Abstract
We compare physiological responses of the crustacean copepod Calanus pacificus and pelagic pteropod mollusk Limacina helicina to ocean temperatures and pH by measuring biomarkers of oxidative stress, antioxidant defences, and the activity of the respiratory electron transport system in organisms collected on the 2016 West Coast Ocean Acidification cruise in the California Current System. Copepods and pteropods exhibited strong but divergent responses in the same habitat; copepods had higher oxygen-reactive absorbance capacity, glutathione-S-transferase, and total glutathione content. The ratio between reduced to oxidised glutathione was higher in copepods than in pteropods, indicating lower oxidative stress in copepods. Pteropods showed higher activities of glutathione reductase, catalase, and lipid peroxidation, indicating increased antioxidant defences and oxidative stress. Thus, the antioxidant defence system of the copepods has a greater capacity to respond to oxidative stress, while pteropods already face severe stress and show limited capacity to deal with further changes. The results suggest that copepods have higher adaptive potential, owing to their stronger vertical migration behaviour and efficient glutathione metabolism, whereas pteropods run the risk of oxidative stress and mortality under high CO2 conditions. Our results provide a unique dataset and evidence of stress-inducing mechanisms behind pteropod ocean acidification responses.
Similar content being viewed by others
Introduction
The California Current System (CCS) along the west coast of North America from British Columbia to Baja California is a region characterised by seasonal upwelling. Coastal regions of the CCS are currently experiencing pH levels well below those that are expected for the global surface ocean later in this century1, and conditions are rapidly changing due to anthropogenic carbon uptake2, compromising the health of pelagic calcifiers3. The shallow shelf areas off California-Oregon-Washington coasts are expected to become undersaturated with respect to aragonite with increasing frequency over the next few decades4,5. In addition, the northeast Pacific was subjected to a marine heatwave event that occurred from late 2013 until the end of 20156,7 overlapping with the strong 2015–16 El Niño7. As a result of these climatic phenomena, surface water temperatures in the northeast Pacific reached anomalies of >3 °C above normal8 with lasting ecological consequences over the following years.
With ongoing ocean acidification, marine calcifying organisms such as pteropods will be exposed more frequently to low quality pelagic habitat with respect to aragonite saturation state (Ωarag), an important parameter affecting the ability for pteropods to form their shells. Ocean warming will add further stress to cool-water pteropods, such as Limacina helicina. Pteropod populations have decreased in abundance9 and are today considered important bio-indicators for ocean acidification3. Pacific pteropods L. helicina are essential players in the marine food web, particularly as prey for many commercial fish species10.
Copepods, in contrast, are generally considered fairly robust to climate change. Their resilience could partly be linked to stronger vertical migration behaviour, during which they encounter a large gradient of physico-chemical factors11. Some species such as Calanus spp. diapause at great depths12. Crustacean copepods have one of the most lightly calcified cuticles of all crustaceans13, and therefore are relatively non-calcifying compared to other taxa, particularly compared to pteropods with aragonitic shells. The species Calanus pacificus is an abundant member of the zooplankton community in NE Pacific waters14, and forms a key species throughout the CCS15. C. pacificus is important in food webs, and has declined strongly in abundance during El Niño-related marine heatwaves15,16.
Biomarkers have increased in popularity among ecologists17, predominantly because of interest in the role of oxidative stress in life-history tradeoffs18 and for understanding the mechanisms behind animal behavioural, reproductive, and survival-related performance19. Under steady-state conditions, reactive oxygen species (ROS), such as peroxides and superoxide, formed as a by-product of oxygen (Table 1), are scavenged by various antioxidative defence mechanisms20. The balance between oxidative stress and antioxidant defences can be perturbed as a consequence of warming, acidification, pollution, hypoxia21,22,23, natural variability24, or differential allocation of resources to immunity, reproduction, or growth19. When the equilibrium between pro-oxidants and antioxidants is unbalanced, due to high pro-oxidants or insufficient antioxidant activity, this can lead to lower survival18. An understanding of cellular responses due to oxidative stress would provide important insight to the stress effects and factors that set limits to species’ tolerance to ocean temperature, oxygen, and inorganic carbon system conditions in the natural oceanic habitat.
The primary objective of this study was to relate physiological condition of field- collected pteropods and copepods (Fig. 1) to oceanographic conditions that are expected to change significantly with climate change, including mainly temperature and carbonate biogeochemistry. By relating oxidative stress to field conditions, we can examine biomarkers measured in situ in both taxa over a range of biogeochemical and temperature conditions. To our knowledge, this is the first study to report and compare biomarker outcomes for pteropods and copepods collected over a large gradient of temperature and biogeochemical conditions. Our main hypothesis is that the pelagic pteropod L. helicina shows higher oxidative stress and lower antioxidant defences compared with C. pacificus copepods.
Overview map of the study area showing location and station number of WCOA2016 sampling stations. Copepods and pteropods were sampled for biomarkers at stations represented by empty circles and crosses, respectively. A circle with a cross inside means that both copepods and pteropods were collected at the station. Offshore stations (>200 m water depth) are red, and onshore stations (<200 m) are black. Copepods were collected for ETS (Respiratory Electron Transport System) activity at stations 79, 85, 99, 122, 128, and 129.
Results
The results from the 2016 West Coast Ocean Acidification (WCOA2016) cruise to the northern California Current System (NCCS, Fig. 1) indicated that seawater property distributions reflected the strong influence of nearshore upwelling in the NCCS where pteropods and copepods were collected. The collective signatures of upwelling and the biogeochemical processes stimulated by macronutrients supplied by upwelling were quite different between the surface and 50 m depth (Fig. 2). At 50 m depth, temperatures, aragonite saturation state, and oxygen concentrations were lower nearshore than offshore, due to upwelling of deeper, colder, oxygen-depleted, and nutrient- and CO2-rich waters with low aragonite saturation states. At the surface, this cold water can rapidly exchange heat and gases with the atmosphere while warming, consequently resulting in degassing of CO2, uptake of O2, and elevating the aragonite saturation state. These changes take place along with the micro-algal uptake of CO2 and release of O2 in the nutrient-rich, well-lit surface ocean waters. As a result of biological cycling and gas exchange, the average apparent oxygen utilisation observed on the WCOA2016 cruise between 40 and 50°N went from as much as −30 µmol kg−1 O2 at the surface (0–10 m) to 90 µmol kg−1 O2 at mid depth (between 45 and 55 m). The highest surface aragonite saturation states were observed in nearshore surface waters where biological drawdown of CO2 was intense. Surface temperatures in May and June varied around 12–13 °C in the northern part of the study area, whereas temperatures were slightly higher (13–15 °C) off the Oregon-Washington coasts (Fig. 2). Temperatures were lower at the 50 m depth horizon and varied between 8–11 °C in the study area. Dissolved oxygen concentrations were high in the surface waters varied between 250–450 µmol kg−1. At 50 m depth, the dissolved oxygen concentrations were considerably lower, in the range of 100 and 280 µmol kg−1. Surface Ωarag ranged from 2.0 to 3.5, but at 50 m depth, Ωarag values were much lower, ranging from 0.5 to 1.5 in nearshore waters and 1.0–2.0 in offshore waters. Farther away from the coast, Ωarag was approximately 1.5 at 50 m depth (Fig. 2).
Temperature, oxygen, and aragonite saturation state in the surface waters (A–C) and at 50 m depth (D–F) of the Northern California Current System (Oregon-Washington-British Columbia). Carbonate chemistry gradients reflect a range of conditions relevant to ocean acidification, along which biological samples were taken at selected stations represented by green dots. Ω = 1 indicates that carbonate minerals are in equilibrium with the surrounding seawater.
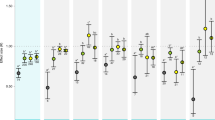
All biomarkers including both oxidative stress and antioxidant defences, except Superoxide dismutase (SOD), were significantly different between pteropods L. helicina and copepods C. pacificus (Linear Mixed Model, usually p < 0.001; Fig. 3, Table 2), apart from GP (glutathione peroxidase) and ETS (electron transport system) that were measured only in copepods. Glutathione system (GST) activity, total GSH (glutathione) concentration, Oxygen Radical Absorbance Capacity (ORAC), and reduced to oxidised glutathione (GSH:GSSG) ratio were higher in copepods compared with pteropods. Glutathione reductase (GR) and catalase (CAT) activities and the lipid peroxidation (LPX) were higher, and ORAC:LPX ratio was lower in pteropods than in copepods (Fig. 3). ORAC:LPX and GSH:GSSG ratios (the lower ratios, the more oxidative stress) in pteropods decreased from north to south, while LPX concentration and SOD activity increased towards the south (Fig. 3). For copepods, there were no significant major changes in biomarkers with latitude.
Biomarkers of copepods and pteropods by station (latitude; i.e., south S to north N), graphically presenting species differences analysed by Linear Mixed Models (Table 2). Each point represents a single sample. Units are given as: GPx, GR, GST, and CAT in µmol min−1 mg−1 protein, SOD as % inhibition, and total glutathione given as µM mg−1 protein, LPX as µM cumene-hydroperoxide equivalents mg−1 protein, and ETS as µ1 O2 h−1 ind−1. Abbreviations as in Table 2.
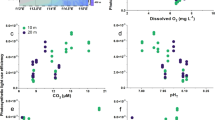
Concerning environmental conditions, the response by species measured as GST and CAT activities was significantly affected by pH (p < 0.05, and p < 0.01, respectively), whereas differences in SOD activity were significantly affected by pCO2 (p < 0.05). The response by species measured as LPX showed a significant effect by pCO2 (p < 0.001). In addition, we found that LPX concentrations in species were different due to temperature and pCO2 (analysed as interactions between species and temperature p < 0.05, or pCO2 p < 0.0001). pCO2 concentrations affected also the ORAC activities by the two species (p < 0.01) (Fig. 4).
Significant relationships of pteropod and copepod biomarkers in relation to selected environmental variables. (a) LPX vs. pCO2, (b) LPX vs. temperature, (c) ORAC vs. temperature, (d) SOD vs. pCO2, (e) GST vs. pH, and (f) CAT vs. pH. The graphs present the mixed model results (Table 2). Abbreviations as in Table 2.
Biomarkers and environmental variables
In the copepod principal component analysis (PCA), GST, ORAC, and CAT were positively associated with Principal Component 1 (PC1), with pCO2, aragonite saturation, salinity and total alkalinity (TA) as main environmental drivers. PC2 was positively associated with SOD and negatively associated with GR, LPX, GSH:GSSG ratio and glutathione peroxidase (GP), with temperature and oxygen as the significant environmental variables. Of the total observed variation, 97.9% was explained by the first two principal components, 87.0% by PC1 and 10.9% by PC2 (Fig. 5a, Table 3a).
Schematic graphs of Principal Component Analysis (PCA) scores for Components 1 and 2 for copepods Calanus pacificus (a), and for pteropods Limacina helicina (b). The corresponding loading plots (biomarker variables and environmental variables) are superimposed on the scores plot. Biomarker abbreviations as in Table 3. Aragonite saturation state (Ara), Temperature (Temp), Salinity (Sal), Dissolved oxygen concentrations (O2), partial pressure of Carbon dioxide in seawater (pCO2), and Total Alkalinity (TA). Environmental data were integrated over the upper 100 m.
In the PCA using pteropod data, ORAC, LPX, and CAT were positively associated with PC1, whereas GST had an inverse relationship with these variables. PC2 was positively associated with GR and GSH:GSSG ratio, and negatively associated with SOD. For pteropods, temperature was the most important environmental factor, and aragonite saturation Ωarag as second most important factor in the PCA. The two main components explained in total > 90% of the observed variance in the PCA: 75.2% by PC1 and 18.4% by PC2 (Fig. 5b, Table 3b).
Discussion
We present a unique dataset as first evidence to demonstrate that biomarkers measured in copepods and pteropods in the Northern California Current System show strong, yet differential, relationships with temperature and pH/pCO2. Our results clearly show higher oxidative damage (elevated LPX and decreased GSH:GSSG ratio) in pelagic pteropods compared to the copepods, whereas for most antioxidants, especially the glutathione system (GST and total glutathione), copepods have higher activities.
The ORAC:LPX ratio indicates oxidative stress, with a low ratio expressing a higher level of oxidative stress25, and the ratio was approximately one tenth as high in Limacina pteropods as in Calanus copepods (Fig. 3a). The ORAC:LPX ratio of pteropods decreased towards the south, and despite the ratio did not have a significant relationship with environmental conditions, ORAC was significantly related to temperature. There was also interactions between species and temperature or pCO2 measured as LPX, showing that pteropods responded more strongly to thermal stress and increasing pCO2 (Fig. 4), of which the latter was observed to be as high as 725 µatm off the Oregon coast. Also, pH, oxygen and temperature were significant factors affecting antioxidant defences and oxidative stress levels (Figs 4 and 5; Tables 2 and 3), showing that several factors combined are stressful for pteropods. In the PCA, temperature, in addition to Ωarag, was by far the most important environmental factor influencing pteropod stress levels. These results suggest harmful changes on a cellular level in pteropods are already occurring at some locations in the NCCS. Low Ωarag combined with higher temperatures could cause the cumulative effects observed in pteropods26, and result in elevated lipid peroxidation and antioxidant enzymes27,28,29. Vehmaa et al.25 reported a decreasing ORAC:LPX ratio as a response to manipulated warming and pH in Acartia copepods. Baselines of biomarkers measured in copepods can be difficult to rely on, as spatio-temporal conditions vary greatly among field studies.
Accounting for this, reported LPX concentration ranges from previous copepod studies are 0.1–21 µM cumene-hydroperoxide equivalents mg−1 in Limnocalanus macrurus30, 29–53 in Acartia sp.31, and 0.3–3.3 µM cumene-hydroperoxide equivalents mg−1 in Calanus finmarchicus (exposed to severe manipulated ocean acidification, unpubl. data). In the current study Calanus spp. LPX concentrations were low, around 0.5 µM cumene-hydroperoxide equivalents mg−1 (cf. Fig. 3), suggesting that the copepods can defend themselves against external stress factors at the cellular level, and keep the balance between ROS and antioxidant defences stable.
Biomarkers (CAT, LPX, SOD, GST) that showed a significant relationship with pH or pCO2 showed the most extreme values in samples collected in the region of regularly occurring upwelling of CO2-rich waters off the Washington-Oregon-California coast1,4. Upwelling exposes organisms to corrosive waters that are depleted in carbonate ion, a central component for CaCO3 shell building3,26,32. If pteropod habitat is undersaturated with respect to aragonite (Ωarag ≤ 1), it will lead to shell thinning and dissolution, which increases vulnerability to predation and imposes increased energetic costs33,34, especially when undersaturation stress is interacting with other in situ stressors, such as thermal stress and hypoxia35. The observed pteropod LPX content also showed a relationship with oxygen concentrations in the PCA (Fig. 5b), which would be expected as pteropods are sensitive to hypoxia, in combination with other stressors36. The energetic cost for pteropods to cope with acidified conditions has not been determined37, but Lischka & Riebesell37 suggest that decreased growth causes decreased activity and metabolism, leading to decreased internal lipids, slower calcification, and discontinued development of gonads.
The GSH:GSSG ratio, which is lower for organisms experiencing oxidative stress, decreased in pteropods with decreasing latitude, suggesting that the environmental conditions in the south are more harmful for them than in the north. As GSH:GSSG ratio decreased in pteropods (Fig. 3), GR should increase. As the function of GR is to reduce GSSG to GSH30, and thereby elevate the GSH:GSSG ratio, this suggests the enhanced stress in the southern NCCS is exceeding the pteropods’ ability to cope by increasing their GR activity. In mammals, a cell in resting phase has a GSH:GSSG ratio exceeding 100:1, whereas a cell suffering from severe oxidative stress has a ratio ~10:1, or 1:130,38. A low GSH:GSSG ratio indicates cellular toxicity and is a biomarker that works across different functional groups, from plankton to mammals30,38. The consequences of a low GSH:GSSG ratio (Figs 3 and 5), in combination with low antioxidant capacity, has so far not been investigated considering pteropod life cycle and mortality risk, but is the focus of our recent efforts35. Two thirds of the pteropod GSH:GSSG ratio samples were <1 in the current paper, suggesting serious oxidative stress30,38.
Considering differences in habitat use of pteropods vs. copepods as a potential explanation for their differences in biomarkers, Calanus pacificus and L. helicina seem to perform diel vertical migration (DVM) across fairly similar depth ranges in the California Current (~100 m)39,40,41,42, although relatively limited data on pteropod DVM are available to characterise the depth they regularly reach. A potentially important difference is that Calanus spp. diapause at depths > 1000 m12 whereas Limacina does not diapause. Lewis et al.11 observed that adult Calanus sp. in the Arctic, which migrate daily across a >140 μatm gradient of pCO2 in their natural habitat, exhibit little response to high CO2 in the laboratory, indicating a potential resilience to high CO2 conferred by their daily exposure. Thus, perhaps via carry-over effects (i.e., effects of previous experience that persist through the life cycle43), copepods that are exposed to a large range of physico-chemical conditions that elicit production of ROS over their life cycle may have higher base levels of antioxidants for efficient elimination of ROS than do pteropods.
In addition, copepods have an efficient glutathione metabolism44 (see Fig. 5), which responds rapidly to changing hydrography30,31. Calanus showed higher antioxidant defences (GSTs, total GSH) than pteropods, indicating a larger capacity for ROS scavenging45. This is also supported by our observations of elevated ORAC in copepods30, detected in warmer conditions and in low pH waters during a laboratory study. Detoxification is a fundamental defence mechanism, which allows the organism to survive or even thrive despite oxidative stress, with the GST superfamily, a set of enzymes involved in the detoxification process, being involved in this44. Previous work suggests that copepods are more sensitive to warming than to acidification25,46,47, and shown by using biomarkers in Vehmaa et al.25. Also, a review by Havenhand48 suggests that copepods will likely be resilient to near-future OA.
Catalase (CAT) is an antioxidant enzyme that directly converts hydrogen peroxide (H2O2), a harmful substance for the cell, to oxygen and water. CAT activities were much higher (33–43 µmol min−1 mg−1) in pteropods than in copepods (4–14 µmol min−1 mg−1), suggesting much more ROS in pteropods to be detoxified by CAT. In contrast, CAT activity increased in copepods when pH decreased, whereas this was not the case in pteropods. This suggests that copepods have the capacity to respond to changes in carbonate chemistry, encountered for example during vertical migration, by increasing their antioxidant capacity49, whereas pteropods show no indication of further increasing their response to additional stress, suggesting combined thermal and acidification stress may push their antioxidative activity to the limits. While SOD was the only biomarker that was not significantly different between copepods and pteropods, SOD activity increased towards the south in both species, again pointing to more severe conditions requiring a response of the antioxidant defence system.
Pinheiro & Oliveira50 showed that crustacean CAT and SOD that are in the first line of antioxidant defence against H2O2 and superoxide, respectively, respond sensitively to changes in environmental conditions, such as thermal stress. Similarly to CAT, glutathione peroxidase (GP) converts H2O2 into oxygen and water. Vuori et al.30 showed that GP in Baltic Limnocalanus copepods (0.01–0.29 µmol min−1 mg−1) was inversely related to lipid peroxidation, which, in turn, suggests the presence of lingering ROS. GP activities in Calanus in the CCS were 0.003–0.01 µmol min−1 mg−1, not revealing any acute stress in copepods. In the PCA of our data, many of the antioxidant biomarkers (Table 3, Fig. 5) were strongly associated with each other, but not negatively associated with LPX, as one would expect cf.30. The PCA emphasises the differences in the stress responses of copepods and pteropods with respect to various environmental stressors. LPX increased at higher temperature and decreased at higher salinity in copepods, whereas in pteropods, LPX showed a stronger relationship with higher TA and salinity. In copepods the GP activity and GSH:GSSG ratio were higher in high O2 and low pCO2, whereas for pteropods there does not seem to be such a good relationship. Temperature seems to affect glutathione recycling in copepods, while in pteropods the increase in ORAC and CAT indicates that other antioxidant routes are activated instead of the glutathione system.
Since all pteropods were collected from deeply towed nets whereas some copepods were collected from relatively short near-surface tows, differences due to the effects of sampling stress also cannot be ruled out. Nets were pulled slowly, but deep tows were pulled for fairly long time periods, and this may have caused increases in enzymes that handle ROS directly and that have a fairly rapid turnover. These procedures may have caused higher variability in the results.
In the present study, the ORAC increased only in pteropods with elevated temperature. Several papers suggest that both copepods and pteropods are sensitive to ocean warming37,47. For example, in copepods, egg production and hatching can decrease with increasing temperature25,51, whereas in pteropods (Limacina helicina, L. retroversa), warming can amplify the effects of pCO2 on shell degradation and increase respiration and mortality33.
ETS reflects maximum potential respiratory rates, and was measured in Calanus copepods at five different stations in the present work. The measured rates of 0.06–1.23 µL O2 h−1 ind.−1 compare well with rates of 0.14–4.46 µL O2 h−1 ind.−1 reported for Calanoides in the northern Benguela upwelling system52. ETS did not show a relationship with in situ temperature, which was opposite to what we had expected53. This result could be due to the fact that the in situ temperature we considered was integrated over the upper 100 m of the water column, but the time the copepods spend over those depths is not distributed evenly as they spend the day at depth and are at the surface feeding during the relatively short nights.
Another result of depth integrating temperature is that the range among stations was only 1 °C, which is much less than they experience during DVM. Many other factors can also influence ETS, such as food availability. Further, differences in body mass, depth of occurrence, and general animal activity could explain up to 90% of the variance in respiratory rates52. Calanus is a fast-moving copepod and its fairly high ETS level could partly be explained by general activity.
To conclude, copepods and pteropods responded differently to environmental conditions, such as warming, hypoxia, and low pH. Copepods showed responses in measured biomarkers indicative of stress in relation to temperature, pCO2, pH, and O2, by increasing their antioxidant defence enzymes that scavenge harmful reactive oxygen species. Pteropods also responded to temperature, Ωarag, and pH and were severely affected by oxidative stress that can lead to cell damage and death. Pörtner & Farrell54 demonstrated that synergistic stressors like ocean acidification and hypoxia narrow thermal windows according to species-specific sensitivities, i.e., climate change will differentially favour species with wide thermal windows, short generation times, and a range of genotypes among its populations. Our results suggest that copepods are more tolerant than pteropods to current conditions, which can be related to their vertical migration behaviour11. Copepods also have efficient glutathione metabolism, allowing fast responses to changing ROS conditions30,31. Our data suggest that pteropods are sensitive to changes to temperature, Ωarag, pH, and oxygen. Further studies are needed to investigate biomarkers of these two groups that both form major prey for commercial fish species.
Methods
Sampling
This study took place in the northern California Current System (NCCS) during the fifth NOAA West Coast Ocean Acidification cruise (WCOA2016, https://www.nodc.noaa.gov/oceanacidification/data/0169412.xml), which was conducted throughout the entire CCS in May–June 2016. The samples were collected on-board the NOAA Ship Ronald H. Brown, from which a total of 19 stations were sampled from 39.5 to 53°N, 124 to 131°W (Fig. 1, Supplementary Table 1). Copepod Calanus pacificus and the pteropod Limacina helicina were targeted. Organisms were collected mainly using a Bongo net (⌀ 60 cm, 333 µm mesh) with a non-filtering cod end, towed obliquely over the upper 100 m at a speed of 2 knots, usually for 25–40 min. Additional copepod samples were collected opportunistically from surface tows using a 0.5 × 1.0 m, 333 µm mesh neuston net towed at the surface (0–2 m) for 15 min. The two nets were used to maximise the catch. After each net tow, the animals were transferred to 20L coolers filled with seawater from the subsurface, and stored at ambient temperature. Other organisms (i.e., fish or crab larvae, and jellyfish) were removed from the cooler to avoid stress to target animals. We sorted both taxa on ice under binocular microscopes, each individual picked with a pipette to a petri dish and transferred with tweezers to a 2 mL plastic Eppendorf tube, then flash-frozen in liquid nitrogen. Limacina helicina and Calanus pacificus (Number of samples = 1–7 per taxonomic group) were picked per station depending on availability. Each copepod sample consisted of ~15 adult females (females selected as they reproduce), and each pteropod sample consisted of ~20 individuals. In total, 60 samples were collected (35 copepod, 25 pteropod samples), and they were transported by air on dry ice to Finland, and stored in −80 °C until biomarker analysis was conducted.
Chemical measurements
As part of the WCOA2016 cruise, which included 17 cross-shelf transects, we obtained measurements of conductivity-temperature-depth (CTD) and oxygen from a Sea-Bird SBE 911 plus CTD system. At each station, water samples were collected in modified Niskin-type bottles, poisoned with HgCl2 and analysed on-board the ship for dissolved inorganic carbon (DIC), total alkalinity (TA), pH, chlorophyll a (chla), and oxygen using the methods described in Alin et al.55). The ship-based DIC and TA data are both precise and accurate to within 2 µmol kg−1.
Biomarkers
In the present study, we used lipid peroxidation LPX and total GSH (glutathione) concentrations; Catalase CAT, Superoxide dismutase SOD, Glutathione S-transferase GST, Glutathione peroxidase GP, Glutathione reductase GR, ETS activities, Oxygen Radical Absorbance Capacity ORAC; and reduced to oxidised glutathione GSH:GSSG ratio (Table 1), as biomarkers of stress responses in copepods and pteropods collected along the NCCS. To prevent oxidative stress and damage to biomolecules, such as LPX, organisms use antioxidants to counteract ROS (reactive oxygen species). SOD catalyses the dismutation of superoxide O•−2 to oxygen and H2O2, which is further catalysed by CAT to ground state O2 and water by GP. GSH is one of the most important and active non- enzymatic antioxidant defence in biological systems and it acts as cofactor for GP, or react with ROS independently. In the reaction with GP, GSH is oxidised, forming GSSG, which can be reduced by GR. The ratio between the reduced and oxidised forms of glutathione has been considered an important indicator of the redox status of cells. GSH is also part of xenobiotic metabolism, by acting as a conjugant in a reaction catalysed by GST enzymes. Some GSTs can catalyse the reaction of organic peroxides with GSH, thus preventing LPX56.
Zooplankton samples were homogenised on ice in 150 µL (Calanus pacificus) or 100 µL (Limacina helicina) of 0.1 M K2HPO4 + 0.15 M KCl buffer (pH 7.4) using a Tissue Lyser II bead mill (Qiagen). 25 µL of raw homogenate was directly frozen in liquid nitrogen and stored at −80 °C for lipid peroxide determination (LPX). Then, the homogenate was centrifuged at 10,000 g for 15 min at 4 °C and the resulting supernatant was divided into aliquots for GST, GR, GP, CAT and SOD enzyme activity determination, ORAC assay and for glutathione sample preparation (GSH, GGSG). The glutathione sample was deproteinised by adding 5% sulfosalicylic acid (SSA). The sample was incubated on ice for 10 min and centrifuged for 10 min at 10,000 g at 4 °C. The supernatant was divided into two different tubes for reduced (GSH) and oxidised glutathione (GSSG) and 33 mM M2VP (1-methyl-2-vinylpyridinium trifluoromethanesulfonate, Sigma Chemicals) in 0.1 M HCl that is a scavenger of GSH, was added to the GSSG sample. The sample homogenate aliquots and glutathione samples were frozen in liquid nitrogen and stored at −80 °C until further analysis.
GSH and GSSG were analysed with Glutathione 384-well plate Fluorescent Detection Kit (Arbor Assays) and intracellular soluble antioxidant capacity with OxiSelectTM Oxygen Radical Antioxidant Capacity (ORAC) Activity Assay (Cell Biolabs), following the manufacturers’ instructions, except for adjusting the reaction volumes for 384-well plate when needed. GST, GR, CAT, SOD, and LPX were determined as described in Vuori & Kanerva57,58,59,60,61. GP was only measured for copepods according to the protocol of Vuori & Kanerva62.
The enzyme activities, lipid hydroperoxides, and total GSH were normalised to the sample protein content, which was determined with PierceTM BCA Protein Assay (Thermo Scientific) with bovine serum albumin (Sigma) as the standard. The ORAC Assay is a tool for measuring the general antioxidant capacity of samples. Antioxidant systems of the sample work to block the peroxyl radical oxidation (caused by addition of free radical initiator) of the fluorescent probe until antioxidant activity in the sample is depleted. All samples, standards and blanks were analysed in triplicate. For all assays in this study, the mean coefficient of variation (CV%) of the technical replicates ranged between 2.77 and 7.30% (copepods) and between 3.43 and 5.33% (pteropods).
For Respiratory Electron Transport System (ETS), adult female Calanus copepods were identified to genus under the microscope and 7–8 individuals per sample were flash frozen on liquid nitrogen and stored at −70 °C. The ETS activity was measured according to Owens & King63, modified by Gómez et al.64, and adapted for a 96-well plate. Briefly, samples were homogenised in a Teflon glass grinder at 2 °C for 1.5 min in 20 mM Tris Buffer (pH 7.8), then centrifuged at 1503 × g for 10 min at 2 °C. ETS activity was measured via INT (2-(p-iodophenyl)-3-(p-nitrophenyl)-5-phenyl tetrazolium chloride) reduction to formazan by the change in absorbance, measured kinetically at 490 nm with spectrophotometer (SpectraMax M2, Molecular Devices). For each assay, 30 µL of the homogenate was added to 90 µL substrate solution (1.7 mM NADH and 0.25 mM NADPH dissolved in phosphate buffer), and the reaction was initiated by adding 30 µL INT (0.2%, pH 8.5). Blank measurements were taken using phosphate buffer (0.1 M phosphate buffer pH 8.5, 0.2% v/v Triton x-100, 0.15% w/v polyvinylpyrrolidone, 75 µM MgSO4) without added substrates. Assays and blanks were measured in triplicate at 24 °C and corrected to in situ temperatures (depth integrated from 0–100 m) using the Arrhenius equation with an activation energy of 15 kcal/mol65; potential respiration was calculated according to Packard & Christensen66.
Statistical analyses
We used linear mixed models (LMMs), based on restricted maximum likelihood estimation, for comparing biomarker levels between species and for analysing effects of environmental conditions. Biomarker levels were treated as the response variable, species as the fixed-effect (categorical) factor and environmental conditions (integrated over the upper 100 m) as fixed-effect co-variates, and CTD (i.e., conductivity-temperature-depth) station was a random effect. Multicollinearity was evidenced by correlations between environmental variables exceeding 0.7; such variables were not included in the same model. Selection between collinear variables was based on comparing the Akaike’s information criterion (AIC) of single-variable candidate models explaining biomarker levels, with lower values of AIC being preferred67. These candidate models were fitted using maximum likelihood to allow AIC-based comparison of models with different fixed effects. The variable with the lowest AIC was therefore selected for the final model (aragonite was not considered, as it is important only for one of the study species, L. helicina). To facilitate models convergence, only two co-variates were included per model. In cases of unfavourably low ratios of observations to variables, indicating overfitting, we omitted two-way interactions from the model. For LMMs, the package ‘lmerTest’ was used in the free software R68, version 3.4.3 (www.R-project.org). pH was transformed to [H+] prior to analyses, as pH is a logarithmic unit.
In order to visualise the associations among measured antioxidant defence and oxidative stress variables and environmental data, a three-table ordination method, the species traits analysis (=RLQ analysis)69, was used. This involves the construction of three data tables: a table with variables describing the environmental conditions at the stations (R), a table containing traits (e.g., biomarkers) of the species (Q), and a table with presence-absence values for species at a series of stations (L). First, a principal component analysis (PCA) was performed for biomarkers and the environmental data, then a correspondence analysis on the sample groups (i.e., CTD stations). These three analyses were passed to the RLQ function of ‘ade4’ package in R, version 3.4.3 (www.R-project.org).
References
Feely, R. A. et al. Chemical and biological impacts of ocean acidification along the west coast of North America. Estuar. Coast. Shelf Sci. 183, 260–270 (2016).
Gruber, N. et al. Rapid progression of ocean acidification in the California Current System. Science 337(6091), 220–223 (2012).
Bednaršek, N. et al. Exposure history determines pteropod vulnerability to ocean acidification along the US West Coast. Sci. Rep. 7, 4526 (2017).
Feely, R. A., Sabine, C. L., Hernandez-Ayon, J. M., Ianson, D. & Hales, B. Evidence for upwelling of corrosive” acidified” water onto the continental shelf. Science 320(5882), 1490–1492 (2008).
Feely, R. A. et al. The combined effects of acidification and hypoxia on pH and aragonite saturation in the coastal waters of the California current ecosystem and the northern Gulf of Mexico. Cont. Shelf Res. 152, 50–60 (2018).
Di Lorenzo, E. & Mantua, N. Multi-year persistence of the 2014/15 North Pacific marine heatwave. Nat. Clim. Change 6(11), 1042–1047 (2016).
Gentemann, C. L., Fewings, M. R. & García‐Reyes, M. Satellite sea surface temperatures along the West Coast of the United States during the 2014–2016 northeast Pacific marine heat wave. Geophys. Res. Lett. 44(1), 312–319 (2017).
Amaya, D. J., Bond, N. E., Miller, A. J. & DeFlorio, M. J. The evolution and known atmospheric forcing mechanisms behind the 2013-2015 North Pacific warm anomalies. US Clivar Var. 14(2), 1–6 (2016).
Mackas, D. L. & Galbraith, M. D. Pteropod time-series from the NE Pacific. ICES J. Mar. Sci. 69(3), 448–459 (2011).
Sturdevant, M. V., Orsi, J. A. & Fergusson, E. A. Diets and trophic linkages of epipelagic fish predators in coastal Southeast Alaska during a period of warm and cold climate years, 1997–2011. Mar. Coast. Fish. 4(1), 526–545 (2012).
Lewis, C. N., Brown, K. A., Edwards, L. A., Cooper, G. & Findlay, H. S. Sensitivity to ocean acidification parallels natural pCO2 gradients experienced by Arctic copepods under winter sea ice. Proc. Natl. Acad. Sci. 110(51), E4960–E4967 (2013).
Johnson, C. L. & Checkley, D. M. Jr. Vertical distribution of diapausing Calanus pacificus (Copepoda) and implications for transport in the California undercurrent. Prog. Oceanogr. 62(1), 1–13 (2004).
Wærvågen, S. B., A Rukke, N. & Hessen, D. O. Calcium content of crustacean zooplankton and its potential role in species distribution. Freshwat. Biol. 47, 1866–1878 (2002).
Bradford, J. M. & Jillett, J. B. A revision of generic definitions in the Calanidae (Copepoda, Calanoida). Crustaceana 27, 5–16 (1974).
Peterson, W. T. & Keister, J. E. Interannual variability in copepod community composition at a coastal station in the northern California Current: a multivariate approach. Deep-Sea Res. Part II–Top. Stud. Oceanogr. 50, 2499–2517 (2003).
Mullin, M. M. Interannual and interdecadal variation in California Current zooplankton: Calanus in the late 1950s and early 1990s. Glob. Change Biol. 4(1), 115–119 (1998).
Costantini, D. Redox physiology in animal function: The struggle of living in an oxidant environment. Curr. Zool. 56(6), 687–702 (2010).
Monaghan, P., Metcalfe, N. B. & Torres, R. Oxidative stress as a mediator of life history trade‐offs: mechanisms, measurements and interpretation. Ecol. Lett. 12(1), 75–92 (2009).
Dowling, D. K. & Simmons, L. W. Reactive oxygen species as universal constraints in life-history evolution. Proc. R. Soc. Lond. Ser. B 276(1663), 1737–1745 (2009).
Philipp, E. E. R., Lipinski, S., Rast, J. & Rosenstiel, P. Immune defense of marine invertebrates: the role of reactive oxygen and nitrogen species, in Oxidative stress in aquatic ecosystems (eds. Abele, D., Vázquez- Medina, J. P. & Zenteno-Savín, T.) (Wiley-Blackwell, 2012).
Matozzo, V., Chinellato, A., Munari, M., Bressan, M. & Marin, M. G. Can the combination of decreased pH and increased temperature values induce oxidative stress in the clam Chamelea gallina and the mussel Mytilus galloprovincialis? Mar. Pollut. Bull. 72(1), 34–40 (2013).
Vidal, M. L., Bassères, A. & Narbonne, J. F. Influence of temperature, pH, oxygenation, water-type and substrate on biomarker responses in the freshwater clam Corbicula fluminea (Müller). Comp. Biochem. Physiol. C–Toxicol. Pharmacol. 132(1), 93–104 (2002).
Lushchak, V. I. Environmentally induced oxidative stress in aquatic animals. Aquat. Toxicol. 101(1), 13–30 (2011).
Martínez, M., Rodríguez-Graña, L., Santos, L., Denicola, A. & Calliari, D. Oxidative damage and vital rates in the copepod Acartia tonsa in subtropical estuaries with contrasting anthropogenic impact. J. Exp. Mar. Biol. Ecol. 487, 79–85 (2017).
Vehmaa, A. et al. Projected marine climate change: effects on copepod oxidative status and reproduction. Ecol. Evol. 3(13), 4548–4557 (2013).
Peck, V. L., Tarling, G. A., Manno, C., Harper, E. M. & Tynan, E. Outer organic layer and internal repair mechanism protects pteropod Limacina helicina from ocean acidification. Deep-Sea Res. Part II–Top. Stud. Oceanogr. 127, 41–52 (2016).
Bocchetti, R. et al. Seasonal variations of exposure biomarkers, oxidative stress responses and cell damage in the clams, Tapes philippinarum, and mussels, Mytilus galloprovincialis, from Adriatic sea. Mar. Environ. Res. 66, 24–26 (2008).
Tomanek, L., Zuzow, M. J., Ivanina, A. V., Beniash, E. & Sokolova, I. M. Proteomic response to elevated pCO2 level in eastern oysters, Crassostrea virginica: evidence for oxidative stress. J. Exp. Biol. 214, 1836–1844 (2011).
Matoo, O. B., Ivanina, A. V., Ullstad, C., Beniash, E. & Sokolova, I. M. Interactive effects of elevated temperature and CO2 levels on metabolism and oxidative stress in two common marine bivalves (Crassostrea virginica and Mercenaria mercenaria). Comp. Biochem. Physiol. A–Mol. Integr. Physiol. 164(4), 545–553 (2013).
Vuori, K. A. et al. Oxidative stress biomarkers in the copepod Limnocalanus macrurus from the northern Baltic Sea: effects of hydrographic factors and chemical contamination. Mar. Ecol. Prog. Ser. 538, 131–144 (2015).
Glippa, O., Engström-Öst, J., Kanerva, M., Rein, A. & Vuori, K. Oxidative stress and antioxidant defense responses in Acartia copepods in relation to environmental factors. PLoS One 13(4), e0195981 (2018).
Comeau, S., Gattuso, J. P., Nisumaa, A. M. & Orr, J. Impact of aragonite saturation state changes on migratory pteropods. Proc. R. Soc. Lond. Ser. B 279(1729), 732–738 (2012).
Bednaršek, N., Johnson, J. & Feely, R. A. Comment on Peck et al: Vulnerability of pteropod (Limacina helicina) to ocean acidification: shell dissolution occurs despite an intact organic layer. Deep-Sea Res. Part II-Top. Stud. Oceanogr. 127, 53–56 (2016).
Peck, V. L., Oakes, R. L., Harper, E. M., Manno, C. & Tarling, G. A. Pteropods counter mechanical damage and dissolution through extensive shell repair. Nat. Commun. 9(1), 264 (2018).
Bednaršek, N., Feely, R. A., Beck, M., Glippa, O., Kanerva, M. & Engström-Öst, J. El Niño-related thermal stress coupled with ocean acidification negatively impacts cellular to population-level responses in pteropods along the California Current System with implications for increased bioenergetic costs. Front. Mar. Sci. 5, 486 (2018).
Bednaršek, N., Harvey, C. J., Kaplan, I. C., Feely, R. A. & Možina, J. Pteropods on the edge: Cumulative effects of ocean acidification, warming, and deoxygenation. Prog. Oceanogr. 145, 1–24 (2016).
Lischka, S. & Riebesell, U. Synergistic effects of ocean acidification and warming on overwintering pteropods in the Arctic. Glob. Change Biol. 18(12), 3517–3528 (2012).
Zitka, O. et al. Redox status expressed as GSH: GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncol. Lett. 4(6), 1247–1253 (2012).
Peterson, W. T., Miller, C. B. & Hutchinson, A. Zonation and maintenance of copepod populations in the Oregon upwelling zone. Deep-Sea Res. Part A–Oceanogr. Res. Pap. 26(5), 467–494 (1979).
Frost, B. W. Variability and possible adaptative significance of diel vertical migration in Calanus pacificus, a planktonic marine copepod. Bull. Mar. Sci. 43, 675–694 (1988).
Palomares-García, R. J., Gómez-Gutiérrez, J. & Robinson, C. J. Winter and summer vertical distribution of epipelagic copepods in the Gulf of California. J. Plankton Res. 35(5), 1009–1026 (2013).
Maas, A. E., Lawson, G. L. & Wang, Z. A. The metabolic response of thecosome pteropods from the North Atlantic and North Pacific Oceans to high CO2 and low O2. Biogeosciences 13(22), 6191 (2016).
Anger, K. Contributions of larval biology to crustacean research: a review. Invertebr. Reprod. Dev. 49(3), 175–205 (2006).
Roncalli, V., Jungbluth, M. J. & Lenz, P. H. Glutathione S-Transferase regulation in Calanus finmarchicus feeding on the toxic dinoflagellate Alexandrium fundyense. PLoS One 11(7), e0159563 (2016).
Cailleaud, K., Maillet, G., Budzinski, H., Souissi, S. & Forget-Leray, J. Effects of salinity and temperature on the expression of enzymatic biomarkers in Eurytemora affinis (Calanoida, Copepoda). Comp. Biochem. Physiol. A–Mol. Integr. Physiol. 147, 841–849 (2007).
Kurihara, H. & Ishimatsu, A. Effects of high CO2 seawater on the copepod (Acartia tsuensis) through all life stages and subsequent generations. Mar. Pollut. Bull. 56(6), 1086–1090 (2008).
Richardson, A. J. In hot water: zooplankton and climate change. ICES J. Mar. Sci. 65, 279–295 (2008).
Havenhand, J. N. How will ocean acidification affect Baltic Sea ecosystems? An assessment of plausible impacts on key functional groups. Ambio 41(6), 637–644 (2012).
Vehmaa, A. et al. Ocean acidification challenges copepod phenotypic plasticity. Biogeosciences 13(22), 6171 (2016).
Pinheiro, L. C. & Oliveira, G. T. Oxidative status profile in different tissues of Parastacus brasiliensis promatensis (Crustacea, Decapoda, Parastacidae) over a seasonal cycle. J. Exp. Zool. A–Ecol. Integr. Physiol. 325(5), 318–328 (2016).
Vehmaa, A., Brutemark, A. & Engström-Öst, J. Maternal effects may act as an adaptation mechanism for copepods facing pH and temperature changes. PLoS One 7(10), e48538 (2012).
Bode, M., Schukat, A., Hagen, W. & Auel, H. Predicting metabolic rates of calanoid copepods. J. Exp. Mar. Biol. Ecol. 444, 1–7 (2013).
Simčič, T. & Brancelj, A. Respiratory electron transport system (ETS) activity as an estimator of the thermal tolerance of two Daphnia hybrids. J. Plankton Res. 26, 525–534 (2004).
Pörtner, H. O. & Farrell, A. P. Physiology and climate change. Science 322(5902), 690–692 (2008).
Alin, S. R. et al. Dissolved inorganic carbon, total alkalinity, pH on total scale, and other variables collected from profile and discrete sample observations using CTD, Niskin bottle, and other instruments from NOAA Ship Ronald H. Brown in the U.S. West Coast California Current System from 2016-05-08 to 2016-06-06 (NCEI Accession 0169412). Version 1.1. NOAA National Centers for Environmental Information. Dataset [access date: 8 March 2019], https://doi.org/10.7289/V5V40SHG (2017).
Halliwell, B. & Gutteridge, J. M. C. Free radicals in biology and medicine (Oxford University Press, 2007).
Vuori, K. & Kanerva, M. Glutathione-S-Transferase (GST) activity assay for zooplankton samples, https://doi.org/10.17504/protocols.io.mwrc7d6 (2018a).
Vuori, K. & Kanerva, M. Glutathione reductase (GR) activity assay for zooplankton samples, https://doi.org/10.17504/protocols.io.mjfc4jn (2018b).
Vuori, K. & Kanerva, M. Catalase (CAT) activity assay for zooplankton samples, https://doi.org/10.17504/protocols.io.mwsc7ee (2018c).
Vuori, K. & Kanerva, M. Superoxide dismutase (SOD) activity assay for zooplankton samples, https://doi.org/10.17504/protocols.io.mv7c69n (2018d).
Vuori, K & Kanerva, M. Lipid peroxidation (LPX) assay for zooplankton homogenates, https://doi.org/10.17504/protocols.io.m4vc8w6 (2018e).
Vuori, K. & Kanerva, M. Glutathione peroxidase (GP) activity assay for zooplankton samples, https://doi.org/10.17504/protocols.io.mv8c69w (2018f).
Owens, T. G. & King, F. D. The measurement of respiratory electron-transport system activity in marine zooplankton. Mar. Biol. 30, 27–36 (1975).
Gómez, M., Torres, S. & Hernández-León, S. Modification of the electron transport system (ETS) method for routine measurements of respiratory rates of zooplankton. S. Afr. J. Mar. Sci. 16, 15–20 (1996).
Packard, T. T., Devol, A. H. & King, F. D. The effect of temperature on the respiratory electron transport system in marine plankton. Deep-Sea Res. 22, 237–249 (1975).
Packard, T. T. & Christensen, J. P. Respiration and vertical carbon flux in the Gulf of Maine water column. J. Mar. Res. 62, 93–115 (2004).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference - A Practical Information-Teoretic Approach. (Springer, 2002).
Dray, S. & Dufour, A. B. The ade4 package: implementing the duality diagram for ecologists. J. Stat. Softw. 22, 1–20 (2007).
Dolédec, S., Chessel, D., ter Braak, C. J. F. & Champely, S. Matching species traits to environmental variables: a new three-table ordination method. Environ. Ecol. Stat. 3, 143–166 (1996).
Acknowledgements
This project took place in the framework of the NOAA Ocean Acidification Program and WCOA16 cruise. We thank officers, crew and scientists on-board the NOAA Ship Ronald H. Brown, and Prof. Mikko Nikinmaa (University of Turku) for his hospitality during biomarker analyses, and collaboration with the lab personnel. We thank Markus Öst for statistical advice. The funding from Academy of Finland (nr. 276947) and Waldemar von Frenckell Foundation is greatly appreciated. This is PMEL contribution number 4807.
Author information
Authors and Affiliations
Contributions
J.E. and O.G. collected the samples, analysed the data, and conceived the manuscript. R.F. and S.A. organised and led the research cruise and provided the environmental data, M.K. and K.V. did the biomarker analyses, J.K. provided data and valuable advice, B.C. analysed the data, A.M. did the ETS analysis, N.B. supervised the pteropod study. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Engström-Öst, J., Glippa, O., Feely, R.A. et al. Eco-physiological responses of copepods and pteropods to ocean warming and acidification. Sci Rep 9, 4748 (2019). https://doi.org/10.1038/s41598-019-41213-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41213-1
This article is cited by
-
Compound marine heatwaves and ocean acidity extremes
Nature Communications (2022)
-
The effects of low pH on the taste and amino acid composition of tiger shrimp
Scientific Reports (2021)
-
Biogeochemical extremes and compound events in the ocean
Nature (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.