboviruses and subsequent outbreaks, serving as a gateway for the introduction and spread of arboviruses to the U.S.
The introduction of arboviruses inadvertently carried by human movements into and from endemic areas is unavoidable49,50,51,52,53. For example, in 2017, around 4 billion people were transported globally by airlines, with the U.S. alone being responsible for transporting 719 million people in domestic flights and 104 million in international flights54,55. Brazil receives around 2 million tourists from Europe every year, with many of them visiting areas with ongoing active VBD transmission. In 2018 only, several cases of YFV were reported among European travelers returning from Brazil to Europe, including two deaths56,57.
Aedes aegypti has been proven an excellent vector of YFV58, and there is growing evidence that Ae. albopictus is a competent vector, thus important for the maintenance of the YFV cycle in suburban and urban areas59. Furthermore, specimens of Ae. albopictus were found naturally infected with YFV in a transmission hotspot in Brazil corroborating its importance in the patterns of YFV transmission60. Evidence points to a single introduction of YFV in the Southeast region of Brazil to be responsible for the current outbreak61. Furthermore, Ae. aegypti and Ae. albopictus from Brazil and Florida are similarly competent to transmit arboviruses62, exposing the high-risk scenario for Florida residents and tourists.
Continued surveillance, public education, environmental ordinance, and active control of mosquito populations are critical for the prevention of VBD outbreaks. The current scientific consensus is pointing to the increased risk of VBD outbreaks due to many factors, including human behavior and global warming, highlighting even more the need for better mosquito control strategies and an increase in awareness of the general public to the needs for well-established mosquito control operations.
This study serves as a stepping stone for future studies that are needed to uncover the population dynamics patterns of the mosquito species present in Miami-Dade and to assess the risk they pose to both residents and tourists. In our opinion, future studies should focus three main foci: (i) determine how populations of mosquito vector species are largely regulated by the availability of essential resources for their survival in habitats at the local neighborhood level; (ii) Investigate why several recently introduced invasive vector mosquito species such as Culex coronator and Culex panocossa, and historically less common vector species are now locally abundant in habitats in the urban built environment of Miami-Dade County; and (iii) determine how the effectiveness of different vector control methods, considering the development of resistance to insecticides by vector mosquitoes, varies in urban habitats, and how the proliferation of vector mosquitoes can be locally reduced by targeting or modifying specific features of the urban environment.
Public health attention should not be diverted from finding, monitoring and controlling the major known mosquito vectors, but extended towards the neglected and invasive species as well. Furthermore, continuous monitoring is mandatory for effectively guiding mosquito control operations.
Methods
Miami-Dade is the most populous and third largest County in Florida. It has almost 3 million people and more than 6 million km2, spreading from the Everglades on the west to the Biscayne Bay on the east63. The surveillance grid was designed to have at least one trap per 1.6 km2 in the urbanized areas of Miami, as well as traps at the city limits bordering the Everglades. Additional traps were also deployed in points of interest such as touristic areas, shopping malls and areas with outdoor activities, where residents and tourist would be more exposed to mosquitoes.
The Miami-Dade County Mosquito Control surveillance grid is comprised of 191 traps, being 157 BG-Sentinel (Biogents AG, Regensburg, Germany) and 34 CDC traps (Fig. 5). Each trap was deployed weekly for 24 hours from August 2016 to November 2018. All traps were baited with CO2 using a container filled with 1 Kg of dry ice pellets. All collected mosquitoes were transported to the Miami-Dade County Mosquito Control Laboratory and subsequently morphologically identified using taxonomic keys64. The surveillance database does not include data of male mosquitoes since both BG-Sentinel and CDC traps mainly attract females seeking for blood feeding and, therefore, male mosquitoes collected were considered accidental catches and were not considered informative for surveillance purposes.
Since this study posed less than minimal risk to participants and did not involve endangered or protected species the Institutional Review Board at the University of Miami determined that the study be exempt from institutional review board assessment (IRB Protocol Number: 20161212).
Analyzes were carried out for all collected mosquitoes, and subsequently sub-setted for mosquitoes collected using BG-Sentinel and CDC traps. To compare mosquito diversity in samples with different sizes, to provide an estimation of the number of species in samples with fewer specimens and to estimate sampling sufficiency, analyzes of individual rarefaction curves were carried out. Subsequently, plots of cumulative profiles of species log abundance (ln S), Shannon index (H) and log evenness (ln E) (SHE) were calculated for all samples. This model calculates the ln S, H and ln E values individually for each sample, repeating the process for the next sample and so on consecutively until the last one. The results can be interpreted based on deviations from the straight line and are useful to help to grasp subtle trends in species composition and variations in the mosquito assembly65.
Analyses were carried out with 10,000 randomizations without replacement and a 95% confidence interval using Past software (v.3.16)66,67. Figures 1 and 4 were produced using ArcGIS (v.10.2) using maps freely available at www.census.gov.
References
Wilder-Smith, A. et al. Epidemic arboviral diseases: priorities for research and public health. Lancet Infect Dis 17, e101–e106 (2017).
Bhatt, S. et al. The global distribution and burden of dengue. Nature 496, 504–507 (2013).
PAHO/WHO. Zika cases and congenital syndrome associated with Zika virus reported by countries and territories in the Americas (Cumulative Cases), 2015–2017. World Health Organization. Available at: https://www.paho.org/hq/index.php?option = com_content&view = article&id = 12390:zika-cumulative-cases&Itemid = 42090&lang = en.
Delaney, A. et al. Population-based surveillance of birth defects potentially related to Zika virus infection — 15 States and U.S. Territories, 2016. MMWR. Morb. Mortal. Wkly. Rep. 67, 91–96 (2018).
Shapiro-Mendoza, C. K. et al. Pregnancy outcomes after maternal Zika virus infection during pregnancy? U.S. Territories, January 1, 2016 – April 25, 2017. MMWR. Morb. Mortal. Wkly. Rep. 66, 615–621 (2017).
Rosenberg, R. et al. Vital Signs: Trends in reported vector-borne disease cases — United States and Territories, 2004–2016. MMWR. Morb. Mortal. Wkly. Rep. 67, 496–501 (2018).
Paules, C. I. & Fauci, A. S. Yellow Fever — Once again on the radar screen in the Americas. N. Engl. J. Med. 376, 1397–1399 (2017).
Fauci, A. S. & Morens, D. M. Zika virus in the Americas — Yet another arbovirus threat. N. Engl. J. Med. 374, 601–604 (2016).
Etienne, C. et al. Zika virus disease in the Americas: A storm in the making. Am. J. Trop. Med. Hyg. 97, 16–18 (2016).
Poletti, P. et al. Transmission potential of chikungunya virus and control measures: The case of Italy. PLoS One 6, e18860 (2011).
Gould, E. A., Gallian, P., De Lamballerie, X. & Charrel, R. N. First cases of autochthonous dengue fever and chikungunya fever in France: From bad dream to reality! Clin. Microbiol. Infect. 16, 1702–1704 (2010).
Gjenero-Margan, I. et al. Autochthonous dengue fever in Croatia, August–September 2010. Euro Surveill. 16, 1–4 (2011).
Gould, E., Pettersson, J., Higgs, S., Charrel, R. & de Lamballerie, X. Emerging arboviruses: Why today? One Heal. 4, 1–13 (2017).
Lorenz, C. et al. Impact of environmental factors on neglected emerging arboviral diseases. PLoS Negl. Trop. Dis. 11, e0005959 (2017).
Weaver, S. C. Prediction and prevention of urban arbovirus epidemics: A challenge for the global virology community. Antiviral Res. 156, 80–84 (2018).
Rosa, T. D. et al. Oropouche virus: Clinical, epidemiological, and molecular aspects of a neglected orthobunyavirus. Am. J. Trop. Med. Hyg. 96, 1019–1030 (2017).
Amarilla, A. A. et al. Ilheus and Saint Louis encephalitis viruses elicit cross-protection against a lethal Rocio virus challenge in mice. PLoS One 13, 1–12 (2018).
Pauvolid-Corrêa, A. et al. Serological evidence of widespread circulation of West Nile Virus and other Flaviviruses in equines of the Pantanal, Brazil. PLoS Negl. Trop. Dis. 8, e2706 (2014).
Lopes, O. S., Sacchetta, L. A., Francy, D. B., Jakob, W. L. & Calisher, C. H. Emergence of a new arbovirus disease in Brazil. Am. J. Epidemiol. 113, 122–125 (1981).
Roiz, D. et al. Integrated Aedes management for the control of Aedes-borne diseases. PLoS Negl. Trop. Dis. 12, e0006845 (2018).
Lizzi, K. M., Qualls, W. A., Brown, S. C. & Beier, J. C. Expanding integrated vector management to promote healthy environments. Trends Parasitol. 30, 394–400 (2014).
Wilke, A. B. B., Beier, J. C. & Benelli, G. Transgenic mosquitoes – Fact or fiction? Trends Parasitol. 34, 456–465 (2018).
Likos, A. et al. Local mosquito-borne transmission of Zika virus — Miami-Dade and Broward Counties, Florida, June–August 2016. MMWR. Morb. Mortal. Wkly. Rep. 65, 1032–1038 (2016).
Kottek, M., Grieser, J., Beck, C., Rudolf, B. & Rubel, F. World map of the Köppen-Geiger climate classification updated. Meteorol. Zeitschrift 15, 259–263 (2006).
Miami-Dade County, Florida building permits. Available at: http://www.miamidade.gov/permits/ (2018).
Wilke, A. B. B., Vasquez, C., Mauriello, P. J. & Beier, J. C. Ornamental bromeliads of Miami-Dade County, Florida are important breeding sites for Aedes aegypti (Diptera: Culicidae). Parasit. Vectors 11, 283 (2018).
Danauskas, J. X., Ehrenkranz, N. J., Davies, J. E. & Pond, W. L. Arboviruses and human disease in South Florida. Am. J. Trop. Med. Hyg. 15, 205–210 (1966).
Gill, J., Stark, L. M. & Clark, G. G. Dengue surveillance in Florida, 1997–98. Emerg Infect Dis 6, 30–35 (2000).
Rey, J. Dengue in Florida (USA). Insects 5, 991–1000 (2014).
Vitek, C. J., Richards, S. L., Mores, C. N., Day, J. F. & Lord, C. C. Arbovirus transmission by Culex nigripalpus in Florida, 2005. J. Med. Entomol. 45, 483–93 (2008).
Messenger, A. M. et al. Serological evidence of ongoing transmission of dengue virus in permanent residents of Key West, Florida. Vector Borne Zoonotic Dis. 14, 783–787 (2014).
Patterson, K. D. Yellow fever epidemics and mortality in the United States, 1693–1905. Soc. Sci. Med. 34, 855–865 (1992).
Grubaugh, N. D. et al. Genomic epidemiology reveals multiple introductions of Zika virus into the United States. Nature 546, 401–405 (2017).
McKinney, M. L. Urbanization as a major cause of biotic homogenization. Biol. Conserv. 127, 247–260 (2006).
Knop, E. Biotic homogenization of three insect groups due to urbanization. Glob. Chang. Biol. 22, 228–236 (2016).
Johnson, M. T. J. & Munshi-South, J. Evolution of life in urban environments. Science 358, eaam8327 (2017).
Wilke, A. B. B., Wilk-da-Silva, R. & Marrelli, M. T. Microgeographic population structuring of Aedes aegypti (Diptera:Culicidae). PLoS One 12, e0185150 (2017).
Multini, L. C., Wilke, A. B. B., Suesdek, L. & Marrelli, M. T. Population genetic structure of Aedes fluviatilis (Diptera:Culicidae). PLoS One 11, e0162328 (2016).
Wilke, A. B. B., de Carvalho, G. C. & Marrelli, M. T. Retention of ancestral polymorphism in Culex nigripalpus (Diptera:Culicidae) from São Paulo, Brazil. Infect. Genet. Evol. 65, 333–339 (2018).
Louise, C., Vidal, P. O. & Suesdek, L. Microevolution of Aedes aegypti. PLoS One 10, e0137851 (2015).
Medeiros-Sousa, A. R., Fernandes, A., Ceretti-Junior, W., Wilke, A. B. B. & Marrelli, M. T. Mosquitoes in urban green spaces: using an island biogeographic approach to identify drivers of species richness and composition. Sci. Rep. 7, 17826 (2017).
Multini, L. C., Wilke, A. B. B. & Marrelli, M. T. Urbanization as a driver for temporal wing-shape variation in Anopheles cruzii (Diptera: Culicidae). Acta Trop. 190, 30–36 (2019).
Alto, B. W., Connelly, C. R., O’Meara, G. F., Hickman, D. & Karr, N. Reproductive biology and susceptibility of Florida Culex coronator to infection with West Nile virus. Vector-Borne Zoonotic Dis. 14, 606–614 (2014).
Debboun, M., Kuhr, D. D., Rueda, L. M. & Pecor, J. E. First record of Culex (Culex) coronator in Louisiana, USA. J. Am. Mosq. Control Assoc. 21, 455–7 (2005).
Connelly, C. R., Alto, B. W. & O’Meara, G. F. The spread of Culex coronator (Diptera: Culicidae) throughout Florida. J. Vector Ecol. 41, 195–199 (2016).
Trimm, A., Insch, A. & Carlson, T. First Record of Culex coronator In Shelby County, Tennessee. J. Am. Mosq. Control Assoc. 33, 345–347 (2017).
Blackmore, C. G. M. et al. Surveillance results from the first West Nile virus transmission season in Florida, 2001. Am. J. Trop. Med. Hyg. 69, 141–150 (2003).
Vontas, J. et al. Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic. Biochem. Physiol. 104, 126–131 (2012).
Ibañez-Justicia, A. et al. The first detected airline introductions of yellow fever mosquitoes (Aedes aegypti) to Europe, at Schiphol International airport, the Netherlands. Parasit. Vectors 10, 603 (2017).
Gething, P. W. et al. A Long neglected world malaria map: Plasmodium vivax endemicity in 2010. PLoS Negl. Trop. Dis. 6 (2012).
Medlock, J. M. & Leach, S. A. Effect of climate change on vector-borne disease risk in the UK. Lancet Infect. Dis. 15, 721–730 (2015).
Semenza, J. C. & Suk, J. E. Vector-borne diseases and climate change: A European perspective. FEMS Microbiol. Lett. 365, 1–9 (2018).
Ogden, N. H. & Lindsay, L. R. Effects of climate and climate change on vectors and vector-borne diseases: Ticks are different. Trends Parasitol. 32, 646–656 (2016).
Bureau of Transportation Statistics. 2016 Annual and December U.S. Airline Traffic Data. Available at: https://www.bts.gov/newsroom/2017-traffic-data-us-airlines-and-foreign-airlines-us-flights (2017).
International Air Transport Association. Worldwide annual air passenger numbers. Available at: https://www.iata.org/pressroom/pr/Pages/2018-09-06-01.aspx (2017).
Javelle, E., Gautret, P. & Raoult, D. Towards the risk of yellow fever transmission in Europe. Clin. Microbiol. Infect. 8–10 (2018).
Hamer, D. H. et al. Fatal yellow fever in travelers to Brazil, 2018. MMWR. Morb. Mortal. Wkly. Rep. 67, 340–341 (2018).
Couto-Lima, D. et al. Potential risk of re-emergence of urban transmission of yellow fever virus in Brazil facilitated by competent Aedes populations. Sci. Rep. 7, 4848 (2017).
Amraoui, F. et al. Experimental adaptation of the yellow fever virus to the mosquito Aedes albopictus and potential risk of urban epidemics in Brazil, South America. Sci. Rep. 8, 14337 (2018).
PAHO/WHO. Epidemiological update yellow fever. World Health Organization. 1–4 (2017).
Rezende, I. M. d. e. et al. Persistence of yellow fever virus outside the Amazon Basin, causing epidemics in Southeast Brazil, from 2016 to 2018. PLoS Negl. Trop. Dis. 12, e0006538 (2018).
Honório, N. A., Wiggins, K., Câmara, D. C. P., Eastmond, B. & Alto, B. W. Chikungunya virus vector competency of Brazilian and Florida mosquito vectors. PLoS Negl. Trop. Dis. 12, 1–16 (2018).
United States Census Bureau. Income and Poverty in the United States 2016. Available at: https://www.census.gov/topics/income-poverty/income.html.
Darsie, R. F. Jr. & Morris, C. D. Keys to the adult females and fourth-instar larvae of the mosquitoes of Florida (Diptera, Culicidae). 1st ed. Vol. 1. Tech Bull Florida Mosq Cont Assoc (2000).
Buzas, M. A. & Hayek, L. A. C. SHE analysis for biofacies identification. J. Foraminifer. Res. 28, 233–239 (1998).
Morris, E. K. et al. Choosing and using diversity indices: Insights for ecological applications from the German Biodiversity Exploratories. Ecol. Evol. 4, 3514–3524 (2014).
Hammer, Ø., Harper, D. A. T. T. & Ryan, P. D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 4, 9 (2001).
CDC Arbovirus Catalog. Centers for Disease Control and Prevention. Available at: https://wwwn.cdc.gov/Arbocat/Default.aspx (2018).
Bourtzis, K., Lees, R. S., Hendrichs, J. & Vreysen, M. J. B. More than one rabbit out of the hat: Radiation, transgenic and symbiont-based approaches for sustainable management of mosquito and tsetse fly populations. Acta Trop. 157, 115–130 (2016).
Mackay, I. M. & Arden, K. E. Mayaro virus: a forest virus primed for a trip to the city? Microbes Infect. 18, 724–734 (2016).
Vasconcelos, P. F. C. et al. Aedes aegypti, dengue and re-urbanization of yellow fever in Brazil and other South American countries - past and present situation and future perspectives. Dengue Bull. 23, 55–66 (1999).
CDC. Mosquito species in which West Nile virus has been detected. Centers for Disease Control and Prevention Available at: https://www.cdc.gov/westnile/resources/pdfs/Mosquito%20Species%201999-2012.pdf (2017).
Boyer, S., Calvez, E., Chouin-Carneiro, T., Diallo, D. & Failloux, A.-B. An overview of mosquito vectors of Zika virus. Microbes Infect. 20, 646–660 (2018).
WHO. Yellow fever – Brazil. World Health Organization Available at: https://www.who.int/csr/don/09-march-2018-yellow-fever-brazil/en/ (2018).
Le Duc, J. W. et al. Ecology of California Encephalitis Viruses on the Del Mar Va Peninsula. Am. J. Trop. Med. Hyg. 24, 118–123 (1975).
Roberts, D. R. & Scanlon, J. E. The Ecology and behavior of Aedes Atlanticus D. & K. and other species with reference to Keystone virus in the Houston area, Texas. J. Med. Entomol. 12, 537–546 (1975).
Shroyer, D. A. Preliminary Studies of Aedes bahamensis as a host and potential vector of St-Louis Encephalitis-Virus. J. Am. Mosq. Control Assoc. 7, 63–65 (1991).
Ferreira Fé, N., Vale Barbosa, M. D. G., Andrade Fé, F. A., De Farias Guerra, M. V. & Duarte Alecrim, W. Culicidae insect fauna from rural zone in Amazonas state with incidence of sylvatic yellow fever. Rev. Soc. Bras. Med. Trop. 36, 343–348 (2003).
Mitchell, C. J., Forattini, O. P. & Miller, B. R. Vector competence experiments with Rocio virus and three mosquito species from the epidemic zone in Brazil. Rev. Saude Publica 20, 171–177 (1986).
Cardoso, J. & da, C. et al. Yellow fever virus in Haemagogus leucocelaenus and Aedes serratus mosquitoes, Southern Brazil, 2008. Emerg. Infect. Dis. 16, 1918–1924 (2010).
Coffey, L. L. & Weaver, S. C. Susceptibility of Ochlerotatus taeniorhynchus and Culex nigripalpus for Eeverglades virus. Am. J. Trop. Med. Hyg. 73, 11–6 (2005).
Sinka, M. E. et al. A global map of dominant malaria vectors. Parasit. Vectors 5, 69 (2012).
Barrera, R. et al. Mosquito vectors of West Nile virus during an epizootic outbreak in Puerto Rico. J. Med. Entomol. 47, 1185–1195 (2010).
Jensen, T., Dritz, D. A., Fritz, G. N., Washino, R. K. & Reeves, W. C. Lake Vera revisited: Parity and survival rates of Anopheles punctipennis at the site of a malaria outbreak in the Sierra Nevada foothills of California. Am. J. Trop. Med. Hyg. 59, 591–594 (1998).
Molaei, G. et al. Host feeding pattern of Culex quinquefasciatus (Diptera: Culicidae) and its role in transmission of West Nile virus in Harris County, Texas. Am. J. Trop. Med. Hyg. 77, 73–81 (2007).
Bradley, G. H. & Kitchen, S. F. Anopheles walkeri Theobald as a vector of Plasmodium falciparum (Welch). Am. J. Trop. Med. Hyg. 16, 579–581 (1936).
Sakkas, H., Bozidis, P., Franks, A. & Papadopoulou, C. Oropouche fever: A review. Viruses 10, 1–16 (2018).
Acknowledgements
We would like to thank the staff of Miami-Dade County Mosquito Control Division for their help with field collections. We would also like to thank the residents of Miami-Dade County that graciously allowed us to enter their properties. This research was supported by CDC (https://www.cdc.gov/) grant 1U01CK000510-03: Southeastern Regional Center of Excellence in Vector-Borne Diseases: The Gateway Program. CDC had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
C.V., J.M. and A.C. were responsible for the mosquito collection and taxonomic identification. C.V., J.M., A.C., A.B.B.W., W.P. and J.B. conceived of and designed the study. C.V., J.M., A.C. and A.B.B.W. were responsible for the implementation of the study. C.V., J.M., A.C. and A.B.B.W. developed the study methodology and data analysis methodologies. A.B.B.W. collected and analyzed the data, prepared the original figures, and wrote the original draft of the paper. All authors contributed to reviewing and editing the paper. C.V., W.P. and J.B. were responsible for the project administration, funding acquisition, resources, supervision and validation of this study.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wilke, A.B.B., Vasquez, C., Medina, J. et al. Community Composition and Year-round Abundance of Vector Species of Mosquitoes make Miami-Dade County, Florida a Receptive Gateway for Arbovirus entry to the United States. Sci Rep 9, 8732 (2019). https://doi.org/10.1038/s41598-019-45337-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45337-2
This article is cited by
-
Presence and abundance of malaria vector species in Miami-Dade County, Florida
Malaria Journal (2024)
-
Automated differentiation of mixed populations of free-flying female mosquitoes under semi-field conditions
Scientific Reports (2024)
-
Study on the temporal and spatial distribution of Culex mosquitoes in Hanoi, Vietnam
Scientific Reports (2024)
-
Evaluation of the effectiveness of BG-Sentinel and CDC light traps in assessing the abundance, richness, and community composition of mosquitoes in rural and natural areas
Parasites & Vectors (2022)
-
Island biogeography and human practices drive ecological connectivity in mosquito species richness in the Lakshadweep Archipelago
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.