Abstract
Self-initiated voluntary acts, such as pressing a button, are preceded by a surface-negative electrical brain potential, the Bereitschaftspotential (BP), that can be recorded over the human scalp using electroencephalography (EEG). While the BP’s early component (BP1, generated in the supplementary and cingulate motor area) was linked to motivational, intentional and timing properties, the BP’s late component (BP2, generated in the primary motor cortex) was found to be linked to motor execution and performance. Up to now, the BP required to initiate voluntary acts has only been recorded under well-controlled laboratory conditions, and it was unknown whether possible life-threatening decision making, e.g. required to jump into a 192-meter abyss, would impact this form of brain activity. Here we document for the first time pre-movement brain activity preceding 192-meter bungee jumping. We found that the BP’s spatiotemporal dynamics reflected by BP1 and BP2 are comparable before 192-meter bungee jumping and jumping from 1-meter. These results, possible through recent advancements in wireless and portable EEG technology, suggest that possible life-threatening decision-making has no impact on the BP’s spatiotemporal dynamics.
Similar content being viewed by others
Introduction
The decision to perform a self-initiated act, such as releasing an arrow during Kyūdō (Japanese art of archery), pushing off for the first glide in alpine (downhill) skiing or jumping from a rock in cliff diving, requires integration and synchronization of internal and external sensory information1, as well as a sense for the opportune moment for action, a moment termed καιρός in Ancient Greek (e.g. Ilias IV, 185)2. In Greek mythology, καιρός, the youngest son of Zeus, was worshiped as a deity depicted as a young man with winged feet holding a Libra in one hand to weigh the right moment in time and a knife in the other hand to cut off ties to the past. Having only some tuft of hair on the front head while being bald on his occiput and constantly whirling around, one had to seize the right moment to grasp him by his tuft, or the right moment would be missed, possibly for ever.
The notion that success of a purposeful, self-initiated complex action depends on the right timing has its analogy in findings showing that perceptual sensitivity in the visual and auditory domain depends on brain oscillatory phase angles at which stimuli are either optimally processed or hampered. While the neural substrates of exteroceptive and interoceptive processes, such as visual and auditory perception or common sensation (“Gemeingefühl”)3, were extensively studied4, the neural substrate underlying the sense for the opportune moment for action involving integration of extero- and interoceptive processes are still widely unknown. Developing a sense for the opportune moment for a self-initiated act in a particular context (e.g., in platform or cliff diving competitions) often requires many years of training, particularly when motor initiation involves possible life-threatening decision making.
For a long time, the neural origins of self-initiated acts remained an enigma. While neurophysiological experimentations up to the 1960ies were mainly influenced by behaviourism focusing on stimulus-response paradigms, the discovery of the Bereitschaftspotential (BP) in 1964 (engl. readiness potential)5,6 signified an entirely new direction in neurophysiological research investigating the neural substrate of self-initiated voluntary acts, i.e. acts that are not triggered by external stimuli.
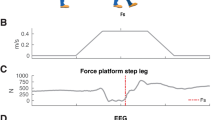
Due to the non-stationary and dynamic nature of brain activity, stimulus-response paradigms usually require averaging of brain activity over multiple trials. This can be easily achieved by using the stimulus as the common starting point (trigger) for averaging. Due to lack of a known starting point, however, such approach cannot be applied to unpredictable self-initiated movements. Building on early EEG systems that used ferromagnetic tapes to store the recorded signals, an important technological innovation and prerequisite for the discovery of the BP was to reverse such tapes and use the onset of electromyographic (EMG) activity related to a volitional act, e.g. moving a finger, as marker for reverse computation (“Rückwärtsanalyse”) of the EEG signal7. In the original experiment, EEG signals of approximately 250 trials were averaged and evidenced a surface-negative electric potential building up approximately 1.5 s before the onset of EMG activity (Fig. 1A). Study participants had to sit still in a Faraday cage with their head reclined into a headrest to avoid EEG artefacts (Fig. 1B). The recorded brain potential was most evident at the vertex electrode (i.e. at electrode position Cz according to the international 10/20 convention) and reached amplitudes of up to 10–15 μV5,6. Based on the waveform and involved generators, two phases or components of the BP can be distinguished: the “early BP” (BP1) generated by the pre-supplementary motor area (pre-SMA), SMA and lateral premotor cortex bilaterally that begins about 1.5 s before movement onset, and the “late BP” (BP2) with a steep surface-negative slope generated by the primary motor cortex (M1) and lateral premotor cortex with precise somatotopy that begins approximately 400 ms before movement onset8,9. Due to the somatotopic organization of the cortex, the BP of bilateral feet movements, e.g. during jumping, is usually not lateralized showing its maximum amplitude at Cz10.
(A) Reverse-computation („Rückwärtsanalyse“) of electroencephalographic (EEG) signals before self-initiated voluntary finger movements (right index finger flexion) evidenced a surface-negative potential shift beginning approximately 1.5 s before finger movement-related electromyographic (EMG) activity could be detected. This characteristic EEG signal was termed „Bereitschaftspotential“ (BP) (in engl. also readiness potential) by Kornhuber and Deecke5 and provided first important insights to the neural origins of self-initiated acts5,6 (superposition of 6 experiments of the same participant at 6 different days with 250 trials each, i.e. 1500 self-initiated finger movements (right index finger flexion) of the same participant B.L., adapted from Deecke, et al.39). (B) Participants had to sit still in a Faraday cage with the head reclined into a headrest to avoid EEG artefact (photograph from the original experimental setup used in Kornhuber and Deecke5, provided by Lüder Deecke, Austria).
Using a multimodal neuroimaging approach based on functional magnet resonance imaging (fMRI) and electroencephalography (EEG), Cunnington et al.11 showed a specific correlation between global electrical field power of pre-movement neural activity recorded at Cz and the metabolic (i.e. blood-oxygen-level dependent, BOLD) activity of the anterior mid-cingulate cortex (aMCC). By applying dynamic causal modelling, they found strong reciprocal interactions between the aMCC and the supplementary motor areas (SMA) that they identified to be important for the sustained activity during the early BP (BP1). Moreover, besides involvement of the cortico-basal ganglia-thalamo-cortical loop (motor loop), a number of studies indicate that the prefrontal cortex (PFC), a region that was found to be tightly linked to the concept of willpower12 (i.e. the capacity to control one’s own thoughts and actions, as well as the ability to override an unwanted thought, feeling or impulse), decision making and executive control13, plays a critical role for initiation of voluntary movements14. Particularly the medial part of the PFC (mPFC) was shown to be important for regulating neuronal circuits linked to fear and anxiety15.
While it was shown that the magnitude and waveform of the BP depend on various factors, such as force exerted, speed and precision of movement as well as pace of movement repetitions or complexity of movement16, it was unknown whether the BP also underlies movement initiation in possible life-threatening decision making, such as self-initiating a jump into a 192-meter abyss. The main reason for this gap in knowledge is that, despite numerous replications and several thousand publications since its discovery, the BP has never been recorded in a real-life situation outside the laboratory where motor initiation in possible life-threatening decision making can be investigated.
Moreover, it was unknown whether the affective anticipation associated with the decision to self-initiate a jump into a 192-meter abyss impacts the BP’s spatiotemporal dynamics. A number of studies showed that affective anticipation is signified by increased fronto-central surface-negativity17,18 found to be generated in the insular cortex19. This fear-induced surface-negativity, however, involved anticipation of an external stimulus, for instance anticipation of a mild electric shock17. Brain electric activity preceding self-initiated movements involving fearful and potentially life-threatening decision making was not studied yet.
Today, more than 50 years after the discovery of the BP, EEG technology has improved across multiple domains, particularly in terms of miniaturization, digitization, signal quality and wireless signal transmission. These advancements fostered the development of innovative neurotechnologies, such as brain-machine interfaces (BMIs) that translate brain activity into control commands of external machines, robots or computers. Recently, it was shown, for example, that severely paralyzed quadriplegics can operate an EEG-controlled brain/neural hand-exoskeleton (B/NHE) to perform activities of daily living, such as eating and drinking in an outside restaurant20. We thus reasoned that, under optimal conditions, recording of the BP outside the laboratory might be feasible, even in extreme real-life scenarios such as 192-meter bungee jumping.
Based on the model that the BP results from an unequal ratio of surface-positive and surface-negative potential shifts accumulating towards a decision threshold21,22, a threshold at which initiation of muscle contractions is inevitable (the “point-of-no-return” found to be reached approximately 200 ms before detection of electromyographic activity)23,24, we expected that affective anticipation related to possible life-threatening decision making should not impact the BP’s spatiotemporal dynamics reflected by BP1 and BP2. We thus hypothesized that the onset of BP1, the maximal amplitude of BP2 as well as waveforms of BP1 and BP2 at Cz reflecting the BP’s spatiotemporal dynamics are comparable before 192-meter bungee jumping and jumping from 1-meter.
Deriving from an ancient ritual on Vanuatu, an island in the South Pacific Ocean, bungee jumping, i.e. jumping from a tall structure while fixed to an elastic cord, has become a popular and commercialized activity with millions of jumps since the 1980ies. Besides representing a test of courage (bungee jumping was seen as an expression of boldness in ancient Vanuatu), it was shown that bungee jumping can result in a marked increase of euphoria ratings and concentration of beta-endorphin (by more than 200%) measured immediately after a bungee jump25. The risk associated with bungee jumping, which was estimated to be lower than the risk involved in riding a bicycle or dancing26, stands in stark contrast to the bungee jumper’s fear and subjective impression that jumping from such height is potentially life threatening.
Here, we report successful recording of the BP across two semi-professional cliff divers who performed several bungee jumps from a 192-meter bungee platform (the second highest bungee platform in Europe). To evaluate the impact of possible life-threatening decision making on the BP’s spatiotemporal dynamics, such as onset, maximal amplitude and waveform of components, BPs recorded before bungee jumping were compared to BPs recorded before the same participants jumped from 1-meter.
Results
Surface-negative EEG deflection before self-initiated 192-meter bungee jumping
Analysis of EEG recordings at Cz showed a surface-negative electrical trend beginning approximately 1.5 s and a clear surface-negative deflection 400 ms before movement onset of self-initiated 192-meter bungee jumping (Fig. 2A,B) evidencing successful detection of BP (Table 1) (participant 1: mean EEG deflection (±standard error): −11.63 ± 2.71 µV, t(14) = −4.29, p < 0.001, d = 1.11; participant 2: −8.37 ± 3.86 µV, t(11) = −2.17, p = 0.027, d = 0.63).
Electroencephalographic (EEG) recordings before 192-meter bungee jumping (blue) and before jumping from 1-meter (orange) evidenced a surface-negative potential shift over the vertex electrode (Cz) with the characteristic features of the Bereitschaftspotential (BP) (A) participant 1, (B) participant 2; average over all trials). The standard error is illustrated as orange and blue shaded areas, respectively. Please note that the BP has been plotted with the y-axis inverted for easier comparison with the BP illustrated by Deecke, et al.39 (Fig. 1A). (C) Movement onset before bungee jumping was detected by an accelerometer integrated into the EEG system. The solid line shows the averaged accelerometer signal across all trials of both jumpers. The standard error is indicted by the shaded area. Reverse-computation of the pre-bungee jumping BPs was time-locked to the detected movement onset. (D) One of the semi-professional cliff divers in pre-bungee jumping posture. 192-meter bungee jumps were initiated by coming up on the toes and bending forward. The EEG system was attached to the jumper’s occiput using a customized electrode cap (EasyCap®, Herrsching, Germany), adhesive tape and an elastic net dressing.
Spatiotemporal dynamics recorded before 192-meter bungee jumping vs. 1-meter jumping
Analysis of EEG recordings at Cz 400 ms before movement onset of self-initiated 1-meter jumps showed a clear surface-negative deflection (Table 1) (participant 1: mean EEG deflection (± standard error): −8.53 ± 1.68 µV, t(15) = −5.08, p < 0.001, d = 1.27; participant 2: −9.08 ± 3.70 µV, t(13) = −2.45, p = 0.015, d = 0.65). Visualization of the recorded BP’s spatiotemporal dynamics with topographical representations at selected latencies (average over all trials) showed a characteristic surface-negative deflection without any lateralization due to the bilaterality of the jumping movement (Fig. 3A,C) (Movie 1).
Left panels: Averaged EEG signals over all trials of all 8 recorded electrodes during 192-meter bungee jumping (A) and during 1-meter jumping (C). BP’s spatiotemporal dynamics of participant 1 are visualized with topographical plots at selected latencies: at −2.3 s representing the reference range, at −0.4 s characterising the transition point between ‘early’ (BP1) and ‘late BP’ (BP2), and at movement onset (0.0 s). Right panels: BP with single-trial EEG variation of participant 1 at Cz during 192-meter bungee jumping (B) and during 1-meter jumping (D). The averaged EEG signals (resulting BPs) are indicated by solid lines whereas the variation of single-trial EEGs is visualised by transparent lines.
Comparison of BP onset of BPs recorded before 192-meter bungee jumping (participant 1: median (Mdn) = −1.53 s, interquartile range (IQR) = 0.94 s; participant 2: Mdn = −1.17 s, IQR = 1.38 s) and BPs recorded before 1-meter jumping (participant 1: Mdn = −1.19 s, IQR = 0.77 s; participant 2: Mdn = −1.54 s, IQR = 1.37 s) showed no difference (participant 1: U = 93, p = 0.29, r = 0.19; participant 2: U = 94, p = 0.62, r = 0.10) (Table 2, Fig. 4).
Similarly, maximal amplitudes of BPs recorded before 192-meter bungee jumping (participant 1: mean amplitude (± standard error): −21.61 ± 2.89 µV; participant 2: −21.44 ± 6.17 µV) and before 1-meter jumping (participant 1: −19.99 ± 2.13 µV; participant 2: −20.37 ± 2.93 µV) were comparable (participant 1: t(29) = −0.46, p = 0.65, d = 0.16; participant 2: t(16) = −0.16, p = 0.88, d = 0.07) (Table 3, Fig. 5).
Differences between maximal BP amplitude recorded at Cz before 192-meter bungee jumping and jumping from 1-meter for both participants. No differences were found between jumping heights. Centerlines of boxplots show the median, while dots show the mean. Box limits indicate the 25th and 75th percentiles.
Calculating Pearson’s correlation coefficient between BP waveforms preceding 192-meter bungee jumping and 1-meter jumping evidenced highly correlated BP components for each participant (participant 1: BP1: r = 0.98, p < 0.001, BP2: r = 0.97, p < 0.001; participant 2: BP1: r = 0.88, p < 0.001, BP2: r = 0.97, p < 0.001) (Table 4) indicating that possible life-threatening decision-making does not impact BP’s spatiotemporal dynamics recorded at Cz.
Discussion
To our knowledge, this is the first report on successful recordings of the BP outside the laboratory and in the context of self-initiating a voluntary act in possible life-threatening decision making. We found that the BP can be successfully detected by averaging EEG data from less than 16 trials documenting feasibility of such recordings outside the laboratory. We found that BP onsets and maximal amplitudes recorded before self-initiated 192-meter bungee jumps and before 1-meter jumps were comparable. High correlation between BP waveforms recorded before 192-meter bungee jumping and 1-meter jumping suggests that possible life-threatening decision making has no impact on the BP’s spatiotemporal dynamics.
Catalysed by Benjamin Libet’s experiments in the 1980ies27 indicating that the conscious decision for a self-initiated act occurs not earlier than 200–250 ms before the onset of EMG activity, the discovery of the BP led to a vivid and controversial discussion about free will28 (it should be noted that neither Kornhuber and Deecke29 nor Libet ever called human freedom or free will into question30. While Kornhuber and Deecke admit that “absolute freedom from nature is an impossibility”29 leaving humans relative freedom, or freedom in degrees, Libet argued that, even if a volitional process is initiated unconsciously, the conscious function can still control the outcome by exerting a veto). While it is unlikely that neurophysiological experiments can decisively contribute to solving this controversy, feasibility of BP recordings in real-life scenarios allow now for investigating the neural mechanisms underlying self-initiated voluntary acts under realistic conditions. In this context, EEG recordings using a larger number of electrodes (≥32) and application of advanced signal processing techniques, such as connectivity14 or entropy measures31,32, may provide important insights regarding the involved brain networks and their interactions. Moreover, implementation of real-time signal processing within the framework of brain-computer interfaces (BCIs) allows answering some profound research questions, for example determining the point in time at which people are still able to cancel (“veto”) a movement after the elicitation of a BP23,24. Moreover, establishing BCIs based on motor-related cortical potentials (MRCP)33 that include the BP to detect movement intentions in everyday life environments may enhance existing BCI systems, e.g. to restore activities of daily living to quadriplegics20 or in the context of neurorehabilitation34.
The fact that emotional anticipation or fear did not impact the BP’s onset and amplitude suggests that BP might be particularly suitable to detect movement preparation even in emotionally engaging situations. Besides further substantiating that neurotechnologies, i.e. technological tools to interact with the brain, are now about to enter everyday life environments35, our report shows that brain physiological assessments previously regarded unfeasible, e.g. assessment of the BP outside the laboratory, are viable and can extend the possibility to test scientific hypotheses that were previously un-testable.
It could be argued that, in most lab-based investigations, decision-making, i.e. the decision to obey the instruction to self-initiate a particular movement, e.g. to press a button, was already concluded before the beginning of the actual experiment. If so, participants have only to decide when and how to press and not whether to press. In bungee jumping or other extreme activities that involve possibly life-threatening decision making, this decision-making process cannot be assumed to be concluded at any point in time before the actual jump. Many bungee jumping novices interrupt their first attempt and need an external trigger to overrule their instinct not to jump. Even bungee jumpers with year-long experience and dozens of jumps report that each jump requires them to overcome this instinct suggesting a decisive role of the PFC coordinating neuronal circuits related to willpower, executive functions and fear responses. While our data indicate that this coordination process does not influence the onset or waveform of the pre-bungee jumping BP, the limited number of electrodes and trials did not allow for analyses of fronto-parietal cortico-cortical interactions. Further studies are needed to elucidate these possible network interactions and their relationship to the generation of the BP. Moreover, it would be interesting to also evaluate post-movement brain potentials and their components9, an endeavour particularly challenging due to movement-related EEG artefacts (e.g. related to eye movements or muscle activity5).
The neurophysiological data reported here were recorded from semi-professional cliff divers who have never engaged in bungee jumping before. Both participants reported that they had a very strong inner resistance to jump before each trial suggesting that no relevant desensitization due to over-familiarity occurred. Due to the necessary averaging of EEG signals, however, a possible desensitization or habituation effect cannot be fully ruled out, and it is conceivable that neurophysiological data recorded from individuals who are not accustomed to high altitudes or individuals with acrophobia show different results. While the necessarily low number of trials demonstrates that the pre-bungee jumping BP is very well expressed, the small number of participants and trials constrains the possibility to exclude habituation effect and to generalize these findings towards other real-life scenarios. In this context, use of virtual reality (VR) technology may be particularly helpful as a complementary approach, not only to increase the number of trials and validate generalizability of the reported results, e.g. towards other real-life scenarios, but also to further investigate the neural origins of self-initiated acts in a controlled environment.
Methods
Experimental setup
Two semi-professional cliff divers (both males, 19 years each) who have never engaged in bungee jumping performed self-initiated voluntary jumps from two different heights: first outside the laboratory from a 192-meter bungee jumping platform (Fig. 2D) (operator: Rupert Hirner Bungee Jumping GmbH, Europa Bridge in Innsbruck, Austria; http://www.europabruecke.at/) and second under well-controlled laboratory conditions from 1-meter. Before 192-meter bungee jumping, both participants were informed by the platform operator that they were free to jump as often as they liked, and that all jumps were entirely voluntary and could be aborted at any time. Each participant performed up to 16 jumps from both heights (Table 1).
Before the jumps, specific instructions on how to perform the jumps without generating excessive muscle artefacts while generating a clear trigger signal for reverse computation were provided (e.g. keeping head motions and blinking to a minimum, relaxing the arms and trunk, initiating the jump by coming up on the toes and bending forward). Besides allowing for precise detection of movement onset due to an initial upward movement, muscle artefacts were kept at a minimum by initiating the jump with the toe extensors (the most distant muscles from the EEG recording sites).
Portable electroencephalography (EEG) with built-in accelerometer
For EEG recordings, a wireless and portable 8-channel EEG system (LiveAmp®, Brain Products GmbH, Gilching, Germany) in combination with an active electrode system (actiCAP®, Brain Products GmbH) was used. Use of active electrodes, i.e. electrodes with build-in readout circuitry that locally amplify and buffer EEG signals before transmitting the signals through cablings, ensured that the signal in the cables was insensitive to interference, e.g. due to motion36. EEG was recorded from eight conventional recording sites (Fz, FC1, FC2, C3, Cz, C4, CP1, CP2 according to international 10/10 system). Ground and reference electrodes were placed at Fpz and the mastoid, respectively. Impedance of all electrodes was kept below 5 kΩ to ensure high EEG signal quality. Since the onset of self-initiated movements is unpredictable, a built-in three-axis acceleration sensor (±2 g) provided a clear trigger signal for reverse computation. Unlike other possible movement detection methods, e.g. measuring electromyography (EMG) from leg muscles, the applied method reduced the necessary equipment and cabling to a minimum. The EEG amplifier and acceleration sensor [dimensions (w × d × h): 83 × 51 × 14 mm, overall weight: approx. 60 g (incl. built-in battery)] were fixed to the jumper’s occiput using customized electrode caps (EasyCap®, Herrsching, Germany), adhesive tape and an elastic net dressing. Besides allowing for EEG recordings for up to 3 hours, the system transmitted the recorded signals wirelessly via Bluetooth to a signal processing unit (Sony Vaio Duo 13® equipped with an Intel Core i7® processor).
Signal processing
EEG and accelerometer data were sampled at 250 Hz and band-pass-filtered at 0.1 to 3 and 5 Hz, respectively. Based on studies indicating that a band-pass filter with a lower edge of 0.1 Hz (corresponds to a time constant of 1.6 s) yields the best detection accuracy for movement related cortical potentials (MRCPs, a group of brain potentials that includes BP)37, the EEG band pass-filter was set to 0.1–3 Hz. EEG signals recorded from Cz before each jump (defined as trial) were time locked to the onset of an accelerometer signal exceeding two standard deviations recorded during movement preparation at rest (defined as detected movement onset) (Fig. 2C) and then epoched into 3-second windows (for illustration of single-trial EEG signals at Cz recorded before 192-meter bungee jumping and before 1-meter jumping see Fig. 3B,D). Epochs ranged from −2.5 s to +0.5 s with a baseline correction relative to the first 0.5 s (reference window: −2.5 s to −2.0 s). Epochs with non-physiological signal amplitudes exceeding ±100 µV or large drifts before detected movement onset were excluded from further analysis. Topographical plots of the averaged EEG signals were visualized at selected latencies (−2.3 s, −0.4 s and 0.0 s).
Outcome measures and statistics
As suggested by Shibasaki and Hallett9, successful detection of a BP was defined as surface-negative deflection of the EEG signal 400 ms before movement onset relative to a mean reference value recorded at −2.5 s to −2.0 s across all trials. To test whether there was a difference in the BP’s spatiotemporal dynamics between 192-meter bungee jumping and 1-meter jumping, averaged BP onsets, maximal BP amplitudes and BP waveforms were compared. BP onset was defined as the time point at which the EEG signal showed a continuous surface-negative deflection without further zero-crossing for more than 500 ms occurring after the reference window (−2.0 s) and movement onset (0.0 s). Maximal BP amplitude was defined as the maximal surface-negative deflection at movement onset ±100 ms. While the BP’s early component (BP1) was defined as surface-negative EEG deflection from −1.5 s to −0.4 s, the BP’s late component (BP2) was defined as surface-negative EEG deflection from −0.4 s to 0.0 s.
High comparability of averaged BP waveforms was defined as a Pearson’s r correlation coefficient higher than 0.6 (i.e. strong correlation). Normality of data was tested using a Shapiro-Wilk test. Provided normality of data, parametric Student’s t-tests were performed (one-tailed paired-sample t-tests as well as two-sample t-tests). In case normality of data could not be assumed, non-parametric Mann-Whitney-U tests were performed. Effect sizes as measured by Cohen’s d for parametric tests and Wilcoxon’s r for non-parametric tests were calculated38. For all tests, significance level was set to p ≤ 0.05.
Data Availability
All datasets are available from the corresponding author on reasonable request.
References
Gold, J. I. & Shadlen, M. N. The neural basis of decision making. Annu Rev Neurosci 30, 535–574, https://doi.org/10.1146/annurev.neuro.29.051605.113038 (2007).
Kornhuber, H. H. & Deecke, L. The Will and Its Brain: An Appraisal of Reasoned Free Will. (University press of America, 2012).
Weber, E. & Wagner, A. Handworterb. Der physi ologye 3, 481 (1846).
Craig, A. D. How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 3, 655–666, https://doi.org/10.1038/nrn894 (2002).
Kornhuber, H. H. & Deecke, L. Hirnpotentialänderungen bei Willkürbewegungen und passiven Bewegungen des Menschen: Bereitschaftspotential und reafferente Potentiale. Pflügers Arch 284, 1–17 (1965).
Kornhuber, H. H. & Deecke, L. Brain potential changes in voluntary and passive movements in humans: readiness potential and reafferent potentials. Pflugers Arch – Eur J Physiol 468, 1115–1124, https://doi.org/10.1007/s00424-016-1852-3 (2016).
Kornhuber, H. H. & Deecke, L. Hirnpotentialänderungen beim Menschen vor und nach Willkürbewegungen, dargestellt mit Magnetbandspeicherung und Rückwärtsanalyse. Pflügers Arch 281, 52 (1964).
Deecke, L. & Kornhuber, H. H. In The Bereitscha ftspotential, movement-related cortical potentials (eds Jahanshahi, M. & Hallett, M.) 283–320 (Kluwer Academic/Plenum Publishers New York, 2003).
Shibasaki, H. & Hallett, M. What is the Bereitschaftspotential? Clin Neurophysiol 117, 2341–2356, https://doi.org/10.1016/j.clinph.2006.04.025 (2006).
Boschert, J. & Deecke, L. Cerebral potentials preceding voluntary toe, knee and hip movements and their vectors in human precentral gyrus. Brain Res 376, 175–179 (1986).
Nguyen, V. T., Breakspear, M. & Cunnington, R. Reciprocal interactions of the SMA and cingulate cortex sustain premovement activity for voluntary actions. J Neurosci 34, 16397–16407, https://doi.org/10.1523/JNEUROSCI.2571-14.2014 (2014).
Knoch, D. & Fehr, E. Resisting the power of temptations: the right prefrontal cortex and self-control. Ann N Y Acad Sci 1104, 123–134, https://doi.org/10.1196/annals.1390.004 (2007).
Domenech, P. & Koechlin, E. Executive control and decision-making in the prefrontal cortex. Curr Opin Behav Sci 1, 101–106, https://doi.org/10.1016/j.cobeha.2014.10.007 (2015).
Kim, K., Kim, J. S. & Chung, C. K. Increased Gamma Connectivity in the Human Prefrontal Cortex during the Bereitschaftspotential. Front Hum Neurosci 11, 180, https://doi.org/10.3389/fnhum.2017.00180 (2017).
Tovote, P., Fadok, J. P. & Luthi, A. Neuronal circuits for fear and anxiety. Nat. Rev. Neurosci. 16, 317–331, https://doi.org/10.1038/nrn3945 (2015).
Benecke, R., Dick, J. P., Rothwell, J. C., Day, B. L. & Marsden, C. D. Increase of the Bereitschaftspotential in simultaneous and sequential movements. Neurosci Lett 62, 347–352 (1985).
Rockstroh, B., Elbert, T., Canavan, A., Lutzenberger, W., Birbaumer, N. Slow Cortical Potentials and Behaviour. 267 pp (Urban and Schwarzenberg, 1989).
Lang, P. J., Bradley, M. M. & Cuthbert, B. N. Emotion, attention, and the startle reflex. Psychol Rev 97, 377–395 (1990).
Bocker, K. B., Brunia, C. H. & van den Berg-Lenssen, M. M. A spatiotemporal dipole model of the stimulus preceding negativity (SPN) prior to feedback stimuli. Brain Topogr 7, 71–88 (1994).
Liew, S. L. et al. Improving Motor Corticothalamic Communication After Stroke Using Real-Time fMRI Connectivity-Based Neurofeedback. Neurorehab Neural Re 30, 671–675, https://doi.org/10.1177/1545968315619699 (2016).
Jo, H. G., Hinterberger, T., Wittmann, M., Borghardt, T. L. & Schmidt, S. Spontaneous EEG fluctuations determine the readiness potential: is preconscious brain activation a preparation process to move? Exp Brain Res 231, 495–500, https://doi.org/10.1007/s00221-013-3713-z (2013).
Schurger, A., Mylopoulos, M. & Rosenthal, D. Neural Antecedents of Spontaneous Voluntary Movement: A New Perspective. Trends Cogn Sci 20, 77–79, https://doi.org/10.1016/j.tics.2015.11.003 (2016).
Schultze-Kraft, M. et al. The point of no return in vetoing self-initiated movements. Proceedings of the National Academy of Sciences, USA 113, 1080–1085, https://doi.org/10.1073/pnas.1513569112 (2016).
Deecke, L. & Soekadar, S. R. Beyond the point of no return: Last-minute changes in human motor performance. P Natl Acad Sci USA 113, E2876, https://doi.org/10.1073/pnas.1604257113 (2016).
Hennig, J., Laschefski, U. & Opper, C. Biopsychological changes after bungee jumping: β-endorphin immunoreactivity as a mediator of euphoria? Neuropsychobiology 29, 28–32 (1994).
Lief, E. Bungee Jumping And The Art Of Risk Assessment, https://www.acsh.org/news/2016/08/19/bungee-jumping-and-the-art-of-risk-assessment (2016).
Libet, B., Gleason, C. A., Wright, E. W. & Pearl, D. K. Time of conscious intention to act in relation to onset of cerebral activity (readiness-potential) the unconscious initiation of a freely voluntary act. Brain 106, 623–642 (1983).
Dennett, D. C. Freedom Evolves. (Penguin Books, 2004).
Deecke, L. There Are Conscious and Unconscious Agendas in the Brain and Both Are Important—Our Will Can Be Conscious as Well as Unconscious. Brain sci 2, 405–420 (2012).
Libet, B. Do we have free will? Journal of consciousness studies 6, 47–57 (1999).
Schreiber, T. Measuring information transfer. Phys Rev Lett 85, 461–464, https://doi.org/10.1103/PhysRevLett.85.461 (2000).
Robinson, S. E., Mandell, A. J. & Coppola, R. Spatiotemporal imaging of complexity. Front Comput Neurosci 6, 101, https://doi.org/10.3389/fncom.2012.00101 (2012).
Mrachacz-Kersting, N. et al. The effect of type of afferent feedback timed with motor imagery on the induction of cortical plasticity. Brain Res 1674, 91–100, https://doi.org/10.1016/j.brainres.2017.08.025 (2017).
Soekadar, S. R., Birbaumer, N., Slutzky, M. W. & Cohen, L. G. Brain-machine interfaces in neurorehabilitation of stroke. Neurobiol Dis 83, 172–179, https://doi.org/10.1016/j.nbd.2014.11.025 (2015).
Clausen, J. et al. Help, hope, and hype: Ethical dimensions of neuroprosthetics. Science 356, 1338–1339, https://doi.org/10.1126/science.aam7731 (2017).
Xu, J., Mitra, S., Van Hoof, C., Yazicioglu, R. F. & Makinwa, K. A. A. Active Electrodes for Wearable EEG Acquisition: Review and Electronics Design Methodology. IEEE Rev Biomed Eng 10, 187–198, https://doi.org/10.1109/RBME.2017.2656388 (2017).
Jiang, N., Mrachacz-Kersting, N., Xu, R., Dremstrup, K. & Farina, D. In Brain-Computer Interface Research: A State-of-the-Art Summary 3 (eds Christoph Guger, Theresa Vaughan, & Brendan Allison) 47–61 (Springer International Publishing, 2014).
Cohen, J. A power primer. Psychol Bull 112, 155–159 (1992).
Deecke, L., Grözinger, B. & Kornhuber, H. H. Voluntary finger movement in man: cerebral potentials and theory. Biol Cybern 23, 99–119 (1976).
Acknowledgements
We thank Manuel Halbisch and Pascal Pollin for their willingness to have their brain activity recorded before their bungee jumps, and Patrick Britz, Luke Enge, Liam Scannell and Thomas Emmerling (Brain Products GmbH, Gilching, Germany) for their support in preparing and conducting the reported physiological recordings. We’d also like to thank Rupert Hirner and his team for their exceptional support during the measurements. S.R.S. received special support by the European Research Council (ERC-2017-STG-759370), Einstein Foundation Berlin, and Brain & Behavior Research Foundation as 2017 NARSAD Young Investigator Grant recipient and P&S Fund Investigator. M.N. was supported by the Baden-Württemberg Stiftung (NEU007/1). Open access to this work was supported by the Deutsche Forschungsgemeinschaft (DFG) and Open Access Publishing Fund of the University of Tübingen.
Author information
Authors and Affiliations
Contributions
M.N. collected and analysed the data. M.N., L.D. and S.R.S. interpreted the data, performed the literature search and wrote the manuscript. M.N. and S.R.S created the figures. M.N., L.G.C., L.D. and S.R.S. edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nann, M., Cohen, L.G., Deecke, L. et al. To jump or not to jump - The Bereitschaftspotential required to jump into 192-meter abyss. Sci Rep 9, 2243 (2019). https://doi.org/10.1038/s41598-018-38447-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-38447-w
This article is cited by
-
Free will and neurosurgical resections of the supplementary motor area: a critical review
Acta Neurochirurgica (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.