Abstract
Olfactory perception determines food selection behavior depending on energy homeostasis and nutritional status. The mechanisms, however, by which metabolic signals in turn regulate olfactory perception remain largely unclear. Given the evidence for direct insulin action on olfactory neurons, we tested olfactory performance (olfactory threshold, olfactory discrimination) in 36 subjects of normal- and overweight after administration of three different insulin doses (40 I.U., 100 I.U., 160 I.U.) or corresponding placebo volume in a within-subject design. Poor peripheral insulin sensitivity as quantified by HOMA-IR in baseline condition and increases in systemic insulin levels reactive to intranasal administration predicted poor olfactory performance. In contrast, intranasal insulin enhanced odor perception with a dose-dependent improvement of olfactory threshold. These findings indicate a new diametric impact of insulin on olfactory perception depending on peripheral or central availability.
Similar content being viewed by others
Introduction
The sense of smell impacts food selection behavior. The olfactory system does not only help to identify the chemical composition of food but also serves as an internal sensor for the nutritional status1,2. Recent observations that odor perception even directly impacts central nervous circuitries3,4 and peripheral metabolism5 have clearly emphasized the relevance of the olfactory system for energy homeostasis, but the underlying mechanisms are still poorly understood.
Olfactory neurons interact with central nervous circuitries regulating food intake and energy expenditure6. Interestingly, already the smell of hidden food stimuli activates AgRP and POMC neurons in the hypothalamus3,4 suggesting a strong influence of olfactory inputs on hypothalamic regulation of energy homeostasis. Olfactory sensitivity, in turn, is dynamically modulated by the nutritional status. Fasting enhances olfactory acuity, while satiation attenuates olfactory performance7,8,9,10,11. In long term, overconsumption of high fat food causes a loss of olfactory sensory neurons, impairs olfactory functioning and alters olfaction driven behavior12,13. However, it remains unclear how odor perception is modulated by global energy homeostasis.
Insulin is a key candidate for mediating this interaction. Secreted after food intake, blood insulin levels dynamically reflect body fuel availability. In fact, the highest density of central insulin receptors and the highest insulin concentration are found on the olfactory bulb14,15,16. The transport across the blood brain barrier is higher in this area compared to the rest of the brain17 suggesting direct modulation of olfactory signaling by insulin. Indeed, acute euglycaemic hyperinsulinaemia deteriorates olfactory performance18. A similar effect was observed after intracerebroventricular insulin application in rats19. Intranasal insulin application in humans, however, provided so far inconsistent results: While decreased olfactory performance was reported in normosmic subjects20, better olfactory sensitivity was demonstrated after intranasal insulin administration in anosmic subjects21 and hyposmic subjects22.
Here, we investigated the role of peripheral insulin sensitivity, intranasal insulin and reactive blood insulin changes after intranasal administration in the regulation of olfactory performance in humans. To consider variability in peripheral insulin sensitivity, we recruited a sample of healthy- and overweight subjects (BMI 20–30 kg/m2). Given the evidence that insulin sensitivity is a better indicator for the metabolic state than body weight23,24, and that the homeostasis model assessment of insulin resistance (HOMA-IR) is a good proxy for peripheral insulin sensitivity, we hypothesized that poor insulin sensitivity assessed by HOMA-IR will predict poor olfactory performance. Second, we explored the modulatory effect of central insulin action on olfactory perception through intranasal administration of different doses. Intranasal insulin applications are associated with reactive blood insulin changes25 that might in turn interact with olfactory perception and mask the intranasal insulin effects. Therefore, we hypothesized that correcting for reactive changes of blood insulin levels will reveal a dose-dependent modulation of olfactory perception by intranasal insulin with stronger effects of higher doses.
Research Design and Methods
Participants
This article provides findings obtained from a sample of 36 healthy weight and overweight male volunteers (BMI 24.98 ± 2.6 kg/m²), who underwent an intranasal intervention study to test for insulin effects on olfactory acuity. All participants gave written informed consent to participate in the experiment. The informed consent forms and all experimental protocols were approved by the local ethics committee of the Medical Faculty of the University of Cologne (Cologne, Germany). All methods were performed according to relevant guidelines and regulations.
The participants were recruited from a pre-existing database of volunteers maintained at the Max-Planck-Institute for Metabolism Research. We only employed male participants to exclude variations of hormonal effects on the olfactory performance during the menstrual cycle26. All participants were non-smokers without any history of neurological, psychiatric, gastrointestinal or eating disorders, and without any special diets or medical treatments. We had to exclude three subjects in the course of the data analysis: two subjects exceeded the cut-off value in a depression score (BDI > 13), and one subject showed an outlying variance of test performance across testing days (Cochran C test, C(7,34) = 0.14, p = 0.001). In total, 33 subjects (BMI: 24.87 ± 2.6 kg/m2, age: 25.6 ± 2.9 yrs, HOMA-IR: 1.68 ± 0.9) were included in the complete data analysis.
Experimental design
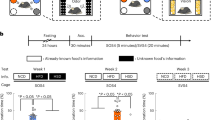
The study was carried out in a single-blinded, placebo-controlled, crossover design (Supplementary Fig. 1). Each volunteer participated in seven experimental sessions. Two subsequent sessions were 4–21 days apart from each other and started around the same time of the day (either at 8:00 a.m. or 10:00 a.m.). On each testing day, participants arrived fasted with the last meal before 10 p.m. of the previous day.
On the first testing day the subjects performed the olfactory tests without any intervention to measure the baseline olfactory performance. On the next six testing days each participant received on different testing days either 40 units (0.4 ml), 100 units (1.0 ml), or 160 units (1.6 ml) of insulin (I.U.; Huminsulin® Normal KwikPen 100 I.U./ml; Lilly Germany GmbH) or 0.4, 1.0 or 1.6 ml of placebo (Saline KwikPen, Lilly Germany GmbH) in a counterbalanced order. As placebo, the vehicle solution with the same conservations agents was used and, hence, was indistinguishable by smell or sensation from insulin (Supplementary Fig. 4). To directly reach the central nervous system and circumvent the blood brain barrier the intervention was administered intranasally. Insulin and placebo were administered with a precision air pump (Aero Pump, Hochheim, Germany) alternating between both nostrils with an interval of one minute to allow sufficient time for absorption. Each puff of the air pump contained 0.1 ml of solution (equal to 10 I.U.). During the administration the volunteers were sitting upright in a separate room, observed by the test leader. On each testing day, an intravenous catheter was inserted into the left forearm vein and a blood sample was drawn in baseline condition as well as 10, 25, and 70 min post-intervention to identify the effect of intranasal insulin on glucose, insulin, c-peptide, and cortisol level.
Thirty minutes post-intervention two subtests of the “Sniffin’ Sticks” test battery (Burghart Instruments, Wedel, Germany) were used to assess the olfactory threshold and discrimination. The Sniffin’ Sticks test kit using odor dispensing pen-like devices is a standard approach to assess nasal chemosensory performance in research and clinical setting with a high test-retest reliability (0.61 for threshold, 0.54 for discrimination testing)27 and validity compared to other assessment batteries such as the “Cross-Cultural Smell Identification Test” and the “Connecticut Chemosensory Clinical Research Center Test”27,28,29. In particular, the olfactory threshold test, that assesses the concentration where half of the stimuli are detected while the other half remain undetectable, does not involve the identification of the exact odorant stimulus and is therefore unrelated to episodic and sematic memory and cognitive performance30.
To determine the olfactory threshold a single staircase, three alternative forced choice (3-AFC) procedure was used with 16 different dilutions of n-butanol (BUT)27,28. The olfactory threshold is defined as the lowest odor concentration that can be reliably detected31. More precisely, the test starts with the lowest BUT concentration that has to be identified among three presented pens (the other two pens are impregnated with a solvent). A wrong choice is followed by the next triplet of pens with increased BUT concentration until the subject identifies the correct pen. After a BUT pen with a certain concentration is correctly identified two times in a row the examiner continues with the triplet of pens, which includes the pen with the immediately lower BUT concentration. This is defined as the first turning point. After the next observed error (second turning point) a higher concentration is then presented again. The olfactory threshold detection score is determined by the mean of the last four turning points. The higher the olfactory detection score - called olfactory threshold performance in our results - the lower the olfactory threshold. For the discrimination test, subjects were presented 16 triplets of common everyday odorants like vanilla or cinnamon with the same concentration in a 3-AFC procedure in a counterbalanced order27,31. Here, two pens of each triplet contain the same odor, while the third pen is impregnated with a different smell with the same concentration. The subject has to identify the pen that differs from the other two. Olfactory discrimination is defined as the number of correct responses. See the Supplementary information for the data table on olfactory performance (Supplementary Data 2).
At the beginning of each testing day as well as before and after the olfactory testing subjects were asked to rate their hunger, satiety, tiredness and the feeling of a stuffy nose on a continuous 10 cm visual analogue scale (0 = “not hungry at all/not satiated at all/not tired at all”, and 10 = “extremely hungry/extremely satiated/extremely tired”). At the end of each testing day, subjects were further asked to rate how difficult they perceived the olfactory test on a visual analogue scale (0 = “extremely easy” and 10 = ”extremely difficult”). Additionally, on the first testing day subjects had to answer to a set of questionnaires comprising the Beck Depression Inventory (BDI-II)32 and the Baratt Impulsiveness Scale (BIS-1)33.
Statistical analysis
The effect of insulin intervention on biochemical data, ratings and olfactory test results was assessed by linear mixed effect models using the R package ‘NLME’ 3.134. Linear mixed effect models allowed to account for both within- and between-subjects sources of variance and offered an equivalent yet more versatile alternative to repeated measures ANOVA35. In addition to a general error term, all models included a random intercept for each participant to account for the repeated measures within participants (denoted “error ~1|participant” in Wilkinson notation). All other effects were modeled as fixed effects. In the description of each linear mixed effect model below we provide the characteristics of the factors tested. Main and interaction effects were both tested, but only effects that were significant at a critical p-value of 0.05 were reported. Post-hoc tests were corrected for multiple comparisons using the Dunnett correction (R ‘LSMEANS’ package36).
In this study, we investigated the olfactory performance after intranasal application of different doses of insulin and the corresponding placebo volumes (independent, manipulated variable). In addition, we assessed the relation between olfactory threshold and the pre-experimental variable insulin sensitivity (quantified by HOMA-IR) and the reactive variable blood insulin change after intranasal insulin application.
To investigate the relation between body weight, insulin sensitivity, and olfactory performance (both threshold performance and discrimination performance), we first performed a Pearson correlation analysis between BMI, average baseline HOMA-IR and average test performance across all seven testing days. To further control for the pharmacological manipulation, we implemented a linear mixed effect model assessing the effect of baseline HOMA-IR and condition (baseline, three different insulin interventions, three different placebo interventions) on olfactory performance (olfactory threshold or olfactory discrimination). Accordingly, we tested the effect of BMI and conditions on olfactory performance. The homeostasis model assessment of insulin resistance (HOMA-IR)37,38 is a proxy for insulin sensitivity and calculated as
Higher values indicate a lower degree of insulin sensitivity. As the HOMA-IR was initially not normally distributed, we applied a logarithmic transformation prior to analysis.
In the following we only used linear mixed effect models including the random effect for the factor participant.
To start with, we tested the effect of intervention (categorical factor, 2 levels: insulin, placebo) and test modality (categorical factor, 2 levels: olfactory threshold, olfactory discrimination) on olfactory performance. We beforehand z-scored test-performances to allow the comparison of olfactory performances across subjects and test modalities (olfactory threshold and olfactory discrimination).
Next, to assess the dose dependent modulation of blood insulin and blood glucose by intranasal insulin we analyzed the effect of intervention dose (categorical factor, 4 levels: 40 I.U., 100 I.U., 160 I.U, average placebo) and time (categorical factor, 4 levels: baseline, 10, 25, 70 min post intervention) on either blood insulin or blood glucose.
We then tested whether reactive blood insulin changes after intranasal insulin administration modulated olfactory threshold performance by setting up a model testing the effect of intranasal insulin dose (categorical factor, 3 levels: 40 I.U., 100 I.U. and 160 I.U.) and blood insulin level prior to olfactory testing in the insulin compared to the placebo condition (Δ blood insulin(insulin–placebo), continuous factor) on the olfactory threshold performance in the insulin compared to placebo condition (Δ olfactory threshold performance(insulin-placebo), continuous factor).
Finally, we corrected the olfactory threshold values by regressing out the blood insulin level prior to olfactory assessment (25 min post intervention) and consequently analyzed the effect of intervention (categorical factor, 2 levels: placebo, insulin) and insulin dose (categorical factor, 3 levels: 40 I.U, 100 I.U., 160 I.U.) on the corrected olfactory threshold performance.
We controlled for differences in internal states by testing the effect of time (baseline, 10 min, 25 min and 70 min) and intervention dose on the subjects’ ratings of satiety, hunger, and tiredness.
The order of the testing days was counterbalanced by pseudo-randomly assigning the participants to one of five groups, each group being associated with a different carefully predetermined order of the six interventions. To analyze a potential order effect we used a linear mixed model analyzing the effect of group (categorical factor, 5 levels) on either olfactory threshold or olfactory discrimination.
All data are reported as the mean ± SEM.
Results
To investigate the effect of intranasal insulin application on olfactory performance, we tested 33 male volunteers in a weight range between normal- and overweight.
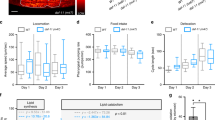
First, we investigated the effect of baseline insulin sensitivity on olfactory performance. While there was no correlation with BMI, average performance of both olfactory threshold (r(31) = −0.455, p = 0.008) as well as odor discrimination (r(31) = −0.352, p = 0.04) were correlated with average HOMA-IR at baseline level. Moreover, to account for the different conditions across testing days (baseline, three different insulin and placebo interventions) we performed linear mixed effect models testing the effect of baseline HOMA-IR and conditions on olfactory performance (olfactory threshold/olfactory discrimination). In line with the correlation analysis, beside the main effect of condition (F(6,185) = 2.67, p < 0.016) we found a main effect of baseline HOMA-IR (F(1,185) = 6.86, p = 0.009) on olfactory threshold. For olfactory discrimination, there was a main effect of condition (F(6,185) = 2.83, p = 0.011) and a statistical trend for the main effect of baseline HOMA-IR HOMA-IR (F(1,185) = 2.28, p = 0.100). In contrast, testing the effect of BMI and conditions on olfactory performance (olfactory threshold/olfactory discrimination) we could not find a main effect of BMI. In other words, higher HOMA-IR, i.e. lower systemic insulin sensitivity, rather than increased body weight per se was associated with poor performance in olfactory tasks (Fig. 1).
Baseline insulin sensitivity and olfactory performance: Average baseline HOMA-IR across all seven testing days was correlated with both (a) average olfactory threshold (r(31) = −0.455, p = 0.008) and (b) average olfactory discrimination (r(31) = −0.352, p = 0.04) performance across testing days. (Baseline HOMA-IR was converted by logarithmic transformation).
Next, we explored the effect of local intranasal insulin application on olfactory functioning. Surprisingly, intranasal insulin improved the sense of smell (Fig. 2). This effect was specific to olfactory threshold performance, while the discrimination of olfactory stimuli remained unchanged after intranasal insulin application (main effect of intervention: F(1,360) = 4.970, p = 0.0264, interaction effect intervention × test type: F(1,360) = 6.031, p = 0.01).
Improved olfactory performance after intranasal insulin: The effect of intranasal insulin (across all intervention doses) was specific to olfactory modality. It improved olfactory threshold, while olfactory discrimination remained unchanged. (Means ± SEM, ***p ≤ 0.001, **p ≤ 0.01, *p ≤ 0.05, the values are z-transformed).
To further identify possible dose dependent effects of intranasally applied insulin on olfactory performance, we compared the olfactory performance after application of 40 I.U., 100 I.U. and 160 I.U. intranasal insulin.
Before going further, it is important to note that in line with previous reports intranasal insulin application induced reactive blood insulin changes. It increased blood insulin (no significant main effect, interaction effect of time × dose: F(9,380) = 6.595, p < 0.0001) and reduced blood glucose (no significant main effect, interaction effect of time × dose: F(9,380) = 16.044, p < 0.0001) level in a time- and dose-dependent manner (Fig. 3a,b). This observation raised the question whether the shift in blood insulin level prior to olfactory testing in the insulin compared to the placebo condition (Δ blood insulin, insulin - placebo) affected the olfactory performance. Hence, we assessed the effect of Δ blood insulin on the olfactory threshold performance. Indeed, we found that both blood insulin and intranasal insulin intervention dose had an effect on olfactory threshold (main effect of blood insulin: F(2,61) = 4.772, p = 0.03), main effect of intervention dose: F(2,61) = 5.081, p = 0.009). Higher Δ blood insulin predicted poorer olfactory threshold performance (Fig. 3c). To better delineate the effect of local insulin on olfactory threshold performance, we then tested the effect of intranasal insulin after correcting for (i.e. regressing out) the effect corresponding to the blood insulin level prior to olfactory testing (25 minutes post-intervention). This analysis showed a main effect of intervention (F(1,160) = 17.351, p = 0.001) and revealed an interaction of intervention and dose (F(2,160) = 3.144, p = 0.04). Strikingly, post-hoc tests showed that 100 I.U. (p = 0.0001, t = −4.0) and 160 I.U. (p = 0.0076, t = −2.7) of intranasal insulin induced a strong improvement of olfactory threshold performance compared to the corresponding placebo dose (Fig. 4). In other words, while higher systemic insulin levels predicted poorer olfactory performance, higher intranasal insulin doses improved olfactory threshold.
Effects of systemic insulin on olfactory threshold: Intranasal insulin administration caused a time- and dose-dependent modulation of (a) systemic insulin and (b) glucose level. (Means ± SEM, ***p ≤ 0.001, **p ≤ 0.01, *p ≤ 0.05). (c) Both systemic and intranasal insulin had an effect on olfactory threshold: Higher systemic Δ insulin predicted poor Δ olfactory threshold performance. (Δ insulin = blood insulin level 25 min post intervention in insulin compared to placebo condition, Δ Olfactory threshold performance = threshold test in insulin compared to placebo condition).
Improved olfactory threshold after higher doses of intranasal insulin: Intranasal insulin showed a dose dependent effect on olfactory threshold performance. We found an interaction of intervention and intervention dose. 100 I.U. and 160 I.U. intranasal insulin improved olfactory threshold compared to the corresponding placebo condition (Means ± SEM, ***p ≤ 0.001, **p ≤ 0.01, the values are z-scored).
To control for differences in internal states, we instructed the participants to rate their hunger, satiety, and tiredness at baseline and immediately before and after olfactory testing on each day. Interestingly, hunger (main effect of time: F(2,254) = 16.24, p < 0.0001) and tiredness (main effect of time: F(2,254) = 7.50, p = 0.0007) increased and satiety dropped (main effect of time: F(2,254) = 12.63, p < 0.0001) over time, but we could not find an effect of intranasal insulin application on these parameters (Supplementary Fig. 3). In addition, we tested for a potential order effect, as all participants performed olfactory tests for seven times. We could not find an effect of order neither on olfactory threshold (F(1,164) = 1.54, p = 0.22) nor on olfactory discrimination (F(1,164) = 0.98, p = 0.32) performance.
Discussion
Recent findings indicate a strong impact of metabolic state and body weight on olfactory perception, however very little information has been gathered on the underlying mechanisms1,39,40,41. Given the evidence for direct insulin action on neuronal activity42,43 and cell regeneration in the olfactory system44, we systematically investigated the modulatory effects of intranasal insulin on olfactory performance. In addition we characterized the role of insulin-sensitivity and reactive blood insulin changes after intranasal insulin application on olfactory threshold. Our findings suggest that systemic and local insulin modulate smell perception in opposing direction.
The olfactory bulb is considered as an internal sensor for the nutritional status. Besides other gastrointestinal signals, insulin has been shown to directly act on olfactory neurons43,45. This is particularly interesting, since systemic insulin levels dynamically reflect body fuel availability. We demonstrate that a transient increase in blood insulin modulated olfactory performance. Consistent with previous hyperinsulinemic clamp data18, higher blood insulin predicted poorer olfactory performance. These findings extend to the assumption that changes of olfactory sensitivity are induced by satiety signals18,19,31.
With increasing body weight insulin homeostasis is impaired, leading to hyperinsulinemia and insulin resistance. Previous studies reported olfactory dysfunction in morbidly obese subjects39,46 and diabetes47. Odor sensitivity has been found to decrease with increasing BMI40,41. We discovered that not body weight itself but blood insulin level and insulin sensitivity determine olfactory threshold. Higher baseline HOMA-IR or circulating baseline insulin levels were correlated with poorer olfactory performances. Future studies need to clarify whether normalization of peripheral insulin sensitivity leads to a restored olfactory acuity. Interestingly, our pilot assessment of nine morbidly obese subjects (BMI: 42.8 ± 5.24 kg/m2, Supplementary Data 1) undergoing a weight loss program indicated an improvement of olfactory acuity with decreasing peripheral insulin levels (Supplementary Fig. 7).
Surprisingly, intranasal insulin had a contrary effect. We found enhanced olfactory performance after intranasal insulin administration. The olfactory threshold was selectively improved, while odor discrimination remained unchanged. Interestingly, this effect was dose-dependent. We discovered best improvement after 100 I.U. and 160 I.U. intranasal insulin.
At the first glance, these results seem contradictory to the assumption that insulin signals satiety and reduces odor sensitivity. However, it is established that centrally applied insulin often results in opposite effects compared to peripheral application suggesting a counter-regulatory mechanism48. Blood borne insulin crosses the blood brain barrier (BBB) through a saturable transport mechanism17,49. Blood and olfactory bulb insulin levels are elevated in sated compared to fasted state, but these levels are not correlated and the ratio changes with increasing blood insulin level19. Intranasal application bypassing the BBB results in unphysiological elevation of central insulin levels50 that may evoke opposing effects on olfactory sensitivity. This is further supported by the fact that we found a stronger improvement for high intervention doses. However, it is important to note that intranasal insulin does not specifically target the olfactory bulb but may also cause activity changes in other components of the olfactory system such as the olfactory epithelium and the olfactory cortex.
Intranasal insulin application is widely considered as the standard procedure to mimic central nervous insulin actions in humans. However, in line with previous literature25 we demonstrate a profound dose-dependent increase of blood insulin with subsequent reductions in glucose concentration. Previous human and animal work suggested several mechanisms underlying these reactive changes such as spill over effects, vagally transmitted insulin secretion51, suppression of endogenous glucose production52 or even classical conditioning53. However, the short interval between intranasal insulin administration and peripheral insulin rise in our study suggest spill-over effects. Our results clearly indicate the relevance of controlling for fasting state, blood insulin level and peripheral insulin sensitivity when testing the effects of intranasal insulin. Inconsistencies in these parameters might explain the contradictory reports on intranasal insulin effects on olfactory threshold so far20,21,45.
In conclusion, our findings indicate opposing effects of central and blood insulin on olfactory threshold performance. While intranasal insulin dose-dependently improved olfactory threshold, increasing blood insulin levels and poor insulin sensitivity predicted poor olfactory performance. Given the evidence, that olfactory sensitivity directly impacts central homeostatic regulation3,4 and peripheral metabolism5, being able to modulate olfactory acuity through intranasal insulin application represents a highly attractive potential for clinical application.
Data Availability
The data on olfactory performance and blood parameters are included in the Supplementary Information files. Further datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Palouzier-Paulignan, B. et al. Olfaction under Metabolic Influences. Chem Senses 37, 769–797, https://doi.org/10.1093/chemse/bjs059 (2012).
Julliard, A. K., Al Koborssy, D., Fadool, D. A. & Palouzier-Paulignan, B. Nutrient Sensing: Another Chemosensitivity of the Olfactory System. Front Physiol 8, 468, https://doi.org/10.3389/fphys.2017.00468 (2017).
Chen, Y., Lin, Y. C., Kuo, T. W. & Knight, Z. A. Sensory Detection of Food Rapidly Modulates Arcuate Feeding Circuits. Cell 160, 829–841, https://doi.org/10.1016/j.cell.2015.01.033 (2015).
Chen, Y. & Knight, Z. A. Making Sense of the Sensory Regulation of Hunger Neurons. Bioessays 38, 316–324, https://doi.org/10.1002/bies.201500167 (2016).
Riera, C. E. et al. The Sense of Smell Impacts Metabolic Health and Obesity. Cell Metab 26, 198–211.e195, https://doi.org/10.1016/j.cmet.2017.06.015 (2017).
Gascuel, J. et al. Hypothalamus-Olfactory System Crosstalk: Orexin a Immunostaining in Mice. Front Neuroanat 6, 44, https://doi.org/10.3389/fnana.2012.00044 (2012).
Aime, P. et al. Fasting Increases and Satiation Decreases Olfactory Detection for a Neutral Odor in Rats. Behav Brain Res 179, 258–264, https://doi.org/10.1016/j.bbr.2007.02.012 (2007).
Cameron, J. D., Goldfield, G. S. & Doucet, E. Fasting for 24 H Improves Nasal Chemosensory Performance and Food Palatability in a Related Manner. Appetite 58, 978–981, https://doi.org/10.1016/j.appet.2012.02.050 (2012).
Soria-Gomez, E. et al. The Endocannabinoid System Controls Food Intake Via Olfactory Processes. Nat Neurosci 17, 407–415, https://doi.org/10.1038/nn.3647 (2014).
Critchley, H. D. & Rolls, E. T. Hunger and Satiety Modify the Responses of Olfactory and Visual Neurons in the Primate Orbitofrontal Cortex. J Neurophysiol 75, 1673–1686, https://doi.org/10.1152/jn.1996.75.4.1673 (1996).
Ramaekers, M. G., Verhoef, A., Gort, G., Luning, P. A. & Boesveldt, S. Metabolic and Sensory Influences on Odor Sensitivity in Humans. Chem Senses 41, 163–168, https://doi.org/10.1093/chemse/bjv068 (2016).
Thiebaud, N. et al. Hyperlipidemic Diet Causes Loss of Olfactory Sensory Neurons, Reduces Olfactory Discrimination, and Disrupts Odor-Reversal Learning. J Neurosci 34, 6970–6984, https://doi.org/10.1523/jneurosci.3366-13.2014 (2014).
Lacroix, M. C. et al. Long-Lasting Metabolic Imbalance Related to Obesity Alters Olfactory Tissue Homeostasis and Impairs Olfactory-Driven Behaviors. Chem Senses 40, 537–556, https://doi.org/10.1093/chemse/bjv039 (2015).
Marks, J. L., Porte, D. Jr., Stahl, W. L. & Baskin, D. G. Localization of Insulin Receptor Mrna in Rat Brain by in Situ Hybridization. Endocrinology 127, 3234–3236, https://doi.org/10.1210/endo-127-6-3234 (1990).
Havrankova, J., Brownstein, M. & Roth, J. Insulin and Insulin Receptors in Rodent Brain. Diabetologia 20(Suppl), 268–273, https://doi.org/10.1007/BF00254492 (1981).
Baskin, D. G., Porte, D. Jr., Guest, K. & Dorsa, D. M. Regional Concentrations of Insulin in the Rat Brain. Endocrinology 112, 898–903, https://doi.org/10.1210/endo-112-3-898 (1983).
Banks, W. A., Kastin, A. J. & Pan, W. Uptake and Degradation of Blood-Borne Insulin by the Olfactory Bulb. Peptides 20, 373–378, https://doi.org/10.1016/S0196-9781(99)00045-5 (1999).
Ketterer, C. et al. Acute, Short-Term Hyperinsulinemia Increases Olfactory Threshold in Healthy Subjects. Int J Obes (Lond) 35, 1135–1138, https://doi.org/10.1038/ijo.2010.251 (2011).
Aime, P. et al. A Physiological Increase of Insulin in the Olfactory Bulb Decreases Detection of a Learned Aversive Odor and Abolishes Food Odor-Induced Sniffing Behavior in Rats. PLoS One 7, e51227, https://doi.org/10.1371/journal.pone.0051227 (2012).
Brunner, Y. F., Benedict, C. & Freiherr, J. Intranasal Insulin Reduces Olfactory Sensitivity in Normosmic Humans. J Clin Endocrinol Metab 98, E1626–1630, https://doi.org/10.1210/jc.2013-2061 (2013).
Schopf, V., Kollndorfer, K., Pollak, M., Mueller, C. A. & Freiherr, J. Intranasal Insulin Influences the Olfactory Performance of Patients with Smell Loss, Dependent on the Body Mass Index: A Pilot Study. Rhinology 53, 371–378, https://doi.org/10.4193/Rhin15.065 (2015).
Rezaeian, A. Effect of Intranasal Insulin on Olfactory Recovery in Patients with Hyposmia: A Randomized Clinical Trial. Otolaryngol Head Neck Surg 158, 1134–1139, https://doi.org/10.1177/0194599818764624 (2018).
Utzschneider, K. M. et al. Insulin Resistance Is the Best Predictor of the Metabolic Syndrome in Subjects with a First‐Degree Relative with Type 2 Diabetes. Obesity 18, 1781–1787, https://doi.org/10.1038/oby.2010.77 (2010).
Owei, I., Umekwe, N., Provo, C., Wan, J. & Dagogo-Jack, S. Insulin-Sensitive and Insulin-Resistant Obese and Non-Obese Phenotypes: Role in Prediction of Incident Pre-Diabetes in a Longitudinal Biracial Cohort. BMJ Open Diabetes Research and Care 5, e000415, https://doi.org/10.1136/bmjdrc-2017-000415 (2017).
Schmid, V. et al. Safety of Intranasal Human Insulin: A Review. Diabetes Obes Metab 20, 1563–1577, https://doi.org/10.1111/dom.13279 (2018).
Hummel, T., Gollisch, R., Wildt, G. & Kobal, G. Changes in Olfactory Perception During the Menstrual Cycle. Experientia 47, 712–715, https://doi.org/10.1007/BF01958823 (1991).
Hummel, T., Sekinger, B., Wolf, S. R., Pauli, E. & Kobal, G. ‘Sniffin’ Sticks’: Olfactory Performance Assessed by the Combined Testing of Odor Identification, Odor Discrimination and Olfactory Threshold. Chem Senses 22, 39–52, https://doi.org/10.1093/chemse/22.1.39 (1997).
Kobal, G. et al. “Sniffin’ Sticks”: Screening of Olfactory Performance. Rhinology 34, 222–226, https://doi.org/10.1093/chemse/22.1.39 (1996).
Hummel, T. et al. Position Paper on Olfactory Dysfunction. Rhinol Suppl 54, 1–30, https://doi.org/10.4193/Rhin16.248 (2017).
Hedner, M., Larsson, M., Arnold, N., Zucco, G. M. & Hummel, T. Cognitive Factors in Odor Detection, Odor Discrimination, and Odor Identification Tasks. J Clin Exp Neuropsychol 32, 1062–1067, https://doi.org/10.1080/13803391003683070 (2010).
Albrecht, J. et al. Olfactory Detection Thresholds and Pleasantness of a Food-Related and a Non-Food Odour in Hunger and Satiety. Rhinology 47, 160–165 (2009).
Beck, A. T., Steer, R. A. & Brown, G. K. Beck Depression Inventory-Ii. San Antonio 78, 490–498 (1996).
Patton, J. H., Stanford, M. S. & Barratt, E. S. Factor Structure of the Barratt Impulsiveness Scale. J Clin Psychol 51, 768–774, https://doi.org/10.1002/1097-4679(199511)51:6%3C768::AID-JCLP2270510607%3E3.0.CO;2-1 (1995).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & Team, R. C. Nlme: Linear and Nonlinear Mixedeffects Models. R Package Version 3.1–128. 2016. R software (2017).
Gelman, A. & Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models. (Cambridge university press, 2006).
Lenth, R. V. Least-Squares Means: The R Package Lsmeans. Journal of statistical software 69, 1–33 (2016).
Matthews, D. et al. Homeostasis Model Assessment: Insulin Resistance and Β-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 28, 412–419, https://doi.org/10.1007/BF00280883 (1985).
McAuley, K. A., Mann, J. I., Chase, J. G., Lotz, T. F. & Shaw, G. M. Point: Homa—Satisfactory for the Time Being: Homa: The Best Bet for the Simple Determination of Insulin Sensitivity, until Something Better Comes Along. Diabetes Care 30, 2411–2413, https://doi.org/10.2337/dc07-1067 (2007).
Richardson, B. E., Vanderwoude, E. A., Sudan, R., Leopold, D. A. & Thompson, J. S. Gastric Bypass Does Not Influence Olfactory Function in Obese Patients. Obes Surg 22, 283–286, https://doi.org/10.1007/s11695-011-0487-x (2012).
Simchen, U., Koebnick, C., Hoyer, S., Issanchou, S. & Zunft, H. J. Odour and Taste Sensitivity Is Associated with Body Weight and Extent of Misreporting of Body Weight. Eur J Clin Nutr 60, 698–705, https://doi.org/10.1038/sj.ejcn.1602371 (2006).
Skrandies, W. & Zschieschang, R. Olfactory and Gustatory Functions and Its Relation to Body Weight. Physiol Behav 142, 1–4, https://doi.org/10.1016/j.physbeh.2015.01.024 (2015).
Lacroix, M. C. et al. Expression of Insulin System in the Olfactory Epithelium: First Approaches to Its Role and Regulation. J Neuroendocrinol 20, 1176–1190, https://doi.org/10.1111/j.1365-2826.2008.01777.x (2008).
Fadool, D. A., Tucker, K. & Pedarzani, P. Mitral Cells of the Olfactory Bulb Perform Metabolic Sensing and Are Disrupted by Obesity at the Level of the Kv1.3 Ion Channel. PLoS One 6, e24921, https://doi.org/10.1371/journal.pone.0024921 (2011).
Lacroix, M. C. et al. Insulin but Not Leptin Protects Olfactory Mucosa from Apoptosis. J Neuroendocrinol 23, 627–640, https://doi.org/10.1111/j.1365-2826.2011.02154.x (2011).
Kuczewski, N. et al. Insulin Modulates Network Activity in Olfactory Bulb Slices: Impact on Odour Processing. The Journal of physiology 592, 2751–2769, https://doi.org/10.1113/jphysiol.2013.269639 (2014).
Richardson, B. E., Vander Woude, E. A., Sudan, R., Thompson, J. S. & Leopold, D. A. Altered Olfactory Acuity in the Morbidly Obese. Obes Surg 14, 967–969, https://doi.org/10.1381/0960892041719617 (2004).
Gouveri, E. et al. Olfactory Dysfunction in Type 2 Diabetes Mellitus: An Additional Manifestation of Microvascular Disease? Angiology 65, 869–876, https://doi.org/10.1177/0003319714520956 (2014).
Banks, W. A. The Source of Cerebral Insulin. Eur J Pharmacol 490, 5–12, https://doi.org/10.1016/j.ejphar.2004.02.040 (2004).
Schwartz, M. W. et al. Kinetics and Specificity of Insulin Uptake from Plasma into Cerebrospinal Fluid. Am J Physiol 259, E378–383, https://doi.org/10.1152/ajpendo.1990.259.3.E378 (1990).
Born, J. et al. Sniffing Neuropeptides: A Transnasal Approach to the Human Brain. Nat Neurosci 5, 514–516, https://doi.org/10.1038/nn849 (2002).
Chen, M., Woods, S. C. & Porte, D. Effect of Cerebral Intraventricular Insulin on Pancreatic Insulin Secretion in the Dog. Diabetes 24, 910–914, https://doi.org/10.2337/diab.24.10.910 (1975).
Dash, S., Xiao, C., Morgantini, C., Koulajian, K. & Lewis, G. F. Intranasal Insulin Suppresses Endogenous Glucose Production in Humans Compared with Placebo in the Presence of Similar Venous Insulin Concentrations. Diabetes 64, 766–774, https://doi.org/10.2337/db14-0685 (2015).
Stockhorst, U., De Fries, D., Steingrueber, H.-J. & Scherbaum, W. Unconditioned and Conditioned Effects of Intranasally Administered Insulin Vs Placebo in Healthy Men: A Randomised Controlled Trial. Diabetologia 54, 1502–1506, https://doi.org/10.1007/s00125-011-2111-y (2011).
Acknowledgements
S.E.T. analyzed data and performed experiments together with V.H. L.R. contributed to the analysis methods and statistics. R.H. provided support with recruiting subjects. S.E.T., J.C.B. and M.T. conceived the study and wrote the manuscript. All authors agreed on the final version of the manuscript. M.T. is the guarantor of this study. The authors are grateful to Lisa Marie Koch, Laura Emmrich and Vera Kocher, all Max Planck Institute for Metabolism Research, for excellent technical assistance. S.E.T., M.T. and J.C.B. are supported by of the German Research Foundation in the Transregional Colloborative Research Center 134. J.C.B. and M.T. are supported by funding of the German Center for Diabetes Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Edwin Thanarajah, S., Hoffstall, V., Rigoux, L. et al. The role of insulin sensitivity and intranasally applied insulin on olfactory perception. Sci Rep 9, 7222 (2019). https://doi.org/10.1038/s41598-019-43693-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43693-7
This article is cited by
-
Mechanism and treatment of olfactory dysfunction caused by coronavirus disease 2019
Journal of Translational Medicine (2023)
-
Internal state affects local neuron function in an early sensory processing center to shape olfactory behavior in Drosophila larvae
Scientific Reports (2022)
-
Intranasal Insulin Enhances Intracerebroventricular Streptozotocin–Induced Decrease in Olfactory Discriminative Learning via Upregulation of Subventricular Zone–Olfactory Bulb Neurogenesis in the Rat Model
Molecular Neurobiology (2021)
-
Olfaction is a Marker of Severity but Not Diagnosis in Anorexia Nervosa: a Systematic Review and Meta-Analysis
Neuropsychology Review (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.