Abstract
Measures characterizing the complexity of heart rate (HR) dynamics have been informative in predicting age- and disease-related decline in cardiovascular health, but few studies have evaluated whether mind-body exercise can impact HR complexity. This study evaluated the effects of long-term Tai Chi (TC) practice on the complexity of HR dynamics using an observational comparison of TC experts and age- and gender-matched TC-naïve individuals. Shorter-term effects of TC were assessed by randomly assigning TC-naïve participants to either TC group to receive six months of TC training or to a waitlist control group. 23 TC experts (age = 63.3 ± 8.0 y; 24.6 ± 12.0 y TC experience) and 52 TC-naïve (age = 64.3 ± 7.7 y) were enrolled. In cross-sectional analyses, TC experts had a higher overall complexity index (CI, p = 0.004) and higher entropy at multiple individual time scales (p < 0.05); these findings persisted in models accounting for age, gender, body mass index (BMI), and physical activity levels. Longitudinal changes in complexity index did not differ significantly following random assignment to six months of TC vs. a waitlist control; however, within the TC group, complexity at select time scales showed statistically non-significant trends toward increases. Our study supports that longer-term TC mind-body training may be associated with increased complexity of HR dynamics.
Similar content being viewed by others
Introduction
Human heart beat dynamics are influenced by the interaction of numerous factors, including the parasympathetic and sympathetic branches of the autonomic nervous system, hormones, body temperature, physical activity, digestion and circadian rhythms1. These processes impact heart rate dynamics across a broad range of time scales. Consequently, heart rate dynamics are exceedingly complex2. Indeed, a large body of research supports the concept that healthy heart rate rhythms exhibit features of complex systems reflecting dynamic, non-stationary, nonlinear properties3. Research also demonstrates that the complexity of heart rate dynamics decreases with age and disease4,5. For example, studies using multiscale entropy (MSE)––one widely used metric of complexity––observe age-related decline in heart rate complexity in healthy adults above the age of 40 years6,7. Compared to healthy adults, lower MSE of heart rate dynamics has also been associated with the presence of cardiac disease8,9,10,11, post-surgical complications12,13, mood disorders14,15, heart failure16,17, and overall cardiovascular related mortality in elderly adults18. In many of these and other studies, complexity-based metrics of heart rate dynamics have demonstrated better prognostic power compared with traditional measures of heart rate variability (HRV). However, to date, little research has evaluated whether the loss of cardiovascular physiological complexity associated with aging and age-related disease can be attenuated or restored with non-pharmacological interventions.
Tai Chi is a multimodal mind-body exercise with historical roots in traditional Chinese medicine. Its goal of impacting multiple physiological systems and integrating their dynamics make it especially well-suited for evaluation within a complexity framework19,20,21. Tai Chi is reported to improve or attenuate decline in many age-related health issues, including cardiovascular health22,23. Meta-analyses and systematic reviews support that Tai Chi may benefit multiple risk factors and clinically relevant outcomes related to cardiovascular health including: lowering systolic and diastolic blood pressure24; lowering levels of triglycerides, low-density lipoprotein (LDL), and serum B-type natriuretic peptide (BNP)25,26; and improving exercise capacity27,28,29,30, disease-related quality of life31, mood32, sleep quality33, and exercise self-efficacy27,34. Tai Chi may also reduce inflammatory markers known to contribute to coronary artery disease (CAD)35,36. A handful of smaller studies have also evaluated the impact of Tai Chi on a variety of time and frequency domain measures of HRV, however, the conclusions of these studies are mixed and difficult to interpret37,38,39,40,41. It remains unknown whether Tai Chi can impact complexity-based measures of heart rate dynamics.
As part of a larger study evaluating the impact of Tai Chi19 on physiological complexity and healthy aging, the current work evaluates the impact of both long- and short-term Tai Chi training on MSE of heart rate dynamics monitored during 5 minute bouts of natural breathing under controlled experimental conditions. Potential long-term effects of TC training were evaluated through cross-sectional comparisons of healthy Tai Chi naïve adults and age- and gender-matched expert Tai Chi practitioners. Tai Chi short-term effects were evaluated by random assignment of the Tai Chi naïve healthy participants to either six months of Tai Chi plus usual care or to usual care alone. Based on our previously completed studies, we hypothesized that (1) Tai Chi experts may have greater MSE of heart rate dynamics, compared to age- and gender-matched controls; and (2) Tai Chi naïve older adults who were randomized to six months of Tai Chi would subsequently show greater MSE of heart rate dynamics compared to a wait-list control. For comparison with MSE, traditional time domain measures of HRV were also evaluated.
Results
Participant characteristics and flow through the trial
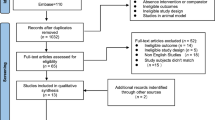
Recruitment of both Tai Chi experts (N = 27) and Tai Chi naïve individuals (N = 60) took place between March 2011 and March 2013, and follow-up assessments were completed in September 2013. Of the 27 Tai Chi experts, data from 4 were discarded due to artifacts in the ECG signals providing an analyzed sample of 23 individuals. The average training experience of Tai Chi experts was 24.6 ± 12.0 years (median 20 yrs, range 10–50 yrs). Tai Chi naïve subjects were overall well-matched with Tai Chi experts in respect to age, reported presence of hypertension, and global cognitive status. However, Tai Chi experts showed higher physical activity levels, lower body mass index (BMI), and were represented by a slightly higher proportion of men and Asians. Tai Chi naïve subjects who were subsequently randomized to the Tai Chi group versus the usual care control group were comparable at baseline (Table 1).
Figure 1 summarizes the flow of participants in the randomized controlled trial included in this study. As a previously published study reported42, sixty healthy adults were enrolled. Among them, 28 of 29 (97%) individuals in the Tai Chi group, and 27 of 31 (87%) individuals in the usual care group completed the 6-month follow-up assessment. Data from 8 Tai Chi naïve participants were excluded due to the loss of follow-up (n = 5) or the low ECG quality (artifacts or excessive ectopic beats, n = 3), resulting in 26 subjects randomized to each of the Tai Chi and waitlist control groups with analyzable baseline and 6 month data.
Cross-sectional comparisons
Compared to the novices, Tai Chi experts exhibited an overall higher MSEmod complexity index over 20 scales (CI1–20) compared to Tai Chi naïve controls (CI1–20 of 1.69 ± 0.13 vs. 1.57 ± 0.18, p = 0.004) (Fig. 2). This group difference remained statistically significant in linear models adjusting for age, BMI, and physical activity (P = 0.002). Secondary analyses evaluating individual scales of the MSEmod suggest significant differences at most scales (scale 3–17) in analyses not including Bonferroni adjustments for multiple comparisons. Differences at scales 3–7 remained significant following Bonferroni adjustments. Similar findings were observed in models adjusting for age, BMI, and physical activity. Tai Chi experts also exhibited longer breathing intervals (p = 0.003). After adjustment for age, BMI, activity level, and breathing intervals, between groups differences in complexity index were still significant (p = 0. 018).
Baseline complexity compared between experts and naïve, presented by mean and standard error of the group. (a) Unadjusted p values calculated from t-test. (b) Adjusted p values calculated from linear model adjusting for age, BMI, and physical activity. (c) Adjusted p values calculated from linear model adjusting for age, BMI, physical activity and BI. Abbreviations: BI, intervals of breathing peaks; NN, normal-to-normal heartbeat intervals; CI1–20, complexity index calculated by the mean entropy of 20 scales. Complexity index (CI) curves for Tai Chi experts and Tai Chi naïve evaluated cross-sectionally at baseline. Values represent mean and standard error for each scale. Embedded table summarizes statistics for comparisons. Significantly different comparisons at individual scales (following Bonferroni adjustments) are denoted with an asterisk (*).
In contrast, conventional time domain parameters did not show any difference between Tai Chi experts and naïve controls, even after adjusting for age, BMI, level of activity and breathing (Table 2).
Longitudinal comparisons
There was a trend towards a greater increase in the overall MSEmod complexity index (CI1–20) in healthy TC naïve adults randomly assigned to 6 months of Tai Chi (1.52 ± 0.2 to 1.58 ± 0.13) versus usual care group (1.63 ± 0.13 to 1.65 ± 0.14). However, within (baseline vs. 6 month) and between (Tai Chi vs. naïve) group differences were not statistically significant based on all linear models evaluated. When MSEmod was further explored and analyzed on individual scales (Fig. 3), within group differences in those randomized to Tai Chi exhibited trends towards increased entropy on a subset of larger scales (scales 11–17), with differences being statistically significant at scales 13 (p = 0.047), 14 (p = 0.024) and 15 (p = 0.034) in analyses not including Bonferroni adjustments for multiple comparisons. However, after adjustments for multiple comparisons, these differences were no longer significant. In contrast, no notable changes in individual complexity scales in the control group were observed after 6 months.
There was a trend towards longitudinal increases in breathing intervals (i.e. slower breathing) in the Tai Chi group (5.0 ± 1.87 to 5.53 ± 3.16 seconds), compared to the control group (5.28 ± 1.66 to 4.97 ± 1.61 seconds). However, within- and between-group differences were not statistically significant in any of the models.
In contrast to MSEmod, conventional time domain parameters of HRV did not show any trends when longitudinal comparisons were made between those randomly assigned to Tai Chi versus usual care (Table 3).
Discussion
In our cross-sectional comparisons, we observed that long-term training in TC is associated with higher (i.e., more complex) MSEmod of heart rate dynamics across a wide range of scales. Importantly, statistical differences between TC experts and TC naive controls persisted even when multiple potential confounders were taken into account. In our randomized trial, we observed that a 6-month exposure to Tai Chi resulted in statistically non-significant within-group trends towards increases in MSE at a subset of MSE scales, however, between-group differences in longitudinal changes were not significant. Of note, no differences were found in average heart rate or conventional time domain HRV parameters for either long-term or short-term Tai Chi exposures. Together, these findings add to a growing body of research supporting that Tai Chi can have positive effects on markers and risk factors associated with cardiovascular health43,44. Moreover, along with other recent studies reporting an association of Tai Chi training with enhanced physiological complexity of balance45,46 and gait dynamics42, our findings support the general value of complexity-based indices for informing the impact of Tai Chi on health.
Ageing and disease are commonly associated with multi-system impairment4, resulting in complex changes in physiological dynamics and function5,47. For this reason, it has been argued that complexity-based metrics that provide indices related to the magnitude of physiological ‘cross-talk’, feedback and multi-scale regulation, might serve as sensitive and meaningful biomarkers of age- and disease-related decline5. While many studies have confirmed the value of complexity-based measures for characterizing and predicting age- and disease-related decline based on the dynamics of a variety of physiological signals such as heart rate48,49,50, blood pressure51,52,53, center of pressure45,46,54, stride time42, and gait variability55, surprisingly few studies to date have evaluated the potential of interventions to attenuate or restore age- or disease-related decline in complexity. With respect to heart rate dynamics, one 8-week study evaluated administration of the anti-psychotic clozapine for treatment-resistant schizophrenia. This study reported that positive clinical responses were correlated with increases in complexity of heart rate dynamics measured with approximate and sample entropy56. Another small study in children affected by a spectrum of neurological disorders reported that short-term exposure to hippotherapy (therapeutic horse riding) led to greater post-treatment sample entropy of HRV57. A third non-controlled study evaluating a meditation technique reported higher sample entropy in heart beat dynamics following an individual meditation session58. Our study extends this small body of research by both evaluating a novel mind-body exercise intervention, and by demonstrating potential impacts on heart rate dynamics well beyond acute, single-session exposure effects.
Compared to robust cross-sectional differences observed between Tai Chi experts and naive controls in MSEmod, we observed only modest and statistically non-significant differences over time in MSEmod for TC naive adult participants randomized to six months of TC training vs. a waitlist control. This smaller effect may reflect inadequate dose (too short a duration and/or too low an intensity of training) of Tai Chi to alter fundamental HR dynamics. The fact that the population in our study was very healthy and already physically active may have further contributed to the observed small effect. These findings parallel those observed in gait and balance dynamics outcomes in the same population, with only modest effects observed after 6 months of Tai Chi exposure42,45,55,59. Future studies assessing the impact of TC practice on HR complexity might consider evaluating less healthy populations, and administering greater intensities and longer exposures of Tai Chi training.
The complexity of physiologic control systems is believed to be important, as it affords the organism resilience leading to better adaptation to a specific task or an external stress4,60. To date, existing evidence to support this theory is very limited. In elderly people, studies have shown that the magnitude of complexity exhibited within center of pressure dynamics during quiet standing is correlated with the ability to balance while challenged with a mental dual task (i.e., counting backwards)61. With regards to heart rate dynamic measures evaluated in this study, future studies might also explore if the Tai Chi-related increases in MSEmod are associated with enhanced resiliency in heart rate or blood pressure responses during sit-to-stand challenges or acute bouts of aerobic exercise.
In contrast to the observed associations between Tai Chi training and complexity of heart rate dynamics, we observed no long- or short-term effects of Tai Chi on traditional time domain measures of HRV. Indices of HRV based on time domain methods have been shown to be predictive of adverse outcomes in patients with heart disease62,63,64 and in the general population. However, the evidence that Tai Chi and related mind-body studies enhance time domain measures of HRV are quite mixed37, and many rely only on short-term (post single session) acute effects39,41,65,66. In studies evaluating the longer-term impact of Tai Chi training, some studies report increased HRV67,68, while others report no effect27,40,69, or even reduced variability70. Part of this heterogeneity may be attributed to the confounding effects of respiratory rate on traditional HRV measure. It is well established that changes in respiratory frequency can affect tidal volume (e.g., lower respiratory rate is associated with greater tidal volumes) and thus the amplitude of heart rate oscillations via a respiratory sinus arrhythmia (RSA) mechanism. Traditionally, this vagally-mediated process is captured in the high frequency (HF) component of HRV. However, in our cohort of Tai Chi experts, the mean breathing interval was 6.79 s corresponding to a rate of 0.147 Hz or 8.8 breaths/min, compared to the 5.17 seconds or 0.193 Hz (11.6 breaths/min) seen in the control group. As a consequence, the vagally-mediated RSA would be categorized into the low frequency (LF) component (0.04–0.15 Hz) of HRV for many within the Tai Chi group, whereas it would be appropriately categorized into the HF HRV component (0.15–0.40 Hz) for most in the control group. The reduced respiratory frequency observed in the Tai Chi group not only increases RSA-associated HRV but can also lead to mis-categorization of the parasympathetic nervous activity into the LF band. This confounding effect of respiration on HRV may explain the variability in the reported effects of Tai Chi on HRV, and indeed, the majority of studies evaluating the impact of Tai Chi on HRV do not account for breathing65,66,67,69.
Multiscale Entropy, on the other hand, remains relatively robust to respiratory rate as evidenced by the sustained statistically significant difference in MSEmod, even after accounting for differences in breathing intervals in the Tai Chi experts group and TC naïve group (Fig. 2). The MSEmod complexity index (CI1–20) employed in this study is essentially a composite score that theoretically represents the summative effects of physiological control (including, but not exclusive to, respiratory contributions) of HRV in the time domain of ~0.8 to 25 seconds (depending on the mean HR). As a result, the application of MSE does not require an additional process to correct for respiratory confounding – such as paced breathing which mandates an artificially forced condition onto the body. In addition, the scales utilized in MSE are not a function of time, but rather a function of the number of RR (or NN) intervals. For instance, Scale 2 SampEn is derived by utilizing the coarse-grained time series for averages of two consecutive RR-intervals. MSE utilizes heart beat (and not time) as the frame of reference, and thus avoids the need for an arbitrarily-chosen range of frequencies to capture specific physiological processes. Whether this approach yields more physiologically-consonant results compared to a time-based approach remains to be further tested. Finally, MSE has the additional benefit of extracting information from the sequential order of RR-intervals. Traditional HRV measures, both time- and frequency-based, are linear measures that simply reveal the distribution of the data, whether it be the spectral power or number of RR-intervals in a specific time range. The failure to account for the sequential order (i.e., what RR interval follows a previous one) may explain why traditional HRV measures are not able to effectively capture differences in health-states when compared to complexity-based metrics such as MSE.
This study may have a number of limitations. First, sample sizes were small for both cross-sectional comparisons and randomized controlled trial (RCT). In addition, as with any observational study, comparisons may be confounded by differences between groups other than exposure to Tai Chi. Although our statistical models accounted for multiple confounders, there may still be other factors not measured or included. There was not an active control in our RCT, thus is possible that observed effects were due to social support and/or placebogenic beliefs. Future studies evaluating Tai Chi on complexity measures of heart rate dynamics should consider employing active comparative controls. Additionally, it may be that six months of Tai Chi training is an insufficient dose to significantly change heart rate dynamics in healthy adults. Finally, given the 5-minute duration of the RR-interval data, the physiological significance of MSEmod at higher scales (up to Scale 20) remain unclear, and the full dynamics of the heart rate variability at these lower frequencies may be under-represented in our results and analyses.
Conclusions
Long-term Tai Chi mind-body training may be associated with increased complexity of HR dynamics in healthy adults, as measured with MSEmod. Compared to traditional time domain HRV measures of cardiovascular health, MSEmod appears to be a more sensitive in evaluating the impact of a multi-modal mind-body exercise.
Materials and Methods
Study Design
Design features of this study have been reported elsewhere19,42,45,59. This study was approved by the Institutional Review Boards of Beth Israel Deaconess Medical Center and Brigham and Women’s Hospital, and conducted in accordance with the relevant guidelines and regulations. The randomized trial component of this study was registered at ClinicalTrials.gov (NCT01340365, date of registration: April 22nd, 2011).
Our study design that integrated a two-arm RCT with an cross-sectional comparison group at baseline19. Reports of other outcomes from this study have already been published34,42,45,55,59: The Tai Chi naïve group was composed of 60 adults, age 50–79 years, and reporting no consistent training in Tai Chi in the prior 5 years. Exclusion criteria included: 1) chronic medical condition (e.g. cardiovascular disease, cancer; neurological conditions); 2) acute conditions requiring hospitalization in the prior 6 months; 3) inability to walk unassisted and continuously for 15 minutes; and 4) engagement in physical exercise four or more times in a given week. Eligible participants provided written informed consent and were randomized (1:1) to either 6 months of Tai Chi training in combination with usual healthcare, or usual healthcare alone. Randomization was age-stratified (50–59, 60–69, 70–79 years) utilizing a permuted-blocks scheme with randomly varying block sizes. Outcomes were assessed at baseline and 6 months. Staff overseeing and conducting the assessments were blinded to treatment assignment. Particpants assigned to the Tai Chi group were required to attend at least 2 study-sanctioned community-based Tai Chi classes each week and engage in self-practice at home for 30 min on 2 additional days each week17. Tai Chi class attendance monitored by instructors and compliance with home practice was tracked with self-completed logs.
The TC expert group included 27 adults (age 50–79 yrs) currently engaged in ongoing Tai Chi training, each with at least 5 years of experience. Other than the restriction on Tai Chi practice, procedures for identifying and enrolling Tai Chi experts were identical to the Tai Chi naïve group.
Measurements
All testing was performed in the Syncope and Falls in the Elderly (SAFE) laboratory, located in Beth Israel Deaconess Medical Center (Boston, MA). Outcomes related to heart rate dynamics were just one components of a more comprehensive battery of tests (including balance45, gait55, and cognition59). Characterization of heart rate dynamics employing MSE was defined a priori as a primary outcome measure19. Tai Chi experts evaluated in longer-term comparisons were assessed at only one point in time. Tai Chi naive participants were assessed at baseline and at 6 months following randomization to either Tai Chi or a wait list control. For all subjects, demographic, anthropometric, and relevant medical histories data were obtained at screening visits.
Heart rate dynamics were assessed using a standardized testing protocol employed in prior studies71,72. Subjects were asked to refrain from caffeine on the day of testing. Following a 10-minute resting period, electrocardiogram (ECG) and respiratory signals from CO2 recordings were collected while participants laid supine and were instructed to breathe naturally for 5 minutes. ECG and respiratory signals were sampled at 500 Hz. Consecutive R peaks were detected from raw ECG recordings and the time series were extracted as RR intervals for analyses proposed in this study50. Ectopic beats were detected and removed from calculations as in previous studies50. ECG data with arrhythmias or artifacts that comprised greater than 5% of the total epoch were removed. Qualified clean data were used for analyses in the presented study. Respiration cycles were detected by identifying consecutive peaks of the respiratory signal, from which mean breathing intervals (BI) were calculated73,74. Breathing intervals were recorded to inform potential differences in heart rate complexity between participants with and without exposure to Tai Chi, which typically includes instruction in breathing. Since lower breathing frequencies are associated with increased tidal volumes, increased breathing intervals are typically associated with increased respiratory sinus arrhythmia, which may ultimately affect the overall HR complexity value.
Estimate of Multiscale Entropy of Heart Rate Dynamics
Multiscale entropy (MSE) of HR dynamics utilizes Sample Entropy (SampEn) to quantify the degree of irregularity within a time-series (e.g. RR or NN intervals) across multiple time scales45. For relatively longer time series (e.g., hours), MSE first divides the original signal into non-overlapping segments of length τ, then averages the data points inside each segment, which is the “coarse-graining” process48. SampEn is calculated for each coarse-grained time series at different scales. The commonly used parameters for MSE are m = 2, r = 15% of RR intervals’ standard deviation. The traditional coarse-graining process shortens the time series data with each increasing time scale and thus limits the extent to which SampEn can accurately be calculated at larger time scales. Based on the data we collected in this study, we calculated MSE using a validated modified approach, referred to as modified multiscale entropy (MSEmod)75,76. MSEmod was developed to specifically be suitable for shorter-term time series and to provide stable, reliable, and precise estimates for short-term time series. MSEmod adheres to most of the conventional MSE algorithm, but replaces the coarse-graining procedure with a moving average procedure, and a time delay was incorporated for constructing template vectors in calculating sample entropy75. This ensures that sufficient data are maintained for entropy calculation, even at large scales (i.e., lower frequencies) and for shorter time series data (Supplementary file 1). In this study, parameters were set as m = 2, r = 15% of RR intervals’ standard deviation. Entropies were calculated up to 20 scales, and a complexity index was calculated as mean of the entropy values of scale 1–20 (CI1–20).
Time Domain Parameters of Heart Rate Variability
Analyzed time domain parameters included mean NN intervals (NN), standard deviation of NN intervals (SDNN), mean heart rate (HR), root mean square of the successive differences (RMSSD), percentage of adjacent NN intervals that differ by greater than 20 milliseconds (pNN20) and 50 milliseconds (pNN50).
Baseline characteristics
Sociodemographic variables collected at baseline included age, height, weight, race and ethnicity, medical history, current medication use, diet, smoking and alcohol history, education, marital status, employment status, and level of physical activity. Physical activity level at baseline was evaluated by the physical activity status scale (PASS)77. Baseline global cognitive function was evaluated using the Mini Mental State Exam59.
Statistical analyses
Statistical analyses were performed using SPSS 19.0 (IBM SPSS Statistics). Continuous data was tested for normality of the distribution, and descriptive statistics were presented as mean ± standard deviation for normal distributed data. Categorical data were reported as umber (percentage). Between-group comparisons of baseline characteristics were assessed by chi-square test for categorical variables. Cross-sectional comparisons were assessed by linear regression models including three levels of potential confounders between TC experts and TC naïve control subjects. Model 1 was based on raw unadjusted data. Model 2 incorporated the potential confounding effects of age, BMI, and levels of physical activity following analytic methods employed to evaluate other outcomes based on this same experimental design and study population42,45,55,59. Model 3 included breathing intervals in addition to Model 2 variables.
Longitudinal comparisons in the randomized trial of this study were evaluated by baseline-adjusted linear regression models including three levels of potential confounders. Model 1 was based on raw unadjusted data. Model 2 included the potential confounders of age and gender as in our prior studies42,45,55,59. Model 3 included breathing intervals in addition to Model 2 variables.
References
Goldberger, A. L. Is the normal heartbeat chaotic or homeostatic? News in physiological sciences: an international journal of physiology produced jointly by the International Union of Physiological Sciences and the American Physiological Society 6, 87–91 (1991).
Kaplan, D. T. et al. Aging and the complexity of cardiovascular dynamics. Biophysical journal 59, 945–949 (1991).
Stanley, H. E. et al. Fractal landscapes in biological systems: Long-range correlations in DNA and interbeat heart intervals. Physica A: Statistical Mechanics and its Applications 191, 1–12 (1992).
Lipsitz, L. A. Aging as a process of complexity loss. Complex systems science in biomedicine, 641–654 (2006).
Goldberger, A. L., Peng, C. K. & Lipsitz, L. A. What is physiologic complexity and how does it change with aging and disease? Neurobiology of aging 23, 23–26 (2002).
Beckers, F., Verheyden, B. & Aubert, A. E. Aging and nonlinear heart rate control in a healthy population. American journal of physiology. Heart and circulatory physiology 290, H2560–2570 (2006).
Pikkujamsa, S. M. et al. Cardiac interbeat interval dynamics from childhood to senescence: comparison of conventional and new measures based on fractals and chaos theory. Circulation 100, 393–399 (1999).
Angelini, L. et al. Multiscale analysis of short term heart beat interval, arterial blood pressure, and instantaneous lung volume time series. Artificial intelligence in medicine 41, 237–250 (2007).
Gomez-Garcia, J. A., Martinez-Vargas, J. D. & Castellanos-Dominguez, G. Complexity-based analysis for the detection of heart murmurs. Conference proceedings. Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Conference 2011, 2728–2731 (2011).
Norris, P. R., Stein, P. K. & Morris, J. A. Jr. Reduced heart rate multiscale entropy predicts death in critical illness: a study of physiologic complexity in 285 trauma patients. Journal of critical care 23, 399–405 (2008).
Vikman, S. et al. Altered complexity and correlation properties of R-R interval dynamics before the spontaneous onset of paroxysmal atrial fibrillation. Circulation 100, 2079–2084 (1999).
Makikallio, T. H. et al. Heart rate dynamics before spontaneous onset of ventricular fibrillation in patients with healed myocardial infarcts. The American journal of cardiology 83, 880–884 (1999).
Makikallio, T. H. et al. Dynamic analysis of heart rate may predict subsequent ventricular tachycardia after myocardial infarction. The American journal of cardiology 80, 779–783 (1997).
Leistedt, S. J. et al. Decreased neuroautonomic complexity in men during an acute major depressive episode: analysis of heart rate dynamics. Translational psychiatry 1, e27 (2011).
Nardelli, M., Lanata, A., Bertschy, G., Scilingo, E. P. & Valenza, G. Heartbeat Complexity Modulation in Bipolar Disorder during Daytime and Nighttime. Scientific reports 7, 17920 (2017).
Ho, K. K. et al. Predicting survival in heart failure case and control subjects by use of fully automated methods for deriving nonlinear and conventional indices of heart rate dynamics. Circulation 96, 842–848 (1997).
Ho, Y. L., Lin, C., Lin, Y. H. & Lo, M. T. The prognostic value of non-linear analysis of heart rate variability in patients with congestive heart failure–a pilot study of multiscale entropy. PloS one 6, e18699 (2011).
Lin, Y. H. et al. Multi-scale symbolic entropy analysis provides prognostic prediction in patients receiving extracorporeal life support. Critical care (London, England) 18, 548 (2014).
Wayne, P. M. et al. A systems biology approach to studying Tai Chi, physiological complexity and healthy aging: design and rationale of a pragmatic randomized controlled trial. Contemporary clinical trials 34, 21–34 (2013).
Ma, Y., Sun, S. & Peng, C. K. Applications of dynamical complexity theory in traditional Chinese medicine. Frontiers of medicine 8, 279–284 (2014).
Ma, Y., Zhou, K., Fan, J. & Sun, S. Traditional Chinese medicine: potential approaches from modern dynamical complexity theories. Frontiers of medicine 10, 28–32 (2016).
Taylor-Piliae, R. E. Tai Chi as an adjunct to cardiac rehabilitation exercise training. J Cardiopulm Rehabil 23, 90–96 (2003).
Yeh, G. et al. Effects of Tai-Chi mind-body movement therapy on functional status and exercise capacity in patients with chronic heart failure: a randomized controlled trial. Am J Med 117, 541–548 (2004).
Yeh, G. Y., Wang, C., Wayne, P. M. & Phillips, R. S. The effect of tai chi exercise on blood pressure: a systematic review. Preventive cardiology 11, 82–89 (2008).
Yeh, G. Y., Wayne, P. M. & Phillips, R. S. T’ai Chi exercise in patients with chronic heart failure. Medicine and sport science 52, 195–208 (2008).
Yeh, G. Y. et al. Effects of tai chi mind-body movement therapy on functional status and exercise capacity in patients with chronic heart failure: a randomized controlled trial. The American journal of medicine 117, 541–548 (2004).
Yeh, G. Y. et al. Tai chi exercise in patients with chronic heart failure: a randomized clinical trial. Archives of internal medicine 171, 750–757 (2011).
Yeh, G. Y. et al. Tai chi exercise for patients with chronic obstructive pulmonary disease: a pilot study. Respiratory care 55, 1475–1482 (2010).
Yeh, G. Y. et al. Tai chi mind-body exercise in patients with COPD: study protocol for a randomized controlled trial. Trials 15, 337 (2014).
Yeh, G. Y., Wang, C., Wayne, P. M. & Phillips, R. Tai chi exercise for patients with cardiovascular conditions and risk factors: A SYSTEMATIC REVIEW. Journal of cardiopulmonary rehabilitation and prevention 29, 152–160 (2009).
Wayne, P. M. et al. Can Tai Chi improve vestibulopathic postural control? Archives of physical medicine and rehabilitation 85, 142–152 (2004).
Yeung, A. et al. Tai chi treatment for depression in Chinese Americans: a pilot study. American journal of physical medicine & rehabilitation/Association of Academic Physiatrists 91, 863–870 (2012).
Ma, Y. et al. The Effects of Tai Chi on Sleep Quality in Chinese American Patients With Major Depressive Disorder: A Pilot Study. Behavioral sleep medicine, 1–17 (2016).
Yeh, G. Y., Chan, C. W., Wayne, P. M. & Conboy, L. The Impact of Tai Chi Exercise on Self-Efficacy, Social Support, and Empowerment in Heart Failure: Insights from a Qualitative Sub-Study from a Randomized Controlled Trial. PloS one 11, e0154678 (2016).
Bower, J. E. & Irwin, M. R. Mind-body therapies and control of inflammatory biology: A descriptive review. Brain, behavior, and immunity 51, 1–11 (2016).
Morgan, N., Irwin, M. R., Chung, M. & Wang, C. The effects of mind-body therapies on the immune system: meta-analysis. PloS one 9, e100903 (2014).
Cole, A. R., Wijarnpreecha, K., Chattipakorn, S. C. & Chattipakorn, N. Effects of Tai Chi exercise on heart rate variability. Complementary therapies in clinical practice 23, 59–63 (2016).
Lu, X., Hui-Chan, C. W. & Tsang, W. W. Changes of heart rate variability and prefrontal oxygenation during Tai Chi practice versus arm ergometer cycling. Journal of physical therapy science 28, 3243–3248 (2016).
Wei, G. X. et al. Tai Chi Chuan modulates heart rate variability during abdominal breathing in elderly adults. PsyCh journal 5, 69–77 (2016).
Sato, S., Makita, S., Uchida, R., Ishihara, S. & Masuda, M. Effect of Tai Chi training on baroreflex sensitivity and heart rate variability in patients with coronary heart disease. International heart journal 51, 238–241 (2010).
Lu, W. A. & Kuo, C. D. The effect of Tai Chi Chuan on the autonomic nervous modulation in older persons. Medicine and science in sports and exercise 35, 1972–1976 (2003).
Gow, B. J. et al. Can Tai Chi training impact fractal stride time dynamics, an index of gait health, in older adults? Cross-sectional and randomized trial studies. PloS one 12, e0186212 (2017).
Hartley, L., Flowers, N., Lee, M. S., Ernst, E. & Rees, K. Tai chi for primary prevention of cardiovascular disease. The Cochrane database of systematic reviews, CD010366 (2014).
Lan, C., Chen, S. Y., Lai, J. S. & Wong, A. M. Tai chi chuan in medicine and health promotion. Evidence-based complementary and alternative medicine: eCAM 2013, 502131 (2013).
Wayne, P. M. et al. Complexity-Based Measures Inform Effects of Tai Chi Training on Standing Postural Control: Cross-Sectional and Randomized Trial Studies. PloS one 9, e114731 (2014).
Manor, B., Lipsitz, L. A., Wayne, P. M., Peng, C. K. & Li, L. Complexity-based measures inform Tai Chi’s impact on standing postural control in older adults with peripheral neuropathy. BMC complementary and alternative medicine 13, 87 (2013).
Lipsitz, L. A. Age-related changes in the “complexity” of cardiovascular dynamics: A potential marker of vulnerability to disease. Chaos (Woodbury, N.Y.) 5, 102–109 (1995).
Costa, M., Goldberger, A. L. & Peng, C. K. Multiscale entropy analysis of complex physiologic time series. Physical review letters 89, 068102 (2002).
Costa, M., Goldberger, A. L. & Peng, C. K. Multiscale entropy analysis of biological signals. Physical review. E, Statistical, nonlinear, and soft matter physics 71, 021906 (2005).
Ma, Y. et al. Cardiac Autonomic Alteration and Metabolic Syndrome: An Ambulatory ECG-based Study in A General Population. Scientific reports 7, 44363 (2017).
Fujiwara, Y. et al. Preoperative ultra short-term entropy predicts arterial blood pressure fluctuation during the induction of anesthesia. Anesthesia and analgesia 104, 853–856 (2007).
Schoenenberger, A. W. et al. Prediction of hypertensive crisis based on average, variability and approximate entropy of 24-h ambulatory blood pressure monitoring. Journal of human hypertension 22, 32–37 (2008).
Tejera, E., Areias, M. J., Rodrigues, A. I., Nieto-Villar, J. M. & Rebelo, I. Blood pressure and heart rate variability complexity analysis in pregnant women with hypertension. Hypertension in pregnancy 31, 91–106 (2012).
Ko, J. H. & Newell, K. M. Aging and the complexity of center of pressure in static and dynamic postural tasks. Neuroscience letters 610, 104–109 (2016).
Wayne, P. M. et al. Tai Chi Training may Reduce Dual Task Gait Variability, a Potential Mediator of Fall Risk, in Healthy Older Adults: Cross-Sectional and Randomized Trial Studies. Frontiers in human neuroscience 9, 332 (2015).
Kim, J. H., Yi, S. H., Lee, J. & Kim, Y. S. Effects of clozapine on heart rate dynamics and their relationship with therapeutic response in treatment-resistant schizophrenia. Journal of clinical psychopharmacology 33, 69–73 (2013).
Cabiddu, R. et al. Hippotherapy acute impact on heart rate variability non-linear dynamics in neurological disorders. Physiology & behavior 159, 88–94 (2016).
An, H., Kulkarni, R., Nagarathna, R. & Nagendra, H. Measures of heart rate variability in women following a meditation technique. International journal of yoga 3, 6–9 (2010).
Walsh, J. N. et al. Impact of Short- and Long-term Tai Chi Mind-Body Exercise Training on Cognitive Function in Healthy Adults: Results From a Hybrid Observational Study and Randomized Trial. Global advances in health and medicine 4, 38–48 (2015).
Lipsitz, L. A. & Goldberger, A. L. Loss of ‘complexity’ and aging. Potential applications of fractals and chaos theory to senescence. JAMA: the journal of the American Medical Association 267, 1806–1809 (1992).
Manor, B. & Lipsitz, L. A. Physiologic complexity and aging: implications for physical function and rehabilitation. Progress in neuro-psychopharmacology & biological psychiatry 45, 287–293 (2013).
Huikuri, H. V. & Stein, P. K. Heart rate variability in risk stratification of cardiac patients. Progress in cardiovascular diseases 56, 153–159 (2013).
Melillo, P. et al. Automatic prediction of cardiovascular and cerebrovascular events using heart rate variability analysis. PloS one 10, e0118504 (2015).
Tsuji, H. et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 94, 2850–2855 (1996).
Lu, W. A. & Kuo, C. D. Breathing frequency-independent effect of Tai Chi Chuan on autonomic modulation. Clinical autonomic research: official journal of the Clinical Autonomic Research Society 24, 47–52 (2014).
Vaananen, J. et al. Taichiquan acutely increases heart rate variability. Clinical physiology and functional imaging 22, 2–3 (2002).
Audette, J. F. et al. Tai Chi versus brisk walking in elderly women. Age and ageing 35, 388–393 (2006).
Lu, W. A., Chen, Y. S. & Kuo, C. D. Increased first and second pulse harmonics in Tai Chi Chuan practitioners. BMC complementary and alternative medicine 16, 87 (2016).
Chang, R. Y. et al. The effect of t’ai chi exercise on autonomic nervous function of patients with coronary artery disease. Journal of alternative and complementary medicine (New York, N.Y.) 14, 1107–1113 (2008).
Lu, W. A. & Kuo, C. D. Comparison of the effects of Tai Chi Chuan and Wai Tan Kung exercises on autonomic nervous system modulation and on hemodynamics in elder adults. The American journal of Chinese medicine 34, 959–968 (2006).
Novak, V. et al. Altered cerebral vasoregulation in hypertension and stroke. Neurology 60, 1657–1663 (2003).
Novak, V., Hu, K., Vyas, M. & Lipsitz, L. A. Cardiolocomotor coupling in young and elderly people. The journals of gerontology. Series A, Biological sciences and medical sciences 62, 86–92 (2007).
Hoppenbrouwers, T., Hodgman, J. E., Harper, R. M. & Sterman, M. B. Respiration during the first six months of life in normal infants: IV. Gender differences. Early human development 4, 167–177 (1980).
Hoyer, D., Frasch, M. G., Eiselt, M., Hoyer, O. & Zwiener, U. Validating phase relations between cardiac and breathing cycles during sleep. IEEE engineering in medicine and biology magazine: the quarterly magazine of the Engineering in Medicine & Biology. Society 20, 101–106 (2001).
Wu, S.-D., Wu, C.-W., Lee, K.-Y. & Lin, S.-G. Modified multiscale entropy for short-term time series analysis. Physica A: Statistical Mechanics and its Applications 392, 5865–5873 (2013).
Humeau-Heurtier, A. The multiscale entropy algorithm and its variants: a review. Entropy 17, 3110–3123 (2015).
Baumgartner, T., Jackson, A., Mahar, M. & Rowe, D. Measurement for Evaluation in Physical Education and Exercise Science: New York, McGraw-Hill 560 p (2006).
Acknowledgements
This work was supported by multiple grants from the National Institutes of Health (K24AT009282, R21 AT005501, R01AG025037–09, T32AT000051, K24AT009465), and from grant number UL1 RR025758 from the National Center for Research Resources supporting the Harvard Clinical and Translational Science Center. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NCCIH, NIA, NCRR, or the NIH.
Author information
Authors and Affiliations
Contributions
P.M.W. and C.K.P. designed the study, supervised the analysis, contributed to the discussion, and contributed to the drafting of the manuscript. Y.M. and C.W.W. analyzed all data. Y.M. drafted and revised the original manuscript and contributed to the discussion. A.A., S.M.B., L.A.L., G.Y.Y., B.M., V.N., J.H.H., and B.G. contributed significantly to the elements of data analysis plan, data interpretation, and review and discussion of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
Peter Wayne is the founder and sole owner of the Tree of Life Tai Chi Center. His interests were reviewed and managed by the Brigham and Women’s Hospital and Partner’s HealthCare in accordance with their conflict of interest policies. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ma, Y., Wu, Cw., Peng, CK. et al. Complexity-Based Measures of Heart Rate Dynamics in Older Adults Following Long- and Short-Term Tai Chi Training: Cross-sectional and Randomized Trial Studies. Sci Rep 9, 7500 (2019). https://doi.org/10.1038/s41598-019-43602-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43602-y
This article is cited by
-
Enhanced declarative memory in long-term mindfulness practitioners
Psychological Research (2023)
-
Detecting cardiac pathologies via machine learning on heart-rate variability time series and related markers
Scientific Reports (2020)
-
Electrocardiogram-based sleep analysis for sleep apnea screening and diagnosis
Sleep and Breathing (2020)
-
Research on Function and Mechanism of Tai Chi on Cardiac Rehabilitation
Chinese Journal of Integrative Medicine (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.