Abstract
Fine-scale topographic complexity creates important microclimates that can facilitate species to grow outside their main distributional range and increase biodiversity locally. Enclosed depressions in karst landscapes (‘dolines’) are topographically complex environments which produce microclimates that are drier and warmer (equator-facing slopes) and cooler and moister (pole-facing slopes and depression bottoms) than the surrounding climate. We show that the distribution patterns of functional groups for organisms in two different phyla, Arthropoda (ants) and Tracheophyta (vascular plants), mirror this variation of microclimate. We found that north-facing slopes and bottoms of solution dolines in northern Hungary provided key habitats for ant and plant species associated with cooler and/or moister conditions. Contrarily, south-facing slopes of dolines provided key habitats for species associated with warmer and/or drier conditions. Species occurring on the surrounding plateau were associated with intermediate conditions. We conclude that karst dolines provide a diversity of microclimatic habitats that may facilitate the persistence of taxa with diverse environmental preferences, indicating these dolines to be potential safe havens for multiple phyla under local and global climate oscillations.
Similar content being viewed by others
Introduction
Environmental heterogeneity is often positively related to biodiversity1,2,3,4. Topography and biological structures can create microhabitats with unique microclimates5,6,7, which species may depend on for survival8,9. These microhabitats may be warmer, drier, cooler and/or moister than the prevailing regional climate5,10, creating a mosaic of microclimates that can allow species to survive changes in their environment by migrating short distances between microhabitats10,11. These fine-scale mosaics improve a species’ chances to persist in a landscape, calling into question the results of large-scale (resolutions ≥1 km2) species distribution models7,9,12,13. As a result, attempts to regionally model climatic data at 50–100 m resolution have been made7,13.
Furthermore, topographic complexity can create habitats that remain environmentally more stable through time, even as regional climate changes. Such habitats may therefore facilitate the persistence of biodiversity and are known as refugia14,15,16. Refugia are important for conservation planning and may offer the only chance of in situ survival for many species17,18,19. Microrefugia are small areas that provide such suitable pockets of relatively stable microclimate20,21,22. For instance, areas of high topographic convergence (e.g. local depressions and valleys)23,24,25 retain cooler microclimates when regional climates warm through drainage of cold air currents at night26,27,28.
Karst areas cover about 20% of the earth’s dry land surface29 and their complex topography provides various ecological niches for plants and wildlife, playing a crucial role in the maintenance of world’s biodiversity30,31. Karst systems support unique microclimates in several microhabitats, such as south- and north-facing slopes, enclosed depressions (‘dolines’) and ravines21,32,33,34. Where dolines are associated with cave entrances and/or sinks, the cave and/or sink will further influence the microclimate35. Several studies indicate that karst areas in Europe have supported the populations of both cool- and warm-adapted taxa outside their main distribution area (i.e. climate relicts) during past Quaternary climate oscillations36,37,38,39.
A number of studies indicate that dolines may play a crucial role in maintaining biodiversity. They harbour unique taxa that are rare or absent in the surrounding areas (e.g. endemic and relict species)40,41,42 and are characterised by high plant, genetic and habitat diversity43,44,45,46. Cool-adapted species from various phyla (e.g. Arthropoda, Bryophyta, Mollusca and Tracheophyta) have been documented from dolines47,48,49,50. In addition, dolines may also provide key habitats for warm-adapted species35. Documenting the distribution of both cool- and warm-adapted taxa with respect to microclimate inside and adjacent to dolines may therefore help us to better understand the potential of dolines to function as microrefugia.
Here we investigate the distribution patterns of organisms in four different microhabitats inside (south-facing slopes, bottoms and north-facing slopes) and outside of dolines (plateau) in the Bükk Mountains, northern Hungary. We illustrate that dolines can support a wide range of microclimatic conditions (both warmer and cooler than the surrounding plateau) and that they have the capacity to support diverse ant and plant assemblages. Furthermore, we show that the distributions of different functional groups (cool- and moist-adapted versus warm- and dry-adapted) of both, ants and plants, respond to the fine-scale microclimatic differences among the microhabitats in a similar manner. Therefore, our results show that dolines can be crucial safe havens for species from various phyla and highlight that investigating climate change responses will require high (~10 m) resolution environmental data for some taxa.
Results
Microclimate
Temperatures were higher on south-facing slopes (T24 = 25.9 °C; Td = 33.8 °C; Tn = 13.8 °C) than in other microhabitats (Figs 1 and 2B). The mean daily temperature was similar on north-facing slopes (T24 = 20.1 °C) and in doline bottoms (T24 = 20.6 °C), while night temperatures were lowest in bottoms (Tn = 9.78 °C). Night temperatures were similar on north-facing slopes (Tn = 11.5 °C) and the plateau (Tn = 11.8 °C). The mean daytime temperature was higher in bottoms (Td = 27.7 °C) than on north-facing slopes (Td = 25.7 °C). Temperatures on the plateau were intermediate (T24 = 24.0 °C; Td = 31.8 °C; Tn = 11.8 °C). Mean daily relative humidity was lowest on south-facing (RH24 = 68.0%) and highest on north-facing slopes (RH24 = 78.4%). The values were intermediate in bottoms (RH24 = 76.2%) and on the plateau (RH24 = 70.0%). At night, relative humidity was similar (RHn = 92.9–94.0%) in all microhabitats. However, daytime relative humidity was usually higher on north-facing slopes (RHd = 68.2%) than in bottoms (RHd = 65.2%), on south-facing slopes (RHd = 51.7%) and on the plateau (RHd = 55.1%). A small, intermittent temperature increase in all microhabitats was recorded around 1:30 hours and indicated an inflow of warmer air from the surrounding lower altitudes.
Study sites and study design. (A) Location of the Bükk Mountains in Hungary. (B) Study area and studied karst dolines (1–3). (C) Different parts of doline 1. (D) Early morning fog in dolines. (E) Semi-dry grassland on the plateau between dolines. (F) Wet meadow in the bottom of a doline. (G) Location of the study sites (site 1–6) in and around a doline. (H) Set-up of bait stations in a cross-shaped pattern.
Schematic illustration of the differentiation of microhabitats within karst dolines in Bükk, Hungary, with regard to (A) abundant ant and plant taxa and (B) microclimate. North-facing slopes and bottoms of dolines are consistently cooler and moister, while south-facing slopes are consistently warmer and drier than their surroundings. Abundant taxa of both phyla differ among microhabitats.
Species composition
A total of 14 ant (nine from baiting and another five from hand collecting) and 145 plant species (from the plots) were recorded in our sites (Fig. 1G; Supplementary Tables S1 and S2). Hand collection yielded 52 ant individuals, 13 were found on south-facing slopes, five in bottoms, 11 on north-facing slopes and 23 on the plateau. In terms of diagnostic species, south-facing slopes had two ant and 13 plant species, while two ant and 20 plant species were identified for the plateau. North-facing slopes and bottoms had 15 and nine plant species, respectively, and one ant species (Fig. 2A; Table 1). Dry grasslands dominated the south-facing slope of dolines 1 and 3, while semi-dry grasslands covered the south-facing slope of doline 2 and the major part of the plateau. North-facing slopes and doline bottoms were dominated by wet meadows.
NMDS ordinations of bait stations (stress factor: 0.119) and vegetation plots (stress factor: 0.064) showed that the species composition of ant and plant assemblages differed among microhabitats (Fig. 3A,B). These differences were significant, except between ant assemblages of bottoms and north-facing slopes (PERMANOVA; Table 2). Plant assemblages of all microhabitats displayed highly significant differences (p < 0.001). T24, Td and RHd were significantly related to the ordination of ant, and T24, Td, RH24 and RHd to the ordination of plant assemblages (Supplementary Table S3), with assemblages on south-facing slopes being associated with higher temperatures and lower humidity, and assemblages on north-facing slopes and in doline bottoms being related to lower temperatures and higher humidity.
Non-metric multidimensional scaling (NMDS) ordination for (A) ant and (B) plant assemblages in different microhabitats (south-facing slopes, bottoms and north-facing slopes of dolines, and the plateau) with fitted vectors of mean daily temperature (T24) and relative humidity (RH24), mean daytime temperature (Td) and relative humidity (RHd), and mean night temperature (Tn) and relative humidity (RHn). Vector length indicates the strength of correlation (see Supplementary Table S3). Microclimate variables that were significantly correlated to the ordination (T24, Td, RH24 and RHd) are indicated in boldface.
Functional groups of species
Functional groups of both ants and plants showed significant preferences for certain microhabitats (Figs 4 and 5; Supplementary Tables S4 and S5). On north-facing slopes, 64% of the hand-collected ants (individuals) were species adapted to cooler conditions, and 82% of the individuals were species adapted to intermediate moisture conditions. In other microhabitats, most of the hand-collected ants (100% on south-facing slopes, 80% in bottoms and 52% on the plateau) were species adapted to warmer and drier conditions. Ant (collected from bait stations) and plant species adapted to warmer and/or drier conditions occurred more frequently on south-facing slopes than on north-facing slopes and in doline bottoms (Figs 4 and 5; Supplementary Tables S4 and S5). Conversely, ant and plant species adapted to cooler and/or moister conditions were generally most frequent on north-facing slopes and in doline bottoms compared to south-facing slopes and the plateau. We did not find any ant species adapted to moister conditions and no plant species adapted to cooler conditions on south-facing slopes.
Occurrences of ant and plant species (mean ± SE) belonging to different functional groups of temperature requirements (warmer, intermediate and cooler) in different microhabitats (S: south-facing slopes, B: bottoms and N: north-facing slopes of dolines, and P: the plateau). Significant differences detected using mixed-effect models (see Supplementary Table S4) are indicated by different lower case letters (a–c).
Occurrences of ant and plant species (mean ± SE) belonging to different functional groups of moisture requirements (drier, intermediate and moister) in different microhabitats (S: south-facing slopes, B: bottoms and N: north-facing slopes of dolines, and P: the plateau). Significant differences detected using mixed-effect models (see Supplementary Table S5) are indicated by different lower case letters (a–c).
Discussion
Karst dolines are topographically complex environments that provide a variety of microhabitats. We have demonstrated that these habitats can be cooler and moister or warmer and drier than the surrounding plateau, providing high environmental heterogeneity at very fine scales. We further documented that the fine-scale distributions of functional groups in two different phyla (Arthropoda and Tracheophyta) correspond to these different microhabitats. To our knowledge, this is the first study to illustrate that the fine-scale topography of dolines provides microhabitats for diverse functional groups (cool- and moist-adapted versus warm- and dry-adapted) of both ants and vascular plants within tens of meters.
Topographic complexity increases the climatic variability in an area over fine scales24,51,52. Despite recording temperature and relative humidity for only a relatively short period of time (24 hours), we found that karst dolines introduce great variation in microclimates. For instance, mean daytime temperatures were more than 8 °C warmer on south- than on north-facing slopes (Fig. 2B). Previous microclimatic studies over longer time periods, i.e. from a few days to a year, also indicate north-facing slopes and bottoms of dolines to be cooler and more humid than south-facing slopes and the surrounding plateau23,35,53,54,55,56. In addition to receiving less solar radiation, doline bottoms tend to receive more water57, likely contributing to the higher relative humidity and lower temperatures recorded in these habitats58,59. North-facing slopes and bottoms of dolines may retain snow cover longer than south-facing slopes and plateaus60.
Fine-scale variation in environmental conditions affects the distributions of organisms61,62,63. Both ant and vascular plant species responded to microclimatic variation in our study. The cooler and moister north-facing slopes and bottoms of dolines in Bükk acted as key habitats for plants adapted to cooler and/or moister conditions (e.g. Bupleurum longifolium and Iris sibirica) and ants adapted to cooler conditions (e.g. Myrmica ruginodis) (Fig. 2A). On the other hand, south-facing slopes acted as key habitats both for ants (e.g. Myrmica sabuleti) and plants (e.g. Iris variegata) adapted to warmer and/or drier conditions, while many ant (e.g. Lasius niger) and plant species (e.g. Galium verum) found on the plateau indicated intermediate temperature and/or moisture conditions (Fig. 2A). The ant species recorded are not known to form strict trophic relationships with plants, and the main drivers of the observed patterns seem to be their temperature and moisture preferences. Our results underline the importance of considering fine-scale environmental variation when investigating the distribution of biodiversity63,64. Even the highest resolution (50–100 m) climate data currently available7,13 would likely be insufficient to detect the environmental heterogeneity provided by the karst dolines in our study, which are <200 m wide and <20 m deep.
We have documented a strong, concerted response to fine-scale topography by the distributions of microclimate and species in two major taxonomic groups (ants, Arthropoda; and vascular plants, Tracheophyta). However, other taxa also display distribution patterns reflecting changes in environmental conditions in dolines over short distances. For instance, the increased soil moisture content on north-facing slopes and bottoms of solution dolines in the Aggtelek Karst area (Hungary) has been shown to provide suitable habitats for several cool-adapted species of land snails (Mollusca)49 and terrestrial isopods (Arthropoda)65. Dolines in Mexico, Australia and China (‘cenotes’ and ‘tiankengs’) have also been shown to maintain populations of rare taxa in various phyla46,66,67,68, highlighting dolines as important safe havens for biodiversity on a global scale.
Species may respond to climate changes by range-shifting69 or by persisting in environmentally stable habitats70,71. In karst dolines, species could potentially track suitable microclimates over a wide range of regional climatic changes with minimal movement because cooler/moister and warmer/drier microclimates vary over very short distances. As few areas can buffer opposing trends in environmental conditions72, karst dolines may be particularly important for maintaining biodiversity through time. Therefore, they could be considered high-capacity microrefugia73. The highest-capacity microrefugia for cool-adapted taxa can usually be found in cold, humid and topographically complex areas74,75.
Therefore, karst dolines may play important roles in facilitating the persistence of different phyla under global warming, which poses a serious threat to global biodiversity76. Regional predictions of climate change suggest that warming in East-Central Europe will continue in the coming decades77. These changes are already impacting the distributions of ants78 and vascular plants79 in a sand-dune area in Hungary, with drought-tolerant species replacing dune slack species over the last decades. Therefore, species adapted to warmer and/or drier conditions are expected to expand their distribution from south-facing slopes of dolines to surrounding areas. However, north-facing slopes and bottoms of dolines could provide important microrefugia from global warming by facilitating the persistence of species adapted to cooler and/or moister and to intermediate conditions. The retention of cooler microclimates in these habitats may be facilitated by lower solar radiation, thicker soil layer, higher soil moisture and cool-air pooling27,58,80.
Although our data supports dolines to be safe havens for relict plant species (e.g. Aconitum variegatum, Bupleurum longifolium and Dracocephalum ruyschiana in Bükk) in the current climate, future studies should aim to confirm the status of dolines as refugia (i.e. places providing environmental conditions that are comparatively stable over long time periods) under global warming. While our 24-hour data demonstrate that dolines are currently providing cooler and warmer microclimates than the surrounding plateau, this does not necessarily prove stability and long-term monitoring would be needed for this. Available data from north-facing slopes in the Northern Hemisphere does suggest that such habitats undergo slower changes under global warming81,82. Alternatively, the microclimate of dolines could be investigated along a temperature gradient using a space-for-time substitution approach to determine if north-facing slopes and bottoms of dolines indeed retain more stable microclimates. Finally, functional traits can be reflective of long-term environmental stability and therefore could provide important eco-evolutionary information about refugia83.
We conclude that enclosed depressions in karst surfaces provide a diversity of microclimates that have the potential to enable the persistence of various taxa in different phyla and under various climatic trends. These dolines may be vital for facilitating the in situ persistence of numerous species under local and global climate oscillations. This implies that modelling of climate change impacts on the distribution of biodiversity will need to consider fine-scale topographic variation occurring within tens of meters to arrive at accurate predictions.
Methods
Study area
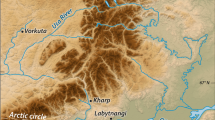
Our study area was located on the karst plateau of the Bükk Mountains (48°04′31″N, 20°29′57″E), in northern Hungary, at an altitude of approximately 780 m (Fig. 1A,B) in the beech (Fagus sylvatica) forest zone. This mountain range is believed to be an important refugial area in Hungary, supporting relict plant populations from both warmer (e.g. Clinopodium thymifolium, Cotinus coggygria and Ferula sadleriana) and cooler (e.g. Aconitum variegatum, Bupleurum longifolium and Dracocephalum ruyschiana) periods84,85. The plateau has a cool and humid climate, with a mean annual temperature of 6.3 °C and a mean annual precipitation of 800 mm. The plateau has typical karst landform features, such as solution dolines86,87,88, with a bowl-shaped geometry (Fig. 1C) and unique microclimate. At night, cold-air pooling occurs in these depressions, and the occurrence of frost or fog is possible all year round55 (Fig. 1D). According to some researchers89, the coldest areas in Hungary can be found in the non-forested dolines of Bükk. Previous investigations showed that north-facing slopes of these dolines are consistently cooler and moister than the surrounding plateau, and that south-facing slopes provide the warmest microhabitats54,90. The study area is well known for its unique wildlife and is part of the strictly protected area network of the Bükk National Park. The entire area is fenced to prevent overgrazing, soil erosion and the illegal collection of wild plants. Semi-dry grasslands and wet meadows are the dominant vegetation types within the fenced area91 (Fig. 1E,F).
Sampling design
Three large solution dolines were selected (Fig. 1B,C). Dolines 1 and 2 had diameters of 100 and 70 m and depths of 17 and 15 m, respectively. The longer diameter of doline 3 was 190 m, while the shorter was 65 m, its depth was 13 m. Six sampling sites were established per each doline (18 sites in total), one on the south-facing slope, one in the bottom, one on the north-facing slope and three on the surrounding plateau. The sites were located at least 20 m from each other (Fig. 1G). Sampling and microclimate measurements were carried out in August, under clear weather conditions, at the peak of the growing season.
Ants and plants were selected as focal taxa because ant colonies and plants share many similarities92. Both groups usually ‘nest’ in or on the ground and use their modules (e.g. plant roots and ant workers) to forage in the surrounding habitat93,94. In addition, due to the relatively fixed location of ant colonies and plants, competition in both groups is confined to well-defined zones. Similarities also exist in their functional roles in a given community (e.g. subordinate, specialist and cryptic species). Finally, ant foundresses (i.e. colony-founding queens) can be considered analogous to dispersing plant seeds93,95.
Considering that the study area is a strictly protected nature reserve, we used only non-destructive sampling methods such as baiting and hand collecting to assess the species diversity and relative abundance of ants. At each site, we placed five bait stations in a cross-shaped pattern at 5-m intervals (90 bait stations in total) (Fig. 1H). Baits were plastic discs (8 cm in diameter) with a quarter-teaspoon of a mixture of tuna and honey as a food reward. Foraging activity on baits was monitored every 40 minutes from 7:00 to 9:40, overlapping with the daily period of peak ant activity. During each observation, we recorded the presence and number of workers of each ant species on the bait. Baits were replenished as necessary. In addition, we also performed hand collecting to sample those ant species that may have not visited the baits. We visually searched the ground surface in each site for 5 minutes, hand collecting any individuals (workers, incipient queens, etc.) found. Ants were identified to morphospecies or genus level in the field, and representatives were collected and preserved in 95% ethanol for later species determination96. Field-collected specimens were identified in the laboratory using the keys of Seifert97 and Czechowski et al.98. All the collected specimens were deposited at the Department of Ecology, University of Szeged. We used Bolton’s catalogue99 and the Hymenoptera Name Server100 to determine the valid names of all ant species.
For plants, five randomly selected 1 m × 1 m plots were established in each site (90 plots in total). We recorded the presence/absence of all vascular plant species in all plots. Nomenclature follows ‘The Plant List’ (www.theplantlist.org).
To provide information on the microclimate of the study area, air temperature (T) and relative air humidity (RH) were recorded every minute for 24 hours using Voltcraft DL-121TH data loggers. Sensors were suspended 10 cm above the ground to allow sufficient wind flow to ensure that no humidity was trapped by the sensor casing and actual air temperature and humidity were measured.
Species grouping
We classified all ant and plant species according to their temperature and moisture requirements following the methods of Czechowski et al.98 for ants and Borhidi101 for plants (Supplementary Table S6 and Table S7). These were reduced to six functional groups applied to both ants and plants: (1) species adapted to warmer conditions, (2) species adapted to cooler conditions, (3) species adapted to intermediate temperature conditions, (4) species adapted to drier conditions, (5) species adapted to moister conditions, and (6) species adapted to intermediate moisture conditions (see details in Supplementary Tables S6 and S7). We did not analyse combined groups (temperature + moisture) because that would have made the interpretation of results difficult (with many groups), especially in the case of plants. All six main groups of plants and five main groups of ants (groups 1–4 and 6; ants adapted to moister conditions were absent in our study) were analysed. Because the temperature and moisture requirements of ant and plant species are not the same, direct comparisons between them were not possible.
Data analysis
The temperature and relative humidity data were averaged over 10-minute intervals across all sites of south-facing slopes, bottoms, north-facing slopes and the plateau, respectively, and plotted using a line graph. Extreme environmental values are generally more informative regarding the distribution of organisms, but maximum relative humidity values in our case often reached 100%, therefore we considered mean values more suitable for differentiating between the microclimatic properties of microhabitats than extreme values.
From the site-averaged data, we calculated the mean daily temperature (T24) and relative humidity (RH24), mean daytime temperature (Td) and relative humidity (RHd), and mean night temperature (Tn) and relative humidity (RHn). We also calculated these microclimate variables separately for each site, and used them in multivariate analyses.
The diagnostic ant and plant species of the microhabitats were determined by calculating the phi (Φ) coefficient of association between species and habitat. Calculations for ants were based on data from bait stations. Species with Φ > 0.2 were considered diagnostic for ants and species with Φ > 0.3 for plants. Different threshold values for ants and plants were used due to the differences in the total number of species within each of these taxonomic groups. Non-diagnostic species were excluded with Fisher’s exact test (p < 0.05) following Tichý and Chytrý102. Fidelity measures were calculated using the JUICE program103.
We used permutational multivariate analysis of variance (PERMANOVA) to test the effect of microhabitats (south-facing slopes, bottoms, north-facing slopes and the plateau) on the species composition of ant and plant assemblages. We used the raw presence/absence data of species for each sampling plot in the source matrices. We applied the Jaccard dissimilarity, performed 5000 permutations and also accounted for the nested design of the data set. When a PERMANOVA yielded significant results, we calculated pairwise PERMANOVAs among the microhabitat types. PERMANOVAs were calculated in R104 using the adonis function of the ‘vegan’ package105. We used the FDR (false discovery rate) method to adjust p values for multiple comparisons (p.adjust function). We prepared non-metric multidimensional scaling (NMDS) ordinations to visually illustrate compositional differences. To remove the confounding effect of the nested data structure on the resulting point clouds, we lumped data from the different sampling plots of each site of each doline together and used the frequency data, ranging from one to five, of the species in the source matrices. We used Euclidean distances and two dimensions (after assessing stress factors for one to five dimensions). NMDS ordinations were done using the metaMDS function of the ‘vegan’ package. To assess the relationships between microclimate variables (T24, RH24, Td, RHd, Tn and RHn) and species assemblages, we fitted environmental vectors onto the ordination space using the envfit function and calculated correlations between ordination values and fitted vectors.
We used mean-and-whisker plots to illustrate the distribution of the various functional groups in different microhabitats. To test if these differences were significant we used generalized linear mixed-effects models (GLMM). Calculations for ants were based on data from bait stations. Five models were built for ants (three for temperature and two for moisture) and six models for plants (three for temperature and three for moisture). In the full models, different microhabitats were included as fixed factors, the number of ant and plant species as dependent variables, and location (i.e. doline 1, 2 and 3) as the random factor. We transformed the data of ants to binary scale (presence/absence) and used a binomial error term because each functional group had a high preference for one or a few sites and were very rare in other sites, leading to zero inflation of the data. No transformation was needed for plants, and we used Poisson or, if overdispersion was detected, negative binomial error term. GLMMs were performed in R using the glmer function of the ‘lme4’ package106. Full models were tested for significance with analysis of variance, using the Anova function of the ‘car’ package107. Pairwise comparisons of factor levels were undertaken with the relevel function and the FDR method (p.adjust function) was used to correct p values for multiple comparisons.
Data Availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Burnett, M. R., August, P. V., Brown, J. H. & Killingbeck, K. T. The influence of geomorphological heterogeneity on biodiversity. Conserv Biol 12, 363–370 (1998).
Stein, A., Gerstner, K. & Kreft, H. Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecol Lett 17, 866–880 (2014).
Keppel, G., Gillespie, T. W., Ormerod, P. & Fricker, G. A. Habitat diversity predicts orchid diversity in the tropical south-west Pacific. J Biogeogr 43, 2332–2342 (2016).
Fitzsimons, J. A. & Michael, D. R. Rocky outcrops: A hard road in the conservation of critical habitats. Biol Conserv 211, 36–44 (2017).
Ashcroft, M. B., Gollan, J. R., Warton, D. I. & Ramp, D. A novel approach to quantify and locate potential microrefugia using topoclimate, climatic stability, and isolation from the matrix. Glob Change Biol 18, 1866–1879 (2012).
Keppel, G., Anderson, S., Williams, C., Kleindorfer, S. & O’Connell, C. Microhabitats and canopy cover moderate high summer temperatures in a fragmented Mediterranean landscape. PLoS One 12, e0183106 (2017).
Meineri, E. & Hylander, K. Fine-grain, large-domain climate models based on climate station and comprehensive topographic information improve microrefugia detection. Ecography 40, 1003–1013 (2017).
Schmalholz, M. & Hylander, K. Microtopography creates small-scale refugia for boreal forest floor bryophytes during clear-cut logging. Ecography 34, 637–648 (2011).
Keppel, G. et al. A low-altitude mountain range as an important refugium for two narrow endemics in the Southwest Australian Floristic Region biodiversity hotspot. Ann Bot-London 119, 289–300 (2017).
Scherrer, D. & Körner, C. Infra-red thermometry of alpine landscapes challenges climatic warming projections. Glob Change Biol 16, 2602–2613 (2010).
Gaüzère, P., Princé, K. & Devictor, V. Where do they go? The effects of topography and habitat diversity on reducing climatic debt in birds. Glob Change Biol 23, 2218–2229 (2017).
Franklin, J. et al. Modeling plant species distributions under future climates: how fine scale do climate projections need to be? Glob Change Biol 19, 473–483 (2013).
Maclean, I. M. D., Suggitt, A. J., Wilson, R. J., Duffy, J. P. & Bennie, J. J. Fine‐scale climate change: modelling spatial variation in biologically meaningful rates of warming. Glob Change Biol 23, 256–268 (2017).
Taberlet, P. & Cheddadi, R. Quaternary refugia and persistence of biodiversity. Science 297, 2009–2010 (2002).
Keppel, G. et al. Refugia: identifying and understanding safe havens for biodiversity under climate change. Global Ecol Biogeogr 21, 393–404 (2012).
Tapper, S.-L. et al. Isolated with persistence or dynamically connected? Genetic patterns in a common granite outcrop endemic. Divers Distrib 20, 987–1001 (2014).
Keppel, G. & Wardell-Johnson, G. W. Refugia: keys to climate change management. Glob Change Biol 18, 2389–2391 (2012).
Tapper, S.-L. et al. Prolonged isolation and persistence of a common endemic on granite outcrops in both mesic and semi-arid Environments. J Biogeogr 41, 2032–2044 (2014).
Morelli, T. L. et al. Managing climate change refugia for climate adaptation. Plos One 11, e0159909 (2016).
Rull, V. Microrefugia. J Biogeogr 36, 481–484 (2009).
Dobrowski, S. Z. A climatic basis for microrefugia: the influence of terrain on climate. Glob Change Biol 17, 1022–1035 (2010).
Keppel, G. & Wardell-Johnson, G. W. Refugial capacity defines holdouts, microrefugia and stepping-stones: a response to Hannah et al. Trends Ecol Evol 30, 233–234 (2015).
Šegina, E., Benac, Č., Rubinić, J. & Knez, M. Morphometric analyses of dolines – the problem of delineation and calculation of basic parameters. Acta Carsologica 47, 23–33 (2018).
Hofierka, J., Gallay, M., Bandura, P. & Šašak, J. Identification of karst sinkholes in a forested karst landscape using airborne laser scanning data and water flow analysis. Geomorphology 308, 265–277 (2018).
Telbisz, T. et al. The advantage of lidar digital terrain models in doline morphometry compared to topographic map based datasets – Aggtelek karst (Hungary) as an example. Acta Carsologica 45, 5–18 (2016).
Whiteman, C. D., Haiden, T., Pospichal, B., Eisenbach, S. & Steinacker, R. Minimum temperatures, diurnal temperature ranges, and temperature inversion in limestone sinkholes of different sizes and shapes. J Appl Meteorol 43, 1224–1236 (2004).
Daly, C., Conklin, D. R. & Unsworth, M. H. Local atmospheric decoupling in complex topography alters climate change impacts. Int J Climatol 30, 1857–1864 (2010).
Novick, K. A., Oishi, A. C. & Miniat, C. F. Cold air drainage flows subsidize montane valley ecosystem productivity. Glob Change Biol 22, 4014–4027 (2016).
White, W. B., Culver, D. C., Herman, J. S., Kane, T. C. & Mylroie, J. E. Karst Lands. The dissolution of carbonate rock produces unique landscapes and poses significant hydrological and environmental concerns. Am Sci 83, 450–459 (1995).
Culver, D. C. Hotspots of subterranean biodiversity in caves and wells. J Cave Karst Stud 62, 11–17 (2000).
Clements, R. et al. Using biogeographical patterns of endemic land snails to improve conservation planning for limestone karsts. Biol Conserv 141, 2751–2764 (2008).
Antonić, O., Hatic, D. & Pernar, R. DEM-based depth in sink as an environmental estimator. Ecol Model 138, 247–254 (2001).
Wezel, A. Changes between 1927 and 2004 and effect of rock climbing on occurrence of Saxifraga paniculata and Draba aizoides, two glacial relicts on limestone cliffs of the Swabian Jura, southern Germany. J Nat Conserv 15, 84–93 (2007).
Molnár, A., Végvári, Z. & Tóthmérész, B. Identification of floral relicts based on spatial distance of isolation. Forests 8, 459 (2017).
Růžička, V. et al. Invertebrates of the Macocha Abyss (Moravian Karst, Czech Republic). Acta Carsologica 45, 71–84 (2016).
Reisch, C. Glacial history of Saxifraga paniculata (Saxifragaceae): molecular biogeography of a disjunct arctic-alpine species from Europe and North America. Biol J Soc 93, 385–398 (2008).
Erdős, L., Gallé, R., Bátori, Z., Papp, M. & Körmöczi, L. Properties of shrubforest edges: a case study from South Hungary. Cent Eur J Biol 6, 639–658 (2011).
Redžić, S., Barudanović, S., Trakić, S. & Kulijer, D. Vascular plant biodiversity richness and endemo-relictness of the karst mountains Prenj, Čvrsnica and Čabulja in Bosnia and Herzegovina (W. Balkan). Acta Carsologica 40, 527–555 (2011).
Kováč, L., Parimuchová, A. & Miklisová, D. Distributional patterns of cave Collembola (Hexapoda) in association with habitat conditions, geography and subterranean refugia in the Western Carpathians. Biol J Linn Soc 119, 571–592 (2016).
Sánchez, M., Alcocer, J., Escobar, E. & Lugo, A. Phytoplankton of cenotes and anchialine caves along a distance gradient from the northeastern coast of Quintana Roo, Yucatan Peninsula. Hydrobiologia 467, 79–89 (2002).
Surina, Ž. M. & Surina, B. Snowbed vegetation in Croatia: Phytosociology, ecology and conservation status. Plant Biosyst 144, 74–768 (2010).
Bátori, Z. et al. The conservation value of karst dolines for vascular plants in woodland habitats of Hungary: refugia and climate change. Int J Speleol 43, 15–26 (2014).
Favretto, D. & Poldini, L. The vegetation in the dolinas of the karst region near Trieste (Italy). Studia Geobotanica 5, 5–18 (1985).
Özkan, K., Gulsoy, S., Mert, A., Ozturk, M. & Muys, B. Plant distribution-altitude and landform relationships in karstic sinkholes of Mediterranean region of Turkey. J Environ Biol 31, 51–61 (2010).
Kobal, M., Bertoncelj, I., Pirotti, F., Dakskobler, I. & Kutnar, L. Using lidar data to analyse sinkhole characteristics relevant for understory vegetation under forest cover – case study of a high karst area in the Dinaric Mountains. Plos One 10, e0122070 (2015).
Su, Y., Tang, Q., Mo, F. & Xue, Y. Karst tiankengs as refugia for indigenous tree flora amidst a degraded landscape in southwestern China. Sci Rep-UK 7, 4249 (2017).
Beck v. Mannagetta, G. Die Umkehrung der Pflanzenregionen in den Dolinen des Karstes. Sitzungsberichte der Kaiserliche Akademie der Wissenschaften in Wien 65, 3–4 (1906).
Pericin, C. & Hürlimann, H. Beobachtungen zur vertikalen Verteilung der Moosarten in der Doline Sterna-Filaria im Karstgebiet von Buje/Buie in Istrien (Kroatien). Bauhinia 15, 91–96 (2001).
Kemencei, Z. et al. Microhabitat associations of land snails in forested dolinas: implications for coarse filter conservation. Community Ecol 15, 180–186 (2014).
Raschmanova, N., Miklisova, D., Kovac, L. & Sustr, V. Community composition and cold tolerance of soil Collembola in a collapse karst doline with strong microclimate inversion. Biologia 70, 802–811 (2015).
Bennie, J., Huntley, B., Wiltshire, A., Hill, M. O. & Baxter, R. Slope, aspect and climate: spatially explicit and implicit models of topographic microclimate in chalk grassland. Ecol Model 216, 47–59 (2008).
Lenoir, J. et al. Local temperatures inferred from plant communities suggest strong spatial buffering of climate warming across Northern Europe. Glob Change Biol 19, 1470–1481 (2013).
Shanks, R. E. & Norris, F. H. Microclimatic variation in a small valley in eastern Tennessee. Ecology 31, 532–539 (1950).
Wagner, R. D. T. Der Tagesgang der Lufttemperatur einer Doline im Bükk-Gebirge. Acta Climatologica 2-3, 49–79 (1963).
Bárány-Kevei, I. Microclimate of karstic dolines. Acta Climatologica 32–33, 19–27 (1999).
Clements, C. B., Whiteman, C. D. & Horel, J. D. Cold-air-pool structure and evolution in a mountain basin: Peter Sinks, Utah. J Appl Meteorol 42, 752–768 (2003).
Bátori, Z., Gallé, R., Erdős, L. & Körmöczi, L. Ecological conditions, flora and vegetation of a large doline in the Mecsek Mountains (South Hungary). Acta Bot Croat 70, 147–155 (2011).
Fridley, J. D. Downscaling climate over complex terrain: high finescale (<1000 m) spatial variation of near-ground temperatures in a montane forested landscape (Great Smoky Mountains). J Appl Meteorol Clim 48, 1033–1049 (2009).
Ashcroft, M. B. & Gollan, J. R. Moisture, thermal inertia, and the spatial distributions of near-surface soil and air temperatures: Understanding factors that promote microrefugia. Agr Forest Meteorol 176, 77–89 (2013).
Gargano, D., Vecchio, G. & Bernardo, L. Plant-soil relationships in fragments of Mediterranean snow-beds: ecological and conservation implications. Plant Ecol 207, 175–189 (2010).
Schmalholz, M. & Hylander, K. Microtopography creates small‐scale refugia for boreal forest floor bryophytes during clear‐cut logging. Ecography 34, 637–648 (2011).
Frey, S. J., Hadley, A. S. & Betts, M. G. Microclimate predicts within‐season distribution dynamics of montane forest birds. Divers Distrib 22, 944–959 (2016).
Bátori, Z. et al. Microclimate-vegetation relationships in natural habitat islands: species preservation and conservation perspectives. Időjárás 118, 257–281 (2014).
Bátori, Z. et al. Large- and small-scale environmental factors drive distributions of cool-adapted plants in karstic microrefugia. Ann Bot-London 119, 301–309 (2017).
Vilisics, F. et al. Small scale gradient effects on isopods (Crustacea: Oniscidea) in karstic sinkholes. Biologia 66, 499–505 (2011).
Wilkens, H. Regressive evolution and phylogenetic age: The history of colonization of freshwaters of Yucatan by fish and Crustacea. Association of Mexican Cave Studies Bulletin 8, 237–243 (1982).
Jaume, D., Boxshall, G. A. & Humphreys, W. F. New stygobiont copepods (Calanoida; Misophrioida) from Bundera Sinkhole, an anchialine cenote in north-western Australia. Zool J of the Linn Soc-Lond 133, 1–24 (2001).
Boxshall, G. A., Zylinski, S., Jaume, D., Iliffe, T. M. & Suárez-Morales, E. A new genus of speleophriid copepod (Copepoda: Misophrioida) from a cenote in the Yucatan, Mexico with a phylogenetic analysis at the species level. Zootaxa 3821, 321–336 (2014).
Wilson, R. J. et al. Changes to the elevational limits and extent of species ranges associated with climate change. Ecology Lett 8, 1138–1146 (2005).
Willis, K. J., Rudner, E. & Sümegi, P. The full-glacial forests of Central and southeastern Europe. Quaternary Res 53, 203–213 (2000).
McLaughlin, B. C. et al. Hydrologic refugia, plants, and climate change. Glob Change Biol 23, 2942–2961 (2017).
Mokany, K. et al. Past, present and future refugia for Tasmania’s palaeoendemic flora. J Biogeogr 44, 1537–1546 (2017).
Keppel, G. et al. The capacity of refugia for conservation planning under climate change. Front Ecol Environ 13, 106–112 (2015).
Totland, Ø. & Alatalo, J. M. Effects of temperature and date of snowmelt on growth, reproduction, and flowering phenology in the arctic/alpine herb, Ranunculus glacialis. Oecologia 133, 168–175 (2002).
Gentili, R. et al. Potential warm-stage microrefugia for alpine plants: Feedback between geomorphological and biological processes. Ecol Complex 21, 87–99 (2015).
Bellard, C., Bertelsmeier, C., Leadley, P., Thuiller, W. & Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol Lett 15, 365–377 (2012).
Bartholy, J., Pongrácz, R., Gelybó, G. & Szabó, P. Analysis of expected climate change in the Carpathian Basin using the PRUDENCE results. Időjárás 112, 249–264 (2008).
Gallé, L. Climate change impoverishes and homogenizes ants’ community structure: a long term study. Community Ecol 18, 128–136 (2017).
Tölgyesi, C. & Körmöczi, L. Structural changes of a Pannonian grassland plant community in relation to the decrease of water availability. Acta Bot Hung 54, 413–431 (2012).
Iijima, Y. & Shinoda, M. Seasonal changes in the cold-air pool formation in a subalpine hollow, central Japan. Int J Climatol 20, 1471–1483 (2000).
Maclean, I. M., Hopkins, J. J., Bennie, J., Lawson, C. R. & Wilson, R. J. Microclimates buffer the responses of plant communities to climate change. Global Ecol Biogeogr 24, 1340–1350 (2015).
Winkler, M. et al. The rich sides of mountain summits – a pan‐European view on aspect preferences of alpine plants. J Biogeogr 43, 2261–2273 (2016).
Keppel, G. et al. Towards an eco-evolutionary understanding of endemism hotspots and refugia. Ann Bot-London 122, 927–934 (2018).
Vojtkó, A. A Bükk hegység flórája. (Sorbus Kiadó, Eger, 2001).
Lazarević, P., Lazarević, M., Krivošej, Z. & Stevanović, V. On the distribution of Dracocephalum ruyschiana (Lamiaceae) in the Balkan Peninsula. Phytologia Balcanica 15, 175–179 (2009).
De Waele, J., Plan, L. & Auda, P. Recent developments in surface and subsurface karst geomorphology: An introduction. Geomorphology 106, 1–8 (2009).
Williams, P. Dolines in Encyclopedia of caves and karst science (ed. Gunn, J.) 628–642 (Fitzroy Dearborn, New York & London, 2004).
Veress, M. Solution DOLINE development on GLACIOKARST in alpine and Dinaric areas. Earth-Sci Rev 173, 31–48 (2017).
Bacsó, N. & Zólyomi, B. Mikroklíma és növényzet a Bükkfennsíkon. Az Időjárás 38, 177–196 (1934).
Jakucs, L. A karsztok morfogenetikája. A karsztfejlődés varienciái. (Akadémiai Kiadó, Budapest, 1971).
Borhidi A., Kevey, B. & Lendvai, G. Plant communities of Hungary. (Akadémiai Kiadó, Budapest, 2012).
Brian, M. V. Social insect populations. (Academic Press, London, 1965).
Andersen, A. N. Parallels between ants and plants: implications for community ecology in Ant-Plant interactions (eds Huxley, C. R. & Cutler, D. F.) 539–558 (Oxford University Press, Oxford, 1991).
López, F., Serrano, J. M. & Acosta, F. J. Parallels between the foraging strategies of ants and plants. Trends Ecol Evol 9, 150–153 (1994).
Johnson, R. A. Foundress survival and brood production in the desert seed-harvester ants Pogonomyrmex rugosus and P. barbatus (Hymenoptera, Formiicidae). Insectes Soc 45, 255–266 (1998).
Lőrinczi, G. Szubmediterrán erdei hangyaközösségek (Hymenoptera: Formicidae) tér- és időbeli szerveződése. (PhD értekezés, Szeged, 2014).
Seifert, B. Die Ameisen Mittel- und Nordeuropas. (Lutra & Vertriebsgesellschaft, Görlitz, 2007).
Czechowski, W., Radchenko, A., Czechowska, W. & Vepsäläinen, K. The ants (Hymenoptera, Formicidae) of Poland with reference to the myrmecofauna of Europe. (MIZ PAS, Warszawa, 2012).
Bolton, B. An online catalog of the ants of the world, http://antcat.org (2017).
Johnson, N. F. Hymenoptera Name Server (version 1.5), http://osuc.biosci.ohio-state.edu/ (2007).
Borhidi, A. Social behaviour types, the naturalness and relative ecological indicator values of the higher plants in the Hungarian flora. Acta Bot Hung 39, 9–181 (1995).
Tichy, L. & Chytry, M. Statistical determination of diagnostic species for site groups of unequal size. J Veg Sci 17, 809–818 (2006).
Tichý, L. JUICE, software for vegetation classification. J Veg Sci 13, 451–453 (2002).
R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, http://www.R-project.org/ (2018).
Oksanen, J. et al. Vegan: Community ecology, http://CRAN.R-project.org/package=vegan (2018).
Bates, D., Maechler, M. & Bolker, B. lme4: linear mixedeffects models using S4 classes. R package version 0.999999-2. http://cran.r-project.org/package=lme4 (2013).
Fox, J. & Weisberg, S. An {R} Companion to applied regression, Second Edition. Thousand Oaks CA: Sage, http://socserv.socsci.mcmaster.ca/jfox/Books/Companion (2011).
Acknowledgements
This research was supported by the NKFIH K 124796 grant. Z.B. was supported by the TÁMOP 4.2.4. A/2-11-1-2012-0001 ‘National Excellence Program’. G.K. was partially supported by an Alexander von Humboldt fellowship. G.M. was supported by the UNKP-18-3-I-SZTE-74 ‘New National Excellence Program’ of the Ministry of Human Capacities of Hungary. We are thankful to Anna Tenyér, Gábor Horváth and Imola Bóni for their help in field works. Thanks to the Bükk National Park for permission to carry out this work on land under their care. We thank to Gema Trigos-Peral for assistance in ant identification.
Author information
Authors and Affiliations
Contributions
Z.B., A.V., G.L. and I.E.M. conceived and designed the study. Z.B., T.F., P.J.K., O.J., N.K., I.E.M., G.M., G.L, E.T. and A.V. performed field surveys and collected the data. A.V., T.F. and Z.B. identified the plants; G.L. identified the ants. Z.B., C.T., I.E.M., L.E. and D.J.A. analysed the data; Z.B., G.K., C.T. and I.E.M. prepared the figures, and wrote the manuscript. All the authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bátori, Z., Vojtkó, A., Maák, I.E. et al. Karst dolines provide diverse microhabitats for different functional groups in multiple phyla. Sci Rep 9, 7176 (2019). https://doi.org/10.1038/s41598-019-43603-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43603-x
This article is cited by
-
Forest age and topographic position jointly shape the species richness and composition of vascular plants in karstic habitats
Annals of Forest Science (2023)
-
The influence of illumination regimes on the structure of ant (Hymenoptera, Formicidae) community composition in urban habitats
Insectes Sociaux (2023)
-
Functional composition of ant assemblages in habitat islands is driven by habitat factors and landscape composition
Scientific Reports (2021)
-
Plant adaptability in karst regions
Journal of Plant Research (2021)
-
Anthropogenic disturbances alter the conservation value of karst dolines
Biodiversity and Conservation (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.