Abstract
Impairment of cognitive functions in advanced age leads to a reduced quality of life and impaired ability to perform everyday tasks. The positive impact of physical exercise on the quality of life and well-being, also at a later age, is well established. However, the effect of endurance exercises, including long distance running and cycling, on cognitive function and mental health within the elderly population has still to be elucidated. To this end, elderly active marathoners (N = 50) aged over 60 years and non-athlete controls (N = 49) were followed for four years. Cognitive function was assessed using the CERAD test battery. In addition, the Short Form Health Survey (SF-36) was applied to assess self-reported physical, mental, and emotional health. Except for age, sex and education-corrected z-values of the test “Word list recall”, with marathon runners showing a decline compared to an improvement in controls (p < 0.05), there was no statistically significant difference in time trend between groups. In contrast, concerning self-reported health, scores in all eight domains of the SF-36 remained stable over time and, in nearly all of them, marathon runners showed higher self-reported health than controls. The results indicated that extensive endurance exercise is associated with improved subjective health but does not lead to better scores in cognitive performance tests in elderly persons.
Similar content being viewed by others
Introduction
There is general consensus that moderate physical exercise is a healthy undertaking at all ages, including the elderly. As the population in the industrialized world continues to age, and cognitive functions slowly decline at an advanced age, this raises the question of whether physical exercise might combat cognition decay. Although light exercises, including yoga and aerobics, have been shown to improve performance in cognitive tests over the short term1,2,3, the question of whether endurance sports in the elderly lead to similar improvements in the long term has not yet been addressed. Hence, in light of the importance of guiding the elderly population towards a lifestyle that would increase the likelihood of maintaining cognitive performance through advanced age, there is a need to assess whether more intensive exercise would represent a way to conserve cognitive function.
The aforementioned demographic change observed in industrialized countries is mainly due to improvements in the quality of healthcare and general living standards. According to predictions by the World Health Organization, the population aged over 60 years will almost double from 12% in 2015 to 22% in 20504. A similar demographic change is anticipated in Austria: the proportion of the population aged 65 years and older has been estimated to increase from 18% in 2013 to 24% in 2030. In 2013, the average life expectancy in Austria for males was 78.45 years and 83.56 years for females and was expected to increase by 2030 to 80.45 years for males and 85.53 years for females5.
Accompanied senescence of body tissues and functions, known to be associated with a number of chronic age-related diseases, including metabolic syndromes, osteoporosis, cardiovascular diseases, and cancer also has an impact on cognitive function and may result in severe cognitive impairment and dementia6. Chronic diseases impose huge burdens for individuals but also on the social welfare system and cause increased healthcare expenditures7,8. In 2016, nearly 44 million people worldwide were living with dementia9. Associated healthcare costs were US$ 604 billion in 2010, increased significantly by 35% to US$ 818 billion in 2015 and are expected to keep rising in the future10. Therefore, WHO declared in 2012 that dementia is a public health priority11 and pointed out that primary prevention would be the most effective and affordable way to reduce the onset of dementia12.
There is a continuing debate about how physical and mental health can be maintained at an advanced age, and which interventions might be cost-effective to prevent or delay age-related physical and mental degradation. It is well known that low to moderate intensity physical activity is beneficial to overall health and especially regarding cognitive function13,14,15. In this regard, there is growing evidence that physical exercising is neuroprotective and enhances immune competence16,17. Besides that, physical activity ameliorates depressive symptoms, even if genetic susceptibility is present18,19, and improves subjective well-being and quality of life20,21 Accordingly, general recommendations on physical activity for elderly above the age of 65 years propose at least 150 min/week of moderate-intensity aerobic exercise, if a substantial health benefit is desired22. The underlying mechanisms are not yet fully understood, although different modes of action were proposed. These include the induction of neuro- and synaptogenesis, the release of neurotrophins23 and also the modulation of gut microbiota24.
Likewise, there is very little knowledge about how high intensity endurance training might affect cognitive functions and well-being in the elderly. This form of training includes high intensity, regular, and long-term physical activity such as long distance running and cycling. So far, some authors reported a positive effect of vigorous activity on mental health21,25. Here, we aimed to increase the spare existing knowledge on the interplay between vigorous physical activity, cognitive functions and mood by studying a cohort of elderly marathon runners and long-distance cyclists, as well as a control group with low physical activity during almost four years in a prospective, controlled observational trial. Both groups were assessed regarding changes in cognitive functions as measured by the German Plus-version of neuropsychological test battery designed by the Consortium to Establish a Registry for Alzheimer’s disease (CERAD). This test battery is a reliable measure to identify patients with any type of dementia and covers a broad range of cognitive domains, including language, memory, and other executive functions, which are amongst the earliest functions impaired in Alzheimer’s disease)26,27. Moreover, the battery provides standard values corrected for age and years of education, enabling the calculation of individual deviations from the normal range (“z-values”)26.
Results
Cohort description
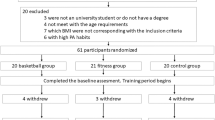
Overall, 99 participants (50 athletes, 49 controls) were included in the present analysis comprising 86% of the initially enrolled individuals (Fig. 1). The mean age at baseline was 66 ± 4 years for the control group and 66 ± 5 years for the athletes. No statistically significant differences were observed for gender and years of education (original matching variables) and family history of Alzheimer’s disease, current medication, and alcohol consumption. Body mass index (BMI) differed significantly (pBH <0.0001) between the two groups (controls 27.6 ± 4.9 and athletes 23.7 ± 2.6 kg/m²). A significantly lower number of athletes reported themselves as smokers, or to suffer from a relevant disease (all pBH <0.05 Table 1).
Relative Physical performance
General physical performance of the participants is given in Table 2. Athletes performed almost 50% better than individuals of the control group, both at baseline and during the follow-up exam in 2012 (p < 0.001, Table 2). Also, the decline in absolute physical performance was less expressed in athletes than in controls, although this observation lost statistical significance after correction for multiple hypothesis testing.
Cognitive performance is not better in athletes than in non-athletes
When analyzing z-scores of the subtests of the CERAD Plus neuropsychological test battery using general linear models, it seemed that both athletes and controls improved in the S-word test assessing phonemic verbal fluency (p = 0.011). In contrast, z-scores in constructional praxis declined in both groups between baseline and follow-up measurements (p = 0.045), whereby athletes presented with a higher z-score than controls (p = 0.033). However, all three results did not maintain statistical significance after correction according to Benjamini and Hochberg. The only significant difference in temporal developments of z-scores between athletes and controls were registered for the subtest “Word list recall” assessing episodic memory. In this subscale, values of controls improved, while z-scores of athletes slightly deteriorated (athletes: z = −0.31 ± 1.24 vs. −0.52 ± 1.30, controls: −0.66 ± 1.24 vs. −0.20 ± 1.29, pBH <0.05). Detailed numbers are given in Table 3.
Hence, it appeared that athletes were not better than non-athletes in neuropsychological surrogates of all cognitive domains. On the contrary, controls presented with a significant improvement of a memory-related subtest, while athletes declined. In a next step, we aimed to assess whether both groups differed in subjectively perceived health and wellbeing.
Self-reported health is higher in endurance athletes compared to non-athletes
To assess whether endurance sports had any impact on subjective well-being, athletes and controls were asked to self-evaluate their health by the SF-36 at both time points. In general, athletes evaluated their health as better than non-athletes in the following categories: Physical functioning (pBH <0.001), bodily pain (pBH <0.05), physical role functioning (pBH <0.01), general health perceptions (pBH <0.01), vitality (pBH <0.01), social functioning (pBH <0.01), and emotional role functioning (pBH <0.05). Solely the differences in mental health perception (p = 0.016) were no longer statistically significant after p-value correction (Table 4).
Our results indicated that high intensity endurance training is associated with better subjective health perception and wellbeing.
Discussion
In this study, we investigated the effect of high intensity endurance training on cognitive and physical performance as well as well-being in healthy older people. We found no strong evidence that long-term, high intensity endurance training reduced cognitive impairment in later age. However, elderly athletes reported a significantly better quality of life.
Previous interventional and cohort studies had investigated the protective effect of physical activity on cognitive performance in the elderly. Most of the published interventional studies focused on light to moderate sports activities, including walking, gymnastics, yoga, and aerobic exercises28,29. The results of these earlier studies indicated that the dosage and intensity of the training might be important for preventing the deterioration of cognitive function. The World Health Organization recommends that being active is better than remaining sedentary. Even a small amount of movement, including daily activities and walking, can have a positive impact on cognitive functioning and preventing its impairment in later life30,31. It was also claimed that moderate- and high intensity-persistence exercise programs have an equally beneficial effect on cognitive function32,33. In contrast, Kimura K et al. found that short-term (12 weeks) training has a positive effect on health-related quality of life (HRQOL), but did not affect executive functions34.
In our study, at the four-year follow-up stage, both the athletes as well as the non-athlete controls scored lower in four out of 15 tasks of the test battery designed by the Consortium to Establish a Registry for Alzheimer’s disease (data not shown), however, there was no statistically significant decline, when z-values (corrected for sex, age and years of education) were compared instead of raw scores. Hence, the observed cognitive decline might be related to the natural aging process.
In detail, during follow-up, Word List Recall appeared improved in controls, but deteriorated slightly in athletes. In both groups, performance in the S-word test increased. A slight decline was also noted for Constructional praxis, but this was likely due to a regression to the mean effect because, in 2008, performance was at the maximum for almost all participants. However, both developments (S-words improvement and decline of Constructional praxis) did not remain statistically significant after correction for multiple hypothesis testing. The same was true for the apparent dominance of athletes in Constructional praxis, which lost statistical significance as well.
In contrast, athletes reported better physical and cognitive health and well-being in nearly all subscales of the SF-36, which is accompanied by significantly lower depressive symptoms, as reported before18,19,35. This is in-line with previous findings among all age groups and types of exercise; in law students, e.g., the degree of mental distress correlated well with their levels of exercise36. For sedentary older adults, it was reported that Yoga interventions markedly improved physical function and well-being37. As underlying mechanisms, neuromodulatory effects were proposed. In this regard, it was demonstrated that exercise might stem the reduction in hippocampal neurogenesis as it is seen in depressive patients38. In fact, exercise therapy has meanwhile become a standard treatment for mild-to-moderate depression39, especially in patients without physical co-morbidity40.
Our present results appear at first glance to be counterintuitive, as one would assume that endurance athletes would enjoy not only better health but also slower cognitive decline, which was apparently not evident here. This is indeed in line with previous observations41,42. We hypothesize that daily high intensity endurance training requires a great deal of time, which is no longer available for social contact. There is a considerable volume of literature demonstrating a relationship between cognitive function and social interaction. These studies emphasize that an active social life in older age is essential for maintaining healthy cognitive function43,44. For instance, social engagements with friends offer protection against cognitive decline in women43, and a higher social activity is associated with better cognitive function44.
An additional potential explanation for the observed reduction in athletes’ cognitive performance might be attributed to high amounts of free radicals produced during high intensity endurance training, which at a senior age might not be as effectively counteracted due to reduced anti-oxidative capacity, thereby leading to damage to the central nervous system. It has been reported previously that myeloperoxidase, produced as a result of oxidative stress, increases to threefold higher levels after running a marathon45,46.
Consequently, it is important to ensure the optimal amount of physical activity and antioxidant intake in older age, since the ability of endogenous antioxidant production during this phase of life is reduced47. The American College of Sports Medicine clearly states that an optimal dosage of sport and other physical activities is essential in older age47.
This pioneer longitudinal investigation, which studied the effect of high intensity endurance training on cognitive performance in a later age, comes with a few limitations. First, our sample size is comparably moderate, and the portion of female participants was rather low. Therefore, we could not compare variables between male and female participants. Secondly, we did not investigate daytime activities including gardening, walking, reading, and moderate sports. Therefore, the control group cannot be considered as absolutely sedentary, but as non-athletic. Furthermore, we did not aim to investigate any social effects on cognition. Thus, further studies should be performed focusing on the mutual effect of socioeconomic and physical activity on cognition. Moreover, a more detailed quantification of the control cohort’s actual physical activity level might increase the validity of future investigations.
To conclude, our study provided novel evidence that high intensity endurance training might not be supportive of maintaining cognitive function in the elderly. Future studies will be required to determine whether aged athletes engaged in team sports (rather than in individual endurance sports) will perform better in tests for cognitive functions. Nevertheless, our results suggest that elderly endurance athletes enjoy greater subjective health and well-being.
Methods
Study design
In 2008, 136 individuals aged 60 years or older were screened for participation in the Vienna Marathon Study APSOEM (Austrian Prospective Cohort Study in Cognitive Function of Elderly Marathon-runners, clinicaltrials.gov NC M01045031, which was designed as a prospective cohort study. Baseline data were collected in 200818,19,35,46, the first follow-up examinations took place in 201248,49.
The protocol, as well as related amendments, were reviewed and approved by the local ethics committee of the Medical University of Vienna (EK № 401/2005). All medical procedures followed institutional guidelines. All participants gave written informed consent before participation in the study.
Participants
At baseline, 56 athletes and 58 controls were enrolled. During the follow-up period, 15 participants dropped out due to various reasons (quit physical activity, refused to participate or died). Hence, a total of 99 participants (50 athletes, 49 controls) could be re-evaluated in 2012. The applied inclusion and exclusion criteria are described below. For control participants, which were recruited via personal contact, as well as via newspaper advertisements, the same inclusion and exclusion criteria applied, except for the necessity to have participated in a marathon and the minimum weekly amount of physical training. Instead, control participants had to specify that they did not undergo any strenuous endurance training on a regular basis. Selection of controls was carried out in a frequency-matching process, where sex, age, and years of education served as matching criteria.
Inclusion criteria
Subjects aged ≥60 in the high intensity endurance training group had to participate in one of the following competitions during the previous three years: Wachau half marathon (21.5 km), Vienna City Marathon (43 km), Carinthian marathon (180 km). Their current amount of physical activity per week must not have been less than 2 hours of intensive training.
Exclusion criteria
Exposure to neurotoxic substances (queried by occupation anamnesis), a first language other than German (since the neuropsychological test battery was available only in German), the presence of clinically manifest diseases affecting the central nervous system (e.g., ischemia, dementia), cardiovascular diseases or depression or chronic alcoholism and unwillingness to participate in the study led to exclusion.
Medical Evaluation
Participants underwent a detailed medical check-up including anamnesis, physical examinations and a physical performance test (treadmill test). Moreover, blood was drawn for assessment of hematological, biomedical, genetic, and immunological parameters. Additional blood samples were taken and stored at the MedUni Wien Biobank for subsequent analyses50.
Assessment of cognitive function
Cognitive function was assessed by the German version of a neuropsychological test battery developed by the Consortium to Establish a Registry for Alzheimer’s disease (CERAD). The CERAD Plus test battery contains 15 subtests: Verbal Fluency, Boston Naming Test, Wordlist Encoding, Wordlist Recall, Word list recognition, Constructional Practise and recall, Intrusions, Word list savings, Figure savings, Trail Making Test A, B and BA, S-words and Mini-Mental State Examination. It was mainly designed for the evaluation of executive, frontal and subcortical function51,52. All psychological tests were conducted by trained and certified psychologist.
Health-related quality of life (SF-36)
Additionally, individuals self-assessed their health-related quality of life and well-being status by the Short Form health survey SF-3653.
Treadmill test
Relative physical performance was calculated as the individual maximum workload achieved given as a percentage of the final wattage relative to tabulated sex, age and body surface-specific performance references54. The workload was raised every two minutes in steps of 25 W, starting with 25 W and move on until exhaustion. This test is done under monitored electro-cardiograph and blood pressure. The individual physical working capacity (PWC) was calculated as the maximal individual power (WATTmax) in per cent of a reference value (WATTref): PWCind = 100 × WATTmax/WATTref.
Statistical analysis
Statistical analyses were performed using SPSS 23 (IBM Corp., NY, USA) and Stata 13.0 (StataCorp TX, USA). Results are reported as means and standard deviations (SD). General linear models with repeated measurements design were applied to assess differences between baseline and follow-up (within-subject factor), between groups, or the interaction between time and group (i.e., whether there are differences in time trend between groups). Individuals’ responses to the question about the frequency of conversation with others were used as a covariate to control for possible effects of social communication in the analyses of cognitive performance. P-values were corrected for multiple testing according to Benjamini and Hochberg and given as pBH. For all statistical tests, p-values below 0.05 were considered significant.
Data Availability
Raw data will be available for interested researchers upon request from the corresponding author.
References
Eyre, H. A. et al. A randomized controlled trial of Kundalini yoga in mild cognitive impairment. Int Psychogeriatr 29, 557–567, https://doi.org/10.1017/s1041610216002155 (2017).
Karssemeijer, E. G. A. et al. Positive effects of combined cognitive and physical exercise training on cognitive function in older adults with mild cognitive impairment or dementia: A meta-analysis. Ageing Res Rev 40, 75–83, https://doi.org/10.1016/j.arr.2017.09.003 (2017).
Klimova, B., Valis, M. & Kuca, K. Cognitive decline in normal aging and its prevention: a review on non-pharmacological lifestyle strategies. Clin Interv Aging 12, 903–910, https://doi.org/10.2147/cia.s132963 (2017).
World Health Organization. World report on ageing and health, http://www.who.int/ageing/events/world-report-2015-launch/en/ (2015).
Statistics Austria. Demographisches Jahrbuch (2014).
Vaupel, J. W. Biodemography of human ageing. Nature 464, 536–542, https://doi.org/10.1038/nature08984 (2010).
World Health Organization. Global health, http://www.who.int/ageing/publications/global_health.pdf (2011).
Kankeu, H. T., Saksena, P., KeXu & Evans, D. B. The financial burden from non-communicable diseases in low- and middle-income countries: a literature review. Health Research Policy and Systems 11, 31 (2013).
GBD 2016 Dementia Collaborators. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol, https://doi.org/10.1016/s1474-4422(18)30403-4 (2018).
Prince, M. et al. The World Alzheimer Report 2015 (Alzheimer’s Disease International (ADI), 2015).
World Health Organization. Dementia is priority of public health (2012).
Wu, Y. et al. Dementia in western Europe: epidemiological evidence and implications for policy making. Lancet Neurol 15, 116–124, https://doi.org/10.1016/S1474-4422(15)00092-7 (2015).
Zheng, G., Xia, R., Zhou, W., Tao, J. & Chen, L. Aerobic exercise ameliorates cognitive function in older adults with mild cognitive impairment: a systematic review and meta-analysis of randomised controlled trials. Br J Sports Med 50, 1443–1450, https://doi.org/10.1136/bjsports-2015-095699 (2016).
Loprinzi, P. D., Blough, J., Ryu, S. & Kang, M. Experimental effects of exercise on memory function among mild cognitive impairment: systematic review and meta-analysis. Phys Sportsmed, 1–6, https://doi.org/10.1080/00913847.2018.1527647 (2018).
Northey, J. M., Cherbuin, N., Pumpa, K. L., Smee, D. J. & Rattray, B. Exercise interventions for cognitive function in adults older than 50: a systematic review with meta-analysis. Br J Sports Med 52, 154–160, https://doi.org/10.1136/bjsports-2016-096587 (2018).
Phillips, C. & Fahimi, A. Immune and Neuroprotective Effects of Physical Activity on the Brain in Depression. Front Neurosci 12, 498, https://doi.org/10.3389/fnins.2018.00498 (2018).
Valdiglesias, V. et al. Immune biomarkers in older adults: Role of physical activity. J Toxicol Environ Health A 80, 605–620, https://doi.org/10.1080/15287394.2017.1286898 (2017).
Haslacher, H. et al. Physical exercise counteracts genetic susceptibility to depression. Neuropsychobiology 71, 168–175, https://doi.org/10.1159/000381350 (2015).
Haslacher, H. et al. rs6295 [C]-Allele Protects Against Depressive Mood in Elderly Endurance Athletes. J Sport Exerc Psychol 37, 637–645, https://doi.org/10.1123/jsep.2015-0111 (2015).
Garatachea, N. et al. Exercise attenuates the major hallmarks of aging. Rejuvenation Res 18, 57–89, https://doi.org/10.1089/rej.2014.1623 (2015).
Wicker, P. & Frick, B. Intensity of physical activity and subjective well-being: an empirical analysis of the WHO recommendations. J Public Health (Oxf) 39, e19–e26, https://doi.org/10.1093/pubmed/fdw062 (2017).
Sparling, P. B., Howard, B. J., Dunstan, D. W. & Owen, N. Recommendations for physical activity in older adults. BMJ 350, h100, https://doi.org/10.1136/bmj.h100 (2015).
Hotting, K. & Roder, B. Beneficial effects of physical exercise on neuroplasticity and cognition. Neurosci Biobehav Rev 37, 2243–2257, https://doi.org/10.1016/j.neubiorev.2013.04.005 (2013).
Codella, R., Luzi, L. & Terruzzi, I. Exercise has the guts: How physical activity may positively modulate gut microbiota in chronic and immune-based diseases. Dig Liver Dis 50, 331–341, https://doi.org/10.1016/j.dld.2017.11.016 (2018).
Iso-Markku, P., Waller, K., Kujala, U. M. & Kaprio, J. Physical activity and dementia: long-term follow-up study of adult twins. Ann Med 47, 81–87, https://doi.org/10.3109/07853890.2014.994675 (2015).
Schmid, N. S., Ehrensperger, M. M., Berres, M., Beck, I. R. & Monsch, A. U. The Extension of the German CERAD Neuropsychological Assessment Battery with Tests Assessing Subcortical, Executive and Frontal Functions Improves Accuracy in Dementia Diagnosis. Dement Geriatr Cogn Dis Extra 4, 322–334, https://doi.org/10.1159/000357774 (2014).
Twamley, E. W., Ropacki, S. A. & Bondi, M. W. Neuropsychological and neuroimaging changes in preclinical Alzheimer’s disease. J Int Neuropsychol Soc 12, 707–735, https://doi.org/10.1017/s1355617706060863 (2006).
Etgen, T. et al. Physical Activity and Incident Cognitive Impairment in Elderly Persons. Arch Intern Med 170, 186–193 (2010).
Brunner, D., Abramovitch, A. & Etherton, J. A yoga program for cognitive enhancement. PLoS One 12, e0182366, https://doi.org/10.1371/journal.pone.0182366 (2017).
Barnes, D. E. et al. Cognition in older women: the importance of daytime movement. J Am Geriatr Soc 56, 1658–1664, https://doi.org/10.1111/j.1532-5415.2008.01841.x (2008).
Martinez-Velilla, N. et al. Effect of Exercise Intervention on Functional Decline in Very Elderly Patients During Acute Hospitalization: A Randomized Clinical Trial. JAMA Intern Med, https://doi.org/10.1001/jamainternmed.2018.4869 (2018).
Quintero, A. P. et al. Acute effect of three different exercise training modalities on executive function in overweight inactive men: A secondary analysis of the BrainFit study. Physiol Behav 197, 22–28, https://doi.org/10.1016/j.physbeh.2018.09.010 (2018).
Liu-Ambrose, T. & Donaldson, M. G. Exercise and cognition in older adults: is there a role for resistance training programmes? Br J Sports Med 43, 25–27, https://doi.org/10.1136/bjsm.2008.055616 (2009).
Kimura, K. et al. The Influence of Short-term Strength Training on Health-related Quality of Life and Executive Cognitive Function. Journal of Physiological Antrophology 29, 95–101, https://doi.org/10.2114/jpa2.29.95 (2010).
Winker, R. et al. Cognitive function in elderly marathon runners: cross-sectional data from the marathon trial (APSOEM). Wien Klin Wochenschr 122, 704–716, https://doi.org/10.1007/s00508-010-1485-z (2010).
Skead, N. K. & Rogers, S. L. Running to well-being: A comparative study on the impact of exercise on the physical and mental health of law and psychology students. Int J Law Psychiatry 49, 66–74, https://doi.org/10.1016/j.ijlp.2016.05.012 (2016).
Noradechanunt, C., Worsley, A. & Groeller, H. Thai Yoga improves physical function and well-being in older adults: A randomised controlled trial. J Sci Med Sport 20, 494–501, https://doi.org/10.1016/j.jsams.2016.10.007 (2017).
Yuan, T. F. et al. Neural Mechanisms of Exercise: Anti-Depression, Neurogenesis, and Serotonin Signaling. CNS Neurol Disord Drug Targets 14, 1307–1311 (2015).
Kok, R. M. & Reynolds, C. F. 3rd. Management of Depression in Older Adults: A Review. Jama 317, 2114–2122, https://doi.org/10.1001/jama.2017.5706 (2017).
Frost, R., Bauernfreund, Y. & Walters, K. Non-pharmacological interventions for depression/anxiety in older adults with physical comorbidities affecting functioning: systematic review and meta-analysis. Int Psychogeriatr, 1–16, https://doi.org/10.1017/s1041610218001564 (2018).
Sabia, S. et al. Physical activity, cognitive decline, and risk of dementia: 28 year follow-up of Whitehall II cohort study. BMJ 357, j2709, https://doi.org/10.1136/bmj.j2709 (2017).
Etnier, J. L., Nowell, P. M., Landers, D. M. & Sibleyc, B. A. A meta-regression to examine the relationship between aerobic fitness and cognitive performance. Brain Res Rev 52, 119–130, https://doi.org/10.1016/j.brainresrev.2006.01.002 (2006).
Zunzunegui, M. A., Alvarado, B. E., Del Ser, T. & Otero, A. Social networks, social integration, and social engegament determine cognitive decline in community dwelling spanish older adults. Journal of Gerentology: Social sciences 58B, S93–S100 (2003).
Krueger, K. R. et al. Social engagement and cognitive function in old age. Exp Aging Res 35, 45–60, https://doi.org/10.1080/03610730802545028 (2009).
Melanson, S. E., Green, S. M., Wood, M. J., Neilan, T. G. & Lewandrowski, E. L. Elevation of myeloperoxidase in conjunction with cardiac-specific markers after marathon running. Am J Clin Pathol 126, 888–893, https://doi.org/10.1309/1D62H6KRFTVQRJ0A (2006).
Haslacher, H. et al. Myeloperoxidase levels predict executive function. Int J Sports Med 33, 1034–1038, https://doi.org/10.1055/s-0032-1304637 (2012).
Polidori, M. C., Mecocci, P., Cherubini, A. & Senin, U. Physical Activity and Oxidative Stress During Aging. Int J Sports Med 21, 154–157 (2000).
Haslacher, H. et al. A combination of routine blood analytes predicts fitness decrement in elderly endurance athletes. PLoS One 12, e0177174, https://doi.org/10.1371/journal.pone.0177174 (2017).
Haslacher, H. et al. Low Vitamin D Levels Do Not Predict Hyperglycemia in Elderly Endurance Athletes (but in Controls). PLoS One 11, e0157695, https://doi.org/10.1371/journal.pone.0157695 (2016).
Haslacher, H. et al. Usage Data and Scientific Impact of the Prospectively Established Fluid Bioresources at the Hospital-Based MedUni Wien Biobank. Biopreserv Biobank, https://doi.org/10.1089/bio.2018.0032 (2018).
Tractenberg, R. E. et al. What the CERAD Battery Can Tell Us about Executive Function as a Higher-Order Cognitive Faculty. Curr Gerontol Geriatr Res, 510614, https://doi.org/10.1155/2010/510614 (2010).
Morris, J. C. et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part I. Clinical and neuropsychological assessment of Alzheimer’s disease. Neurology 39, 1159–1165 (1989).
Ware, J. E. Jr. & Sherbourne, C. D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30, 473–483 (1992).
Böhm, H. et al. Empfehlungen für eine standardisierte Ergometrie. Öst Ärzteztg 33, 333–344 (1978).
Acknowledgements
This work was supported by the Anniversary Fund of the Austrian National Bank (No. 14511, No. 12979). The authors thank Professor Aner Gurvitz for English language editing.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study R.W., H.H., J.L., M.K., E.P. Performed the experiments and collected the data R.W., H.H., I.L., E.P. and D.B.. Analysed the data M.K., D.B., H.H. and R.W. Wrote the paper D.B., M.K., H.H. and R.W. Critically reviewed and approved the manuscript: all authors.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Batmyagmar, D., Kundi, M., Ponocny-Seliger, E. et al. High intensity endurance training is associated with better quality of life, but not with improved cognitive functions in elderly marathon runners. Sci Rep 9, 4629 (2019). https://doi.org/10.1038/s41598-019-41010-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41010-w
This article is cited by
-
Long-term consequences of COVID-19 on mental health and the impact of a physically active lifestyle: a narrative review
Annals of General Psychiatry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.