Abstract
Iron is an essential mineral required for most forms of life. However, very little is known in relation to the different forms of dietary iron on the development of NAFLD. The aims of this study were to investigate the effects of iron intake from different food types on risk of NAFLD and whether this effect may be modified by other factors. We conducted a hospital-based case–control study including 1,273 NAFLD cases and 1,273 gender and age-matched controls. We conducted in-person interviews while participants completed a questionnaire on food habits. We assessed animal- and plant-derived intake of iron and fat. We observed that animal-derived iron intake (>4.16 mg/day) was positively associated with augmented NAFLD risk in a Chinese population (ORadjusted = 1.66 in the highest quartile compared with the lowest, 95% confidence interval [CI] = 1.01–2.73). In contrast, a high consumption of iron (>16.87 mg/day) from plant-based foods was associated with a decreased NAFLD risk (ORadjusted = 0.61 in the highest quartile compared with the lowest; 95% CI = 0.40–0.935). In addition, high intake of fat or being overweight may exacerbate this effect. Reduced consumption of iron and fat from animal sources could reduce NAFLD risk, as would weight loss.
Similar content being viewed by others
Introduction
Non-alcoholic fatty liver disease (NAFLD) has been defined as the hepatic accumulation of lipids (mainly triglyceride) in the absence of significant alcohol intake (<20 g/day), or other secondary causes. It encompasses a spectrum of disease ranging from uncomplicated steatosis to non-alcoholic steatohepatitis (NASH), and augments the risk of cirrhosis and hepatocellular carcinoma1,2. With the escalating incidence of obesity and unhealthy dietary patterns worldwide, NAFLD is currently recognized as a major health burden3,4. However, the pathogenesis of NAFLD remains to be elucidated. The pathophysiological mechanisms for the development and progression of NAFLD are both complex and multi-factorial. A number of parallel factors (including insulin resistance, adipose-driven hormones, nutritional factors, gut flora, and genetic factors), all act together in genetically predisposed individuals with induction of NAFLD5. Recently, interest has focused on the association of iron overload and NAFLD6,7,8.
Iron is an essential mineral for most forms of life. Its importance lies in its ability to mediate electron transfer and it plays a vital role in a number of essential biological functions, including transport of oxygen and cell respiration. However, iron is also potentially toxic, as it can act as a pro-oxidant. Iron has been reported as the initial precipitant in promoting liver steatosis through lipid interference9 or glucose metabolism10. Excess iron is associated with oxidative stress and leads to fat peroxidation11,12, B-cell dysfunction, and impaired glucose metabolism13. In accordance with this, previous studies have reported that approximately one third of patients with NAFLD exhibit disturbed iron homeostasis as demonstrated by high serum ferritin with normal or mildly elevated transferrin saturation14. While iron depletion has been shown to insulin resistance, it has also been associated with improvement in liver damage in patients with NAFLD15,16. Furthermore, hepatic iron may be a determinant of serum ferritin in NAFLD patients17. Recently, a meta-analysis revealed that serum ferritin was increased in NAFLD patients (NAFL and/or NASH) compared with control patients18. However, very little is known in relation to the different forms of dietary iron on the development of NAFLD in populations. Dietary iron comes in two different forms; non-heme and heme iron, both which can be derived from multiple sources. Heme iron mainly exists in animal-based products, while in both plant and animal-based foods is present as non-heme iron19. Heme iron is the less prevalent form of dietary iron, however, it is absorbed four to five times more easily than non-heme iron20, and it has been suggested that in a typical western diet the heme pool contributes up to 50% of the total iron21. Results from a recent epidemiological study showed that red meat consumption was associated with NAFLD risk22, and heme iron has partly attributed to this association23. Here, we explored the the effects of intake of iron from several food sources on NAFLD risk, and interactions between iron from the diet and other established risk factors of NAFLD. We did this by performing a large case-control study among a population in China, where foods have yet to be fortified with iron.
Results
Selected characteristics of study participants
Cases and controls differed only in: some demographic factors, established risk factors for NAFLD, and dietary factors (Table 1). There were no significant differences in gender, age, and marital status between the two groups. Compared to controls, NAFLD patients tended to be less educated, were more likely to smoke, rarely drank alcohol, and had lower income. Cases had higher blood pressure, increased body mass index (BMI), higher fasting plasma glucose (FPG), increased uric acid (UA), triglyceride (TG), total cholesterol (TC), aspartate aminotransferase (AST), alanine aminotransferase (ALT), and ApoB but had lower levels of HDL-C and ApoA (each P < 0.05). Finally, total fat intake was significantly greater among NAFLD patients than among controls.
The associations of dietary iron intake with NAFLD
The association of dietary iron consumption and risk of NAFLD is presented in Table 2. Total daily iron intake and plant-derived iron intake (non-heme iron) was similar in the two groups but daily intake of animal-derived iron (heme iron) in patients with NAFLD [(7.84 ± 4.11) mg/d] was significantly higher (P = 0.013) than in controls [(7.24 ± 4.04) mg/d]. Considering that alcohol intake impacts upon the severity of steatosis and also on circulating iron levels, it is possible that alcohol inhibits hepcidin synthesis and thus ameliorates dietary iron absorption. Therefore, subjects who consumed alcohol to excess were excluded. A similar positive association between iron derived from animal products and NAFLD was observed among subjects without alcohol consumption (shown in Table S1). In order to adjust for potential confounding factors, controls were categorized into quartiles by consumption of total iron, heme iron, and non-heme iron (Table 3). Elevated total iron consumption was not associated significantly with NAFLD risk. However, clear differences were observed in the effects of iron derived respectively from plant and animal sources. After adjusting for potential confounders, high consumption of iron (>4.16 mg/day) from animal-sourced foods was associated strongly with increased risk of NAFLD among all participants (OR Q4 vs. Q1 = 1.66; 95% CI = 1.01–2.73; Ptrend = 0.003). High consumption of iron (>16.87 mg/day) from plant-based products was associated with reduced NAFLD risk (OR Q4 vs. Q1 = 0.61; 95% CI = 0.40–0.94; Ptrend = 0.013). When the fully adjusted model was stratified by gender, a significant positive association was demonstrated between animal-derived iron and NAFLD, and this effect was observed only among men (OR = 2.29; 95% CI = 1.27–4.13 in the highest quartile compared with the lowest; Ptrend < 0.001). However, a significant inverse association was found between plant-derived iron and NAFLD, which was observed only for female (ORadjusted = 0.33, 95% CI = 0.15–0.71 in the highest quartile compared with the lowest, Ptrend = 0.043). There was no evidence of effect modification by age for the association between total dietary iron, heme iron, or non-heme iron intake and NAFLD (data not shown).
Clinical parameters and their association with different quartiles of animal-derived iron from foods in NAFLD patients and controls
We assessed the relationship between quartiles of animal-derived iron, BMI, fasting plasma glucose (FPG), and serum lipids in NAFLD and control subjects, respectively. Among NAFLD patients, subjects with higher animal-derived iron intake tended to be associated with higher levels of BMI, UA, and ALT. Whereas in controls, subjects with higher animal-derived iron consumption tended to be associated with higher levels of serum UA and lower levels of HDL-C (shown in Table S2).
The combined effects of animal-derived iron and selected risk factors on NAFLD risk
We also explored the combined effects of animal-derived iron and established risk factors of NAFLD (Table 4). We did not observe significant interactions between animal-derived iron, moderate alcohol consumption, or smoking on NAFLD risk were observed. However, animal-derived iron appeared to have a synergistic effect with dietary fat intake (P for interaction = 0.041) and being overweight or obese (P for interaction = 0.006) in precipitating NAFLD risk. Specifically, subjects with the highest intake of animal-derived iron and higher levels of total dietary lipid, corresponding to >9.71 mg iron/day and >44 mg lipid/day from the diet had the greatest risk of NAFLD compared with subjects ingesting low levels of animal-derived iron and total fat (OR adjusted = 2.08, 95% CI:1.50–2.89). Similarly, subjects with the highest intake of animal-derived iron and BMI ≥24 kg/m2 had the highest NAFLD risk compared with subjects consuming low levels of iron derived from animals and BMI < 24 kg/m2 (adjusted OR = 9.15, 95% CI: 5.96–14.07).
Discussion
In this large case-control study of a Han Chinese population, high daily consumption of iron (>4.16 mg/day) derived from animal sources was associated with significantly augmented risk of NAFLD, particularly in men. However, a significant inverse association was found between a high daily intake of plant-source iron (>16.87 mg/day) and NAFLD, particularly among women. We also observed a statistically significant synergistic interaction between animal-derived iron and BMI ≥24 kg/m2. To our knowledge, this is the first case-control study to ever evaluate the independent and combined effects of different iron forms from dietary and selected risk factors on the risk of NAFLD.
Iron is an essential mineral, which is required for vital functions throughout the body. It is utilized as an enzyme cofactor, involved in oxygen transport, plays an essential role in ATP generation, and it supports growth24. An excess of iron is also, however, a potent cause of cellular injury from oxidative stress due to the generation of ROS mediated by the Fenton reaction25. A strong association between excess iron that is not related to several metabolic syndrome (MetS) features and hereditary hemochromatosis (HHC), has been demonstrated26,27,28,29. However, most studies have focused on serum iron markers (ferritin or hepcidin) and NAFLD risk, where disturbed serum levels of these elements were induced by conditions such as insulin resistance and hyperglycemia30,31. Thus, serum levels of these elements can be both contributors to the development of NAFLD and/or the result of metabolic distress in NAFLD. However, in the present work, we evaluated the association between intake of iron from different food sources with risk of NAFLD, rather than serum levels of iron. After adjusting for potential confounders, we found that animal-derived iron was associated strongly with increased risk of NAFLD among all participants, while high consumption of iron from plant-based products was associated with decreased NAFLD risk. Our observations for dietary iron largely reflect those found in another Chinese population conducted by Zheng et al.32, who demonstrated a significantly positive association between dietary heme-iron intake and NAFLD. However, dietary intake of non-heme iron was not associated with NAFLD. Our findings were also supported by experimental results noted in mice where a high-iron diet was positively associated with elevated hepatic cholesterol synthesis leading to hyperglycemia, hyperinsulinemia, and IR, which are risk factors for NAFLD33,34. In addition, NAFLD has been regarded as a “hepatic manifestation of the metabolic syndrome (MetS)”. Therefore, our findings were indirectly supported by several studies that revealed a positive association between heme iron consumption and risk of type 2 diabetes mellitus and cardiovascular disease; these share common metabolic parameters with NAFLD29,35,36,37. The unusual effects of animal-derived iron and plant-iron consumption on NAFLD risk may be explained by the differences in bioavailability and their effects on iron stores within the body38. Iron obtained from animal products mainly consists of heme iron, which has high bioavailability, and is the main contributor to stored body iron in humans. Non-heme iron is less bioavailable but also mainly obtained from fruit, vegetable, whole grains, and fortified cereals. These are commonly considered to be beneficial foods that contain some healthy components such as plant proteins, fiber and others antioxidants. Furthermore, dietary patterns may alter the gut microbiota39, which contributes to pathogenesis of NAFLD through multiple mechanisms40.
Previous studies on iron-NAFLD risk either assessed one gender or did not provide a stratified analysis32. In the present study, when the model was stratified by gender, a significant positive association was found between animal-derived iron and NAFLD only for men. In contrast, a significant inverse association was found between plant-derived iron and NAFLD only for women. The gender difference observed in this study may be explained by the heterogeneity in iron absorption. Studies have reported that women generally have higher iron requirements, and they absorb approximately twice as much iron as men from the same dietary intake of this mineral41. Nonetheless, further studies are needed to validate the gender heterogeneity in the aforementioned association between heme iron and NAFLD, and potential mechanisms should be explored further.
Increased hepatic iron associated with the MetS has been commonly observed as a syndrome and has been named as ‘dysmetabolic iron overload syndrome42,43. In this study, we also evaluated the influence of animal-derived iron on metabolic syndrome components and we observed that subjects with higher animal-derived iron intake tended to be associated with higher levels of BMI and UA among NAFLD patients. In controls, subjects with higher animal-derived iron intake tend to be associated with higher levels of serum UA and lower levels of HDL-C. Previous studies have reported that high levels of BMI and serum UA were associated with NAFLD risk. Therefore, we speculate that high animal-derived iron intake is associated with NAFLD risk by increased BMI and/or serum UA. Nevertheless, more studies are required to confirm our findings.
The underlying mechanisms for the development of NAFLD are complex and multi-factorial. We also found that animal-derived iron appeared to have a combined effect with dietary fat intake and being overweight or obese with respect to increased NAFLD risk. We postulate that one possible mechanism for the interaction between iron and fat is that particular fats and fatty acids might significantly enhance transport of reactive forms of iron into cells44. Another reason for the interaction may be explained by results from an in vivo study, which showed that a high-fat diet and additional iron acted synergistically in exacerbating hyperglycemia, hyperinsulinemia, and IR. Furthermore, such a diet/supplementation may induce synthesis of lipids, and thereby increase hepatic TG and cholesterol levels, resulting in hepatic steatosis33,34. In addition, we also found that subjects with BMI ≥24 kg/m2 experienced an enhanced risk of NAFLD associated with animal-derived iron intake than their non-overweight peers. The mechanisms underlying the effect modification by BMI in this study have yet to be elucidated; therefore, future studies are needed to evaluate the potential mechanisms.
Our study had both strengths and limitations. The strength of our study was the large sample size, which could have reduced type II errors. Secondly, detailed collection of data through face-to-face interviews using a FFQ and extensive information on anthropometrics and lifestyle factors were also collected in this study, which allowed us to potentially adjust for confounding factors. The study had enough power to allow investigation of interactions between animal-derived iron and total lipid; there is a plausible mechanism for such interactions. Nonetheless, several limitations of our study require consideration when interpreting study findings. First, selective participation and recall bias are potential concerns in our study as with other epidemiologic studies of this nature. Second, because dietary iron intake was self-reported, we cannot exclude the possibility of reporting bias. Therefore, our study can only provide etiological clues in exploring the association between dietary heme iron intake and NAFLD. It cannot confirm whether the difference in heme iron intake between NAFLD subjects and controls is really biologically relevant. Third, we found it difficult to obtain samples from individuals who attended for a regular medical health check-up. Thus, we only examined dietary intake rather than measurement of circulating iron levels, and so we cannot accurately predict what level of iron was absorbed. Fourth, as with all observational studies, although we adjusted for known confounders, it is not possible to exclude others confounding of unknown covariates. Finally, our study was performed on a health examination of the Han Chinese population. Therefore, extrapolation of our findings to the general population or other ethnicities should be done with caution. Further population-based studies are warranted to verify our results, especially those that include different ethnic groups or populations that eat predominantly plant-based products e.g. vegetarians.
Conclusions
In this Chinese population, we found that high consumption of animal-derived iron was associated with significantly increased risk of NAFLD, particularly among men. This effect may be augmented by high total fat intake or being overweight. While confirmation of these findings in other populations is required, and further exploration of possible underlying biological mechanisms of observed associations is warranted, our study suggests that animal-derived iron and fats or BMI have combined effects in the development of NAFLD. We suggest reductions in consumption of animal-derived iron and fat (or weight loss) to reduce NAFLD risk.
Methods
Study design
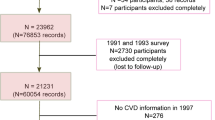
We performed a case-control study for NAFLD in health examination centers at two teaching hospitals of Fujian Medical University (the Union Hospital and Affiliated Nanping First Hospital, China), from June of 2011 to May of 2017. As described in our previous work45, criteria for inclusion of cases in the study was permanent residency in urban Fujian being 20–74 years of age, no secondary causes of steatosis, including alcohol misuse in the past year (weekly ethanol consumption ≥140 g for men, or ≥70 g for women), total parenteral nutrition, infection with hepatitis B or hepatitis C virus, or the use of drugs known to precipitate hepatic steatosis prior to diagnosis (cases) or study enrollment (controls). Incident cases were diagnosed by liver ultrasonography using established criteria46. Controls with no liver steatosis by ultrasonography were randomly selected from the same center during the same study period. Eligibility criteria for controls were identical to those for cases with the exception of liver steatosis diagnosis. Controls were frequency matched to cases on age (within 5-yr intervals), gender, ethnicity, occupation and region of origin. Additionally, cases and controls with any of the following were excluded from the study: type II diabetes, hypertension, hyperlipidemia, autoimmune hepatitis, drug-induced liver disease, primary biliary cirrhosis, and primary sclerosing cholangitis. 1,273 eligible cases and controls were recruited to the study and completed an interview. All enrolled subjects were of Chinese Han ethnicity. No significant differences were found for the mean age and gender distribution between cases and controls. All subjects who participated in this study provided written informed consent and the study was approved by the local ethics committees of Fujian Medical University. In addition, all methods were performed in accordance with the relevant guidelines and regulations of the University.
Physical, anthropometric, and biochemical evaluation
After an overnight fast, all subjects underwent a complete physical examination in the morning. The examination included anthropometric measurements, abdominal ultrasonography, and biochemical determinations. Body weight, height, diastolic arterial blood pressure (DABP) and systolic arterial blood pressure (SABP) were measured using standardized procedures47. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. As described in our previous work48, hypertension was defined as systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg, or if the subject was taking anti-hypertensive medications. Venous blood samples were taken immediately after the anthropometric examinations. Total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C), TGs, apolipoprotein A (ApoA1), apolipoprotein B (ApoB), serum uric acid (UA), fasting plasma glucose (FPG), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were measured by standard clinical laboratory techniques.
Dietary Assessment
Dietary information on typical food consumption was collected by an evaluated 125- item semiquantitative food frequency questionnaire (SFFQ), which had been specifically developed and validated for the southern Chinese population49. Participants were asked to estimate information on the average frequency of consumption of selected foods (rarely, < once/month, 1–3 times/month, 1–2 times/week, 3–4 times/week, 5–6 times/week, once/day, twice/day and >twice/day). Ignoring any recent changes, serving sizes in lians (1 lian equals 50 g) or jins (where 1 jin equals 500 g) over the previous year were considered. These frequencies were then multiplied by 0, 0.03, 0.07, 0.22, 0.50, 0.79, 1, 2, 3/day, respectively to obtain the intake of each food item. When seasonal foods were assessed, subjects described their intake based on availability and number of months per year in which they consumed the food. A lifestyle questionnaire was used to collect information relating to socio-demographic characteristics and lifestyles. The information collected included age, gender, family income, marital status, education level, tea drinking, smoking status, light alcohol consumption, physical activity, and medical history.
Assessment of iron intake
The quantity of iron for each food item was calculated from Chinese food composition tables50. For each food and beverage queried, the product of daily frequency scores and standard serving size (in grams) was multiplied by the iron content per 100 g of each food to obtain the daily iron intake. Daily iron intake from various foods was obtained by adding up all the food items. We did not have sufficient data on iron supplementation therefore we could not measure supplemental iron intake. We similarly estimated daily total energy intake (kcal) by calculating the energy intake for individual food items. We also used the FFQ to assess heme iron intake, which was estimated by calculating iron intake that was exclusively derived from animal foods. These included beef, pork, sausage, chicken, liver, fish, dried fish, and boiled fish paste. Non-heme iron intake was calculated as total iron minus heme iron intake.
Statistical analysis
χ2 tests were used to compare study participant characteristics, including gender, family income, marital status, education level, tea drinking, smoking status, alcohol consumption, and physical activity according to NAFLD status. Independent sample t-tests were used to compare mean values according to NAFLD status. Consumption of total iron, heme iron, and non-heme iron was categorized among controls in quartiles. The association between NAFLD risk and each quartile of iron intake was determined by unconditional logistic regression models [calculating odds ratios (OR) and their 95% confidence intervals (CIs)], using the lowest quartile as the reference category. Independent risk factors for NAFLD—age, gender, marital status, family income, educational attainment, body mass index (BMI), regular physical activity (yes/no), current cigarette smoking, biochemistry, total dietary fat intake, and total energy intake—were included in all models. By conducting stratified analyses and evaluating interaction terms, we evaluated whether associations between dietary animal-derived iron consumption and NAFLD risk were modified by other established risk factors of NAFLD. These factors included BMI (<24 or ≥24 kg/m2), moderate alcohol consumption (yes or no), current cigarette smoking (yes or no), and dietary fat intake (low or high). All data analyses were performed using IBM SPSS Statistics Release Version 19.0.0.1 (IBM SPSS, 2010, Chicago, IL).
References
Barshop, N. J., Sirlin, C. B., Schwimmer, J. B. & Lavine, J. E. Review article: epidemiology, pathogenesis and potential treatments of paediatric non-alcoholic fatty liver disease. Aliment Pharmacol Ther 28, 13–24 (2008).
Hoekstra, M. et al. The expression level of non-alcoholic fatty liver disease-related gene PNPLA3 in hepatocytes is highly influenced by hepatic lipid status. J Hepatol 52, 244–251 (2010).
Fan, J. G. & Cao, H. X. Role of diet and nutritional management in non-alcoholic fatty liver disease. J Gastroenterol Hepatol 28(Suppl 4), 81–87 (2013).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84 (2016).
Buzzetti, E., Pinzani, M. & Tsochatzis, E. A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 65, 1038–1048 (2016).
Sabrina, N. et al. Serum Iron:Ferritin Ratio Predicts Healthy Body Composition and Reduced Risk of Severe Fatty Liver in Young Adult Women. Nutrients 9 (2017).
Kowdley, K. V. et al. Serum ferritin is an independent predictor of histologic severity and advanced fibrosis in patients with nonalcoholic fatty liver disease. Hepatology 55, 77–85 (2012).
Aigner, E. et al. Pathways underlying iron accumulation in human nonalcoholic fatty liver disease. Am J Clin Nutr 87, 1374–1383 (2008).
Ahmed, U., Latham, P. S. & Oates, P. S. Interactions between hepatic iron and lipid metabolism with possible relevance to steatohepatitis. World J Gastroenterol 18, 4651–4658 (2012).
Dongiovanni, P. et al. Dietary iron overload induces visceral adipose tissue insulin resistance. Am J Pathol 182, 2254–2263 (2013).
Lasheras, C. et al. Plasma iron is associated with lipid peroxidation in an elderly population. J Trace Elem Med Biol 17, 171–176 (2003).
King, S. M. et al. Daily supplementation with iron increases lipid peroxidation in young women with low iron stores. Exp Biol Med (Maywood) 233, 701–707 (2008).
Liu, Q. et al. Role of iron deficiency and overload in the pathogenesis of diabetes and diabetic complications. Curr Med Chem 16, 113–129 (2009).
Datz, C., Muller, E. & Aigner, E. Iron overload and non-alcoholic fatty liver disease. Minerva Endocrinol 42, 173–183 (2017).
Valenti, L. et al. Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study. Am J Gastroenterol 102, 1251–1258 (2007).
Valenti, L. et al. A randomized trial of iron depletion in patients with nonalcoholic fatty liver disease and hyperferritinemia. World J Gastroenterol 20, 3002–3010 (2014).
Ryan, J. D. et al. Hepatic iron is the major determinant of serum ferritin in NAFLD patients. Liver Int 38, 164–173 (2018).
Du, S. X. et al. Association of serum ferritin with non-alcoholic fatty liver disease: a meta-analysis. Lipids Health Dis 16, 228 (2017).
Hurrell, R. & Egli, I. Iron bioavailability and dietary reference values. Am J Clin Nutr 91, 1461S–1467S (2010).
Rajpathak, S. N. et al. The role of iron in type 2 diabetes in humans. Biochim Biophys Acta 1790, 671–681 (2009).
Jiang, R. et al. Dietary iron intake and blood donations in relation to risk of type 2 diabetes in men: a prospective cohort study. Am J Clin Nutr 79, 70–75 (2004).
Zelber-Sagi, S. et al. High red and processed meat consumption is associated with non-alcoholic fatty liver disease and insulin resistance. J Hepatol (2018).
Carpenter, C. E. & Mahoney, A. W. Contributions of heme and nonheme iron to human nutrition. Crit Rev Food Sci Nutr 31, 333–367 (1992).
Huang, X. Iron overload and its association with cancer risk in humans: evidence for iron as a carcinogenic metal. Mutat Res 533, 153–171 (2003).
Babitt, J. L. & Lin, H. Y. The molecular pathogenesis of hereditary hemochromatosis. Semin Liver Dis 31, 280–292 (2011).
Park, S. K. et al. Clinical significance of serum ferritin level as an independent predictor of insulin resistance in Korean men. Diabetes Res Clin Pract 107, 187–193 (2015).
O’Brien, J. & Powell, L. W. Non-alcoholic fatty liver disease: is iron relevant? Hepatol Int 6, 332–341 (2012).
Chang, J. S. et al. Serum ferritin and risk of the metabolic syndrome: a population-based study. Asia Pac J Clin Nutr 22, 400–407 (2013).
Talaei, M., Wang, Y. L., Yuan, J. M., Pan, A. & Koh, W. P. Meat, Dietary Heme Iron, and Risk of Type 2 Diabetes Mellitus: The Singapore Chinese Health Study. Am J Epidemiol 186, 824–833 (2017).
Zheng, Y., Li, X. K., Wang, Y. & Cai, L. The role of zinc, copper and iron in the pathogenesis of diabetes and diabetic complications: therapeutic effects by chelators. Hemoglobin 32, 135–145 (2008).
Swaminathan, S., Fonseca, V. A., Alam, M. G. & Shah, S. V. The role of iron in diabetes and its complications. Diabetes Care 30, 1926–1933 (2007).
Zheng, Q., Wu, Y., Ye, Q., Li, J. & Zhao, Y. Relation between dietary iron intake and nonalcoholic fatty liver disease. Wei Sheng Yan Jiu 44, 527–531 (2015).
Choi, J. S., Koh, I. U., Lee, H. J., Kim, W. H. & Song, J. Effects of excess dietary iron and fat on glucose and lipid metabolism. J Nutr Biochem 24, 1634–1644 (2013).
Silva, M., Silva, M. E., de Paula, H., Carneiro, C. M. & Pedrosa, M. L. Iron overload alters glucose homeostasis, causes liver steatosis, and increases serum triacylglycerols in rats. Nutr Res 28, 391–398 (2008).
Dos Santos Vieira, D. A., Hermes Sales, C., Galvao Cesar, C. L., Marchioni, D. M. & Fisberg, R. M. Influence of Haem, Non-Haem, and Total Iron Intake on Metabolic Syndrome and Its Components: A Population-Based Study. Nutrients 10 (2018).
Eshak, E. S., Iso, H., Maruyama, K., Muraki, I. & Tamakoshi, A. Associations between dietary intakes of iron, copper and zinc with risk of type 2 diabetes mellitus: A large population-based prospective cohort study. Clin Nutr 37, 667–674 (2018).
Quintana Pacheco, D. A. et al. Red meat consumption and risk of cardiovascular diseases-is increased iron load a possible link? Am J Clin Nutr 107, 113–119 (2018).
Bao, W., Rong, Y., Rong, S. & Liu, L. Dietary iron intake, body iron stores, and the risk of type 2 diabetes: a systematic review and meta-analysis. BMC Med 10, 119 (2012).
Al-Muzafar, H. M. & Amin, K. A. Efficacy of functional foods mixture in improving hypercholesterolemia, inflammatory and endothelial dysfunction biomarkers-induced by high cholesterol diet. Lipids Health Dis 16(1), 194 (2017).
Jia, W. & Rajani, C. The Influence of Gut Microbial Metabolism on the Development and Progression of Non-alcoholic Fatty Liver Disease. Adv Exp Med Biol 1061, 95–110 (2018).
Grauer, A. L. S.-M. P. Sex and Gender in Paleopathological Perspective. In:Grauer AL, Stuart-Macadam P, eds. Iron deficiency anemia: exploring the difference. Cambridge, UK: Cambridge University Press, 54 (2005).
Dongiovanni, P., Fracanzani, A. L., Fargion, S. & Valenti, L. Iron in fatty liver and in the metabolic syndrome: a promising therapeutic target. J Hepatol 55, 920–932 (2011).
Moirand, R. et al. A new syndrome of liver iron overload with normal transferrin saturation. Lancet 349, 95–97 (1997).
Yao, D. et al. Fatty acid-mediated intracellular iron translocation: a synergistic mechanism of oxidative injury. Free Radic Biol Med 39, 1385–1398 (2005).
Peng, X. E., Wu, Y. L., Lu, Q. Q., Hu, Z. J. & Lin, X. MTTP polymorphisms and susceptibility to non-alcoholic fatty liver disease in a Han Chinese population. Liver International 34, 118–28 (2014).
Zeng, M. D. et al. Guidelines for the diagnosis and treatment of nonalcoholic fatty liver diseases. J Dig Dis 9, 108–112 (2008).
Valenti, L. et al. Alpha 1-antitrypsin mutations in NAFLD: high prevalence and association with altered iron metabolism but not with liver damage. Hepatology 44, 857–864 (2006).
Peng, X. E. et al. Genetic Variants in PNPLA3 and Risk of Non-Alcoholic Fatty Liver Disease in a Han Chinese Population. PLoS ONE 7, e50256 (2012).
Ke, L. et al. Relatie validity of a semi-quantitative food frequency questionnaire versus 3 day weighed diet records in middle-aged inhabitants in Chaoshan area, China. Asian Pac J Cancer Prev 6, 376–381 (2005).
Yang, Y. H. M. P. X. China Food Composition Table. Peking: China Institute of Nutriton andFood Safety China CDC (2002).
Acknowledgements
We thank all study participants for their cooperation and acknowledge all the efforts of our recruiting and technical staff. This work was supported by grants from the National Natural Science Foundation of China (No. 81473047), Joint funds for the innovation of science and technology, Fujian province (No. 2017Y9104), and the Training Program Foundation for Middle-aged and Young Talents from Sanitation System of Fujian province (2014-ZQN-ZD-23).
Author information
Authors and Affiliations
Contributions
X.E.P. conceived and supervised the project. S.H.X. and W.J.L. performed in-person interviews. X.E.P., Z.L. and Z.J.H. prepared the database and directed data analysis. X.E.P. and X.L. conducted the quality assurance and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests as defined by Nature Research, or other interests that might be perceived to influence the results and/or discussion reported in this paper.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Peng, XE., Xu, SH., Liu, W. et al. Independent and combined effects of dietary iron composition and selected risk factors on the risk of NAFLD in a Chinese population. Sci Rep 9, 4069 (2019). https://doi.org/10.1038/s41598-019-40449-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40449-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.