Abstract
Predatory behavior and top-down effects in marine ecosystems are well-described, however, intraguild interactions among co-occurring marine top predators remain less understood, but can have far reaching ecological implications. Killer whales and white sharks are prominent upper trophic level predators with highly-overlapping niches, yet their ecological interactions and subsequent effects have remained obscure. Using long-term electronic tagging and survey data we reveal rare and cryptic interactions between these predators at a shared foraging site, Southeast Farallon Island (SEFI). In multiple instances, brief visits from killer whales displaced white sharks from SEFI, disrupting shark feeding behavior for extended periods at this aggregation site. As a result, annual predations of pinnipeds by white sharks at SEFI were negatively correlated with close encounters with killer whales. Tagged white sharks relocated to other aggregation sites, creating detectable increases in white shark density at Ano Nuevo Island. This work highlights the importance of risk effects and intraguild relationships among top ocean predators and the value of long-term data sets revealing these consequential, albeit infrequent, ecological interactions.
Similar content being viewed by others
Introduction
High trophic-level consumers, or top predators, play an important ecological role through top-down forcing1,2,3. Regulation of prey density via direct consumption by predators is the most common form of top-down control documented in ecological literature4,5,6. Yet there is also increasing recognition for non-lethal or behaviorally mediated mechanisms, which can similarly shape ecosystem function and structure7,8,9,10,11. Non-consumptive mechanisms include ‘risk effects’ in which prey are not removed, but respond behaviorally to the presence of a predator by reducing activity or shifting habitats to reduce risk9. Risk effects can result in the ecological equivalent of density reduction and may have negative impacts on fitness including decreased reproductive success through loss of foraging opportunities, increased stress, and increased energy demands associated with predator avoidance12,13,14. Top-down forcing can result in trophic cascades where changes in predator forcing alter the densities of intermediate and lower level consumers down through multiple trophic levels15. Risk effects can also initiate trophic cascades8,16,17. For instance, under threat from a potential predator, behavioral responses of a risk-averse intermediate consumer can result in the local release of its food base18.
In marine ecosystems predator-prey interactions and resulting ecological effects have received relatively more focus2,6,19, whereas much less is known about the interactions among top consumers20, which likely have similar top-down implications for marine ecosystems21. Large-bodied upper trophic-level consumers have few natural predators. However, competition within predator guilds can lead to complex interactions and strongly affect the distribution and abundance of the predator populations5,21,22,23. Relatively common in terrestrial systems, intraguild predation among top predators can potentially reduce exploitation competition for food resources and confer energetic benefits for the prevailing consumer23,24. Even in cases where the rate of killing is very low8, indirect dominance effects can profoundly influence the behavior and fitness of sub-dominant predators25,26,27. In spite of their ubiquity where well-studied22,23,28, the frequency and importance of lethal and sub-lethal interactions among top marine predators remain difficult to measure in the oceanic realm and therefore are potentially underrepresented3.
Many marine top predators exhibit migratory behavior and seasonal aggregations at foraging areas29. Concentrated seasonal foraging is crucial in supporting migratory behavior in many consumers30,31 and, conversely, the seasonal influx of predators can have strong regulatory and behavioral effects on local prey populations3. Perturbations in such predator-prey systems may therefore be impactful for both prey and predator with potentially cascading effects1. In cases of co-occurrence between top predators at such sites, the effect of intraguild interactions on local ecosystem dynamics remains relatively unknown.
Here we document and investigate interactions between two top ocean predators, white sharks (Carcharodon carcharias) and killer whales (Orcinus orca). In the northeastern Pacific (NEP) white sharks aggregate seasonally at Southeast Farallon Islands (SEFI), Año Nuevo Island (ANI), and other pinniped rookeries off the west coast of North America32,33. The sharks’ timing and observed foraging is associated with the seasonal haulout of juvenile elephant seals (Mirounga angustirostrous), a preferred prey34,35,36,37 consumed prior to offshore migration32,38. Although white sharks also forage or scavenge on cetaceans39, teleosts, other elasmobranchs40 and various pinnipeds, their seasonal targeting of elephant seals41 provides a consistent source of caloric capital to fuel extended oceanic migrations42,43.
White sharks and killer whales exhibit a high degree of niche overlap along the western shores of North America. The coastal distribution of NEP white sharks extends from northern Mexico to Canada (and in El Nino years up into Alaskan waters), entirely within the coastal distribution of NEP killer whales that range from Mexico to the Aleutian islands of Alaska44 (Fig. 1). NEP killer whale populations form distinct and stable social groups (pods), which differ in specialization of prey choice (ecotypes). Three recognized NEP ecotypes, ‘resident,’ ‘transient,’ and ‘offshore’, exhibit genetic and phenotypic differentiation45,46,47,48. Transient pods typically feed on marine mammals including elephant seals and sea lions, whereas resident pods target teleosts, mainly salmonids45,49. The offshore ecotype is least known, but thought to primarily target teleosts including salmonids and elasmobranchs such as Pacific sleeper sharks45,50. All three killer whale ecotypes overlap spatially with NEP white sharks and share similar prey resources, and thus may be considered part of the same ecological guild51. Regional overlap is highest during fall and early winter, when NEP white sharks show high site fidelity and extended residency periods at SEFI or other coastal aggregation sites near pinniped rookeries32 for approximately 4–4.5 months (Fig. 1).
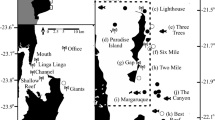
Spatial and temporal overlap of two top predators, white sharks (Carcharodon carcharias), and killer whales (Orcinus orca), and their shared prey, juvenile elephant seals (Mirounga angustirostrous), in the Northeastern Pacific (see Supplement) and at Southeast Farallon Islands (SEFI). Seasonally concentrated activity of each species at SEFI (*) evident from (A) weekly M. angustirostrous counts between March and December (1987–2013), (B) daily mean number of tagged C. carcharias detected (2007–2013) with shaded standard error and, (C) monthly frequency of O. orca observed (1987–2013). Note the two predators co-occur only during the fall peak. Map was created using R software (v3.5.1; https://www.R-project.org/).
Despite an extensive overlap in distributional range and trophic niche, observations of direct interactions between killer whales and white sharks are extremely rare, but have been recorded off California, South Africa, and Southern Australia52,53. A clear understanding of the ecological relationship between these two top predators has remained elusive. An interaction between these top predators in the NEP was documented on Oct 4, 1997, at SEFI in which a white shark was killed and partially consumed (liver only) by transient killer whales. Immediately following this event, observations of white sharks during regular surveys at SEFI declined precipitously; only two predations by sharks were observed in the remaining eight weeks of study at SEFI52.
In the current study we use a combination of an extensive electronic tagging dataset of white sharks throughout Central California together with long-term observational surveys of shark-pinniped predatory interactions and killer whale occurrences at SEFI to elucidate the frequency and consequences of rarely observed intraguild interactions between white sharks and killer whales. We reveal in detail the immediate behavioral nature of the predator interactions, as well as resulting effects that white shark redistribution has on the predator-prey relationship between white sharks and elephant seals.
Materials and Methods
White shark tagging
Between 2006 and 2013, we tagged 165 white sharks (Carcharodon carcharias) with acoustic tags (V16-4H-A69, Vemco; transmitting at 158 Db every 60–180 s for >1400 days) in the vicinities of Southeast Farallon Island, Tomales Point, and Año Nuevo Island using previously described methods32. Briefly, white sharks were attracted to the research boat using a seal decoy made from outdoor carpet. Upon investigation by a shark, the decoy was retrieved using a fishing rod, reel and monofilament line. A small amount of olfactory attractant served to retain the shark near the boat, while the tag was applied using 3 to 4 m pole to insert a titanium dart tethered to the tag beneath the sharks’ dorsal skin. All methods were carried out in accordance with relevant guidelines and regulations, and all experimental protocols were approved under Stanford University animal care protocol 10765. The annual probability of tag loss (shedding) was estimated at 0.32 (95% CI = 0.26, 0.39)54, and annual incremental tagging (average 20 per year) provided a relatively constant flux of tagged individuals in the study system. We maintained continuous coverage in tag detection throughout the study period using sub-surface moored acoustic receivers (VR3, Vemco) stationed at the same three tagging locations32. Equivalent acoustic receivers (VR3, Vemco) deployed off Point Reyes by other research teams provided opportunistic detections supplementing the data collected at the primary shark aggregation sites.
Long-term surveys at Southeast Farallon Island
We recorded the number of hauled-out elephant seals from weekly elephant seal population census surveys conducted throughout the duration of the study (1987–2013) under NMFS Permit No. 373-1868. Surveys occurred between 1000 and 1600, throughout the year. The number of individuals hauled-out on SEFI were tallied with respect to age class as previously described in depth55. During the same period, we recorded the number of predations by white sharks from annual ‘shark watch’ surveys conducted from a lighthouse platform at the highest peak of SEFI (elevation 90 m). Between September 1 and November 30, trained observers continually scanned the waters around the islands for the occurrence of predation events during all daylight hours, unless visibility dropped below 1.6 km, winds exceeded 25 knots, or rain was persistent37. Observation hours averaged 569.7 hours per year (SD = 93.6). Predation observations included prey species when identifiable. Observational survey data were non-experimental, and carried out in accordance with relevant guidelines and regulations.
Killer whale observations were aggregated from a variety of sources, including the ‘shark watch’ surveys, island cetacean surveys56, opportunistic island-based observations, and reports received via a network of wildlife-viewing tour boats during daily radio contact. Killer whale observations were not procedurally standardized, however were relatively consistent throughout the year, except between September 1 and November 30 each year when observation effort increased during the standardized ‘shark watch’ survey. When possible, an estimate of the minimum distance between killer whale pods from the nearest point of the island was recorded. If observation notes indicated killer whales were inside one of the island bays, then a minimum distance of <1 km was assigned. For survey observations with no killer whale sightings, we assumed a distance value of >15 km, a distance beyond the maximum range included in SEFI cetacean surveys56. Where distance was missing from killer whale sightings, we assumed an average distance obtained from all other distance values. Accounts of killer whale behavior and photographs of individual killer whales including dorsal fin and saddle pigmentation were collected from tour boat operators and Island staff. Photos were compared to killer whale ID catalogues to match individuals and determine the pod’s ecotype and size.
Analysis
We hypothesized that shark predations on elephant seals would decline in years when killer whales occurred at SEFI. We initially fit a log-log regression model to the positive predator-prey relationship37 between annual predation rate (number of predations per observation hour) by sharks and mean fall (September 1–November 30) elephant seal counts to determine if deviations corresponded with killer whale occurrences. To test whether the presence of killer whales disrupted the seasonality of shark predations on elephant seals, we modeled daily predation events using a Generalized Additive Mixed Model. We assumed that the realized number of predation events followed a Poisson distribution and that the expected number of predations per day was a smooth function (cyclic cubic regression spline) of date included as ordinal days within a season from September 1 to November 30. We also constrained the spline to start and end at similar values. We treated sighting distance as a factor variable and as a quadratic function of survey observation effort in hours37. We also hypothesized that the shape of the functional relationship between daily shark predations on seals and date would change in relation to the distance of sighted killer whales, and consequently affect the average seasonal predation rate by white sharks. Therefore we included an interaction term between the spline of date and distance. We also expected to have between-year variability in predation rate. This could arise from annual variability in the number or behavior of individual sharks and seals. To control for this variability and ensure unbiased parameter estimates on the other terms, we included season (categorical) as a random effect, modeled as a smooth term with a random effect spline basis57. For model fitting we used a restricted maximum likelihood approach with the package “mgcv” in R57.
To test the hypothesis that killer whale activity in close proximity to SEFI elicited avoidance behavior, we looked at the number of tagged sharks detected per day at each site (SEFI, ANI, and TOM) between 2006 and 2013. We compared this metric during periods when killer whales were observed at SEFI against the mean value for all other years.
Results and Discussion
Multi-predator community at Southeast Farallon Island (SEFI)
Long-term intensive monitoring surveys, combined with electronic tagging and observational studies, revealed the frequency and modality of cryptic interactions amongst marine predator populations at SEFI. Seasonality in white shark and killer whale presence matched annual cycles in prey aggregations, namely juvenile (age 0–3) northern elephant seals (Mirounga angustirostris) that first haulout during spring molt (peaking in April and May) and then again in the fall (peaking in October and November) (Fig. 1). An estimated 219 adult and sub-adult white sharks ((130, 275) 95% credible intervals) aggregate and feed at SEFI and adjacent elephant seal rookeries around Point Reyes during this fall haulout period58. Long-term (1972–2010) birth rates of elephant seals at SEFI are variable (median = 198 births/year; mean = 232; SD = 132) and have decreased to a relatively stable level over the past decade, while the regional population continues to rapidly increase59. Additionaly, California sea lions (Zalophus californianus), Steller sea lions (Eumetopias jubatus), harbor seals (Phoca vitulina ricardii), and northern fur seals (Callorhinus ursinus) also haul out at SEFI at various times of the year55.
Killer whale pod observations occurred year-round on 57 occasions between 1987 and 2013 (Fig. 1C). These sparse occurrences peaked in May, concurrent with gray whale calf migrations, followed by a secondary fall peak during October and November (Fig. 1C). The co-occurrence of white sharks and killer whales was confined to fall, coincident with the peak in adult white shark activity at SEFI (Fig. 1B). During the fall overlap (September–November) killer whale pods were recorded during daily surveys ( mean = 7.7 hr/day; weather permitting) from the island lighthouse at various distances from SEFI on 18 out of 1998 survey days in eight different years: 1992, 1995–1998, 2000, 2001, 2009, and 2013 (Table 1). The recorded duration of these 18 visits ranged from a maximum of 5.5 hours to less than an hour. When killer whale ecotype could reliably be identified (n = 5), mammal-eating transient pods were the most common visitors to SEFI (four of five), while offshore individuals were identified on a single occasion in 2009 when both offshores and transients were observed (Table 1).
White sharks aggregated at SEFI annually, where the observed number of predations by sharks peaked during October and November (Fig. 2B). During the fall surveys, a mean of 40 observed predations (±16 SD; N = 27 years) by sharks occurred annually on elephant seals and unidentified pinniped prey. In years when killer whales were not observed or were sighted 3 or more km from shore (N = 19), the distribution of predation events on pinnipeds peaked between mid-October and mid-November (Fig. 2B). In years when killer whales were sighted <3 km from shore (N = 9), this predation rate was depressed and truncated (Fig. S3 and Table S1). Therefore, annual predation rates on pinnipeds were significantly impacted when killer whale activity occurred at a distance threshold <3 km from the SEFI seal haul-out.
Predator-prey relationship between white sharks (Carcharodon carcharias) and elephant seals (Mirounga angustirostrous) altered by the presence of killer whales (Orcinus orca) at Southeast Farallon Island (SEFI). (A) Annual predation rate by C. carcharias as a function of mean fall (Sept. – Nov.) M. angustirostrous counts fit with a log-log regression line (dashed black line) showing confidence interval (dashed blue lines). Points are years where no flight response was detected, and triangles are the years in which a flight response was observed, near or before the peak of the C. carcharias season (≤November 2, inverted triangles), and near the end of the season (≥November 19, upright triangles). For comparison, an equivalent regression fit excluding flight years is shown with red dotted lines. (B) Seasonal C. carcharias kill rate as a function of the observed distance of O. orca activity to SEFI. The distribution of observed predations was reduced and truncated as a function of O. orca proximity to the common foraging ground (distance given in legend in km).
Overall, the observed annual rate of predation by sharks was positively correlated with the abundance of elephant seals present (R2 = 0.191, p = 0.023) (Fig. 2A). However, in years when killer whales occurred in close proximity to the island during or before peak shark abundance, the observed rate of predation by sharks deviated most from this relationship dropping 3.5 to 7-fold from the long-term average of 6.02 ± 2.4 predations per 100hrs (SD), to 1.73, 1.29, and 0.84 respectively in 1997, 2009, and 2013 (Fig. 2B). In 2000 by contrast, killer whales also occurred close to SEFI, but much later in the season (November 18; Table 1) resulting in no deviation from the expected annual predation rate (Fig. 2A).
Displacement of white sharks and flight response
Acoustic tag detections documented the abrupt and consistent flight of white sharks from SEFI in 2009, 2011, and 2013 (Figs 3 and SI). In the best-documented instance, killer whales from two separate pods (offshore and transient ecotypes; Table 1) arrived at SEFI on November 2, 2009, when 17 previously tagged white sharks were present. Killer whales were present at SEFI for just over 2.5 hours between 12:48 and 15:30 local time, remained on the western side of SEFI during approach and initiated three separate killing bouts on pinnipeds, then departed to the north. There were no observations of direct predation on white sharks, and all tagged animals were later confirmed alive through acoustic detections; still predations on untagged white sharks could not be ruled out.
The flight response of white sharks (Carcharodon carcharias) triggered by the presence of killer whales (Orcinus orca) at a common foraging site, Southeast Farallon Islands (SEFI). (A) Mean daily number of acoustic tagged C. carcharias detected (2007–2013; excluding 2009; shaded standard error) at Central California receivers colored by location: Tomales Point (green), Southeast Farallon Islands (orange and orange/yellow), Año Nuevo Island (blue), and Point Reyes (purple). (B) The number of tagged C. carcharias detected per day at each site (respectively colored) during the 2009 season showing the sudden departure of all tagged individuals from SEFI in response to O. orca (Nov 2) presence. Note the subsequent influx around Año Nuevo Island where the shaded orange area represents individuals present at SEFI during killer whale interactions. (C) Detections of each tagged shark at color-coded locations are shown along the horizontal timeline illustrating the abrupt departure from SEFI by tagged C. carcharias following O. orca presence (between vertical black lines) and subsequent avoidance. Solid orange diamonds indicate the western SEFI receiver while orange with yellow centers indicate the eastern receiver. (D) Precise receiver locations are indicated by the right corner of each solid diamond and the left corner of the yellow filled diamond.
Desertion of SEFI by all tagged sharks followed the foraging behavior of killer whales close to SEFI. Regular daily detections of 17 tagged animals at two stationary acoustic receivers moored on eastern and western sides of SEFI (SI) discontinued abruptly following the appearance of killer whales (Fig. 3). Overall, the mean number of white sharks detected per day at SEFI declined from a seasonal maximum to zero for the remainder of the season. Declines in detections followed a spatial gradient, immediately subsiding at the western receiver most proximal to killer whale observations, followed by a tapering of detections over the following hours at the eastern receiver (Fig. 3). Seven hours and 50 minutes following the event, no tagged sharks remained within receiver range at SEFI and 16 individuals (of 17 displaced tagged sharks) were not detected at SEFI again until the following season (July 2010 or later). One individual returned a week later (November 8), and was detected at SEFI three times over 73 minutes, before departing and being re-detected at Año Nuevo Island (ANI) on November 24.
Anomalous shark absences at SEFI for the remainder of the 2009 season resulted in influxes of displaced individuals at mainland aggregation sites. Within 2 to 13 days of departing SEFI following the killer whale disturbance at SEFI, seven tagged individuals relocated nearly 90 km to the south at ANI. Three individuals were redetected at Tomales Point for extended periods, before two of these continued to ANI (Fig. 3). These influxes at mainland sites resulted in daily totals of individual sharks detected at ANI increasing sharply from 4 day−1 on 2 November to 10 day−1 by 14 November and peaking at 16 day−1 by 23 November. In contrast, SEFI remained virtually shark-free for the remaining season. Three tagged individuals not initially present during the killer whale event were detected subsequently at SEFI, though for abbreviated durations (0.25, 1, and 11 hours, respectively) compared to mean SEFI residency periods of 35 days32. Two of these three sharks were then subsequently detected at mainland aggregation sites (Fig. 3).
Acoustic tag records provided a clear ‘signature’ for understanding and estimating flight responses of sharks relative to other killer whale occurrences at SEFI during years when sufficient active tags were present (see Table 1). The well-documented incident in 2009 was consistent with previous observations of cessation of seal predation at SEFI by white sharks following brief killer whale visits52,60. Acoustic tag records revealed two additional similar signatures at SEFI: November 20, 2011, with 10 tagged individual present, and October 31, 2013, with 3 tagged individuals present (see SI). Inclement weather resulting in poor visibility precluded visual confirmation of killer whales for the former event (no surveys that day) in 2011. On the 2013 occasion, 13 killer whales were observed during regular shark visual surveys from SEFI. In both cases following typical flight responses, no further tag detections were recorded at SEFI for the remainder of the season (see SI). Similarly, no further predations of pinnipeds by white sharks were observed during the remaining visual surveys in 2011 and 2013, and only a single predation during the remaining season in 2009 near Mid-Farallon Island, 3.4 km northwest of SEFI. In summary, a white shark flight response from SEFI related to killer whale occurrence was identified in four separate years, along with a fifth flight response with unconfirmed attribution (Table 1). While intensive observer survey data are lacking at TOM and ANI, no equivalent flight response was ever apparent in acoustic tagging data with continuous coverage between 2006 and 2013.
Ecological roles and context of killer whale-shark interactions
Transient killer whales were present in the two flight response years when ecotypes could be reliably determined (1997, 2009), whereas offshore individuals were identified in addition to transients in the 2009 disturbance. In determining the ecological relationship between white sharks and killer whales, understanding whether their interactions are defined by predator-prey or competitive aggression interactions depends on killer whale ecotype. Mammal-eating transient killer whales45 are direct competitors, but also pose a predatory threat as illustrated in the 1997 event52 (see introduction). Interactions with the offshore ecotype are potentially predatory, as well as competitive. Offshore killer whales are known to forage on teleosts and elasmobranchs, the latter forming a potentially important dietary component as evidenced by apical teeth worn flat, presumably from the abrasive shark skin50, and observations of repeated feeding on Pacific sleeper sharks, Somniosus pacificus61. Residents are likely a weak competitor (for teleosts) and potentially not a predation threat45,49. Whether white sharks might distinguish a predatory versus competitive threat remains unknown, but the result may be the same. Like predation pressure, interspecific competitive aggression can similarly drive behavior that reduces encounter rates, shape habitat use, and shift activity schedules8,62. Only one direct predation on white sharks by killer whales was ever confirmed on white sharks at SEFI52, yet white sharks vacating SEFI, effectively freed up potential pinniped resources for the killer whales and restricted white shark access to those resources.
Risk effects among top ocean predators
This study demonstrates the occurrence of risk effects among upper trophic level marine predators. The key interactions surrounding the phenomena remained cryptic and rarely observed despite intensive long-term visual surveys and multi-year continuous electronic tracking coverage. In the rare instances when both predators co-occurred at SEFI, antagonistic interactions between them resulted in the extended displacement of foraging white sharks via risk effects (Fig. 3), and in turn reduced local predation pressure on seals (Fig. 2). Despite exceptionally brief killer whale visits to SEFI (2.4–5 hr) during well-documented events near the peak white shark foraging season, the observed predation rate on pinnipeds by white sharks during those years decreased (Fig. 2). It is unlikely that killer whale predation on pinnipeds could compensate for the reduction in predation by white sharks following their displacement. Of the predation events observed at the surface in 15,383 hours of lighthouse surveys between 1987 and 2013, 912 were attributed to white sharks and only 5 events on 3 dates to killer whales.
Killer whales exert top-down effects in various systems by directly reducing meso-predator density through consumption1,4 as well as eliciting shifts in prey behaviors and distributions due to risk effects63,64. Similarly, large sharks can have a direct regulatory influence over their prey populations6,65,66, and induce food-safety tradeoffs67 including avoidance behavior68,69. This study suggests that intraguild interactions between killer whales and white sharks may result in cascading effects at lower trophic levels by reducing consumptive (and possibly non-consumptive) effects on elephant seals. Quantifying the indirect population-level effects killer whales induce on white sharks may have on elephant seals locally and regionally remains an important future direction. Northern elephant seals are undergoing rapid habitat expansion and population growth, following long-term human exploitation and extreme depletion59. Any population regulatory effects white sharks, killer whales, and their interactions have on elephant seals could become more significant as elephant seals approach an equilibrium level.
Occasional consumption of the highly-caloric liver of white sharks may confer ancillary energetic benefits to the killer whale. The fitness loss to white sharks from direct lethal interactions with killer whales is unambiguous. But avoidance behaviors in response to killer whale presence could also impact white shark fitness by restricting spatiotemporal access and activity to habitats that are sub-optimal or more competitive (more densely populated by conspecifics). Intimidation and predation risk pervasively affects entire populations, not just the individuals directly killed16. Potential consequences of displacement to white sharks should be evaluated within the ecological context of their migratory phenology. Fall-time aggregations and site fidelity of NEP white sharks along the central California coast immediately precede extensive offshore migrations to relatively oligotrophic waters32,60,70. Despite spending one third of their time in coastal California habitats, adults assimilate nearly half their protein from coastal foraging41. Concentrated energy acquisition during this coastal phase is stored in the oil-rich liver mass and is expended during long migrations (1000–3000 km) to seasonal offshore subtropical habitats43, where males increase diving activity extensively32,38. Disruptions of foraging prior to migration is known to negatively impact migratory performance in numerous long-distance migrating species30,71,72. Future efforts should aim to measure the impact and ecological implications of these risk effects on white shark fitness and elephant seal population dynamics.
Data Availibility
Data underlying this study and described above in Methods are archived (open access) at https://osf.io/b7su4/?view_only=f4f874c19e044ea5951a9ac355954d9f. These include (1) aggregated phenological acoustic tag detection data (2007–2013), and raw tag detection data (October 14 to November 30, 2009) (2) weekly census data of juvenile elephant seals at SEFI (1987–2013), and (3) killer whale observations at SEFI aggregated by month (1987–2013).
References
Estes, J. A., Tinker, M. T., Williams, T. M. & Doak, D. F. Killer Whale Predation on Sea Otters Linking Oceanic and Nearshore Ecosystems. Science 282, 473–476 (1998).
Estes, J. A. et al. Trophic Downgrading of Planet Earth. Science 333, 301–306 (2011).
Heithaus, M. R., Frid, A., Wirsing, A. J. & Worm, B. Predicting ecological consequences of marine top predator declines. Trends in Ecology & Evolution 23, 202–210 (2008).
Williams, T. M., Estes, J. A., Doak, D. F. & Springer, A. M. Killer Appetites: Assessing the Role of Predators in Ecological Communities. Ecology 85, 3373–3384 (2004).
Ritchie, E. G. & Johnson, C. N. Predator interactions, mesopredator release and biodiversity conservation. Ecology Letters 12, 982–998 (2009).
Baum, J. K. & Worm, B. Cascading top-down effects of changing oceanic predator abundances. Journal of Animal Ecology 78, 699–714 (2009).
Werner, E. E. & Peacor, S. D. A review of trait-mediated indirect interactions in ecological communities. Ecology 84, 1083–1100 (2003).
Creel, S. & Christianson, D. Relationships between direct predation and risk effects. Trends in Ecology & Evolution 23, 194–201 (2008).
Heithaus, M. R., Wirsing, A. J., Burkholder, D., Thomson, J. & Dill, L. M. Towards a predictive framework for predator risk effects: the interaction of landscape features and prey escape tactics. Journal of Animal Ecology 78, 556–562 (2009).
Laundré, J. W., Hernández, L. & Ripple, W. J. The landscape of fear: ecological implications of being afraid. Open Ecology Journal 3, 1–7 (2010).
Burkholder, D. A., Heithaus, M. R., Fourqurean, J. W., Wirsing, A. & Dill, L. M. Patterns of top-down control in a seagrass ecosystem: could a roving apex predator induce a behaviour-mediated trophic cascade? Journal of Animal Ecology 82, 1192–1202 (2013).
Clinchy, M., Zanette, L., Boonstra, R., Wingfield, J. C. & Smith, J. N. M. Balancing food and predator pressure induces chronic stress in songbirds. Proceedings of the Royal Society of London B: Biological Sciences 271, 2473–2479 (2004).
Mukherjee, S., Heithaus, M. R., Trexler, J. C., Ray-Mukherjee, J. & Vaudo, J. Perceived Risk of Predation Affects Reproductive Life-History Traits in Gambusia holbrooki, but Not in Heterandria formosa. PLOS ONE 9, e88832 (2014).
Allen, A. N., Schanze, J. J., Solow, A. R. & Tyack, P. L. Analysis of a Blainville’s beaked whale’s movement response to playback of killer whale vocalizations. Marine Mammal Science 1, 154–168 (2013).
Pace, M. L., Cole, J. J., Carpenter, S. R. & Kitchell, J. F. Trophic cascades revealed in diverse ecosystems. Trends in Ecology & Evolution 14, 483–488 (1999).
Preisser, E. L., Bolnick, D. I. & Benard, M. F. Scared to death? The effects of intimidation and consumption in predator-prey interactions. Ecology 86, 501–509 (2005).
Suraci, J. P., Clinchy, M., Dill, L. M., Roberts, D. & Zanette, L. Y. Fear of large carnivores causes a trophic cascade. Nature Communications 7, 10698 (2016).
Beckerman, A. P., Uriarte, M. & Schmitz, O. J. Experimental evidence for a behavior-mediated trophic cascade in a terrestrial food chain. PNAS 94, 10735–10738 (1997).
McCauley, D. J. et al. Marine defaunation: Animal loss in the global ocean. Science 347, 1255641 (2015).
Heithaus, M. R. Predator–prey and competitive interactions between sharks (order Selachii) and dolphins (suborder Odontoceti): a review. Journal of Zoology 253, 53–68 (2001).
Polis, G. A. & Holt, R. D. Intraguild predation: The dynamics of complex trophic interactions. Trends in Ecology & Evolution 7, 151–154 (1992).
Polis, G. A., Myers, C. A. & Holt, R. D. The Ecology and Evolution of Intraguild Predation: Potential Competitors That Eat Each Other. Annual Review of Ecology and Systematics 20, 297–330 (1989).
Palomares, F. & Caro, T. M. Interspecific Killing among Mammalian Carnivores. The American Naturalist 153, 492–508 (1999).
Glen, A. S. & Dickman, C. R. Complex interactions among mammalian carnivores in Australia, and their implications for wildlife management. Biological Reviews 80, 387–401 (2005).
Laundré, J. W., Hernández, L. & Altendorf, K. B. Wolves, elk, and bison: reestablishing the ‘landscape of fear’ in Yellowstone National Park, USA. Can. J. Zool. 79, 1401–1409 (2001).
Heithaus, M. R. & Dill, M. L. Does tiger shark predation risk influence foraging habitat use by bottlenose dolphins at multiple spatial scales? Oikos 114, 257–264 (2006).
Wirsing, A. J., Heithaus, M. R., Frid, A. & Dill, L. M. Seascapes of fear: evaluating sublethal predator effects experienced and generated by marine mammals. Marine Mammal Science 24, 1–15 (2008).
Lourenço, R., Penteriani, V., Rabaça, J. E. & Korpimäki, E. Lethal interactions among vertebrate top predators: a review of concepts, assumptions and terminology. Biol Rev 89, 270–283 (2014).
Block, B. A. et al. Tracking apex marine predator movements in a dynamic ocean. Nature 475, 86–90 (2011).
Dingle, H. Migration: the biology of life on the move. (Oxford University Press, USA, 1996).
Newton, I. The migration ecology of birds. (Academic Press, 2010).
Jorgensen, S. J. et al. Philopatry and migration of Pacific white sharks. Proc R Soc B 277, 679–688 (2010).
Hoyos-Padilla, E. M., Klimley, A. P., Galván-Magaña, F. & Antoniou, A. Contrasts in the movements and habitat use of juvenile and adult white sharks (Carcharodon carcharias) at Guadalupe Island, Mexico. Animal Biotelemetry 4, 14 (2016).
Le Beouf, B. J. & Laws, R. M. Elephant Seals: An Introduction to the Genus. In Elephant Seals: Population Ecology, Behavior, and Physiology 1–26 (University of California Press, 1994).
Klimley, A. P. et al. The hunting strategy of white sharks (Carcharodon carcharias) near a seal colony. Mar Biol 138, 617–636 (2001).
Anderson, S. D., Becker, B. H. & Allen, S. G. Observations and Prey of White Sharks, Carcharodon carcharias, at Point Reyes National Seashore: 1982-2004. California Fish and Game 94, 33 (2008).
Brown, A. C., Lee, D. E., Bradley, R. W. & Anderson, S. Dynamics of White Shark Predation on Pinnipeds in California: Effects of Prey Abundance. Copeia 2010, 232–238 (2010).
Jorgensen, S. J. et al. Eating or Meeting? Cluster Analysis Reveals Intricacies of White Shark (Carcharodon carcharias) Migration and Offshore Behavior. PLoS ONE 7, e47819 (2012).
Long, D. J. & Jones, R. E. White shark predation and scavenging on cetaceans in the eastern North Pacific Ocean. Great white sharks: the biology of Carcharodon carcharias 293–307 (1996).
Klimley, A. P. The areal distribution and autoecology of the white shark, Carcharodon carcharias, off the west coast of North America. Memoirs of the Southern California Academy of Sciences 9, 15–40 (1985).
Carlisle, A. B. et al. Using stable isotope analysis to understand the migration and trophic ecology of northeastern Pacific white sharks (Carcharodon carcharias). PLoS ONE 7, e30492 (2012).
Carey, F. G. et al. Temperature and activities of a white shark, Carcharodon carcharias. Copeia 1982, 254–260 (1982).
Raye, G. D., Jorgensen, S. J., Krumhansl, K., Ezcurra, J. M. & Block, B. A. Travelling light: white sharks (Carcharodon carcharias) rely on body lipid stores to power ocean-basin scale migration. Proc. R. Soc. B 280, 20130836 (2013).
Herman, D. P. et al. Feeding ecology of eastern North Pacific killer whales Orcinus orca from fatty acid, stable isotope, and organochlorine analyses of blubber biopsies. Mar Ecol Prog Ser 302, 275–291 (2005).
Ford, J. K. B. et al. Dietary specialization in two sympatric populations of killer whales (Orcinus orca) in coastal British Columbia and adjacent waters. Can J Zool 76, 1456–1471 (1998).
Morin, P. A. et al. Complete mitochondrial genome phylogeographic analysis of killer whales (Orcinus orca) indicates multiple species. Genome Res. 20, 908–916 (2010).
de Bruyn, P. J. N., Tosh, C. A. & Terauds, A. Killer whale ecotypes: is there a global model? Biological Reviews 88, 62–80 (2013).
Hoelzel, A. R. & Moura, A. E. Resource specialisation and the divergence of killer whale populations. Heredity (Edinb) 115, 93–95 (2015).
Ford, J. K. B., Ellis, G. M. & Balcomb, K. C. Killer whales: The natural history and genealogy of Orcinus orca in British Columbia and Washington. (Univ of Washington Pr, 2000).
Ford, J. K. et al. Shark predation and tooth wear in a population of northeastern Pacific killer whales. (2011).
Simberloff, D. & Dayan, T. The Guild Concept and the Structure of Ecological Communities. Annual Review of Ecology and Systematics 22, 115–143 (1991).
Pyle, P., Schramm, M. J., Keiper, C. & Anderson, S. D. Predation on a white shark (Carcharodon carcharias) by a killer whale (Orcinus orca) and a possible case of competitive displacement. Marine Mammal Science 15, 563–568 (1999).
Best, P. B., Meÿer, M. A. & Lockyer, C. Killer whales in South African waters—a review of their biology. African Journal of Marine Science 32, 171–186 (2010).
Chapple, T. K. et al. A novel application of multi-event modeling to estimate class segregation in a highly migratory oceanic vertebrate. Ecology 97, 3494–3502 (2016).
Sydeman, W. J. & Allen, S. G. Pinniped population dynamics in central California: correlations with sea surface temperatures and upwelling indices. Marine Mammal Science 15, 446–461 (1999).
Pyle, P. & Gilbert, L. Occurrence patterns and trends of cetaceans recorded from Southeast Farallon Island, California, 1973 to 1994. Northwestern Naturalist 1–8 (1996).
Wood, S. N. Generalized Additive Models: An Introduction with R, Second Edition. (CRC Press, 2017).
Chapple, T. K. et al. A first estimate of white shark, Carcharodon carcharias, abundance off Central California. Biol lett 7, 581–583 (2011).
Lowry, M. S. et al. Abundance, Distribution, and Population Growth of the Northern Elephant Seal (Mirounga angustirostris) in the United States from 1991 to 2010. Aquatic Mammals 40, 20–31 (2014).
Weng, K. C. et al. Migration and habitat of white sharks (Carcharodon carcharias) in the eastern Pacific Ocean. Mar Biol 152, 877–894 (2007).
Deecke, V. B., Ford, J. K. B. & Slater, P. J. B. The vocal behaviour of mammal-eating killer whales: communicating with costly calls. Animal Behaviour 69, 395–405 (2005).
Peiman, K. S. & Robinson, B. W. Ecology and Evolution of Resource-Related Heterospecific Aggression. The Quarterly Review of Biology 85, 133–158 (2010).
Jefferson, T. A., Stacey, P. J. & Baird, R. W. A review of Killer Whale interactions with other marine mammals: predation to co-existence. Mammal Review 21, 151–180 (1991).
Breed, G. A. et al. Sustained disruption of narwhal habitat use and behavior in the presence of Arctic killer whales. PNAS 114, 2628–2633 (2017).
Wirsing, A. J., Heithaus, M. R. & Dill, L. M. Tiger shark (Galeocerdo cuvier) abundance and growth in a subtropical embayment: evidence from 7 years of standardized fishing effort. Mar Biol 149, 961–968 (2006).
Heithaus, M. R., Wirsing, A. J. & Dill, L. M. The ecological importance of intact top-predator populations: a synthesis of 15 years of research in a seagrass ecosystem. Mar. Freshwater Res. 63, 1039–1050 (2012).
Wirsing, A. J., Heithaus, M. R. & Dill, L. M. Fear factor: do dugongs (Dugong dugon) trade food for safety from tiger sharks (Galeocerdo cuvier)? Oecologia 153, 1031–1040 (2007).
Wirsing, A. J., Cameron, K. E. & Heithaus, M. R. Spatial responses to predators vary with prey escape mode. Animal Behaviour 79, 531–537 (2010).
Moxley, J. H. The Abundance and Behavioral Ecology of Cape Cod Gray Seals Under Predation Risk From White Sharks. (2016).
Boustany et al. Expanded niche for white sharks. Nature 415, 35–36 (2002).
Fontaine, M. Physiological Mechanisms in the Migration of Marine and Amphihaline Fish. Advances in Marine Biology 13, 241–355 (1976).
Alerstam, T., Hedenström, A. & Åkesson, S. Long-distance migration: evolution and determinants. Oikos 103, 247–260 (2003).
Acknowledgements
All research activities were conducted under permits and authorizations granted by the Greater Farallones National Marine Sanctuary, the California department of Fish and Wildlife, U.S. department of fish and Wildlife, and U.S. National Park Service, in accordance with relevant guidelines and regulations. We would like to thank numerous individuals who assisted in field work, lab work, data processing, editing and inspiration including J. Moskito, A. Shulman-Janiger, J. Benson, J. Barlow, R. Elliot, A. Carlisle, C. Perle, A. Brown, C. Logan, J. Cornelius, B. Becker, S. McAfee, C. Farwell, C. Harrold, J. O’Sullivan, J. Ganong, A. Swithenbank, M. Castleton, A.P. Klimley, T. Brandt, R. Repass and many more. We thank Wyliecat, R. Repass, T. O’Leary, Bodega Marine Lab, and Farallon Patrol for vessel assistance. We also thank Farallon Islands National Wildlife Refuge staff, and biologists who collected data over several decades. The research on SEFI was made possible by a cooperative agreement with the United States Fish and Wildlife Service. Financial support was provided by the Monterey Bay Aquarium, Sloan, Moore and David and Lucile Packard Foundations and Stanford University. Additional funding for Point Blue’s Farallon research was provided by the Shark Trust, Baker Trust, and the Bently, Marisla, Mead, Campini, Bernice Barbour, Kimball, Volgenau, and RHE Charitable Foundations, and by individual donors. FF acknowledges the Lenfest Ocean Program and the Bertarelli Foundation. For JT and RWB this is Point Blue Contribution # 2051.
Author information
Authors and Affiliations
Contributions
S.J.J., S.A., B.A.B., R.W.B., T.C. and J.R.T. designed the study. S.J.J., S.A., B.A.B., R.W.B., T.C., P.K. and J.R.T. conducted field work. S.J.J., F.F. and J.H.M. analyzed the data and drafted the figures. S.J.J. wrote the manuscript. All authors contributed data and edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jorgensen, S.J., Anderson, S., Ferretti, F. et al. Killer whales redistribute white shark foraging pressure on seals. Sci Rep 9, 6153 (2019). https://doi.org/10.1038/s41598-019-39356-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39356-2
This article is cited by
-
First detailed footage of orcas hunting great white sharks
Nature Africa (2022)
-
Premières images détaillées d'orques chassant des grands requins blancs
Nature Africa (2022)
-
North Pacific warming shifts the juvenile range of a marine apex predator
Scientific Reports (2021)
-
New insights into the trophic ecology of the scalloped hammerhead shark, Sphyrna lewini, in the eastern tropical Pacific Ocean
Environmental Biology of Fishes (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.