Abstract
Among the genetic variations in the monoamine oxidase A (MAOA) gene, upstream variable number tandem repeats (uVNTRs) of the promoter have been associated with individual differences in human physiology and aggressive behaviour. However, the evidence for a molecular or neural link between MAOA uVNTRs and aggression remains ambiguous. Additionally, the use of inconsistent promoter constructs in previous studies has added to the confusion. Therefore, it is necessary to demonstrate the genetic function of MAOA uVNTR and its effects on multiple aspects of aggression. Here, we identified three MAOA alleles in Koreans: the predominant 3.5R and 4.5R alleles, as well as the rare 2.5R allele. There was a minor difference in transcriptional efficiency between the 3.5R and 4.5R alleles, with the greatest value for the 2.5R allele, in contrast to existing research. Psychological indices of aggression did not differ among MAOA genotypes. However, our electroencephalogram and electrocardiogram results obtained under aggression-related stimulation revealed oscillatory changes as novel phenotypes that vary with the MAOA genotype. In particular, we observed prominent changes in frontal γ power and heart rate in 4.5R carriers of men. Our findings provide genetic insights into MAOA function and offer a neurobiological basis for various socio-emotional mechanisms in healthy individuals.
Similar content being viewed by others
Introduction
Aggression is a ubiquitous phenomenon that arises from anger or antipathy and often results in hostile or violent behaviour in humans. Pathological aggression related to antisociality or violence is highly heritable1,2,3,4,5, and previous work estimated that genetic factors account for approximately 50% of the variation in aggression6. Among candidate genes for aggression, mutations, deletions, single-nucleotide polymorphisms (SNPs), and variable number of tandem repeats (VNTRs) in the X-linked monoamine oxidase A (MAOA) gene were the first known genetic deficiencies in animal and human models7,8,9. The gene product monoamine oxidase A, which is expressed in specific cells of the human brain and peripheral tissues10,11,12, metabolizes monoamines, such as the neurotransmitters serotonin, norepinephrine, and dopamine, which are involved in the regulation of emotions13,14,15.
The 30-base pair (bp) upstream variable number tandem repeat (uVNTR) polymorphism of the MAOA promoter region is a major focus of studies on genetic associations with numerous phenotypes of aggression, such as antisocial behaviour16 and impulsivity17, as well as neuropsychiatric disorders including alcoholism18,19. In vitro gene fusion and transfection assays initially indicated that MAOA uVNTR variants may cause differential transcription of the MAOA mRNA; the 2-repeat (2R) and 3-repeat (3R) low-MAOA (L-MAOA) alleles are associated with lower transcriptional efficiency than the 4-repeat (4R) high-MAOA (H-MAOA) allele, with corresponding lower enzyme expression and higher predicted downstream neurotransmitter levels20,21,22,23. In addition, studies showing that the aggressive propensity of L-MAOA carriers becomes more apparent following traumatic experiences, such as childhood maltreatment, underscore the importance of gene × environment interactions in modulating the role of MAOA in aggression16,24,25,26. However, conflicting results suggest that the MAOA uVNTR alone or its interactions with the environment have no or opposing relationships with aggressive phenotypes27,28,29,30,31. In addition, studies have shown that MAOA uVNTR variants do not correspond to monoamine oxidase A levels in post-mortem brains with brain monoamine oxidase A activity in healthy men32,33,34, and the in vitro transcriptional efficiency of monoamine oxidase A was inconsistent and depended on the transfected constructs35. These findings suggest that factors such as post-translational factors or the modulation of protein levels and activity, and possibly compensatory mechanisms between the different neurotransmitter systems regulated by MAOA, may work together in modulating the role of MAOA uVNTR genotype on aggression.
While studies using positron emission tomography (PET) and [11C]clorgyline did not find an association between MAOA genotype and brain MAOA activity in healthy men32,33, studies using functional magnetic resonance imaging (fMRI) support an effect of the MAOA genotype on aggression-related functional changes in the nervous system. The MAOA genotype has been associated with the level or distribution of neural activity in cortical regions of the brain, such as the dorsal anterior cingulate cortex or the ventromedial prefrontal cortex, in response to Go/No-Go tasks or programmes including social exclusion36,37. In addition, heathy L-MAOA carriers showed prominent structural and functional changes in the corticolimbic network, which plays a crucial role in regulating emotional states during the recall of negative events17. Recent work showed that L-MAOA carriers are more susceptible to pharmacological regulation by serotonin, with changes in the corticolimbic network, indicating a possible link between the serotoninergic system and MAOA genotype38. The effects of MAOA genotype on the dopaminergic system remain controversial. For example, the dopamine levels in response to a violent video was not dependent on MAOA genotype39. As previously indicated, the conflicting results obtained by different studies could be due to the broad role of MAOA in modulating different neurotransmitter systems, the heterogeneity in measuring neural function and human behaviour, the in vitro system used to measure the effect of uVNTR on MAOA expression, and to the effect of gene × environmental interactions40,41,42,43. The results of such studies in women are also perplexing. It remains unclear whether MAOA is an X-inactivated gene, making it challenging to evaluate enzyme expression or activity in heterozygous genotypes44.
Beyond the conflicting results, historical evidence points towards an association of MAOA genotype to neural function in aggressive behaviour. Thus, in the present study, we investigated the effects of the MAOA genotype on multiple aspects of aggression, including neural and cardiac activities. In addition, given the fact that allelic distribution varies across ethnic/racial groups, we characterized the MAOA genotype distribution in the Korean population for the first time22. Transcriptional efficiency was assessed using in vitro reporter gene assays based on full MAOA promoter sequences to more accurately test the transcriptional contribution of the uVNTR. We also investigated the functional relationship between MAOA genotypes and aggression by analysing electroencephalogram (EEG)- and electrocardiogram (ECG)-based neurobiological responses to commonly encountered aggression stimuli in both men and women. In particular, EEG and ECG are useful tools for studying differences in neural activity because they are capable of measuring real-time responses and are highly accessible, with fine temporal resolution across the spectrum range of EEG and the direct autonomic reactivity range of ECG. Moreover, many EEG and ECG studies have shown that specific forms of oscillatory activity are associated with antisocial behaviour or aggressive tendencies45,46,47,48,49,50,51,52,53,54,55,56. In particular, frontal EEG asymmetry, which is a representative functional feature of aggression45,46, is affected by dopamine regulation57. Nevertheless, few studies of the relationship between MAOA genotypes and aggression have used EEG and ECG approaches in a pool of healthy adults.
Results
Genetic variation of MAOA in the Korean population
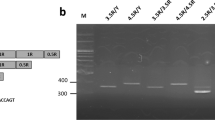
To characterize genetic variation of MAOA in the Korean population, we identified MAOA uVNTR genotypes and allelic frequencies. Although many studies have followed the original method, which classifies alleles as 2R, 3R, 4R, and 5R22,23,58, some groups have suggested a new classification method for the sequence repeats, namely, 2.5R, 3.5R, 4.5R, and 5.5R59,60,61,62. The difference lies in the first 15 bp half-repeat sequence (−1141/−1127 bp), which is next to the repeated 30 bp sequence (−1262/−1142 bp)63; the redefined classification includes this sequence (Fig. 1a). Seven different MAOA genotypes were detected in our population, including two genotypes in men and five genotypes in women (Table 1). The polymerase chain reaction (PCR) products of the seven observed types of MAOA uVNTRs are shown in Figs 1b and S7a. Among the three MAOA uVNTR alleles detected, the 3.5R allele was the most prevalent (Table 1). The 2.5R allele appeared only in heterozygous genotypes, such as 2.5R/3.5R and 2.5R/4.5R in women, and at a very low frequency. The genotype distribution in our study was not in Hardy-Weinberg equilibrium (χ2 = 15.59, df = 4, χ2/df = 3.90, p (χ2 > 15.59) = 0.0036).
Genomic structure of the MAOA uVNTR polymorphism. (a) Nucleotide sequence alignment of the MAOA uVNTR alleles 2.5R, 3.5R, and 4.5R. 1R of each MAOA uVNTR allele consisted of a 30 bp “ACCGGCACCGGCACCAGTACCCGCACCAGT” sequence, followed by the first half-sequence of “ACCGGCACCGGCACC” in the last 0.5R. (b) Visualization of the different MAOA genotypes of the Korean population in 3% agarose gel. Lanes 1 and 2 are 3.5R/Y and 4.5R/Y from men; lanes 3, 4, 5, 6 and 7 are 3.5R/3.5R, 4.5R/4.5R, 2.5R/3.5R, 2.5R/4.5R, and 3.5R/4.5R from women, respectively. The full gel is presented in Fig. S7a.
Functional analysis of the promoter activity of MAOA uVNTR alleles
To characterize MAOA uVNTR function in vitro, the transcriptional efficiency of the observed alleles was assessed in SH-SY5Y and JAR cells using reporter gene constructs containing the MAOA promoter region fused to the firefly luciferase gene. For each promoter construct, we used a full 1.47 kb upstream fragment from −1470 to +34 bp63, which encompasses three different MAOA uVNTR alleles (2.5R, 3.5R, and 4.5R) containing transcription-regulatory regions of MAOA (Fig. 2a). Reporter gene expression levels gradually decreased with each allele in the order of 2.5R, 3.5R, and 4.5R in both SH-SY5Y and JAR cells (Fig. 2b,c). Statistically significant differences were detected between the negative control condition and each allele (F, 65.41; p < 0.0001 for 2.5R; p, 0.0002 for 3.5R; and p, 0.0261 for 4.5R in SH-SY5Y cells; F, 20.14; p < 0.0001 for 2.5R; p, 0.0001 for 3.5R; p, 0.0239 for 4.5R in JAR cells). In a comparison among three alleles, 3.5R showed significantly higher MAOA transcriptional efficiency than 4.5R (F, 65.41, p, 0.0365), and the 2.5R allele showed the highest value, with statistical significance in SH-SY5Y cells (F, 65.41, p < 0.0001). There was also a significant difference in MAOA transcriptional efficiency between the 2.5R and 4.5R alleles in JAR cells (F, 20.14, p, 0.0003). Since the transcriptional efficiency decreased as the MAOA uVNTR sequence increased in length, we performed additional reporter gene assays with nine alleles spanning 2R-6R to determine the length-dependent effect of MAOA uVNTR on transcription. The PCR products of the promoters carrying each of the MAOA uVNTR allele-fused constructs, along with the corresponding observed MAOA genotypes, are shown (Figs S1a,b and S7b,c). Although the range of absolute values differed, the pattern of transcriptional efficiency for the 2.5R, 3.5R, and 4.5R alleles was preserved (F, 191.8; p < 0.0001 between 2.5R and 3.5R; p < 0.0001 between 2.5R and 4.5R; and p < 0.0351 between 3.5R and 4.5R in SH-SY5Y cells; F, 57.74; p < 0.0001 between 2.5R and 3.5R; p, 0.0345 between 2.5R and 4.5R; and p, ns between 3.5R and 4.5R in JAR cells); in addition, the 5.5R allele, which was found in other ethnic/racial groups20,22, showed a relatively higher value than those of the 3.5R and 4.5R alleles (Fig. S1c–f). However, no length-dependent effect of the MAOA uVNTR on transcription was observed. Nevertheless, our results showed higher transcriptional efficiency for the shorter uVNTR variants, 2.5R and 3.5R, that was more evident in SH-SY5Y cell line as compared to JAR. Due to the low frequency of the 2.5R allele in the population, we focused the next experiments on subject carrying the 3.5R and 4.5R alleles.
Transcriptional efficiency of MAOA promoter fusions. (a) Schematic diagram of the 1.5 kb promoter organization with the 4.5R allele. Sites from −1262 to −1127 are for the position of MAOA uVNTR alleles and sites −1470 to +35 are for cloning into the promoter fusion vector. The sites for the core promoter and three GRE/AREs (grey blocks) are also shown. GRE, Glucocorticoid response element; ARE, Androgen response element. (b,c) Reporter gene assays. MAOA promoter constructs carrying the 2.5R, 3.5R, or 4.5R alleles fused to firefly luciferase were co-transfected with a plasmid containing Renilla luciferase into SH-SY5Y cells and JAR cells. Empty pcDNA vector was used as a negative control (−). Data are presented as the ratio of firefly to Renilla luciferase activity (mean ± SEM) from one of three independent experiments with 4–8 wells. *, **, and *** above the horizontal lines indicate statistically significant differences among MAOA uVNTR alleles at p < 0.05, 0.01, and 0.001, respectively, by one-way ANOVA with Tukey’s post hoc test; * and ** above the bar graph indicate statistically significant differences between the negative control group and MAOA uVNTR alleles at p < 0.05 and 0.01, respectively, by one-way ANOVA with Tukey’s post hoc test.
Effects of MAOA genotypes on questionnaire scores
To test the relationship between MAOA genotypes and psychological aggression and to compare hemi- and homozygous 3.5R and 4.5R carriers, the scores of three self-report questionnaires (the Buss-Durkee Hostility Inventory (BDHI), the Buss-Perry Aggression Questionnaire (BPAQ), and the Peer Conflict Scale (PCS)) were analysed. No significant differences in the mean scores of either the 3.5R/Y and 4.5R/Y genotypes in men or the 3.5R/3.5R and 4.5R/4.5R genotypes in women were found on any of the questionnaires (Fig. S2). Similarly, no significant differences in the mean BPAQ and PCS subcategory scores were observed between genotypes. Therefore, the effect of the MAOA genotype on psychological aggression was negligible in our study population.
Effects of MAOA genotypes on EEG response
We analysed EEG-based neurobiological responses to aggression-inducing stimuli to examine associations between MAOA allelic variation and neural or functional differences. When aggression-inducing stimuli were applied through the F8 channel, the power spectral density was displayed as a heat map according to the frequency interval (Fig. 3a). We assessed significant difference between the genotypes during S only for the EEG values with significant differences between NS and S in the analysis. Mean square error (MSE) analysis, which indicates the oscillatory variability of the time (neutral stimulus (NS) and stimulus (S)) and frequency (α, β, γ, θ, and δ waves) intervals, revealed that the MSE value of the γ wave in the F8 channel during S was significantly larger than that during NS in both men and women, except for the 3.5R/3.5R group (t, 2.425, p, 0.0416 for 3.5R/Y; t, 6.125, p < 0.0001 for 4.5R/Y; t, 3.692, p, 0.018 for 4.5R/4.5R; and t, 3.711, p, 0.0017 for heterozygous group) (Fig. 3b,c). Moreover, the MSE value of the γ wave in the F8 channel also differed significantly between MAOA genotypes during S in men; specifically, the MSE values of the 4.5R/Y group were larger than those of the 3.5R/Y group (t, 2.375, p, 0.0408). However, for the relative power value of the γ wave in the F8 channel during S, the values did not differ significantly by MAOA genotype both in men and women (Fig. 3d,e). In addition, for the other four frequency intervals in the F8 channel and the five frequency intervals in the Fp2 channel, the MSE values during S did not show significant differences between genotypes (Figs. S3 and S4). For the relative power value of the five frequency intervals in the Fp2 and F8 channels, significant differences between genotypes were not observed as well (Figs. S3 and S4). Therefore, when aggression-inducing stimuli were applied, the oscillatory change in the γ wave in the F8 channel was greater in hemizygous 4.5R carriers than in hemizygous 3.5R carriers.
Comparisons of the EEG γ response at F8 across MAOA genotypes. (a) Power spectral density of the γ wave at F8. The panels represent power spectral density by time (10–117.75 s) and frequency (30–90 Hz) intervals across MAOA genotypes. At the bottom of each panel, the first arrow represents the start of the NS (19 s), and the second arrow represents the start of S (stimulus, 28 s). (b,c) represent the MSE value of the γ wave at F8 during NS and S between MAOA genotypes in men and women, respectively. (d,e) represent the relative power value of the γ wave at F8 during NS and S between MAOA genotypes in men and women, respectively. All values are represented as the means ± SEM. The sample size for EEG measurement was 84, including 36 men (21 for 3.5R/Y, 15 for 4.5R/Y) and 48 women (23 for 3.5R/3.5R, 5 for 4.5R/4.5R, and 20 for the heterozygous group). *, **, and *** above the horizontal lines indicate statistically significant differences between MAOA genotypes at p < 0.05, 0.01, and 0.001, respectively, by repeated measures of two-way ANOVA with Bonferroni post hoc test; * and ** above the graph bars indicate statistically significant differences between NS and S at p < 0.05 and 0.01, respectively, by repeated measures of two-way ANOVA with Bonferroni post hoc test.
Effects of MAOA genotype on ECG responses
We analysed the heart rates (HRs) from the ECG results to investigate autonomic reactivity related to aggression according to the MAOA genotype. Since low HR in the resting state is a marker for aggression48,64,65,66,67, we assessed significant differences between the genotypes during NS only for the average HR values, with significant differences between NS and S in the analysis. The average HR was significantly lower for 4.5R/Y than for 3.5R/Y during NS (t, 2.604, p, 0.0234), and the values were significantly higher in both genotypes during NS than during S in men (Fig. 4a). However, the average HR was not significantly different either between genotypes or between scenes (NS vs. S) in women (Fig. 4b).
Comparison of ECG across MAOA genotypes. ECG signals were used to calculate HR (unit: BPM) and were converted to values relative to baseline. (a,b) represent average HR during NS and S between MAOA genotypes in men and women, respectively. (c,d) represent ΔHR during NS and S between MAOA genotypes in men and women, respectively. (e,f) represent variance in HR during NS and S between MAOA genotypes in men and women, respectively. All values are represented as the means ± SEM. The ECG sample size was 70, including 30 men (18 for 3.5R/Y, 12 for 4.5R/Y) and 40 women, (20 for 3.5R/3.5R, 3 for 4.5R/4.5R, and 17 for the heterozygous group). * and ** above the horizontal line indicate statistically significant differences between MAOA genotypes at p < 0.05 and 0.01, respectively, by repeated measures of two-way ANOVA with Bonferroni post hoc test; *, **, and *** above the graph bar indicate statistically significant differences between NS and S at p < 0.05, 0.01, and 0.001, respectively, by repeated measures of two-way ANOVA with Bonferroni post hoc test.
Since ΔHR and HR variance indicate the degree of change in autonomic reactivity, we assessed significant differences between the genotypes during S only for the values, with significant differences between NS and S in the analysis. ΔHR was significantly higher during S than during NS in all genotypes (t, 4.975 p < 0.0001 for 3.5R/Y; t, 6.929 p < 0.0001 for 4.5R/Y; t, 8.333, p < 0.0001 for 3.5R/3.5R; t, 3.716, p, 0.0020 for 4.5R/4.5R; and t, 7.537, p < 0.0001 in the heterozygous group) (Fig. 4c,d). In particular, ΔHR was significantly higher in the 4.5R/Y group than in the 3.5R/Y group during S in men (t, 2.364, p, 0.0426) (Fig. 4c). However, no such difference in ΔHR between genotypes was observed in women (Fig. 4d). In addition, the variance of HR was not significantly different either between genotypes or between scenes both in men and women (Fig. 4e,f). Taken together, the ΔHR by aggression-inducing stimuli was greater in hemizygous 4.5R carriers than in hemizygous 3.5R carriers, while average HR by neutral stimuli was lower in hemizygous 4.5R carriers than in hemizygous 3.5R carriers in men.
Discussion
MAOA has long been a gene of particular interest for studying the association between genetic variation and behavioural or neurobiological characteristics associated with aggression16,17,24,25,26,36,37,38,68. However, despite numerous publications on this matter, studies are still needed to investigate whether some relationships are present in all racial groups69. Our research found that the frequencies of the common 3.5R and 4.5R alleles in Koreans, 63.8% and 34.8%, respectively, differed from the observed frequencies of 35–39% and 59–63% for 3R and 4R, respectively, in Caucasians (assumed to be equivalent to 3.5R and 4.5R in our classification)22,60,70. The allele frequency observed here was similar to those previously reported for Chinese (54.5–64.7% and 34.8–44.2% for 3R and 4R, respectively)35,69,71 and Japanese (62% and 38% for 3R and 4R, respectively)72 individuals. Although not based on sequence analyses, these data provide a basis for the representative genetic distribution of MAOA uVNTR in Asians. We verified the allele frequency difference in MAOA uVNTR by race and geographic location22,70,73,74. Previous studies showed that 2R was rare in Asians35,71,72 and that 5R was found only in Caucasians22,60. In our study, we observed 2.5R at a very low frequency, which corresponds to 2R in previous studies.
The molecular basis of the difference in aggression between MAOA genotypes has largely been pursued in terms of expression levels using in vitro systems. These studies began with Sabol’s initial finding in vitro, which provided evidence for classifying MAOA genotypes as H-MAOA and L-MAOA22. The results for the most frequent alleles, which demonstrated lower promoter activities for 3R than for 4R, were replicated by others20,23, but these studies also showed inconsistent results for the relative promoter activity of the 5R allele, which were not noted due to its rare frequency20,22. We found that the differences in promoter activity among the three alleles were inconsistent with previous findings20,22,23; in our results, the difference between the 3.5R and 4.5R alleles was minor and dependent on the cell line used, with the 2.5R allele showing the highest transcriptional efficiency. Although our results are consistent with one previous report, this result was not discussed further due to the use of glioblastoma cell lines rather than neuroblastoma35. These discrepancies could be attributed to the different MAOA-luciferase reporter gene constructs used and to the length of the promoter studied. Most studies include the MAOA promoter fragment up to 1.3 kb upstream of the transcription start site, including the core transcriptional regulatory regions of MAOA, such as the four known Sp1 sites (−239/−14 bp), the SRY-binding site (−117/−111 bp), and the GR/AR response element (−289/−275 bp)63,75,76,77,78,79. However, it remains unclear whether an upstream region beyond 1.3 kb affects the transcriptional efficiency of MAOA uVNTR alleles; in particular, the results obtained using extended sequences (−1470/+34 bp, in our study; −1396/0 bp35) reached conclusions that differed from those of other studies (−1308/−6 bp22; −1336/−64 bp23; −1370/−7 bp20) in terms of the difference between the 3R and 4R alleles. Similar results have been reported regarding the 5R allele (−1308/−6 bp22 vs. −1370/−7 bp20; −1470/+34 bp, in our study; –1396/0 bp35). Additionally, the GR/AR response element (−1350/−1335 bp) and its upstream region in the MAOA promoter affect basal luciferase activity but do not respond to glucocorticoids or androgens78. Our results show that, using the full promoter under in vitro conditions, the promoter activity is higher in shorter uVNTR variants, although there is no a length dependent effect for variants ranging from 2.5R up to 6R. This finding suggests that the MAOA uVNTR may not be the only cis-regulatory element modulating MAOA transcriptional efficiency. For instance, epigenetic modification of the genome, such as DNA methylation, can be proposed as a potential mechanism that contributes to MAOA expression32,33. Indeed, MAOA promoter methylation in white blood cells has been associated with brain MAOA activity measured by PET. Future studies of MAOA promoter methylation and transcriptional efficiency will provide more reliable results and mechanisms related to the role of the MAOA uVNTR in transcription.
Beyond the ongoing studies to decipher the molecular mechanisms modulating MAOA levels, many studies have demonstrated functional phenotypes associated with H-MAOA and L-MAOA carriers17,36,37,38. We also discovered that EEG and ECG responses related to aggression differed with respect to the MAOA genotype. We found that hemizygous 4.5R carriers showed greater oscillatory variability in the right frontal γ wave in response to aggression-inducing stimuli. The EEG signal in the frontal region has mainly been studied with respect to the asymmetrical activity pattern of the α wave, which is related to specific types of aggression, such as positive or negative affect45,46,49. However, no effects were observed for the α wave of the right hemisphere in our results. Therefore, we can raise the possibility of other candidates. In early investigation, enhanced cortical δ wave activity was associated with violence or antisociality50,80. Studies of the γ wave in relation to aggression are relatively rare, but some reports suggested that the frontal γ wave shows increased activity following unpleasant stimuli81 and is also involved in impulse control in relation to addiction82. Thus, the greater γ responses of 4.5R carriers in our results might be associated with impulsive tendencies. Even though many neuroimaging studies have also shown that neural activity in the prefrontal/frontal cortex or corticolimbic neural circuits are related to aggressive behaviours, such as impulsivity83,84,85,86, this evidence is insufficient to causally link individual brain areas to specific EEG frequencies, including the γ wave. Moreover, variations in the γ wave might be induced during visual processing. Growing evidence indicates that this oscillatory variation can be driven by micro-saccadic movement or cross-frequency coupling of θ waves caused by this movement87,88. First, transient increases in γ power due to micro-saccadic movement generally occur in the 200–300 ms range88. However, the latency and duration of this effect have differed among some MEG studies89,90,91. Second, there is neural communication related to induced γ oscillation in visual cortical areas87. However, whether γ synchronization occurs between the visual cortex and the prefrontal/frontal cortex is unclear. Future research on γ dynamics caused by visual cues should be supplemented with eye-tracking systems or correction methods, such as averaging trials. Taken together, our results suggest that the level of oscillatory change in the frontal γ wave under aggression-inducing stimuli can be another functional phenotype that varies with the MAOA genotype in relation to aggression.
Furthermore, we discovered differences in ECG responses to aggression-inducing stimuli between MAOA genotypes. Previous studies proposed that a low HR during resting state is among the best-replicated physiological characteristics of aggression48,64,65,66,67, which may also indicate a fearless temperament92. We found a lower average HR under NS in hemizygous 4.5R carriers than in 3.5R carriers in men. Therefore, the physiological phenotypes of HR in 4.5R carrier will often be closely interrelated aggressive tendencies. Meanwhile, in our results, the average HR decreased in response to stimuli in men. Comparisons of average HR in response to emotional stimuli in adults with more and less aggressive tendencies or with and without a history of violent behaviour have revealed inconsistencies across studies65,80,93,94. Since HR variabilities are regulated by interactions between the vagal (which reduces HR) and sympathetic (which increases HR) systems95, these values have been used to measure emotional regulation capacity during psychological processes80,96,97. Then we remain focused on interpreting HR variabilities rather than average HR in the analyses under aggression stimulation. The correlation between HR variabilities and antisocial behaviour is not yet clear98; the possible connection between 4.5R carriers with larger HR variabilities under aggression stimuli and higher aggression is open to debate. Nevertheless, the different HR variability dynamics in MAOA-deficient mice support the feasibility of differences in HR variabilities in humans according to MAOA uVNTR genotype99. Therefore, this result implies that HR variabilities in response to aggression stimuli may be a neurobiological phenotype associated with genetic variations in MAOA. Furthermore, the use of human cardiomyocytes which contain MAOA100,101 has potential for studying enzyme expression and activity in the heart according to MAOA genotype indirectly. Although MAOA studies in human cardiomyocytes have still focused on the role of myocardial regulation of ROS102,103, there is evidence that MAOA is involved in the catabolism of norepinephrine in mouse cardiomyocytes104. This system can be suitable for predicting intracellular mechanism of MAOA in heart which corresponds to the phenotypes of HR between MAOA genotypes.
As the MAOA gene can be expected to follow the principles of X-linked gene expression, research in women is likely to have multiple outcomes with regard to aggression. Our EEG and ECG results in women with homozygous MAOA genotypes were inconsistent with those in men, as well as statistical significance was not obtained for women because of the low number of homozygous 4.5R carriers. Moreover, our findings showed no significant neurobiological response in women with heterozygous MAOA genotypes, similar to other studies105,106. This difference highlights the need for research at the cellular level regarding whether X-inactivation occurs in heterozygous MAOA genotypes to better understand genetic effects on aggression in women.
In this study, we identified new functional phenotypes of oscillatory EEG and ECG activities induced by aggression-related stimuli that vary with MAOA genotypes: greater reactivity of the γ signals and greater HR variabilities in 4.5R carriers than in 3.5R carriers. This result suggests that MAOA does have some genetic effects on neural mechanisms and may extend to emotional control mechanisms related to aggression. We also attempted to provide a basis for the neural mechanism indirectly through transcriptional expression according to MAOA uVNTR alleles via a reporter gene assay in vitro. Our study calls for further investigation into the contribution of uVNTR and additional mechanisms to MAOA transcription. Future studies should address two points. First, we must clarify whether MAOA uVNTR modulates the expression and activity of the enzyme, as well as neurotransmitter levels, neural activity and behaviour. Second, further studies are required to understand the differences in oscillatory activity from cognitive and emotional perspectives. Addressing these points will facilitate the interpretation of various human socio-emotional behaviours, such as depression107, anxiety108, and prosociality109, as well as cognitive abilities such as memory110,111, with regards to genetic variations in MAOA.
Methods
Study subjects
In the present study, 524 college and high school students (260 men and 264 women, age: 18.0 ± 1.4) in Korea were recruited and provided oral epithelial cells for genotyping. All subjects had no record of pathological behaviour and no criminal record. Of the total sample, 334 participants (169 men and 165 women) completed three self-report questionnaires. Of the participants who completed the questionnaires, 94 participants were randomly selected, and their neurobiological signals were measured. Since, the difference in brain wiring between right-handed and left-handed individuals remains unclear112,113,114,115, to avoid bias, we decided to select only right-handed individuals as our subjects. We collected valid data from 84 participants (36 men and 48 women) using EEG measurements and from 70 participants (30 men and 40 women) using ECG measurements. EEG samples with poor electrode attachment or subject movement were excluded from the analysis. ECG samples that were not consistent with PQRST waveforms, i.e., the series of the electrical events of heart rhythms (i.e., the P wave represents atrial depolarization, the QRS complex represents ventricular depolarization and contraction, and the T wave represents repolarization of ventricles)116 were also excluded from the analysis. This study was approved by the Institutional Review Board (IRB) of DGIST [Number: DGIST_170614-HR-009-04]. All participants provided written informed consent after hearing a detailed explanation of the experimental procedures, in accordance with the Declaration of Helsinki.
Identification of genotypes
Genomic DNA was extracted from epithelial cells using the G-spin™ Total DNA Extraction Kit (7046, BOCA Scientific, USA). Polymerase chain reaction (PCR) fragments were amplified using the following primers: MAOA forward (−1386, 5′-GCTGGTCTCTAAGAGTGGGTAC-3′, −1366) and MAOA reverse (−1019, 5′-GAACGGACGCTCCATTCGGAC-3′, −1038)35,63. For MAOA genotyping, PCR was performed in 50 μL reactions containing 1 μL of each primer (10 pmol/μL), approximately 100 ng of genomic DNA, and 2X TaKaRa Ex Taq PCR Premix (RR001A, Takara, Korea). Amplification was performed under the following conditions: 34 cycles of 30 s denaturing at 95 °C and 120 s annealing and elongation at 68 °C, in accordance with the manufacturer’s instructions. In addition, all PCR products were purified using a PureLink™ Quick Gel Extraction and PCR Purification Combo Kit (Invitrogen, USA). The purified amplicons were split into two aliquots; one was submitted to an automated Sanger’ sequencing service (Cosmogenetech, Korea), and the other was separated via 3% agarose gel electrophoresis and visualized to confirm all genotypes twice, especially for heterozygous genotypes. The alleles of the MAOA uVNTR-repeated region were genetically identified based on a redefined genetic characterization method that include copies of the 1R sequence and the first half of 0.5R22,59,60,61,62. We followed the naming convention of 2.5R, 3.5R, and 4.5R in this study, which is assumed to be equivalent to 2R, 3R, and 4R in other studies22,23,58.
Construct generation
DNA fragments containing the 2R, 2.5R, 3R, 3.5R, 4R, 4.5R, 5R, 5.5R, and 6R alleles of the MAOA gene promoter were generated by DNA synthesis (Cosmogenetech, Korea) based on the MAOA DNA sequence63 (GenBank accession number m89636). The alleles were PCR-amplified using two primers containing SacI (−1470, 5′-ACTGGCCGGTACCTGAGCTCCTGCAGCGAGCG-3′) and HindIII (+35, 5′-CCGGATTGCCAAGCTTGCTTTGGCTGAC-3′) restriction sites. The 1442–1577 bp DNA fragments containing the MAOA gene promoter were digested with restriction enzymes and ligated into the pGL4.10 [luc] vector (Promega, USA). All constructs were verified by sequencing (Cosmogenetech, Korea).
Cell lines, transfection, and reporter gene assays
We used the brain-derived SH-SY5Y human neuroblastoma cell line and the JAR human placental choriocarcinoma cell line that have been previously used to the study the in vitro functional characteristics of MAOA22. SH-SY5Y cells exhibit prominent features of catecholaminergic neurons, in which MAOA is predominantly found117, and are used as an in vitro model of the dopaminergic system, which expresses tyrosine hydroxylase and dopamine-beta-hydroxylase, as well as the dopamine transporter118,119. SH-SY5Y cells are also driven towards adrenergic phenotypes under certain growth conditions120,121. JAR cells are derived from placental trophoblasts, in which MAOA is expressed122,123, and are used to study the serotonergic system, especially the serotonin transporter124,125. Firefly DNA and control TK DNA (pGL4.74, Promega, USA) were co-transfected into SH-SY5Y (ATCC, CRL2266) and JAR (ATCC, HTB144) cells using the SF Cell Line 4D-Nucleofector® X Kit (Lonza, Switzerland) using a 4D-Nucleofector system (Lonza, Switzerland) according to the manufacturer’s instructions. pcDNA and control TK DNA were co-transfected as negative controls. For each transfection, 2 × 106 cells were resuspended in 100 µL of Nucleofector solution, 1 µg of firefly DNA and 1 µg of control DNA was added in a single NucleocuvetteTM, and the electroporation settings recommended for the cell line by 4D-Nucleofector system were applied (Pulse code: CM137). Transfected cells were plated at a density of 1 × 104 cells/well in 96-well plates with culture medium and were cultured at 37 °C and 5% CO2 for 48 h and then lysed with 50 µL of PLB solution in each well following the instructions of the Dual-Luciferase Reporter Assay System (Promega, USA). Next, 100 µL of LAR II solution was added to 20 µL of transferred PLB lysate in a luminometer plate, and firefly luciferase activity was measured. After reading, 100 µL of STOP & Glo reagent was added, and Renilla luciferase activity was measured. Luminescence was measured on a Spark 10 M (Tecan, Switzerland), and the results are shown as the ratio of firefly luciferase to Renilla luciferase. Tests were repeated in 6–12 wells using the transfected cells, and two or three independent experiments were performed.
Self-report questionnaires
The following three self-report questionnaires on aggression were used in this study: K-BDHI126, adapted from the Buss-Durkee Hostility Inventory (BDHI)127; K-BPAQ128, adapted from the Buss-Perry Aggression Questionnaire (BPAQ)129, which includes plan failure scenarios130,131; and K-PCS132, adapted from the Peer Conflict Scale (PCS)133. The peer-to-peer conflict scales for adolescents were classified into four factors: overt/relational and proactive/reactive. All questionnaires were validated in the Korean population134,135,136.
Stimuli and procedure
Experiments were conducted in a soundproof room maintained at 25 °C and constant humidity, with one subject and one experimenter in pairs. Neurobiological signals were measured while the subject watched a 118-s-long video that consisted of ‘fixation cross (4 s) - black - neutral scene (5 s) - black - scene 1 (39 s) - black - scene 2 (32 s) - black - scene 3 (11 s) - end’ (Fig. S5). We used a fixation cross set against a white background, as well as four black scenes between the other scenes, as the baseline. We used a scene of an empty school classroom, which is a common sight in the daily lives of the subjects, as the neutral stimulus (NS). Three scenes (scene 1, scene 2, and scene 3) related to aggression were used as the stimulus (S). Scene 1 showed an abusive argument between a driver and bossy passenger on a bus, as verbal abuse is reported to be related to verbal aggression137. Scene 2 showed a chat screen of peer conflict, in which one student is bullied and burdened with all of the assignment. Bullying has been reported to trigger proactive and reactive aggression in adolescents138,139. Scene 3 showed a person scratching a blackboard with their nails, which has been reported to lead to irritability140,141.
Data acquisition, processing, and analysis
Neurobiological signals were measured using an MP36 (BIOPAC, CA, USA) with a 4-channel system (sampling rate = 1000 Hz, bandpass filter = 0.5–100 Hz), which is useful for quick and simple measurements in large populations (sampling rate = 1000 Hz, bandpass filter = 0.5–100 Hz). In studies analysing the neurobiological responses of aggression using EEG, frontal α asymmetry is a well-known signal pattern for aggression45,46. Reportedly, relatively greater left frontal α asymmetry at resting state is an indicator of approach motivation, while greater right asymmetry is an indicator of avoidance motivation45,142. Similar results have also been obtained for the β signal49. Our subjects, Koreans, tend to show indirect emotional expressions such as avoidance of irritability compared with Westerners in several cross-cultural studies143. Additionally, studies of EEG responses to neutral, happy, and fear-related sounds in adult Korean men showed differences in α, β, and γ power values between emotional stimuli, especially in the right frontal region (Fp2 and F8 channels), compared with the left frontal region (F3, Fp1, F7)144. Therefore, we decided to analyse individual differences in the EEG signals concentrated on the right frontal region, Fp2 and F8, rather than analysing the asymmetric difference between the left and right frontal regions. Moreover, in control experiments, Fp2 and F8 showed prominent responses to the stimuli, while the differences in responses from P7 and P8 on stimulation were insignificant (Fig. S6). These two channels are not only accessible but are also related to aggression46. In our study, EEG was recorded through two electrodes on Fp2 (right side of prefrontal cortex) and F8 (right side of dorsolateral prefrontal cortex), with ground and reference electrodes on the neck, in accordance with the BIOPAC EEG electrode placement guidelines and the International 10–20 EEG system145,146,147. For EOG (Electrooculogram), an active electrode was placed above the left eye, with a ground electrode on the mid-forehead and a reference electrode on the left cheek bone for removal of artefacts from eye-movement or gross muscle movement. These artefacts were removed by independent component analysis using BIOPAC AcqKnowledge program version 4.2. For ECG, an active electrode was place on the right wrist, with a ground electrode on the right foot and a reference electrode on the left wrist, following Einthoven’s triangle. For data analysis, we targeted all frequency intervals, as many studies have shown that α, β, γ, and θ signals in the frontal regions are related to aggression45,46,49,82,148. For the signals from the Fp2 and F8 channels of each subject, discrete Fourier transforms were performed using the fast Fourier transform algorithm in MATLAB. Power spectral density was calculated in the area of each frequency interval (0.5 and 4 (δ), 4 and 8 (θ), 8 and 13 (α), 13 and 30 (β), and 30 and 90 (γ)) by the trapezoidal method. The mean value of the power spectral density of each frequency interval during neutral stimulus or stimulus was normalized to the mean value at baseline and was then expressed as the relative power value. We performed a 2000-point fast Fourier transform by dividing the data into 0.25 s intervals to trace dynamic changes in EEG over time. The values from each participant were normalized to area of the whole frequency domain (0–90 Hz). The mean values from all participants were then visualized by heat-map analysis. Apart from mean values, we calculated the power spectral density of each participant149. The average squared difference between the power spectral density for all 0.25 s intervals and measured values for each 0.25 s interval during NS or S at frequency intervals were presented as MSE. The ECG signal was corrected by detrending using the method of least squares, and R-R intervals (unit: ms) were obtained by calculating the local maximum value per heartbeat period. These were converted to HR values (unit: BPM) by 60000/(R-R intervals)150. The standard deviation of HR was calculated as the variance of HR, and the difference between the maximum and minimum values was calculated as ΔHR. The level of HR variabilities was then analysed using the variance of HR and ΔHR.
Statistical analysis
All statistical analyses were performed using Prism 8.0. One-way ANOVA with Tukey’s post hoc test was used to identify statistically significant differences in transcriptional efficiency values between allelic groups or between allelic groups and the negative control group. Since MAOA is located on the X chromosome, the results from men and women were analysed separately. To identify significant differences in the scores of questionnaires and their subcategories between genotypes, we used two-way ANOVA with the Bonferroni post hoc test. EEG results were analysed by separating the five frequency intervals (α, β, γ, θ, and δ). Repeated measures of two-way ANOVA with the Bonferroni post hoc test was used to determine statistically significant differences in EEG and ECG values between NS and S for each genotype. To determine significant differences in EEG and ECG values between genotypes during NS or S, we also used repeated measures of two-way ANOVA with the Bonferroni post hoc test. Statistical significance was set at p < 0.05.
Change history
26 June 2019
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Grove, W. M. et al. Heritability of substance abuse and antisocial behavior: A study of monozygotic twins reared apart. Biol. Psychiatry 27, 1293–1304 (1990).
DiLalla, L. F. & Gottesman, I. I. Biological and genetic contributors to violence: Widom’s untold tale. Psychological Bulletin 109, 125–129 (1991).
Miles, D. R. & Carey, G. Genetic and environmental architecture on human aggression. Journal of Personality and Social Psychology 72, 207–217 (1997).
Rhee, S. H. & Waldman, I. D. Genetic and environmental influences on antisocial behavior: a meta-analysis of twin and adoption studies. Psychol. Bull. 128, 490–529 (2002).
Godar, S. C., Fite, P. J., McFarlin, K. M. & Bortolato, M. The role of monoamine oxidase A in aggression: Current translational developments and future challenges. Prog. Neuro-Psychopharmacology Biol. Psychiatry 69, 90–100 (2016).
Moffitt, T. E. The new look of behavioral genetics in developmental psychopathology: Gene-environment interplay in antisocial behaviors. Psychol. Bull. 131, 533–554 (2005).
Cases, O. et al. Aggressive behavior and altered amounts of brain serotonin and norepinephrine in mice lacking MAOA. Science (80-.). 268, 1763–1766 (1995).
Brunner, H. G., Nelen, M., Breakefield, X. O., Ropers, H. H. & van Oost, B. A. Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science 262, 578–580 (1993).
Eccles, D. A., Macartney-Coxson, D., Chambers, G. K. & Lea, R. A. A unique demographic history exists for the MAO-A gene in Polynesians. J. Hum. Genet. 57, 294–300 (2012).
Westlund, K. N., Krakower, T. J., Kwan, S.-W. & Abell, C. W. Intracellular distribution of monoamine oxidase A in selected regions of rat and monkey brain and spinal cord. Brain Res. 612, 221–230 (1993).
Konradi, C. et al. Topographic immunocytochemical mapping of monoamine oxidase-A, monoamine oxidase-B and tyrosine hydroxylase in human post mortem brain stem. Neuroscience 26, 791–802 (1988).
Thorpe, L. W., Westlund, K. N., Kochersperger, L. M., Abell, C. W. & Denney, R. M. Immunocytochemical localization of monoamine oxidases A and B in human peripheral tissues and brain. J. Histochem. Cytochem. 35, 23–32 (1987).
Raine, A. From Genes to Brain to Antisocial Behavior. Curr. Dir. Psychol. Sci. 17, 323–328 (2008).
Buckholtz, J. W. & Meyer-Lindenberg, A. MAOA and the neurogenetic architecture of human aggression. Trends Neurosci. 31, 120–129 (2008).
Bortolato, M., Chen, K. & Shih, J. C. Monoamine oxidase inactivation: From pathophysiology to therapeutics. Adv. Drug Deliv. Rev. 60, 1527–1533 (2008).
Caspi, A. et al. Role of Genotype in the Cycle of Violence in Maltreated Children. Science (80-.). 297, 851 LP–854 (2002).
Meyer-Lindenberg, A. et al. Neural mechanisms of genetic risk for impulsivity and violence in humans. Proc. Natl. Acad. Sci. 103, 6269–6274 (2006).
Shih, J. C. & Thompson, R. F. Monoamine Oxidase in Neuropsychiatry and Behavior. Am. J. Hum. Genet, https://doi.org/10.1086/302562 (1999).
Tikkanen, R. et al. MAOA Alters the Effects of Heavy Drinking and Childhood Physical Abuse on Risk for Severe Impulsive Acts of Violence Among Alcoholic Violent Offenders. Alcohol. Clin. Exp. Res. 34, 853–860 (2010).
Deckert, J. et al. Excess of High Activity Monoamine Oxidase A Gene Promoter Alleles in Female Patients with Panic Disorder. Hum. Mol. Genet. 8, 621–624 (1999).
Denney, R. M., Koch, H. & Craig, I. W. Association between monoamine oxidase A activity in human male skin fibroblasts and genotype of the MAOA promoter-associated variable number tandem repeat. Hum. Genet. 105, 542–551 (1999).
Sabol, S. Z., Hu, S. & Hamer, D. A functional polymorphism in the monoamine oxidase A gene promoter. Hum Genet 103, 273–279 (1998).
Guo, G., Ou, X. M., Roettger, M. & Shih, J. C. The VNTR 2 repeat in MAOA and delinquent behavior in adolescence and young adulthood: Associations and MAOA promoter activity. Eur. J. Hum. Genet. 16, 626–634 (2008).
Byrd, A. L. & Manuck, S. B. MAOA, Childhood Maltreatment, and Antisocial Behavior: Meta-analysis of a Gene-Environment Interaction. Biol. Psychiatry 75, 9–17 (2014).
Haberstick, B. C. et al. MAOA Genotype, Childhood Maltreatment, and Their Interaction in the Etiology of Adult Antisocial Behaviors. Biol. Psychiatry 75, 25–30 (2014).
Taylor, A. & Kim-Cohen, J. Meta-analysis of gene-environment interactions in developmental psychopathology. Dev. Psychopathol. 19, 1029–1037 (2007).
Reif, A. et al. Nature and Nurture Predispose to Violent Behavior: Serotonergic Genes and Adverse Childhood Environment. Neuropsychopharmacology 32, 2375 (2007).
Frazzetto, G. et al. Early trauma and increased risk for physical aggression during adulthood: The moderating role of MAOA genotype. PLoS One 2 (2007).
Fergusson, D. M., Boden, J. M., Horwood, L. J., Miller, A. L. & Kennedy, M. A. MAOA, abuse exposure and antisocial behaviour: 30-year longitudinal study. Br. J. Psychiatry 198, 457–463 (2011).
Weder, N. et al. MAOA Genotype, Maltreatment, and Aggressive Behavior: The Changing Impact of Genotype at Varying Levels of Trauma. Biol. Psychiatry 65, 417–424 (2009).
Cervera-Juanes, R. et al. MAOA expression predicts vulnerability for alcohol use. Mol. Psychiatry 21, 472 (2015).
Fowler, J. S. et al. Evidence That Brain MAO A Activity Does Not Correspond to MAO A Genotype in Healthy Male Subjects. Biol. Psychiatry 62, 355–358 (2007).
Shumay, E., Logan, J., Volkow, N. D. & Fowler, J. S. Evidence that the methylation state of the monoamine oxidase A (MAOA) gene predicts brain activity of MAO A enzyme in healthy men. Epigenetics 7, 1151–1160 (2012).
Balciuniene, J., Emilsson, L., Oreland, L., Pettersson, U. & Jazin, E. Investigation of the functional effect of monoamine oxidase polymorphisms in human brain. Hum. Genet. 110, 1–7 (2002).
Pai, C., Chou, S. & Huang, F. Assessment of the role of a functional VNTR polymorphism in MAOA gene promoter: a preliminary Study. Forensic Sci. J. 6, 37–43 (2007).
Passamonti, L. et al. Monoamine oxidase-A genetic variations influence brain activity associated with inhibitory control: New insight into the neural correlates of impulsivity. Biol. Psychiatry 59, 334–340 (2006).
Eisenberger, N. I., Way, B. M., Taylor, S. E., Welch, W. T. & Lieberman, M. D. Understanding Genetic Risk for Aggression: Clues From the Brain’s Response to Social Exclusion. Biol. Psychiatry 61, 1100–1108 (2007).
Eisner, P. et al. Cortico-limbic connectivity in MAOA-L carriers is vulnerable to acute tryptophan depletion. Hum. Brain Mapp. 38, 1622–1635 (2016).
Schlüter, T. et al. MAOA-VNTR polymorphism modulates context-dependent dopamine release and aggressive behavior in males. Neuroimage 125, 378–385 (2016).
Haberstick, B. C. et al. Monoamine oxidase A (MAOA) and antisocial behaviors in the presence of childhood and adolescent maltreatment. Am. J. Med. Genet. - Neuropsychiatr. Genet. 135 B, 59–64 (2005).
Huizinga, D. et al. Childhood Maltreatment, Subsequent Antisocial Behavior, and the Role of Monoamine Oxidase A Genotype. Biol. Psychiatry 60, 677–683 (2006).
Fossella, J. et al. Assessing the molecular genetics of attention networks. BMC Neurosci. 3, 1–11 (2002).
Alia-Klein, N. et al. Brain Monoamine Oxidase A Activity Predicts Trait Aggression. J. Neurosci. 28, 5099–5104 (2008).
Kim-Cohen, J. et al. MAOA, maltreatment, and gene-environment interaction predicting children’s mental health: New evidence and a meta-analysis. Mol. Psychiatry 11, 903–913 (2006).
Harmon-Jones, E., Gable, P. A. & Peterson, C. K. The role of asymmetric frontal cortical activity in emotion-related phenomena: A review and update. Biol. Psychol. 84, 451–462 (2010).
Coyiuto, C. Resting EEG Asymmetries and Levels of Irritability. Student Libr. Res. Award. Wellesley Coll. Paper 16 (2016).
Scarpa, A. & Raine, A. Psychophysiology of Anger and Violent Behavior. Psychiatr. Clin. 20, 375–394 (2018).
Lorber, M. F. Psychophysiology of aggression, psychopathy, and conduct problems: A meta-analysis. Psychol. Bull. 130, 531–552 (2004).
Schutter, D. J. L. G., de Weijer, A. D., Meuwese, J. D. I., Morgan, B. & van Honk, J. Interrelations between motivational stance, cortical excitability, and the frontal electroencephalogram asymmetry of emotion: A transcranial magnetic stimulation study. Hum. Brain Mapp. 29, 574–580 (2007).
Volavka, J. Aggression, Electroencephalography, and Evoked Potentials: A Critical Review. Cogn. Behav. Neurol. 3 (1990).
Raine, A. & Dunkin, J. J. The genetic and psychophysiological basis of antisocial behavior: Implications for counseling and therapy. J. Couns. Dev. 68, 637–644 (1990).
Davidson, R. J. What does the prefrontal cortex ‘do’ in affect: Perspectives on frontal EEG asymmetry research. Biol. Psychol. 67, 219–233 (2004).
Ferguson, C. J. & Rueda, S. M. The Hitman Study. Eur. Psychol. 15, 99–108 (2010).
Schaffer, C. E., Davidson, R. J. & Saron, C. Frontal and parietal electroencephalogram asymmetry in depressed and nondepressed subjects. Biol. Psychiatry 18, 753–762 (1983).
Heller, W. In Psychological and biological approaches to emotion. 167–211 (Lawrence Erlbaum Associates, Inc, 1990).
Harmon-Jones, E., Lueck, L., Fearn, M. & Harmon-Jones, C. The effect of personal relevance and approach-related action expectation on relative left frontal cortical activity. Psychol. Sci. 17, 434–440 (2006).
Wacker, J. & Smillie, L. D. Trait Extraversion and Dopamine Function. Soc. Personal. Psychol. Compass 9, 225–238 (2015).
Huang, Y. Y. et al. An association between a functional polymorphism in the monoamine oxidase A gene promoter, impulsive traits and early abuse experiences. Neuropsychopharmacology 29, 1498–1505 (2004).
Jorm, A. F. et al. Association of a functional polymorphism of the monoamine oxidase A gene promoter with personality and psychiatric symptoms. Psychiatr. Genet. 10, 87–90 (2000).
Malan, C. Allelic diversity of selected human neurotransmitter genes in South African ethnic groups, scholarly journal. M.Sc. Thesis. Univ. Free State (2014).
Das, M. et al. MAOA promoter polymorphism and attention deficit hyperactivity disorder (ADHD) in Indian children. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 141, 637–642 (2006).
Laubscher, N. The role of emotional intelligence and a functional polymorphism in the MAO-A gene on aggression in humans (2012).
Zhu, Q. S., Grimsby, J., Chen, K. & Shih, J. C. Promoter organization and activity of human monoamine oxidase (MAO) A and B genes. J. Neurosci. 12, 4437–46 (1992).
Suls, J. & Wan, C. K. The relationship between trait hostility and cardiovascular reactivity: A quantitative review and analysis. Psychophysiology 30 (1993).
Gottman, J. M. et al. The Relationship Between Heart Rate Reactivity, Emotionally Aggressive Behavior, and General Violence in Batterers. J. Fam. Psychol. 9 (1995).
Ortiz, J. & Raine, A. Heart rate level and antisocial behavior in children and adolescents: a meta-analysis. J. Am. Acad. Child Adolesc. Psychiatry 43, 154–162 (2004).
Scarpa, A. & Raine, A. Psychophysiology of anger and violent behavior. Psychiatr. Clin. North Am. 20, 375–394 (1997).
McDermott, R., Tingley, D., Cowden, J., Frazzetto, G. & Johnson, D. D. P. Monoamine oxidase A gene (MAOA) predicts behavioral aggression following provocation. Proc. Natl. Acad. Sci. 106, 2118–2123 (2009).
Zhang, Y. et al. Gene-Gene-Environment Interactions of Serotonin Transporter, Monoamine Oxidase A and Childhood Maltreatment Predict Aggressive Behavior in Chinese Adolescents. Front. Behav. Neurosci. 11, 1–10 (2017).
Beaver, K. M., DeLisi, M., Vaughn, M. G. & Barnes, J. C. Monoamine oxidase A genotype is associated with gang membership and weapon use. Compr. Psychiatry 51, 130–134 (2010).
Lu, R. et al. No association of the MAOA gene with alcoholism among Han Chinese males in Taiwan $. 26, 457–461 (2002).
Kunugi, H. et al. A functional polymorphism in the promoter region of monoamine oxidase-A gene and mood disorders. Mol. Psychiatry 4, 393–395 (1999).
Rosenberg, S. et al. The association of DNA sequence variation at the MAOA genetic locus with quantitative behavioural traits in normal males. Hum. Genet. 120, 447–459 (2006).
Widom, C. S. & Brzustowicz, L. M. MAOA and the ‘Cycle of Violence:’ Childhood Abuse and Neglect, MAOA Genotype, and Risk for Violent and Antisocial Behavior. Biol. Psychiatry 60, 684–689 (2006).
Wong, M.-L. & Licinio, J. Research and treatment approaches to depression. Nat. Rev. Neurosci. 2, 343–351 (2001).
Chen, K., Holschneider, D. P., Wu, W., Rebrini, I. & Shih, J. C. A spontaneous point mutation produces monoamine oxidase A/B knock-out mice with greatly elevated monoamines and anxiety-like behavior. J. Biol. Chem. 279, 39645–39652 (2004).
Wu, J. B. & Shih, J. C. Valproic Acid Induces Monoamine Oxidase A via Akt / Forkhead. Cell 1, 714–723 (2011).
Ou, X. M., Chen, K. & Shih, J. C. Glucocorticoid and androgen activation of monoamine oxidase a is regulated differently by R1 and Sp1. J. Biol. Chem. 281, 21512–21525 (2006).
Wong, W. K., Chen, K. & Shih, J. C. Regulation of human monoamine oxidase B gene by Sp1 and Sp3. Mol. Pharmacol, https://doi.org/10.1124/mol.59.4.852 (2001).
Patrick, C. J. Psychophysiological correlates of aggression and violence: an integrative review. Philos. Trans. R. Soc. Lond. B. Biol. Sci, https://doi.org/10.1098/rstb.2008.0028 (2008).
Martini, N. et al. The dynamics of EEG gamma responses to unpleasant visual stimuli: From local activity to functional connectivity. NeuroImage 60 (2012).
Choi, J.-S. et al. Resting-state beta and gamma activity in Internet addiction. Int. J. Psychophysiol. 89, 328–333 (2013).
Anderson, S. W., Bechara, A., Damasio, H., Tranel, D. & Damasio, A. R. Impairment of social and moral behavior related to early damage in human prefrontal cortex. Nat. Neurosci. 2, 1032 (1999).
Volkow, N. D. et al. Brain glucose metabolism in violent psychiatric patients: a preliminary study. Psychiatry Res. Neuroimaging 61, 243–253 (1995).
Coccaro, E. F., McCloskey, M. S., Fitzgerald, D. A. & Phan, K. L. Amygdala and Orbitofrontal Reactivity to Social Threat in Individuals with Impulsive Aggression. Biol. Psychiatry 62, 168–178 (2007).
Davidson, R. J., Putnam, K. M. & Larson, C. L. Dysfunction in the neural circuitry of emotion regulation - A possible prelude to violence. Science (80-.). 289 (2000).
Lowet, E., Roberts, M. J., Bosman, C. A., Fries, P. & De Weerd, P. Areas V1 and V2 show microsaccade-related 3–4-Hz covariation in gamma power and frequency. Eur. J. Neurosci. 43, 1286–1296 (2015).
Yuval-Greenberg, S., Tomer, O., Keren, A. S., Nelken, I. & Deouell, L. Y. Transient Induced Gamma-Band Response in EEG as a Manifestation of Miniature Saccades. Neuron 58, 429–441 (2008).
Hoogenboom, N., Schoffelen, J.-M., Oostenveld, R., Parkes, L. M. & Fries, P. Localizing human visual gamma-band activity in frequency, time and space. Neuroimage 29, 764–773 (2006).
Vidal, J. R., Chaumon, M., O’Regan, J. K. & Tallon-Baudry, C. Visual Grouping and the Focusing of Attention Induce Gamma-band Oscillations at Different Frequencies in Human Magnetoencephalogram Signals. J. Cogn. Neurosci. 18, 1850–1862 (2006).
Wyart, V. & Tallon-Baudry, C. Neural Dissociation between Visual Awareness and Spatial Attention. J. Neurosci. 28, 2667 LP–2679 (2008).
De Wied, M. et al. Facial EMG and heart rate responses to emotion-inducing film clips in boys with disruptive behavior disorders. Psychophysiology, https://doi.org/10.1111/j.1469-8986.2009.00851.x (2009).
Meehan, J. C., Holtzworth-Munroe, A. & Herron, K. Maritally violent men’s heart rate reactivity to marital interactions: A failure to replicate the Gottman et al. (1995) typology. Journal of Family Psychology 15, 394–408 (2001).
Babcock, J. C., Green, C. E., Webb, S. A. & Graham, K. H. A Second Failure to Replicate the Gottman et al. (1995) Typology of Men Who Abuse Intimate Partners…and Possible Reasons Why. Journal of Family Psychology 18, 396–400 (2004).
Susman, E. J. Psychobiology of persistent antisocial behavior: Stress, early vulnerabilities and the attenuation hypothesis. Neurosci. Biobehav. Rev. 30, 376–389 (2006).
Juujärvi, P., Kaartinen, J., Pulkkinen, L., Vanninen, E. & Laitinen, T. Controlling reactive aggression through cognitive evaluation of proactive aggression cues. Cogn. Emot. 20, 759–784 (2006).
Posthumus, J. A., Böcker, K. B. E., Raaijmakers, M. A. J., Van Engeland, H. & Matthys, W. Heart rate and skin conductance in four-year-old children with aggressive behavior. Biol. Psychol. 82, 164–168 (2009).
Ashouri, F. P., Hamadiyan, H., Parvizpanah, A. & Rasekhi, S. Associations Between Resting Heart Rate and Antisocial Behavior. Int. Electron. J. Med. 5, 33–36 (2016).
Holschneider, D. P., Scremin, O. U., Chialvo, D. R., Chen, K. & Shih, J. C. Heart rate dynamics in monoamine oxidase-A- and -B-deficient mice. Am. J. Physiol. - Hear. Circ. Physiol. 282, H1751–H1759 (2002).
Sivasubramaniam, S. D., Finch, C. C., Rodriguez, M. J., Mahy, N. & Billett, E. E. A comparative study of the expression of monoamine oxidase-A and -B mRNA and protein in non-CNS human tissues. Cell Tissue Res, https://doi.org/10.1007/s00441-003-0765-6 (2003).
Richards, J. G., Saura Marti, J., Cesura, A. M. & Da Prada, M. Quantitative enzyme radioautography with [3H]Ro 19-6327: Localization of MAO-B in rat CNS, peripheral organs and human brain. Pharmacol. Res. Commun, https://doi.org/10.1016/S0031-6989(88)80557-5 (1988).
Mialet-Perez, J., Bianchi, P., Kunduzova, O. & Parini, A. New insights on receptor-dependent and monoamine oxidase-dependent effects of serotonin in the heart. In Journal of Neural Transmission, https://doi.org/10.1007/s00702-007-0695-7 (2007).
Kaludercic, N., Mialet-Perez, J., Paolocci, N., Parini, A. & Di Lisa, F. Monoamine oxidases as sources of oxidants in the heart. Journal of Molecular and Cellular Cardiology, https://doi.org/10.1016/j.yjmcc.2013.12.032 (2014).
Kaludercic, N. et al. Monoamine oxidase a-mediated enhanced catabolism of norepinephrine contributes to adverse remodeling and pump failure in hearts with pressure overload. Circ. Res, https://doi.org/10.1161/CIRCRESAHA.109.198366 (2010).
Meyer-Lindenberg, A. & Weinberger, D. R. Intermediate phenotypes and genetic mechanisms of psychiatric disorders. Nat. Rev. Neurosci. 7, 818–27 (2006).
Eme, R. MAOA and male antisocial behavior: A review. Aggress. Violent Behav. 18, 395–398 (2013).
Dannlowski, U. et al. Reduced amygdalaprefrontal coupling in major depression: Association with MAOA genotype and illness severity. Int. J. Neuropsychopharmacol. 12, 11–22 (2009).
Tadic, A. et al. Association of a MAOA gene variant with generalized anxiety disorder, but not with panic disorder or major depression. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 117B, 1–6 (2003).
Mertins, V., Schote, A. B., Hoffeld, W., Griessmair, M. & Meyer, J. Genetic susceptibility for individual cooperation preferences: The role of monoamine oxidase a gene (MAOA) in the voluntary provision of public goods. PLoS One 6, 1–9 (2011).
Mueller, S. C. et al. Evidence of MAOA genotype involvement in spatial ability in males. Behav. Brain Res. 267, 106–110 (2014).
Ross, R. S., Smolen, A., Curran, T. & Nyhus, E. MAO-A Phenotype Effects Response Sensitivity and the Parietal Old/New Effect during RecognitionMemory. Front. Hum. Neurosci. 12, 1–12 (2018).
Hopkins, W. D. Comparative and familial analysis of handedness in great apes. Psychological Bulletin 132, 538–559 (2006).
Klar, A. J. S. Genetic models for handedness, brain lateralization, schizophrenia, and manic-depression. Schizophr. Res. 39, 207–218 (1999).
Lake, D. A. & Bryden, M. P. Handedness and sex differences in hemispheric asymmetry. Brain and Language 3, 266–282 (1976).
Good, C. D. et al. Cerebral asymmetry and the effects of sex and handedness on brain structure: a voxel-based morphometric analysis of 465 normal adult human brains. Neuroimage 14, 685–700 (2001).
editor, L. S. L. Pathophysiology of heart disease: a collaborative project of medical students and faculty. (Third edition. Philadelphia: Lippincott Williams & Wilkins, [2003] ©2003).
Shih, J. et al. Monoamine oxidase: from genes to behavior. Annu Rev Neurosci 22: 197–217. Annual review of neuroscience 22 (1999).
Lopes, F. M. et al. Comparison between proliferative and neuron-like SH-SY5Y cells as an in vitro model for Parkinson disease studies. Brain Res. 1337, 85–94 (2010).
Korecka, J. A. et al. Phenotypic Characterization of Retinoic Acid Differentiated SH-SY5Y Cells by Transcriptional Profiling. PLoS One 8, e63862 (2013).
Scott, I. G., Åkerman, K. E. O., Heikkilä, J. E., Kaila, K. & Andersson, L. C. Development of a neural phenotype in differentiating ganglion cell-derived human neuroblastoma cells. J. Cell. Physiol. 128, 285–292 (1986).
Kovalevich, J. & Langford, D. In (eds Amini, S. & White, M. K.) 9–21, https://doi.org/10.1007/978-1-62703-640-5_2 (Humana Press, 2013).
Egashira, T., Ekstedt, B. & Oreland, L. Inhibition by clorgyline and deprenyl of the different forms of monoamine oxidase in rat liver mitochondria. Biochem. Pharmacol. 25, 2583–2586 (1976).
Cawthon, R. M., Pintar, J. E., Haseltine, F. P. & Breakefield, X. O. Differences in the structure of A and B forms of human monoamine oxidase. J. Neurochem. 37, 363–372 (1981).
Heils, A. et al. Allelic Variation of Human Serotonin Transporter Gene Expression. J. Neurochem. 66, 2621–2624 (1996).
Jayanthi, L. D., Ramamoorthy, S., Mahesh, V. B., Leibach, F. H. & Ganapathy, V. Calmodulin-dependent regulation of the catalytic function of the human serotonin transporter in placental choriocarcinoma cells. J. Biol. Chem. 269, 14424–14429 (1994).
No, A. The effects of assertive training on the reduction of aggression and anxiety in juvenile delinquents. Dep. Psychol. Grad. Sch. Seoul Natl. Univ (1983).
Buss, A. H. & Durkee, A. An inventory for assessing different kinds of hostility. J. Consult. Psychol. 21 (1957).
Jeong, D. A buffering effects of social support on children’s school stress and maladjustments. Dep. Educ. Grad. Sch. Korea Univ (1995).
Buss, A. H. & Perry, M. The Aggression Questionnaire. J. Pers. Soc. Psychol. 63 (1992).
Kuppens, P. & Van Mechelen, I. Interactional appraisal models for the anger appraisals of threatened self-esteem, other-blame, and frustration. Cogn. Emot. 21 (2007).
Lee, S. Relations of psychopathy and narcissism with aggression: focusing on differential triggers. Dep. Psychol. Grad. Sch. Sungkyunkwan Univ (2015).
Han, Y. The relation of psychological variables to relational aggression in early adolescence. Dept. Psychol. Grad. Sch. Ajou Univ (2008).
Marsee, M. A., Kimonis, E. R. & Frick, P. J. Peer Conflict Scale. Unpubl. Rat. scale. Univ. New Orleans (2004).
Kim, Y. The relationship among the parent’s verbal control type the aggression of children and the self-esteems of children. Dep. Educ. Grad. Sch. Ewha Womans Univ (1997).
Seo, S. & Kwon, S. Validation study of the Korean version of the Aggression Questionnaire. Korean J. Clin. Psychol. 21, 487–501 (2002).
Ha, M. & Kim, J. The validation of the Korean peer conflict scale(K-PCS) for use with young adolescents. Stud. Korean Youth 23, 71–101 (2013).
Infante, D. A. & Wigley, C. J. Verbal aggressiveness: An interpersonal model and measure. Commun. Monogr. 53, 61–69 (1986).
Salmivalli, C. & Nieminen, E. Proactive and reactive aggression among school bullies, victims, and bully-victims. Aggress. Behav. 28, 30–44 (2001).
Jara, N., Casas, J. A. & Ortega-Ruiz, R. Proactive and reactive aggressive behavior in bullying: The role of values. Int. J. Educ. Psychol. 6, 1 (2017).
Ely, D. J. Aversiveness without pain: Potentiation of imaginai and auditory effects of blackboard screeches. Bull. Psychon. Soc. 6, 295–296 (1975).
Halpern, D. L., Blake, R. & Hillenbrand, J. Psychoacoustics of a chilling sound. Percept. Psychophys. 39, 77–80 (1986).
Denson, T. F., O’Dean, S. M., Blake, K. R. & Beames, J. R. Aggression in Women: Behavior, Brain and Hormones. Front. Behav. Neurosci. 12, 81 (2018).
Merkin, R. S. Cross-cultural communication patterns - Korean and American Communication. J. Intercult. Commun. may (2009).
Du, R. & Lee, H. J. Power spectral performance analysis of EEG during emotional auditory experiment. In 2014 International Conference on Audio, Language and Image Processing 64–68, https://doi.org/10.1109/ICALIP.2014.7009758 (2014).
Khazi, M., Kumar, A. & Vidya, M. J. Analysis of eeg using 10:20 electrode system. Int. J. Innov. Res. Sci. Eng. Technol. 1 (2012).
Torres, G., Cinelli, M., T Hynes, A., S Kaplan, I. & Leheste, J. Electroencephalogram Mapping of Brain States. Journal of Neuroscience and Neuroengineering 3 (2014).
Seeck, M. et al. The standardized EEG electrode array of the IFCN. Clin. Neurophysiol. 128, 2070–2077 (2017).
Jensen, O. et al. On the human sensorimotor-cortex beta rhythm: Sources and modeling. Neuroimage 26, 347–355 (2005).
Yeom, H.-G., Han, C.-H., Kim, H.-D. & Sim, K.-B. Human Emotion Recognition using Power Spectrum of EEG Signals: Application of Bayesian Networks and Relative Power Values TT - Human Emotion Recognition using Power Spectrum of EEG Signals: Application of Bayesian Networks and Relative Power Values. J. Korean Inst. Intell. Syst. 18, 251–256 (2008).
Park, K. & Jeong, H. Assessing Methods of Heart Rate Variability. Korean J. Clin. Neurophysiol. 16, 49–54 (2014).
Acknowledgements
We thank Mr. Won-Seok Kang in DGIST for technical advice on the neurobiological experiments. We also thank Professors Joohan Kim and Hyosang Kang in DGIST for their invaluable discussions and feedbacks upon statistics and mathematics related to our data. And, we are deeply indebted to Professor Youngsun Jin in Kyungpook National University for discussions and comments about our EEG and ECG results to improve our manuscript. This study was supported by the DGIST fund from the Korean government; “Laboratory Expenses of Academic Support”, “Infrastructure for Undergraduate Studies of Academic Infrastructure Establishment”, Education Innovation Activity Fund – 2018010154, Undergraduate Research Program 2017 grant from Korea Foundation for the Advancement of Science & Creativity, Undergraduate Group Research Program 2018 grant from DGIST, and Ministry of Science, ICT and Future Planning & DGIST (18-BD-0402, DGIST Convergence Science Center).
Author information
Authors and Affiliations
Contributions
S.Y.I. and Ji.J. performed the major analyses and wrote the paper. G.J. coded the programs used in our paper. J.Y. and Ja.J. collected and analysed physiological data. S.-iL. collected data and wrote the paper. J.C., S.L., Y.L. and D.K. collected data from human subjects. M.B. and J.H. performed the experiments and assisted in the research. C.M. wrote the paper. C.L. designed the research and wrote the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Im, S., Jeong, J., Jin, G. et al. MAOA variants differ in oscillatory EEG & ECG activities in response to aggression-inducing stimuli. Sci Rep 9, 2680 (2019). https://doi.org/10.1038/s41598-019-39103-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39103-7
This article is cited by
-
Interaction effects of the 5-HTT and MAOA-uVNTR gene variants on pre-attentive EEG activity in response to threatening voices
Communications Biology (2022)
-
From warrior genes to translational solutions: novel insights into monoamine oxidases (MAOs) and aggression
Translational Psychiatry (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.