Abstract
The most severe visual impairments due to age-related macular degeneration (AMD) are frequently caused by the occurrence of choroidal neovascularization (CNV). Although photodynamic therapy with verteporfin (PDT-V) is currently a second-line treatment for neovascular AMD, it can be conveniently combined with drugs acting against vascular endothelial growth factor (anti-VEGF) to reduce the healthcare burden associated with the growing necessity of anti-VEGF intravitreal re-injection. Because the common 677 C > T polymorphism of the methylenetetrahydrofolate reductase gene (MTHFR-C677T; rs1801133) has been described as predictor of satisfactory short-term responsiveness of AMD-related CNV to PDT-V, we retrospectively examined the outcomes of 371 Caucasian patients treated with standardized, pro-re-nata, photodynamic regimen for 24 months. Responder (R) and non-responder (NR) patients were distinguished on the basis of the total number of scheduled PDT-V (TN-PDT-V) and change of best-corrected visual acuity (∆-BCVA). The risk for both TN-PDT-V and ∆-BCVA to pass from R to NR group was strongly correlated with CT and TT genotypes of MTHFR-C677T variant resulting, respectively, in odd ratios of 0.19 [95% CI, 0.12–0.32] and 0.09 [95% CI, 0.04–0.21] (P < 0.001), and odd ratios of 0.24 [95% CI, 0.15–0.39] and 0.03 [95% CI, 0.01–0.11] (P < 0.001). These pharmacogenetic findings indicate a rational basis to optimize the future clinical application of PDT-V during the combined treatments of AMD-related CNV, highlighting the role of thrombophilia to be aware of the efficacy profile of photodynamic therapy.
Similar content being viewed by others
Introduction
Age-related macular degeneration (AMD) [MIM 603075] is the most common cause of central blindness or low vision in the elderly population of the industrialized countries, i.e. in the areas defined by the World Health Organization as AMR-A (Amr, Region of the Americas), Eur-A (Eur, European Region), and WPR-A (WPR, Western Pacific Region)1,2,3. Caucasian populations are largely more affected by AMD than other ethnic groups4,5,6,7. There are two different clinical forms of sight-threatening AMD: (i) atrophic AMD (or dry AMD), characterized by atrophic changes of photoreceptors, retinal pigment epithelium (RPE), Bruch’s membrane, and choriocapillaris; and (ii) neovascular AMD (or wet/exudative AMD), complicated by choroidal neovascularization (CNV) and associated with RPE detachment, hemorrhages, exudates and/or cystoid macular edema8,9,10,11,12,13,14,15,16,17. At present, neovascular AMD is considered the most common cause of legal blindness in the Western countries18,19,20,21. In this fast-progressive form of AMD, CNV is caused by endothelial cell migration coming from choriocapillaris through weaker areas in Bruch’s membrane. The activation of several signaling pathways leads to a proliferating unit whose aberrant vessels, lacking of gap junctions, leak so that exudates and blood can spread through retinal layers8,10,13,17. Two therapeutic strategies are currently available to switch off AMD-related CNV and reduce its dramatic effects on patient’s vision: the intravitreally administrated drugs acting against vascular endothelial growth factor (anti-VEGF) and, as second-line treatment, the photodynamic therapy with verteporfin (PDT-V)22,23,24,25,26,27,28,29,30,31. In fact, during the last years, the care management of neovascular AMD is advanced from treatments able to minimize the vision loss or stabilize visual acuity (i.e. as-needed quarterly PDT-Vs)22,23,24,25,26, to those that allow a vision improvement (i.e. monthly, pro-re-nata or proactive regimes of intravitreal injections of anti-VEGF drugs)27,28,29,30,31. However, because of the critical drawback due to the growing burden of anti-VEGF drugs employment during the real-life clinical practice32,33,34, PDT-V has been combined with anti-VEGF in several different manners, allowing variable diminutions of anti-VEGF re-treatments35,36,37,38,39,40,41,42,43,44 but, in some protocols, also increasing the number of patients who experienced a minor visual gain than those treated with anti-VEGF alone36,37,38. Despite a huge debate on the need of improving the therapeutic management of neovascular AMD, the possible customization of PDT-V has not been fully explored to improve the decision-making process about this photothrombotic procedure targeting AMD-related CNV32,33. Therapeutic action of PDT-V is realized by a laser-light-induced shutdown of the neovascular complex that was previously photosensitized by the intravenous injection of verteporfin45,46,47,48,49,50. Even if demographic and clinical predictors, such as age of patient, baseline best-corrected visual acuity (BCVA) and baseline CNV dimension, have been evaluated to explain the remarkable heterogeneity of PDT-V outcomes in patients with neovascular AMD51,52,53, no unequivocal data were recorded also after the differentiation between different CNV subtypes which are traditionally categorized by fluorescein angiography (FA) in: (i) classic CNV (C-CNV), characterized by a well-demarcated hyperfluorescent area with an hypofluorescent margin in FA early phase and dye leakage obscuring the boundaries of the neovascular lesion during the late phase; (ii) predominantly classic CNV (PC-CNV), with the classic component occupying 50% or more of the entire neovascular lesion that includes occult CNV and all the fluorescence-blocking constituents; (iii) minimally classic CNV (MC-CNV), with the classic component occupying less than 50% of the neovascular complex; (iv) occult CNV (O-CNV), with no classic component22,23,24,25. In fact, within patients treated with PDT-V for AMD-related CNV, other factors appear to be implicated in those differences of therapeutic responsiveness, which are clearly noticeable reviewing the final outcomes of both randomized controlled trials22,23,24,25,26 and real-life clinical studies54,55,56. Focusing on a pharmacogenetic predictive approach, our previous investigations have pointed out the role of thrombophilic and anti-thrombophilic single nucleotide polymorphisms (SNPs) in modifying the result of PDT-V in Caucasian patients with neovascular macular degenerations57,58,59,60. In fact, several coagulation-balance SNPs are able to influence the response to PDT-V as a consequence of the multifaceted photo-thrombotic mechanism triggered by this therapeutic strategy within the neovascular complex61,62,63,64,65. Although the common 677 C > T polymorphism of the methylenetetrahydrofolate reductase gene (MTHFR-C677T; rs1801133) has been described as positive pro-thrombotic factor for the closing of C- and PC-CNVs secondary to AMD after a single PDT-V procedure57, its role in determining the long-term outcomes of photodynamic protocol has not yet been evaluated. The present retrospective study verified whether, during a 2-year period of PDT-V regimen, the carriers of MTHFR-C677T polymorphism with AMD-related C- or PC-CNV required less photodynamic re-treatments than patients without this SNP, also comparing the final differences of BCVA change between these two study clusters.
Results
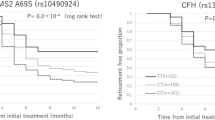
The study cluster consisted of 371 eyes of 371 patients treated with standardized PDT-V protocol, according to the procedures of the Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) study22,23, after the diagnosis of treatment-naïve AMD complicated by subfoveal C- or PC-CNV. In the course of the retrospectively selection of the study population, 98 patients were ruled out from the final statistical analyses owing to lack in follow-up visits (38 cases), not exact adhesion to the check timing (22 cases), not unequivocal interpretation of FA reading (14 cases), incomplete data collection (13 cases), or PDT-V complications such as RPE tear, massive retinal hemorrhage, and acute severe visual acuity decrease (11 cases). At baseline, the demographic and clinical attributes of these excluded patients were not statistically different with respect to those of the study cluster. In Table 1 are summarized the demographic and clinical characteristics of the analyzed cluster of patients, separated in responder (R) and non-responder (NR) to represent the percentage and mean values of the examined variables. In the final study population, patients with CC wild genotype of MTHFR-C677T polymorphism were 151, whereas 220 were carriers of polymorphic genotypes (CT in 176 cases and TT in 44 cases).
The baseline comparisons between demographic and clinical characteristics of these CC, CT, and TT patients are shown in Table 2. No significant statistical differences were found among the three genotypic groups. No significant deviations from Hardy-Weinberg equilibrium and genotype distribution were observed comparing MTHFR-C677T polymorphism among a control group of healthy Caucasian individuals [n = 200; CC = 93 (46.5%); CT = 88 (44.0%); TT = 19 (9.5%)] and the study group [n = 371; CC = 151 (40.7%); CT = 176 (47.4%); TT = 44 (11.9%)].
In our cluster of patients with classic or predominantly classic AMD-related CNV, the mean number of PDT-V ± standard deviation (SD) administered through the 24-month follow-up period, including the baseline application and the subsequent as-needed treatments, was 4.6 ± 1.6 (range: from 1 to 8). This average of total PDT-V is rather lower in comparison with that observed at the end of the 24-month follow-up period in TAP trial, i.e. 5.6 treatments from the study onset23. After the standardized photodynamic protocol, among carriers of MTHFR-C677T wild CC genotype, the mean number of PDT-V ± SD was 5.4 ± 1.5 (range: from 1 to 8), whereas in patients with CT and TT genotypes the averages of PDT-V ± SD were, respectively, 4.1 ± 1.4 (range: from 1 to 8) and 3.5 ± 1.4 (range: from 1 to 7). The statistical evaluation of the influence of MTHFR-C677T polymorphism on the responsiveness to PDT-V in terms of total number of treatments scheduled in each patient during the 24-month follow-up period (TN-PDT-V) has been accomplished also considering patient’s age per 3-year increment, baseline BCVA and baseline CNV area. Multivariate logistic regression analysis showed that polymorphic CT and TT genotypes were strongly correlated with, respectively, a 5- and 10-time decreased risk for TN-PDT-V of passing from R to NR group, i.e. odds ratios (OR) equal to 0.19 [95% confidence intervals (CI), 0.12–0.32] and 0.09 [95% CI, 0.04–0.21] (P < 0.001). Furthermore, lower patient’s age (OR, 1.13 [95% CI, 1.02–1.26]; P < 0.02) and greater baseline CNV area (OR, 0.80 [95% CI, 0.68–0.96]; P < 0.01) performed as weak positive predictors for good responsiveness to PDT-V with regard to the extent of re-treatment necessity in the course of our as-needed therapeutic protocol (Table 3). Likewise, the same statistical analysis concerning the change of BCVA from baseline to final visit (∆-BCVA), pointed out that CT and TT genotypes were also associated with, respectively, a 4- and 30-time decreased risk for ∆-BCVA of passing from R to NR group, i.e. ORs equal to 0.24 [95% CI, 0.15–0.39] and 0.03 [95% CI, 0.01–0.11] (P < 0.001). No other putative predictors emerged as significant factors influencing the responsiveness to PDT-V with regard to the degree of satisfactory vision outcomes, i.e. recovery, stabilization or slight worsening of BCVA (Table 3). Finally, when ∆-BCVA was analyzed on a continuous scale, patients with a CC genotype worsened by 0.23 logMAR [95%CI, 0.20–0.26 logMAR], those with CT genotype worsened by 0.09 logMAR [95%CI, 0.06–0.12 logMAR; P < 0.001 vs. CC genotype] and patients with TT genotype remained stable [0.01 logMAR; 95%CI, 0.06-0.07 logMAR; P < 0.001 vs. CC genotype]. These estimates did not change after adjusting for age, BCVA and CNV area at baseline.
Discussion
The current study pointed out that the thrombophilic MTHFR-C677T polymorphism, already recognized as positive predictor in short-term angiographic response to PDT-V of AMD-related C- and PC-CNVs57, get better the long-term outcomes of standardized, pro-re-nata, PDT-V protocol among patients with neovascular AMD. As a matter of fact, after two years of follow-up, the carriers of CT or TT genotype of MTHFR-C677T variant experienced a minor reduction of final BCVA as compared to non-carriers, showing a very low risk to pass from R to NR patients. Similarly, these polymorphic MTHFR-C677T genotypes are predictive of a minor probability to receive a greater number of PDT-V to deactivate CNV thus having to be included within NR patients. In particular, the chance to pass from R- to NR-status related to CT heterozygosis was remarkably higher than that related to TT homozygosis, indicating the existence of strong gene-dosage effect where the possibility of good response to PDT-V appears to be proportional to the number of the polymorphic T-alleles. Our retrospective investigation failed to document that demographic or clinical factors were important predictors of PDT-V outcomes in patients with neovascular AMD51,52,53. In fact, only considering the amount of the photodynamic re-treatments but not in regards to the final BCVA change, patient’s age and baseline CNV dimension weakly influenced the responsiveness to PDT-V. Besides, within our clusters of Caucasian ethnicity, the difference in genotype distribution of MTHFR-C677T between healthy controls and patients with neovascular AMD was appreciable even if not significant, in line with the data previously reported in Asians by Tanaka and coworkers66.
Genotypic predictors act as factors influencing PDT-V effect because they modify the photo-thrombotic and immuno-inflammatory interactions between PDT-V targets (i.e. CNV with its blood content and endothelial wall) and degenerated retina-RPE structures in which the neovascular complex develops57,58,59,60,61,62,63,64,65,67,68,69. Considering the photo-thrombotic action of PDT-V on CNV45,46,47,48,49, a comprehensive appraisal of the thrombophilic predisposition of vascular endothelium induced by MTHFR-C677T-related hyperhomocysteinemia (HHcy) provides explanation about the rationale of this pharmacogenetic correlation62,63,64. Experimental and clinical findings point out that CNV therapeutic occlusion after PDT-V is obtained by the laser-light-triggered thrombosis of photosensitized neovascular network by means of three synergistic mechanisms of action: (i) cellular, (ii) vascular, and (iii) immunological45,46,47,48,49. PDT-V efficacy is due to the preferential binding of a specific photosensitizer, i.e. verteporfin, to the endothelium of CNV in comparison with that covering the walls of normal retino-choroidal vasculature of macular area. Verteporfin couples with low-density lipoproteins (LDL) to form a complex that is prevalently up-taken into neovascular endothelial cells because of their over-expression of LDL receptors. Post-PDT-V changes of neovascular endothelium are caused by the photo-oxidative action of several reactive oxygen species (ROS), which act as triggers able to achieve the therapeutic hemostasis inside CNV. ROS-related exposure of vascular basement membrane activates adhesion, degranulation, and aggregation of the platelets, followed by the release of vasoactive mediators that amplify platelet activation, thrombosis, vasoconstriction, and increased vascular permeability. This series of events finally causes blood stasis, tissue hypoxia, and variable extent of CNV occlusion45, which is also related to iatrogenic damages of choriocapillaris because of non-selective hemodynamic drop of sub-retinal micro-vasculature48. During PDT-V, the photochemical changes triggered at the level of CNV endothelium can be strengthened by the MTHFR-677 T-allele62,63,64, as supported by our results of good therapeutic responsiveness among carriers of CT and TT genotypes characterized by an evident gene-dosage effect. MTHFR-C677T polymorphism is a very common folate-pathway genotypic variant heterogeneously distributed in the various ethnic groups70,71,72,73. The environmental selective pressure modifies this SNP distribution reliably because of different protein-intake habits: in fact, TT homozygosity is particularly represented in Mexico, South Italy and North China with an evident continental north/south gradient in Western Countries conversely to a south/north gradient in Eastern ones70,74. Pro-thrombotic consequences of the MTHFR-677 T-allele are mainly due to its HHcy-related effect. This polymorphic allele affects homocysteine (Hcy) metabolism elevating its plasma level and altering both vascular wall structure and blood coagulation system, to result in a structural dysfunction that alters normal homeostatic properties of endothelium, including its role in regulation of vascular tone, hemostasis, and inflammation75,76,77,78,79,80. In particular, the HHcy thrombotic diathesis is related to the tissue factor, a membrane glycoprotein generating the coagulation process through thrombin boost62,81,82. Genetic predisposition to HHcy can justify the variable efficacy of phototrombotic treatments, such as PDT-V. The mechanisms by which a photodynamic procedure elicits its therapeutic effects triggering CNV endothelium are basically overlapped with those causing pro-thrombotic phenomena due to hyperhomocysteinemic, folate-related, gene variants. These SNPs produce functional damages in MTHFR and also in other enzymes regulating the methionine-homocysteine metabolism, reducing its activity and inducing thrombophilia by means of endothelial cells and platelets hyper-activation45,62,75,76,77,78,79,80. HHcy causes oxidative and inflammatory changes in blood vessels as consequence of ROS-related stimulation, followed by lipid peroxidation in membranes of endothelial cells and in circulating LDL, over-expression of lectin-like oxidized LDL receptor-1 and aberrant platelet activation. The importance of these interactions among HHcy- and PDT-V-related effects are also corroborated by a rational interpretation of the pharmacogenetic correlation observed in the present study and in a previously published series of patients treated with PDT-V for neovascular AMD, both indicative of an intriguing gene-environment relationship between photodynamic action and MTHFR-C677T polymorphism57,62,83,84,85,86,87,88,89,90,91. This interaction has been described as one of main factor causative of acute vision loss associated with a photodynamic procedure92 and, of course, it should be taken into account in case of unexpected, PDT-V-related, adverse events such as severe BCVA decrease possibly owing to a non-selective hyper-thrombotic response of the normal macular vasculature93,94,95,96. On the other hand, also when patients were repeatedly treated with PDT-V, several cases of atrophic RPE changes have been reported in the irradiated areas with previously normal RPE, indicating an iatrogenic damage due to choriocapillaris hypo-perfusion with consequent RPE damage especially occurred during long-term PDT-V regimen48,97,98,99,100. Likewise, macular RPE atrophy have been also observed after anti-VEGF intravitreal administrations101,102,103 and, as reported by Abdelfattah and coworkers, its extent is proportionally correlated with the number of the injections101.
Although neovascular AMD is traditionally considered one of the leading causes of irreversible vision loss in the developed Countries, the large-scale utilization of anti-VEGF drugs, particularly evident in the last 10 years, allowed an outstanding reduction of AMD patients eligible for legal blindness certification104,105,106, preserving or restoring their vision-related quality of life107,108,109. However, in the real-life clinical practice, none of these benefits can be obtained without burdening the healthcare system that is weighted down by the continuous increase of the necessity for both monitoring and retreating of the patients with neovascular AMD110. In order to balance costs with benefits, one of the most promising solution is the synergistic or additive use of anti-VEGF drugs and PDT-V111. At present, these are the therapeutic strategies approved for the care of AMD-related CNV representing, in the clinical practice, the only treatments that can be simultaneously utilized to minimize both the irreversible vision loss caused by neovascular AMD and the socio-sanitary burden of this sight threatening disease. However, anti-VEGF/PDT-V combination is also characterized by an increased risk of detrimental effect in patient’s visual acuity secondary to RPE atrophy in macular area98,99,100,101,102,103.
Clinical investigations comparing the anti-VEGF/PDT-V combination with the anti-VEGF monotherapy always showed a lower necessity of anti-VEGF re-injection in the combination study groups35,36,37,38,39,40,41,42,43,44,112, whereas the mean BCVA gain has been often higher in the groups of patients exclusively treated with anti-VEGF drugs in comparison with those undergoing combined approach36,37,38,112. An ideal therapeutic management of a pathologic condition requiring a long-term patient’s take-in-charge, such as AMD-related CNV, should be able to individually select the most efficient strategies and the most efficient regimen for each of them, to reduce the gap between the need of patient-centered care and the actual care’s feasibility inside the healthcare provider.
Although PDT-V is no longer used as first-line therapy for neovascular AMD, the appraisal of its role as adjunctive treatment to anti-VEGF drugs has not yet led to a harmonized decision-making process32,33,110, generating heterogeneous clinical data35,36,37,38,39,40,41,42,43,44,112 which could find meaning through a new personalized pharmacogenetic approach for PDT-V application that might be inclusive of fundus autofluorescence imaging to inspect RPE atrophy98,99,100,101,102,103 and optical coherence tomography angiography to decide on single or combined retreatment. Consequently, predictive data on CNV response to PDT-V mono-therapy can represent the bases on which rationally expand our knowledge to optimize the modalities of synergistic combination between anti-VEGF and PDT-V effects32,50,111,113,114. In a perspective of translational medicine, the present findings on the common MTHFR-C677T polymorphism should be considered: (i) for the assessment of microvascular thrombus formation and expansion-rate in patients treated with photodynamic therapy to reduce the growth of solid tumors115,116,117; and, together with other consolidated pharmacogenetic data on anti-VEGF agents118,119, (ii) for the revision of the multifaceted responsiveness time-to-time reported in patients treated with anti-VEGF/PDT-V combinations for neovascular AMD.
Methods
Patients enrolled in the study
In the course of this multicenter study, the clinical records of Caucasian patients exclusively treated with PDT-V for the occurrence of newly diagnosed AMD-related CNV were retrospectively examined, exclusively including those homogeneous data which had been collected in six Eye Clinics from March 2004 to September 2017. All phenotypic and genotypic findings were computed and analyzed at the University of Ferrara (Italy). In the majority of these patients (268 of 371), the 24-month PDT-V protocol had been completed before anti-VEGF drugs (pegaptanib, ranibizumab, aflibercept, and bevacizumab) become available in the normal clinical practice. Conversely, in each of the other cases subsequently treated (103 of 371), contraindications to the intravitreal administration of anti-VEGF drugs, objective compliance’s troubles or patient’s refusal of this invasive therapy were present. All the selected patients had undergone both FA and indocyanine green angiography (ICGA) at baseline examination to finalize an accurate differential diagnosis between C-, PC-, MC- and O-CNV. The CNV classification was based on definitions from the Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) study, the Visudyne in Photodynamic Therapy study, and the Visudyne in Minimally Classic Choroidal Neovascularization study22,23,24. For our investigative purposes focusing the MTHFR-C677T pharmacogenetic aspect of PDT-V-related photothrombosis, only treatment-naïve patients suffering from AMD complicated by C- or PC-CNV were computed together to achieve a homogeneous study cluster, because of the lack of any significant difference in PDT-V responsiveness between these two similar CNV patterns22,23,25,57,62. On the other hand, considering the lack of any plausible correlation between PDT-V responsiveness and MTHFR-C677T polymorphism in patients with MC- and O-CNV58,62, these latter CNV patterns have been excluded from our data revision. As well, also subjects who had no regularly and/or correctly completed PDT-V protocol for a 24-month follow-up period have been ruled out. In particular, we have analyzed only the data of patients in whom the standardized PDT-V protocol was uneventfully completed in accordance with TAP study procedures22,23, also excluding patients with serious PDT-V complications such as RPE tear, massive retinal hemorrhage, and acute severe visual acuity decrease. Re-treatments were scheduled according to the international guidelines for PDT-V application, which recommend a patient’s examination at 3-month intervals and an additional course of treatment in case of persistent angiographic signs of CNV activity; each photodynamic treatment was scheduled within one week after baseline or follow-up angiographic exams22,23. Inclusion criteria are summarized as follows: (i) diagnosis of AMD in Caucasian patients with more than 50 years; (ii) BCVA better than 20/200 Snellen equivalent; (iii) angiographic diagnosis of C- or PC-CNV secondary to AMD after the examination of both FA and ICGA; (iv) active CNV under the geometric center of the foveal avascular zone; (v) greatest linear dimension of entire neovascular complex less than 5400 microns. On the other hand, exclusion criteria are listed herein: (i) history of any other anti-CNV treatment before and/or during the 24-month PDT-V protocol; (ii) angiographic diagnosis of MC- or O-CNV secondary to AMD after the examination of both FA and ICGA; (iii) any other possible cause of CNV different from AMD, such as pathologic myopia, angioid streaks, chorioretinal inflammatory diseases, hereditary retinal disorders, presumed ocular histoplasmosis syndrome, and/or severe ocular trauma; (iv) ascertained or suspected diagnosis of retinal angiomatous proliferation or polypoidal choroidal vasculopathy; (v) intraocular surgery and any laser-treatment of the eye during the 6 months before or the 3 months after the 24-month PDT-V protocol; (vi) presence of any significant condition, side effect and/or event possibly influencing the outcome of each PDT-V. At baseline and follow-up visits, these patients had undergone complete clinical examination, including medical and ophthalmologic anamneses, auto-refraction, BCVA test, slit-lamp biomicroscopy of the anterior segment, applanation tonometry, 60-diopter lens ophthalmoscopy, FA and ICGA. BCVA was measured using a standard logarithmic chart at a test distance of three meters. BCVA values (Snellen equivalent) were converted to the logarithm of the minimum angle of resolution (logMAR) scale for the statistical analyses. After a detailed description of the aims and procedures, patients gave their written informed consent to participate. In each studied patient, blood sample was collected for genotyping from September 2015 to December 2017. Genomic DNA was isolated from peripheral blood using standard proteinase K treatment, followed by phenol-chloroform extraction and ethanol precipitation. In a Peltier Thermal Cycler apparatus, samples were polymerase chain reaction (PCR)-genotyped for MTHFR-C677T polymorphism according to our earlier report120. MTHFR-C677T genotypes were confirmed by re-genotyping a random selection of samples. No discrepancies were found between genotypes determined in duplicate. All examinations were carried out in a blinded fashion in respect to the clinical data of each patient. The study followed the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of the University of Ferrara (protocol identification code PRUA2-2013-00002008; subgroup analysis of the version V3 amended on 12 June 2015 and approved on 10 September 2015).
Statistical analysis
Considering the typology of the investigated parameters, the sample size calculation, accomplished for the amount of the selected patients (371 cases), provided a value constantly upper than 85%. This test was performed using the PASS 97 statistical program (NCSS Inc., Kaysville, UT, USA). In the study population, the expected genotype distribution of MTHFR-C677T polymorphism was checked by Hardy-Weinberg equilibrium test, and compared with a cluster of normal individuals matched for sex, age and ethnicity with the study group. At baseline, demographic and clinical characteristics were compared between patients with CC, CT, and TT MTHFR-C677T genotypes. A t-test, corrected for groups, was employed to compare the mean of continuous measures, and the χ2 test was used to compare proportions of categorical measures. In the present study, the main outcome measures were: (i) the total number of PDT-V scheduled during the 24-month therapeutic protocol (TN-PDT-V); and (ii) change of BCVA from baseline to 24 months after the first PDT-V (∆-BCVA). For our analytical purposes regarding the influence of MTHFR-C677T polymorphism on PDT-V efficacy, these main outcome measures have been considered as dependent variables and were arbitrarily categorized in two clinical levels of therapeutic responsiveness distinguishing responder (R) and non-responder (NR) patients on the basis of both TN-PDT-V and final ∆-BCVA (Table 4). Considering the expected mean clinical outcomes obtainable by an as-needed quarterly, standardized PDT-V regimen in patients with subfoveal AMD-related C- or PC-CNV, a case has been labeled as responder if a maximum of four TN-PDT-V in 24 months were able to control the disease, because this is the first number lower than both TAP-study average of 5.623 and our average of 4.6 treatments. On the other hand, owing to the fact that in these patients the maximum estimated mean benefit of PDT-V protocol on ∆-BCVA is a slight vision loss or a visual acuity stabilization25,26, a case has been labeled as responder if a maximum of 0.1 logMAR BCVA reduction occurred at the end of 2-year follow-up. Odds ratios (OR) and 95% confidence intervals (CI) for combined comparisons were calculated by binary logistic regression models. Linear regression was used to estimate differences in ∆-BCVA across genotype subgroups. Multivariate analyses were performed to determine which factors were predictive of different response to PDT-V, using R/NR TN-PDT-V and R/NR ∆-BCVA as binary dependent variables. In these regression models, putative predictors were included according to the clinical plausibility of their possible influence on the dependent variables, i.e. TN-PDT-V and ∆-BCVA. Therefore, the following parameters were collectively examined as PDT-V predictors: patient’s age per 3-year increment, baseline BCVA, baseline CNV area, CT and TT genotypes of MTHFR-C677T51,52,53,57. Statistical analyses were performed by SySTAT V.5.0 (SySTAT Inc., Evanston, IL, USA) and SPSS Statistical Package (SPSS Inc., Chicago, IL, USA). A probability of P 0.05 was considered significant. During the investigation, the clinicians, laboratory personnel, and statistician were completely masked to both therapeutic interventions and clinical/genotyping outcomes regarding each enrolled patient.
Data Availability
All authors agree to make materials, data and associated protocols promptly available to readers.
References
Resnikoff, S. et al. Global data on visual impairment in the year 2002. Bull. World Health Organ. 82, 844–851 (2004).
Stevens, G. A. et al. Global prevalence of vision impairment and blindness: magnitude and temporal trends, 1990–2010. Ophthalmology 120, 2377–2384 (2013).
Wong, W. L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob. Heal. 2 (2014).
Klein, R., Peto, T., Bird, A. & Vannewkirk, M. R. The epidemiology of age-related macular degeneration. Am. J. Ophthalmol. 137, 486–495 (2004).
Friedman, D. S. et al. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 122, 564–572 (2004).
Klein, R. et al. Prevalence of age-related macular degeneration in 4 racial/ethnic groups in the multi-ethnic study of atherosclerosis. Ophthalmology 113, 373–380 (2006).
Velez-Montoya, R. et al. Current knowledge and trends in age-related macular degeneration: genetics, epidemiology, and prevention. Retina 34, 423–441 (2014).
Campa, C. et al. Inflammatory mediators and angiogenic factors in choroidal neovascularization: pathogenetic interactions and therapeutic implications. Mediators Inflamm. 2010, 546826 (2010).
Parmeggiani, F. et al. Mechanism of inflammation in age-related macular degeneration. Mediators Inflamm. 2012, 546786 (2012).
Bhutto, I. & Lutty, G. Understanding age-related macular degeneration (AMD): relationships between the photoreceptor/retinal pigment epithelium/Bruch’s membrane/choriocapillaris complex. Mol. Aspects Med. 33, 295–317 (2012).
Parmeggiani, F. et al. Mechanism of inflammation in age-related macular degeneration: an up-to-date on genetic landmarks. Mediators Inflamm. 2013, 435607 (2013).
Gallenga, C. E., Parmeggiani, F., Costagliola, C., Sebastiani, A. & Gallenga, P. E. Inflammaging: should this term be suitable for age related macular degeneration too? Inflamm. Res. 63, 105–107 (2014).
Kauppinen, A., Paterno, J. J., Blasiak, J., Salminen, A. & Kaarniranta, K. Inflammation and its role in age-related macular degeneration. Cell. Mol. Life Sci. 73, 1765–1786 (2016).
Blasiak, J., Reiter, R. J. & Kaarniranta, K. Melatonin in retinal physiology and pathology: the case of age-related macular degeneration. Oxid. Med. Cell. Longev. 2016, 6819736 (2016).
Handa, J. T., Cano, M., Wang, L., Datta, S. & Liu, T. Lipids, oxidized lipids, oxidation-specific epitopes, and Age-related Macular Degeneration. Biochim. Biophys. Acta - Mol. Cell Biol. Lipids 1862, 430–440 (2017).
Tan, P. L., Bowes Rickman, C. & Katsanis, N. AMD and the alternative complement pathway: genetics and functional implications. Hum. Genomics 10, 23 (2016).
Chirco, K. R., Sohn, E. H., Stone, E. M., Tucker, B. A. & Mullins, R. F. Structural and molecular changes in the aging choroid: implications for age-related macular degeneration. Eye (Lond) 31, 10–25 (2017).
Ferris, F. L., Fine, S. L. & Hyman, L. Age-related macular degeneration and blindness due to neovascular maculopathy. Arch. Ophthalmol. 102, 1640–1642 (1984).
Soubrane, G. et al. Burden and health care resource utilization in neovascular age-related macular degeneration: findings of a multicountry study. Arch. Ophthalmol. 125, 1249–1254 (2007).
Bandello, F., Lafuma, A. & Berdeaux, G. Public health impact of neovascular age-related macular degeneration treatments extrapolated from visual acuity. Invest. Ophthalmol. Vis. Sci. 48, 96–103 (2007).
Wong, T. et al. The natural history and prognosis of neovascular age-related macular degeneration. a systematic review of the literature and meta-analysis. Ophthalmology 115, 116–126 (2008).
Treatment of Age-related Macular Degeneration With Photodynamic Therapy Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials - TAP Report 1. Arch. Ophthalmol. 117, 1329–1345 (1999).
Bressler, N. M. et al. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: two-year results of 2 randomized clinical trials - TAP Report 2. Arch. Ophthalmol. 119, 198–207 (2001).
Azab, M. et al. Verteporfin therapy of subfoveal minimally classic choroidal neovascularization in age-related macular degeneration: 2-year results of a randomized clinical trial. Arch. Ophthalmol. 123, 448–457 (2005).
Chakravarthy, U. et al. Evolving European guidance on the medical management of neovascular age related macular degeneration. Br. J. Ophthalmol. 90, 1188–1196 (2006).
Wormald, R., Evans, J., Smeeth, L. & Henshaw, K. Photodynamic therapy for neovascular age-related macular degeneration. Cochrane Database Syst. Rev. 3, CD002030 (2007).
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group; Martin, D.F. et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 119, 1388–1398 (2012).
Heier, J. S. et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119, 2537–2548 (2012).
Chakravarthy, U. et al. IVAN study investigators. Alternative treatments to inhibit VEGF in age-related choroidal neovascularisation: 2-year findings of the IVAN randomised controlled trial. Lancet 382, 1258–1267 (2013).
Solomon, S. D., Lindsley, K., Vedula, S. S., Krzystolik, M. G. & Hawkins, B. S. Anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Cochrane Database Syst. Rev. 8, CD005139 (2014).
Arnold, J. J. et al. Two-year outcomes of “treat and extend” intravitreal therapy for neovascular age-related macular degeneration. Ophthalmology 122, 1212–1219 (2015).
Colquitt, J. et al. Ranibizumab and pegaptanib for the treatment of age-related macular degeneration: a systematic review and economic evaluation. Health Technol. Assess. 12, iii–iv, ix-201 (2008).
Reeves, B. C. et al. Verteporfin photodynamic therapy for neovascular age-related macular degeneration: cohort study for the UK. Health Technol. Assess. 16, i–xii, 1–200 (2012).
Schmier, J. K. & Hulme-Lowe, C. K. Cost-effectiveness models in age-related macular degeneration: issues and challenges. Pharmacoeconomics 34, 259–272 (2016).
Kaiser, P. K. Verteporfin photodynamic therapy combined with intravitreal bevacizumab for neovascular age-related macular degeneration. Ophthalmology 116, 747–755 (2016).
Bashshur, Z. F. et al. Ranibizumab monotherapy versus single-session verteporfin photodynamic therapy combined with as-needed ranibizumab treatment for the management of neovascular age-related macular degeneration. Retina 31, 636–644 (2011).
Kaiser, P. K. et al. Verteporfin plus ranibizumab for choroidal neovascularization in age-related macular degeneration: twelve-month results of the DENALI study. Ophthalmology 119, 1001–1010 (2012).
Krebs, I. et al. Comparison of ranibizumab monotherapy versus combination of ranibizumab with photodynamic therapy with neovascular age-related macular degeneration. Acta Ophthalmol. 91, e178–183 (2013).
Si, J. K. et al. Combination of ranibizumab with photodynamic therapy vs ranibizumab monotherapy in the treatment of age-related macular degeneration: a systematic review and meta-analysis of randomized controlled trials. Int. J. Ophthalmol. 7, 541–549 (2014).
Datseris, I. et al. Prospective comparison of low-fluence photodynamic therapy combined with intravitreal bevacizumab versus bevacizumab monotherapy for choroidal neovascularization in age-related macular degeneration. Semin. Ophthalmol. 30, 112–117 (2015).
Hatz, K. et al. Ranibizumab plus verteporfin photodynamic therapy in neovascular age-related macular degeneration: 12 months of retreatment and vision outcomes from a randomized study. Ophthalmologica 233, 66–73 (2015).
Rishi, E., Rishi, P., Sharma, V., Koundanya, V. & Athanikar, R. Long-term outcomes of combination photodynamic therapy with ranibizumab or bevacizumab for treatment of wet age-related macular degeneration. Oman J. Ophthalmol. 9, 87–92 (2016).
Tong, Y. et al. Comparison of the efficacy of anti-VEGF monotherapy versus PDT and intravitreal anti-VEGF combination treatment in AMD: a meta-analysis and systematic review. Int. J. Ophthalmol. 9, 1028–1037 (2016).
Dong, Y. et al. Effect of anti-VEGF drugs combined with photodynamic therapy in the treatment of age-related macular degeneration. Exp. Ther. Med. 12, 3923–3926 (2016).
Schmidt-Erfurth, U. & Hasan, T. Mechanisms of action of photodynamic therapy with verteporfin for the treatment of age-related macular degeneration. Surv. Ophthalmol. 45, 195–214 (2000).
Schlötzer-Schrehardt, U. et al. Dose-related structural effects of photodynamic therapy on choroidal and retinal structures of human eyes. Graefe’s Arch. Clin. Exp. Ophthalmol. 240, 748–757 (2002).
Schmidt-Erfurth, U., Michels, S., Barbazetto, I. & Laqua, H. Photodynamic effects on choroidal neovascularization and physiological choroid. Invest. Ophthalmol. Vis. Sci. 43, 830–841 (2002).
Michels, S. & Schmidt-Erfurth, U. Sequence of early vascular events after photodynamic therapy. Invest. Ophthalmol. Vis. Sci. 44, 2147–2154 (2003).
Schmidt-Erfurth, U., Niemeyer, M., Geitzenauer, W. & Michels, S. Time course and morphology of vascular effects associated with photodynamic therapy. Ophthalmology 112, 2061–2069 (2005).
Schmidt-Erfurth, U., Kiss, C. & Sacu, S. The role of choroidal hypoperfusion associated with photodynamic therapy in neovascular age-related macular degeneration and the consequences for combination strategies. Prog. Retin. Eye Res. 28, 145–154 (2009).
Blinder, K. J. et al. Effect of lesion size, visual acuity, and lesion composition on visual acuity change with and without verteporfin therapy for choroidal neovascularization secondary to age-related macular degeneration: TAP and VIP report no. 1. Am. J. Ophthalmol. 136, 407–418 (2003).
Axer-Siegel, R. et al. Photodynamic therapy for age-related macular degeneration in a clinical setting: visual results and angiographic patterns. Am. J. Ophthalmol. 137, 258–264 (2004).
Arias, L. et al. Impact of lesion size on photodynamic therapy with verteporfin of predominantly classic lesions in age related macular degeneration. Br. J. Ophthalmol. 89, 312–315 (2005).
Incorvaia, C. et al. 12-month retrospective study and review of photodynamic therapy with verteporfin for subfoveal choroidal neovascularization in age-related macular degeneration. Retina 28, 289–297 (2008).
Murjaneh, S. et al. Observational prospective study of the effectiveness in routine clinical practice of verteporfin photodynamic therapy in patients with neovascular age-related macular degeneration. Br. J. Ophthalmol. 93, 468–473 (2009).
Mataix, J., Desco, M., Palacios, E., Garcia-Pous, M. & Navea, A. Photodynamic therapy for age-related macular degeneration treatment: epidemiological and clinical analysis of a long-term study. Ophthalmic. Surg. Lasers Imaging 40, 277–284 (2009).
Parmeggiani, F. et al. Predictive role of coagulation-balance gene polymorphisms in the efficacy of photodynamic therapy with verteporfin for classic choroidal neovascularization secondary to age-related macular degeneration. Pharmacogenet. Genomics 17, 1039–1046 (2007).
Parmeggiani, F. et al. Coagulation gene predictors of photodynamic therapy for occult choroidal neovascularization in age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 49, 3100–3106 (2008).
Parmeggiani, F. et al. Impact of coagulation-balance gene predictors on efficacy of photodynamic therapy for choroidal neovascularization in pathologic myopia. Ophthalmology 117, 517–523 (2010).
Parmeggiani, F. et al. Effect of factor XIII-a G185T polymorphism on visual prognosis after photodynamic therapy for neovascular macular degeneration. Int. J. Mol. Sci. 16, 19796–19811 (2015).
Parmeggiani, F., Gemmati, D., Costagliola, C., Sebastiani, A. & Incorvaia, C. Predictive role of gene polymorphisms affecting thrombin-generation pathway in variable efficacy of photodynamic therapy for neovascular age-related macular degeneration. Recent Pat. DNA Gene Seq. 3, 114–122 (2009).
Parmeggiani, F., Gemmati, D., Costagliola, C., Sebastiani, A. & Incorvaia, C. Predictive role of C677T MTHFR polymorphism in variable efficacy of photodynamic therapy for neovascular age-related macular degeneration. Pharmacogenomics 10, 81–95 (2009).
Parmeggiani, F., Costagliola, C., Incorvaia, C., Sebastiani, A. & Gemmati, D. Pharmacogenetic aspects in therapeutic management of subfoveal choroidal neovascularisation: role of factor XIII-A 185 T-allele. Curr. Drug Targets 12, 138–148 (2011).
Parmeggiani, F. et al. Genetic predictors of response to photodynamic therapy. Mol. Diagn. Ther. 15, 195–210 (2011).
Gemmati, D. et al. Coagulation factor XIIIA (F13A1): novel perspectives in treatment and pharmacogenetics. Curr. Pharm. Des. 22, 1449–1459 (2016).
Tanaka, K. et al. Analysis of candidate genes for age-related macular degeneration subtypes in the Japanese population. Mol. Vis. 17, 2751–2758 (2011).
Tsuchihashi, T. et al. Prognostic phenotypic and genotypic factors associated with photodynamic therapy response in patients with age-related macular degeneration. Clin. Ophthalmol. 8, 2471–2478 (2014).
Chen, H., Yu, K. D. & Xu, G. Z. Association between variant Y402H in age-related macular degeneration (AMD) susceptibility gene CFH and treatment response of AMD: a meta-analysis. PLoS One 7, e42464 (2012).
Tsuchihashi, T. et al. Complement factor H and high-temperature requirement A-1 genotypes and treatment response of age-related macular degeneration. Ophthalmology 118, 93–100 (2011).
Peng, F., Labelle, L. A., Rainey, B. J. & Tsongalis, G. J. Single nucleotide polymorphisms in the methylenetetrahydrofolate reductase gene are common in US Caucasian and Hispanic American populations. Int. J. Mol. Med. 8, 509–511 (2001).
Rosenberg, N. et al. The frequent 5,10-methylenetetrahydrofolate reductase C677T polymorphism is associated with a common haplotype in Whites, Japanese, and Africans. Am. J. Hum. Genet. 70, 758–762 (2002).
Sachdeva, S. et al. MTHFR C677T polymorphism among three mendelian populations: a study from north India. Biochem. Genet. 50, 893–897 (2012).
Wang, X., Fu, J., Li, Q. & Zeng, D. Geographical and ethnic distributions of the MTHFR C677T, A1298C and MTRR A66G gene polymorphisms in Chinese populations: a meta-analysis. PLoS One 11, e0152414 (2016).
Wilcken, B. et al. Geographical and ethnic variation of the 677C> T allele of 5,10 methylenetetrahydrofolate reductase (MTHFR): findings from over 7000 newborns from 16 areas world wide. J. Med. Genet. 40, 619–625 (2003).
Rozen, R. Genetic predisposition to hyperhomocysteinemia: deficiency of methylenetetrahydrofolate reductase (MTHFR). Thromb. Haemost. 78, 523–526 (1997).
Castro, R., Rivera, I., Blom, H. J., Jakobs, C. & Tavares de Almeida, I. Homocysteine metabolism, hyperhomocysteinaemia and vascular disease: an overview. J. Inherit. Metab. Dis. 29, 3–20 (2006).
Moll, S. & Varga, E. A. Homocysteine and MTHFR mutations. Circulation 132, e6–e69 (2015).
Lai, W. K. C. & Kan, M. Y. Homocysteine-induced endothelial dysfunction. Ann. Nutr. Metab. 67, 1–12 (2015).
Craige, S. M., Kant, S. & Keaney, J. F. Jr. Reactive oxygen species in endothelial function - from disease to adaptation. Circ. J. 79, 1145–1155 (2015).
Sorrentino, F., Matteini, S., Bonifazzi, C., Sebastiani, A. & Parmeggiani, F. Diabetic retinopathy and endothelin system: microangiopathy versus endothelial dysfunction. Eye (Lond) 32, 1157–1163 (2018).
Nemerson, Y. The tissue factor pathway of blood coagulation. Smin. Hematol. 1992, 29, 170–176
Fryer, R. H., Wilson, B. D., Gubler, D. B., Fitzgerald, L. A. & Rodgers, G. M. Homocysteine, a risk factor for premature vascular disease and thrombosis, induces tissue factor activity in endothelial cells. Arterioscler. Thromb. 13, 1327–1333 (1993).
Kanani, P. M. et al. Role of oxidant stress in endothelial dysfunction produced by experimental hyperhomocyst(e)inemia in humans. Circulation 100, 1161–1168 (1999).
Weiss, N. Mechanisms of increased vascular oxidant stress in hyperhomocys-teinemia and its impact on endothelial function. Curr. Drug Metab. 6, 27–36 (2005).
Holven, K. B. et al. Hyperhomocysteinemic subjects have enhanced expression of lectin-like oxidized LDL receptor-1 in mononuclear cells. J. Nutr. 133, 3588–3591 (2003).
Sakurai, K. & Sawamura, T. Stress and vascular responses: endothelial dysfunction via lectin-like oxidized low-density lipoprotein receptor-1: close relationships with oxidative stress. J. Pharmacol. Sci. 91, 182–186 (2003).
Holven, K. B., Aukrust, P., Pedersen, T. M., Ose, L. & Nenseter, M. S. Enhanced platelet activation in hyperhomocysteinemic individuals. J. Thromb. Haemost. 5, 193–195 (2007).
Papatheodorou, L. & Weiss, N. Vascular oxidant stress and inflammation in hyperhomocysteinemia. Antioxid. Redox Signal. 9, 1941–1958 (2007).
Abais, J. et al. Nod-like receptor protein 3 (NLRP3) inflammasome activation and podocyte injury via thioredoxin-interacting protein (TXNIP) during hyperhomocysteinemia. J. Biol. Chem. 289, 27159–27168 (2014).
Pang, X. et al. Homocysteine induces the expression of C-reactive protein via NMDAr-ROS-MAPK-NF-κB signal pathway in rat vascular smooth muscle cells. Atherosclerosis 236, 73–81 (2014).
Hung, C. H., Chan, S. H., Chu, P. M. & Tsai, K. L. Homocysteine facilitates LOX-1 activation and endothelial death through the PKCβ and SIRT1/HSF1 mechanism: relevance to human hyperhomocysteinaemia. Clin. Sci. 129, 477–487 (2015).
Parmeggiani, F. et al. Vision loss after PDT. Ophthalmology 113, 157 (2006).
Arnold, J. J. et al. Acute severe visual acuity decrease after photodynamic therapy with verteporfin: case reports from randomized clinical trials-TAP and VIP report no. 3. Am. J. Ophthalmol. 137, 683–696 (2004).
Isola, V., Pece, A. & Battaglia Parodi, M. Choroidal ischemia after photodynamic therapy with verteporfin for choroidal neovascularization. Am. J. Ophthalmol. 142, 680–683 (2006).
Koizumi, H. & Hatanaka, H. Severe retinal vascular infarction after photodynamic therapy with verteporfin using the standard protocol. Arch. Ophthalmol. 128, 259–262 (2010).
Parmeggiani, F., Gemmati, D. & Costagliola, C. Thrombophilia in the occurrence of retinal vascular infarction after photodynamic therapy with verteporfin using the standard protocol. Arch. Ophthalmol. 128, 1632 (2010).
Weinberger, D. et al. Competitive quenching: a possible novel approach in protecting RPE cells from damage during PDT. Curr. Eye Res. 30, 269–277 (2005).
Wachtlin, J., Behme, T., Heimann, H., Kellner, U. & Foerster, M. H. Concentric retinal pigment epithelium atrophy after a single photodynamic therapy. Graefe’s Arch. Clin. Exp. Ophthalmol. 241, 518–521 (2003).
Battaglia Parodi, M., Da Pozzo, S. & Ravalico, G. Retinal pigment epithelium changes after photodynamic therapy for choroidal neovascularization in pathological myopia. Acta Ophthalmol. Scand. 85, 50–54 (2007).
Dewi, N. A., Yuzawa, M., Tochigi, K., Kawamura, A. & Mori, R. Effects of photodynamic therapy on the choriocapillaris and retinal pigment epithelium in the irradiated area. Jpn. J. Ophthalmol. 52, 277–281 (2008).
Abdelfattah, N. S., Zhang, H., Boyer, D. S. & Sadda, S. R. Progression of macular atrophy in patients with neovascular age-related macular degeneration undergoing antivascular endothelial growth factor therapy. Retina 36, 1843–1850 (2016).
Christenbury, J. G. et al. Progression of macular atrophy in eyes with type 1 neovascularization and age-related macular degeneration receiving long-term intravitreal anti–vascular endothelial growth factor therapy. Retina 38, 1276–1288 (2018).
Munk, M. R. et al. Macular atrophy in patients with long-term anti-VEGF treatment for neovascular age-related macular degeneration. Acta Ophthalmol. 94, e757–e764 (2016).
Bressler, N. et al. Estimated cases of legal blindness and visual impairment avoided using ranibizumab for choroidal neovascularization: non-hispanic white population in the United States with age-related macular degeneration. Arch. Ophthalmol. 129, 709–717 (2011).
Bloch, S. B., Larsen, M. & Munch, I. C. Incidence of legal blindness from age-related macular degeneration in Denmark: year 2000 to 2010. Am. J. Ophthalmol. 153, 209–213 (2012).
Keenan, T. D. L. et al. Incidence and baseline clinical characteristics of treated neovascular age-related macular degeneration in a well-defined region of the UK. Br. J. Ophthalmol. 97, 1168–1172 (2013).
Finger, R. P., Wiedemann, P., Blumhagen, F., Pohl, K. & Holz, F. G. Treatment patterns, visual acuity and quality-of-life outcomes of the WAVE study - a noninterventional study of ranibizumab treatment for neovascular age-related macular degeneration in Germany. Acta Ophthalmol. 91, 540–546 (2013).
Rakic, J. M. et al. Real-world variability in ranibizumab treatment and associated clinical, quality of life, and safety outcomes over 24 months in patients with neovascular age-related macular degeneration: the HELIOS study. Clin. Ophthalmol. 7, 1849–1858 (2013).
Finger, R. P., Guymer, R. H., Gillies, M. C. & Keeffe, J. E. The impact of anti-vascular endothelial growth factor treatment on quality of life in neovascular age-related macular degeneration. Ophthalmology 121, 1246–1251 (2016).
Han, D. Age-related macular degeneration, anti-VEGF therapy, and ophthalmic imaging: is there a best practice? JAMA Ophthalmol. 131, 1124–1126 (2013).
Englander, M. & Kaiser, P. K. Combination therapy for the treatment of neovascular age-related macular degeneration. Curr. Opin. Ophthalmol. 24, 233–238 (2013).
Larsen, M. et al. Verteporfin plus ranibizumab for choroidal neovascularization in age-related macular degeneration: twelve-month MONT BLANC study results. Ophthalmology 119, 992–1000 (2012).
Amoaku, W. M. et al. Defining response to anti-VEGF therapies in neovascular AMD. Eye 29, 721–731 (2015).
Lindsley, K., Li, T., Ssemanda, E., Virgili, G. & Dickersin, K. Interventions for age-related macular degeneration: are practice guidelines based on systematic reviews? Ophthalmology 123, 884–897 (2016).
Maeda, H., Tsukigawa, K. & Fang, J. A retrospective 30 years after discovery of the enhanced permeability and retention effect of solid tumors: next-generation chemotherapeutics and photodynamic therapy - problems, solutions, and prospects. Microcirculation 23, 173–182 (2016).
Acker, G., Palumbo, A., Neri, D., Vajkoczy, P. & Czabanka, M. F8-SIP mediated targeted photodynamic therapy leads to microvascular dysfunction and reduced glioma growth. J. Neurooncol. 129, 33–38 (2016).
Sirotkina, M. A. et al. Photodynamic therapy monitoring with optical coherence angiography. Sci. Rep. 7, 41506 (2017).
Wu, M. et al. Association between VEGF-A and VEGFR-2 polymorphisms and response to treatment of neovascular AMD with anti-VEGF agents: a meta-analysis. Br. J. Ophthalmol. 101, 976–984 (2017).
Yamashiro, K. et al. A prospective multicenter study on genome wide associations to ranibizumab treatment outcome for age-related macular degeneration. Sci. Rep. 7, 9196 (2017).
Gemmati, D. et al. Common gene polymorphisms in the metabolic folate and methylation pathway and the risk of acute lymphoblastic leukemia and non-Hodgkin’s lymphoma in adults. Cancer Epidemiol. Biomarkers Prev. 13, 787–794 (2004).
Acknowledgements
The Authors thank Mr. Giuseppe Gilli for his assistance in statistical analyses.
Author information
Authors and Affiliations
Contributions
Contributions were as follows: F.P., C.C., F.S., M.R.R., D.G., F.S.S.: conception and design; F.P., C.E.G., C.C., F.S., M.R.R., R.D.O., A.R., K.D.N., D.G., S.D.A., E.B., F.S.S.: collection and assembly of data; F.P., C.E.G., D.G.: data analysis and interpretation; and F.P., C.E.G., K.D.N., F.S.S.: writing manuscript text and preparing tables. All Authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Parmeggiani, F., Gallenga, C.E., Costagliola, C. et al. Impact of methylenetetrahydrofolate reductase C677T polymorphism on the efficacy of photodynamic therapy in patients with neovascular age-related macular degeneration. Sci Rep 9, 2614 (2019). https://doi.org/10.1038/s41598-019-38919-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-38919-7
This article is cited by
-
From resveratrol to ISIDE11: how to activate SIRT1 and improve endothelial function? New therapeutic insights for methylenetetrahydrofolate reductase deficiency
Cellular and Molecular Life Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.