Abstract
During pregnancy, the woman’s body undergoes tremendous changes in immune system adaptation. The immunological shifts that occur in pregnancy can partially be explained by alterations in hormonal levels. Furthermore, during pregnancy, many autoimmune diseases go into remission, only to flare again in the early postpartum period. Given these important changes in the clinical course of a number of autoimmune disorders, surprisingly little has been done to investigate the inflammatory profile changes across pregnancy and the postpartum period. Thus, the aim of this study was to describe how inflammatory and anti-inflammatory markers change from late pregnancy to the early postpartum period, using a multiplexed assay consisting of both well-known as well as exploratory proteins. Two-hundred-and-ninety women were included in this study and donated a total of 312 blood samples; 198 in late pregnancy (~gw38) and 114 in the postpartum period (~w8). The plasma blood samples were analyzed for 92 immune system related protein markers using Proseek Multiplex Inflammation I panel, a high-sensitivity assay based on proximity extension assay technology. Fifty-six inflammatory and anti-inflammatory markers were significantly different between pregnancy and the postpartum, of which 50 survived corrections for multiple comparisons. Out of these 50 markers, 41 decreased from pregnancy to postpartum, while the remaining 9 increased in the postpartum period. The top five markers with the greatest decrease in the postpartum period were Leukemia inhibitory factor receptor (LIF-R), Latency-associated peptide Transforming growth factor beta-1 (LAP TGF-beta-1), C-C motif chemokine 28 (CCL28), Oncostatin M (OSM) and Fibroblast growth factor 21 (FGF21). Top three markers that increased in the postpartum period were Tumor necrosis factor ligand superfamily member 11 (TRANCE), Tumor necrosis factor ligand superfamily member 12 (TWEAK), and C-C motif chemokine/Eotaxin (CCL11). This study revealed that the majority of the markers decreased from pregnancy to postpartum, and only a few increased. Several of the top proteins that were higher in pregnancy than postpartum have anti-inflammatory and immune modulatory properties promoting pregnancy progress. These results clearly reflect the tremendous change in the immune system in the pregnancy to postpartum transition.
Similar content being viewed by others
Introduction
During pregnancy, the woman’s body undergoes tremendous changes in immune system adaptation. At the same time as the fetus needs to be protected against pathogens, the female body needs to maintain a tolerance to paternal alloantigens in order to prevent rejection of the fetus1.
The immunological shifts that occur in pregnancy can partially be explained by alterations in hormonal levels, including progesterone, estradiol, and other proteins like leukemic inhibitory factor (LIF), as well as prostaglandins2,3. While pregnancy mainly is characterized by an anti-inflammatory immunological tolerance, inflammatory events take place during different phases of pregnancy, including implantation, placentation and in preparation for delivery. Implantation is characterized by increased levels of pro-inflammatory chemokines, cytokines and growth factors4. The immune system response at this stage mainly involves mast cells, dendritic cells, monocytes and macrophages. Of importance, macrophages can present in different forms, M1-macrophages and M2-macrophages, and the switch from M1- to M2- decidual macrophages is mediated by interleukin (IL)-10 and macrophage colony-stimulating factor (M-CSF)5. Thus, the first stage of pregnancy is dominated by M1-macrophages, which promote inflammation by metabolizing the amino acid arginine to nitric oxide, a molecule toxic to pathogens, and by secreting a number of pro-inflammatory cytokines such as Tumor necrosis factor (TNF)-α, IL-6 and IL-1β4,6.
During placental development, the previous dominance of M1-macrophages is succeeded by a more anti-inflammatory M2-milieu7. M2-macrophages secrete IL-10 and Transforming growth factor (TGF)-β and promote tissue-healing8. The M2-milieu continues into the second and third trimester with anti-inflammatory dominance, and the second trimester is characterized by rapid fetal growth and protection against preterm contractions7. In addition to the importance of the M1-M2 balance, the T lymphocyte profile plays an important role in the maintenance of pregnancy. Both hormonal changes and placental trophoblast immunomodulatory molecules are believed to play a role in the switch to a predominantly T helper type 2 (Th2) cell profile. Th2 and regulatory T (Treg) cells inhibit, by production of IL-4 and IL-10, the allograft rejection promoted by T helper type 1 (Th1) and T helper type 17 (Th17) cells. Moreover, Tregs are important in the maintenance of pregnancy, probably due to their production of IL-10 and TGF-β9,10. Further, pregnancy, with its associated hormones rising, might negatively regulate sub-populations of B cell development to avoid autoimmunity and rejection of the fetus, while enhancing antibody production in other sub-populations of B cells responsible for protection against pathogens11.
In preparation of delivery, another shift back to a pro-inflammatory state occurs. Cells of the immune system start migrating into the myometrium and high levels of pro-inflammatory cytokines have been found both in the cervical tissue and in the peripheral blood12,13,14,15. M1 macrophages in the uterus contribute to an inflammatory profile promoting uterine contractions, delivery of the baby, expulsion of the placenta, and uterine involution16,17.
In women, the reproductive years coincide with the period when several autoimmune disorders have their peak onset. Furthermore, during pregnancy, many autoimmune diseases go into remission, only to flare again in the early postpartum period. Typically, Th1 and Th17-type autoimmune disorders improve as a result of the rise in Th2-type cytokines in pregnancy (e.g. rheumatoid arthritis, Graves’ disease, the autoimmune neuromuscular myasthenia gravis (MG) and multiple sclerosis (MS)18,19,20,21), whereas Th2-type autoimmune disorders may deteriorate in pregnancy (e.g. systemic lupus erythematosus22). In addition, immunosuppressive regulatory B cells have been shown to increase during pregnancy, of potential relevance for the clinical course of the autoimmune disorders23. After delivery, the immune system returns to a non-pregnant state. A shift towards Th1 dominance, and a fall in Th2 and Treg cells, followed by altered cytokine pattern in the first weeks following delivery have been reported1,24,25. All of these changes may result in the worsening of Th1 and Th17-type autoimmune diseases in the postpartum period9.
Given these important changes in the clinical course of a number of autoimmune disorders, surprisingly little has been done to investigate the inflammatory profile changes across pregnancy and the postpartum period. For instance, previous studies have merely focused on a limited number of inflammatory and anti-inflammatory markers, primarily IL-6, IL-8 TNF-α, IL-1β and IL-1026,27,28,29 and a more comprehensive picture of differences in inflammatory profile in pregnancy versus postpartum is still missing. Thus, the aim of this study was to describe how inflammatory and anti-inflammatory markers change from late pregnancy to the early postpartum period, using a high-sensitivity multiplexed assay consisting of both well-known and exploratory proteins.
Materials and Methods
Participants
This sub-study is part of an ongoing longitudinal cohort-project, started in 2009, named the BASIC-study (Biology, Affect, Stress, Imaging and Cognition) with the aim to study peripartum depression. All Swedish speaking pregnant women over 18 years of age without confidential personal data, scheduled for a routine ultrasound at Uppsala University hospital are asked to participate in the study.
Following informed consent, information on ongoing medical and obstetric conditions, use of medication, smoking habits, pre-pregnancy body mass index and life style factors are collected by online surveys filled in by the women at gestational week 17 and 32 and at 6 weeks and 6 months postpartum. In addition to the background characteristics, the Edinburgh postnatal depression scale (EPDS) is included in all surveys.
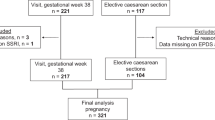
For the purpose of the present study, we used blood samples obtained from two different sources within the BASIC framework, obtained between 2010 and 2013. First, blood samples from women, participating in a nested case-control sub-study, in gestational week 38 (median 19 (IQR 24-13) days before partus) or at 8 weeks (median 69.5 (IQR 62–77) days after partus) postpartum, or both, were used. Women invited for the sub-study comprised a selection of women with depressive symptoms according to the online EPDS survey, women on antidepressant treatment, and a corresponding number of women without depressive symptoms. Upon inclusion, the women were asked to fill in the EPDS again and were interviewed with MINI International Neuropsychiatric Interview30. Blood samples were drawn after at least 90 minutes fasting. The second source of blood samples were derived from women in the BASIC-study who underwent planned cesarean section (CS), in gestational week 38 (median 8 (IQR 11–5) days before ultrasound-determined date of partus), at Uppsala University hospital. In these women, venous blood samples were collected prior to surgery, following overnight fasting. Independent of source, all blood samples were centrifuged at 1500 RCF for 10 minutes and stored in −70 °C within one hour. Further, all blood samples were collected between 8:00 and 15:00, with the majority of samples drawn around 9:00 or 13:00. These conditions were similar for the samples obtained during pregnancy and postpartum.
Five hundred–and-thirty plasma samples from these two parts of the BASIC-study were analyzed, representing 482 unique women. As previous studies in our group had found profound changes in inflammatory and anti-inflammatory markers in women with antenatal depression31, we excluded 139 women with EPDS scores ≥13 at any time-point during pregnancy or ≥12 at any time-point during the postpartum period32,33. In line with this reasoning, we also excluded 33 women who were on antidepressant treatment at the time of blood sampling and 8 women with missing information on EPDS scores. Further, we excluded two duplex pregnancies, two women on oral corticosteroid treatment, one woman with preeclampsia, and finally 7 samples that could not be analyzed for technical reasons. Women with inflammatory/autoimmune disorders (celiac disease, psoriatic arthritis, ulcerative colitis and Crohn’s disease) in remission, i.e. not requiring corticosteroid treatment, were kept in the study but were adjusted for in the statistical analyses.
All in all, 290 women were included in this sub-study and donated a total of 312 blood samples; 198 in late pregnancy (129 in the case-control sub-study and 69 from the CS group) and 114 in the postpartum period. Given this, 22 women had donated samples in both pregnancy (19 in the case-control sub-study and 3 from the CS group) and postpartum.
The study procedures were in accordance with ethical standards for human experimentation and the study was approved by the Regional Ethical Review Board in Uppsala.
Proximity extension assay
The plasma blood samples were analyzed for 92 immune system related protein markers at the Clinical Biomarker Facility at SciLifeLab Uppsala using Proseek Multiplex Inflammation I panel (Olink Bioscience, Sweden) (https://www.olink.com/products/inflammation/biomarkers/), which is based on proximity extension assay (PEA) technology34,35. The PEA uses two oligonucleotide-labeled antibodies for each target protein. When the two antibodies are in close proximity, a new polymerase chain reaction (PCR) target sequence is formed by proximity-dependent DNA polymerization. The resulting sequence is subsequently detected and quantified using real-time PCR.
Plasma samples were transferred to 96-well plates, each consisting of 90 samples and 6 controls (three negative controls (buffer) and three interplate controls). Each plasma sample (1 µL) was mixed with 3 µL incubation mix containing 92 antibody pairs, spiked in with two incubation controls (green fluorescent protein and phycoerythrin), one extension control and one detection control, and allowed to incubate at 4 °C overnight. Further, 96 µL extension mix, containing PEA enzyme and PCR reagents, was added, and the samples were incubated for 5 min at room temperature before the plate was transferred to the thermal cycler for an initial DNA extension at 50 °C for 20 min, followed by 17 cycles of DNA amplification. A 96.96 Dynamic Array IFC (Fluidigm, South San Francisco, CA, USA) was prepared and primed according to the manufacturer’s instructions. In a new plate, 2.8 µL of sample mixture was mixed with 7.2 µL detection mix from which 5 µL was loaded into the right side of the primed 96.96 Dynamic Array IFC.
Results were presented as Normalized protein expression (NPX), which corresponds to log2(expression), obtained in GenEx software using Olink Wizard by normalizing Cq-values against extension control, interplate control and a correction factor, and NPX corresponds to relative quantification between samples. The extension control is subtracted from the Cq-value of every sample in order to correct for technical variation and the interplate control is subtracted to compensate for possible variation between runs. Finally, the NPX is determined by normalization against a calculation correction factor that accounts for background noise.
The assay has sensitivity down to fg/mL and detects relative protein values that can be used for comparison between groups, although not for absolute quantification. Limit of detection (LOD) was determined for each biomarker based on the mean value of triplicate negative controls analyzed in each run. According to a notification from the company, brain-derived neurotrophic factor (BDNF) has been excluded from the Inflammation panel I because of a potential technical issue. For this reason, we discarded the results from the BDNF assay in our study.
Statistics
Only markers that had NPX values higher than the limit of detection (LOD) for more than 50% of the women in pregnancy and postpartum were used in the analyses, leaving 70 markers for the statistical analyses. Included markers are presented in Supplementary Table 1, and excluded markers in Supplementary Table 2. The markers are presented with the percentage of samples with detectable protein levels. For handling of values below level of detection (LOD), the NPX values <LOD were replaced by LOD/square root(2)36.
Demographic data were compared using Chi-square tests, T-test and Mann-Whitney U-test as suited. Inflammatory and anti-inflammatory markers were compared between pregnancy and postpartum using Linear Mixed models, by which the cross-sectional and longitudinal design of the study was fully utilized. These analyses were adjusted for age (continuous), pre-pregnancy BMI (continuous), use of antibiotics at blood sampling (yes/no), and chronic inflammatory or rheumatic diseases (yes/no). Bonferroni correction was applied to test for multiple comparisons. The results from the Linear Mixed model were validated by Wilcoxon paired tests in the subset of 22 women who had donated a blood sample both during pregnancy and in the postpartum period.
All statistical analyses were performed using IBM SPSS statistics version 24.0. P-values below 0.05 were considered a statistical significant difference.
Results
Demographic and clinical variables of the study population are presented in Table 1. The mean age of the women in this study was approximately 32 years, and the mean body mass index (BMI) before pregnancy was around 23 kg/m2. No differences were found in parity, assisted reproduction in terms of in vitro fertilization, smoking habits, inflammatory/rheumatoid diseases, use of antibiotics, preterm birth, or offspring gender. EPDS scores were slightly higher in the pregnancy group although within the range considered non-depressed. Among the postpartum women, 107 (93.8%) were breastfeeding. Breastfeeding status did not affect the overall results obtained from this study, data not shown.
Fifty-six inflammatory and anti-inflammatory markers were significantly different between pregnancy and the postpartum, of which 50 survived corrections for multiple comparisons (Tables 2 and 3). Out of these 50 markers, 41 were higher in pregnancy (Table 2), while the remaining 9 were higher in the postpartum period (Table 3).
Among the 22 participants who contributed with samples in both late pregnancy and postpartum, all but two markers (Chemokine (C-X-C motif) ligand 9 (CXCL9) and Cluster of Differentiation 6 (CD6)) were confirmed significant in the Wilcoxon test.
The top five markers with the greatest decrease in the postpartum period were Leukemia inhibitory factor receptor (LIF-R), Latency-associated peptide Transforming growth factor beta-1 (LAP TGF-beta-1), C-C motif chemokine 28 (CCL28), Oncostatin M (OSM) and Fibroblast growth factor 21 (FGF21) (top three markers shown in Fig. 1a). Other well-studied markers that also decrease in the postpartum period include IL-6 and IL-10.
Top three markers that increased in the postpartum period were Tumor necrosis factor ligand superfamily member 11 (TRANCE), Tumor necrosis factor ligand superfamily member 12 (TWEAK), and C-C motif chemokine/Eotaxin (CCL11) (Fig. 1b).
Discussion
In this study we have statistically analyzed 70 inflammatory and anti-inflammatory markers in non-depressed women and found that the NPX values of 56 markers significantly differed from late pregnancy to postpartum. After correcting for multiple testing, 41 markers significantly decreased while nine markers increased in the postpartum period. The results of the paired analyses further indicate the robustness of the results. The results of this largely exploratory study on inflammatory and anti-inflammatory markers confirm, using high-sensitive proximity extension assays, the tremendous changes in immune function and inflammation status during the perinatal period. Further emphasizing the change, these decreases in inflammatory and anti-inflammatory markers were evident despite the 20% increase in plasma volume during third trimester pregnancy.
The pro- or anti-inflammatory profile of individual markers is not always easy to establish, as many of the markers have differential profiles in distinct cell-signaling pathways, tissues, or even physiological conditions37. Nevertheless, some typical pro-inflammatory markers are TNF-α, IL-6 and IL-1β, while IL-4, IL-10 and TGF-β on the other hand have a more prominent anti-inflammatory profile37. Even though the literature on inflammatory and anti-inflammatory marker changes from pregnancy to postpartum is limited, some of our results are in line with previous studies. A decrease of CXCL10 and an increase of CXCL9 from late pregnancy to early postpartum have been described previously, which is in line with our study results38. Decreasing levels of IL-10 from pregnancy to postpartum have been reported in an allergen-induced IL-10 production in vitro model39. A non-significant difference in levels of IL-8 has been described27, while another study found IL-8 to have a U-shaped curve with increasing levels postpartum28. The findings from previous studies of IL-6 are inconclusive. IL-6 has been reported, in line with our results, to decrease27, but also in contradiction to our study, to increase28 or to show no significant difference26 from pregnancy to postpartum. However, the differences in the direction of levels of inflammatory and anti-inflammatory markers from pregnancy to postpartum observed, might be due to the different sampling time points. Unfortunately, some of the markers mentioned in the literature (TNF-α and IL-1β26,27,28) were excluded from further analyses due to NPX values below LOD for more than 50% of the women in this study (IL-6), or not even included in the panel (IL-1β).
Pregnancy requires substantial alterations of multiple physiological systems in the female body, and in particular the immune system. In the postpartum period, these systems return to baseline levels. The top five markers that decreased postpartum were LIF-R, LAP TGF-beta-1, CCL28, OSM, and FGF21. LIF-R acts as a receptor for the cytokines LIF and OSM, both members of the IL-6 family40. LIF has anti-inflammatory properties and stimulates T regulatory cells (Tregs)41, and acts in a pleiotropic manner via the LIF-R on pituitary corticotrophic cells, macrophages, blastocysts, embryos, and in the placenta42 suggesting that higher levels of LIF-R and OSM are needed during pregnancy. LIF is described within reproduction to play an important role in implantation, embryo development, and by sufficient levels preserving the early pregnancy42,43. OSM seems to have both pro-inflammatory and anti-inflammatory properties44. OSM also possess pleiotropic actions, but seems less important within reproduction, as OSM knock-out mice appear to stay fertile45. OSM has been suggested to act balancing on adipogenesis/lipogenesis in peripheral organs46, and treatment with OSM seems to improve for example obesity and adipose tissue inflammation in mice47. High levels of LIF-R48,49 and low levels of LIF49 have previously been reported in pregnancy. As a whole, the LIF-R and its ligands are involved in metabolism, which may be of importance also within reproduction and preservation of an early pregnancy, which further supports the hypothesis regarding the need for higher levels of LIF-R and OSM during pregnancy than postpartum. LAP TGF-beta-1 refers to the latency-associated peptide to the TGF-beta-1 protein, that is required for several functions such as efficient secretion, prevention of binding to cell surface receptors, and extracellular availability50,51. Previous studies describe both LAP TGF-beta-1 and TGF-beta-1 as having immunosuppressive properties52,53. TGF-beta-1 has been shown to be involved in processes important in pregnancy, such as trophoblast invasion and proliferation, angiogenesis, and tolerance to the semi-allogeneic fetus54, suggesting even in this case a mechanism involving elevated levels during pregnancy. CCL28 (Mucosa associated epithelial chemokine (MEC)) is a chemokine that has antimicrobial and immunomodulatory properties, and is suggested to act as a bridge between the innate and the adaptive immunity55. Expression of CCL28 by epithelial cells, followed by an induction of pro-inflammatory cytokines/infection, helps in the recruitment of T-lymphocytes, and leads to accumulation of for example Tregs on mucosal surfaces56. CCL28 is suggested to have anti-inflammatory properties by limiting autoimmunity and inflammation at these sites. Elevated levels of CCL28 are also described in inflammatory diseases, such as rheumatoid arthritis57. CCL28 has been found to induce apoptosis in decidual stromal cells, and a higher expression of the receptors of CCL28 (CCR3, CCR10), in these cell populations in spontaneous abortion is displayed prior to a pro-inflammatory stimulation58. Interestingly, in mouse studies, CCL28 has been shown to be upregulated during lactation59, while in the current study, blood levels were lower in the postpartum period. FGF21 acts as an anti-inflammatory and immunoregulatory agent, both in vivo and in vitro60. Elevated plasma levels of FGF21 have been reported in pregnant women, and a positive impact of FGF21 on cardiac development in mice embryos has been described61. Further, FGF21 has been suggested to improve metabolism of lipids and glucose during pregnancy62, which could explain a need for increased levels observed during pregnancy.
The postpartum period implies extraordinary physiological processes of wound healing, lactation and sleep deprivation. The top three markers that increased postpartum were TRANCE, TWEAK, and CCL11. TRANCE is a member of the tumor necrosis factor (TNF) superfamily (TNFSF11) and can also be named RANKL63. TRANCE/RANKL is secreted by T cells and regulates osteoclastogenesis and bone remodeling64 and have been found elevated in mice induced with sleep deprivation65. TRANCE/RANKL secreted from human trophoblasts has also been found to polarize decidual macrophages from M1 to M2 phenotype, and decreased levels of RANKL has been noted in decidual cells from women with miscarriage66. Further, TRANCE is suggested to have an important role in the function of the mammary glands and for lactation in mice67, supporting the evidence of elevated levels of TRANCE observed postpartum. TWEAK is also a member of the tumor necrosis factor (TNF) superfamily (TNFSF12), and has been described within chronic inflammation, angiogenesis and fibrosis68. TWEAK is a cytokine expressed by leucocytes, monocytes, dendritic cells and natural killer cells69 and is suggested to be essential in tissue repair following injury70 which might explain the elevated levels observed in postpartum women in this study. However, in mice TWEAK is expressed by decidual NK cells, and a down-regulation of TWEAK may contribute to uterine NK cell cytotoxicity and fetal rejection71. CCL11 (Eotaxin-1) is a cytokine that recruits eosinophils and therefore involved in allergic responses72. While CCL11 has pro-invasive properties and promotes migration and invasion of trophoblasts into the uterine wall73,74 during pregnancy, CCL11 recruits eosinophils, neutrophils and basophiles75 which all play important roles in the vital tissue healing and remodeling during the postpartum period.
Notably, the two excluded markers Programmed cell death 1 ligand 1 (PD-L1) and Artemin (ARTN) switched from detectable in late pregnancy to almost undetectable in the postpartum period. This could either be due to some unknown technical issue regarding the sampling of postpartum samples, or these two markers have the most drastic decrease from pregnancy to postpartum. PD-L1 has a role in regulating T-cell homeostasis and promoting Tregs for maintenance of peripheral tolerance in pregnancy76. ARTN has been found in the maternal reproductive tract and embryos, suggesting its’ importance in early embryo development and pregnancy77.
The profound immunological alterations that occur from late pregnancy to postpartum in both pro-and anti-inflammatory proteins could play a role in the cyclic pattern of either improvement or onset/deterioration of several autoimmune diseases during pregnancy or after childbirth. In particular, Tregs are known to be defect in the autoimmune neuromuscular disorder myasthenia gravis (MG), where the risk of onset is dramatically increased in the postpartum period78. Even though none of the MG specific inflammatory proteins79 were found to be elevated in the postpartum period of healthy women, five of these proteins (TGF-α, β-NGF, IL-6, IL-17C and IL-10) were significantly higher in late pregnancy in the present study. However, the possible involvement of these inflammatory proteins in the elevated risk for postpartum onset of MG would have to be confirmed by prospective studies. Additionally, disease activity in the neuroimmune disorders MS, MG and neuromyelitis optica undergoes pronounced shifts during and after pregnancy78,80. The most established example is a reduction in relapse rates in the last trimester by 70–80%. Nevertheless, disease activity reappears in the first few months after delivery, temporarily overshooting pre-pregnancy levels. There are also differences in the different trimesters of pregnancy; since clinical disease activity is usually aggravated in Graves’ hyperthyroidism and MG in early pregnancy and worsening may also occur after delivery. Patients with rheumatoid arthritis experience improved clinical status during pregnancy, which is accompanied by a reduced pro-inflammatory profile81. Since the numbers of participants with inflammatory or rheumatic diseases in this study were very low (6 in pregnancy and 1 postpartum), we refrained from analyzing their inflammatory status separately. Thus, clarifying different autoimmune disease spectra of pro-and anti-inflammatory proteins between pregnancy and postpartum were out of the scope of the current study.
The sensitive PEA method used in combination with the large number of markers investigated, are among the strengths of this study. Cytokines are difficult to measure in the circulation due to their low concentrations. Hopefully, the highly sensitive method we used provides more robust results than previous studies, most of them being based on immunoassays. The major limitation of this study is the cross-sectional design with a low number of women followed prospectively. Additional sampling time-points could have given strengths to the study.
In conclusion, this study revealed 41 markers to decrease from late pregnancy to postpartum while nine markers increased. These results clearly reflect the tremendous change in the immune system in the pregnancy to postpartum transition. Several of the top proteins that were higher in pregnancy than postpartum have anti-inflammatory and immune modulatory properties promoting pregnancy progress. LIF-R and LAP TGF-beta-1, the top two markers, are well-characterized proteins within reproductive immunology and of much interest in peripartum research.
With greater understanding of the inflammatory changes the pregnant body undergoes, greater effort could be made in finding causes and treatment to pregnancy and postpartum complications and diseases thought to be related to immune function and inflammation, such as preeclampsia82, preterm birth15 and perinatal depression26, but even autoimmune disorders.
Data Availability
The data generated during the current study are available in the Zenodo repository 10.5281/zenodo.1249367.
References
La Rocca, C., Carbone, F., Longobardi, S. & Matarese, G. The immunology of pregnancy: regulatory T cells control maternal immune tolerance toward the fetus. Immunol Lett 162, 41–48, https://doi.org/10.1016/j.imlet.2014.06.013 (2014).
Mjosberg, J., Berg, G., Jenmalm, M. C. & Ernerudh, J. FOXP3+ regulatory T cells and T helper 1, T helper 2, and T helper 17 cells in human early pregnancy decidua. Biol Reprod 82, 698–705, https://doi.org/10.1095/biolreprod.109.081208 (2010).
Sykes, L., MacIntyre, D. A., Yap, X. J., Teoh, T. G. & Bennett, P. R. The Th1:th2 dichotomy of pregnancy and preterm labour. Mediators Inflamm 2012, 967629, https://doi.org/10.1155/2012/967629 (2012).
Mor, G., Aldo, P. & Alvero, A. B. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol 17, 469–482, https://doi.org/10.1038/nri.2017.64 (2017).
Svensson, J. et al. Macrophages at the fetal-maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J Immunol 187, 3671–3682, https://doi.org/10.4049/jimmunol.1100130 (2011).
Mills, C. D., Kincaid, K., Alt, J. M., Heilman, M. J. & Hill, A. M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol 164, 6166–6173 (2000).
Brown, M. B., von Chamier, M., Allam, A. B. & Reyes, L. M1/M2 macrophage polarity in normal and complicated pregnancy. Front Immunol 5, 606, https://doi.org/10.3389/fimmu.2014.00606 (2014).
Mantovani, A., Biswas, S. K., Galdiero, M. R., Sica, A. & Locati, M. Macrophage plasticity and polarization in tissue repair and remodelling. The Journal of pathology 229, 176–185, https://doi.org/10.1002/path.4133 (2013).
Piccinni, M. P. et al. How pregnancy can affect autoimmune diseases progression? Clin Mol Allergy 14, 11, https://doi.org/10.1186/s12948-016-0048-x (2016).
Svensson-Arvelund, J. et al. The human fetal placenta promotes tolerance against the semiallogeneic fetus by inducing regulatory T cells and homeostatic M2 macrophages. J Immunol 194, 1534–1544, https://doi.org/10.4049/jimmunol.1401536 (2015).
Muzzio, D., Zenclussen, A. C. & Jensen, F. The role of B cells in pregnancy: the good and the bad. Am J Reprod Immunol 69, 408–412, https://doi.org/10.1111/aji.12079 (2013).
Dubicke, A. et al. Pro-inflammatory and anti-inflammatory cytokines in human preterm and term cervical ripening. J Reprod Immunol 84, 176–185, https://doi.org/10.1016/j.jri.2009.12.004 (2010).
Malmstrom, E. et al. The importance of fibroblasts in remodelling of the human uterine cervix during pregnancy and parturition. Mol Hum Reprod 13, 333–341, https://doi.org/10.1093/molehr/gal117 (2007).
Sennstrom, M. B. et al. Human cervical ripening, an inflammatory process mediated by cytokines. Mol Hum Reprod 6, 375–381 (2000).
Fransson, E. et al. Negative emotions and cytokines in maternal and cord serum at preterm birth. Am J Reprod Immunol 67, 506–514, https://doi.org/10.1111/j.1600-0897.2011.01081.x (2012).
Zhang, Y. H., He, M., Wang, Y. & Liao, A. H. Modulators of the Balance between M1 and M2 Macrophages during Pregnancy. Front Immunol 8, 120, https://doi.org/10.3389/fimmu.2017.00120 (2017).
Hamilton, S. et al. Macrophages infiltrate the human and rat decidua during term and preterm labor: evidence that decidual inflammation precedes labor. Biol Reprod 86, 39, https://doi.org/10.1095/biolreprod.111.095505 (2012).
Weetman, A. P. Immunity, thyroid function and pregnancy: molecular mechanisms. Nat Rev Endocrinol 6, 311–318, https://doi.org/10.1038/nrendo.2010.46 (2010).
de Man, Y. A., Dolhain, R. J. & Hazes, J. M. Disease activity or remission of rheumatoid arthritis before, during and following pregnancy. Current opinion in rheumatology 26, 329–333, https://doi.org/10.1097/bor.0000000000000045 (2014).
Pozzilli, C., Pugliatti, M. & Paradig, M. S. G. An overview of pregnancy-related issues in patients with multiple sclerosis. Eur J Neurol 22(Suppl 2), 34–39, https://doi.org/10.1111/ene.12797 (2015).
Airas, L. Hormonal and gender-related immune changes in multiple sclerosis. Acta Neurol Scand 132, 62–70, https://doi.org/10.1111/ane.12433 (2015).
Jara, L. J. et al. Risk factors of systemic lupus erythematosus flares during pregnancy. Immunol Res 60, 184–192, https://doi.org/10.1007/s12026-014-8577-1 (2014).
Muzzio, D., Zygmunt, M. & Jensen, F. The role of pregnancy-associated hormones in the development and function of regulatory B cells. Front Endocrinol (Lausanne) 5, 39, https://doi.org/10.3389/fendo.2014.00039 (2014).
Saito, S., Nakashima, A., Shima, T. & Ito, M. Th1/Th2/Th17 and regulatory T-cell paradigm in pregnancy. Am J Reprod Immunol 63, 601–610, https://doi.org/10.1111/j.1600-0897.2010.00852.x (2010).
Saito, S. et al. Quantitative analysis of peripheral blood Th0, Th1, Th2 and the Th1:Th2 cell ratio during normal human pregnancy and preeclampsia. Clin Exp Immunol 117, 550–555 (1999).
Simpson, W., Steiner, M., Coote, M. & Frey, B. N. Relationship between inflammatory biomarkers and depressive symptoms during late pregnancy and the early postpartum period: a longitudinal study. Rev Bras Psiquiatr 38, 190–196, https://doi.org/10.1590/1516-4446-2015-1899 (2016).
Gillespie, S. L., Porter, K. & Christian, L. M. Adaptation of the inflammatory immune response across pregnancy and postpartum in Black and White women. J Reprod Immunol 114, 27–31, https://doi.org/10.1016/j.jri.2016.02.001 (2016).
Christian, L. M. & Porter, K. Longitudinal changes in serum proinflammatory markers across pregnancy and postpartum: effects of maternal body mass index. Cytokine 70, 134–140, https://doi.org/10.1016/j.cyto.2014.06.018 (2014).
Sherer, M. L., Posillico, C. K. & Schwarz, J. M. An examination of changes in maternal neuroimmune function during pregnancy and the postpartum period. Brain Behav Immun 66, 201–209, https://doi.org/10.1016/j.bbi.2017.06.016 (2017).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. The Journal of clinical psychiatry 59 Suppl 20, 22–33, quiz 34–57 (1998).
Edvinsson, A. et al. Lower inflammatory markers in women with antenatal depression brings the M1/M2 balance into focus from a new direction. Psychoneuroendocrinology 80, 15–25, https://doi.org/10.1016/j.psyneuen.2017.02.027 (2017).
Rubertsson, C., Borjesson, K., Berglund, A., Josefsson, A. & Sydsjo, G. The Swedish validation of Edinburgh Postnatal Depression Scale (EPDS) during pregnancy. Nord J Psychiatry 65, 414–418, https://doi.org/10.3109/08039488.2011.590606 (2011).
Wickberg, B. & Hwang, C. P. The Edinburgh Postnatal Depression Scale: validation on a Swedish community sample. Acta psychiatrica Scandinavica 94, 181–184 (1996).
Assarsson, E. et al. Homogenous 96-plex PEA immunoassay exhibiting high sensitivity, specificity, and excellent scalability. PLoS One 9, e95192, https://doi.org/10.1371/journal.pone.0095192 (2014).
Lundberg, M., Eriksson, A., Tran, B., Assarsson, E. & Fredriksson, S. Homogeneous antibody-based proximity extension assays provide sensitive and specific detection of low-abundant proteins in human blood. Nucleic acids research 39, e102, https://doi.org/10.1093/nar/gkr424 (2011).
NHANES, N. H. a. N. E. S. 2011–2012 DataDocumentation, Codebook, and Frequencies., http://wwwn.cdc.gov/nchs/nhanes/2011-2012/PP_G.htm (2013).
Cavaillon, J. M. Pro- versus anti-inflammatory cytokines: myth or reality. Cellular and molecular biology (Noisy-le-Grand, France) 47, 695–702 (2001).
Abelius, M. S. et al. Th2-like chemokine levels are increased in allergic children and influenced by maternal immunity during pregnancy. Pediatric allergy and immunology: official publication of the European Society of Pediatric Allergy and Immunology 25, 387–393, https://doi.org/10.1111/pai.12235 (2014).
Abelius, M. S. et al. Pregnancy modulates the allergen-induced cytokine production differently in allergic and non-allergic women. Pediatric allergy and immunology: official publication of the European Society of Pediatric Allergy and Immunology 28, 818–824, https://doi.org/10.1111/pai.12802 (2017).
Nicola, N. A. & Babon, J. J. Leukemia inhibitory factor (LIF). Cytokine & growth factor reviews 26, 533–544, https://doi.org/10.1016/j.cytogfr.2015.07.001 (2015).
Metcalfe, S. M. LIF and multiple sclerosis: One protein with two healing properties. Mult Scler Relat Disord 20, 223–227, https://doi.org/10.1016/j.msard.2018.01.018 (2018).
Lass, A., Weiser, W., Munafo, A. & Loumaye, E. Leukemia inhibitory factor in human reproduction. Fertil Steril 76, 1091–1096 (2001).
Stewart, C. L. et al. Blastocyst implantation depends on maternal expression of leukaemia inhibitory factor. Nature 359, 76–79, https://doi.org/10.1038/359076a0 (1992).
Pelletier, J. P. & Martel-Pelletier, J. Oncostatin M: foe or friend? Arthritis Rheum 48, 3301–3303, https://doi.org/10.1002/art.11348 (2003).
Hermanns, H. M. & Oncostatin, M. and interleukin-31: Cytokines, receptors, signal transduction and physiology. Cytokine & growth factor reviews 26, 545–558, https://doi.org/10.1016/j.cytogfr.2015.07.006 (2015).
Stephens, J. M. & Elks, C. M. Oncostatin M: Potential Implications for Malignancy and Metabolism. Current pharmaceutical design. https://doi.org/10.2174/1381612823666170704122559 (2017).
Komori, T. et al. Oncostatin M is a potential agent for the treatment of obesity and related metabolic disorders: a study in mice. Diabetologia 58, 1868–1876, https://doi.org/10.1007/s00125-015-3613-9 (2015).
Layton, M. J. et al. A major binding protein for leukemia inhibitory factor in normal mouse serum: identification as a soluble form of the cellular receptor. Proceedings of the National Academy of Sciences of the United States of America 89, 8616–8620 (1992).
Pitard, V. et al. The presence in human serum of a circulating soluble leukemia inhibitory factor receptor (sgp190) and its evolution during pregnancy. European cytokine network 9, 599–605 (1998).
Miyazono, K. & Heldin, C. H. Latent forms of TGF-beta: molecular structure and mechanisms of activation. Ciba Found Symp 157, 81–89; discussion 89–92 (1991).
Barcellos-Hoff, M. H. Latency and activation in the control of TGF-beta. J Mammary Gland Biol Neoplasia 1, 353–363 (1996).
Zhang, Y. et al. Mammary-tumor-educated B cells acquire LAP/TGF-beta and PD-L1 expression and suppress anti-tumor immune responses. Int Immunol 28, 423–433, https://doi.org/10.1093/intimm/dxw007 (2016).
Minciullo, P. L. et al. Inflammaging and Anti-Inflammaging: The Role of Cytokines in Extreme Longevity. Arch Immunol Ther Exp (Warsz) 64, 111–126, https://doi.org/10.1007/s00005-015-0377-3 (2016).
Moore, T. & Dveksler, G. S. Pregnancy-specific glycoproteins: complex gene families regulating maternal-fetal interactions. Int J Dev Biol 58, 273–280, https://doi.org/10.1387/ijdb.130329gd (2014).
Mohan, T., Deng, L. & Wang, B. Z. CCL28 chemokine: An anchoring point bridging innate and adaptive immunity. Int Immunopharmacol 51, 165–170, https://doi.org/10.1016/j.intimp.2017.08.012 (2017).
Eksteen, B. et al. Epithelial inflammation is associated with CCL28 production and the recruitment of regulatory T cells expressing CCR10. J Immunol 177, 593–603 (2006).
Chen, Z. et al. Characterising the expression and function of CCL28 and its corresponding receptor, CCR10, in RA pathogenesis. Ann Rheum Dis 74, 1898–1906, https://doi.org/10.1136/annrheumdis-2013-204530 (2015).
Sun, C. et al. Chemokine CCL28 induces apoptosis of decidual stromal cells via binding CCR3/CCR10 in human spontaneous abortion. Mol Hum Reprod 19, 676–686, https://doi.org/10.1093/molehr/gat038 (2013).
Wilson, E. & Butcher, E. C. CCL28 controls immunoglobulin (Ig)A plasma cell accumulation in the lactating mammary gland and IgA antibody transfer to the neonate. The Journal of experimental medicine 200, 805–809, https://doi.org/10.1084/jem.20041069 (2004).
Li, J. Y. et al. FGF-21 Elevated IL-10 Production to Correct LPS-Induced Inflammation. Inflammation. https://doi.org/10.1007/s10753-018-0729-3 (2018).
Redondo-Angulo, I. et al. Fgf21 is required for cardiac remodeling in pregnancy. Cardiovasc Res 113, 1574–1584, https://doi.org/10.1093/cvr/cvx088 (2017).
Li, S. M. et al. Fibroblast growth factor 21 expressions in white blood cells and sera of patients with gestational diabetes mellitus during gestation and postpartum. Endocrine 48, 519–527, https://doi.org/10.1007/s12020-014-0309-8 (2015).
Hanada, R., Hanada, T., Sigl, V., Schramek, D. & Penninger, J. M. RANKL/RANK-beyond bones. J Mol Med (Berl) 89, 647–656, https://doi.org/10.1007/s00109-011-0749-z (2011).
Kong, Y. Y. et al. Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature 402, 304–309, https://doi.org/10.1038/46303 (1999).
Geng, W. et al. Sleep deprivation induces abnormal bone metabolism in temporomandibular joint. International journal of clinical and experimental medicine 8, 395–403 (2015).
Meng, Y. H. et al. RANKL-mediated harmonious dialogue between fetus and mother guarantees smooth gestation by inducing decidual M2 macrophage polarization. Cell Death Dis 8, e3105, https://doi.org/10.1038/cddis.2017.505 (2017).
Walsh, M. C. & Choi, Y. Biology of the TRANCE axis. Cytokine Growth Factor Rev 14, 251–263 (2003).
Burkly, L. C. TWEAK/Fn14 axis: the current paradigm of tissue injury-inducible function in the midst of complexities. Semin Immunol 26, 229–236, https://doi.org/10.1016/j.smim.2014.02.006 (2014).
Maecker, H. et al. TWEAK attenuates the transition from innate to adaptive immunity. Cell 123, 931–944, https://doi.org/10.1016/j.cell.2005.09.022 (2005).
Burkly, L. C., Michaelson, J. S., Hahm, K., Jakubowski, A. & Zheng, T. S. TWEAKing tissue remodeling by a multifunctional cytokine: role of TWEAK/Fn14 pathway in health and disease. Cytokine 40, 1–16, https://doi.org/10.1016/j.cyto.2007.09.007 (2007).
Qi, X. et al. Endogenous TWEAK is critical for regulating the function of mouse uterine natural killer cells in an immunological model of pregnancy loss. Immunology 148, 70–82, https://doi.org/10.1111/imm.12588 (2016).
Garcia-Zepeda, E. A. et al. Human eotaxin is a specific chemoattractant for eosinophil cells and provides a new mechanism to explain tissue eosinophilia. Nat Med 2, 449–456 (1996).
Chau, S. E. et al. Control of extravillous trophoblast function by the eotaxins CCL11, CCL24 and CCL26. Hum Reprod 28, 1497–1507, https://doi.org/10.1093/humrep/det060 (2013).
Sharma, S., Godbole, G. & Modi, D. Decidual Control of Trophoblast Invasion. Am J Reprod Immunol 75, 341–350, https://doi.org/10.1111/aji.12466 (2016).
Menzies-Gow, A. et al. Eotaxin (CCL11) and eotaxin-2 (CCL24) induce recruitment of eosinophils, basophils, neutrophils, and macrophages as well as features of early- and late-phase allergic reactions following cutaneous injection in human atopic and nonatopic volunteers. J Immunol 169, 2712–2718 (2002).
Zhang, Y. H., Tian, M., Tang, M. X., Liu, Z. Z. & Liao, A. H. Recent Insight into the Role of the PD-1/PD-L1 Pathway in Feto-Maternal Tolerance and Pregnancy. Am J Reprod Immunol 74, 201–208, https://doi.org/10.1111/aji.12365 (2015).
Gomez, E. et al. Expression and localization of ARTEMIN in the bovine uterus and embryos. Theriogenology 90, 153–162, https://doi.org/10.1016/j.theriogenology.2016.12.007 (2017).
Boldingh, M. I. et al. Increased risk for clinical onset of myasthenia gravis during the postpartum period. Neurology 87, 2139–2145, https://doi.org/10.1212/WNL.0000000000003339 (2016).
Molin, C. J., Westerberg, E. & Punga, A. R. Profile of upregulated inflammatory proteins in sera of Myasthenia Gravis patients. Sci Rep 7, 39716, https://doi.org/10.1038/srep39716 (2017).
Klawiter, E. C. et al. High risk of postpartum relapses in neuromyelitis optica spectrum disorder. Neurology 89, 2238–2244, https://doi.org/10.1212/WNL.0000000000004681 (2017).
Youssef, P. & Kennedy, D. Arthritis in pregnancy: the role and safety of biological agents. Obstet Med 2, 134–137, https://doi.org/10.1258/om.2009.090023 (2009).
Matthiesen, L. et al. Immunology of preeclampsia. Chemical immunology and allergy 89, 49–61, https://doi.org/10.1159/000087912 (2005).
Acknowledgements
This study was supported by grants from the Swedish research council (VR:521-2013-2339, 523-2014-2342), and the Marianne and Marcus Wallenberg Foundation (2011/Skalkidou).
Author information
Authors and Affiliations
Contributions
E.B. conducted laboratory visits. Å.E. prepared the samples. E.B. and Å.E. analyzed the data and wrote the main manuscript. I.S.P. and A.S. designed the study, and supervised the work. A.R.P., I.S.P., and A.S. made substantial contribution to the interpretation of results. Å.E. and E.B. revised the manuscript, with additional help from A.R.P., I.S.P., and A.S.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bränn, E., Edvinsson, Å., Rostedt Punga, A. et al. Inflammatory and anti-inflammatory markers in plasma: from late pregnancy to early postpartum. Sci Rep 9, 1863 (2019). https://doi.org/10.1038/s41598-018-38304-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-38304-w
This article is cited by
-
Bidirectional association between autoimmune disease and perinatal depression: a nationwide study with sibling comparison
Molecular Psychiatry (2024)
-
Autonomic function and inflammation in pregnant women participating in a randomized controlled study of Mindfulness Based Childbirth and Parenting
BMC Pregnancy and Childbirth (2023)
-
A cross-sectional study evidences regulations of leukocytes in the colostrum of mothers with obesity
BMC Medicine (2022)
-
Inflammatory biomarkers in pregnant women with COVID-19: a retrospective cohort study
Scientific Reports (2021)
-
Endogenous control of inflammation characterizes pregnant women with asymptomatic or paucisymptomatic SARS-CoV-2 infection
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.