Abstract
Low-density lipoprotein Receptor Related Protein 1B (LRP1B) is homologous to the gigantic lipoprotein receptor-related protein 1 that belongs to the family of Low-density lipoprotein receptors. Previous genetic association studies of the LRP1B gene have shown its genetic association with obesity. Through exome sequencing of the LRP1B gene from a childhood severe obesity cohort (n = 692), we found novel single nucleotide polymorphism (rs431809) in intron 4, which has been significantly correlated with both body mass index (BMI) and waist-hip-ratio (WHR). Three methylations of CpG sites (cg141441481, cg01852095 and cg141441470) in the same intron were also significantly correlated with BMI and WHR. All CpG methylations had bimodal patterns, and were dependent on rs431809 genotypes. The genetic influences of obesity on the LRP1B gene may be linked to the interplay of CpG methylations in the same intron. Heritability of SNP interacts with epigenetic crosstalk in LRP1B. Genetic and epigenetic crosstalk of LRP1B gene may be implicated in the prevention and therapeutic approach to childhood obesity.
Similar content being viewed by others
Introduction
Low-density lipoprotein Receptor Related Protein 1B (LRP1B) is highly homologous to the gigantic lipoprotein receptor-related protein 1 that belongs to the family of low-density lipoprotein receptors (LRP)1. LRP is widely expressed in several tissues and plays important roles in lipoprotein catabolism, blood coagulation, cell adhesion and migration, neuronal process outgrowth, and the pathogenesis of Alzheimer’s disease2,3,4. Most of the newly identified LRP1B ligands are well-known factors in blood coagulation and lipoprotein metabolism, suggesting a possible role of LRP1B in atherosclerosis5.
Genetic association studies of novel obesity-related gene variants in large populations have reported LRP1B as a factor in obesity susceptibility6,7,8,9. For example, intergenic rs2890652 single nucleotide polymorphism (SNP) of LRP1B has been associated with a BMI increase of 0.09 kg/m2 with a P value of 1e-106. In a genome-wide association study (GWAS) of body mass index (BMI) in Japanese people (n = 173,430), intronic SNP of LRP1B was shown to be significantly associated with BMI8. Another GWAS study in European ancestry cohorts showed that rs7583748 of LRP1B was strongly associated with BMI10. Burgdorf et al. reported that the rs2890652 C allele of LRP1B was associated with insulin resistance in a population-based sample of 6,039 Danish individuals11. A study examining the effect of genetic obesity loci on cognitive restraint and uncontrolled and emotional eating showed that the BMI-increasing variant of fat mass and obesity-associated protein (FTO) was positively associated with cognitive restraint while LRP1B was inversely associated with cognitive restraint12.
LRP1B is also candidate involved in the pathogenesis of cancer13,14,15,16,17,18. In one study, LRP1B mutation was a predictive marker for the presence of chronic obstruction pulmonary disease in patients with lung adenocarcinoma18. LRP1B is a novel candidate tumor suppressor gene that is inactivated by genetic and transcript alterations in nearly 50% of non-small-cell lung cancer cell lines.
Epigenetic regulation of LRP1B results in changes to the tumor environment, such as the aberrant methylation of LRP1B in acute lymphoblastic leukemia and in gastric cancer13,14,16. In colon cancer tissues, downregulation of LRP1B inhibits the growth, migration and metastasis of colon cancer cells17.
Here, we investigated the genetic polymorphisms of LRP1B in obesity by exome sequencing in a childhood obesity sample. We found an intronic SNP which was associated with Body Mass Index (BMI) and waist-hip-ratio (WHR). To understand the functional role of SNP located in this intron, we also investigated nearby CpG methylations (cg141441470, cg01852095 and cg141441481) for obesity.
In this study, we investigated the integrated effect of genetic and epigenetic changes in childhood obesity. We performed the genetic and epigenetic association study of LRP1B gene with obesity-related traits, as well as the sequence dependent methylation patterns linked to childhood obesity.
Experimental Procedures
All methods are approved by the committee of Korea National Institute of Health (KNIH) and carried out in accordance with relevant guidelines and regulations of KNIH. All the analysis was performed anonymously.
Study subjects
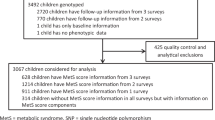
Study subjects were selected from the Korean Child-Adolescent Cohort Study, which followed a cohort of Korean students annually, at four elementary schools in Gwacheon, from entry into school at age 7, in 200519. The overall objective of the cohort study was to identify early risk factors for obesity and associated metabolic diseases in Korean children, and details have been published previously20. Severe childhood obesity is defined as a BMI-for-age of ≥1.2 times the 95th percentile value or above a BMI of ≥35 kg/m2 according to the recently proposed definition by the Centers for Disease Control and Prevention. For analysis, we used DNA samples from 305 children (159 males) with severe obesity (average age: 13.94 ± 0.84, average BMI: 31.71 ± 3.94 kg/m2) and from 387 lean controls (199 males) (average age: 13.93 ± 0.77, average BMI: 19.44 ± 1.34 kg/m2). Informed consents were obtained from parents of participants, and IRB of Korea National Institute of Health was approved (IRB approved number, 2014-08EXP-05-P-A). Detailed subject criteria are described in Table 1.
Exome Sequencing
All exons were captured by the SureSelect Human Exon V4 (Agilent Technologies, Carlsbad, CA, USA) according to the manufacturer protocols. The captured DNA was sequenced on the Illumina HiSeq. 2500. The sequence data were assembled using the UCSE Genome Browser (GRCh37/hg19) and mapped by BWA (http://bio-bwa.sourceforge.net). Variant detection was performed using SAMTOOLS (http://samtools.sourceforge.net). An average depth of coverage was ~67.2 fold21.
Quantitation of CpG DNA methylation and genotyping by pyrosequencing analyses
Polymerase chain reactions (PCRs) of LRP1B were performed for the purpose of pyrosequencing. The genomic position and gene positions of three target CpG sites analyzed via pyrosequencing are described in Fig. 1A. The PCR cycling conditions and the sequences for all primers designed by MethPrimer (Biotage AB) for cg141441470, cg01852095 and cg141441481 are described in Fig. 1B. Pyrosequencing assays were designed, optimized, performed on the PSQ HS 96 A System (Biotage AB) according to the manufacturer’s specifications (Pyrosequencing, Westborough, MA, USA). We measured the quantitation of cg141441470, cg01852095 and cg141441481 methylations. Pyrosequencing was conducted using bisulfite treated DNA. Three CpG methylation sites are located 570 kb upstream from rs431809. Technical controls for pyrosequencing revealed median methylations of 0~10% (unmethylated control), and >90% (methylated control), thereby defining detection limits of EpiTect PCR control DNA set (QIAGEN, USA). Finally, we measured DNA methylations 638 samples (numbers of controls:386, numbers of cases:252). Fifty-four samples were failed for pyrosequencing.
The schematic diagrams of LRP1B locus and Pyrosequencing. (A) The schematic diagrams of LRP1B locus with rs431809 and three CpG sites (cg141441470 cg01852095 and cg141441481) in intron 4 is diagramed. The biological position of each site was indicated by arrows. (B) The primer sequences and PCR conditions for pyrosequencing. (C) The pyrogram for the experiment was shown.
Statistical analyses for genetic association study
For genetic association study, quality control was implemented in PLINK22. Single nucleotide polymorphisms (SNPs) were excluded for any of the following reasons: (1) minor allele frequency (MAF) < 1%; (2) genotype call rate <99%; and (3) Hardy–Weinberg equilibrium (HWE) P value < 1e−03. Rare genetic variants (MAF < 1%), were excluded to test for association study, because of underpowered by small sample size. Before frequency and genotyping pruning, there are 235 SNPs. 3 markers to be excluded based on HWE test (P < = 1e-3). Total genotyping rate in remaining individuals is 0.999256. 4 SNPs failed missingness test (GENO > 0.01). 170 SNPs failed frequency test (MAF < 1%). After frequency and genotyping pruning, there are 61 SNPs. A genetic association was tested using the additive model in PLINK22. Age and sex were used as the covariates for the study performed with linear and logistic regression analysis. Raw P values < 0.05 was considered statistically significant.
Statistical analyses for DNA methylation study
Statistical analysis for DNA methylation study was performed by non-parametric methods because of bimodal data distribution. Wilcoxon signed-rank test, Kruskal–Wallis test and Spearman’s correlation test were adopted. Age and sex were used as the covariates for the study performed with regression analysis. Statistical analysis was performed using R version 3.2.1 and data was visualized using ggplot2.
Results
Genetic association study of rs431809 with obesity related traits
The general characteristics of obese children and controls are summarized in Table 1. Childhood obese cases (n = 305) with an average BMI of 31.71 ± 3.94 and lean controls (n = 387) with an average BMI of 19.4 ± 1.34 were categorized. There was no significant difference between the two groups in age (P > 0.05). The average of waist-hip-ratio (WHR) were also listed with significant differences between cases and controls in Table 1.
We performed exome sequencing of LRPB1 gene from 692 blood samples to find any genotype which associated with BMI and WHR. Before frequency and genotyping pruning, exome sequencing showed 235 single nucleotide variants existed at LRP1B (chr2: 140,988,992 - 142,889,270, hg19). After pruning, 61 SNPs were left for the analysis. The pruning criteria were described in details at Materials and Methods. By linear regression analysis of 61 SNPs with BMI and WHR, two SNPs for each trait were significant (raw P values < 0.05). Only rs431809 was significantly associated with both traits (Table 2). By case-control analysis with 387 controls and 305 obese cases, rs431809 was significantly associated with childhood obesity, with an odd ratio of 1.3165 (raw P values = 0.03). Age and sex were used as covariates for the study.
The rs431809 was located in intron 4 and had minor allele frequency was 0.1792. Major dominant genotype (GG) was 67.4%, heterozygous genotype (GT) was 29.2% and minor recessive genotype (TT) was 3.3% in all subjects (n = 692). According to rs431809 genotypes, BMI and WHR were graphed at Fig. 2. An analysis of variance test (ANOVA) of the data presented in each bar graph showed that BMI (Fig. 2A) and WHR (Fig. 2B) significantly differed in the three groups according to genotype (P < 0.05).
The bar graph of BMI and WHR depending on rs431809 genotype, GG (homozygous for major genotype), GT (heterozygous genotype), TT (homozygous for minor genotype). (A) BMI, (B) WHR. The statistical significance by ANNOVA test was indicated on the left top. The statistic significances below P value < 0.05 were also marked as a star (*).
The relationship of BMI and WHR with the rs431809 genotypes (GG: homozygous for the major genotype, GT: heterozygous, TT: homozygous for the minor genotype) are graphed in Fig. 2. There were significant changes of BMI and WHR by rs431809 with positive directions to minor genotype (TT), respectively. The differences of BMI and WHR between GG genotype to TT genotype were 1.4 kg/m2 and 0.018 (Fig. 2). rs431809 TT is minor genotype, and so minor genotype has tended to have higher BMI and WHR.
CpG methylation of childhood obesity
Mechanistic studies for rs431809 genetic association with obesity are being carried out by the investigation of CpG methylations in same intron which may be important for gene regulation. We examined three CpG sites (cg141441481, cg01852095 and cg141441470) in an intron 4 of LRP1B by pyrosequencing using bisulfite treated DNA. The methylations between the controls and the obese cases of each CpG site were graphed at cg141441481, cg01852095 and cg141441470 sites in Fig. 3. Hypermethylation from the obese group was significant in cg141441481, cg01852095 and cg141441470 (Wilcoxon test, P < 0.05). Our case-control study found that three CpG methylations were hypermethylated in obese cases by 0.76% in cg141441481, 0.34% in cg01852095, and 0.97% in cg141441470 (Fig. 3).
We interrogated the CpG methylations with obesity related traits (n = 638). The regression values with beta and P values of the methylation of cg141441481, cg01852095 and cg141441470 with BMI and WHR were listed in Table 3, respectively. All three CpG methylations were significantly association with BMI and WHR (P < 0.05). The correlation of CpG methylations with BMI and WHR was all positive, which indicate hypermethylation drive the increase of obesity related traits (Table 3).
The relationship of the CpG methylation with the rs431809 genotypes in intron 4 of LRP1B gene
To investigate genetic and epigenetic interaction, we further analyzed three CpG methylations (cg141441481, cg01852095 and cg141441470) in intron 4 of LRP1B gene. The relationship of the CpG methylation with the rs431809 genotypes (GG: homozygous for the major genotype, GT: heterozygous, TT: homozygous for the minor genotype) are graphed in Fig. 4. CpG methylations were altered dependent on the genotype of rs431809. cg141441481 and cg01852095 methylations significantly depended on rs431809 genotypes (Kruskal–Wallis test, P < 0.05), but not cg141441470. The methylation changes of three CpG sites from GG genotype to TT genotypes were the negative direction (hypomethylation) in all CpG sites, respectively (Fig. 4A–C). The difference of CpG methylation between GG genotype to TT genotype were 2.07% in cg141441481, 1.23% in cg01852095 and 1.58% in cg141441470 on the average.
The distribution of 3 CpG methylations by the genotype of rs431809. A diagram of the distribution of DNA methylation percentages of 3 CpG sites depending on the genotypes of rs431809, GG (homozygous for major genotype), GT (heterozygous genotype), TT (homozygous for minor genotype). The methylation percentage of (A) cg141441481 (B) cg01852095 (C) cg141441470 depending on the rs431809 genotypes.
The correlation significance and strength of the CpG methylation with BMI differ in rs431809 genotype
We investigated if the correlation of CpG methylations with BMI, would be dependent on rs431809 genotypes. BMI significantly correlated with cg141441481 (Fig. 5A), cg01852095 (Fig. 5B), and cg141441470 (Fig. 5C) in all subjects (Spearman test, P < 0.05). Correlational strengths were similar and positive, in all CpG sites, 0.19 in cg141441481, 0.17 in cg01852095 and 0.19 in cg141441470.
Correlations of three CpG methylations with BMI, by rs431809 genotype, TT: homozygous major genotype, GT: heterozygous genotype, GG: homozygous minor genotypes. In (A) cg141441481 (B) cg01852095 (C) cg141441470 methylation in totals, GG, GT, TT group. Correlation lines fitted on graphs with correlation coefficients (Corr.c) indicated in the top of each graph. The statistic significances below P value < 0.05 were also marked as a star (*).
We investigate CpG methylation was different on the subject’s genotype in the same intron. After dividing by rs431809 genotype, strength and significance of correlations differed by genotype (Fig. 5A–C). For correlation coefficients of GG (n = 430), GT (n = 187) and TT (n = 21) group, three CpG methylations significantly correlated with BMI in both GG and GT group. In TT group, the correlations with BMI were insignificant in three CpG sites (Spearman test, P > 0.05), and very low in cg141441481 and cg141441470 (Corr.c ≤ 0.01). Small sample size (n = 21) and lower mean value of CpG methylation in TT group may affect the significance and strength of correlation. Each CpG methylation may have different genetic dependency of the rs431809 genotype, respectively.
Discussion
Previous genetic association study of LRP1B showed strong association with obesity6,7,8,10,11. In this study, we identified novel BMI-associated SNP, rs431809. We compared the effect size of rs431808 with those of previously reported BMI-related SNPs of LRP1B gene. Effect size of rs431809 in our severe childhood obesity study was larger as 0.95 kg/m2 BMI increase compared with intronic rs12617004 with 0.02, intergenic rs2121279 with 0.024 and intergenic rs2890652 with 0.09, in adult Japanese and European studies6,7,8.
Bimodality patterns were clearly visible in three CpG sites (Figs 3 and 5). Distribution of three CpG methylations were scattered, which was classified into two predefined methylation states (high-methylated and low-methylated). rs431809 is cis-methylation quantitative trait loci of three CpG sites in same intron. A link between genetic variation and CpG methylation represent maximum level of heritability in methylation. Differential DNA methylation changes close to BMI-associated genetic loci of LRP1B may drive epigenetic change in certain environments, through epigenetic regulation of chromatin structure.
Since rs431809 is located in intron 4, its functional consequences are not clear. Intronic genetic polymorphisms with certain traits might be explained by its epigenetic regulation for gene expression in target cell23,24,25. LRP1B is a functional tumor suppressor gene in gastric cancer, and is regulated by DNA methylation13,16. Epigenetic changes including DNA methylation may be important for the expression of LRP1B in certain cells, such as the human brain, thyroid gland and skeletal muscle5. Therefore, further analysis of obesity-related differential methylation region and genetic variations of LRP1B, may explain where and how sophisticated epigenetic regulations changes come from.
In conclusion, strong correlations of rs431809 genotype with closely located CpG sites trigger the epigenetic changes, which had a dynamic role in gaining weight differed by environmental factor. This study is the first study to validate the association of rs431809 in childhood obesity related to CpG methylation. Our data suggest genetic context of the LRP1B is directly correlated with epigenetic modification of BMI-related CpG methylation. Genetic and epigenetic interplay of LRP1B gene may be implicated in the prevention and therapeutic approach to childhood obesity.
References
Liu, C. X., Musco, S., Lisitsina, N. M., Yaklichkin, S. Y. & Lisitsyn, N. A. Genomic organization of a new candidate tumor suppressor gene, LRP1B. Genomics 69, 271–274, https://doi.org/10.1006/geno.2000.6331 (2000).
Howell, B. W. & Herz, J. The LDL receptor gene family: signaling functions during development. Curr Opin Neurobiol 11, 74–81 (2001).
Ulery, P. G. & Strickland, D. K. LRP in Alzheimer’s disease: friend or foe? J Clin Invest 106, 1077–1079, https://doi.org/10.1172/JCI11455 (2000).
Hussain, M. M., Strickland, D. K. & Bakillah, A. The mammalian low-density lipoprotein receptor family. Annu Rev Nutr 19, 141–172, https://doi.org/10.1146/annurev.nutr.19.1.141 (1999).
Haas, J. et al. LRP1b shows restricted expression in human tissues and binds to several extracellular ligands, including fibrinogen and apoE-carrying lipoproteins. Atherosclerosis 216, 342–347, https://doi.org/10.1016/j.atherosclerosis.2011.02.030 (2011).
Speliotes, E. K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet 42, 937–948, https://doi.org/10.1038/ng.686 (2010).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206, https://doi.org/10.1038/nature14177 (2015).
Akiyama, M. et al. Genome-wide association study identifies 112 new loci for body mass index in the Japanese population. Nat Genet 49, 1458–1467, https://doi.org/10.1038/ng.3951 (2017).
Garver, W. S. et al. The genetics of childhood obesity and interaction with dietary macronutrients. Genes Nutr 8, 271–287, https://doi.org/10.1007/s12263-013-0339-5 (2013).
Namjou, B. et al. EMR-linked GWAS study: investigation of variation landscape of loci for body mass index in children. Front Genet 4, 268, https://doi.org/10.3389/fgene.2013.00268 (2013).
Burgdorf, K. S. et al. Association studies of novel obesity-related gene variants with quantitative metabolic phenotypes in a population-based sample of 6,039 Danish individuals. Diabetologia 55, 105–113, https://doi.org/10.1007/s00125-011-2320-4 (2012).
Cornelis, M. C. et al. Obesity susceptibility loci and uncontrolled eating, emotional eating and cognitive restraint behaviors in men and women. Obesity (Silver Spring) 22, E135–141, https://doi.org/10.1002/oby.20592 (2014).
Taylor, K. H. et al. Large-scale CpG methylation analysis identifies novel candidate genes and reveals methylation hotspots in acute lymphoblastic leukemia. Cancer Res 67, 2617–2625, https://doi.org/10.1158/0008-5472.CAN-06-3993 (2007).
Lu, Y. J. et al. Aberrant methylation impairs low density lipoprotein receptor-related protein 1B tumor suppressor function in gastric cancer. Genes Chromosomes Cancer 49, 412–424, https://doi.org/10.1002/gcc.20752 (2010).
Prazeres, H. et al. Chromosomal, epigenetic and microRNA-mediated inactivation of LRP1B, a modulator of the extracellular environment of thyroid cancer cells. Oncogene 30, 1302–1317, https://doi.org/10.1038/onc.2010.512 (2011).
Qu, Y., Dang, S. & Hou, P. Gene methylation in gastric cancer. Clin Chim Acta 424, 53–65, https://doi.org/10.1016/j.cca.2013.05.002 (2013).
Wang, Z. et al. Down-regulation of LRP1B in colon cancer promoted the growth and migration of cancer cells. Exp Cell Res 357, 1–8, https://doi.org/10.1016/j.yexcr.2017.04.010 (2017).
Xiao, D. et al. Integrative analysis of genomic sequencing data reveals higher prevalence of LRP1B mutations in lung adenocarcinoma patients with COPD. Sci Rep 7, 2121, https://doi.org/10.1038/s41598-017-02405-9 (2017).
Yoo, S., Lee, S. Y., Kim, K. N. & Sung, E. Obesity in Korean pre-adolescent school children: comparison of various anthropometric measurements based on bioelectrical impedance analysis. Int J Obes (Lond) 30, 1086–1090, 0803327
Bahk, J. & Khang, Y. H. Trends in Measures of Childhood Obesity in Korea From 1998 to 2012. J Epidemiol 26, 199–207, https://doi.org/10.2188/jea.JE20140270 (2016).
Koh, I. U., Lee, H. J., Hwang, J. Y., Choi, N. H. & Lee, S. Obesity-related CpG Methylation (cg07814318) of Kruppel-like Factor-13 (KLF13) Gene with Childhood Obesity and its cis-Methylation Quantitative Loci. Sci Rep 7, 45368, https://doi.org/10.1038/srep45368 (2017).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81, 559–575, https://doi.org/10.1086/519795 (2007).
Cheng, A. J., Mao, Y. M. & Cui, R. Z. The effect of gene polymorphism in promoter and intron 1 on human ApoA I expression. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 23, 610–613 (2006).
Hejjas, K. et al. Molecular and behavioral analysis of the intron 2 repeat polymorphism in the canine dopamine D4 receptor gene. Genes Brain Behav 8, 330–336, https://doi.org/10.1111/j.1601-183X.2008.00475.x (2009).
Zhou, Q. et al. EGFR Intron 1 polymorphism in Asian Populations and its correlation with EGFR gene expression and amplification in breast tumor tissues. Cancer Biol Ther 5, 1445–1449 (2006).
Acknowledgements
This work was supported by intramural grants (2014-NI73002-00, 2014-ND73001-00, 2016-NI73004-00 and 2017-NI73003-00) from the Korean National Institute of Health.
Author information
Authors and Affiliations
Contributions
S.L. conceived of the study and secured funding. S.L. analyzed the data and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The author declares no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, S. The genetic and epigenetic association of LDL Receptor Related Protein 1B (LRP1B) gene with childhood obesity. Sci Rep 9, 1815 (2019). https://doi.org/10.1038/s41598-019-38538-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-38538-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.