Abstract
Preterm premature rupture of membranes (PPROM) is associated with an increased risk of serious maternal, fetal, and neonatal morbidities. We compared neonatal outcomes of women with PPROM before 34+0 weeks of gestation according to inpatient or outpatient management policy. 587 women with PPROM >48 hours, 246 (41.9%) in the group with an inpatient care policy (ICP) and 341 (58.1%) in the group with an outpatient care policy (OCP), were identified in France, from 2009 to 2012. Neonatal outcomes were compared between the two groups using logistic regression. A second analysis was performed to compare inpatient care and effective outpatient care (discharge from hospital) through propensity score matching. The outcome was a neonatal composite variable including one or more of the neonatal morbidity complications. The perinatal composite outcome was 14.6% with the ICP and 15.5% with the OCP (p = 0.76). After using the 1:1 ratio propensity score matching, effective outpatient care was not associated with a significantly higher risk of the perinatal composite outcome (OR 0.88, CI 0.35 to 2.25; p = 0.80) compared with inpatient care. Outpatient care is not associated with an increased rate of obstetric or neonatal complications and can be an alternative to hospital care for women with uncomplicated PPROM.
Similar content being viewed by others
Introduction
Preterm premature rupture of membranes (PPROM) before 34 weeks of gestation (WG) occurs in 1% of pregnancies1,2,3,4,5,6. PPROM is associated with an increased risk of serious maternal, fetal, and neonatal morbidities, especially the risks of preterm delivery, and infectious complications such as chorioamnionitis, sepsis and neonatal infection1,7,8,9,10. Expectant management in outpatient care may be appropriate in the absence of complication risk factors11 and improves women’s well-being and reduces health costs12. However, this practice is controversial because deliveries at home have been reported13. Moreover, obstetricians fear not managing fast enough serious complications such as home delivery, cord prolapse and placental abruption in an outpatient policy14.
There is currently no guideline concerning outpatient or inpatient management. In recent years, some centers have evaluated outpatient care management in PPROM and suggested that it is an acceptable option with comparable maternal and neonatal outcomes14,15,16,17. However, those retrospective studies had limitations, such as small sample size (18 to 61 patients) and comparison of care within the same center, which increases the risk of selection bias.
The Cochrane review on outpatient versus hospital care following PPROM includes two randomized trials with a total of 116 women18. It concluded that outpatient care is associated with fewer days in hospital and less cost. Both the American College19 and Royal College statements20 on PPROM highlight the lack of data to guide recommendations regarding hospital or outpatient care. As a comparison of different center policies has never been performed, we opted for this study design in order to limit the risk of bias.
Our aim was therefore to compare neonatal outcomes for women with PPROM before 34 WG, firstly according to their management policies - inpatient and outpatient care - and secondly between inpatients and effective outpatients (discharge from hospital).
Material and Methods
Study design, context
This retrospective study was carried out in six French tertiary care referral centers. Three centers had an inpatient care policy (ICP) (the maternity units of the Robert Debré, Intercommunal de Créteil and Angers Hospitals) and three centers had an outpatient care policy (OCP) (the maternity units of the Antoine Béclère, Bicêtre and Louis Mourier Hospitals). The study was conducted over a 4-year period between January 1, 2009 and December 31, 2012.
Data sources
Women were identified through the electronic database of each center. Data were then collected from obstetric and neonatal hospitalization reports.
Inclusion/exclusion criteria
Inclusion criteria were all patients admitted to hospital with a diagnosis of PPROM between 24+0 and 33+6 WG not delivered within the 48 hours following PPROM. Exclusion criteria were twin pregnancies, cases with fetal abnormalities conditioning prognosis and women who delivered in another maternity unit.
Description of management in each group
Gestational age was determined using the routine ultrasound examination performed in the first trimester. In the OCP, criteria for outpatient care were absence of infectious signs (maternal fever (> = 38 °C), uterine tenderness, fetal tachycardia (>160 beats/min) and foul odor of amniotic fluid), signs of labor (regular uterine contractions, cervical dilatation over 3 cm), and patient acceptance.
A patient eligible for outpatient care was discharged from the second day of hospitalization with the following management: daily fetal cardiotocography, a laboratory blood sample twice a week and a weekly clinical and ultrasound exam. In the ICP group, the same protocol was applied, but the patient was not discharged until delivery.
All patients received betamethasone (two intramuscular doses of 12 mg 24 hours apart) for fetal lung maturation and antibiotic prophylaxis at PPROM (ampicillin or cefixime) according to the unit’s protocol. Antibiotic therapy was amoxicillin in five centers and cefotaxime in one center. The administration of tocolysis for a duration of 48 hours was not systematic and depended on the center’s practices. All patients were managed expectantly until spontaneous labor, signs of chorioamnionitis, fetal heart rate anomalies, or acute complications (placental abruption, cord prolapse). If not delivered at 37 WG, induction of labor or cesarean section was performed.
Maternal demographic data were collected: maternal age, weight, size, parity, history of PPROM, history of late miscarriage (abortion between 14+6 and 23+6 WG) or prematurity, previous cesarean section, cervical cerclage, and pregnancy pathology (abnormal placental insertion such as placenta previa, intrauterine growth restriction or gestational diabetes mellitus).
Data upon admission were collected: gestational age at PPROM, vaginal bacterial culture, maternal serum levels of inflammatory markers (C-reactive protein and white blood cell count21), fetal presentation, and amount of amniotic fluid evaluated by the single deepest pocket. The cervix was not systematically measured by ultrasound and this was left to the clinician’s discretion. In centers reporting an OCP, inpatient or outpatient care following the initial period in hospital and duration of hospitalization were collected.
Outcomes
The main variable of interest was a composite measure of neonatal outcome, including one or more of the following criteria: perinatal death, early neonatal infection with sepsis (defined as sepsis in a neonate with a positive blood or cerebrospinal culture in the first 48 hours of life), respiratory distress syndrome, bronchopulmonary dysplasia at discharge (oxygen requirement for a minimum of 28 days)22, grade 3 or 4 intraventricular hemorrhage23, and necrotizing enterocolitis24. Other neonatal outcomes included birth weight, gestational age at birth, and Apgar scores.
Other obstetric outcomes included gestational age at delivery, latency duration (i.e., the time interval between PPROM and delivery), chorioamnionitis, placental abruption, cord prolapse, and delivery mode. Chorioamnionitis was defined by the presence of maternal fever (> = 38 °C) with one of the following signs: uterine tenderness, fetal tachycardia (>160 beats/min) and foul odor of amniotic fluid10. Leukocytosis was defined as a white blood cell count above 15 G/L. Elevated CRP at PPROM was defined as above 15 mg/L. Maternal sepsis was defined by a suspected infection with at least two of the following criteria: a temperature above 38 °C or below 36 °C, a respiratory rate above 20 per minute (or PaCO2 < 32 mm Hg), a heart rate above 90 per minute, and a white blood cell count above 12,000 per mm3 or below 4,000 per mm3 25. Post-partum endometritis was defined by the association of fever with foul lochia or a painful uterus at mobilization.
Statistical analysis
Firstly, outcomes were compared according to the management policy of the center. The comparison of continuous variables was performed using Student’s t-test. Other variables were compared using the chi-square test and the Fisher exact test, where appropriate. Continuous variables are expressed as mean and standard deviation or median and interquartile range and categorical variables as percentages. All tests were two-sided at a significance level of 0.05. Neonatal morbidity and mortality were compared using a univariate logistic regression. Results were expressed as crude odds ratios (ORs) with their 95% confidence intervals (95% CIs), considering the inpatient policy group as the reference group.
Secondly, neonatal outcomes were compared between inpatients and effective outpatients using propensity score matching. Propensity score matching is a technique in which quasi-case/control pairs are produced from a retrospective cohort. In our study, propensity score is the probabilistic measure that reflects the propensity of the patient, based on other characteristics, to have outpatient management. For each group, the propensity to have outpatient management was calculated using multivariate logistic regression with outpatient as the dependent variable and all available patient characteristics as the independent variables. So, propensity score is used to reduce confounding variables and thus includes variables thought to be related to both outpatient management and outcome.
Effective outpatient was the actual discharge from hospital (because some patients were candidates for outpatient management but were not discharged from hospital for various reasons). This statistical strategy was used to minimize the effects of covariables related to differences in the two groups of patients26. Propensity score was constructed with the following variables: maternal age, ethnic group, smoking, BMI >25 kg/m2, a history of cesarean section, gestational age at PPROM, cervical length <25 mm at PPROM, anhydramnios at PPROM, administration of tocolysis, and cerclage during pregnancy.
Once the propensity score has been calculated for each observation, one must ensure that there is overlap in the range of propensity scores across inpatient and outpatient groups (called “common support”)26.
The analysis was based on propensity score matching with a 1:1 matching algorithm without replacement. The ORs for neonatal morbidity (early neonatal infection with sepsis, respiratory distress syndrome, bronchopulmonary dysplasia at discharge, grade 3 or 4 intraventricular hemorrhage, and necrotizing enterocolitis) and mortality were analyzed in the matched sample. Statistical analysis was performed using Stata 14 Software (StataCorp LP, College Station, TX, USA).
Ethics
The study was approved by the Institutional Review Board (IRB, CEROG Comité d’éthique de la recherche en Obstétrique et Gynécologie) (protocol number 2018-OBS-0207). The need to obtain informed consent was waived by the ethics committee. An information sheet explained the purpose and the design of the study to the patients. The original identification of each patient in the database has been encrypted and replaced with surrogate identification. All methods were performed in accordance with the relevant guidelines and regulations.
Details of ethics approval
The procedures of the study received ethical approval from the institutional review board (IRB, CEROG Comité d’éthique de la recherche en Obstétrique et Gynécologie) (protocol number 2018-OBS-0207).
Results
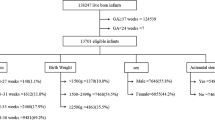
During the study period, 74,195 patients were delivered in the six maternity units. The prevalence of PPROM among singleton births before 34+0 WG in the population was 2.2% (1621/74195), (Fig. 1). Six hundred sixteen patients with PPROM between 24+0 and 33+6 delivered 48 hours after PPROM. Twenty-nine patients (4.7%, 29/616) were excluded because they delivered in another maternity unit. Five hundred eighty-seven patients with PPROM between 24+0 and 33+6 WG were included, 246 in centers with an inpatient care policy (ICP) group and 341 of them in centers with an outpatient care policy (OCP) group. In the centers with outpatient care, the effective rate of outpatient was 19.4% (66/341 patients).
First part: comparison according to the management policy of the center
Maternal demographic characteristics and obstetric history for women with PPROM between 24+0 and 33+6 WG in the ICP and OCP are presented in Table 1. There was no significant between-group difference except for parity (p = 0.04), with significantly more nulliparous women in the outpatient group. The median gestational age at PPROM did not differ between the two groups: 29+2 WG [interquartile range (IQR) 26+3–31+4] versus 29+6 WG (IQR 27+2–31+6; p = 0.14) in the ICP and OCP, respectively. No between-group difference was found for leukocytosis or CRP at admission (Table 2). The rate of sonographic anhydramnios and median cervical length was similar in the two groups.
We also compared inpatients and outpatients among the OCP group for maternal and obstetrical characteristics (Table S1). Gestational age at PPROM was significantly lower for outpatients. Inpatients were more likely to have an elevated CRP (>15 mg/L) at PPROM, and a cervical length <25 mm.
Table 2 summarizes management and delivery characteristics. In the ICP, significantly more tocolytic treatment was administered than in centers with an OCP: 82.5% (203/246) versus 56.6% (193/341), respectively (p < 0.001).
Initial duration of hospitalization was significantly higher for ICP: 14.9 ± 0.5 days versus 11.4 ± 1.0 days in the OCP (p = 0.001). There was no significant between-group difference in latency duration, gestational age at birth, induction of labor, or delivery mode (Table 2).
However, the rate of births after 32 WG was lower in the ICP group, with 47.3% versus 55.4% in the OCP group (p = 0.05). The rate of births before 28 WG was higher in the ICP group, with 18.9% versus 12.9% in the OCP group (p = 0.05). The median birth weight was lower for neonates in the ICP group, with 1632 g versus 1790 g in the OCP group (p = 0.04).
There was no difference in the neonatal composite outcome between the two groups: 14.6% (36/246) versus 15.5% (53/341) in the ICP and OCP, respectively; p = 0.76. The chorioamnionitis rate was similar in the two groups: 12.0% (41/246) versus 9.8% (24/341) in the ICP and OCP groups, respectively; p = 0.39 (Table 2). There was no difference concerning intrauterine death, placental abruption, maternal sepsis, or endometritis rates between the two groups. The cord prolapse rate was significantly increased in the ICP group compared with the OCP group: 4.5% versus 1.5%, respectively (p = 0.03).
Second part: comparison between inpatients and effective outpatients using propensity score matching
Propensity scores among the 66 patients in the effective outpatient care group and the 246 in the inpatient group were calculated. They take into account the possible criteria for in- or outpatient management. Gestational age at birth was not considered in the models because it can be a result of management.
Distribution of the propensity score before and after matching is displayed in Figs S1 and S2. Mean propensity score and covariates were balanced across in- and outpatients. Moreover, standardized differences in the weighted samples were less than 10% (Table S2). The matched groups were found to be well balanced for all recorded variables (Table S2). Thus, the quality of the matching was acceptable. The distribution of standardized differences after propensity scores matching is displayed in Figs S3 and S4. Each effective outpatient is matched only with control inpatients whose propensity score is within a predefined neighborhood of the propensity score of the outpatient. Therefore, 132 patients could be matched, with 66 in each group, inpatients and outpatients.
After propensity score matching, in the matched sample with a 1:1 ratio propensity score, the risk of neonatal morbidity assessed using the composite criteria among inpatients was comparable to the risk among effective outpatients (OR 0.88, 95% CI 0.35–2.25; p = 0.80) (Table 3). In addition, the risk of chorioamnionitis was comparable in the two groups (OR 0.54, 95% CI 0.15–1.95; p = 0.35) (Table 4). Risks of prolapse cord and placental abruption were no longer significantly different between the two groups.
Discussion
Our results show that an OCP in cases of PPROM between 24+0 and 33+6 is not associated with higher neonatal morbidity or mortality than an ICP. Severe acute complications such as placental abruption, cord prolapse or intrauterine death in centers were not more frequent with an outpatient policy. After propensity score matching, the risk of neonatal morbidity and neonatal sepsis was not increased for patients receiving outpatient care.
Three retrospective studies have shown that maternal outcomes (chorioamnionitis, mode of delivery) and neonatal outcomes (hospitalization in intensive care units, respiratory distress syndrome, intraventricular hemorrhage) were comparable between outpatients and inpatients, but these studies suffer from selection bias14,27,28,29. Indeed, most of the women in outpatient care were different from inpatients because of specific eligibility criteria with better prognostic factors. Our study compared inpatient to outpatient care using propensity score matching to equalize the women’s characteristics. Furthermore, two studies were conducted in a single center with small sample sizes14,27.
The Cochrane review reported data from two randomized trials with a total of 116 patients (55 patients in Carlan et al. and 61 in Ryan et al.)18. Inclusion criteria were singleton pregnancy, no signs of infection, amniotic fluid above 2 cm, cephalic presentation, cervical dilatation less than 4 cm, and resident in the country. These trials compared neonatal and maternal morbidity and perinatal mortality between planned outpatient and inpatient care in patients with PPROM before 37 WG. The risk of neonatal morbidity and mortality was not increased for outpatients. But the trials did not have sufficient statistical power to detect meaningful differences between inpatient and outpatient groups18.
The risk of acute complications such as placental abruption and cord prolapse was suggested as the main argument for the need for inpatient management14. Ellestad et al.13 assessed the outcome of pregnancies of patients hospitalized for PPROM who would have received outpatient care according to the criteria defined by Carlan et al.: PPROM before 37 WG, 48 to 72 hours of monitoring in hospital, without signs of infection or of the start of labor16. They concluded that because of the obstetric emergency or the premature birth of a child, being at home could have prevented adequate and sufficiently rapid care in a gestational age-appropriate center13. This is not in agreement with our results, which show that placental abruption rate was similar in the two groups and that the cord prolapse rate was significantly higher in the ICP. However, after propensity score matching, these differences were no longer significant.
One of the strengths of our study is that it relates to a large sample of 587 patients with PPROM with a non-selected population and a low rate of loss to follow-up (4.7%). The patients lost to follow-up gave birth in another maternity hospital, which could be the one where they were initially managed. The study design consisted of two complementary analyses. Firstly, we opted for an original study design with comparison of center policies. The center policy was applied to all patients in the centers even if effective outpatient care concerned only 19.4% of cases. Secondly, we compared effective outpatients with inpatients using propensity score matching. This strategy reduces selection bias due to nonrandom outpatient care assignment in the case of our observational data.
Our study, though, has several limitations. Its multicenter nature allowed for large enrollment but induced differences in management. Differences in protocol mainly involved the administration of tocolysis for centers with an inpatient policy, as well as the type of antibiotic therapy administered. However, those characteristics were taken into account by using the propensity score and, moreover, Combs et al. concluded that tocolysis longer than 48 hours did not increase latency or decrease neonatal complications compared with no tocolysis or tocolysis of less than 48 hours30. Lorthe et al. recently showed that tocolysis in cases of PPROM is not associated with improved obstetric or neonatal outcomes31. In addition, few patients were managed in outpatient care in the outpatient group (19.4%), which could have resulted in a lack of power. Our study was conducted between 2009 and 2012. Although obstetric management of PROM has not changed substantially since 2012, neonatal practice may have shifted since then, which may have implications for the contemporary applicability of the study’s findings. Finally, the retrospective nature of our study did not allow assessment of patient satisfaction and well-being, which are benefits of outpatient care16.
In our series, the observed rate of outpatients in centers declaring an outpatient policy was 19.4%, which is comparable to the rate of 11% to 17% reported in the literature11,12. Moreover, in the participant centers with an OCP, outpatient rates ranged from 14.8% to 30.9%, indicating variability in centers stating the same practices. This variability is multifactorial and is linked to the patient’s socioeconomic status, to the obstetrician, and to the center. Furthermore, patients can choose to refuse outpatient care management, after having received information from an obstetrician on PPROM, about risks, modalities of management and care provision. When the criteria for outpatient care management are not clearly defined, the outpatient decision remains subjective and is left to the discretion of the obstetrician. In addition, it should be noted that some obstetricians are reluctant to allow the discharge of these patients.
To answer the question of the comparison between these two management strategies, a randomized trial would be necessary, including an economic cost analysis and assessment of patient satisfaction. Due to the low prevalence of obstetric and neonatal complications, a very large number of subjects would be necessary and it would hardly be acceptable for patients to be selected randomly, which means that such a trial would be unrealistic. Moreover, current economic restrictions make it difficult to maintain these patients in conventional hospitalization in some centers. There is insufficient evidence to claim that outpatient management should currently be standard care for PPROM. For this purpose, we need more retrospective studies and a large national prospective study.
Conclusion
Outpatient care remains an alternative to conventional inpatient care for PPROM as a result of prolonged care for patients who understand the issues involved.
Data Availability
The datasets analyzed during the current study are not publicly available due to patient privacy but are available from the corresponding author on reasonable request.
References
Mercer, B. Preterm premature rupture of the membranes. Obstet. Gynecol. 101, 178–93 (2003).
Shree, R., Caughey, A. B. & Chandrasekaran, S. Short interpregnancy interval increases the risk of preterm premature rupture of membranes and early delivery. J. Matern. Fetal. Neonatal Med. 1–7, https://doi.org/10.1080/14767058.2017.1362384 (2017).
Roberts, C. L. et al. Childhood outcomes following preterm prelabor rupture of the membranes (PPROM): a population-based record linkage cohort study. J. Perinatol. 37, 1230–1235 (2017).
Mercer, B. M. et al. The Preterm Prediction Study: prediction of preterm premature rupture of membranes through clinical findings and ancillary testing. The National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Am. J. Obstet. Gynecol. 183, 738–45 (2000).
Schaaf, J., Mol, B., Abu-Hanna, A. & Ravelli, A. Trends in preterm birth: singleton and multiple pregnancies in the Netherlands, 2000–2007. BJOG An Int. J. Obstet. Gynaecol. 118, 1196–1204 (2011).
Parry, S. & Strauss, J. F. Premature rupture of the fetal membranes. N. Engl. J. Med. 338, 663–70 (1998).
Tsafrir, Z. et al. Conservative management of preterm premature rupture of membranes beyond 32 weeks’ gestation: Is it worthwhile? J. Obstet. Gynaecol. 1–6, https://doi.org/10.3109/01443615.2014.990432 (2015).
Dammann, O., Leviton, A., Gappa, M. & Dammann, C. E. L. Lung and brain damage in preterm newborns, and their association with gestational age, prematurity subgroup, infection/inflammation and long term outcome. BJOG 112(Suppl), 4–9 (2005).
Goldenberg, R. L., McClure, E. M., Saleem, S. & Reddy, U. M. Infection-related stillbirths. Lancet (London, England) 375, 1482–90 (2010).
Osmanağaoğlu, M. A., Unal, S. & Bozkaya, H. Chorioamnionitis risk and neonatal outcome in preterm premature rupture of membranes. Arch. Gynecol. Obstet. 271, 33–9 (2005).
Goya, M. et al. Premature rupture of membranes before 34 weeks managed expectantly: maternal and perinatal outcomes in singletons. J. Matern. neonatal Med. 26, 290–293 (2013).
Mc Call Jones, P. Patient satisfaction with home care after early postpartum hospital discharge. Home Care Provid. 2, 235–241 (1997).
Ellestad, S. C. et al. Preterm premature rupture of membrane management–inpatient versus outpatient: a retrospective review. Am. J. Perinatol. 25, 69–73 (2008).
Ayres, A. W. Home management of preterm premature rupture of membranes. Int. J. Gynaecol. Obstet. 78, 153–5 (2002).
Huret, E., Chanavaz-Lacheray, I., Grzegorczyk-Martin, V. & Fournet, P. Home care of premature rupture of membranes prior to 37 weeks’ gestation. Gynécologie, Obs. Fertil. 42, 222–8 (2014).
Carlan, S. J., O’Brien, W. F., Parsons, M. T. & Lense, J. J. Preterm premature rupture of membranes: a randomized study of home versus hospital management. Obstet. Gynecol. 81, 61–4 (1993).
Ryan, G. et al. Randomized controlled trial of inpatient vs. outpatient management of PPROM. Am. J. Obstet. Gynecol. 180, S95 (1999).
Abou El Senoun, G., Dowswell, T. & Mousa, H. A. Planned home versus hospital care for preterm prelabour rupture of the membranes (PPROM) prior to 37 weeks’ gestation. Cochrane database Syst. Rev. CD008053, https://doi.org/10.1002/14651858.CD008053.pub2 (2010).
American College of Obstetricians and Gynecologists, A. ACOG Practice Bulletin No.80: premature rupture of membranes. Clinical management guidelines for obstetrician-gynecologists. Obstet. Gynecol. 109, 1007–19 (2007).
Royal College of Obstetricians and Gynaecologist. Preterm prelabour rupture of membranes: Guidelines. Green top guideline (2010).
Hvilsom, G. B., Thorsen, P., Jeune, B. & Bakketeig, L. S. C-reactive protein: a serological marker for preterm delivery? Acta Obstet. Gynecol. Scand. 81, 424–9 (2002).
Jobe, A. H. & Bancalari, E. Bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 163, 1723–9 (2001).
Volpe, J. J. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 8, 110–124 (2009).
Bell, M. J. et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 187, 1–7 (1978).
Bone, R. C., Sibbald, W. J. & Sprung, C. L. The ACCP-SCCM consensus conference on sepsis and organ failure. Chest 101, 1481–3 (1992).
Garrido, M. M. et al. Methods for Constructing and Assessing Propensity Scores. Health Serv. Res. 49, 1701–1720 (2014).
Garabedian, C. et al. Preterm rupture of membranes: Is home care a safe management? J. Gynecol. Obstet. Biol. la Reprod., https://doi.org/10.1016/j.jgyn.2015.02.018 (2015).
Beckmann, M. & Gardener, G. Hospital versus outpatient care for preterm pre-labour rupture of membranes. Aust. N. Z. J. Obstet. Gynaecol. 53, 119–24 (2013).
Palmer, L. et al. A retrospective cohort study of hospital versus home care for pregnant women with preterm prelabor rupture of membranes. Int. J. Gynaecol. Obstet. 137, 180–184 (2017).
Combs, C. A., McCune, M., Clark, R. & Fishman, A. Aggressive tocolysis does not prolong pregnancy or reduce neonatal morbidity after preterm premature rupture of the membranes. Am. J. Obstet. Gynecol. 190, 1723–8; discussion 1728–31 (2004).
Lorthe, E. et al. Tocolysis after preterm premature rupture of membranes and neonatal outcome: a propensity-score analysis. Am. J. Obstet. Gynecol. 217, 212.e1–212.e12 (2017).
Acknowledgements
We are grateful for the cooperation of the maternity and neonatal units. We thank David Marsh, BSc (Biophysics, First Class Honours, University of Leeds, UK) PhD (Biophysics, King’s College London, UK), freelance copyeditor/proofreader, for language editing.
Author information
Authors and Affiliations
Contributions
H.B., G.K., C.D. and M.V.S. were involved in the conception and planning of the study. H.B. and C.D. acquired the data. H.B. carried out the analysis and drafted the manuscript. H.B., G.K. and M.V.S. interpreted the data. G.K., M.V.S., T.S., L.S. and A.B. reviewed the article critically, and revised it for important intellectual content. All authors approved the final version to be published and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing Interests
Dr. Sentilhes carried out consultancy work and has been a lecturer for Ferring Laboratories. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bouchghoul, H., Kayem, G., Schmitz, T. et al. Outpatient versus inpatient care for preterm premature rupture of membranes before 34 weeks of gestation. Sci Rep 9, 4280 (2019). https://doi.org/10.1038/s41598-019-40585-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40585-8
This article is cited by
-
Obstacles to Accepting Care: Understanding Why Obstetric Patients Leave against Medical Advice
Maternal and Child Health Journal (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.