Abstract
The high vapor pressure deficit (VPD) in some arid and semi-arid climates creates undesirable conditions for the growth of tomato plants (Solanum lycopersicum L., cv. Jinpeng). The global CO2 concentration ([CO2]) has also risen in recent years to levels above 400 μmol·mol−1. However, the coordinated effect of VPD and [CO2] on tomato plant growth remains unclear, especially at VPDs of 5–6 kPa or even higher that are extremely detrimental to plant growth. Here, we explore the interaction of VPD and [CO2] on plant water status, stomatal characteristics, and gas exchange parameters in summer greenhouses in a semi-arid area. Plants were grown in four adjacent glass greenhouses with different environmental conditions: (i) high VPD + low [CO2] representing natural/control conditions; (ii) high VPD + high [CO2] representing enriched CO2; (iii) low VPD + low [CO2] representing reduced VPD; and (iv) low VPD + high [CO2] representing reduced VPD and enriched CO2. Reducing the VPD alleviated the water stress of the plant and increased the gas exchange area of the leaf, which was beneficial to the entry of CO2 into the leaf. At this time, the increase of [CO2] was more beneficial to promote the photosynthetic rate and then improve the water use efficiency and yield.
Similar content being viewed by others
Introduction
Atmospheric drought is a very common phenomenon in arid and semi-arid areas, which usually have a high vapor pressure deficit (VPD) that sometimes exceeds the appropriate level for plant growth and impacts water transport and water balance1. Reduced transpiration rate (Tr) at high VPD is observed in most crop species2,3,4. As a result, guard cells may be especially vulnerable to turgor loss under conditions of high evaporation when the low water flux into the stem is insufficient to meet the high Tr. Consequently, stomata close at high VPD, resulting in a decrease in the photosynthetic rate5.
A decrease in Tr at high VPD due to the partial closure of the stomata will help in conserving the soil water, but CO2 assimilation declines due to the water vapor and CO2 exchange synchronization by leaves and canopies6. Premature restriction of Tr will result in a decrease in the ability to transport CO2 to the leaves7, which will have a negative impact on yield8.
In conjunction with global warming and climate change, the atmospheric CO2 concentration ([CO2]) is gradually increasing; levels have risen from about 340 μmol·mol−1 in the 1980s to the current level about 410 μmol·mol−1, with signs this trend will continue9. CO2 diffuses through stomata into the intercellular spaces of leaves and then spreads through the mesophyll to carboxylation sites in the chloroplasts10. Elevated [CO2] can overcome its diffusion limitations11,12, reduce photorespiration13, enhance RuBp regeneration14, and promote efficient assimilation. On the other hand, elevated [CO2] can compensate for the effects of drought on water status15. In C3 plants, rising [CO2] reduces the sensitivity of assimilation rates caused by high VPD to partial stomatal closure16.
Stomata regulate the CO2 uptake and water loss of leaves17. The stomatal responses to VPD are actively driven by the phytohormone abscisic acid (ABA)18 and regulated by rapid gene expression19. Stomatal morphology parameters are also significantly affected by VPD20. Active physiological stomatal control in response to [CO2] will eventually initiate ion pumping to alter the turgor pressure of guard cells21. These mechanisms are closely associated with the capacity to sustain stomatal conductance (gs) and to optimize photosynthesis. VPD, [CO2], and their interaction affect plant transpiration and assimilation22. Angiosperm stomata usually exhibit short-term rapid responses; for example, light irradiance promotes openness but increased [CO2] and VPD promote closure23. The sensitivity of stomatal conductance to VPD is reduced at elevated [CO2]24.
Our research has shown that controlling VPD can benefit protected cultivation systems20,25,26,27, similar to other findings28,29. However, most such studies focus on VPD levels below 4 kPa and few consider VPD up to or even beyond 5–6 kPa30, yet this represents the climate characteristics of most arid and semi-arid areas, such as northwest China. The highest VPD in a summer greenhouse in this region can be up to 6 kPa, but a suitable VPD for tomato growth is less than 2 kPa31. Little research has been done on reducing VPD while increasing [CO2] under these circumstances.
We hypothesized that: (1) lowering the VPD would effectively alleviate the water stress of the leaves, prevent the excessive transpiration that leads to stomatal closure, and thus maintain the water balance of plants and (2) under low VPD conditions, the morphology or density of the stomata may change, increasing the gas exchange area between the interior of the leaves and the outside environment to better balance water loss and CO2 absorption so CO2 can quickly enter the leaves and promote photosynthesis. We tested this hypothesis in a greenhouse study of tomato plants (Solanum lycopersicum L., cv. Jinpeng) considering four different environmental conditions: (i) high VPD + low [CO2] representing natural/control conditions (HVPD-LCO2); (ii) high VPD + high [CO2] representing enriched CO2 (HVPD-HCO2); (iii) low VPD + low [CO2] representing reduced VPD (LVPD-LCO2); and (iv) low VPD + high [CO2] representing reduced VPD and enriched CO2 (LVPD-HCO2). The transpiration rates of the plants exposed to low- and high-VPD were compared. The water status of the leaves was also determined to verify whether excessive transpiration was avoided and the whether stomatal openings were prevented from fully or partially closing. The ability of the plant to photosynthesize was measured to improve the understanding of how the VPD and CO2 have a coordinated effect on plant growth as well as what conditions are more conducive for CO2 to enter the leaves. Accordingly, the biomass, yield, and water use efficiency were measured and compared.
Results
Environmental data
The experimental design successfully achieved very different VPD values in the high- and low-VPD greenhouses. Based on recording VPD changes from 8:00 to 20:00 h every day, the mean of the highest daily VPD values was 4.44 kPa for high VPD conditions and 1.90 kPa for low VPD conditions (Fig. 1). The VPD exceeded 4, 5, 6, and 7 kPa for 36, 20, 10, and 3 days, respectively. Greenhouses with low VPD conditions had levels maintained below 2 kPa.
Coordination between VPD and CO2 on leaf stomatal traits after long-term acclimation
Reducing the VPD under ambient and elevated [CO2] conditions significantly increased the stomatal density (SD) by 24.80 and 42.90%, respectively. Stomatal length and width respectively increased by 12.40 and 13.20% in plants exposed to the LVPD + LCO2 environment and by 6.0 and 4.8% in plants exposed to the LVPD + HCO2 environment compared to the control. No significant differences in stomatal characteristics were observed between plants exposed to high- and low-[CO2] environments under high VPD conditions (Table 1).
Coordination between VPD and CO2 on plant water status
Regulation of VPD and [CO2] strongly affected plant water status. Reducing VPD significantly increased the relative water content (RWC) and water potential (ψleaf) of plant leaves (Fig. 2). Reduced VPD at atmospheric [CO2] resulted in increases in the RWC and ψleaf by 2.33 and 78.65%, respectively; corresponding increments in the high-[CO2] environment were 6.89 and 85.39%. The leaves maintained a higher water status under low VPD conditions, which may be beneficial for CO2 entrance.
Coordination between VPD and CO2 on leaf gas exchange
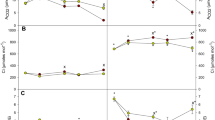
The intercellular CO2 concentration (Ci) significantly increased in plants exposed to HVPD + HCO2 or LVPD + HCO2 conditions (Fig. 3). The photosynthesis rate (Pn) significantly increased by 63.52, 55.11, and 57.64% on day 20, 40, and 60 for plants exposed to the HVPD + HCO2 environment, respectively; by 79.27, 77.88, and 78.41% on day 20, 40, and 60 for plants exposed to the LVPD + HCO2 environment, respectively; and by 29.10% on day 20 for the LVPD + LCO2 environment. The gs increased significantly by 32.26 and 190.63% for plants exposed to LVPD + LCO2 conditions and by 96.77 and 215.63% for plants exposed to LVPD + HCO2 conditions on day 40 and 60, respectively. Corresponding decreases in Tr were 72.22 and 88.24% for plants exposed to LVPD + LCO2 conditions and 77.78 and 82.35% for plants exposed to LVPD + HCO2 conditions. As a result, the RWC and ψleaf for plants exposed to LVPD + LCO2 conditions and LVPD + HCO2 conditions both increased significantly (Fig. 2). The instantaneous water use efficiency (WUEi) significantly increased by 74.77 and 412.75% at 20 and 60 days for plants exposed to LVPD + LCO2 conditions and by 114.49, 811.34, and 833.33% at 20, 40, and 60 days for plants exposed to LVPD + HCO2 conditions (Fig. 3).
Coordination between VPD and CO2 on CO2 response curves and light response curves
Determination of photosynthetic physio-ecological characteristics of plants through CO2 and light response curves has become a hot issue in scientific research32. The Pmax-CO2 (maximum photosynthesis rate on the CO2 response curve) increased by 10.12 and 35.20% in plants exposed to HVPD + HCO2 and LVPD + HCO2 environments, respectively, but decreased by 2.08% in plants exposed to the LVPD + LCO2 environment compared to the control. The interval between the CO2 compensation point (CCP) and CO2 saturation point (CSP) was used to characterize the range of [CO2] that plants could use for net photosynthesis33. The interval was 900.64 μmol·m−2·s−1 for plants exposed to HVPD + LCO2 conditions, 785.17 μmol·m−2·s−1 for HVPD + HCO2 conditions, 797.27 μmol·m−2·s−1 for LVPD + LCO2 conditions, and 921.52 μmol·m−2·s−1 for LVPD + HCO2 conditions. This result shows elevated [CO2] can decrease the interval at high VPD conditions but increases the interval under low VPD conditions. The interval for LVPD + HCO2 conditions was the largest, indicating that plants had the strongest adaptability to different [CO2] for photosynthesis under these conditions.
The order of Pmax-light (maximum photosynthesis rate in light response curve) of plants among treatments was LVPD + HCO2 > HVPD + HCO2 > HVPD + LCO2 > LVPD + LCO2 (Fig. 4). The Pmax-light of plants exposed to HVPD + HCO2 and LVPD + HCO2 conditions significantly increased by 56.16 and 73.92%, respectively, compared to the control. The interval between the light compensation point (LCP) and light saturation point (LSP) indicated the range of light that plants could use for net photosynthesis. This interval was 1622.34 μmol·m−2·s−1 for plants exposed to the HVPD + LCO2 environment, 1720.33 μmol·m−2·s−1 for the HVPD + HCO2 environment, 1438.23 μmol·m−2·s−1 for the LVPD + LCO2 environment, and 1868.25 μmol·m−2·s−1 for the LVPD + HCO2 environment. The interval for the LVPD + HCO2 environment was the largest, indicating that plants had the strongest adaptability to different light intensities for net photosynthesis in these conditions. The dark respiration (Rd) of plants exposed to HVPD + LCO2, HVPD + HCO2, LVPD + LCO2 and LVPD + HCO2 was 2.69, 2.83, 1.18 and 3.73 μmol·m−2·s−1.
Coordination between VPD and CO2 on plant growth
Changes in plant morphology directly reflect adaptability to the growing environment34. Plant stem diameter did not significantly differ under the four treatments considered. The average height of plants exposed to HVPD + LCO2, HVPD + HCO2, LVPD + LCO2, and LVPD + HCO2 environments was 88.2, 93.25, 93.60, and 95.20 cm, respectively. Among these values, plant height in the LVPD + HCO2 conditions was significantly greater than the control (Fig. 5).
Leaves are the largest part of the plant to contact the external environment, and changes in environmental factors directly affect their traits and functions. Leaves play an important role in the relationships between plant water status, light energy interception, and energy balance35,36. The leaf area of plants exposed to HVPD + HCO2 conditions was 2.06% smaller than the control, but for LVPD + LCO2 and LVPD + HCO2 conditions was respectively 6.35 and 11.53% larger than the control (Fig. 5). With growth of the plant, the proportion of root dry weight decreased gradually, of stem dry weight increased gradually, and of leaf dry weight remained unchanged, with no significant differences noted in these values. The leaf and total dry weight of plants exposed to LVPD + LCO2 and LVPD + HCO2 conditions were both greater than the control, with only the latter comparison being significant (Fig. 6). These findings indicate that reducing VPD can increase plant biomass mainly by increasing leaf biomass. Reducing VPD while elevating [CO2] can significantly promote biomass accumulation.
Coordination between VPD and CO2 on water consumption and yield
The average water consumption per plant for plants exposed to HVPD + HCO2 conditions significantly increased (by 14.91%) and for plants exposed to LVPD + LCO2 and LVPD + HCO2 significantly decreased (by 20.83 and 17.18%, respectively) compared to the control (Fig. 7). Water saved per plant for plants exposed to LVPD + LCO2 and LVPD + HCO2 conditions was 2.01 and 1.74 kg, respectively, compared to the control. Average yield per plant for plants exposed to LVPD + LCO2 and LVPD + HCO2 conditions increased significantly (by 11.53 and 16.75%, respectively). Two-factor analysis showed that [CO2] and VPD both had significant effects on plant water consumption and yield, both of which increased under HVPD + HCO2 conditions. Reducing the VPD decreased plant water consumption but increased plant yield and water use efficiency (WUE). The VPD also had a significant effect on WUE.
Discussion
Reduced VPD kept leaves in a higher water state and improved plant water use efficiency
VPD is the driving force for water flow in the soil-plant-atmosphere continuum. Increasing VPD leads to a high evaporation demand. The balance between water supply and evaporative demand is lost under high VPD, causing leaf dehydration and water stress (i.e., leaf wilting, xylem cavitation)37. In this experiment, the low VPD conditions in the greenhouse were effectively maintained below 2 kPa, which is suitable for tomato plant growth. Plants exposed to low VPD conditions did not show excessive transpiration, because of the reduced transpiration pull and the leaves maintained a higher water content and water potential. As such, the changes in Tr were consistent with the changes in RWC and ψleaf. The significant increase in the length and width of the stomata were observed. Together, these indicators indicate that no significant stomatal closure has occurred.
Two-factor analysis showed that VPD, [CO2] and their interactions had significant effects on RWC and leaf water potential. The RWC of plants exposed to LVPD + HCO2 was greater than for plants exposed to LVPD + LCO2. A possible reason is that the greater [CO2] provides more material for plant photosynthesis; the resulting increase in organic matter decreases the osmotic potential of guard cell and increases the amount of water absorbed38.
Both the WUEi and WUE of plants exposed to low VPD conditions increased significantly, indicating low VPD conditions are conducive to water use efficiency. Among them, the increase of WUEi of LVPD + HCO2 is larger, indicating that the application of CO2 under low VPD is more conducive to the instantaneous utilization efficiency of water. The improvement in the WUE of plants exposed to low VPD conditions was due to the increase in yield and decrease in water consumption during plant growth. When irrigation water is scarce, it is feasible to reduce VPD by adding atomized water to the air39. Of course, the water consumption of the atomization system should also be evaluated. The WUEi increase in plants exposed to high [CO2] conditions is consistent with previous research40,41, indicating increased [CO2] can confer drought resistance by reducing carbon starvation42. This is also an important aspect of research under drought conditions.
Coordination of VPD and CO2 stimulated plant growth by enhancing photosynthetic capacity
Arve et al.43 found that, during the light period, stomatal aperture was 25% smaller under low relative humidity (RH) than continuously high RH. In the present experiment, stomatal density, length, and width significantly increased in plants exposed to LVPD + LCO2 or LVPD + HCO2 conditions compared to the control. The gs of plants exposed to HVPD + LCO2 conditions was significantly smaller than those under low VPD conditions after both 40 and 60 days. Similarly, Ramonda nathliate maintained small mesophyll cells and lower gs44 under severe drought conditions to increase the ability to resist drought45. This also indicates that reducing the VPD increases the gas exchange area between the leaves and outside environment. For plants exposed to LVPD + HCO2 conditions, the stomatal limitations (Ls) decreased and gs increased (Fig. 3), which were helpful for CO2 entrance into stomata. The Pn of plants exposed at HVPD + HCO2 and LVPD + HCO2 treatment were both increased significantly, indicating that high [CO2] is beneficial to the photosynthesis of plants, and the larger increase of the latter indicates that low VPD conditions were more conducive to plant CO2 utilization. It was also verified that low VPD environment is more conducive to CO2 entering into stomata.
High [CO2] is favorable for Pn improvement for a short time, but photosynthetic acclimation usually occurs after long-term treatment46,47. However, in the present experiment the Pn of plants exposed to HVPD + HCO2 and LVPD + HCO2 conditions was significantly larger than the control at every stage of plant growth, with no evidence of photosynthetic acclimation. In the former condition, this may be due to the decrease of sensitivity of [CO2] in the late growth stage; in the latter condition, it may be due to the lack of reduction in the demand for CO2 in the later stages of growth.
The Rubisco activation usually has a decrease at high VPD46. In present study, VPD was reduced below 2 kPa at low VPD conditions, the stress of air on plants is alleviated, which can effectively avoid the inactivation of Rubisco. The intervals between CCP and CSP and between LCP and LSP were the largest for plants exposed to LVPD + HCO2 conditions, indicating these plants had the strongest adaptability to the widest [CO2] range and widest light intensity range for net assimilation. Furthermore, elevated [CO2] can increase Pn by inhibiting photorespiration48. The Rd was the highest in plants exposed to LVPD + HCO2 conditions, accompanied by the maximum photosynthetic rate. Similar to previous studies, there was a positive relationship between CO2 assimilation rate and dark respiration rate, that meeting the energy demand for plant growth simultaneously49.
Conclusion
In the present study, the LVPD + HCO2 environment affected plant growth in two aspects. The first effect is related to the water relationship. The water potential and turgor pressure of leaves in low VPD conditions increased due to the reduced transpiration pull. When the products of photosynthesis increase, the osmotic potential of the leaf decreases, the guards cells absorb more water, and the leaves maintain a higher moisture state, which can keep the stomata open50. This is confirmed by the significant increase in stomata size under low VPD conditions along with the increase in both stomatal density and leaf area. These factors all lead to the increase of gas exchange area between the leaves and the external environment. The second is related to the photosynthetic capacity. The increase in gas exchange area is conducive to CO2 entering the leaves and providing more raw material for photosynthesis. At the same time, higher [CO2] can prevent RuBp oxidation and reduce CO2 loss in photorespiration. The CO2 and light response curves indicate the plants exposed to the LVPD + HCO2 environment had the highest utilization ability for CO2 and light and the strongest ability to photosynthesize. The findings of this research are particularly significant with respect to how plants in arid and semi-arid regions will respond to future increases in atmospheric [CO2].
Materials and Methods
Experimental conditions and plant materials
The experiment was carried out from April 1 to June 21, 2017 at Yangling, located in Shannxi province in northwestern China (N 34°15´, E 108°04´, 443.6 m above sea level) in a semi-arid climate zone. Tomato seeds (Solanum lycopersicum L. cv. ‘Jinpeng’) were germinated in an artificial climate chamber with day/night temperatures of 25 °C/18 °C, a 12 h light (400 μmol·m−2·s−1)/12 h dark photoperiod, and RH of 80%. Seedlings were planted in cultivation substrate when plants had four leaves and a center leaf, with pots then placed in four independent but closely connected glass greenhouses, each 3 m wide and 6 m long, for a floor area of 18 m2; this was considered day 0. At midday on a sunny day, the light intensity in the greenhouses reached 800–900 μmol·m−2·s−1 (Fig. 1C). Plant density was 3 plants/m2 25. A total of 30 plants per treatment were randomly sampled at each time point, with at least three repeated measurements taken for each indicator. For each plant, two trusses were used with four fruits per truss. The substrate surface was covered with a black plastic film to prevent water loss due to evaporation and keep the substrate water content at 85–90% of field water capacity. The amount of irrigation water/water consumption equaled the amount of plant transpiration. Pots were weighed at 7 d intervals with an electronic scale. The RH, temperature (T), and light intensity were recorded automatically every 5 minutes using sensors (ZDR-20j, WuGe Instruments Co., Ltd., China). VPD values were calculated using51
Other management considerations were the same as field management.
Environmental design
The two independent variables in this experiment—VPD and [CO2]—were each considered at two levels, for a total of four treatments: natural conditions of high VPD and low [CO2] (HVPD-LCO2) as the control; high VPD with high [CO2] (HVPD-HCO2) representing enriched CO2; low VPD with low [CO2] (LVPD-LCO2) representing reduced VPD; and low VPD with high [CO2] (LVPD-HCO2) representing reduced VPD and enriched CO2. High natural VPD varied with the weather, with the maximum value exceeding 6 kPa. Low VPD was controlled under 2 kPa by a high-pressure industrial humidifier (model BL-C03ZT; BELIN, Shanghai, China); the machine started automatically when the VPD in the greenhouse exceeded 2 kPa. High [CO2] was about 800 ± 50 μmol·mol−1, supplied by a liquefied CO2 cylinder. Natural low [CO2] was about 400 ± 50 μmol·mol−1. VPD and [CO2] were not adjusted on rainy days.
Measurement of leaf water status
Leaves were weighed to determine the leaf fresh weight (Wf), then soaked in distilled water for about 24 hours until they were saturated and weighed again (Wt), then dried at 80 °C to a constant weight (Wd). The RWC was calculated as follows52:
Leaf water potential (ψleaf) was measured between 12:00 and 13:00 h using a pressure chamber (PMS-1000, Corvallis, OR, USA)53.
Measurement of gas exchange parameters
Leaf gas exchange parameters of plants were measured on new fully expanded leaves on sunny days between 9:00 and 11:00 h, using a portable photosynthesis system (LI-6800, Li-Cor, Inc., Lincoln, NE, USA). The environmental parameters of the cuvette were set close to the external environment in the greenhouse during measurement: high VPD conditions were set at 35 °C/30%; low VPD conditions were set at 35 °C/70%. If the VPD is set directly, the range of T or RH may exceed the range of suitable tomato growth even if they meet the VPD setting values. For this reason, we set T and RH directly to conditions suitable for tomato growth instead of setting the VPD directly. Light intensity was set at 1000 µmol·m−2·s−1; high and low [CO2] were set at 800 ± 10 and 400 ± 10 µmol·mol−1, respectively; flow rate was set at 500 µmol·s−1; fan speed was set at 10000 rpm; and the red:blue ratio was set at 9:1. The stomatal limitation (Ls) was calculated according to54:
where Ci is the intercellular [CO2] and Ca is the atmospheric [CO2].
Measurement of CO2 response curves and light response curves
The CO2 and light response curves were measured on the leaves by determining gas exchange parameters. For the CO2 response curves, the CO2 gradient was set at 400, 300, 200, 150, 100, 50, 30, 400, 400, 600, 800, 1000, and 1200 μmol·mol−1, with a light intensity of 1000 μmol·m−2·s−1. For the light response curves, the light intensity gradient was set at 1800, 1500, 1200, 1000, 800, 400, 300, 200, 100, 50, 20, and 0 μmol·m−2·s−1, with high and low [CO2] of 800 ± 10 and 400 ± 10 µmol·mol−1, respectively.
Measurement of stomatal characteristics
Stomatal characteristics were measured on the same leaves used for measurement of gas exchange parameters. A 1-cm2 patch of clear nail polish on the left or right side of the main vein on the abaxial surface of leaves was used to make a transparent imprint. The nail polish was removed with tweezers after it dried and then placed on a glass slide. These leaf peels were then examined using a microscopic imaging system (BX51 + IX71, Olympus, Japan) at both 200× and 400× magnification. For each leaf peel, we selected and photographed 40 views at random at 200× magnification to determine the stomatal density (SD). Measurements of stomatal length and width were derived from photographs taken at 400× magnification55. Images were analyzed using the public domain image processing program ImageJ (ImageJ, U.S. National Institutes of Health, Bethesda, Maryland, USA).
Measurement of plant growth, yield, and WUE
Plant height was measured from the surface of the cultivation substrate to the top of the plant. Stem diameter was measured with a Vernier caliper at 1 cm above the substrate surface. Leaf area was measured on day 0, 30, and 50 using a leaf area meter (LI-COR, Inc., Lincoln, Nebraska, USA). Biomass was measured on day 40 and 60.
Instantaneous water use efficiency (WUEi) was measured by a portable photosynthesis system (LI-6800, Li-Cor, Inc., Lincoln, NE, USA) and calculated as follows56:
The fruit was harvested after the color-turning period and counted to determine yield. WUE was determined from plant yield. Water consumption was calculated from the following equation:
Statistical analysis
Data were analyzed with SPSS (Version 16.0; IBM Institute, USA) using one-way and two-way analysis of variance (ANOVA) to evaluate the effects of VPD, [CO2], and their interaction on all parameters. Differences between treatments were compared using Tukey’s test. Differences were considered significant at P < 0.05.
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available, but can be requested from the corresponding author.
References
Cowan, I. R. Stomatal behaviour and environment. Adv. Bot. Res. 4, 117–228 (1978).
Schoppach, R. & Sadok, W. Differential sensitivities of transpiration to evaporative demand and soil water deficit among wheat elite cultivars indicate different strategies for drought tolerance. Environ. Exp. Bot. 84, 1–10 (2012).
Zaman-Allah, M., Jenkinson, D. M. & Vadez, V. Chickpea genotypes contrasting for seed yield under terminal drought stress in the field differ for traits related to the control of water use. Funct. Plant Biol. 38, 270–281 (2011).
Gholipoor, M., Prasad, P. V. V., Mutava, R. N. & Sinclair, T. R. Genetic variability of transpiration response to vapor pressure deficit among sorghum genotypes. F. Crop. Res. 119, 85–90 (2010).
Sinclair, T. R. et al. Limited-transpiration response to high vapor pressure deficit in crop species. Plant Sci. 260, 109–118 (2017).
Taylor, H. M., Jordan, W. R. & Sinclair, T. R. Limitations to efficient water use in crop production. American Society of Agronomy, Inc., Crop Science Society of America, Inc., and Soil Science Society of America, Inc. (1983).
Schoppach, R., Fleury, D., Sinclair, T. R. & Sadok, W. Transpiration sensitivity to evaporative demand across 120 years of breeding of Australian wheat cultivars. J. Agron. Crop Sci. 203, 219–226 (2016).
Sinclair, T. R. Effective water use required for improving crop growth rather than transpiration efficiency. Front. Plant Sci. 9, 1442 (2018).
NOAA. National Oceanic and Atmospheric Administration, Earth System Research Laboratory. Washington, DC. Ed Dlugokencky and Pieter Tans, NOAA/ESRL, www.esrl.noaa.gov/gmd/ccgg/trends/ (2018).
Terashima, I., Miyazawa, S.-I., Yano, S., Hanba, Y. & Kogami, H. Anatomy of CO2 diffusion in leaf photosynthesis. Sci. Access 3 (2001).
Singh, S. K., Badgujar, G., Reddy, V. R., Fleisher, D. H. & Bunce, J. A. Carbon dioxide diffusion across stomata and mesophyll and photo-biochemical processes as affected by growth CO2 and phosphorus nutrition in cotton. J. Plant Physiol. 170, 801–813 (2013).
Kobza, J. & Edwards, G. E. The photosynthetic induction response in wheat leaves: net CO2 uptake, enzyme activation, and leaf metabolites. Planta 171, 549–559 (1987).
Urban, O. et al. Impact of elevated CO2 concentration on dynamics of leaf photosynthesis in Fagus sylvatica is modulated by sky conditions. Environ. Pollut. 185, 271–280 (2014).
Tomimatsu, H. et al. High CO2 concentration increases relative leaf carbon gain under dynamic light in Dipterocarpus sublamellatus seedlings in a tropical rain forest, Malaysia. Tree Physiol. 34, 944–954 (2014).
Paudel, I. et al. Elevated CO2 compensates for drought effects in lemon saplings via stomatal downregulation, increased soil moisture, and increased wood carbon storage. Environ. Exp. Bot. 148, 117–127 (2018).
Bunce, J. A. Effects of water vapor pressure difference on leaf gas exchange in potato and sorghum at ambient and elevated carbon dioxide under field conditions. F. Crop. Res. 82, 37–47 (2003).
Hetherington, A. M. & Woodward, F. I. The role of stomata in sensing and driving environmental change. Nature 424, 901 (2003).
Bauer, H. et al. The stomatal response to reduced relative humidity requires guard cell-autonomous ABA synthesis. Curr. Biol. 23, 53–57 (2013).
McAdam, S. A. M., Sussmilch, F. C. & Brodribb, T. J. Stomatal responses to vapour pressure deficit are regulated by high speed gene expression in angiosperms. Plant. Cell Environ. 39, 485–491 (2016).
Du, Q., Zhang, D., Jiao, X., Song, X. & Li, J. Effects of atmospheric and soil water status on photosynthesis and growth in tomato. Plant, Soil Environ. 64, 13–19 (2018).
Fujita, T., Noguchi, K. & Terashima, I. Apoplastic mesophyll signals induce rapid stomatal responses to CO2 in Commelina communis. New Phytol. 199, 395–406 (2013).
Wong, S.-C. Interaction between elevated atmospheric concentration of CO2 and humidity on plant growth: comparison between cotton and radish. In CO 2 and Biosphere, 211–221 (Springer, 1993).
Creese, C., Oberbauer, S., Rundel, P. & Sack, L. Are fern stomatal responses to different stimuli coordinated? Testing responses to light, vapor pressure deficit, and CO2 for diverse species grown under contrasting irradiances. New Phytol. 204, 92–104 (2014).
Gunderson, C. A., Sholtis, J. D., Wullschleger, S. D., Tissue, D. T. & Hanson, P. J. Environmental and stomatal control of photosynthetic enhancement in the canopy of a sweetgum (Liquidambar styraciflua L.) plantation during 3 years of CO2 enrichment. Plant Cell Environ. 25, 379–393 (2002).
Zhang, D. et al. Vapour pressure deficit control in relation to water transport and water productivity in greenhouse tomato production during summer. Sci. Rep. 7 (2017).
Du, Q., Xing, G., Jiao, X., Song, X. & Li, J. Stomatal responses to long-term high vapor pressure deficits mediated most limitation of photosynthesis in tomatoes. Acta Physiol. Plant. 40, 149 (2018).
Zhang, D., Jiao, X., Du, Q., Song, X. & Li, J. Reducing the excessive evaporative demand improved photosynthesis capacity at low costs of irrigation via regulating water driving force and moderating plant water stress of two tomato cultivars. Agric. Water Manag. 199, 22–23 (2018).
Li, S., Willits, D. H. & Yunker, C. A. Experimental study of a high pressure fogging system in naturally ventilated greenhouses. In International Symposium on Greenhouse Cooling 719, 393–400 (2006).
Lu, N. et al. Control of vapor pressure deficit (VPD) in greenhouse enhanced tomato growth and productivity during the winter season. Sci. Hortic. (Amsterdam). 197, 17–23 (2015).
Shekoofa, A., Rosas-Anderson, P., Carley, D. S., Sinclair, T. R. & Rufty, T. W. Limited transpiration under high vapor pressure deficits of creeping bentgrass by application of Daconil-Action ®. Planta 243, 421–427 (2016).
Shamshiri, R. et al. Membership function model for defining optimality of vapor pressure deficit in closed-field cultivation of tomato. Acta Hortic. (2017).
Demesin, C. Respiration and photosynthesis characteristics of current-year of Fagus sylvatica: from the seasonal pattern to an annual balance. New Phytol. 158, 465–475 (2003).
Sterck, F. J. & Weemstra, M. Plasticity influencing the light compensation point offsets the specialization for light niches across shrub species in a tropical forest understorey. J. Ecol. 101, 971–980 (2013).
Schlichting, C. D. The evolution of phenotypic plasticity in plants. Annu. Rev. Ecol. Syst. 17, 667–693 (1986).
Roche, P., Díaz-Burlinson, N. & Gachet, S. Congruency analysis of species ranking based on leaf traits: which traits are the more reliable? Plant Ecol. 174, 37–48 (2004).
Vendramini, F. et al. Leaf traits as indicators of resource-use strategy in floras with succulent species. New Phytol. 154, 147–157 (2002).
Tyree, M. T. & Sperry, J. S. Vulnerability of xylem to cavitation and embolism. Annu. Rev. Plant Biol. 40, 19–36 (1989).
Aspelmeier, S. & Leuschner, C. Genotypic variation in drought response of silver birch (Betula pendula): leaf water status and carbon gain. Tree Physiol. 24, 517–528 (2004).
Romero-Aranda, R., Soria, T. & Cuartero, J. Greenhouse mist improves yield of tomato plants grown under saline conditions. J. Am. Soc. Hortic. Sci. 127, 644–648 (2002).
Varga, B., Vida, G., Varga-László, E., Hoffmann, B. & Veisz, O. Combined effect of drought stress and elevated atmospheric CO2 concentration on the yield parameters and water use properties of winter wheat (Triticum aestivum L.) genotypes. J. Agron. Crop Sci. 203, 192–205 (2017).
Varga, B., Bencze, S., Balla, K. & Veisz, O. Effects of the elevated atmospheric CO2 concentration on the water use efficiency of winter wheat. Procedia Environ. Sci. 29, 180–181 (2015).
Drake, B. L., Hanson, D. T., Lowrey, T. K. & Sharp, Z. D. The carbon fertilization effect over a century of anthropogenic CO2 emissions: Higher intracellular CO2 and more drought resistance among invasive and native grass species contrasts with increased water use efficiency for woody plants in the US Southwest. Glob. Chang. Biol. 23, 782–792 (2017).
Arve, L. E. et al. Daily changes in VPD during leaf development in high air humidity increase the stomatal responsiveness to darkness and dry air. J. Plant Physiol. 211, 63–69 (2017).
Rakić, T., Gajić, G., Lazarević, M. & Stevanović, B. Effects of different light intensities, CO2 concentrations, temperatures and drought stress on photosynthetic activity in two paleoendemic resurrection plant species Ramonda serbica and R. nathaliae. Environ. Exp. Bot. 109, 63–72 (2015).
Jiang, Y., Xu, Z., Zhou, G. & Liu, T. Elevated CO2 can modify the response to a water status gradient in a steppe grass: from cell organelles to photosynthetic capacity to plant growth. BMC Plant Biol. 16, 157 (2016).
Shirke, P. A. & Pathre, U. V. Influence of leaf-to-air vapour pressure deficit (VPD) on the biochemistry and physiology of photosynthesis in Prosopis juliflora. J. Exp. Bot. 55, 2111–2120 (2004).
Rogers, A. & Ellsworth, D. S. Photosynthetic acclimation of Pinus taeda (loblolly pine) to long-term growth in elevated pCO2 (FACE). Plant Cell Environ. 25, 851–858 (2010).
Wujeska-Klause, A., Crous, K. Y., Ghannoum, O. & Ellsworth, D. S. Lower photorespiration in elevated CO2 reduces leaf N concentration in mature Eucalyptus trees in field. Glob. Chang. Biol. (2019).
Yamori, W. et al. Enhanced leaf photosynthesis as a target to increase grain yield: insights from transgenic rice lines with variable Rieske FeS protein content in the cytochrome b6/f complex. Plant. Cell Environ. 39, 80–87 (2016).
Wong, S. C. Elevated atmospheric partial pressure of CO2 and plant growth. Photosynth. Res. 23, 171–180 (1990).
Buck, A. L. New equations for computing vapor pressure and enhancement factor. J. Appl. Meterology. 20, 1527–1532 (1981).
Turner, N. C. Techniques and experimental approaches for the measurement of plant water status. Plant Soil 58, 339–366 (1981).
Shackel, K. et al. The relation of midday stem water potential to the growth and physiology of fruit trees under water limited conditions. Acta Hortic. 537, 425–430 (2000).
Duan, Q., Jiang, W., Ding, M., Lin, Y. & Huang, D. Light affects the chloroplast ultrastructure and post-storage photosynthetic performance of watermelon (Citrullus lanatus) plug seedlings. PLoS One 9, e111165 (2014).
Savvides, A., Fanourakis, D. & Van Ieperen, W. Co-ordination of hydraulic and stomatal conductances across light qualities in cucumber leaves. J. Exp. Bot. 63, 1135–1143 (2012).
Zhou, L. et al. Responses of photosynthetic parameters to drought in subtropical forest ecosystem of China. Sci Rep 5, 18254 (2015).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 31471916) and grants from the China Agriculture Research System (No. CARS-23-C-05).
Author information
Authors and Affiliations
Contributions
J.L. conceived and designed the experiments. X.J., X.S., D.Z. and Q.D. performed the experiments. X.J. and J.L. analyzed the data. X.J. contributed to the writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiao, XC., Song, XM., Zhang, DL. et al. Coordination between vapor pressure deficit and CO2 on the regulation of photosynthesis and productivity in greenhouse tomato production. Sci Rep 9, 8700 (2019). https://doi.org/10.1038/s41598-019-45232-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45232-w
This article is cited by
-
Climate change and the growth of Amazonian species seedlings: an ecophysiological approach to Euterpe oleracea
New Forests (2023)
-
The impact of elevated CO2 concentration on fruit size, quality, and mineral nutrient composition in tomato varies with temperature regimen during growing season
Plant Growth Regulation (2023)
-
Coordination of leaf hydraulic, anatomical, and economical traits in tomato seedlings acclimation to long-term drought
BMC Plant Biology (2021)
-
2,4-Epibrassinolide mechanisms regulating water use efficiency and fruit production in tomato plants
Brazilian Journal of Botany (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.