Abstract
The Western honeybee, Apis mellifera, is the most important managed pollinator globally and has recently experienced unsustainably high colony losses. Synergistic interactions among stressors are believed to be primarily responsible. However, despite clear evidence of strong effect on honeybee longevity of widely-employed neonicotinoid insecticides and of the ubiquitous ectoparasitic mite Varroa destructor, no data exist to show synergistic effects between these two stressors. Even though neonicotinoids had no significant impact by themselves, we here show for the first time a synergistic time-lag interaction between mites and neonicotinoids that resulted in significantly reduced survival of long-lived winter honeybees. Even though these mites are potent vectors of viruses, the virus-insecticide interaction had no significant impact. The data suggest a previously overlooked mechanism possibly explaining recent unsustainably high losses of managed A. mellifera honeybee colonies in many regions of the world. Future mitigation efforts should concentrate on developing sustainable agro-ecosystem management schemes that incorporate reduced use of neonicotinoids and sustainable solutions for V. destructor mites.
Similar content being viewed by others
Introduction
As pollinators, bees provide a key ecosystem service that is essential for human food security and maintenance of natural biodiversity1,2. The Western honeybee, Apis mellifera, is the single most important managed pollinator globally3. Therefore, unsustainably high colony mortality experienced by A. mellifera beekeepers in many countries is alarming4.
Interactions among stressors are believed to be primarily responsible for increased A. mellifera colony mortality recently observed5,6. It must be pointed out that such proposed interactions have to yield strong synergistic, or at least additive effects, to have a significant impact on a global scale. To date, comparatively little attention has focussed on the combined effects of two confirmed A. mellifera stressors – the mite Varroa destructor and neonicotinoid insecticides. The former is a nearly ubiquitous ectoparasite of Asian origin that is the main biological cause of honeybee colony mortality worldwide4,7, primarily because of viral infections vectored by this parasite8. The neonicotinoids are among the most commonly employed insecticides globally and are known to elicit wide-ranging lethal and sublethal effects on honeybees9,10. The effects of V. destructor, viruses and neonicotinoids have been intensively studied independently over the past decades7,10,11. Even though the co-occurrence between V. destructor and neonicotinoids is virtually inevitable in honeybee colonies12, possible effects of their interactions have rarely been considered. The few previous studies on the topic have not shown synergistic interactions between these mites and insecticides13,14,15, even though those are often assumed to occur. Moreover, the timing of interactions needs to be taken into account. Indeed, in autumn, colonies produce the usually long-living winter honeybees, which are essential for survival in temperate regions16,17. Therefore, reduced longevity of the winter bees would increase risks for colony death. Besides longevity, previous studies have shown that pathogens and neonicotinoid insecticides can affect adult emergence mass15,18. Since body mass is a proxy for individual performance (e.g. thermoregulation19), this sublethal effect may also yield consequences at the honeybee colony level. To this date, the impact of combined neonicotinoid exposure and V. destructor mite parasitism on winter honeybee weight and life expectancy has not been addressed. Similarly, the reported interactions between neonicotinoids and viruses20 have not been verified in field colonies yet, even though the proportion of honeybee workers with clinical symptoms of deformed wing virus (DWV) is a known predictive marker for colony collapse21. Given synergistic or additive interactions between V. destructor and neonicotinoids reducing winter honeybee longevity and increasing clinical DWV symptoms, a previously overlooked key mechanism contributing to overwintering colony mortality would emerge.
In a fully crossed experiment, we investigated sublethal and lethal effects of neonicotinoids and V. destructor on individual adult honeybee workers. Using a pre-established method22, we exposed honeybee colonies to neonicotinoid contaminated pollen (at field-realistic doses of 4 ppb thiamethoxam and 2 ppb clothianidin) or to control uncontaminated pollen for 42 days in spring, a time when colonies are typically exposed to plant protection products23. Within each group of colonies, workers parasitized by V. destructor during their development or not were collected to obtain a fully crossed design. To investigate possible seasonal effects, we measured the clinical symptoms of DWV and worker mass on emergence and longevity in laboratory cages. This was conducted in summer immediately following neonicotinoid exposure and 16 weeks later in autumn when colonies rear winter bees.
Results
Pollen patty consumption and colony strength
Average daily consumption was measured by weighing the leftover pollen patties (median colony consumption per day was ~70–73 grams); no significant differences were observed between neonicotinoid treated and non-exposed control colonies (Extended Data Table 1). Colony strength assessments revealed no significant differences between control and neonicotinoid insecticide treatment (Extended Data Table 1).
V. destructor infestation levels and DWV clinical symptoms
No significant differences in mite infestation levels obtained by either mite washes, bottom board counts or brood dissection were observed between neonicotinoid-exposed and control colonies, neither for summer nor autumn (Extended Data Tables 1 and 2; Extended Data Fig. 1). Similarly, no significant differences were observed for levels of DWV clinical symptoms between only neonicotinoid-exposed and control workers in summer and autumn (Fig. 1; Extended Data Table 3). In contrast, a significant increase in the proportion of workers with DWV clinical symptoms was observed for the V. destructor parasitism and combined treatment groups compared to controls in both seasons (Fig. 1; Extended Data Table 3). No significant difference was observed between V. destructor parasitism and combined treatment groups (Fig. 1; Extended Data Table 3).
Proportion of honeybee (Apis mellifera) workers exposed to neonicotinoid insecticides and/or Varroa destructor and showing clinical symptoms of Deformed wing virus (DWV). Measurements of DWV clinical symptom levels for each of the four treatment groups were generated by uncapping worker brood cells and investigating wing anatomy for each individual. The level of DWV clinical symptoms was then calculated by dividing the total number of observed workers with symptoms by the total number of uncapped brood cells. The boxplots show the inter-quartile-range (box), the median (black line within box), and outliers (dots). A significant difference (generalized linear mixed model, P < 0.05) between groups is indicated by different letters (a, b).
Worker emergence mass
There was no significant impact of only neonicotinoid treatments on worker emergence mass, in summer or in autumn (Fig. 2A,C; Extended Data Table 3). In contrast, a significant negative effect of V. destructor parasitism on worker emergence mass was observed in both summer and autumn (Fig. 2A,C; Extended Data Table 3). In both seasons, the decrease in emergence mass was strongest when parasitism was combined with neonicotinoids (Extended Data Table 4). Individuals exposed to only V. destructor parasitism or to both stressors in summer weighed less compared to controls, (4.4 and 8.1% reduction in median emergence mass, respectively; Extended Data Tables 4 and 5). A synergy between stressors was observed because the effect of combined stressor exposure (8.1%) was higher than the sum of individual effects (0 + 4.4% of only neonicotinoids and only V. destructor parasitism, respectively). Emergence mass in autumn decreased by 0.3% (only neonicotinoids), 8.6% (only V. destructor parasitism) and 13.2% (combined) on average (Extended Data Table 5). Therefore, synergistic effects were also present in autumn.
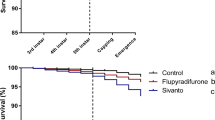
Effects of neonicotinoid insecticides and Varroa destructor on honeybee (Apis mellifera) worker emergence mass and survival. To obtain a fully crossed experimental design, honeybee colonies were exposed to neonicotinoids (4 ppb thiamethoxam and 2 ppb clothianidin) and to natural V. destructor infestation. Measurements of emergence mass were performed in summer and autumn. For each of the four treatment groups generated, body mass (1A, 1C) of 1430 workers originating from ten colonies was measured 24 h prior expected emergence date. The boxplots show the inter-quartile-range (box), the median (black line within box), and outliers (dots). Survival of the workers (N = 1105) kept in cages in groups of 10 was measured until all individuals died. The Kaplan-Meier curves show the survival over time of the four treatment groups (solid lines) as well as the 95% confidence intervals (CI) (shaded areas). A significant difference (generalized linear mixed model, P < 0.05) between groups is indicated by different letters (a, b, c).
Worker survival
Similar to emergence mass, exposure to only neonicotinoids did not yield a significant effect on worker survival in summer and autumn (Fig. 2B,D; Extended Data Table 3), whereas exposure to only V. destructor parasitism and combined stressors resulted in strong negative effects (Fig. 2B,D; Extended Data Table 4). In summer, survival of workers exposed to only V. destructor parasitism (measured as survival curve) was not significantly different compared to workers that were exposed to combined stressors, and resulted in a small increase in hazard rate (HR) of ~153 and ~122% compared to controls, respectively (Fig. 2B, Extended Data Fig. 2A, Extended Data Table 6). In autumn, a significant decrease in the survival of the workers exposed to the combined treatment with respect to only V. destructor parasitism was observed (Fig. 2B,D; Extended Data Table 3). The HR for only V. destructor parasitism and combined stressors in autumn was ~230 and ~272%, respectively (Fig. 2D, Extended Data Fig. 2B, Extended Data Table 6).
With respect to median survival, no significant difference was observed due to only neonicotinoids for both seasons (Extended Data Table 3). In summer, exposure to only V. destructor parasitism and to the combined stressors had a significantly larger effect by reducing the median survival of workers by 68% in both cases (Extended Data Table 5). Since the observed magnitude of the effect of combined stressors is equal to that of the sum of the individual stressors (0 + 68% of only neonicotinoids and only V. destructor parasitism), there was no evidence of synergy in summer (Extended Data Table 5). Evidence for synergism was obtained in autumn, when the large detrimental effects of exposure to only V. destructor parasitism on median survival (47% decrease) were surpassed by those of the combined exposure with neonicotinoids (70% decrease, Extended Data Table 5).
Seasonal effects
Brood infestation rates revealed no significant seasonal effects for both control and neonicotinoid exposed colonies (Extended Data Table 3). Mite wash counts were significantly higher in autumn compared to summer for both control and neonicotinoid exposed colonies (Extended Data Table 3). Likewise, a significant seasonal effect was observed for bottom board counts for the control colonies, whereas no difference was seen for the neonicotinoid exposed colonies (Extended Data Table 3). No significant seasonal effect for the proportion of workers with DWV clinical symptoms was observed between summer and autumn for all treatment groups (Extended Data Table 3). Furthermore, no significant effects for emergence mass were observed between summer and autumn controls or for workers exposed to only neonicotinoids (Fig. 3A,C; Extended Data Table 3). In contrast, workers exposed to only V. destructor parasitism and to combined stressors were significantly lighter in autumn when compared to summer (Fig. 3E,G). For survival, no difference was observed between seasons for control workers (Fig. 3B) or workers exposed to only V. destructor parasitism (Fig. 3F), while seasonal effects were observed for the neonicotinoids and combined treatments (Fig. 3D,H) with survival being lower in autumn.
Seasonal effects of neonicotinoid insecticides and Varroa destructor on honeybee (Apis mellifera) worker emergence mass and survival. For each of the four treatment groups, body mass of 1425 workers originating from ten colonies was measured 24 h prior expected emergence date. The boxplots show the inter-quartile-range (box), the median (black line within box), and outliers (dots). Survival of the previously weighed workers (N = 1100) kept in cages in groups of 10 was measured until all individuals died. The Kaplan-Meier curves show the survival over time of the four treatment groups (solid lines) as well as the 95% CI (shaded areas). A significant difference (P < 0.05) between groups within the same cage assay is indicated by different letters (a, b).
Colony mortality and queen loss
There was no significant difference between control and neonicotinoid treated colonies in either winter mortality (one non-exposed, two neonicotinoid-exposed colonies: Chi-square test, χ2 = 0.56, df = 1, P = 0.45) or queen loss (one in non-exposed, two in neonicotinoid-exposed colonies, two treatment queens: Chi-square test, χ2 = 0.39, df = 1, P = 0.53).
Discussion
Our data clearly show a significant negative synergistic effect of neonicotinoids and V. destructor mites on A. mellifera honeybee body mass and longevity. While negative synergism was observed for body mass in both summer and autumn, it was only observed for survival in autumn. This suggests a time-lag effect of previous neonicotinoid exposure that could contribute to elevated mortality of overwintering honeybee colonies. Even though colony mortality and queen loss were not significantly different between the treatment groups in our study, likely because we routinely treated our colonies against V. destructor, our data nevertheless suggest that the combined exposure to both parasitic mites and neonicotinoids increases the risk for honeybee colony mortality by negatively affecting the survival of winter honeybees.
Non-parasitized workers originating from colonies exposed to only neonicotinoids only, did not differ from the controls at any point, confirming earlier results24. In contrast, honeybee workers exposed to only V. destructor parasitism were consistently lighter and shorter-lived compared to the controls, which is also in line with previous studies18,25. The data also confirm the potent role of V. destructor as a virus vector26 due to the significantly higher proportions of bees with DWV clinical symptoms when infested by the mite. While laboratory data show positive interactions between viruses and insecticides20, the impact of insecticides on the occurrence of DWV clinical symptoms was not significant in our semi-field study (honeybees reared in field colonies but emerged in the laboratory). Had the virus-insecticide interface been a key factor, a significant effect on DWV clinical symptoms would have been observed. The underlying mechanism of the observed effects on mass and longevity is thus likely dominated by the mite-insecticide interface, with a varying impact of the mite-associated viruses. Indeed, this seems inevitable due to the highly variable nature of viruses27.
Interactions between stressors for honeybee health can in principle range from negative, over neutral and additive to synergistic ones28. Our study provides supporting evidence for the long-held hypothesis of synergism between neonicotinoids and V. destructor, constituting a possible mechanism for elevated losses of managed honeybee colonies4. This idea is supported by Blanken et al.15, who found a negative effect of this combination of stressors on a sublethal endpoint of individual bees (flight performance), while there was no effect of the stressors alone, hinting at an additive or synergistic interaction. Our study also showed that such synergism has lethal effects by affecting worker survival. A notable difference between the studies is the opposite effect of the combined stressors on the sublethal endpoint worker mass. Our study showed a negative effect while Blanken et al.15 showed a positive effect of the combined stressors on mass. This could be due to the different exposure routes of the neonicotinoids (pollen vs. sugar water, respectively) or to the different compound tested (thiamethoxam + clothianidin vs. imidacloprid, respectively). These same methodological differences might explain why a recent study by van Dooremalen et al.14 revealed that the combined exposure to a neonicotinoid and V. destructor did not yield significant negative effects on colony size or survival. In conclusion, it appears as if exposure route may be an important parameter to consider in risk assessment tests due to potential diverging consequences.
The synergism between neonicotinoids and V. destructor that we observed could be due to the mite infestation down-regulating the expression of cytochrome P450 genes in its host13. These genes play a crucial role in detoxification of xenobiotics, including neonicotinoids13. Even in the absence of a synergistic effect of these stressors on the expression of a gene from this family13, the effect of its down-regulation due to parasitism alone may lead to a negative synergetic effect on host survival. In absence of the insecticide, the likely reduced detoxification ability might be inconsequential, while in its presence, larger physiological effects than in the presence of the parasite alone might occur.
Synergism between neonicotinoids and V. destructor affected emergence mass in both summer and autumn, whereas it affected worker survival only in autumn. This decrease in survival 16 weeks post neonicotinoid colony exposure reveals a previously overlooked time lag effect of neonicotinoid exposure. The absence of significant neonicotinoid effects on survival of summer bees indicates that direct exposure to residues is unlikely to be responsible for the effect on workers in autumn. It was also not likely due to infestations by V. destructor alone since its effect on our endpoint measurements was constant over the seasons. Instead, this time lag effect most likely resulted from dysfunction within the colony environment (e.g., a reduction in nursing quality) due to the previous neonicotinoids exposure29. Time-lag effects could have also occurred because of an adaptive trait of honeybees to temperate regions. Under temperate climate, honeybee colonies produce two types of worker bees – short-lived summer bees in spring and early summer, and long-lived winter bees in late summer and autumn that are adapted to surviving this period of dearth30. The adaptation can include down-regulation of immune genes31, which might explain why susceptibility to both individual mite and combined stressors increased in autumn. The previously reported reduced longevity of winter bees exposed to V. destructor mites alone32 could be amplified by the here reported negative synergistic interactions between mites and neonicotinoids. Because honeybee colonies must produce sufficient quantities of long-living winter bees to survive33, the observed negative synergistic effects on individual winter honeybee longevity are most likely strongly compromising colony survivorship. Our results therefore imply that synergistic effects between these nearly ubiquitously occurring stressors are likely to contribute to honeybee colony losses over winter.
By providing essential pollination services to agriculture, managed honeybees are often exposed to agrochemicals. Given the ubiquitous nature of V. destructor mites potentially interacting with any agrochemical with a similar mode of action to neonicotinoids, it appears crucial to mitigate the synergistic interactions between such stressors. A reduction of honeybee colony exposure to agrochemicals, combined with more efficient and sustainable V. destructor solutions34,35,36,37, appears sensible in light of our results. In addition, investigating possible interactions between other common stressors of honeybees seem essential for mitigating A. mellifera colony mortality (e.g., between insecticides and malnutrition). This study highlights the possible important role of interactions between an invasive parasite and neonicotinoids in many regions of the world. However, current insecticide risk assessments largely ignore interactions between stressors and thus only provide a fragmentary evaluation of their possible effects38. Future efforts by risk assessors should therefore include possible interactions and time lag effects between agrochemicals and other stressors to safeguard honeybees.
Material and Methods
Set-up
The study was conducted in Bern, Switzerland between April 2014 and May 2015. We employed 20 local honeybee colonies, which were considered healthy based on V. destructor infestation levels and the absence of any obvious clinical symptoms of diseases based on visual inspections39. These colonies were established at the beginning of the experiment using shook swarms (one laying sister queen, ∼1.8 kg workers) and five Dadant frames equipped with organic worker cell wax foundation. Colonies were maintained by local Best Management Practices that included autumn and winter V. destructor mite management using formic (FAM dispenser, 70%) and oxalic (2.7%) acids7. Mite infestation levels were quantified for each colony (29 August 2014 & 31 October 2014) using the soapy water wash and sticky bottom board methods39 and individual worker brood cell infestations39.
Neonicotinoid exposure
Colonies were provided with 100 g pollen paste [55% irradiated honeybee corbicular pollen, 5% brewer’s yeast, and 40% sucrose solution (60% w/v)] on top of the frames on a daily basis, and concurrently fitted with entrance pollen traps to promote feeding on the provided paste22,40,41. Colonies were randomly allocated to one of two treatments (neonicotinoid or no neonicotinoid) in mid May 2014. Pollen paste fed to the colonies belonging to the neonicotinoid treatment contained 4.0 ppb thiamethoxam and 2 ppb of its break-down product clothianidin (both Sigma-Aldrich), which represent field-realistic concentrations of the chemicals found in plant pollen42. For both neonicotinoid and control colonies roughly 90 kg of pollen paste were made. The patties were made in three individual batches by adding the appropriate amount of corbicular pollen, sugar powder and honey, as well as a neonicotinoid solution for the pesticide treatment group. After homogenization, a random sample of pollen paste was taken from each batch for residue analysis. The provided residue levels derive from taking the average value of the three random pollen paste samples from both treatment groups. Pollen patties were stored at −24 °C until they were fed to colonies. The applied concentrations were confirmed (3.9 ppb thiamethoxam and 1.9 ppb clothianidin in neonicotinoid insecticide patties; below limit of quantification for thiamethoxam (less than 0.02 ppb) and clothianidin (less than 0.08 ppb) in control patties) by the French National Centre for Scientific Research using ultra-high performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS). Pollen paste feeding occurred for 42 days to expose colonies for at least two complete worker brood cycles. This ensured that the obtained experimental workers were also reared by workers exposed during their development.
Experimental workers
For all laboratory cage assays, the queen of each colony was restricted for two days to a worker brood comb built by adult workers in the respective colony during treatment exposure. This resulted in known age-cohorts of workers brood43. The experimental frames remained in the respective colonies until ∼48 h prior to adult emergence, when they were transferred to a laboratory incubator maintained at 34.5 °C, 60% RH in complete darkness43. One-day later (i.e., one day before adult emergence) several hundred cells per colony were carefully and individually uncapped to obtain the required number of experimental workers. Each worker was visually examined to estimate appropriate cohort age41, mite infestations39, and clinical symptoms of disease44. The proportion of individuals showing clinical symptoms of DWV (crippled wings and/or shortened abdomens21,45) were recorded and used as a proxy of virus levels45 as well as a predictive marker of colony mortality21. Individuals displaying deformed wings were excluded. Each selected worker was weighed to the nearest 0.1 mg using an analytic scale (Mettler Toledo AT400), and then assigned to one of the four treatment groups based on their previous colony-level exposure to neonicotinoids and individual V. destructor parasitation status: 1. No neonicotinoids/No V. destructor parasitism (control), 2. No neonicotinoids/Yes V. destructor parasitism (only V. destructor), 3. Yes neonicotinoids/No V. destructor parasitism (only neonicotinoids), 4. Yes neonicotinoids/Yes V. destructor parasitism (combined)). Each treatment group consisted of 30 hoarding cages (80 cm³; 3 cages per treatment colony) that each contained 10 workers43. Cages were maintained in complete darkness at 30 °C and ~60% RH43, and provided 50% (w/v) sucrose solution ad libitum. Every 24 h, food was replaced, worker mortality was recorded, and dead honeybees were removed. Each cage assay was terminated when the last worker died.
Honeybee worker health was evaluated by measuring both sublethal (emergence mass) and lethal (survival) health indices, because V. destructor causes both7. This was required although the tested insecticide concentrations are believed to cause sublethal effects only, because the potential interactions with parasitism may be lethal.
Timing
Two hoarding cage assays were performed, starting on 29 June 2014 (summer assay) and 21 September 2014 (autumn assay). These dates corresponded to 6.5 and 16 weeks post initial neonicotinoid exposure and were used to evaluate immediate effects after exposure (summer assay), as well as time-lag effects in autumn (autumn assay).
Colony parameters
The colony strength parameters were assessed using the Liebefeld estimation method that visually quantified bees, capped and uncapped brood, honey, and pollen in each individual colony46,47. Colony strength variables (i.e., total bees, total capped brood surface [cm²], total uncapped brood surface [cm²], total honey surface [cm²], and total pollen surface [cm²]), were first evaluated as percent scores ranging from 0 to 100 of frame coverage. Percent coverage was then converted into absolute values of area [cm²] (e.g., total capped brood surface [cm²]) or weight [kg] (e.g., honey [kg])47. Assessments were carried out six weeks (23.06.2014) and 12 weeks (06.08.2014) post initial treatment exposure.
Bottom board counts, as well as soapy mite wash counts, were performed to determine colony level infestation rates of V. destructor mites7,39 between control and neonicotinoid insecticide exposed treatments for both seasons. In addition, V. destructor brood infestation rates (brood mites)7 were determined during uncapping of the experimental frames (see above) by dividing the total mite-parasitized uncapped cells by the overall total uncapped cells per colony.
Any possible seasonal effects were determined by comparing all three estimates of V. destructor infestation (brood, bottom board, soapy wash)39 and DWV clinical symptom levels between summer and autumn.
Colony mortality and queen loss were recorded throughout the entire experiment.
Statistical analyses
Treatment and seasonal differences were assessed using generalized linear mixed (regression) models (GLMM) fitted using STATA1548. The treatment at the colony level was exposure to neonicotinoids or not. At the individual bee level, we had four treatment groups based on their previous colony-level exposure to neonicotinoids and individual V. destructor parasitism status 1. No neonicotinoids/No V. destructor parasitism (control), 2. No neonicotinoids/Yes V. destructor parasitism (only V. destructor), 3. Yes neonicotinoids/No V. destructor parasitism (only neonicotinoids), 4. Yes neonicotinoids/Yes V. destructor parasitism (combined). Treatment was included as the fixed explanatory variable, where applicable (see Extended Data Table 1). The colony and cage identification numbers were included as random effects to take into account the clustering effects due to every individual’s original colony and our experimental set up in cages49. Therefore, individual workers represented experimental units. In addition, a goodness of fit of the models was assessed by the analysis of residuals, with the STATA function predict [option deviance]. All figures were created using STATA15.
Count data variables (e.g., total bees, bottom board counts, and mite wash counts) were fitted to negative binomial models using the STATA15 function menbreg50. Total capped brood [cm²], total honey [kg], pollen patty consumption [g] or emergence mass [mg]) were modeled with GLMM of the Gaussian or Gamma family (depending on the analysis of residuals) using the STATA15 function meglm. Instead of transforming the outcome varaibles, we opted for the Gamma family that provided good fits (normality of the residuals).
Proportion of mite-infested brood and workers showing DWV clinical symptoms were recorded as a score ranging from 0 to 100% and subsequently an ordered logistic model was applied using the function meologit51. Multiple pairwise comparisons (Bonferroni test) for mite infestation levels and emergence mass were obtained by using the mcompare(bonferroni) function52. Chi-square was used to assess for significant differences between control and neonicotinoid insecticide exposed colony mortality and queen losses.
Worker survival data for all cage assays and treatment groups were fit using the mestreg function for multilevel survival models49,53. Median survival was calculated as the 50th percentile of the survival time25. Furthermore, two different models for adjusting survivor functions for the effects of covariates were used: the proportional hazards (PH) model and the accelerated failure time (AFT) model54,55 (Extended Data Table 6). Both models enable the calculation of a regression coefficient, in our case the hazard rate of a specific treatment group compared to another treatment group (i.e., control). Survival curves (Kaplan Meier plots) and smooth estimated hazard rate plots with 95% CI were visually inspected to determine the appropriate model and are presented in the Extended Data Fig. 2. If the survival curves declined parallel to one another at the same ratio, the PH model (with a Weibull distribution) was applied; if curves diverged, with one showing an increased decline, an AFT model was applied. As its name suggests, the AFT model accounts for an accelerated decrease in the survival rate of the considered treatment with respect to control. Due to the different parameterizations, the coefficients of both models are not directly comparable and conversion is needed. In STATA, the PH model directly calculates a hazard ratio; however, AFT calculates a coefficient’s factor which can then be exponentiated and converted into a hazard ratio56. The relationship between the coefficients of both models (using the PH model with a Weibull distribution) was:
where βAFT is the regression coefficient of the AFT model and p denotes the ancillary parameter (which is estimated in logarithmic metric and displayed in STATA output as \ln_p). The hazard ratios can be interpreted as ‘conditional hazard ratios’ that is conditional on the random effects. A hazard ratio of 1.01 means 1.01 × 100% = 101%, that is a 1% increase in the hazard for each unit of the explanatory variable. In addition, multiple pairwise comparisons (Bonferroni test) for worker survival amongst the different groups were obtained by using the mcompare(bonferroni) function56,57. Whenever possible, every three-level model was compared with its single-level model counterpart using a Likelihood ratio (LR) test58. Likelihood ratio (LR) tests, which did not rely on the assumption of asymptotic normal sampling distributions, were used to demonstrate which model best fit the data.
Interactions
To further identify potential synergistic interactions between V. destructor and neonicotinoids, we employed an additive effects framework28. In this model, synergism and antagonism occur when the combined effect of multiple stressors is greater (synergism) than the sum or smaller (antagonism) than the sum of effects elicited by individual stressors59. Interactive stress effects on emergence body mass and survival were calculated as the percent difference in treated groups relative to controls, whereby the mean emergence mass [g] and median survival [d] were used for the calculations.
Data Availability
The complete raw data can be found on the Dryad repository (https://doi.org/10.5061/dryad.r886083).
References
Garibaldi, L. A. et al. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339(6127), 1608–11 (2013).
Stanley, D. A. et al. Neonicotinoid pesticide exposure impairs crop pollination services provided by bumblebees. Nature 528(7583), 548–550 (2015).
Kleijn, D. et al. Delivery of crop pollination services is an insufficient argument for wild pollinator conservation. Nat. Commun. 6(7414), 1–8 (2015).
Neumann, P. & Carreck, N. Honey bee colony losses. J. Apic. Res. 49, 1 (2010).
Goulson, D., Nicholls, E., Cristina, B. & Rotheray, E. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science. 347(6229) (2015).
Potts, S. G. et al. Global pollinator declines: trends, impacts and drivers. Trends Ecol. Evol. 25(6), 345–53 (2010).
Rosenkranz, P., Aumeier, P. & Ziegelmann, B. Biology and control of Varroa destructor. J. Invertebr. Pathol. 103, S96–119 (2010).
Locke, B., Forsgren, E., Fries, I. & de Miranda, J. R. Acaricide treatment affects viral dynamics in Varroa destructor-infested honey bee colonies via both host physiology and mite control. Appl. Environ. Microbiol. 78, 227–235 (2012).
Hallmann, C. A. et al. Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511(7509), 341–343 (2014).
Blacquière, T., Smagghe, G., van Gestel, C. A. M. & Mommaerts, V. Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology 21, 973–92 (2012).
Aubert, M. et al. Virology and the honey bee. (European Commission, 2005).
Mitchell, E. A. D. et al. A worldwide survey of neonicotinoids in honey. Science. 358(6359), 109–111 (2017).
Gregorc, A., Evans, J. D., Scharf, M. & Ellis, J. D. Gene expression in honey bee (Apis mellifera) larvae exposed to pesticides and Varroa mites (Varroa destructor). J. Insect Physiol. 58, 1042–9 (2012).
van Dooremalen, C., Cornelissen, B., Poleij-Hok-Ahin, C. & Blacquière, T. Single and interactive effects of Varroa destructor, Nosema spp., and imidacloprid on honey bee colonies (Apis mellifera). Ecosphere 9, e02378 (2018).
Blanken, L. J., Langevelde, F. V. & Dooremalen, C. V. Interaction between Varroa destructor and imidacloprid reduces flight capacity of honeybees. Proc. R. Soc. B 282, 1–9 (2015).
Winston, M. L., Taylor, O. R. & Otis, G. W. Some differences between temperate European and tropical African and South American honeybees. Bee World 64, 12–21 (1983).
Seeley, T. D. & Visscher, P. K. Survival of honeybees in cold climates: the critical timing of colony growth and reproduction. Ecol. Entomol. 10, 81–88 (1985).
Bowen-walker, P. L. & Gunn, A. The effect of the ectoparasitic mite, Varroa destructor on adult worker honeybee (Apis mellifera) emergence weights, water, protein, carbohydrate, and lipid levels. Entomol. Exp. Appl. 101, 207–217 (2001).
Heinrich, B. Thermoregulation in endothermic insects. Science. 185(4153), 747–756 (1974).
Di Prisco, G. et al. Neonicotinoid clothianidin adversely affects insect immunity and promotes replication of a viral pathogen in honey bees. Proc. Natl. Acad. Sci. USA 110, 18466–71 (2013).
Dainat, B. & Neumann, P. Clinical signs of deformed wing virus infection are predictive markers for honey bee colony losses. J. Invertebr. Pathol. 112, 278–80 (2013).
Williams, G. R. et al. Neonicotinoid pesticides severely affect honey bee queens. Sci. Rep. 5, 14621 (2015).
Botías, C., David, A., Hill, E. M. & Goulson, D. Contamination of wild plants near neonicotinoid seed-treated crops, and implications for non-target insects. Sci. Total Environ. 567, 269–278 (2016).
Straub, L. et al. Neonicotinoid insecticides can serve as inadvertent insect contraceptives. Proc. R. Soc. B. 283, 20160506 (2016).
Yang, X. & Cox-Foster, D. Effects of parasitization by Varroa destructor on survivorship and physiological traits of Apis mellifera in correlation with viral incidence and microbial challenge. Parasitology 134, 405–12 (2007).
Neumann, P., Fries, I., de Miranda, J. R. & Yañez, O. Varroa invasion and virus adaptation. Trends Parasitol. 28, 353–354 (2012).
Domingo, E. & Holland, J. J. RNA virus mutations and fitness for survival. Annu. Rev. Microbiol. 51, 151–178 (1997).
Folt, C. L., Chen, C. Y., Moore, M. V. & Burnaford, J. Synergism and antagonism among multiple stressors. Limmol. Ocean. 44, 864–877 (1999).
Hatjina, F. et al. Sublethal doses of imidacloprid decreased size of hypopharyngeal glands and respiratory rhythm of honeybees in vivo. Apidologie 44, 467–480 (2013).
Mattila, H. R., Harris, J. L. & Otis, G. W. Timing of production of winter bees in honey bee (Apis mellifera) colonies. Insectes Soc 48, 88–93 (2001).
Steinmann, N., Corona, M., Neumann, P. & Dainat, B. Overwintering is associated with reduced expression of immune genes and higher susceptibility to virus infection in honey bees. PLoS One 10(6), 1–18 (2015).
Dainat, B., Evans, J. D., Chen, P., Gauthier, L. & Neumann, P. Dead or alive: Deformed Wing Virus and Varroa destructor reduce the life span of winter honeybees. Appl. Environ. Microbiol. 78(4), 981–987 (2012).
Martin, S. J. The role of Varroa and viral pathogens in the collapse of honeybee colonies: a modeling approach. J. Appl. Ecol. 38, 1082–1093 (2001).
Locke, B. Natural Varroa mite-surviving Apis mellifera honeybee populations. Apidologie 47, 467–482 (2016).
Oddie, M. et al. Rapid parallel evolution overcomes global honey bee parasite. Sci. Rep. 8(1), 7704 (2018).
Neumann, P. & Blacquière, T. The Darwin cure for apiculture? Natural selection and managed honey bee health. Evol. Appl. 1–5, https://doi.org/10.1111/eva.12448 (2016).
Dietemann, V. et al. Varroa destructor: research avenues towards sustainable control. J. Apic. Res. 51, 125–132 (2012).
Rortais, A. et al. Risk assessment of pesticides and other stressors in bees: Principles, data gaps and perspectives from the European Food Safety Authority. Sci. Total Environ. 587–588, 524–537 (2017).
Dietemann, V. et al. Standard methods for varroa research. J. Apic. Res. 52, 1–54 (2013).
Sandrock, C. et al. Impact of chronic neonicotinoid exposure on honeybee colony performance and queen supersedure. PLoS One 9(8), e103592 (2014).
Human, H. et al. Miscellaneous standard methods for Apis mellifera research. J. Apic. Res. 52, 1–56 (2013).
Pilling, E., Campbell, P., Coulson, M., Ruddle, N. & Tornier, I. A four-year field program investigating long-term effects of repeated exposure of honey bee colonies to flowering crops treated with thiamethoxam. PLoS One 8, e77193 (2013).
Williams, G. R. et al. Standard methods for maintaining adult Apis mellifera in cages under in vitro laboratory conditions. J. Apic. Res. 52, 1–36 (2013).
Williams, G. R. et al. Deformed wing virus in western honey bees (Apis mellifera) from Atlantic Canada and the first description of an overtly-infected emerging queen. J. Invertebr. Pathol. 101, 77–9 (2009).
de Miranda, J. R. & Genersch, E. Deformed wing virus. J. Invertebr. Pathol. 103, S48–S61 (2010).
Gerig, L. & Imdorf, A. Lehrgang zur Erfassung der Volksstärke. Bienen-Zeitung 4, 199–204 (1983).
Delaplane, K. S., Steen, J. VanDer & Guzman-Novoa, E. Standard methods for estimating strength parameters of Apis mellifera colonies. J. Apic. Res. 52, 1–12 (2013).
StataCorp. Stata Statistical Software: Release 15 (2017).
Leckie, G. Centre for multilevel modeling, LEMMA VLE. Centre for Multilevel Modeling (2010).
Garson, G. D. Fundamentals of hierarchical linear and multilevel modeling. In, Hierarchical linear modeling. Guide and applications (pp. 3–25) (Los Angeles: Sage Publications, 2013).
Greene W. H. Econometric analysis, 7th edn. (Englewood Cliffs, NJ: Prentice Hall, 2012).
Mitchell, M. N. Interpreting and visualizing regression models using Stata. (Stata Press, 2012).
Cleves, A. M. How do I analyze multiple failure-time data using Stata? Stata J. (1999).
Kay, R. & Kinnersely, N. On the use of the accelerated failure time model as an alternative to the proportional hazards model in the treatment of time to event data: A case study in influenza. Drug Inf. J. 36, 571–579 (2002).
Wei, L. J. J. The accelerated failure time model: A useful alternative To the Cox regression model in survival analysis. Stat. Med. 11, 1871–1879 (1992).
StataCorp. Stata 15 Base Reference Manual. (Stata Press, 2017).
StataCorp. The Stata Journal. Stata Journal 4 (2004).
Lee, E. T. & Wang, J. Statistical methods for survival data analysis. (John Wiley & Sons, Inc., 2003).
Hay, M. E. Defensive synergisms? Reply to Pennings. Ecology 77, 1950–1952 (1996).
Acknowledgements
The author’s thank Laura Brettell, and the two anonymous reviewers for their valuable and detailed comments which significantly improved our manuscript. Christoph Moor from the BAFU and Inge Werner from the Swiss Centre of Applied Ecotoxicology engaged us with fruitful discussions. Financial support was provided by the Bundesamt für Umwelt (BAFU) to L.S., P.N. and G.R.W., by Agroscope to G.R.W., L.S. and V.D., by the Vinetum Foundation to P.N. and G.R., by ETH Global (People Exchange Grant) to K.K., Chiang Mai University Fund to P.N. and G.R.W., the USDA National Institute of Food and Agriculture Multi-state Hatch project 1014681 to G.R.W., the Swiss National Science Foundation Project 31003A_169751 to G.R.W., as well as the Foundation for Food and Agriculture Research Pollinator Health Fund grant 549003 to G.R.W.
Author information
Authors and Affiliations
Contributions
G.R.W., V.D. and P.N. designed the experiment; L.S., G.R.W., V.D., K.K., A.S. and G.R. carried out the experiment and collected field as well as laboratory data; P.N., V.D. and G.R.W. provided material; B.V. and L.S. designed the statistical analysis and wrote the statistical methods and results; L.S., B.V., G.R.W., V.D. and P.N. analyzed the data. All authors contributed to the interpretation of results and writing of the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Straub, L., Williams, G.R., Vidondo, B. et al. Neonicotinoids and ectoparasitic mites synergistically impact honeybees. Sci Rep 9, 8159 (2019). https://doi.org/10.1038/s41598-019-44207-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44207-1
This article is cited by
-
Positive selection in cytochrome P450 genes is associated with gonad phenotype and mating strategy in social bees
Scientific Reports (2023)
-
Environmental differences explain subtle yet detectable genetic structure in a widespread pollinator
BMC Ecology and Evolution (2022)
-
Acute toxicity of the insecticide abamectin and the fungicide difenoconazole (individually and in mixture) to the tropical stingless bee Melipona scutellaris
Ecotoxicology (2021)
-
Investigating Acute and Subchronic Effects of Neonicotinoids on Northwestern Salamander Larvae
Archives of Environmental Contamination and Toxicology (2021)
-
You are what you eat: relative importance of diet, gut microbiota and nestmates for honey bee, Apis mellifera, worker health
Apidologie (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.