Abstract
Human foods have become a pervasive subsidy in many landscapes, and can dramatically alter wildlife behavior, physiology, and demography. While such subsidies can enhance wildlife condition, they can also result in unintended negative consequences on individuals and populations. Seasonal hibernators possess a remarkable suite of adaptations that increase survival and longevity in the face of resource and energetic limitations. Recent work has suggested hibernation may also slow the process of senescence, or cellular aging. We investigated how use of human foods influences hibernation, and subsequently cellular aging, in a large-bodied hibernator, black bears (Ursus americanus). We quantified relative telomere length, a molecular marker for cellular age, and compared lengths in adult female bears longitudinally sampled over multiple seasons. We found that bears that foraged more on human foods hibernated for shorter periods of time. Furthermore, bears that hibernated for shorter periods of time experienced accelerated telomere attrition. Together these results suggest that although hibernation may ameliorate cellular aging, foraging on human food subsidies could counteract this process by shortening hibernation. Our findings highlight how human food subsidies can indirectly influence changes in aging at the molecular level.
Similar content being viewed by others
Introduction
Human food subsidies, like garbage, crops, and livestock, are a ubiquitous consequence of human development1,2,3. While such food subsidies can enhance nutritional condition and physiological performance of wildlife4, more human food may not always be better. Easily accessible human foods may lack species-specific nutritional requirements5,6, contain lethal toxicological compounds7, or enhance the spread of disease8. Consumption of human foods can also alter animal behavior9, increasing the risk of injury or mortality in human-dominated landscapes10,11. In general though, the consequences of human food subsidies on the individual fitness and longevity of free-ranging animals remain largely unknown.
Torpor, a state of lowered metabolic demand, has evolved as an adaptive response to food limitations and harsh environmental conditions. Although the degree and type of torpor range widely across animal groups, one of the deepest and most extended forms is seasonal hibernation12, which is observed in eight groups of mammals. By lowering body temperatures and reducing metabolic rates, hibernators accrue significant energetic savings and avoid predation, which increases overwinter and annual survival13, with direct implications for longevity14. In particular, small-bodied mammals that can enter hibernation possess lifespans longer than expected from their body size or metabolic rate15. This increased longevity appears to have coevolved with aspects of a relatively slow life history strategy, including delayed onset of senescence13,16. Hibernation, then, not only conserves energy, but may also be adaptive in slowing cellular aging14. Increasingly, researchers are utilizing telomeres – repetitive DNA sequences on the ends of eukaryotic chromosomes17,18 that are lost during cellular replication and from oxidative damage19 – as markers to quantify cellular aging, or aging distinct from chronology20,21,22. Recent studies have found that more time spent in torpor can decelerate telomere attrition, or reduce cellular aging, among small hibernators23,24,25. Although the exact mechanism of hibernation that slows cellular aging in small-bodied mammals is unknown, it appears to be associated either with a reduction in cell turnover rates26 or a reduction in oxidative stress24.
Changes to hibernation strategies and characteristics, then, are likely to have important implications for individual fitness. For example, warmer weather during the winter and spring due to climate change27 has altered the timing of emergence, leading to phenological mismatches with food sources28 and reducing individual fitness29. Expanding human development and increased wildlife access to supplemental food has been linked to delayed or shortened hibernation11,30,31, and even the loss of hibernation for a winter altogether32. Shortened hibernation periods are likely to lead to similar mismatches with local food sources and increased interactions and conflicts with humans11,31. It is unknown what these consequences will have on individual physiology or fitness traits, but given that hibernation is modulated primarily by local food conditions11,12,30,33, natural food availability and human subsidies could indirectly govern senescence by altering rates of cellular aging.
In this study, we investigated the relationship between food subsidies, hibernation, and cellular aging in the American black bear (Ursus americanus). As large-bodied hibernators, bears are sufficiently long-lived to exhibit senescence34,35, but unlike small hibernators, they remain near-euthermic during hibernation in spite of their reduced metabolic rate36 and increased oxidative stress37. Preliminary research suggests that cellular aging in black bears is driven principally by environmental conditions—such as natural food availability—found at different latitudes38. Bears generally hibernate for 4–6 months/year, and denning chronology is driven in part by forage availability – individuals with access to more food tend to enter hibernation later and den for shorter periods11,30,39. Furthermore, black bears often supplement their diet with human food subsidies, especially in years of natural food shortages40,41. Bears that use areas of human development show decreased hibernation periods11,30,31. This altered denning chronology is assumed to result from increased consumption of food subsidies, although this link has not been directly explored. To assess the effects of food subsidies on hibernation and cellular aging, we tracked and sampled a subset of female black bears through several summer and winter seasons as part of a larger study in Durango, Colorado, USA11,40. We analyzed bear stable isotopic signatures (δ13C) as a measure of consumption of human foods41,42, and determined the influence of use of human food on hibernation lengths across individuals. We then assessed the relationship between hibernation length and rates of telomere length to test the role of hibernation in cellular aging. Finally, we examined whether the specific role of oxidative stress associated with hibernation is a potential mechanism mediating telomere length change in bears.
Results
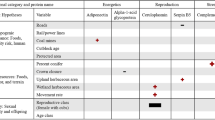
Female black bears (n = 30) averaged 8 years old at first sampling (range: 2 to 24) and hibernation lengths over the study averaged 170 days (range: 134 to 223). Summer sampled bears averaged −20.63 δ13C (range: −22.36 to −18.80). Bear serum exhibited average oxidative damage of 10.8 mg H2O2 dl−1 (range: 4.5 to 18.8) and average antioxidant capacity of 516 μmol HClO ml−1 neutralized (range: 349 to 769). Age was positively correlated with hibernation length (r = 0.73, P < 0.001); however, given the importance of age in determining bear physiology and behavior11, and that the variance inflation factor was only 1.47, we retained age as a covariate in subsequent tests.
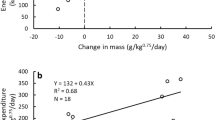
Bears enriched in δ13C during the summer (i.e., those that consumed more human foods), as well as younger bears, hibernated for shorter periods the subsequent winter (Table 1a, Fig. 1A). Telomere lengths on average decreased at a rate of 0.001 RTL/month (σ = 0.01) throughout the study period, but this pattern was inconsistent, as almost half the bears showed increased telomere lengths. We found that the mean monthly rate of telomere change was related to hibernation length; bears that hibernated longer on average experienced a slower rate of telomere attrition or even telomere lengthening during the study (Table 1b, Fig. 1B). There was limited support that telomere length change was related to oxidative stress (antioxidant capacity/oxidative damage; Table 1b; model coefficients are reported in Supplementary Table 1).
(A) Hibernation length for each bear (over one winter) regressed on the δ13C signature of bear hair sampled in the preceding summer (n = 15), showing a relationship between increased enrichment in δ13C and shorter hibernation lengths. (B) Average monthly telomere length (RTL) change regressed against hibernation lengths (days) for each bear (n = 30), exhibiting a relationship between longer hibernation length and slower rate of telomere shortening, and even telomere lengthening.
Oxidative damage (ROM) was related to sampling season, breeding status, and age (Table 2a). Bears exhibited increased oxidative damage during hibernation compared to the summer, and bears that had newborn cubs exhibited reduced oxidative damage compared to those that were barren or had yearlings. We found minimal differences in antioxidant capacity among bears based on our covariates (model coefficients are reported in Supplementary Table 2).
Discussion
Highly accessible and predictable food subsides can alter animal behavior9,43, change population dynamics44, and restructure community assemblages and species interactions45,46. Our study demonstrates that such food subsidies are also associated with cellular aging indirectly via altering hibernation length. Black bears with a greater reliance on human food subsidies were associated with having shorter hibernation lengths, and these shortened hibernation periods were associated with greater telomeric attrition. Consequently, bears that use more food subsidies hibernate less and thereby appear to experience greater cellular aging.
Hibernation chronology is driven by individual energy balance47, which is strongly linked to local weather conditions and food availability11,30. Recent work has shown that bears with access to more food, and bears exhibiting increased use of human development, den later and for a shorter period11,31. Our results demonstrate that greater consumption of human foods is associated with shorter hibernation in black bears. Increased consumption of human foods by bears has been associated with increased body weights and fecundity, but also reduced survival (due to vehicle collisions, lethal management, etc.)10,48. As a result, it has been suggested that urban areas may serve as an ecological trap10,41. This risk may be compounded by increased bear-human interactions resulting from shortened denning31, as well as have further consequences on fitness, through altered hibernation and accelerated telomere loss.
Bears display a remarkable suite of adaptations allowing them to remain immobile during hibernation, yet avoid negative side effects such as bone loss49 and muscle atrophy50. An additional advantage of hibernation appears to be slowed cellular aging; we found that bears with longer average hibernation lengths showed reduced rates of telomere shortening over the study period. Our finding corroborates recent work in small hibernators that effectively demonstrated that longer and deeper bouts of torpor slowed cellular aging23,24,25. Because telomere dynamics reflect accumulated life stress20 and can predict survival and longevity21, altering those dynamics through shortened denning periods may have negative long-term consequences.
Some animals display adaptations to counteract telomeric shortening, such as unusually high levels of the enzyme telomerase, which lengthens telomeres51,52. Although oxidative damage is typically an accelerant of telomere attrition19,53,54, animals that increase their antioxidant capacity might be able to mitigate such effects55. However, we found that although bears exhibited increased oxidative damage during hibernation compared to the active season37, we did not detect a concurrent increased antioxidant capacity. According to these stress measures, it appears that hibernation ameliorates cellular aging in spite of increased oxidative damage, perhaps due to reduced metabolic rate or enhanced somatic maintenance. This lack of a relationship between oxidative stress and telomere attrition could, however, also be influenced by our sampling - telomeres were not measured immediately before and after hibernation, and therefore may be more representative of stress experienced throughout the study period, not only during hibernation.
In addition to seasonal differences, oxidative damage differed among breeding status; females with cubs showed less damage, corroborating a recent study in polar bears56. Reproduction, and lactation in particular, is energetically expensive57,58, and resulting oxidative stress is typically regarded as a cost of reproduction59. The relationship between reduced oxidative damage and reproduction in bears remains unclear; however, researchers have speculated it could result from physiological changes during lactation that allow the off-loading of contaminants that otherwise induce oxidative stress56.
Our study of a free-ranging large hibernator suggests that increased reliance on human food subsidies reduces hibernation lengths. Our study also supports previous work on small hibernators that a benefit of hibernation is decelerated telomere attrition23. Thus, bears consuming more human foods may lose some of the long-term fitness advantages associated with hibernating, in particular rates of cellular aging. Therefore, the continued growth in food subsidies to wildlife are likely to cascade into altered behavior, ultimately with potential molecular consequences for rates of cellular aging.
Methods
Sample collection
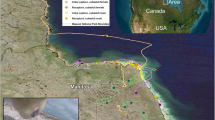
Black bears were captured near Durango, Colorado, from summer 2011 through winter 2015. All captures and animal handling were performed in accordance with relevant guidelines and regulations and approved by Colorado Parks and Wildlife [CPW], Fort Collins, CO (Animal Care and Use Protocol #01-2011)11. Adult females were fitted with GPS collars (Vectronics Globalstar) and subsequently relocated at their winter dens. Thirty bears were included in this study that were sampled a minimum of twice during the study period, twenty-six were sampled ≥3 times. Sampling occurred during initial capture in summer (mainly June – August) and then again during winter den visits (mainly early February – mid-March) in subsequent years; 18 of the bears were sampled in both the summer and winter within the same year.
During captures, bears were immobilized11, and guard hair and blood samples were collected for molecular analyses. At first capture, a premolar was removed to determine chronological age by counting cementum annuli (Matson’s Lab, Milltown, MT)60. Breeding status was also identified by the presence/absence of cubs (or lactation during summer captures when cubs were not always visible) or yearlings, and adult females categorized as “with yearlings”, “with cubs”, or “barren”. Black bear cubs are born during hibernation, and nurse part of that first year, typically staying with their mother through the next winter season; at the start of the second summer, yearlings will disperse.
We used collar activity sensor data to determine den entry and exit dates for each bear on an annual basis11. In 11 observations (out of 58 total), activity data were not available to estimate denning dates. In those cases, we used hourly GPS locations to define den entry as the first day of a 6-day period when a bear was exclusively located within 135 m of her den, and den emergence as the first day of a 6-day period when a bear remained 135 m away from her den61. Hibernation length was calculated as the number of days between den entrance and emergence.
Laboratory analyses
Blood samples for DNA extraction were stored in EDTA tubes; those for oxidative stress analyses were kept in serum-separating tubes. All samples were stored at −20 °C until analyses. We extracted DNA with standard procedures (QIAGEN DNeasy Blood and Tissue Extraction Kit; QIAGEN Inc., Valencia, CA). We quantified relative telomere lengths (RTL) using real-time quantitative polymerase chain reaction (qPCR)62. We previously optimized this method using the HNRPF gene63 and telomere primers telg and telc38,64 (Supplementary Material). We quantified relative telomere lengths from each sample. Because samples were collected once in the summer, and following mid-winters, we accounted for differences between sampling times of individuals by calculating an overall telomere length change for each bear between their first and last capture, averaged over months (n = 30).
Hair samples were prepared for stable isotope analyses as described in Pauli et al. 200965. Results are provided as per mil (‰) ratios relative to international standard, with calibrated internal laboratory standards. Individual foraging was represented by δ13C of hair samples; specifically enrichment in δ13C signifies increased human food in bear diets41,66. Human foods are enriched in δ13C compared to temperate native vegetation because they are dominated by corn and cane sugar derivatives67. Hair samples represent the assimilated diet during hair growth from spring through fall68, though in black bears tend to be highly correlated with stable isotopes in bone collagen, representing overall lifetime diet42.
We measured oxidative damage in bear serum samples, using the d-ROM test (Diacron International, Italy). The d-ROM test measures oxidative damage via the concentration of hydroperoxide, a reactive oxygen metabolite (ROM) that results from an attack of reactive oxygen species on organic substrates (e.g. nucleotides, proteins). The oxy-adsorbent test measures the total antioxidant capacity of the sample by measuring the ability of the serum to oppose the massive oxidative action of a hypochlorous acid (HClO) solution. Oxidative stress or status of an individual sample can be considered the ratio of antioxidant capacity to oxidative damage55,69. We prepared samples following the manufacturer’s protocol (Supplementary Material).
Data analyses
We tested three main hypotheses: (1) bear consumption of human foods reduces hibernation length; (2) reduced hibernation accelerates telomere attrition (i.e., the cellular aging process); (3) increased oxidative stress is a mechanism mediating telomere attrition. To test whether foraging on human food subsidies influenced hibernation length, we used linear regression with hibernation length (days) as the response variable and δ13C of bear hair (sampled in the preceding summer) as an explanatory variable. We also included age as a covariate, to account for the fact that older bears hibernate longer11. We restricted our data to bears sampled in summer and then again in the following winter (n = 15). To test our second and third hypotheses, we explored the relationship between telomeres (rate of telomere change for each individual, standardized as change per month), hibernation length (days within one season for each individual, averaged over multiple seasons), and oxidative stress (ratio of antioxidant capacity to oxidative damage for each individual, averaged over the sampling period; n = 30). Finally, because repeated oxidative stress samples from an individual bear fluctuated throughout the study period, we also examined factors associated with individual measures of oxidative stress (oxidative damage and antioxidant capacity) during sampling, rather than averaged over the study. We examined separately how oxidative damage or antioxidant capacity varied with age, sampling season (summer or winter), and breeding status of bears with linear mixed models; repeated samples from the same bear were accounted for with a random effect (unique bears = 28, samples = 84). For all analyses, we compared linear regression models using Akaike’s Information Criteria corrected for small sample sizes (AICc). The datasets are available from the corresponding author on reasonable request.
References
Oro, D., Genovart, M., Tavecchia, G., Fowler, M. S. & Martínez-Abraín, A. Ecological and evolutionary implications of food subsidies from humans. Ecol. Lett. 16, 1501–1514 (2013).
Ewen, J. G., Walker, L., Canessa, S. & Groombridge, J. J. Improving supplementary feeding in species conservation. Conserv. Biol. 29, 341–349 (2014).
Milner, J., van Beest, F., Schmidt, K., Brook, R. & Storaas, T. To feed or not to feed? Evidence of the intended and unintended effects of feeding wild ungulates. J. Wildl. Manage. 78, 1322–1334 (2014).
Marzluff, J. M. & Neatherlin, E. Corvid response to human settlements and campgrounds: causes, consequences, and challenges for conservation. Biol. Conserv. 130, 301–314 (2006).
Grémillet, D. et al. A junk-food hypothesis for gannets feeding on fishery waste. Proc. R. Soc. B Biol. Sci. 275, 1149–1156 (2008).
Birnie-Gauvin, K., Peiman, K. S., Raubenheimer, D. & Cooke, S. J. Nutritional physiology and ecology of wildlife in a changing world. Conserv. Physiol. 5, cox030 (2017).
Oaks, J. L. et al. Diclofenac residues as the cause of vulture population decline in Pakistan. Nature 427, 630–633 (2004).
Miller, R., Kaneene, J. B., Fitzgerald, S. D. & Schmitt, S. M. Evaluation of the influence of supplemental feeding of white-tailed deer (Odocoileus virginianus) on the prevalence of bovine tuberculosis in the Michigan wild deer population. J. Wildl. Dis. 39, 84–95 (2003).
Yirga, G. et al. Adaptability of large carnivores to changing anthropogenic food sources: diet change of spotted hyena (Crocuta crocuta) during Christian fasting period in northern. J. Anim. Ecol. 81, 1052–1055 (2012).
Beckmann, J. P. & Lackey, C. W. Carnivores, urban landscapes, and longitudinal studies: a case history of black bears. Human-Wildlife Interact. 2, 168–174 (2008).
Johnson, H. E. et al. Human development and climate affect hibernation in a large carnivore with implications for human–carnivore conflicts. J. Appl. Ecol. 55, 663–672 (2017).
Ruf, T. & Geiser, F. Daily torpor and hibernation in birds and mammals. Biol. Rev. 90, 891–926 (2015).
Turbill, C., Bieber, C. & Ruf, T. Hibernation is associated with increased survival and the evolution of slow life histories among mammals. Proc. R. Soc. B Biol. Sci. 278, 3355–3363 (2011).
Lyman, C. P., Brien, R. C. O., Greene, G. C. & Papafrangos, E. D. Hibernation and longevity in the Turkish hamster Mesocricetus brandi. Science 212, 668–670 (1981).
Wu, C. & Storey, K. B. Life in the cold: links between mammalian hibernation and longevity. Biomol. Concepts 7, 41–52 (2016).
Blanco, M. B. & Zehr, S. M. Striking longevity in a hibernating lemur. J. Zool. 296, 177–188 (2015).
Monaghan, P. & Haussmann, M. F. Do telomere dynamics link lifestyle and lifespan? Trends Ecol. Evol. 21, 47–53 (2006).
Meyne, J., Ratiliff, R. L. & Moyzis, R. K. Conservation of the human telomere sequence (TTAGGG)n among vertebrates. Proc. Natl. Acad. Sci. USA 86, 7049–7053 (1989).
von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 27, 339–344 (2002).
Finkel, T. & Holbrook, N. J. Oxidants, oxidative stress and the biology of ageing. Nature 408, 239–247 (2000).
Bize, P., Criscuolo, F., Metcalfe, N. B., Nasir, L. & Monaghan, P. Telomere dynamics rather than age predict life expectancy in the wild. Proc. R. Soc. B Biol. Sci. 276, 1679–1683 (2009).
Young, R. C. et al. Telomere length is a strong predictor of foraging behavior in a long-lived seabird. Ecosphere 6, 1–26 (2015).
Turbill, C., Ruf, T., Smith, S. & Bieber, C. Seasonal variation in telomere length of a hibernating rodent. Biol. Lett. 9, 20121095 (2013).
Turbill, C., Smith, S., Deimel, C. & Ruf, T. Daily torpor is associated with telomere length change over winter in Djungarian hamsters. Biol. Lett. 8, 304–307 (2012).
Hoelzl, F., Cornils, J. S., Smith, S., Moodley, Y. & Ruf, T. Telomere dynamics in free-living edible dormice (Glis glis): the impact of hibernation and food supply. J. Exp. Biol. 219, 2469–2474 (2016).
Koizumi, A., Tsukada, M., Wada, Y., Masuda, H. & Weindruch, R. Mitotic activity in mice is suppresed by energy restriction-induced torpor. J. Nutr. 122, 1446–1453 (1992).
Trenberth, K. E. et al. In Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report (2007).
Inouye, D. W., Barr, B., Armitage, K. B. & Inouye, B. D. Climate change is affecting altitudinal migrants and hibernating species. Proc. Natl. Acad. Sci. 97, 1630–1633 (2000).
Dobson, F. S., Lane, J. E., Low, M. & Murie, J. O. Fitness implications of seasonal climate variation in Columbian ground squirrels. Ecol. Evol. 6, 5614–5622 (2016).
Baldwin, R. A. & Bender, L. C. Denning chronology of black bears in Eastern Rocky Mountain National Park, Colorado. West. North Am. Nat. 70, 48–54 (2010).
Krofel, M., Špacapan, M. & Jerina, K. Winter sleep with room service: Denning behaviour of brown bears with access to anthropogenic food. J. Zool. 302, 8–14 (2017).
Beckmann, J. P. & Berger, J. Rapid ecological and behavioural changes in carnivores: the responses of black bears (Ursus americanus) to altered food. J. Zool. 261, 207–212 (2003).
Melvin, R. G. & Andrews, M. T. Torpor induction in mammals: recent discoveries fueling new ideas. Trends Endocrinol. Metab. 20, 490–498 (2009).
Schwartz, C. C. et al. Reproductive maturation and senescence in the female brown bear. Ursus 14, 109–119 (2003).
Turbill, C. & Ruf, T. Senescence is more important in the natural lives of long- than short-lived mammals. PLoS One 5, e12019 (2010).
Tøien, Ø. et al. Hibernation in black bears: independence of metabolic suppression from body temperature. Science 331, 906–909 (2011).
Chauhan, V. P. S. et al. Increased oxidative stress and decreased activities of Ca2+/Mg2+-ATPase and Na+/K+-ATPase in the red blood cells of the hibernating black bear. Life Sci. 71, 153–61 (2002).
Kirby, R., Alldredge, M. W. & Pauli, J. N. Environmental, not individual, factors drive markers of biological aging in black bears. Evol. Ecol. 31, 571–584 (2017).
Johnson, K. G. & Pelton, M. R. Environmental relationships and the denning period of black bears in Tennessee. J. Mammal. 61, 653–660 (1980).
Johnson, H. E. et al. Shifting perceptions of risk and reward: dynamic selection for human development by black bears in the western United States. Biol. Conserv. 187, 164–172 (2015).
Kirby, R., Alldredge, M. W. & Pauli, J. N. The diet of black bears tracks the human footprint across a rapidly developing landscape. Biol. Conserv. 200, 51–59 (2016).
Kirby, R., MacFarland, D. M. & Pauli, J. N. Consumption of intentional food subsidies by a hunted carnivore. J. Wildl. Manage. 81, 1161–1169 (2017).
Newsome, S. D., Garbe, H. M., Wilson, E. C. & Gehrt, S. D. Individual variation in anthropogenic resource use in an urban carnivore. Oecologia 178, 115–128 (2015).
Fedriani, J. M., Fuller, T. K. & Sauvajot, R. M. Does availability of anthropogenic food enhance densities of omnivorous mammals? An example with coyotes in southern California. Ecography (Cop.). 24, 325–331 (2001).
Newsome, T. M. et al. The ecological effects of providing resource subsidies to predators. Glob. Ecol. Biogeogr. 24, 1–11 (2015).
Rodewald, A. D., Kearns, L. J. & Shustack, D. P. Anthropogenic resource subsidies decouple predator-prey relationships. Ecol. Appl. 21, 936–943 (2011).
Schooley, R. L., McLaughlin, C. R., Matula, G. J. & Krohn, W. B. Denning chronology of female black bears: effects of food, weather, and reproduction. J. Mammal. 75, 466–477 (1994).
McLean, P. K. & Pelton, M. R. Some demographic comparisons of wild and panhandler bears in the Smoky Mountains. Int. Conf. Bear Res. Manag. 8, 105–112 (1989).
Fedorov, V. B. et al. Preservation of bone mass and structure in hibernating black bears (Ursus americanus) through elevated expression of anabolic genes. Funct. Integr. Genomics 12, 357–365 (2012).
Harlow, H. J., Lohuis, T., Beck, T. & Iaizzo, P. A. Muscle strength in overwintering bears. Nature 409, 997 (2001).
Munshi-South, J. & Wilkinson, G. S. Bats and birds: exceptional longevity despite high metabolic rates. Ageing Res. Rev. 9, 12–19 (2010).
Wang, L., McAllan, B. & He, G. Telomerase activity in the bats Hipposideros armiger and Rousettus leschenaultia. Biochemistry 76, 1017–1021 (2011).
Hammers, M. et al. Senescence in the wild: insights from a long-term study on Seychelles warblers. Exp. Gerontol. 71, 69–79 (2015).
Monaghan, P., Metcalfe, N. B. & Torres, R. Oxidative stress as a mediator of life history trade-offs: mechanisms, measurements and interpretation. Ecol. Lett. 12, 75–92 (2009).
Beaulieu, M., Reichert, S., Le Maho, Y., Ancel, A. & Criscuolo, F. Oxidative status and telomere length in a long-lived bird facing a costly reproductive event. Funct. Ecol. 25, 577–585 (2011).
Neuman-Lee, L. A. et al. Demographic and temporal variations in immunity and condition of polar bears (Ursus maritimus) from the southern Beaufort Sea. J. Exp. Zool. 327, 333–346 (2017).
LeBlanc, P. J. et al. Correlations of plasma lipid metabolites with hibernation and lactation in wild black bears Ursus americanus. J. Comp. Physiol. - B Biochem. Syst. Environ. Physiol. 171, 327–334 (2001).
Alonso-Alvarez, C. et al. Increased susceptibility to oxidative stress as a proximate cost of reproduction. Ecol. Lett. 7, 363–368 (2004).
Reichert, S. et al. Increased brood size leads to persistent eroded telomeres. Front. Ecol. Evol. 2, 1–11 (2014).
Willey, C. Aging black bears from first premolar tooth sections. J. Wildl. Manage. 38, 97–100 (1974).
Waller, B. W., Belant, J. L., Young, B. W., Leopold, B. D. & Simek, S. L. Denning chronology and den characteristics of American black bears in Mississippi. Ursus 23, 6–11 (2012).
Cawthon, R. M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 30, e47 (2002).
Fedorov, V. B. et al. Elevated expression of protein biosynthesis genes in liver and muscle of hibernating black bears (Ursus americanus). Physiol. Genomics 37, 108–18 (2009).
Cawthon, R. M. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucleic Acids Res. 37, e21 (2009).
Pauli, J. N., Ben-David, M., Buskirk, S. W., Depue, J. E. & Smith, W. P. An isotopic technique to mark mid-sized vertebrates non-invasively. J. Zool. 278, 141–148 (2009).
Hopkins, J. B., Koch, P. L., Ferguson, J. M. & Kalinowski, S. T. The changing anthropogenic diets of American black bears over the past century in Yosemite National Park. Front. Ecol. Environ. 12, 107–114 (2014).
Jahren, A. H. et al. An isotopic method for quantifying sweeteners derived from corn and sugar cane. Am. J. Clin. Nutr. 84, 1380–1384 (2006).
Hilderbrand, G. et al. Use of stable isotopes to determine diets of living and extinct bears. Can. J. Zool. 74, 2080–2088 (1996).
Stier, A., Reichert, S., Massemin, S., Bize, P. & Criscuolo, F. Constraint and cost of oxidative stress on reproduction: correlative evidence in laboratory mice and review of the literature. Front. Zool. 9, 37 (2012).
Acknowledgements
This work was funded by Colorado Parks and Wildlife, University of Wisconsin-Madison, and an American Society of Mammologists Grant-in-Aid. We thank numerous field technicians that collected data with the Durango bear project, as well as student lab technicians, especially Sonia Petty and Samantha Paddock. We also thank David Lewis for assisting with field data summaries.
Author information
Authors and Affiliations
Contributions
R.K. and J.N.P. wrote the manuscript and performed statistical analyses. H.E.J. and R.K. carried out field and laboratory analyses. R.K., H.E.J., M.W.A., and J.N.P. designed the study and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kirby, R., Johnson, H.E., Alldredge, M.W. et al. The cascading effects of human food on hibernation and cellular aging in free-ranging black bears. Sci Rep 9, 2197 (2019). https://doi.org/10.1038/s41598-019-38937-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-38937-5
This article is cited by
-
Looking Ahead: Predicting the Possible Ecological and Physiological Response of Galago Moholi to Environmental Change
International Journal of Primatology (2023)
-
Hibernation slows epigenetic ageing in yellow-bellied marmots
Nature Ecology & Evolution (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.