Abstract
MicroRNAs (miRNAs) play a critical role in regulation of numerous biological processes and pathogenesis of a variety of diseases. In addition, miRNAs contribute to carcinogenesis by acting as oncogenic or tumor suppressive. Circulating miRNAs including miR-150-5p are associated with colorectal cancer progression, and the putative targets of miR-150-5p include tumor suppressor gene, TP53. Here we sought to investigate the role of miR-150-5p-TP53 signaling pathway in proliferation of colon cancer and to determine expression levels of miR-miR-150-5p and TP53 in colon adenocarcinoma and adjacent non-cancerous tissue samples, or in human colon adenocarcinoma cell lines. MTT assay was used to determine proliferation and apoptosis in cell lines. Furthermore, we used Western blot to determine levels of cell cycle regulators with anti-miR-150-5p or apoptosis with overexpression of TP53. Our results show that expression levels of miR-150-5p were significantly elevated in clinical specimens from cancer patients. We further showed that inhibition of miR-150-5p increased TP53, and in turn, suppression of proliferation of colon adenocarcinoma. Moreover, inhibition of miR-150-5p or overexpression of TP53 caused cell arrest or apoptosis in colon adenocarcinoma. Our results support that miR-150-5p-TP53 pathway plays an important role in regulation of proliferation, cell arrest, and apoptosis in colon cancer, and could be an attractive target for therapy.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is a common malignant tumor with an incidence rate of a million people worldwide every year, and a major cause of mortality and morbidity in developed countries1,2,3. In addition, CRC ranks as the third most common cancer by incidence and the fourth most common cause of cancer-related deaths world-wide. Presently, surgical resection is the only approach for curative treatment of CRC4. Despite numerous efforts in the past few decades, CRC survival rates have not significantly improved, and half of the CRCs relapse and the patients die within five years. As such, identifying novel and more effective drug targets in treatment of CRC are imminent.
MicroRNA (miRNAs) usually silence target genes by binding to their target messenger RNAs at 3′-UTR5. It has been estimated that miRNAs can regulate one-third of all human genes and influence hundreds of potential gene targets6. Dysregulation of miRNA expression is closely linked to cancer initiation and progression. Mechanistically, miRNAs can inhibit oncogenes or tumor suppressor genes to play a tumor suppressor or oncogenic role, respectively7. In addition, miRNAs can serve as biomarkers for early cancer diagnosis, because miRNAs are stable in body fluids, either in secreted microvesicles or bound to carrier proteins, which provides a unique opportunity to exploit the role of miRNAs as biomarkers8. Indeed, expression levels of circulating miRNAs have shown some potential at differentiating cancer patients and healthy controls for prostate9, ovarian10, lung11 and breast12 cancers. Moreover, multiples studies support the prognostic and diagnostic value of some circulating exosomal microRNAs in colon cancer, particularly, miR-150-5p13,14.
The tumor suppressor gene, TP53, is one of the most frequently mutated genes in many cancers including CRC15. TP53 is generally expressed at low levels, in part through negative feedback loops that involve MDM216. Low abundance of T53 expression preserves homoeostasis of the cell cycle and cell death. In response to stress factors, such as hypoxia, UV irradiation, oxidative free radicals, and activation of oncogenes, TP53 protein is activated, and such an activation of p53 can either transactivate or repress downstream target genes that in turn regulate cell cycle arrest, apoptosis, DNA repair, and angiogenesis and even metastasis17.
Although multiple studies have revealed the potential tumor-suppressive effect of TP53 in human tumor, the molecular function of TP53 in CRC has remained further defined. Expression of miR-150-5p has been linked with TP53-downstream target genes15. Interestingly, a preliminary bioinformatics analysis indicates that TP53 is a potential target of miR-150-5p18. In the present study, we sought to investigate the role of miR-150-5p-TP53 signaling in CRC, and we demonstrate that such a signaling plays a critical role in proliferation and progression of CRC.
Materialas and Methods
Subjects
We collected specimens from a total of 10 CRC patients after surgical resection. The study and all methods were carried out in accordance with relevant guidelines and regulations. all experimental protocols were approved by the Ethic Committee of China-Japan Friendship Hospital. The general information for patients is provided in Table 1. Informed consent was obtained from all subjects.
Cell culture
Normal human colon epithelial cell line CCD 841 CoN (ATCC, USA), and human colon carcinoma cell lines HT29, T84, and LS174 (ATCC, USA) were maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum, as recently described19. Cells were cultured in a humidified incubator at 37 °C and 5% CO2.
Reagents
Antibodies to TP53, COX-2, CDK1, Cyclin B1, Caspase-3 and -9 were obtained from Santz Cruz Biotech (TX, USA). TP53 siRNA, miR-150-5p mimics or anti-miR-150-5p inhibitors were purchased from Qiagen (USA).
Quantitative RT-PCR
Total RNAs were extracted by Trizol method (Invitrogen, CA, USA) and re-suspended in nuclease-treated H2O. cDNA synthesis was prepared by the miR-150-5p-, U6 snRNA-specific or oligo-dT primers method using the Superscript II Reverse Transcription kit (Invitrogen, USA). Quantitative PCR was performed using an ABI7300 machine (Applied Biosystems, USA). miR-150-5p or TP53 mRNA levels were determined, relative to U6 or GAPDH expression using the SYBR Green PCR kit (Qiagen, USA), respectively. Fold change in expression was determined by the method of cycle threshold (CT) using the formula of 2−ΔΔCT. U6 forward, 5′-TGCGGGTGCTCGCTTCGGCAGC-3′. U6 reverse, 5′-GGGTCCGAGGTGCACTGGATACGACAAAATATGG-3′; GAPDH forward, 5′-CTCCCGCTTCGCTCTCTG-3′, GAPDH reverse, 5′- CTGGCGACGCAAAAGAAG-3′, TP53 forward: 5′-GGCCCACTTCACCGTACTAA-3′; TP53 forward: 5′-GTGGTTTCAAGGCCAGATGT-3′.
Western blot
Whole cell lysates were suspended in 1 × SDS loading buffer, boiled at 95 °C for 5 min, and centrifuged. Supernatants were separated on SDS–10% PAGE and transferred onto PVDF membranes (Bio-Rad, USA). Membranes were blocked in 5% milk in PBST (10 mM phosphate buffer, pH 7.2; 150 mM NaCl; and 0.1% Tween-20) for 60 min, washed three times, and incubated with appropriate antibody at 4 °C overnight. Membranes were washed three times with PBST, incubated with horseradish peroxidase-conjugated secondary antibody at 1:5,000, and developed by Immun-Star HRP Substrate (Bio-Rad, CA, USA). Blots were probed with β-actin antibody (Sigma-Aldrich, USA) as the loading control.
Dual Luciferase Assay
3′-UTR segment of TP53 gene corresponding to predicted target site was amplified by PCR from human genomic DNA using primers that included a XbaI and EcoRI tails on the 5′ and 3′ strands, respectively, as previously described20. PCR products were restriction digested with both XbaI and EcoRI DNA restriction endonucleases, gel purified, and ligated into pGL3 vector (Promega, USA). HT29 cells were transfected with the firefly luciferase UTR-report vector, control Renilla luciferase pRL-TK vector (Promega, USA) with Lipofectamine 2000 reagent, according to the manufacturer’s protocol (Invitrogen, USA). Twenty-four hours after transfection, cells were lysed with a 1x passive lysis buffer and the activity of both Renilla and firefly luciferases were assayed using the dual-luciferase reporter assay system (Promega, USA), according to the manufacturer’s instructions.
Cell proliferation Assay
MTT [3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazoliumbr-omide] - based assay was performed to estimate the effect of miR-150-5p mimics, anti-miR-150-5p, or TP53 siRNA on human colonic adenocarcinoma cells’ proliferation, as previously described21. Cells were seeded into 96-well plates (5,000 cells/well in 200 µL medium) and incubated for 24 hrs. HT29 cells were transfected with miR-150-5p mimics, anti-miR-150-5p, or TP53 siRNA using Lipofectamine 2000 Reagent (ThermoFisher Scientific, USA) as indicated. Cells cultured in complete medium were used as control. At the end of incubation, 20 µL of 5 mg/ml MTT (Sigma, USA) solution was added per well, and the cells were incubated for another 4 hr at 37 °C. Supernatants were removed and formazan crystals were dissolved in 150 µL of DMSO (Sigma-Aldrich, USA). OD was determined at 490 nm using multi-microplate test system (InfiniteM200Pro,USA).
Statistical analysis
All results were expressed as mean ± standard deviation. We used Student’s t-test or ANONA (One-way) to compare two and three groups, respectively. P < 0.05 was considered significantly different.
Results
miR-150-5p directly targets TP53 in CRC
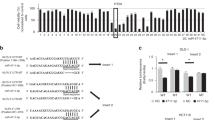
TP53 is a tumor suppressor gene as well as putative target of miR-150-5p18. To investigate whether miR-150-5p targets TP53 in CRC, we first performed bioinformatic analysis, using TargetScan and found that there are multiple miR-150-5p target sites in TP53 mRNA (Fig. 1A).
miR-150-5p directly targets TP53 in CRC. (A) Predicted target site of miR-150-5p in 3′-UTR region of TP53 mRNA. WT, wild-type, Mut, mutation. (B) Equal amounts of empty vector (FL), those with wild-type TP53 3′-UTR or mutant and internal control vector (RL) was transiently transfected into HT29 cells for 48 hrs followed by dual-luciferase assays. (C) HT29 cells were transiently transfected with non-specific (NS) miRNA mimics or anti miR-150-5p (50 nM) for 48 hr and mRNA (left panel) and protein levels (right panel) were determined by Western blot and RT-PCR, respectively. *P < 0.01 (N = 3).
To validate a direct interaction between miR-150-5p and TP53, we cloned a miR-150-5p target site at 3′-UTR of TP53 mRNA upstream of firefly luciferase (FL) reporter gene, so that it could be regulated by miR-150-5p. In addition, a mutated version of this target site was used as the negative control. Colon adenocarcinoma HT29 cells were transfected with either the control FL plasmid or FL plasmid with miR-150-5p target site or the FL plasmid with mutation of miR-150-5p target site. Also, Renilla luciferase reporter plasmid was co-transfected as an internal reference. As shown in Fig. 1B, a significant decrease in FL activity was observed in cells transfected with FL reporter with wild type of miR-150-5p target site. In contrast, no repression of FL activity was obtained in cells transfected with miR-150-5p-mutation FL reporter plasmid (Fig. 2B). Further, we confirmed a direct targeting of TP53 mRNA by miR-150-5p in HT29 cells. We found that TP53 increased at both protein and mRNA levels when cells were treated with miR-150-5p specific inhibitors (Fig. 1C). Taken together, these results indicate that TP53 is a direct target of miR-150-5p in CRC cells.
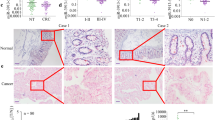
Expression of miR-150-5p and TP53 in colon cancer specimens and cell lines. The expression levels of miR-150-5p (A) or TP53 protein (B) or TP53 mRNA (C) were determined in both colon adenocarcinoma (tumor) and non-cancerous (normal) adjacent tissues. (D) Representative images of immunofluorescence staining, using antibodies against TP53 or COX-2, on isolated cancer cells purified from 3 select specimens. (E) Expression levels of miR-150-5p and TP53 mRNA, as determined by RT-qPCR, in colon adenocarcinoma cell lines (N = 3) and the normal human colon epithelial cell line CCD 841 CoN. *P < 0.01.
Coupled increase of miR-150-5p and decrease of TP53 in CRC
Next, we sought to determine miR-150-5p and TP53 levels in in tissue specimens derived from CRC patients. Total RNAs were extracted from 10 colon cancer and adjacent non-cancerous tissue samples for the assessment of miR-150-5p and TP53 mRNA levels by quantitative RT-PCR. TP53 protein levels were assessed by Western blots as well. In the meantime, the same tissues were subjected to purification of colon cancer cells followed by immunofluorescence staining (IF). We found that miR-150-5p was significantly up-regulated in CRC cancer tissues (Fig. 2A). In contrast, TP53 mRNA and protein levels decreased by an average of >45% (Fig. 2B,C), compared to the non-cancerous adjacent colon mucosa. In addition, weak cytoplasmic staining for TP53 was observed in the cancerous tissues, in contrast to strongly positive staining of COX-2, another biomarker for CRC22 (Fig. 2D).
Next, we used RT-PCR to determine levels of miR-150-5p and TP53 in different colon adenocarcinoma cell lines and similar results were obtained in all three colon adenocarcinoma cell lines as found in the primary tissues (Fig. 2E). Collectively, these results suggest that miR-150-5p and TP53 are negatively correlated in CRC, suggesting a possible regulation of TP53 by miR-150-5p.
miR-150-5p stimulates cell proliferation in vitro
HT29 cells were transfected with non-specific miRNA mimics, anti-miR-150-5p, or anti-miR-150-5p combined with TP53 siRNAs, as indicated, and MTT assays were performed to determine cell proliferation. As shown here, miR-150-5p mimics promoted, but miR-150-5p inhibitors inhibited cancer cell growth (Fig. 3A). Consistent with this, depletion of TP53 also promoted cancer growth, and abolished anti-miR-150-5p mediated inhibition of cell proliferation (Fig. 3B,C). These results further confirm that miR-150-5p directly inhibits TP53 to exert oncogene functions and promote CRC cancer cell proliferation.
Effects of miR-150-5p-TP53 signaling on proliferation of CRC in vitro. (A) HT29 cells were transiently transfected with non-specific miRNA mimics (NS) or antagomirs (anti-miR), or miR-150-5p mimics or anti-miR-150-5p for the time period as indicated. (B) HT29 cells were transiently transfected with non-specific siRNA (NS) or TP53 siRNA (siTP53) for time period, as indicated. (C) HT29 cells were transfected with non-specific antagomirs (NS), or anti-miR-150-5p, or anti-miR-150-5p combined with TP53 siRNA as indicated. Cell proliferation was analyzed by MTT assay. *P < 0.01 (N = 3).
Inhibition of miR-150-5p affects the expression of key cell cycle factors
TP53 has been found to induce cell cycle arrest in G2/M phase in clear cell renal cell carcinoma (ccCRC)23. Here we tested the role of miR-150-5p-TP53 signaling in regulation of cell cycle in CRC. The expression of cell cycle regulators associated with G2/M phase transformation was investigated in HT29 cells treated with either anti-miR-150-5p or the control non-specific miRNA mimics. As shown in Fig. 4, protein levels of cyclinB1 and CDK1 were significantly decreased in cells transfected with anti-miR-150-5p, in contrast to upregulation of TP53. These results support that miR-150-5p-TP53 signaling pathway modulates cell cycle progression via expression of cell cycle regulators.
TP53 triggers cell apoptosis in CRC cells
Evasion of apoptosis is a critical event during malignant transformation, and TP53 has been found to induce apoptosis in various cancer cells24. Hence, we examined the effects of TP53 on induction of CRC cell apoptosis. As shown in Fig. 5, the expression levels of cleaved caspase-3 and caspase-9, which are molecular hallmarks for cell apoptosis, were significantly increased in HT29 cells transfected with pc-DNA3-TP53. Collectively, these results suggest that TP53 overexpression exerts significant effects on the proliferation of CRC cells.
TP53 induces cell apoptosis in CRC cells. HT29 cells were transiently transfected with empty vector, pcDNA3.1 (Emp) or pcDNA3.1 with open reading frame of TP53 (TP53 vect., 1 μg) for 48 hrs. The expression levels of cleaved caspase-3 and caspase-9 were determined and quantified by Western blot. *P < 0.01 (N = 3).
Discussion
The molecular mechanisms underlying CRC initiation and progression remain largely unknown, and, therefore, a comprehensive detailing of all such mechanisms is critical. This alone will facilitate discovery of novel biomarkers for early diagnosis and therapy, thus improving the outcome of CRC patients. Over the past decade, miRNAs have emerged as a new and promising class of gene regulators involved in cancer progression25,26. Here, we show, for the first time, that miR-150-5p plays a critical role in colon tumorigenesis by inhibiting the tumor suppressor gene TP53 to promote cancer proliferation. Our results suggest that miR-150-5p can be used for early stage diagnosis and a therapeutic target for therapy of CRC. These collective findings show an important and new role of miR-150-5p-TP53 signaling pathway in carcinogenesis of CRC, and provide a molecular detaining for the correlation between elevation of miR-150-5p and cell proliferation in CRC. Therefore, our study offers a concrete base for further understanding of the complex biology involved in CRC cancer progression.
miRNAs are well-known to play a role in cancer progression. As demonstrated previously by several groups, the same miRNA might play a distinct role in different types of cancer. For example, miR-146a acts as oncogenic as well as tumor suppressive in distinct cell types27,28. Consistent with this observation, patients with higher miR-150-5p levels usually have a worse survival in melanoma and inhibition of miR-150-5p enhanced cell apoptosis via upregulation of PDCD4-mediated activation of caspase-8 and p2129. However, another study has shown that miR-150 might serve as a potential therapeutic sensitizer through inhibition of the AKT pathway in NK/T cell lymphoma treatment30. Therefore, more studies are needed to elucidate the role of miR-150-5p in other types of cancer.
Our results are consistent with the earlier published literature that TP53 plays a protective role in blocking a decisive step in oncogenesis. Activation of TP53 can induce both the mitochondrial and the death-receptor-induced apoptotic pathways31. Activation of TP53 leads to expression of pro-apoptotic Bcl-2 (B-cell lymphoma-2) family proteins, mainly Bax, Noxa and PUMA, but inhibits the pro-survival Bcl-2, and in turn, permeabilization of outer mitochondrial membrane. Then cytochrome c releases from the mitochondria binds to APAF-1, and induces the activation of the initiator caspase-9, eventually resulting in the activation of executioner caspase-3, -6 and -7. On the other hand, activated TP53 increases some death receptors and form death-inducing signaling complex with caspase-8, to induce apoptosis. The progression of cell cycle is tightly controlled by cyclins and cyclin-dependent kinases (CDK). p21 is one member of CDK inhibitor family, which stimulates cell cycle transition from G1 to S phase, as well as one of the major mediator of p53-induced growth arrest. In response to DNA damage, p53 induces not only cell cycle G1 phase arrest, but also G2/M checkpoint arrest32.
In conclusion, our results support a role of miR-150-5p-TP53 signaling pathway in the proliferation, cell cycle progress, cell apoptosis and invasion/migration of CRC cells. Furthermore, miR-150-5p acts an oncogene and its target TP53 acts as a tumor suppressor in CRC pathogenesis. Due to current lack of appropriate biomarkers and therapeutic targets in CRC, these results suggest that miR-150-5p-TP53 signaling may provide novel insights into the improvement of clinical therapeutic strategies for CRC patients.
Data Availability
All the data is available within this manuscript.
References
Pox, C. P. Controversies in colorectal cancer screening. Digestion 89, 274–281, 000363287 [pii] (2014).
Marley, A. R. & Nan, H. Epidemiology of colorectal cancer. Int J Mol Epidemiol Genet 7, 105–114 (2016).
Arnold, M. et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut 66, 683–691, https://doi.org/10.1136/gutjnl-2015-310912 (2017).
Brenner, H., Kloor, M. & Pox, C. P. Colorectal cancer. Lancet 383, 1490–1502, S0140-6736(13)61649-9 [pii] (2014).
Ha, M. & Kim, V. N. Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol 15, 509–524, https://doi.org/10.1038/nrm3838 (2014).
Lewis, B. P., Burge, C. B. & Bartel, D. P. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120, 15–20, https://doi.org/10.1016/j.cell.2004.12.035 (2005).
Jansson, M. D. & Lund, A. H. MicroRNA and cancer. Mol Oncol 6, 590–610, https://doi.org/10.1016/j.molonc.2012.09.006 (2012).
Pritchard, C. C. et al. Blood cell origin of circulating microRNAs: a cautionary note for cancer biomarker studies. Cancer Prev Res (Phila) 5, 492–497, https://doi.org/10.1158/1940-6207.CAPR-11-0370 (2012).
Hassan, O., Ahmad, A., Sethi, S. & Sarkar, F. H. Recent updates on the role of microRNAs in prostate cancer. J Hematol Oncol 5, 9, https://doi.org/10.1186/1756-8722-5-9 (2012).
Yokoi, A. et al. A combination of circulating miRNAs for the early detection of ovarian cancer. Oncotarget 8, 89811–89823, https://doi.org/10.18632/oncotarget.20688 (2017).
Inamura, K. Diagnostic and Therapeutic Potential of MicroRNAs in Lung Cancer. Cancers (Basel) 9, https://doi.org/10.3390/cancers9050049 (2017).
Hamam, R. et al. Circulating microRNAs in breast cancer: novel diagnostic and prognostic biomarkers. Cell Death Dis 8, e3045, https://doi.org/10.1038/cddis.2017.440 (2017).
Aherne, S. T. et al. Circulating miRNAs miR-34a and miR-150 associated with colorectal cancer progression. BMC Cancer 15, 329, https://doi.org/10.1186/s12885-015-1327-5 (2015).
Hosseini, M. et al. Exosome-Encapsulated microRNAs as Potential Circulating Biomarkers in Colon Cancer. Curr Pharm Des 23, 1705–1709, https://doi.org/10.2174/1381612822666161201144634 (2017).
Slattery, M. L. et al. A comparison of colon and rectal somatic DNA alterations. Dis Colon Rectum 52, 1304–1311, https://doi.org/10.1007/DCR.0b013e3181a0e5df (2009).
Mandinova, A. & Lee, S. W. The p53 pathway as a target in cancer therapeutics: obstacles and promise. Sci Transl Med 3, 64rv61, https://doi.org/10.1126/scitranslmed.3001366 (2011).
Harris, S. L. & Levine, A. J. The p53 pathway: positive and negative feedback loops. Oncogene 24, 2899–2908, https://doi.org/10.1038/sj.onc.1208615 (2005).
Agarwal, V., Bell, G. W., Nam, J. W. & Bartel, D. P. Predicting effective microRNA target sites in mammalian mRNAs. Elife 4, https://doi.org/10.7554/eLife.05005 (2015).
Ding, L., Yu, L. L., Han, N. & Zhang, B. T. miR-141 promotes colon cancer cell proliferation by inhibiting MAP2K4. Oncol Lett 13, 1665–1671, https://doi.org/10.3892/ol.2017.5653 (2017).
Kovalchuk, O. et al. Involvement of microRNA-451 in resistance of the MCF-7 breast cancer cells to chemotherapeutic drug doxorubicin. Mol Cancer Ther 7, 2152–2159, https://doi.org/10.1158/1535-7163.MCT-08-0021 (2008).
He, D. et al. MiR-371-5p facilitates pancreatic cancer cell proliferation and decreases patient survival. PLoS One 9, e112930, https://doi.org/10.1371/journal.pone.0112930.
Wang, D. & Dubois, R. N. The role of COX-2 in intestinal inflammation and colorectal cancer. Oncogene 29, 781–788, https://doi.org/10.1038/onc.2009.421 (2010).
Chen, W. et al. RhoB Acts as a Tumor Suppressor That Inhibits Malignancy of Clear Cell Renal Cell Carcinoma. PLoS One 11, e0157599, https://doi.org/10.1371/journal.pone.0157599 (2016).
Marlow, L. A., Bok, I., Smallridge, R. C. & Copland, J. A. RhoB upregulation leads to either apoptosis or cytostasis through differential target selection. Endocr Relat Cancer 22, 777–792, https://doi.org/10.1530/ERC-14-0302 (2015).
Reddy, K. B. MicroRNA (miRNA) in cancer. Cancer Cell Int 15, 38, https://doi.org/10.1186/s12935-015-0185-1 (2015).
Fayyaz, S. et al. MicroRNA regulation of TRAIL mediated signaling in different cancers: Control of micro steering wheels during the journey from bench-top to the bedside. Semin Cancer Biol, https://doi.org/10.1016/j.semcancer.2019.01.007 (2019).
Raimo, M. et al. miR-146a Exerts Differential Effects on Melanoma Growth and Metastatization. Mol Cancer Res 14, 548–562, https://doi.org/10.1158/1541-7786.MCR-15-0425-T (2016).
Sathyanarayanan, A., Chandrasekaran, K. S. & Karunagaran, D. microRNA-146a inhibits proliferation, migration and invasion of human cervical and colorectal cancer cells. Biochem Biophys Res Commun 480, 528–533, https://doi.org/10.1016/j.bbrc.2016.10.054 (2016).
Wan, J., Yang, J., Huang, Y. & Deng, L. MicroRNA-150 inhibitors enhance cell apoptosis of melanoma by targeting PDCD4. Oncol Lett 15, 1475–1482, https://doi.org/10.3892/ol.2017.7445 (2018).
Wu, S. J., Chen, J., Wu, B., Wang, Y. J. & Guo, K. Y. MicroRNA-150 enhances radiosensitivity by inhibiting the AKT pathway in NK/T cell lymphoma. J Exp Clin Cancer Res 37, 18, https://doi.org/10.1186/s13046-017-0639-5 (2018).
Ryan, K. M., Phillips, A. C. & Vousden, K. H. Regulation and function of the p53 tumor suppressor protein. Curr Opin Cell Biol 13, 332–337 (2001).
Li, X. L., Zhou, J., Chen, Z. R. & Chng, W. J. P53 mutations in colorectal cancer - molecular pathogenesis and pharmacological reactivation. World J Gastroenterol 21, 84–93, https://doi.org/10.3748/wjg.v21.i1.84 (2015).
Author information
Authors and Affiliations
Contributions
F.L. and X.D.W. performed experiments; X.D.W. analyzed results; F.L. put together first draft; X.D.W. developed idea, provided resources and approved manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, F., Di Wang, X. miR-150-5p represses TP53 tumor suppressor gene to promote proliferation of colon adenocarcinoma. Sci Rep 9, 6740 (2019). https://doi.org/10.1038/s41598-019-43231-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43231-5
This article is cited by
-
AIFM2 promotes hepatocellular carcinoma metastasis by enhancing mitochondrial biogenesis through activation of SIRT1/PGC-1α signaling
Oncogenesis (2023)
-
Transcriptomics and Proteomics Approach for the Identification of Altered Blood microRNAs and Plasma Proteins in Parkinson’s Disease
Cellular and Molecular Neurobiology (2023)
-
From inflammatory bowel disease to colorectal cancer: what’s the role of miRNAs?
Cancer Cell International (2022)
-
Effect of combined treatment with lobaplatin and osthole on inducing apoptosis and inhibiting proliferation in human breast cancer MDA-MB-231 cells
Medical Oncology (2022)
-
miR-663b promotes colorectal cancer progression by activating Ras/Raf signaling through downregulation of TNK1
Human Cell (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.