Abstract
Abiotic stresses such as water deficit, salt, and heat are major environmental factors that negatively affect plant growth, development, and productivity. Previous studies showed that overexpression of the Arabidopsis vacuolar H+-pyrophosphatase gene AVP1 increases salt and water deficit stress tolerance and overexpression of the rice SUMO E3 ligase gene OsSIZ1 improves heat and water deficit stress tolerance in transgenic plants. In this report, the effects of co-overexpression of AVP1 and OsSIZ1 in Arabidopsis on abiotic stress tolerance were studied. It was found that AVP1/OsSIZ1 co-overexpressing plants performed significantly better than AVP1-overexpressing plants and OsSIZ1-overexpressing plants, and produced 100% more seed than wild-type plants under single stress or multiple stress conditions. The increased stress tolerance in AVP1/OsSIZ1 co-overexpressing plants was substantially larger than the increased stress tolerance in AVP1-overexpressing plants and OsSIZ1-overexpressing plants under every abiotic stress condition tested. This research provides the proof-of-concept that crop yields might be substantially improved using this approach.
Similar content being viewed by others
Introduction
Water deficit, salinity, and heat are major environmental factors that negatively affect every aspect of plant growth and development, leading to substantial yield losses in agriculture. World population is growing steadily, and it will likely pass 9 billion in 20501. Direct improvement in crop productivity under limited inputs (primarily water) and marginal growing conditions is critical if we are to meet future demands of global food and fiber production. Irregular rainfall, depletion of groundwater supplies, and competition for freshwater with rapidly-expanding urban areas, all place significant constraints on resources for agricultural productivity. Exacerbating these issues is the fact that the average air temperature is projected to increase 1.5–4 °C by the end of the century and for many regions may result in even more unpredictable weather patterns2. It is therefore imperative to develop crops that are more tolerant to drought, heat, and salt stresses, thereby producing sustainable yields under severe stress conditions. Over the last 20 years, many genes have been reported to confer increased stress tolerance when overexpressed in transgenic plants, and some of these genes might be useful in the engineering of crops for higher stress tolerance3. However, the successful implication of this strategy in agriculture is currently very limited. The reason was that most studies used single genes in transgenic plants and the increased stress tolerance was not high enough, or the specificity of increased tolerance was limited to one stress in most cases, which did not lead to substantially increased yields under field conditions3. In production agriculture, abiotic stresses usually do not occur in isolation; instead, they often occur in combinations. For arid and semi-arid production environments, water deficit stress often occurs during heat waves, and a direct response to water deficit is stomatal closure, which results in elevated leave temperatures and thermal stress4. Thus, it is likely necessary to combine the benefits of multiple transgenes to engineer abiotic stress tolerance or improve crop yields under multiple stress conditions.
Salinity affects plants by causing water deficit in plant cells as a result of osmotic effects and inhibition of many enzymes in primary and secondary metabolisms5, and this is achieved through (1) reducing the water potential inside plant cells due to the presence of high concentration of ions such as Na+ and Cl−, (2) causing ionic stress or toxicity by disrupting K+/Na+ homeostasis and inactivation of many important enzymes in plant cells6. Water deficit stress leads to inhibition of root and shoot development due to the reduction in cell division and elongation, alters flowering times and number, and inhibits seed development and production7.
It was well known that increased salt tolerance could be achieved by reducing the cytosolic Na+ concentrations8. Plants can lower the concentration of Na+ by excluding Na+ via plasma membrane-bound Na+/H+ antiporter such as SOS19 or sequestration of Na+ into vacuole via the tonoplast membrane-bound Na+/H+ antiporter such as NHX110. The activity of NHX1 depends on the proton gradient across the tonoplast membrane that is created by proton pumps such as H+-ATPase and vacuolar H+-pyrophosphatase11. The Arabidopsis gene AVP1 encodes a vacuolar H+-pyrophosphatase that pumps H+ into vacuole and overexpression of AVP1 leads to increased salt tolerance, which was attributed to Na+-sequestration into the vacuolar lumen at the expense of protons12. Overexpression of AVP1 also leads to increased auxin polar transport, thereby resulting in robust root development, leading to higher water absorption capacity and increased drought tolerance13. Studies with AVP1-overexpressing tomato14 and cotton15 showed a dramatic increase in root biomass and improved plant survival rate under drought and salt stress conditions. Yang et al. revealed that the overexpression of AVP1 in transgenic plants promotes rhizosphere acidification, root proliferation, and fruit development under phosphate starvation conditions16,17. It has also been demonstrated that overexpression of the H+-PPase gene from Thellungiella halophila in maize could enhance plant response to limiting phosphate concentrations18. Lv et al. reported a significant improvement in growth and photosynthetic rates of cotton plants overexpressing the H+-PPase gene from Thellungiella halophila in response to salt stress19. In addition, the positive correlation between nitrate use efficiency and AVP1 up-regulation in lettuce was shown20. Several studies on overexpression of AVP1 or another type I H+-PPase genes have shown a substantial improvement in salt and drought tolerance in various plants including cotton15,19,21, creeping bentgrass22, maize23, peanut24, sugarcane25, tobacco26, and rice27. Schilling et al. showed that overexpression of AVP1 increased yield and shoot biomass in barely plants grown in saline soil28.
Heat stress is another environmental stress that results in significant losses in crop production worldwide29, and it is predicted to become even more serious in the future30. It has been shown that an increase in air temperature above a plant’s optimum growth temperature decreases net photosynthetic rates in plants31 and affects membrane fluidity, which disrupts ion hemostasis in plant cells32. Therefore, increasing heat tolerance in crops may allow plants to maintain metabolic function and growth under heat stress conditions. Efforts have been made in engineering plants for heat stress tolerance by overexpressing heat-shock protein genes or heat-shock factor genes in transgenic plants over the last 20 years3, yet limited success was made, and so far none was applied in crops.
Recent discoveries might change the landscape of transgenic research on heat-tolerant plants. Overexpression of OsSIZ1, a SUMO E3 ligase gene from rice, was found to confer increased heat and drought tolerance in transgenic creeping bentgrass33 and transgenic cotton34.
SUMO stands for small ubiquitin-like modifier and it can be added to proteins via a post-translational process called sumoylation by a three-enzyme system: SUMO E1 activating enzyme, SUMO E2 conjugating enzyme, and SUMO E3 ligase35. The sumoylation reactions carried out by SUMO E3 ligases are important to many regulatory proteins, as sumoylation might alter protein-protein interactions, change protein activities or subcellular localization of proteins35. The importance of the Arabidopsis SUMO E3 ligase SIZ1 in plant growth, development, and response to hormones, abiotic and biotic stresses have been studied extensively in Arabidopsis35. For examples, and the roles of SIZ1 in sugar signaling36, in plant response to drought37 and cold temperature stress38 were well documented. Other studies also illustrated the function of SIZ1 in regulating phosphate starvation response39 and nitrogen assimilation in Arabidopsis through sumoylation of nitrate reductase40. Yoo et al. showed that siz1 mutants are thermal hypersensitive, indicating that SIZ1-dependent sumoylation is required for heat tolerance41. These studies showed that the Arabidopsis SIZ1 and its homolog in rice, OsSIZ1, are important in the establishment of heat and drought tolerance in plants. Our recent research34 indicates that OsSIZ1-overexpressing cotton plants performed better than any other transgenic cotton plants that we have created over the last 20 years, which includes AtNHX1-overexpressing cotton plants42, AVP1-overexpressing cotton plants15, and IPT-transgenic cotton plants43.
Given the important role of AVP1 and SIZ1 in plant abiotic stress response, we hypothesized that co-overexpression of AVP1 and OsSIZ1 would have an additive effect on tolerance to water deficit, salinity, and high-temperature stress in Arabidopsis. Our findings show that AVP1/OsSIZ1 co-overexpression significantly enhanced tolerance to these stresses and serves as a proof-of-concept that co-overexpression of these genes has the potential to improve yields in crop plants significantly.
Results
Creation and molecular analysis of AVP1/OsSIZ1 co-overexpressing plants
The expression cassette of the pdual35S-AVP1/pUbi-OsSIZ1 was cloned into a pBI121 based binary vector and introduced into Arabidopsis thaliana, using Agrobacterium-mediated “floral dip” transformation44. Forty independent transgenic lines were identified by screening on MS media supplemented with 30 µg/ml of kanamycin. DNA was isolated from kanamycin-resistant lines, and the presence of AVP1 and OsSIZ1 was verified by polymerase chain reaction (PCR). RNA blot analysis was performed to verify transgene expression, and three independent AVP1/OsSIZ1 co-overexpressing lines, AO1, AO2, and AO3 were selected for further experiments (Fig. 1A). The two reference lines expressing either AVP1 or OsSIZ1 were used as positive controls for the RNA blot (Fig. 1A). Also, we conducted DNA blot analysis to confirm the stable integration of transgenes into the Arabidopsis genome and the copy numbers of transgenes in these transgenic plants. Our data indicated that AO1, AO2, and AO3 plants are likely due to three, single, and two T-DNA insertion events, respectively (Fig. 1B).
Molecular analysis of wild-type and AVP1/OsSIZ1 co-overexpressing plants. (A) RNA blot analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants. (B) DNA blot analysis of wild-type and AVP1/OsSIZ1 co-overexpressing plants. WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. M, molecular weight markers.
AVP1/OsSIZ1 co-overexpression results in enhanced tolerance to salt, heat, and water deficit stresses
A series of experiments were conducted to examine the response of AVP1/OsSIZ1 co-overexpressing plants (AO), AVP1-overexpressing plants (A), OsSIZ1-overexpressing plants (O), and wild-type plants (WT) to salt, heat, and water deficit stresses. Plants were grown on MS plates and in potting soil for all experiments and control conditions were defined as PPFD = 175 µmol m−2 s−1, 16 h light/8 h dark at 22/17 °C day/night temperature regime. Supplementary Fig. 1A–E shows no differences in morphology, growth rate, and flowering time among WT, A, O, and AO plants are grown on MS or in potting soil under control conditions. However, AVP1/OsSIZ1 co-overexpressing plants consistently produced more siliques and seeds compared to other genotypes under control conditions. Under salt stress conditions, AVP1/OsSIZ1 co-overexpressing plants were more salt tolerant than OsSIZ1-overexpressing and AVP1-overexpressing plants, and WT plants were the most sensitive to salt stress treatment (Fig. 2 and Supp. Fig. 2). Under normal growth condition (MS alone), there were no apparent differences in root phenotypes (Fig. 2A), whereas in the presence of 125 mM and 150 mM NaCl, AVP1/OsSIZ1 co-overexpressing plants produced significantly longer roots (Fig. 2B,D, and Supp. Fig. 2A).
Analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under salt stress conditions. (A) Phenotypes of wild-type and transgenic plants on MS plate. (B) Phenotypes of wild-type and transgenic plants on MS plate supplemented with 125 mM of NaCl. (C) Phenotypes of wild-type and transgenic plants under salt stress condition (150 mM of NaCl) in soil. (D) Analysis of root length of wild-type and transgenic plants on MS plates shown in A and B above and in Supp. Fig. 2A. (E) Seed yields of wild-type and transgenic plants grown in soil under control and salt stress conditions. Total seed yield per plant grown under normal irrigation (black bar) and salt stress (grey bar) conditions were determined at the end of salt stress treatment. Data are means ± SE (n = 9). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
For studying soil-grown plants under salt stress treatment, seeds were sown into potting soil and grown for three weeks under control conditions, and salt stress was subsequently applied in the amount of 50 mM in the beginning for four days, then increased to 100 mM for four days and 150 mM for four days. After that, regular water was used for irrigation until the end of the experiment. Our soil experiment results show that AO plants out-performed A, O, and WT plants and produced 150% more seed yield compared to WT under salt stress condition (Fig. 2C,E, and Supp. Fig. 2C,D). We also measured the concentration of Na+ and K+ ions in leaves of WT and transgenic plants under control and salt stress conditions, and we found that AO plants irrigated with saline water contained around 20% and 30% more Na+ and K+ ions than WT plants, respectively (Supp. Fig. 3).
Figure 3 shows the response of WT, A, O, and AO to elevated temperature. Figure 3A,B shows the phenotypes of WT and transgenic plants on MS media after moderate heat treatment (i.e., 32 °C) for ten days. AVP1/OsSIZ1 co-overexpressing plants produced significantly longer roots compared to O, A, and WT plants and O had significantly longer roots than A and WT plants. There were no differences in root length between A and WT plants. This was expected as the increased heat tolerance is conferred by overexpression of OsSIZ1, not AVP1. Interestingly, the significant difference in root length observed in AO plants compared to O plants suggests that there is potentially a synergism between AVP1-overexpression and OsSIZ1-overexpression (Fig. 3B). Interestingly, this synergy was not observed when the hypocotyl length of dark-grown plants was analyzed. In this assay, 3-day old seedlings grown on MS plates in darkness were exposed to 45 °C for five h in darkness and then moved to room temperature for another ten days in darkness before the hypocotyl lengths of plants were measured (Fig. 3C). The hypocotyl lengths of AO and O plants were similar, but both lines showed longer hypocotyls compared to A and WT plants (Fig. 3D).
Analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under heat stress conditions. (A) Phenotypes of wild-type and transgenic plants on MS plate exposed to 32 °C for ten days. (B) Root length analysis of plants shown in A. (C) Phenotypes of dark-grown plants on MS plate exposed to 45 °C for 5 h. (D) Hypocotyl length analysis of dark-grown plants at room temperature (black bar) and exposed to 45 °C for 5 h (grey bar). Data are means ± SE (n = 6). (E) Phenotypes of wild-type and transgenic plants grown in soil under heat stress treatment at 37 °C for 5.5 h per day for 40 days. (F) Total seed yields of plants grown under normal growth condition (black bar) and heat stress condition (grey bar). Data are means ± SE (n = 9). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
The performance of soil-grown plants under heat stress was also examined. Seeds were sown into potting soil and grown for three weeks under control conditions and subsequently transferred to a growth chamber where daytime growth temperature was ramped up to 37 °C in the middle of the day for 5.5 h per day for 40 days. No significant differences in growth rate or plant height were observed (Fig. 3E), but AO and O plants produced more shoot branches and more siliques compared to A and WT plants (Fig. 3E), and consequently the highest seed yields (e.g., 110% more in AO plants than in WT plants (Fig. 3F).
Figure 4 shows the response of WT, A, O, and AO plants to water deficit stress. Figure 4A,B shows the phenotype of 7-week old plants after exposure to 2 water deficit stress events. Briefly, plants were grown under control conditions for three weeks followed by a slow-onset water deficit stress for two weeks, irrigation to field capacity; then the second water deficit stress was applied for two weeks. Figure 4A,B shows that AVP1/OsSIZ1 co-overexpressing plants outperformed A, O, and WT plants. Under water deficit stress conditions, AO plants were larger than the other genotypes and had more siliques (Supp. Fig. 4) and showed a 2-fold increase in seed production compared to A and O plants and over 4-fold increase in seed production compared to WT plants (Fig. 4C).
Analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under drought stress condition. (A) Phenotypes of wild-type and transgenic plants grown under reduced irrigation condition. (B) Phenotypes of wild-type and transgenic plants grown in the same pot under reduced irrigation condition. (C) Total seed yields of plants grown under normal growth condition (black bar) and reduced irrigation condition (grey bar). Data are means ± SE (n = 9). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
AVP1/OsSIZ1 co-overexpression results in enhanced tolerance to simultaneous abiotic stress events
We also examined the response of WT, A, O, and AO plants to multiple, simultaneous stress events. Figure 5 shows the growth and seed yield response to multiple stress conditions. Root growth was enhanced in all transgenic lines compared to WT plants grown on MS + 100 mM NaCl and the continuous growth temperature of 32 °C, with AVP1/OsSIZ1 co-overexpressing plants showing over 2-fold increase over A and O and an almost 4-fold increase in root length compared to WT (Fig. 5A,B). Similarly, for soil-grown plants, all transgenic lines produced more above-ground biomass and more seed yield compared to WT plants when grown under heat + salt (Fig. 5C,D and Supp. Fig. 5), heat + water deficit (Fig. 5E,F and Supp. Fig. 6), and salt + water deficit (Fig. 5G,H and Supp. Fig. 7). In all conditions, AVP1/OsSIZ1 co-overexpressing plants produced significantly more biomass and seeds than all genotypes tested (Fig. 5C–H).
Analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under multi-stress conditions. (A) Phenotypes of wild-type and transgenic plants grown under combined heat (32 °C) and salt (100 mM NaCl) stresses for ten days. (B) Root length analysis of plants grown under combined heat and salt stresses as shown in A. (C) Phenotypes of wild-type and transgenic plants grown under combined heat and salt stresses in soil. (D) Total seed yields of plants grown under normal condition (black bar) and combined heat and salt stresses (grey bar). Data are means ± SE (n = 9). (E) Phenotypes of wild-type and transgenic plants grown under combined heat and drought stresses in soil. (F) Total seed yields of plants grown under normal growth condition (black bar) and combined heat and drought stresses (grey bar). Data are means ± SE (n = 9). (G) Phenotypes of wild-type and transgenic plants grown under combined salt and drought stresses in soil. (H) Total seed yields of plants grown under normal condition (black bar) and combined salt and drought stresses (grey bar). Data are means ± SE (n = 9). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
Figure 6 shows the response of WT, A, O, and AO plants to water deficit, salt, and heat stresses (i.e., three-week-old plants were treated with limited irrigation of saline solutions, plus 37 °C for 5.5 h per day until the end of the experiment). Despite severe reductions in total seed yields in all plants due to the harsh conditions created by simultaneous exposure to three stresses, AVP1/OsSIZ1 co-overexpressing plants demonstrated the healthiest phenotype, followed by AVP1-overexpressing and OsSIZ1-overexpressing plants, and WT plants were the worst (Fig. 6A). These differences in growth and appearance were reflected in seed yield with AVP1/OsSIZ1 co-overexpressing plants producing over 160% more seeds than WT, and 31% and 48% more seeds than OsSIZ1-overexpressing and AVP1-overexpressing plants, respectively (Fig. 6B).
Analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under combined drought, heat, and salt stresses. (A) Phenotypes of wild-type and transgenic plants grown under combined heat, salt, and drought stresses in soil. (B) Total seed yields of plants grown under normal growth condition (black bar) and combined heat, salt, and drought stresses (grey bar). Data are means ± SE (n = 9). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
AVP1/OsSIZ1 co-overexpressing plants release more protons into media
Since AVP1/OsSIZ1 co-overexpressing plants displayed better phenotypes under all stress conditions tested, the acidification capacity of these plants might be better than other plants as this should be the property of AVP1-overexpression13. A recent study also showed that the SUMO E3 ligase MdSIZ1 from apple targets MdbHLH104, which modulates the rhizosphere acidification via up-regulation of plasma membrane (PM) H+-ATPase45. We therefore conducted the acidification assay and found that AVP1/OsSIZ1 co-overexpressing plants did have the biggest capacity in acidifying apoplast (see the yellow color in Fig. 7A). AVP1/OsSIZ1 co-overexpressing plants released around 35% more protons into media than AVP1-overexpressing plants. OsSIZ1-overexpressing plants also released more protons into media than WT plants, which might be due to sumoylation of bHLH transcription factor that results in higher PM H+-ATPass-mediated rhizosphere acidification45. As data showed that AVP1/OsSIZ1 co-overexpressing plants have around two-fold increase in the ability to acidify the environment as compared to wild-type plants.
Analysis of root acidification in wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants. (A) Phenotype of seedlings of wild-type and transgenic plants grown on MS plate containing the pH indicator bromocresol purple. The color change from purple to yellow indicates the pH change in the media due to acidification of rhizosphere of plants. (B) Protons released from the roots of wild-type and transgenic plants. Data are means ± SE (n = 5 pools of 10 plants for each pool). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO1 to AO3, three independent AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
Co-overexpression of AVP1/OsSIZ1 significantly alters gene expression under abiotic stresses
To understand the molecular basis of the substantially improved abiotic stress tolerance in AVP1/OsSIZ1 co-overexpressing plants, RNA transcript profiles were generated for WT, A, O, and AO leaf tissues exposed to simultaneous heat and water-deficit stresses. The RNA sequencing data showed that there are 1155 differentially expressed genes (DEGs) in AVP1/OsSIZ1 co-overexpressing plant (AO3) compared to WT plants (Fig. 8A). Among 1226 DEGs in AVP1-overexpressing plants and 2211 DEGs in OsSIZ1-overexpressing plants under combined heat and drought stresses, 628 and 1067 were up-regulated, and 598 and 1144 were down-regulated in AVP1-overexpressing and OsSIZ1-overexpressing plants, respectively (Fig. 8A). The numbers of DEGs in AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing (AO3) plants in comparing to those of WT plants under normal growth condition are presented in Supp. Fig. 8A. The Venn diagrams illustrated in Fig. 8B and Supp. Fig. 8B provide an overview of the distribution of DEGs in transgenic plants in comparing to those of WT plants under combined heat and drought stresses and normal growth conditions. According to these data, 449 and 459 DEGs in AO3 plants vs. WT plants share similar patterns with AVP1-overexpressing and OsSIZ1-overexpressing plants, respectively, while AVP1-overexpressing plants and OsSIZ1-overexpressing plants shared 436 DEGs under combined heat and drought stresses. A total of 200 DEGs were up- or down-regulated in AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under combined heat and drought stress conditions (Fig. 8B). Under normal growth condition (i.e., optimum temperature and regular irrigation), AO3 shared 428 and 403 DEGs with AVP1-overexpressing plants and OsSIZ1-overexpressing plant, respectively, and 142 DEGs were shared in all three plants (Supp. Fig. 8B).
RNA sequencing analysis of wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under combined heat and drought stresses. (A) Number of differentially expressed genes in transgenic plants under combined heat and drought stresses. Grey bars represent down-regulated genes, and black bars represent up-regulated genes. (B) Venn diagram of differentially expressed genes in transgenic plants vs. wild-type plants under combined heat and drought stresses. (C) Heatmap of 44 stress-related differentially expressed genes. WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO3, AVP1/OsSIZ1 co-overexpressing plants. DEGs, differentially expressed genes; ER, environmental response; TF, transcription factor; NT, nutrient transfer; OR, oxidation-reduction.
Among those shared 200 DEGs, we selected 44 DEGs (Supp. Table 1) with the fold-of-change >2, including 21 transcription factor (TF) genes, 13 environmental response (ER) associated genes, 6 nutrient transfer (NT) genes, and 4 oxidative stress response (OR) genes for further analysis (Fig. 8C). Under combined heat and water-deficit stresses, heat shock protein (HSP) genes (e.g., HSP70 and HSP101) were up-regulated in AO3 plant, with significantly higher levels of transcripts (log2-fold >7.5) (Supp. Table 1). A member of small heat shock proteins (sHSPs), HSP17.4, was significantly up-regulated in AO3 plant (log2-fold of 10.3) in comparing to that of WT plants (log2-fold of 8.8). These data are consistent with the functions of HSPs as chaperones in stabilizing proteins and re-folding damaged proteins under stress condition46. The transcript levels of several TF genes (i.e., NAC19, HSFA2, and ABF3) were also altered. Since many TFs are involved in regulating the expression of stress-related genes, any changes in the expression pattern of TF genes will likely result in higher stress tolerance in transgenic plants. The RNA sequencing results also exhibited that, the gene RD28 was highly expressed (log2-fold > 3.6) in AO3 plants in comparing to that in WT plants under combined heat and drought stresses, however, the transcript of ERD4 was slightly downregulated in AO3 plant. Notably, the transcript of NCED3 was much higher in AO3 plants than those of WT, AVP1-overexpressing, and OsSIZ1-overexpressing plants under combined heat and drought stresses, indicating that ABA-dependent pathway might be involved in the improved drought tolerance in AVP1/OsSOZ1 co-overexpressing plant47.
Quantitative real time-PCR analysis confirmed RNA sequencing results
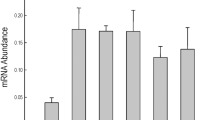
To confirm the RNA sequencing data, we randomly selected nine genes and conducted quantitative real-time (qRT)-PCR using the gene-specific primers. The Actin 8 was used as the internal reference gene. The results of the qRT-PCR experiments showed a highly correlated pattern of transcript levels with the RNA sequencing data (Fig. 9). It appears that the transcript levels of most selected genes were already at much higher levels, which might enable transgenic plants to deal with abiotic stresses more effectively in the very beginning, consequently reducing damages caused by the stresses. Other than the heat shock factor genes and small HSP genes (e.g., HSFA2 and HSP17), most transcripts remain at levels higher than that of WT plants (Fig. 9).
Quantitative real-time PCR (qRT-PCR) analysis of nine stress-related genes in wild-type, AVP1-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under normal growth condition (black bar) and combined heat and drought stresses (grey bar). The Arabidopsis gene Actin8 was used as the internal reference gene. The relative transcript level was calculated using the 2−ΔΔCt method. Data are means ± SE (n = 3). WT, wild-type plant; A, AVP1-overexpressing plant; O, OsSIZ1-overexpressing plant; AO3, AVP1/OsSIZ1 co-overexpressing plants. Samples denoted by different letters are significantly different (P < 0.05, ANOVA, Tukey correction).
Discussion
Since water deficit, heat, and salinity often occur simultaneously, both in nature and crop production settings in arid and semi-arid regions of the world, creating germplasm with enhanced tolerance to these stresses is of great value. In this study, we demonstrate that co-overexpression of the Arabidopsis vacuolar H+-pyrophosphatase gene AVP1 and the rice SUMO E3 ligase gene OsSIZ1 significantly enhances plant tolerance to water deficit, heat, and salt stress in transgenic Arabidopsis plants. The increased tolerances to abiotic stresses are significantly higher than those achieved by overexpression of AVP1 or OsSIZ1 alone, confirming our hypothesis that co-overexpression of AVP1 and OsSIZ1 would further increase tolerance to abiotic stresses, which provides a proof-of-concept that might be applicable to crops, which could potentially lead to substantially increased crop yields in the future.
Previously, Gaxiola et al. showed increased tolerance to drought and salt by overexpressing AVP1 in transgenic Arabidopsis and tomato plants, which was attributed to increased sequestration and accumulation of Na+ in vacuoles and increased auxin polar transport12,13,14, thereby leading to robust root development. The sequestration of more Na+ into the vacuole is likely due to increased activity of Na+/H+ antiporters on tonoplast membranes in response to higher H+ gradient in vacuole in AVP1-overexpressing plants48. Overexpression of OsSIZ1 in transgenic bentgrass and cotton led to increased heat tolerance and drought tolerance in transgenic plants33,34. Furthermore, sumoylation appears to help plants to withstand stress conditions via several pathways like protecting the electron transport system, which reduces the negative impacts of stress on photosynthesis49. When AVP1 and OsSIZ1 were co-overexpressed in Arabidopsis, transgenic plants performed significantly better than AVP1-overexpressing plants and OsSIZ1-overexpressing plants under all stress conditions tested. Our data indicate that AVP1/OsSIZ1 co-overexpressing plants outperformed other plants under salt stress (Fig. 2), heat stress (Fig. 3), drought stress (Fig. 4), combined heat and salt (Fig. 5C,D), combined heat and drought (Fig. 5E,F), combined drought and salt (Fig. 5G,H), and combined heat, drought and salt (Fig. 6). The improved seed yields under these abiotic stress conditions might be due to simple addition of increased beneficial traits of overexpression of AVP1 and OsSIZ1, and the improved abiotic stress tolerance is significantly better than what has been achieved in prior engineering efforts for abiotic stress tolerance, therefore, this discovery has a far-reaching implication in genetic engineering crops for yield improvement. Our previous work on transgenic cotton plants showed that AVP1-overexpression routinely resulted in approximately 20% higher fiber yields than WT cotton when grown under rainfed conditions15. Similarly, OsSIZ1-overexpressing cotton routinely produced 25% higher fiber yields than WT cotton under rainfed conditions34. Based on the research reported here, we predict that AVP1/OsSIZ1 co-overexpressing cotton would produce fiber yields that could exceed the single gene overexpressing cotton under rainfed and low input production scenarios and represents an opportunity to stabilize yields in these highly variable and rapidly expanding production regions.
Since salinity imposes water deficit in plant cells and inhibits many important enzymes in plant cellular metabolisms, it is detrimental to plants. Furthermore, when salinity is combined with drought and/or heat, it becomes more dangerous to plant growth and development4,50. Our AVP1/OsSIZ1 co-overexpressing plants display significantly increased tolerance to multiple stresses, which would allow us to employ this technology to reduce crop losses caused by environmental stresses in the future. In addition to salt and drought tolerances, AVP1-overexpressing plants showed an increased root acidification13. AVP1-overexpressing plants were shown to have increased H+ extrusion from root cells as a function of enhanced H+-ATPase pumps on plasma membrane51. This provides a great advantage to plants in uptaking nutrients like phosphate, particularly if grown under phosphate deprived condition16. The amount of protons released by AVP1-overexpressing plants during the day was found to be approximately 40% higher than that of WT plants51, which results in higher sugar metabolism and ATP synthesis in those transgenic plants. The study by Zhou et al.45 shows that overexpression of MdSIZ1 in Arabidopsis causes in improved growth and chlorophyll production as well as enhanced proton release to medium compared to WT, indicating that SUMO E3 ligase is involved in the up-regulation of PM H+-ATPase. In agreement with these studies, we found that AVP1/OsSIZ1 co-overexpressing plants even have higher capacity in acidifying the rhizosphere (Fig. 7A), hinting at the possibility that the improved growth and yield could be related to better absorption of nutrients, increased photo-assimilate production and sucrose mobility in AVP1/OsSIZ1 co-overexpressing plants.
To better understand the underlying molecular mechanism of substantially increased stress tolerance in AVP1/OsSIZ1 co-overexpressing plants, we conducted a transcriptome analysis of AVP1/OsSIZ1 co-overexpressing plants and non-transgenic plants grown under normal growth and combined heat and drought stress conditions. As expected, transcript levels for HSP genes, i.e., HSP70, HSP101, and HSP17, are higher in AVP1/OsSIZ1 co-overexpressing plants under combined drought and heat stresses (Fig. 8). Heat shock transcription factors (HSFs) control the expression of HSPs52. Our RNA sequencing data showed that the transcripts of HSFA2 and HSFB4 were at higher levels in AVP1/OsSIZ1 co-overexpressing plants in comparing to WT plants under normal growth conditions. It should be noted that the transcript levels of a large number of transcription factor genes were altered in AVP1/OsSIZ1 co-overexpressing plants under combined heat and drought stress conditions. Previous studies illustrated a different pattern of overlap among transcripts of abiotic stress response related genes53,54. In our study, however, we found a considerable overlap between the transcripts altered in AVP-overexpressing, OsSIZ1-overexpressing, and AVP1/OsSIZ1 co-overexpressing plants under combined heat and drought stress conditions (Fig. 8B). Plants exposed to water deprivation accumulate a variety of osmoprotectants such as proline and glycine-betaine55. As it was previously reported by Peng et al.56, the transcript level of the proline dehydrogenase 1 gene (POX1), was down-regulated under drought stress condition and the decrease was more significant in AVP1-overexpressing plants in comparing to that of WT plants.
Transcripts of several drought stress-related genes were also altered in plants exposed to combined heat and drought stresses. Among those, the transcript levels of RD28 and DR4 were at higher levels in AVP1/OsSIZ1 co-overexpressing plants in comparing with those of WT plants, while the transcript of ERD10 was at a lower level in AVP1-overexpressing and AVP1/OsSIZ1 co-overexpressing plants. Previous studies in peanut showed that the transcript level of NCED1 that encodes the 9-cis-epoxycarotenoid dioxygenase 1 was increased under drought condition57. We found that the transcript level of NCED3 was higher in AVP1-overexpressing plants, suggesting that ABA might play a role in the higher stress tolerance of AVP1/OsSIZ1 co-overexpressing plants under combined heat and drought stress conditions.
The results obtained in RNA sequencing analysis were validated by qRT-PCR analysis (Fig. 9), and we found a high correlation in the transcript levels of different genes between qRT-PCR and RNA sequencing studies. It is clear that further analysis of AVP1/OsSIZ1 co-overexpressing plants at gene expression and metabolic levels using transcriptome profiling and bioinformatics tools will continue to give us hints at the molecular mechanism of the immensely improved abiotic stress tolerance observed in AVP1/OsSIZ1 co-overexpressing plants.
Conclusion
The detrimental effects of combined abiotic stresses on agricultural production demonstrate the need to create crops that can withstand multiple stresses. In this study, we show that co-overexpression of AVP1 and OsSIZ1 in Arabidopsis plants could dramatically improve plant performance under multiple stress conditions, especially under combined heat, drought, and salt stresses. If this result could be replicated in staple crops such as rice, maize, wheat, soybeans, significantly increased yields would be expected for rainfed and low-input agricultural production systems, which may lead to another green revolution, stabilizing regional agriculture-based economies, and keeping millions of people fed and clothed.
Materials and Methods
Plasmid construction and arabidopsis transformation
The p35S::AVP1/pUbi::OsSIZ1 construct harboring NptII (kanamycin resistance gene) was cloned into the pBI121 based binary vector, and introduced into Arabidopsis (ecotype Columbia) using Agrobacterium-mediated floral dip method44. The rice SUMO E3 ligase gene OsSIZ1 and the Arabidopsis vacuolar pyrophosphatase gene AVP1 were under the control of the dual 35S promoter and the maize ubiquitin gene promoter, respectively. Transgenic plants were selected by screening seeds on Murashige and Skoog58 (MS) media supplemented with 30 µg/ml of kanamycin. A total of 40 independent putative transgenic plants were obtained on kanamycin plates and were verified by using PCR with AVP1 and OsSIZ1 gene-specific primers. Homozygous transgenic plants were obtained at T3 generation and then used for all experiments.
Plant materials and growth conditions
Wild-type Arabidopsis plants (WT), and two reference lines, OsSIZ1-overexpressing plant59 (O), AVP1-overexpressing plant (A) (generated in our lab), and three independent AVP1/OsSIZ1 co-overexpressing plants (AO1 to AO3) were used in all experiments. Plants were grown in LC1 mix in a growth chamber that was set at 21 °C with a photoperiod of 16 h light/8 h dark. For experiments conducted on MS plates, seeds were surface sterilized using 70% (v/v) ethanol and 15% (v/v) commercial bleach followed by rinsing with sterile distilled water four times, then stratified at 4 °C for three days before sowing seeds on MS plates with or without salts. For experiments conducted in soil, seed collectors were used to avoiding seed loss during harvesting, and all seed yield data were the amount of seeds produced per plant.
RNA and DNA blot analyses
Total RNAs were extracted from leaves of WT and transgenic plants using the TRIzol reagent (Invitrogen, Carlsbad, CA). Ten µg of total RNAs from WT and transgenic plants were separated on 1.2% (W/V) agarose gel and transferred onto BioTrans (+) TM nylon membranes (Ambion, USA). The RNA blot analysis was carried out by hybridizing the membrane with P32-labelled gene-specific probes for OsSIZ1, AVP1, and Actin8, respectively, under the condition as described by Church and Gilbert60. A full-length cDNA for AVP1 was amplified by PCR using the forward and reverse primers of AVP1_F and AVP1_R, and an Actin8 cDNA was amplified using forward and reverse primers of Actin8_F and Actin8_R, respectively. An OsSIZ1 cDNA fragment was amplified as described by Mishra et al.34,59 The PCR amplified DNA fragments were used to make P32-labelled gene-specific probes, using the DECAprimeTM II DNA Labeling Kit (Life Technology, NY, USA).
Genomic DNAs were isolated from Arabidopsis leaves using the cetyltrimethylammonium bromide method with minor modifications61. Fifteen micrograms of genomic DNAs from WT and transgenic plants were digested overnight with Hind III (NEB, USA), then the digested DNAs were run on 0.8% agarose gel followed by transferring it onto a BioTrans (+) TM nylon membrane. The DNA blotting hybridization was performed as previously described by Hu et al.62
The oligonucleotide primers are listed below:
AVP1_F: ATGGGCGAGCTCGGTACC
AVP1_R: GAGAGACTGGTGATTTGCGGAC
Actin8_F: TCACCACAACAGCAGAGCGGG
Actin8_R: GGACCTGCCTCATCATACTCGG
Salt stress treatment
Salt stress treatments were conducted on MS plate and in soil, respectively. Arabidopsis seeds of WT and transgenic plants were surfaced sterilized in 15% bleach once, 70% ethanol once, then rinsed with sterile water four times, then sown on MS plates and kept at 4 °C for three days for stratification. These seeds were allowed to germinate vertically for three days before they were transferred to another MS plate supplemented with 150 mM of NaCl to grow for seven more days, and the number of green cotyledons was counted, and the root lengths were measured. In a different experiment, seeds were directly plated on MS plates supplemented with 125 mM of NaCl. Ten days later the root length and the number of lateral roots were measured. WT plants were grown on MS plates for ten days under the same condition as treated plants. For salt stress treatment in soil, plants were allowed to grow under normal growth condition for three weeks, then saline solution of 50 mM of NaCl was used to irrigate plants (twice in 4 days), then the salt concentration was incrementally increased to 100 mM and 150 mM (each concentration twice, and 4 days at each concentration). After that, plants were irrigated with regular water until the harvest. Seed yields, silique numbers, and shoot heights were measured after salt treatment. Control plants were grown separately in the same growth chamber and irrigated with regular water. For each experiment, three biological replicates for genotype were used, and all experiments were repeated at least three times.
Drought stress treatment
Three-week-old plants under normal growth condition were fully irrigated, then irrigation was stopped for two weeks. After that, plants were fully irrigated again; then in the next two weeks, no irrigation was applied. After two rounds of drought treatment, plants were harvested. The seed yields, silique numbers, and heights of plants were measured. Three technical repeats with three biological replicates were used.
Heat stress treatment
Heat tolerance test was conducted on MS plates. Arabidopsis seeds were surface sterilized as described above and sowed on MS plates, and left at 4 °C for three days for stratification, then kept at 32 °C to grow vertically for ten days. After that, root lengths were measured. In addition, the heat tolerance test was conducted with soil grown plants. In this assay, three weeks old plants grown in soil under normal growth condition (i.e., 25 °C and regular irrigation) were transferred into a growth chamber that was set at 37 °C for 5.5 hours per day until the end of the experiment, and seed yields were analyzed. Plants were irrigated with normal water three times a week during the heat treatment.
Combined heat and drought stresses
Plants were grown in pots in a growth chamber that was set at a photoperiod of 16 h of light and 8 h of darkness per day at 22 °C with regular irrigation for three weeks. Then, plants were fully irrigated before transferring to a growth chamber that was set with the same photoperiod, but with a different temperature regime: 37 °C for 5.5 h from 12:00 pm to 5:30 pm and 22 °C for the rest of time. The irrigation was reduced to half the amount of water used for control plants every five days. For each experiment, three biological replicates for each genotype were used and experiments were repeated three times.
Combined heat and salt stresses
Arabidopsis seeds were surface sterilized and sown on MS plates supplemented with 100 mM of NaCl. Plates were kept at 32 °C, and root lengths were measured, and the phenotype was documented after ten days. For plants grown in soil, three weeks old plants were transferred to a growth chamber that was set at 37 °C for 5.5 h and 22 °C for 18.5 h per day for 25 days. Starting the day after transferring, plants were irrigated with 50 mM of NaCl, which was repeated on the third day of treatment, and followed by 100 mM of NaCl on the 6th and the 9th days of treatment. After that, plants were irrigated with regular water every other day until the end of the experiment. Three biological replicates for each genotype were used, and experiments were repeated three times.
Combined drought and salt stresses
Three weeks old plants grown under normal growth condition were irrigated with half the amount of water used for control plants supplemented with 50 mM of NaCl, which was repeated on the 5th day, then followed by 100 mM of NaCl on the 10th and the 15th days. After that, plants were irrigated with water once every five days until the end of the experiment and seed yields were analyzed. Three biological replicates for each genotype were used, and experiments were repeated three times.
Combined drought, heat and salt stresses
Plants were grown under normal growth conditions for three weeks, then they were moved to a chamber that was set at 37 °C for 5.5 h and 22 °C for 18.5 h per day, irrigated with half the amount of water used for control plants supplemented with 50 mM NaCl on the 1st and the 4th day, and thereafter the concentration of salt increased to 100 mM NaCl in the second week. This was followed by irrigation with half of the amount of normal water used for control plants twice a week until the end of the experiment. Three biological replicates for each genotype were used, and the experiments were repeated three times.
Rhizosphere acidification assay
The amount of protons released by the roots of Arabidopsis plants to the total root fresh weight was calculated using the method described by Pizzio et al.51. Surface sterilized seeds of Arabidopsis were sown on MS plates (pH 5.7) and grown vertically for three days. Thereafter seedlings were transferred to MS plates (pH 6.5) containing 0.004% pH indicator bromocresol purple for 7–10 days. Plates were monitored regularly for color change. The color change from purple to yellow indicates the pH change in the media due to acidification of rhizosphere of plants.
RNA sequencing and differential gene expression analysis
Comparative transcriptome analysis was performed using Arabidopsis plants grown under combined heat and drought stresses and normal growth conditions. Total RNAs were extracted from WT, AVP1-overexpressing, OsSIZ1-overexpressing, and one of the AVP1/OsSIZ1 co-overexpressing (i.e., AO3) plants using the SpectrumTM Plant Total RNA kit (Sigma, USA). High-quality RNA samples were used for cDNA library construction using the NEB Next mRNA Library Prep Master Mix Set for Illumina (NEB, USA) in accordance with the manufacturer instruction. The cDNA libraries were then sequenced with the Illumina HiSeq. 2500 platform (Illumina, Inc., San Diego, CA, USA) using paired-end technology. The clean reads from all samples were mapped onto the reference Arabidopsis genome using Tophat2 (version 2.2.5)63. Transcript levels were measured using EBSeq software64 after normalizing the number of reads with reads per kilo-base of transcript per million mapped reads (RPKM) values from each gene. DEGs were identified by setting a cutoff p-value < 0.001 and the false discovery rate (FDR) < 0.01. DEGs with a fold-change >2 were considered as up-regulated, and those with <0.5 were considered as down-regulated.
Real time quantitative PCR analysis
The validation of DEG data generated from RNA sequencing was carried out with quantitative real time PCR (RT-qPCR). Among those DEGs, we selected 9 DEGs related to heat and/or drought stress response. The primers were designed using Primer Premier 5 (PREMIER Biosoft, CA, USA) (Supplementary Table 2). The reverse transcription was carried out using 1 μg of DNase-treated total RNAs and the SuperScriptTM VILOTM Master Mix (Invitrogen, USA). The cDNA templates were amplified with Applied Biosystems 7500 Real-Time PCR detection system and the SYBR Green JumStartTM Taq ReadyMixTM (Sigma, USA). The Arabidopsis Actin8 was used as the internal reference gene, and the relative transcript level of each gene was calculated using the 2−ΔΔCt method65. Three biological replicates were used for all RT-qPCR reactions.
Statistical analysis
Tukey’s statistical method was performed in all pairwise comparison across different genotypes at the significant level of p = 0.05.
References
Food and Agriculture Organization of the United Nations (FAO). How to feed the world in 2050 (FAO, 2009).
Edgerton, M. D. Increasing crop productivity to meet global needs for feed, food, and fuel. Plant Physiol. 149, 7–13 (2009).
Hu, H. & Xiong, L. Genetic engineering and breeding of drought-resistant crops. Annu. Rev. Plant Biol. 65, 715–741 (2014).
Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 11, 15–19 (2006).
Parida, A. K. & Das, A. B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 3, 324–349 (2005).
Blumwald, E. Sodium transport and salt tolerance in plants. Curr. Opin. Cell Biol. 4, 431–434 (2000).
Boyer, J. S. Plant Productivity and Environment. Science 218, 443–448 (1982).
Munns, R. & Testers, M. Mechanisms of salt tolerance. Annu. Rev. Plant Biol. 59, 651–681 (2008).
Shi, H., Lee, B.-h, Wu, S.-J. & Zhu, J.-K. Overexpression of a plasma membrane Na+/H+ antiporter gene improves salt tolerance in Arabidopsis thaliana. Nat Biotech 1, 81–85 (2003).
Apse, M. P., Aharon, G. S., Snedden, W. A. & Blumwald, E. Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 285, 1256–1258 (1999).
Gaxiola, R. A., Fink, G. R. & Hirschi, K. D. Genetic manipulation of vacuolar proton pumps and transporters. Plant Physiol. 129, 967–973 (2002).
Gaxiola, R. A. et al. Drought- and salt-tolerant plants result from overexpression of the avp1 H+-pump. Proc. Natl. Acad. Sci. USA 20, 11444–11449 (2001).
Li, J. et al. Arabidopsis H+-ppase avp1 regulates auxin-mediated organ development. Science 5745, 121–125 (2005).
Park, S. et al. Up-regulation of a H+-pyrophosphatase (H+-ppase) as a strategy to engineer drought-resistant crop plants. Proc. Natl. Acad. Sci. USA 52, 18830–18835 (2005).
Pasapula, V. et al. Expression of an Arabidopsis vacuolar H+-pyrophosphatase gene (AVP1) in cotton improves drought- and salt tolerance and increases fibre yield in the field conditions. Plant Biotech J 1, 88–99 (2011).
Yang, H. et al. Over-expression of the Arabidopsis proton-pyrophosphatase AVP1 enhances transplant survival, root mass, and fruit development under limiting phosphorus conditions. J. Exp. Bot. 12, 3045–53 (2014).
Yang, H. et al. Enhanced phosphorus nutrition in monocots and dicots over-expressing a phosphorus-responsive type I H+-pyrophosphatase. Plant Biotech J 6, 735–745 (2007).
Pei, L. et al. Overexpression of Thellungiella halophila H+-pyrophosphatase gene improves low phosphate tolerance in maize. PLoS ONE 8, 43501 (2012).
Lv, S. et al. Overexpression of an H+-ppase gene from Thellungiella halophila in cotton enhances salt tolerance and improves growth and photosynthetic performance. Plant Cell Physiol. 8, 1150–1164 (2008).
Paez-Valencia, J. et al. Enhanced proton translocating pyrophosphatase activity improves nitrogen use efficiency in romaine lettuce. Plant Physiol. 3, 1557–1569 (2013).
Lv, S.-L. et al. Overexpression of Thellungiella halophila H+-ppase (TsVP) in cotton enhances drought stress resistance of plants. Planta 4, 899–910 (2009).
Li, Z., Baldwin, C. M., Hu, Q., Liu, H. & Luo, H. Heterologous expression of Arabidopsis H+-pyrophosphatase enhances salt tolerance in transgenic creeping bentgrass (Agrostis stolonifera l.). Plant, Cell Environ. 2, 272–289 (2010).
Li, B., Wei, A., Song, C., Li, N. & Zhang, J. Heterologous expression of the TsVP gene improves the drought resistance of maize. Plant Biotech J 2, 146–159 (2008).
Qin, H. et al. Expression of the Arabidopsis vacuolar H+-pyrophosphatase gene AVP1 in peanut to improve drought and salt tolerance. Plant Biotech Rep. 3, 345–355 (2013).
Kumar, T., Khan, M. R., Abbas, Z. & Ali, G. M. Genetic improvement of sugarcane for drought and salinity stress tolerance using Arabidopsis vacuolar pyrophosphatase (AVP1) gene. Mol. Biotechnol. 3, 199–209 (2014).
Arif, A., Zafar, Y., Arif, M. & Blumwald, E. Improved growth, drought tolerance, and ultrastructural evidence of increased turgidity in tobacco plants overexpressing Arabidopsis vacuolar pyrophosphatase (AVP1). Mol. Biotechnol. 2, 379–392 (2013).
Zhao, F.-Y., Zhang, X.-J., Li, P.-H., Zhao, Y.-X. & Zhang, H. Co-expression of the Suaeda salsa SsNHX1 and Arabidopsis AVP1 confer greater salt tolerance to transgenic rice than the single SsNHX1. Mol. Breed. 4, 341–353 (2006).
Schilling, R. K. et al. Expression of the Arabidopsis vacuolar H+-pyrophosphatase gene (AVP1) improves the shoot biomass of transgenic barley and increases grain yield in a saline field. Plant Biotech J 3, 378–386 (2014).
Battisti, D. S. & Naylor, R. L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science 323, 240–244 (2009).
Ainsworth, E. A. & Ort, D. R. How do we improve crop production in a warming world? Plant Physiol. 154, 526–530 (2010).
Vile, D. et al. Arabidopsis growth under prolonged high temperature and water deficit: Independent or interactive effects? Plant, Cell Environ. 4, 702–718 (2012).
Rivero, R. M. et al. The combined effect of salinity and heat reveals a specific physiological, biochemical and molecular response in tomato plants. Plant, Cell Environ. 5, 1059–1073 (2014).
Li, Z. et al. Heterologous expression of OsSIZ1, a rice sumo E3 ligase, enhances broad abiotic stress tolerance in transgenic creeping bentgrass. Plant Biotech J 4, 432–445 (2013).
Mishra, N. et al. Overexpression of the rice sumo E3 ligase gene OsSIZ1 in cotton enhances drought and heat tolerance, and substantially improves fiber yields in the field under reduced irrigation and rainfed conditions. Plant Cell Physiol. 4, 735–746 (2017).
Miura, K. & Hasegawa, P. M. Sumoylation and other ubiquitin-like post-translational modifications in plants. Trends Cell Biol. 4, 223–232 (2010).
Castro, P. H. et al. SIZ1-dependent post-translational modification by sumo modulates sugar signaling and metabolism in Arabidopsis thaliana. Plant Cell Physiol. 12, 2297–2311 (2015).
Catala, R. et al. The Arabidopsis E3 SUMO ligase SIZ1 regulates plant growth and drought responses. Plant Cell 9, 2952–2966 (2007).
Miura, K. et al. SIZ1-mediated sumoylation of ICE1 controls CBF3/DREB1a expression and freezing tolerance in Arabidopsis. Plant Cell 4, 1403–1414 (2007).
Miura, K. et al. The Arabidopsis SUMO E3 ligase SIZ1 controls phosphate deficiency responses. Proc. Natl. Acad. Sci. USA 21, 7760–7765 (2005).
Park, H. et al. SUMO and Sumoylation in plants. Mol Cells 4, 305–316 (2011).
Yoo, C. Y. et al. SIZ1 small ubiquitin-like modifier E3 ligase facilitates basal thermotolerance in Arabidopsis independent of salicylic acid. Plant Physiol. 4, 1548–1558 (2006).
He, C. et al. Expression of an Arabidopsis vacuolar sodium/proton antiporter gene in cotton improves photosynthetic performance under salt conditions and increase fiber yield in the field. Plant Cell Physiol. 46, 1848–1854 (2005).
Zhu, X. et al. The yield difference between wild-type cotton and transgenic cotton that expresses IPT depends on when water-deficit stress is applied. Sci. Rep. 8, 2538 (2018).
Clough, S. J. & Bent, A. F. Floral dip: A simplified method for agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 6, 735–743 (1998).
Zhou, L. J. et al. The SUMO E3 ligase MdSIZ1 targets MdbHLH104 to regulate plasma memberane H+-ATPase activity and iron homeostasis. Plant Physiol. 179, 88–106 (2019).
Georgopoulos, C. & Welch, W. J. Role of the major heat shock proteins as molecular chaperones. Annu. Rev. Cell Biol. 9, 601–634 (1993).
Iuchi, S. et al. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J 27, 325–333 (2001).
Gaxiola, R. A. et al. The Arabidopsis thaliana proton transporters, AtNHX1 and AVP1, can function in cation detoxification in yeast. Proc. Natl. Acad. Sci. USA 4, 1480–1485 (1999).
Benlloch, R. & Lois, M. L. Sumoylation in plants: mechanistic insights and its role in drought stress. J. Ept. Bot. 69, 4539–4554 (2018).
Hasegawa, P. M., Bressan, R. A., Zhu, J.-K. & Bohnert, H. J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1, 463–499 (2000).
Pizzio, G. A. et al. Arabidopsis type I proton-pumping pyrophosphatase expresses strongly in phloem, where it is required for pyrophosphate metabolism and photosynthate partitioning. Plant Physiol. 4, 1541–1553 (2015).
Morimoto, R. I. Regulation of the heat shock transcriptional response: cross talk between a family of heat shock factors, molecular chaperones, and negative regulators. Genes & Dev. 12, 3788–3796 (1998).
Rizhsky, L., Liang, H., Shuman, J., Shulaev, V. & Davletova, S. When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol. 134, 1683–1696 (2004).
Seki, M. et al. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J. 32, 279–292 (2002).
Hoekstra, F. A., Golovina, E. A. & Buitink, J. Mechanisms of plant desiccation tolerance. Trends Plant Sci. 6, 431–438 (2001).
Peng, Z., Lu, Q. & Verma, D. P. Reciprocal regulation of delta 1-pyrroline-5-carboxylate synthetase and proline dehydrogenase genes controls proline levels during and after osmotic stress in plants. Mol Gen Genet. 253, 334–341 (1996).
Li, X. et al. Identification of rapidly induced genes in the response of peanut (Arachis hypogaea) to water deficit and abscisic acid. BMC Biotechnol. 14, 58–66.
Murashige, T. & Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 3, 473–497 (1962).
Mishra, N., Srivastava, A. P., Esmaeili, N., Hu, W. & Shen, G. Overexpression of the rice gene OsSIZ1 in Arabidopsis improves drought-, heat-, and salt-tolerance simultaneously. PLoS ONE 13(8), e0201716 (2018).
Church, G. M. & Gilbert, W. Genomic sequencing. Proc. Natl. Acad. Sci. USA 7, 1991–1995 (1984).
Paterson, A. H., Brubaker, C. L. & Wendel, J. F. A rapid method for extraction of cotton (gossypium spp.) genomic DNA suitable for RFLP or pcr analysis. Plant Mol. Biol. Rep. 2, 122–127 (1993).
Hu, R., Zhu, Y., Shen, G. & Zhang, H. Tap46 plays a positive role in the abscisic acid insensitive5-regulated gene expression in Arabidopsis. Plant Physiol. 2, 721–734 (2014).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578 (2012).
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods. 5, 621–628 (2008).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 25, 402–408 (2001).
Acknowledgements
This project was supported by USDA-Ogallala Aquifer Program, USDA CRIS 3096-21000-019-00D, and it was also partially supported by a grant to Xiaojie Yang (2016ZX 08005001-005 from the National Transgenic Major Project of China), a grant to Weiping Fang (CARS-15-07 from the Technical System of National Cotton Industry), and grants to Guoxin Shen (2010DS700124-ZZ1809 from the State Key Laboratory Breeding Base for Zhejiang Sustainable Pest and Disease Control, and CYNY 2018005 from the Key laboratory of Creative Agriculture, Ministry of Agriculture of China). We thank Inosha Wijewardene for reading the manuscript.
Author information
Authors and Affiliations
Contributions
N.E., L.S. and H.Z. conceived and designed the experiments; X.Y. and W.F. performed RNA sequencing; N.E., X.Z. and Y.C. performed the experiments; N.E., G.S., P.P., and H.Z. analyzed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Esmaeili, N., Yang, X., Cai, Y. et al. Co-overexpression of AVP1 and OsSIZ1 in Arabidopsis substantially enhances plant tolerance to drought, salt, and heat stresses. Sci Rep 9, 7642 (2019). https://doi.org/10.1038/s41598-019-44062-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44062-0
This article is cited by
-
Functional and structural asymmetry suggest a unifying principle for catalysis in membrane-bound pyrophosphatases
EMBO Reports (2024)
-
‘Omics’ approaches in developing combined drought and heat tolerance in food crops
Plant Cell Reports (2022)
-
Modulation of photosynthesis and other proteins during water–stress
Molecular Biology Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.