Abstract
Considering the already established relationship between the extent of the metabolic dysfunction and the severity of heart failure (HF), it is conceivable that the metabolomic profile of the serum may have a prognostic capacity for 3-month mortality in acute heart failure (AHF). Out of 152 recruited patients, 130 serum samples were subjected to the metabolomic analyses. The 3-month mortality rate was 24.6% (32 patients). Metabolomic profiling by nuclear magnetic resonance spectroscopy found that the serum levels of 2-hydroxybutyrate (2-HB), 3-hydoxybutyrate (3-HB), lactate, citrate, and tyrosine, were higher in patients who died within 3 months compared to those who were alive 3 months after onset of AHF, which was confirmed by univariable logistic regression analyses (p = 0.009, p = 0.005, p = 0.008, p<0.001, and p<0.001, respectively). These associations still remained significant for all tested metabolites except for lactate after adjusting for established prognostic parameters in HF. In conclusion, serum levels of 2-HB, 3-HB, tyrosine, and citrate measured at admission are associated with an increased 3-month mortality rate in AHF patients and might thus be of prognostic value in AHF.
Similar content being viewed by others
Introduction
Heart failure (HF) is a final stage of various cardiovascular diseases and a common cause of disability and death1. The European Society of Cardiology (ESC) defines HF as an abnormality of the cardiac structure and function, which results in a diminished oxygen supply to the metabolizing tissues2,3. Acute heart failure (AHF) is primarily characterized by the rapid onset of symptoms and signs of HF3.
Metabolic dysfunction, an inherent feature of the HF pathophysiology4,5, reflects not only the altered metabolism of the myocardium but rather overall contributions from peripheral tissues and organs6. Hemodynamic impairment and the thereby accompanied tissue hypoperfusion and congestion increase the serum levels of catecholamines7, inflammatory cytokines8, and natriuretic peptides9. These promote lipolysis, proteolysis, and oxidative stress, the hallmarks of metabolic dysfunction in HF5,10 either directly or via the induction of insulin resistance, a principal metabolic feature of the HF pathophysiology11. Metabolic dysfunction and the catabolic dominance in HF are further intensified by a reduced appetite and an impaired intestinal nutrient absorption due to congestion and intestinal edema as well as the diminished biosynthetic capacity of the hypo-perfused and/or congested liver12,13,14,15.
The extent of the metabolic perturbation parallels the hemodynamic impairment, i.e. the severity of HF. Accordingly, it is conceivable that the metabolomic profile of the patients’ serum at admission may have a valuable prognostic potential for 3-month mortality in AHF. Untargeted metabolic profiling using 1H nuclear magnetic resonance (NMR) spectroscopy has been successfully established in the recent years for a wide range of metabolites and biological matrices, including human biofluids16,17,18,19,20. The aim of the present study was therefore to employ NMR spectroscopy to identify serum metabolites that are prognostic for 3-month mortality in patients with AHF.
Results
Clinical characteristics, pre-admission medication and comorbidities
Out of 152 AHF patients recruited in the study21,22,23,24, serum samples of 130 patients were subjected to the metabolomic analyses. Of these, 32 (24.6%) died within three months after onset of AHF. As presented in Table 1, body mass index (BMI) and weight were significantly lower and the liver enlargement and peripheral edema, were considerably more frequently observed in patients who did not survive compared to those who survived the first three months after onset of AHF. These groups were not significantly different regarding age, gender, smoking, New York Heart Association Functional Classification (NYHA), mean arterial pressure (MAP), heart rate, ejection fraction, or systolic pulmonary artery pressure. The incidence of distended jugular veins and ascites as well as pre-admission medication were also similar in both groups (Table 1). Additionally, the groups did not differ with respect to the history of hyperlipidemia, hypercholesterolemia, hypertension, type 2 diabetes mellitus (T2DM), chronic obstructive pulmonary disease, chronic kidney disease, cardiomyopathy, or acute coronary syndrome (Supplementary Table 1).
Laboratory parameters
While glomerular filtration rate (GFR) and total cholesterol were significantly lower, the serum levels of urea, creatinine, N-terminal pro brain natriuretic peptide (NT-proBNP), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were significantly higher in AHF patients who died within three months compared to those who survived three months after onset of AHF (Table 2). The two groups did not differ in the serum levels of proteins, albumin, interleukin-6 (IL-6), C-reactive protein (CRP), as well as low-density lipoprotein cholesterol (LDL cholesterol), high-density lipoprotein cholesterol (HDL-cholesterol), and triglycerides (Table 2).
Identification and prognostic value of metabolites differently abundant in patients who were alive and those who died within three months after onset of AHF
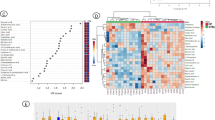
In order to assess metabolic differences between patients that did and did not die within three months after onset of AHF, nuclear magnetic resonance (NMR) metabolic profiling of 130 serum samples was performed. When comparing differences in metabolic fingerprints between patients who were alive and those who died within 3 months after onset of AHF, Orthogonal-Partial Least Squares - Discriminant Analysis (O-PLS-DA) revealed a slight clustering of patient samples with correlation coefficients R2 up to 0.18 and a Q2 of 0.104 (p < 0.01) (Fig. 1a). Reduced NMR spectra revealed altered metabolites in normalized AHF serum samples (Fig. 1b) and indicated that the levels of 2-hydroxybutyrate (2-HB), 3-hydroxybutyrate (3-HB), lactate, alanine, citrate, and tyrosine were higher, whereas valine and glucose were lower in patients who died within three months after onset of AHF.
OPLS-DA plot of AHF serum samples. (a) Multivariate OPLS-DA plot of 3-month mortality. (b) Reduced NMR spectra reveal altered metabolites in normalized AHF serum samples. Positive covariance corresponds to metabolites present at increased concentrations, whereas negative covariance corresponds to decreased metabolite concentrations in patients that died within three months. Predictivity of the model is represented by R2. 1…2-hydroxybutyrate, 2…valine, 3…3-hydroxybutyrate, 4…lactate, 5…alanine, 6…citrate, 7…glucose, 8…tyrosine.
In order to assess the prognostic value of these metabolites, the absolute concentrations were determined. As shown in Fig. 2 the levels of 2-HB, 3-HB, lactate, tyrosine, and citrate, but not of alanine, valine, and glucose, were significantly different (increased) in patients who died compared to those who survived the first three months after onset of AHF. Furthermore, sensitivity and specificity for each metabolite were assessed using Receiver Operating Characteristic (ROC) curves (Fig. 2). The highest values for the area under the curve (AUC), indicating highest predictivity, were obtained for tyrosine and citrate, followed by 3-HB, lactate, 2-HB, alanine, glucose, and valine (Fig. 2).
Serum levels and ROC analysis of altered metabolites. Differences between patients who were alive and those who died within three months after onset of AHF were tested with the Mann-Whitney U test. Absolute concentrations were used to calculate ROC curves for distinct metabolites and to assess the prognostic value of the distinct metabolites for 3-month mortality.
Correlation of 2-HB, 3-HB, lactate, citrate, and tyrosine concentrations with laboratory and clinical parameters
To examine the relationship between the identified metabolites, whose serum levels were significantly higher in patients who did not survive compared to those who survived the first three months after onset of AHF, and clinical and laboratory parameters, correlation analyses were performed. As shown in Table 3, the serum concentration of citrate, but not of any other metabolite, was significantly positively correlated with age, urea and creatinine as well as negatively correlated with fibrinogen, and HDL cholesterol. Both citrate and tyrosine were significantly negatively correlated with BMI, MAP, total cholesterol, LDL cholesterol, and triglyceride levels. Citrate and 3-HB were negatively correlated with GFR but positively with urea. Additionally, 2-HB, 3-HB, citrate, and tyrosine were significantly positively correlated with NT-proBNP. While only 2-HB and lactate were significantly positively correlated with ALT, all tested metabolites were significantly positively correlated with AST. Serum concentrations of 3-HB, citrate, and tyrosine, but not of 2-HB and lactate, were furthermore significantly positively correlated with concentrations of IL-6.
Logistic regression analyses
To further examine the association of the metabolites with 3-month mortality, we performed logistic regression analyses (Table 4) for 2-HB, 3-HB, lactate, tyrosine, and citrate, the metabolites that were significantly higher (Fig. 2) in patients who did not survive three months after onset of AHF as compared to those that did. As shown in Table 4, the univariable analyses showed a significant positive association of the serum levels of all tested metabolites with 3-month mortality. These associations remained significant for all tested metabolites except lactate, upon adjusting for the established prognostic parameters in HF, namely age, sex, BMI, T2DM, NT-proBNP, GFR, MAP, and LDL cholesterol.
Discussion
Despite established multivariable predictive models comprising patients’ characteristics, clinical signs and serum biomarkers, the estimation of risk in AHF is difficult, not accurate and poorly applicable in daily clinical practice25. Therefore, the identification of new biomarkers, which are related to the complex mechanisms of the AHF pathophysiology, may help in identifying high risk patients and initiating timely therapeutic interventions.
Considering a positive relationship between metabolic dysfunction, which is an established inherent feature of HF, and the extent of hemodynamic impairment, serum metabolites might be useful prognostic markers in AHF5,26,27.
In the present study, we show, that serum levels of 2-HB, 3-HB, citrate, and tyrosine are independently associated with 3-months mortality in AHF patients, even after adjusting for other well-known risk factors. Increased levels of these metabolites as well as lactate in AHF patients who did not survive the first three months after onset of AHF strongly indicate a more severe state of the disease and more severe systemic metabolic perturbations in this group of AHF patients, compared to AHF patients who survived 3 months after onset of AHF. Indeed, with the exception of lactate the levels of 2-HB, 3-HB, citrate, and tyrosine were significantly positively correlated with NT-proBNP, reflecting a positive relationship with the severity of HF28.
It is well established that a decreased cardiac output and a subsequent decreased tissue perfusion, a consequence of left-sided HF, congestion, are accompanied by increased serum levels of catecholamines, natriuretic peptides and inflammatory cytokines. These, in turn trigger lipolysis, proteolysis, and oxidative stress, either directly or via the induction of insulin resistance7,8,9,10,11. Increased 2-HB levels have been shown to reflect insulin resistance and tricarboxylic acid (TCA) cycle overload29,30,31 as well as increased oxidative stress32,33. We observed increased serum levels of 2-HB in patients who died within three months after onset of AHF, which argues for the role of insulin resistance and oxidative stress in metabolic perturbations and mortality in our AHF cohort. This is further substantiated by increased 3-HB levels in these patients, since its hepatic synthesis and serum levels are known to be augmented in states of insulin resistance and excess fatty acid supply31. Alternatively or additionally, increased 3-HB serum levels in this group of AHF patients may be a consequence of the decreased utilization of 3-HB as an energy substrate in skeletal muscle. Previous studies have shown decreased 3-HB utilization in skeletal muscle of HF patients compared to healthy controls34.
Elevated serum 3-HB levels may result in an increased uptake and oxidation by the failing heart. This in turn may diminish the uptake and utilization of glucose35, whose energy producing efficiency outperforms that of 3-HB36. Accordingly, by displacing glucose utilization, the elevated 3-HB might be detrimental when oxygen provision is limited due to decreased cardiac tissue perfusion and congestion, as is encountered in the failing heart. Furthermore, an increased intracellular pool of acetyl CoA, secondary to an increased 3-HB oxidation, may facilitate a hyperacetylated state in cardiac myocytes37. This in turn may lead to a posttranslational modification of enzymes involved in cellular energy metabolism, causing detrimental metabolic perturbations in the failing heart.
However, 3-HB has been shown to promote the myocardial blood flow in healthy humans35, and 3-HB is a natural inhibitor of class I histone deacetylases (HDAC)38. Since HDAC inhibitors can block cardiac fibrosis and thus improve diastolic function39,40, elevated 3-HB serum levels might be beneficial for the failing heart. Along these lines, it would be important to examine whether ketogenic diet-induced ketosis, reported to exert positive effects on cardiovascular risk in some but not all studies41, would be beneficial or detrimental in AHF.
Increased serum citrate levels in patients who died within three months after onset of AHF further substantiate an increased supply of the liver and other tissues with fatty acids. Under this condition, an augmented fatty acid ß-oxidation gives rise to the increased levels of TCA cycle intermediates, including citrate. Citrate is a known potent allosteric inhibitor of pyruvate dehydrogenase which is why increased intracellular citrate levels redirect the conversion of pyruvate from acetyl coenzyme A to lactate, consequently leading to increased lactate plasma levels42,43. Indeed, the lactate serum levels were higher in AHF patients who died within three months after onset of AHF compared to those who survived and were significantly positively correlated with citrate serum levels (r = 0.37, p < 0.001). Besides high intracellular citrate levels, diminished oxygen supply to the tissue due to a decreased tissue perfusion and congestion, which are both hemodynamic hallmarks of HF, augments anaerobic glycolysis and lactate production44. In the present study, the association of lactate serum levels with 3-month mortality did not remain significant (p = 0.061) after adjusting for parameters which are associated with mortality in HF patients. This is in contrast with findings of a recent study that showed a significant association of lactate with 1-year mortality in AHF patients45. A possible explanation for this discrepancy is that lactate may have more of a long-term rather than a short-term prognostic capacity for mortality in AHF.
The serum levels of tyrosine at admission were also higher in patients who died within three months compared to those who were alive three months after onset of AHF. In the present study, tyrosine serum levels were significantly positively correlated with the serum levels of NT-proBNP, IL-6, and 2-HB (r = 0.24, P = 0.007), the markers of HF severity, inflammation, and oxidative stress, respectively, which are known to be associated with increased muscle proteolysis and protein turnover46,47. However, the fact that the serum levels of other amino acids were similar in the two patient groups presents an argument against the breakdown of proteins in the skeletal muscle being responsible for increased tyrosine serum levels in AHF patients that did not survive for three months. Therefore, the decreased uptake and use of tyrosine as a substrate for the biosynthesis of various biological molecules, including thyroid hormones, catecholamines, neurotransmitters, or serum proteins48, may better explain higher tyrosine serum levels in the group of patients who died within three months after onset of AHF. We observed a significant positive correlation of the tyrosine serum levels with AST and a significant negative correlation with serum fibrinogen levels in the present study. These strongly argue for a contribution of an impaired liver function and hepatocyte damage, possibly due to hypoperfusion and/or congestion, to the increased tyrosine serum levels. This is in line with results of previous studies showing that a decreased liver function and/or damage to the liver result in increased serum levels of tyrosine and other aromatic amino acids49.
Besides tyrosine, we observed positive correlations of 3-HB, 2-HB, lactate, and citrate with serum levels of liver transaminases, which are known to be increased as a consequence of hepatocyte damage due to reduced perfusion14. This suggests that not only the increased production but also a lower uptake by the liver as well as an augmented release from damaged hepatocytes likely contribute to the increased serum levels of those metabolites44.
The serum levels of citrate and 3-HB were negatively correlated with GFR, suggesting that the renal excretion might be factor in the regulation of the serum levels of these metabolites in AHF. Alternatively, the negative relationship between the levels of these metabolites and GFR may conceivably be a consequence of their opposite regulation by the HF pathophysiology, which most likely also explains the negative correlations of both citrate and tyrosine with various serum lipids, BMI, and MAP, which are frequently decreased in HF50.
This study is not free of limitations: Due to the study design we could not examine the mechanistic relationship between the underlying pathophysiological processes and the serum levels of the identified metabolites. In addition, our metabolic profiles are just a snap shot of the patients’ metabolic state at hospital admission without capturing dynamic changes in the levels of the metabolites during hospitalization. Thus we are unable to assess any temporal development or the impact of therapeutic interventions. Furthermore, we have no data on the patients’ nutritional state, i.e. whether and how long they were fasting before blood collection. Moreover, we profiled only water-soluble metabolites without addressing lipid metabolites. Finally, because the statistical power of our analyses is affected by the moderate number of available serum samples (n = 130), our results need to be confirmed in further and larger studies.
We conclude that serum levels of 2-HB, 3-HB, tyrosine, and citrate are associated with increased 3-month mortality in AHF patients and might thus be of prognostic value in AHF.
Methods
Study design and patients
Details of the study and its patient cohort have already been described in depth21,22,23,24. We conducted a prospective observational single-center study including consecutive hospitalized AHF patients. Written informed consent was obtained from all patients. The study was conducted in adherence to the ethical guidelines of the Declaration of Helsinki51, and was approved by the Ethics Committees of the University Hospital Centre Sisters of Charity, Zagreb, Croatia and the Medical University of Graz, Austria. The patients were treated by local standard operating procedures outlined by the ESC Guidelines for AHF3,52.
Laboratory procedures
The blood sampling and laboratory methods have already been described in previous reports on our AHF cohort21,22,23,24.
NMR metabolic profiling
To remove serum proteins and to quench enzymatic reactions in the samples 200 µL serum were mixed with 400 µL methanol and stored at −20 °C for 7 days until further processing. Afterwards the samples were spun at 17949 rcf at 4 °C for 30 minutes. Supernatants were lyophilized and mixed with 500 μL of NMR buffer in D2O and transferred to 5 mm NMR tubes. Metabolites were measured as described previously53.
Spectral acquisition was performed on a 600 MHz Bruker Avance Neo NMR spectrometer equipped with a TXI 600S3 probehead and processing was performed as previously described53. Spectra pre-processing and data analysis have been carried out as previously described54 using Principle Component Analysis (PCA), O-PLS-DA55, and all associated data consistency checks as well as 7-fold cross-validation. In order to validate the statistical significance of the determined differences between patients that did and did not survive three months, the quality assessment statistic Q2 is reported. This measure provides information about cross-validation and is a qualitative measure of consistency between the predicted and original data, with a maximum value of 1. Processed spectra were imported into MestreNova 12.0.2 in order to quantify metabolites of interest. Glucose quantification was performed using Chenomx Professional 8.0 with the existing Chenomx library.
Statistical analyses
Patients’ baseline characteristics, laboratory parameters, and metabolite serum levels were descriptively analyzed using absolute and relative frequencies or median and range. Differences between patients who did and did not survived the first three months after onset of AHF were assessed either by Fisher’s exact or the Mann-Whitney U test. Correlations between the metabolites and various clinical and laboratory parameters were determined by the Spearman correlation coefficient. Metabolite concentrations were used to perform an ROC curves analysis in MetaboAnalyst 4.056. Additionally, the impact of the metabolites on 3-month mortality was examined using univariable as well as multivariable logistic regression analyses. The latter was adjusted for age, sex, BMI, T2DM, NT-proBNP, GFR, MAP as well as LDL cholesterol. Odds ratios (OR) along with the respective 95% confidence intervals (CI) are presented. In order for the resulting ORs to be on an interpretable scale, all metabolites were log-transformed for the regression analyses. The variance inflation factor was used to assess the degree of multi-collinearity in the models. R version 3.4.4 was used for the statistical analyses.
All methods were carried out in accordance with the relevant guidelines and regulations.
Data Availability
All data generated or analysed during this study are included in this manuscript.
References
Roger, V. L. et al. Trends in heart failure incidence and survival in a community-based population. JAMA 292, 344–350, https://doi.org/10.1001/jama.292.3.344 (2004).
Dickstein, K. et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur Heart J 29, 2388–2442, https://doi.org/10.1093/eurheartj/ehn309 (2008).
McMurray, J. J. et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J 33, 1787–1847, https://doi.org/10.1093/eurheartj/ehs104 (2012).
Neubauer, S. The failing heart–an engine out of fuel. N Engl J Med 356, 1140–1151, https://doi.org/10.1056/NEJMra063052 (2007).
Doehner, W., Frenneaux, M. & Anker, S. D. Metabolic impairment in heart failure: the myocardial and systemic perspective. J Am Coll Cardiol 64, 1388–1400, https://doi.org/10.1016/j.jacc.2014.04.083 (2014).
Ardehali, H. et al. Targeting myocardial substrate metabolism in heart failure: potential for new therapies. Eur J Heart Fail 14, 120–129, https://doi.org/10.1093/eurjhf/hfr173 (2012).
Ryden, M. & Arner, P. Fat loss in cachexia–is there a role for adipocyte lipolysis? Clin Nutr 26, 1–6, https://doi.org/10.1016/j.clnu.2006.09.009 (2007).
Ryden, M. et al. Targets for TNF-alpha-induced lipolysis in human adipocytes. Biochem Biophys Res Commun 318, 168–175, https://doi.org/10.1016/j.bbrc.2004.04.010 (2004).
Sengenes, C., Berlan, M., De Glisezinski, I., Lafontan, M. & Galitzky, J. Natriuretic peptides: a new lipolytic pathway in human adipocytes. FASEB J 14, 1345–1351 (2000).
Szabo, T. et al. Increased catabolic activity in adipose tissue of patients with chronic heart failure. Eur J Heart Fail 15, 1131–1137, https://doi.org/10.1093/eurjhf/hft067 (2013).
Swan, J. W. et al. Insulin resistance in chronic heart failure: relation to severity and etiology of heart failure. J Am Coll Cardiol 30, 527–532 (1997).
von Haehling, S., Doehner, W. & Anker, S. D. Nutrition, metabolism, and the complex pathophysiology of cachexia in chronic heart failure. Cardiovasc Res 73, 298–309, https://doi.org/10.1016/j.cardiores.2006.08.018 (2007).
Marcinkiewicz-Siemion, M., Ciborowski, M., Kretowski, A., Musial, W. J. & Kaminski, K. A. Metabolomics - A wide-open door to personalized treatment in chronic heart failure? Int J Cardiol 219, 156–163, https://doi.org/10.1016/j.ijcard.2016.06.022 (2016).
Cagli, K., Basar, F. N., Tok, D., Turak, O. & Basar, O. How to interpret liver function tests in heart failure patients? Turk J Gastroenterol 26, 197–203, https://doi.org/10.5152/tjg.2015.0086 (2015).
Seeto, R. K., Fenn, B. & Rockey, D. C. Ischemic hepatitis: clinical presentation and pathogenesis. Am J Med 109, 109–113 (2000).
Bray, R. et al. Urinary Metabolic Phenotyping of Women with Lower Urinary Tract Symptoms. J Proteome Res 16, 4208–4216, https://doi.org/10.1021/acs.jproteome.7b00568 (2017).
Chan, Q. et al. Metabolic phenotyping for discovery of urinary biomarkers of diet, xenobiotics and blood pressure in the INTERMAP Study: an overview. Hypertens Res 40, 336–345, https://doi.org/10.1038/hr.2016.164 (2017).
Villasenor, A. et al. 1H NMR global metabolic phenotyping of acute pancreatitis in the emergency unit. J Proteome Res 13, 5362–5375, https://doi.org/10.1021/pr500161w (2014).
Dona, A. C. et al. Precision high-throughput proton NMR spectroscopy of human urine, serum, and plasma for large-scale metabolic phenotyping. Anal Chem 86, 9887–9894, https://doi.org/10.1021/ac5025039 (2014).
Dumas, M. E., Kinross, J. & Nicholson, J. K. Metabolic phenotyping and systems biology approaches to understanding metabolic syndrome and fatty liver disease. Gastroenterology 146, 46–62, https://doi.org/10.1053/j.gastro.2013.11.001 (2014).
Radulovic, B. et al. Hypochloraemia as a predictor of developing hyponatraemia and poor outcome in acute heart failure patients. Int J Cardiol 212, 237–241, https://doi.org/10.1016/j.ijcard.2016.03.081 (2016).
Potocnjak, I. et al. Serum concentrations of asymmetric and symmetric dimethylarginine are associated with mortality in acute heart failure patients. Int J Cardiol 261, 109–113, https://doi.org/10.1016/j.ijcard.2018.03.037 (2018).
Potocnjak, I. et al. Serum Concentration of HDL Particles Predicts Mortality in Acute Heart Failure Patients. Sci Rep 7, 46642, https://doi.org/10.1038/srep46642 (2017).
Degoricija, V. et al. Acute Heart Failure developed as worsening of Chronic Heart Failure is associated with increased mortality compared to de novo cases. Sci Rep 8, 9587, https://doi.org/10.1038/s41598-018-28027-3 (2018).
Passantino, A., Monitillo, F., Iacoviello, M. & Scrutinio, D. Predicting mortality in patients with acute heart failure: Role of risk scores. World J Cardiol 7, 902–911, https://doi.org/10.4330/wjc.v7.i12.902 (2015).
Hunter, W. G., Kelly, J. P., McGarrah, R. W. III, Kraus, W. E. & Shah, S. H. Metabolic Dysfunction in Heart Failure: Diagnostic, Prognostic, and Pathophysiologic Insights From Metabolomic Profiling. Curr Heart Fail Rep 13, 119–131, https://doi.org/10.1007/s11897-016-0289-5 (2016).
Wende, A. R., Brahma, M. K., McGinnis, G. R. & Young, M. E. Metabolic Origins of Heart Failure. JACC Basic Transl Sci 2, 297–310, https://doi.org/10.1016/j.jacbts.2016.11.009 (2017).
Ambrosy, A. P. et al. Clinical course and predictive value of congestion during hospitalization in patients admitted for worsening signs and symptoms of heart failure with reduced ejection fraction: findings from the EVEREST trial. Eur Heart J 34, 835–843, https://doi.org/10.1093/eurheartj/ehs444 (2013).
Gall, W. E. et al. alpha-hydroxybutyrate is an early biomarker of insulin resistance and glucose intolerance in a nondiabetic population. PLoS One 5, e10883, https://doi.org/10.1371/journal.pone.0010883 (2010).
Ferrannini, E. et al. Early metabolic markers of the development of dysglycemia and type 2 diabetes and their physiological significance. Diabetes 62, 1730–1737, https://doi.org/10.2337/db12-0707 (2013).
Karpe, F., Dickmann, J. R. & Frayn, K. N. Fatty acids, obesity, and insulin resistance: time for a reevaluation. Diabetes 60, 2441–2449, https://doi.org/10.2337/db11-0425 (2011).
Lord, R. S. & Bralley, J. A. Clinical applications of urinary organic acids. Part I: Detoxification markers. Altern Med Rev 13, 205–215 (2008).
Landaas, S. & Pettersen, J. E. Clinical conditions associated with urinary excretion of 2-hydroxybutyric acid. Scand J Clin Lab Invest 35, 259–266 (1975).
Janardhan, A., Chen, J. & Crawford, P. A. Altered systemic ketone body metabolism in advanced heart failure. Tex Heart Inst J 38, 533–538 (2011).
Gormsen, L. C. et al. Ketone Body Infusion With 3-Hydroxybutyrate Reduces Myocardial Glucose Uptake and Increases Blood Flow in Humans: A Positron Emission Tomography Study. J Am Heart Assoc 6, https://doi.org/10.1161/JAHA.116.005066 (2017).
Karwi, Q. G., Uddin, G. M., Ho, K. L. & Lopaschuk, G. D. Loss of Metabolic Flexibility in the Failing Heart. Front Cardiovasc Med 5, 68, https://doi.org/10.3389/fcvm.2018.00068 (2018).
Horton, J. L. et al. Mitochondrial protein hyperacetylation in the failing heart. JCI Insight 2, https://doi.org/10.1172/jci.insight.84897 (2016).
Shimazu, T. et al. Suppression of oxidative stress by beta-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 339, 211–214, https://doi.org/10.1126/science.1227166 (2013).
Jeong, M. Y. et al. Histone deacetylase activity governs diastolic dysfunction through a nongenomic mechanism. Sci Transl Med 10, https://doi.org/10.1126/scitranslmed.aao0144 (2018).
Yoon, S. & Eom, G. H. HDAC and HDAC Inhibitor: From Cancer to Cardiovascular Diseases. Chonnam Med J 52, 1–11, https://doi.org/10.4068/cmj.2016.52.1.1 (2016).
Kosinski, C. & Jornayvaz, F. R. Effects of Ketogenic Diets on Cardiovascular Risk Factors: Evidence from Animal and Human Studies. Nutrients 9, https://doi.org/10.3390/nu9050517 (2017).
Alexander, D., Lombardi, R., Rodriguez, G., Mitchell, M. M. & Marian, A. J. Metabolomic distinction and insights into the pathogenesis of human primary dilated cardiomyopathy. Eur J Clin Invest 41, 527–538, https://doi.org/10.1111/j.1365-2362.2010.02441.x (2011).
Zordoky, B. N. et al. Metabolomic fingerprint of heart failure with preserved ejection fraction. PLoS One 10, e0124844, https://doi.org/10.1371/journal.pone.0124844 (2015).
Orn, S. & van Hall, G. Does a normal peripheral lactate value always indicate an aerobic tissue metabolism? Eur J Heart Fail 19, 1034–1035, https://doi.org/10.1002/ejhf.863 (2017).
Zymlinski, R. et al. Increased blood lactate is prevalent and identifies poor prognosis in patients with acute heart failure without overt peripheral hypoperfusion. Eur J Heart Fail, https://doi.org/10.1002/ejhf.1156 (2018).
Pasini, E. et al. Hypercatabolic syndrome: molecular basis and effects of nutritional supplements with amino acids. Am J Cardiol 101, 11E–15E, https://doi.org/10.1016/j.amjcard.2008.02.074 (2008).
Libera, L. D. & Vescovo, G. Muscle wastage in chronic heart failure, between apoptosis, catabolism and altered anabolism: a chimaeric view of inflammation? Curr Opin Clin Nutr Metab Care 7, 435–441 (2004).
Tenori, L. et al. Metabolomic fingerprint of heart failure in humans: a nuclear magnetic resonance spectroscopy analysis. Int J Cardiol 168, e113–115, https://doi.org/10.1016/j.ijcard.2013.08.042 (2013).
Dejong, C. H., van de Poll, M. C., Soeters, P. B., Jalan, R. & Olde Damink, S. W. Aromatic amino acid metabolism during liver failure. J Nutr 137, 1579S–1585S; discussion 1597S–1598S, https://doi.org/10.1093/jn/137.6.1579S (2007).
Kalantar-Zadeh, K., Block, G., Horwich, T. & Fonarow, G. C. Reverse epidemiology of conventional cardiovascular risk factors in patients with chronic heart failure. J Am Coll Cardiol 43, 1439–1444, https://doi.org/10.1016/j.jacc.2003.11.039 (2004).
World Medical, A. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310, 2191–2194, https://doi.org/10.1001/jama.2013.281053 (2013).
Yancy, C. W. et al. ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 62, e147–239, https://doi.org/10.1016/j.jacc.2013.05.019 (2013).
Alkan, H. F. et al. Cytosolic Aspartate Availability Determines Cell Survival When Glutamine Is Limiting. Cell Metab 28, 706–720 e706, https://doi.org/10.1016/j.cmet.2018.07.021 (2018).
Huber, K. et al. N-acetylaspartate pathway is nutrient responsive and coordinates lipid and energy metabolism in brown adipocytes. Biochim Biophys Acta Mol Cell Res 1866, 337–348, https://doi.org/10.1016/j.bbamcr.2018.08.017 (2019).
Maher, A. D. et al. Optimization of human plasma 1H NMR spectroscopic data processing for high-throughput metabolic phenotyping studies and detection of insulin resistance related to type 2 diabetes. Anal Chem 80, 7354–7362, https://doi.org/10.1021/ac801053g (2008).
Chong, J. et al. MetaboAnalyst 4.0: towards more transparent and integrative metabolomics analysis. Nucleic Acids Res, https://doi.org/10.1093/nar/gky310 (2018).
Acknowledgements
This research was supported by the Austrian Science Foundation [P27166-B23 to S.F., P28854 and I3792 to T.M.], the Jubilee Foundation of the Austrian National Bank [15858 to S.F.], the President’s International Fellowship Initiative of CAS (No. 2015VBB045, to T.M.), the National Natural Science Foundation of China (No. 31450110423, to T.M.), the Austrian Research Promotion Agency (FFG: 864690), the Integrative Metabolism Research Center Graz, the Austrian infrastructure program 2016/2017, BioTechMed/Graz, and the OMICS Center Graz. S.S. was trained within the frame of the PhD program Molecular Medicine, Medical University of Graz. The funders had no roles in the design of the study, data collection, analysis, and interpretation, report writing or article submission.
Author information
Authors and Affiliations
Contributions
Experiments conceived and designed by: V.D., M.T., T.M. and S.F. Experiments carried out by: S.S., I.P., V.D., M.T., M.G. and B.R. Contribution of reagents/materials/analysis tools: S.F. and T.M. Contributed significantly to the execution, the analysis or both and interpretation of data: I.P., M.T., G.P., A.B., V.D., T.M., S.S. and S.F. Paper was written by: T.M. and S.F. Participated in reviewing, and/or revising the manuscript for intellectual content: S.S., M.G., V.D., I.P., M.T., B.R., G.P., A.B., T.M. and S.F. The manuscript was approved for submission by: S.S., M.G., V.D., I.P., M.T., B.R., G.P., A.B., T.M. and S.F.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stryeck, S., Gastrager, M., Degoricija, V. et al. Serum Concentrations of Citrate, Tyrosine, 2- and 3- Hydroxybutyrate are Associated with Increased 3-Month Mortality in Acute Heart Failure Patients. Sci Rep 9, 6743 (2019). https://doi.org/10.1038/s41598-019-42937-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-42937-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.