Abstract
Many studies have examined the average effects of ocean acidification and warming on phenotypic traits of reef fishes, finding variable, but often negative effects on behavioural and physiological performance. Yet the presence and nature of a relationship between these traits is unknown. A negative relationship between phenotypic traits could limit individual performance and even the capacity of populations to adapt to climate change. Here, we examined the relationship between behavioural and physiological performance of a juvenile reef fish under elevated CO2 and temperature in a full factorial design. Behaviourally, the response to an alarm odour was negatively affected by elevated CO2, but not elevated temperature. Physiologically, aerobic scope was significantly diminished under elevated temperature, but not under elevated CO2. At the individual level, there was no relationship between behavioural and physiological traits in the control and single-stressor treatments. However, a statistically significant negative relationship was detected between the traits in the combined elevated CO2 and temperature treatment. Our results demonstrate that trade-offs in performance between behavioural and physiological traits may only be evident when multiple climate change stressors are considered, and suggest that this negative relationship could limit adaptive potential to climate change.
Similar content being viewed by others
Introduction
A major goal of climate change research is to determine whether species will withstand environmental change. A wealth of studies has assessed biological responses to projected future conditions in a variety of taxa, providing an indicator of large-scale trends in response to climate change conditions1,2,3. However, it can be advantageous to look beyond mean responses and examine the variation in performance between individuals within a species or population. Once disregarded and considered to be noise or error between measurements, individual variation is now recognized as a metric worth investigating in its own right4,5. In particular, a surge of interest has grown around variation in behavioural and physiological traits for their potential to identify patterns of trait covariation4,6, determine the mechanistic underpinnings of these covariations5,7,8, and predict their evolutionary implications9,10.
The relationship between behavioural and physiological performance could be especially important in the context of climate change. Examining this type of variation can highlight the individuals that are best suited to survival in future climate conditions, as well as reveal correlations between types of performance that can either help or hinder survival at the individual level11. Furthermore, correlations between traits could have implications for the potential of organisms to adapt to climate change11,12. In particular, if the behavioural and physiological traits of interest are heritable, then correlations between them could either accelerate or decelerate adaptive evolution10,13. If the traits are negatively correlated, selection on one would diminish the other, decreasing the rate of adaptation, and vice versa. Thus, identifying correlations among key traits is an important step in predicting species persistence in the face of climate change11,12,14. Importantly, environmental stressors have been shown to alter the relationship between behavioural and physiological traits, either amplifying or masking significant correlations15. Therefore, to improve our understanding of the effects of climate change on fish, it will be necessary to evaluate these relationships not only under current-day conditions, but also under different climate change-relevant scenarios.
Ocean acidification and warming are two of the primary environmental stressors in marine ecosystems16,17. As both are driven by increasing carbon dioxide emissions, they will likely increase in tandem, forcing marine organisms to contend with both stressors simultaneously18,19. The majority of climate change studies on marine organisms have focused on single stressors, although there has been an increasing recognition of the importance of a multi-stressor approach2,20. Multi-stressor experiments more accurately capture likely future scenarios, and can reveal unexpected interactions between stressors. For instance, ocean acidification and warming have been shown to interact both synergistically and antagonistically on the responses of marine organisms20,21. Thus, multi-stressor experiments are crucial, as single-stressor studies could lead to inaccurate predictions about organismal responses to future climate conditions20,22.
Reef fishes can be sensitive to the effects of both elevated CO2 and temperature. Elevated CO2 levels projected for the end of the century have been documented to impact a range of behavioural traits in reef fishes such as lateralization, activity, homing ability, learning, anxiety, and olfactory and auditory discrimination (reviewed by Clements & Hunt23 and Nagelkerken & Munday24). These behavioural changes can make fish more vulnerable to predation and have been linked to higher mortality rates25,26,27. Elevated CO2 has also been documented to affect physiological traits, such as aerobic scope28,29, escape performance27, and reproduction30,31 in some reef fishes, but the physiological effects of CO2 are generally more variable across species than behavioural effects32,33. By contrast, temperature is well known to impact fish physiological performance through its effect on rates of biochemical reactions34. Elevated temperatures predicted for the end of the century have been shown to alter aerobic scope35,36,37, body size38, reproductive output39,40, and swimming ability41 in reef fishes. Behaviourally, both boldness and activity levels have been shown to change in response to temperature increases42,43,44.
In this study, we investigated the effects of elevated CO2 and temperature on behavioural and physiological performance of juvenile spiny chromis, Acanthochromis polyacanthus. We used a full factorial design which consisted of a current-day ambient CO2 level (~500 µatm) and average summer temperature for the study location (29° C), crossed with elevated CO2 (~1000 µatm) and temperature (32° C) based on projections for the end of the century under RCP 8.517,45. The behavioural trait we measured was the percent reduction in feeding strikes after exposure to damage-released olfactory cues for a conspecific (i.e., alarm odours). Conspecifics display an innate aversion to these reliable indicators of predation risk, typically reducing activity and seeking shelter46. However, previous studies have found that when fishes are reared under elevated CO2 levels, they no longer reduce their activity or stop feeding in the presence of these odours47,48. The inability to use alarm odours under elevated CO2 conditions makes prey more vulnerable to predators and can lead to higher mortality at key life-history stages47,49. The physiological trait we determined was aerobic scope. Aerobic scope refers to the total capacity for aerobic activity available to an organism, after accounting for basic maintenance34,50, and is calculated as the difference between the maximal and resting oxygen uptake rates of an organism. Aerobic activities such as growth, reproduction, and swimming are essential life-history processes; therefore, a reduction in aerobic scope could reduce individual performance and fitness1,51,52. Both elevated CO2 and elevated temperature have been predicted to decrease aerobic scope in fishes1,53. However, when tested, elevated temperature often decreases aerobic scope in coral reef fishes35,36, while elevated CO2 has had more variable effects32,33.
By tracking the responses of individual fish when tested for their behavioural and physiological performance, we were able to determine the relationship between these traits at the individual level. We then compared this relationship between all four of the treatment groups to determine how the relationship between these traits might change under different environmental stressors, and ultimately, how adaptation to climate change conditions might be either facilitated or hindered by this relationship.
Results
Behaviour
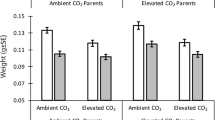
There was a highly significant effect of CO2 treatment on percent reduction in feeding strikes following the addition of the alarm odour (t160 = −3.69, P < 0.001, Fig. 1). Fish from the two current-day control CO2 treatments exhibited a 43.3% greater reduction in feeding strikes than fish from the two elevated CO2 treatments. Temperature treatment did not affect the reduction in feeding strikes (t160 = −0.03, P = 0.98). There was no interaction between CO2 and temperature on percent reduction in feeding strikes (t160 = −0.23, P = 0.82).
Physiology
There was a significant interaction between CO2 and temperature treatments on aerobic scope (t134 = 2.15, P = 0.03; Fig. 2A). Tukey’s post-hoc tests revealed that there was a significant difference in aerobic scope between the control and elevated temperature treatments held at control CO2 (z = −4.03, P = 0.001), but this was not the case when CO2 was elevated (pairwise interactions, P > 0.05). Despite the significant interaction, there was a strong effect of temperature evident in the data, which was supported by a significant main effect of temperature (t134 = −3.73, P < 0.001). Fish from the two elevated temperature treatments exhibited a 20% lower aerobic scope than fish from the two control temperature treatments. There was no main effect of CO2 treatment on aerobic scope (t134 = −1.43, P = 0.15), while mass had a significant effect on aerobic scope (t134 = −5.19, P < 0.001).
The effect of elevated CO2 and temperature treatments on resting and maximal oxygen uptake rates and aerobic scope in juvenile spiny chromis damselfish. Boxplots show median and inter-quartile range for (A) absolute aerobic scope (ṀO2Max - ṀO2Rest); and (B) resting (ṀO2Rest; blue boxes) and maximal oxygen uptake rates (ṀO2Max; orange boxes). Letters represent Tukey’s HSD groups. N = 32–38 per treatment.
There was a significant interaction between CO2 treatment and temperature treatment on ṀO2Max (t140 = 3.47, P < 0.001; Fig. 2B). Tukey’s post-hoc tests revealed that juveniles reared in the current-day control and in the combined elevated CO2 and temperature treatments had a significantly higher ṀO2Max than juveniles from the elevated temperature treatment (control: z = 3.58, P = 0.002; elevated CO2 and temperature: z = 3.61, P = 0.002), while juveniles from the elevated CO2 treatment had a ṀO2Max which was not significantly different from the other three treatments (pairwise interactions, P > 0.05). As was the case with aerobic scope, there a significant main effect of temperature treatment on ṀO2Max (t140 = −3.58, P < 0.001). Fish from the two elevated temperature treatments had an 18.2% lower ṀO2Max than fish from the two control temperature treatments. There was no main effect of CO2 treatment on ṀO2Max (t140 = −1.29, P = 0.20) and mass had a highly significant effect on ṀO2Max (t140 = −3.32, P = 0.001).
There was a significant interaction between CO2 treatment and temperature treatment on ṀO2Rest (t136 = 3.45, P < 0.001; Fig. 2B). Post-hoc Tukey’s tests revealed that juveniles reared in the current-day control, elevated temperature, and elevated CO2 treatments all had similar values for ṀO2Rest (pairwise interactions, P > 0.05), whereas juveniles reared in the elevated CO2 and temperature treatment had a significantly higher ṀO2Rest (pairwise interactions, P < 0.05). Neither CO2 treatment nor mass had a significant effect on ṀO2Rest (t136 = −0.40, P = 0.69 and t136 = 0.05, P = 0.96, respectively).
Relationship between Behavioural and Physiological Performance
There was no significant relationship between percent reduction in feeding strikes and aerobic scope in the juveniles reared in the current-day control (t27 = −0.29, P = 0.77, Fig. 3), elevated CO2 (t26 = 1.72, P = 0.10), or elevated temperature (t27 = 0.22, P = 0.83) treatments. However, there was a significant negative relationship between percent reduction in feeding strikes and aerobic scope in juveniles reared at elevated CO2 and temperature (t27 = −3.09, P = 0.005). This relationship was consistent across all family groups (ANOVA of LM, Family x Reduction in Feeding Strikes interaction, P > 0.05, Fig. 4).
The relationship between percent change in feeding strikes and aerobic scope. Panels represent different treatments, and colours represent different family groups. Trend lines are shown as derived from linear mixed effect models. The relationship is statistically significant for the elevated CO2 and temperature treatment.
Discussion
Our study found a negative relationship between changes in feeding strikes in response to alarm odour and aerobic scope, but only when fish were reared in elevated CO2 and temperature conditions. Our results indicate that when exposed to two climate change stressors, there are no winner and loser individuals– rather, each individual seems constrained along a maximal performance ridge14, such that an individual could maintain a relatively high aerobic scope, or an ecologically appropriate response to alarm cue, but not both. Furthermore, the relationship was consistent across family groups, suggesting that certain families do not hold a distinct advantage over others in dealing with this limitation. This negative correlation could have implications for the adaptive potential of this species. Selection for improved performance of either trait could decrease performance in the other, slowing adaptation. Given the rapid pace at which the global environment is changing, any factor that slows or limits selection for improved performance could have serious implications for individual performance and ultimately, population success.
We detected a significant correlation between traits only in the multi-stressor treatment. This result aligns well with theory that environmental stress can act as a revealing or amplifying factor on correlations between behaviour and physiology15. The mechanism for this pattern is not known, but may be explained by differing sensitivities to the stressors between individuals, which can increase the intraspecific phenotypic variation in the traits54,55. Additionally, the increased demands on performance that are imposed by stressors can emphasize the importance of certain traits, making links between behaviour and physiology more evident under stressful conditions15. Indeed, since we observed behavioural performance to be most affected by elevated CO2 and physiological performance to be most affected by elevated temperature, it is intuitive that the presence of both stressors might be necessary to observe this relationship. Unfortunately, ocean waters will likely become both warmer and more acidic in the future, meaning that future conditions may elicit this negative correlation between behavioural and physiological performance.
This study investigated phenotypic, not genotypic correlations between traits. While phenotypic correlations are often indicative of genetic correlations56, we did not specifically test the genetic basis of the phenotypic traits considered here. Correlations between phenotypic traits might enhance or hinder selection, but ultimately adaptation can only be influenced by genetic covariation between traits. This means that the observed phenotypic variation must be heritable for adaptation to occur. Recent studies demonstrate that aerobic scope has high heritability under elevated temperatures57, and response to alarm odour is heritable, at least under acute exposure to elevated CO258,59. These studies suggest that these traits have a significant amount of additive genetic variation, indicating that the correlations we have detected could indeed affect adaptation.
Our behavioural performance trials demonstrated that juveniles reared under elevated CO2 conditions did not reduce their feeding strikes after the addition of an alarm odour to the same extent as juveniles reared under current-day control conditions. This is consistent with previous studies that have shown elevated CO2 to negatively impact a range of behavioural and sensory traits in reef fishes23,24. A likely mechanism underpinning the behavioural changes we observed is a disruption to neurotransmitter receptor function. Fish have robust acid-base regulatory systems that help them to maintain a stable internal pH60. However, the changed concentration of acid-base relevant ions that is required to maintain internal pH under elevated CO2 conditions can interfere with the function of GABAA neurotransmitter receptors, resulting in altered behaviour and impaired olfactory responses61,62. It is important to note that our behavioural assay does not differentiate between fish with impaired olfactory preferences versus those with increased activity and boldness; rather, the assay encompasses both responses in one ecologically-relevant test. Because ocean acidification has been shown to alter a broad range of cognitive functions in marine fishes, it is likely that it affects central neural processing rather than individual behaviours or sensory systems60,61. Thus, changes to both boldness and olfactory preference likely stem from the same disruption to GABAA functioning, allowing them to be assessed simultaneously in our assay. Through this method, we were able to portray a real-life scenario involving both foraging and anti-predator response in which a fish must respond appropriately to an alarm odour.
In our physiological performance trials, we documented that the aerobic scope of juvenile A. polyacanthus decreased under elevated temperature. This negative effect of elevated temperature on aerobic performance has been previously shown in a range of coral reef fishes35,36,37, although the mechanism by which temperature affects aerobic performance in ectotherms is still not fully understood63. There was also a non-significant trend toward decreased aerobic scope in fish maintained under elevated CO2 conditions. Elevated CO2 has been shown to have mixed effects on aerobic scope in a range of marine fishes, with elevated temperature typically having a greater effect than elevated CO232,33, which aligns well with our results. However, our results contrast with a 38% increase in aerobic scope that was observed in adult A. polyacanthus exposed to similar CO2 levels29. We hypothesize that the differing results primarily stem from the different life-stages of the tested fish (i.e., juvenile vs. sexually-mature adults). It has been suggested that early life stages of fish are more sensitive to changes in pH due to their high surface area-to-volume ratio64,65, which could explain this discrepancy. Our results further underscore the importance of considering life stage when determining species responses to elevated CO2.
This study indicates that there is a negative correlation between behavioural and physiological performance in juvenile damselfish exposed to elevated CO2 and temperature. This relationship could reduce individual performance maxima, as well as limit the potential for adaptive evolution in the population. However, it is important to note that the extent to which this relationship is detrimental will depend upon other factors, such as food availability and predator abundance, and the relative benefits of a higher aerobic scope versus stronger anti-predator response in these environments. Juvenile coral reef fishes are generally considered to live in a high-risk environment, as up to 55% of settlement-stage juveniles are estimated to be consumed within days of reaching a reef66, suggesting that effective anti-predator responses would be under strong selection. Similarly, a high aerobic scope is thought to be beneficial when food is abundant, but less helpful when food is scarce67, and thus its importance will depend upon food availability. Still, in reef fishes there is evidence for heritability of phenotypic variation in physiological and behavioural traits under elevated CO2 and temperature57,58,59, so it is not unreasonable to expect that adaptation can and will act on these traits under future climate conditions.
While we saw a clear correlation in these traits under elevated CO2 and temperature conditions, the proximal cause for the relationship remains unknown. Linkages among metabolic traits and behavioural traits have been well explored7,8,68, but proximal causes are generally more difficult to identify. For instance, the observed negative correlation might be caused by genetic linkages between traits, but could also be explained by a shared hormonal feature69, or a trade-off in energetics between internal pH regulation and thermal tolerance70. This work thus represents an important first step in identifying this correlation, and opens an avenue for future research to identify the mechanistic basis of this relationship.
Materials and Methods
Study species, brood-stock collection and maintenance
The spiny chromis, Acanthochromis polyacanthus, is a tropical damselfish from the western Pacific Ocean. The species has direct development (i.e. eggs hatch into juveniles), and both parents care for the eggs and offspring for up to 45 days post-hatch71. Without a pelagic larval phase, the species is easily bred in captivity. Due to its wide geographic distribution, abundance, and amenability to laboratory experimentation, A. polyacanthus has become a model species for studying the effects of climate change on coral reef fishes (e.g.39,48).
Adult A. polyacanthus were collected using hand nets from the Bramble Reef area (site 1: 18°22′S, 146°40′E; site 2: 18°25′S, 146°40′E) on the Great Barrier Reef in July 2015. Fish were transported to James Cook University (Townsville, Australia), where they were sorted into breeding pairs and housed in 60 L aquaria. Pairs were provided with half a terracotta pot for shelter and as a suitable artificial breeding site. Aquaria were checked daily for the presence of newly laid clutches. Pairs were fed ad libitum on commercial fish feed pellets (INVE Aquaculture Nutrition NRD 12/20) once per day outside the breeding season (July–October) and twice per day during the breeding season (November–May). Starting in October, water temperatures were increased at a rate of 0.5° C per week until the summer breeding temperature of 29° C was reached during the first week of November 2015.
Offspring were fed freshly hatched Artemia spp. nauplii for the first two days post-hatch (dph), then a combination of Artemia spp. nauplii and weaning fish feed (INVE Aquaculture Nutrition Wean-S 200–400 μm) daily for the following three days. They were fed the weaning fish feed from 6–21 dph and then switched to a small pellet fish feed (INVE Aquaculture Nutrition NRD 5/8) at 22 dph.
Carbonate Chemistry
Water for this experiment was supplied via two 8000 L recirculating aquarium systems. The ambient pCO2 level (~500 μatm) in one system was used as the current-day control. The other system was dosed with CO2 to match the end-of-century projection for surface ocean pCO2 under RCP 8.5 (~1000 μatm). The pCO2 level was controlled by an Aqua Medic AT Control System (Aqua Medic, Germany), which dosed CO2 into a 3000 L sump connected to the system whenever the pH in the system rose above the set point. An identical 3000 L sump on the current-day control was not dosed with additional CO2. Temperature was maintained at a current-day control of 29° C by circulating seawater through a SolarWise heater/chiller (Brisbane, Queensland, Australia) on each system. The equilibrated seawater from each system was then either delivered directly into the aquaria, or passed over Toyesi 2.5-kW inline heaters (Prospect, New South Wales, Australia) to raise the temperature to the elevated treatment of 32° C. Water was delivered into fish aquaria at a rate of 1.5 L min−1 in a temperature-controlled room.
The pHNBS and temperature for each system were recorded daily using a pH electrode (SevenGo Pro, Mettler Toledo, Switzerland) and temperature probe (Cormark C26, Norfolk, UK). The pHT was measured weekly with a spectrophotometer following standard operating procedures72 using the indicator dye meta/m-cresol purple (m-cresol purple sodium salt 99%, non-purified, Acros Organic).
Total alkalinity was also estimated weekly by Gran Titration (Metrohm 888 Titrando Titrator Metrohm AG, Switzerland) and using certified reference material from Dr. A.G. Dickson (Scripps Institution of Oceanography). Salinity was measured weekly using a conductivity sensor (HQ15d; Hach, Loveland, CO, USA). All water quality parameters were measured in randomly selected aquaria. The pCO2 was calculated as a function of pHT, temperature, salinity, and total alkalinity in CO2SYS using the constants K1 from Mehrbach et al.73. refit by Dickson & Millero74, and KHSO4 from Dickson75 (Table 1).
Experimental Design
One clutch of offspring from each of four different parental pairs was used for this experiment. At 1 dph, offspring in each clutch were divided into each of the four CO2 X temperature treatment groups: control, elevated CO2, elevated temperature, and elevated CO2 and temperature (Table 1), representing a 2 × 2 factorial design. Behavioural trials were performed at 60–66 dph, and physiological trials were performed on the same individuals at 62–68 dph, allowing at least one day rest between trials. To track individual fish between trials, immediately following the behavioural trial, individuals were placed into labelled PVC pipes (8 cm diameter, 5 cm length) that were covered at both ends by a thin plastic mesh to allow for flow-through of water, and then placed into treatment tanks. All trials were performed during daylight hours only (09:00–18:00) in the fish’s respective treatment water. Research was carried out under approval of the James Cook University animal ethics committee (permit: A2197) and according to the University’s animal ethics guidelines.
Behavioural Assay
The percent change in feeding strikes has been widely used to test the behavioural responses of fishes to conspecific alarm cues44,76,77,78, and this method has been used in previous ocean acidification experiments47,49,79. Using percent change in feeding strikes rather than the absolute feeding rate can account for differences in activity or feeding rate between individuals. The change in feeding strikes in response to alarm odours was tested using methods similar to those described by Ferrari et al.47. Trials were conducted in 13 L (36 × 21 × 20 cm) flow-through aquaria. Each tank contained a small shelter made of half a PVC pipe (8 cm diameter) at one end of the tank, and an airstone at the opposite end. Attached to the airstone was an injection tube for adding food and alarm odours to the tank. The end of the tube was located just above the airstone to ensure rapid dispersal of food and alarm odours throughout the tank. The tank was surrounded on three sides with white plastic to visually isolate the fish, and a black plastic curtain with a small flap separated the tank from the observer and other external stimuli.
Single juvenile A. polyacanthus were placed into tanks filled with their treatment water to habituate overnight (15 h). Each tank received a continuous flow of the relevant treatment water at 0.6 L min−1. A maximum of fourteen fish were tested per day. Ten minutes prior to a trial, the flow-through system to the tank was turned off to prevent the alarm odour from washing out of the tank. At this point, the flap in the observation curtain was opened and a camera placed into the opening, to habituate the fish to the camera. The fish was unable to see the observer, but the fish could be viewed on the camera’s screen. Immediately prior to a trial, 20 mL of water were drawn from the injection tube and discarded to remove any stagnant water that might have collected in the tube. A further 60 mL were drawn from the tube for flushing alarm odours into the tank.
Alarm odours were freshly prepared during the first 10 minutes of each trial, as these cues have been shown to lose potency after 20 minutes when kept at room temperature80. One juvenile A. polyacanthus donor was used for each test fish. The donor fish was euthanized with a quick blow to the head. The alarm odour was then prepared by making eight superficial vertical cuts along each side of the body with a scalpel blade. The cuts were rinsed well with 10 mL of seawater, and the cue water was passed through a filter to remove any solid material or scales47.
To start a trial, the camera (Canon Powershot G15, Canon Powershot G16, or Canon Powershot GX9) was turned on, and the fish was recorded for five minutes to ensure it did not exhibit any abnormal behaviours. Next, 2.5 mL of Artemia solution (containing ~250 individuals per mL) was slowly flushed into the tank with 20 mL of seawater to allow the fish to establish a stable feeding rate. After five minutes, another 2.5 mL of Artemia solution was flushed into the tank with 20 mL of seawater. Finally, after another five minutes, 2.5 mL of Artemia solution, followed by 10 mL of alarm odour, followed by 20 mL of seawater were flushed into the tank. The trial ended five minutes after the addition of the alarm odour, with each trial totalling 20 minutes. The entire trial was filmed for analysis. Following trials, fish were returned to their rearing tanks. Between 8 and 14 individuals from each of four family groups were tested per treatment, for a grand total of 38–47 fish per treatment.
Videos were analysed by a single researcher (T.L.) to determine the feeding rate of the fish before and after the addition of the alarm odour. Video file names were scrambled so that the viewer was unaware of the treatment. Feeding strikes were counted for four minutes in each time period, allowing 30 seconds after the addition of food or alarm odour to ensure a steady feeding rate, and that the alarm odour had fully permeated the tank. The percent change in feeding strikes after the addition of the alarm odour was then calculated.
Physiological Assay
To estimate aerobic scope, the maximum (ṀO2Max) and resting (ṀO2Rest) oxygen uptake rates were measured using intermittent flow respirometry, based on standard respirometry methods81,82. Fish were starved for 24 hours prior to testing to ensure a post-absorptive state83. As A. polyacanthus is a coral reef fish with a relatively sedentary lifestyle, ṀO2Max was measured following a chase to exhaustion (3 min.) and a brief (1 min.) air exposure, as this method has been shown to reliably capture ṀO2Max82. Following the chase and air exposure, fish were then immediately placed into darkened glass respirometry chambers (38 mL total volume including tubing) that were submerged in aquaria containing the fish’s treatment water. Submersible pumps fitted to each chamber supplied a continuous flow (20 mL min−1) of water to the chambers from the surrounding water bath. A digital relay timer (SuperPro Hydroponics Recycling Timer, Xiamen, China) was used to stop water flow for five minutes and then resume flushing for 10 minutes, continuously, for the duration of the trial. Water flow was stopped for five minutes to ensure that O2 did not fall below 80% saturation. Fish remained in chambers for four hours to recover to ṀO2Rest. While adult fish typically remain in chambers for up to 24 hours84, smaller fish recover much more quickly from exhaustive exercise and are commonly measured for only 2–3 hours to minimize stress and the risk of starvation85,86,87,88,89. The temperature-compensated oxygen concentration (mg L−1) of the water in each chamber was continuously recorded every 2 seconds (0.5 Hz) using oxygen-sensitive REDFLASH dye on contactless spots (2 mm) adhered to a glass tube in line with the chamber, and linked to a Firesting Optical Oxygen Meter (Pyro Science e. K., Aachen, Germany) with 2 m fibre-optic cables.
Oxygen uptake rates were calculated using linear least squares regression using LabChart version 7.2.5 (ADInstruments, Colorado Springs, CO, USA). Background microbial respiration was subtracted from total chamber respiration to determine the oxygen uptake rate of the fish, as per Rummer et al.81. The value of ṀO2Max was determined to be the maximum slope (30 second intervals) immediately following the exhaustive chase. The value of ṀO2Rest was calculated as the average of the lowest 10% of slopes during the trial, excluding outliers above or below 2 SD. Aerobic scope was calculated as the difference between ṀO2Max and ṀO2Rest. At the end of each trial, fish were euthanized using an overdose of clove oil. Any excess water was removed by blotting with a paper towel, and the fish’s mass (0.5783 ± 0.1433 g; mean ± SD) and standard length (26.5 ± 0.3 mm; mean ± SD) were recorded. Between trials, the water bath, chambers, and pumps were cleaned with a 10% bleach solution and freshwater to minimize bacterial growth. Between 6 and 14 individuals from each of four family groups were tested per treatment, for a grand total of 37–38 fish per treatment.
Statistical Analyses
Linear mixed-effects models (LME, “nlme” package in R) were used to determine the effect of CO2 and temperature on behavioural and physiological traits. For percent reduction in feeding strikes, the CO2 and temperature treatments were fixed effects, with fish mass and time of day as covariates, allowing for interactions between CO2 treatment, temperature treatment, mass, and time of day. Family was included as a random factor to account for the possibility that sibling responses were more similar to each other than non-sibling responses. Mass and time of day were mean-centred to help with the interpretation of model intercepts. For physiological traits (aerobic scope, ṀO2Rest, and ṀO2Max), CO2 and temperature treatments and mean-centred mass were fixed effects, and family was a random effect. Assumptions of normality and homogeneity of residuals were visually assessed with Q-Q plots and frequency distributions. When the variance of the model residuals increased as the fitted values increased, a power variance function was used to reduce heteroscedasticity. Parameters were estimated using restricted maximum-likelihood. Covariates and interactions between the fixed factor and covariates were dropped when not significant for model simplification and fit. P-values were calculated from the “nlme” package in R, and results were considered statistically significant at P < 0.05. Post hoc multiple comparisons were done using the R function “glht” in the package “multcomp” using Tukey’s HSD contrasts for unequal sample sizes.
The relationship between behavioural and physiological performance was modelled using a linear mixed effects model for each treatment. Here we were interested in describing the relationship between traits, and not necessarily assigning causation. We used linear mixed effect models in order to incorporate additional random effects. For each linear mixed effect model, aerobic scope was the dependent variable and percent reduction in feeding strikes was the independent variable. Mean-centred mass was included as a covariate, and family was included as a random factor. When there was a significant relationship observed, linear regressions were used to determine if there were differences between families, with aerobic scope as the dependent variable and percent reduction in feeding strikes, mass, and family as covariates. All analyses were conducted using R version 3.1.390.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on request or via the Tropical Research Data Hub (doi: 10.25903/5ba8882d93620).
References
Pörtner, H. O. & Farrell, A. Physiology and climate change. Science 322, 690–692 (2008).
Kroeker, K. J. et al. Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 19, 1884–1896 (2013).
Wittmann, A. C. & Pörtner, H. O. Sensitivities of extant animal taxa to ocean acidification. Nat. Clim. Chang. 3, 995–1001 (2013).
Careau, V., Thomas, D., Humphries, M. M. & Réale, D. Energy metabolism and animal personality. Oikos 117, 641–653 (2008).
Williams, T. D. Individual variation in endocrine systems: moving beyond the ‘tyranny of the Golden Mean’. Philos. Trans. R. Soc. B Biol. Sci. 363, 1687–1698 (2008).
Biro, P. A. & Stamps, J. A. Are animal personality traits linked to life-history productivity? Trends Ecol. Evol. 23, 361–368 (2008).
Biro, P. A. & Stamps, J. A. Do consistent individual differences in metabolic rate promote consistent individual differences in behavior? Trends Ecol. Evol. 25, 653–659 (2010).
Metcalfe, N. B., Van Leeuwen, T. E. & Killen, S. S. Does individual variation in metabolic phenotype predict fish behaviour and performance? J. Fish Biol. 88, 298–321 (2016).
Careau, V. & Garland, T. Performance, personality, and energetics: correlation, causation, and mechanism. Physiol. Biochem. Zool. 85, 543–571 (2012).
Lande, R. & Arnold, S. J. The measurement of selection on correlated characters. Evolution 37, 1210–1226 (1983).
Munday, P. L., Warner, R. R., Monro, K., Pandolfi, J. M. & Marshall, D. J. Predicting evolutionary responses to climate change in the sea. Ecol. Lett. 16, 1488–1500 (2013).
McBryan, T. L., Anttila, K., Healy, T. M. & Schulte, P. M. Responses to temperature and hypoxia as interacting stressors in fish: implications for adaptation to environmental change. Integr. Comp. Biol. 53, 648–659 (2013).
Lande, R. Quantitative genetic analysis of multivariate evolution, applied to brain:body size allometry. Evolution 33, 402–416 (1979).
Sunday, J. M. et al. Evolution in an acidifying ocean. Trends Ecol. Evol. 29, 117–125 (2014).
Killen, S. S., Marras, S., Metcalfe, N. B., McKenzie, D. J. & Domenici, P. Environmental stressors alter relationships between physiology and behaviour. Trends Ecol. Evol. 28, 651–658 (2013).
Doney, S. C., Fabry, V. J., Feely, R. A. & Kleypas, J. A. Ocean acidification: the other CO2 problem. Ann. Rev. Mar. Sci. 1, 69–92 (2009).
Collins, M. et al. In Climate Change2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds. Stocker, T.F. et al.) 1029–1136 (Cambridge University Press, 2013).
Doney, S. C. et al. Climate change impacts on marine ecosystems. Ann. Rev. Mar. Sci. 4, 11–37 (2012).
Hoegh-Guldberg, O. & Bruno, J. F. The impact of climate change on the world’s marine ecosystems. Science 328, 1523–1528 (2010).
Riebesell, U. & Gattuso, J. P. Lessons learned from ocean acidification research. Nat. Clim. Chang 5, 12–14 (2015).
Kroeker, K. J., Kordas, R. L., Crim, R. N. & Singh, G. G. Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol. Lett. 13, 1419–1434 (2010).
Gaylord, B. et al. Ocean acidification through the lens of ecological theory. Ecology 96, 3–15 (2015).
Clements, J. C. & Hunt, H. L. Marine animal behaviour in a high CO2 ocean. Mar. Ecol. Prog. Ser. 536, 259–279 (2015).
Nagelkerken, I. & Munday, P. L. Animal behaviour shapes the ecological effects of ocean acidification and warming: moving from individual to community-level responses. Glob. Chang. Biol. 22, 974–989 (2016).
Munday, P. L. et al. Replenishment of fish populations is threatened by ocean acidification. Proc. Natl. Acad. Sci. 107, 12930–12934 (2010).
Ferrari, M. C. O. et al. Putting prey and predator into the CO2 equation – qualitative and quantitative effects of ocean acidification on predator-prey interactions. Ecol. Lett. 14, 1143–1148 (2011).
Allan, B. J. M., Domenici, P., McCormick, M. I., Watson, S.-A. & Munday, P. L. Elevated CO2 affects predator-prey interactions through altered performance. PLoS One 8, e58520 (2013).
Munday, P. L., Crawley, N. E. & Nilsson, G. E. Interacting effects of elevated temperature and ocean acidification on the aerobic performance of coral reef fishes. Mar. Ecol. Prog. Ser. 388, 235–242 (2009).
Rummer, J. L. et al. Elevated CO2 enhances aerobic scope of a coral reef fish. Conserv. Physiol. 1, 1–7 (2013).
Miller, G. M., Watson, S.-A., McCormick, M. I. & Munday, P. L. Increased CO2 stimulates reproduction in a coral reef fish. Glob. Chang. Biol. 19, 3037–3045 (2013).
Welch, M. J. & Munday, P. L. Contrasting effects of ocean acidification on reproduction in reef fishes. Coral Reefs 35, 485–493 (2016).
Hannan, K. D. & Rummer, J. L. Aquatic acidification: a mechanism underpinning maintained oxygen transport and performance in fish experiencing elevated carbon dioxide conditions. J. Exp. Biol. 221 (2018).
Lefevre, S. Are global warming and ocean acidification conspiring against marine ectotherms? A meta-analysis of the respiratory effects of elevated temperature, high CO2 and their interaction. Conserv. Physiol. 4, cow009 (2016).
Fry, F. E. J. The effect of environmental factors on the physiology of fish. Fish Physiol. 6, 1–98 (1971).
Nilsson, G. E., Crawley, N., Lunde, I. G. & Munday, P. L. Elevated temperature reduces the respiratory scope of coral reef fishes. Glob. Chang. Biol. 15, 1405–1412 (2009).
Rummer, J. L. et al. Life on the edge: thermal optima for aerobic scope of equatorial reef fishes are close to current day temperatures. Glob. Chang. Biol. 20, 1055–1066 (2014).
Habary, A., Johansen, J. L., Nay, T. J., Steffensen, J. F. & Rummer, J. L. Adapt, move or die – how will tropical coral reef fishes cope with ocean warming? Glob. Chang. Biol. 23, 566–577 (2017).
Munday, P. L., Kingsford, M. J., O’Callaghan, M. & Donelson, J. M. Elevated temperature restricts growth potential of the coral reef fish Acanthochromis polyacanthus. Coral Reefs 27, 927–931 (2008).
Donelson, J. M., Munday, P. L., McCormick, M. I., Pankhurst, N. W. & Pankhurst, P. M. Effects of elevated water temperature and food availability on the reproductive performance of a coral reef fish. Mar. Ecol. Prog. Ser. 401, 233–243 (2010).
Pankhurst, N. W. & Munday, P. L. Effects of climate change on fish reproduction and early life history stages. Mar. Freshw. Res. 62, 1015–1026 (2011).
Johansen, J. L. & Jones, G. P. Increasing ocean temperature reduces the metabolic performance and swimming ability of coral reef damselfishes. Glob. Chang. Biol. 17, 2971–2979 (2011).
Biro, P. A., Beckmann, C. & Stamps, J. A. Small within-day increases in temperature affects boldness and alters personality in coral reef fish. Proc. R. Soc. B Biol. Sci. 277, 71–77 (2010).
Johansen, J. L., Messmer, V., Coker, D. J., Hoey, A. S. & Pratchett, M. S. Increasing ocean temperatures reduce activity patterns of a large commercially important coral reef fish. Glob. Chang. Biol. 20, 1067–1074 (2014).
Lienart, G. D. H., Mitchell, M. D., Ferrari, M. C. O. & McCormick, M. I. Temperature and food availability affect risk assessment in an ectotherm. Anim. Behav. 89, 199–204 (2014).
Meinshausen, M. et al. The RCP greenhouse gas concentrations and their extensions from 1765 to 2300. Clim. Change 109, 213–241 (2011).
Ferrari, M. C. O., Wisenden, B. D. & Chivers, D. P. Chemical ecology of predator–prey interactions in aquatic ecosystems: a review and prospectus. Can. J. Zool. 88, 698–724 (2010).
Ferrari, M. C. O. et al. Intrageneric variation in antipredator responses of coral reef fishes affected by ocean acidification: implications for climate change projections on marine communities. Glob. Chang. Biol. 17, 2980–2986 (2011).
Welch, M. J., Watson, S.-A., Welsh, J. Q., McCormick, M. I. & Munday, P. L. Effects of elevated CO2 on fish behaviour undiminished by transgenerational acclimation. Nat. Clim. Chang. 4, 1086–1089 (2014).
Chivers, D. P. et al. Impaired learning of predators and lower prey survival under elevated CO2: a consequence of neurotransmitter interference. Glob. Chang. Biol. 20, 515–522 (2014).
Fry, F. E. J. Effects of the environment on animal activity. Publ. Ontario Fish. Res. Lab. 68, 1–62 (1947).
Brett, J. R. The respiratory metabolism and swimming performance of young sockeye salmon. J. Fish. Res. Board Can. 21, 1184–1226 (1964).
Pörtner, H. O. & Peck, M. A. Climate change effects on fishes and fisheries: towards a cause-and-effect understanding. J. Fish Biol. 77, 1745–1779 (2010).
Pörtner, H. O. & Knust, R. Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 315, 95–97 (2007).
Hoffmann, A. A. & Hercus, M. J. Environmental stress as an evolutionary force. Bioscience 50, 217–226 (2000).
Killen, S. S., Marras, S., Ryan, M. R., Domenici, P. & Mckenzie, D. J. A relationship between metabolic rate and risk-taking behaviour is revealed during hypoxia in juvenile European sea bass. Funct. Ecol. 26, 134–143 (2012).
Lynch, M. & Walsh, B. Genetics and Analysis of Quantitative Traits. (Sinauer Associates, Inc., 1998).
Munday, P. L., Donelson, J. M. & Domingos, J. A. Potential for adaptation to climate change in a coral reef fish. Glob. Chang. Biol. 23, 307–317 (2017).
Schunter, C. et al. Molecular signatures of transgenerational response to ocean acidification in a species of reef fish. Nat. Clim. Chang. 6, 1014–1018 (2016).
Welch, M. J. & Munday, P. L. Heritability of behavioural tolerance to high CO2 in a coral reef fish is masked by nonadaptive phenotypic plasticity. Evol. Appl. 10, 682–693 (2017).
Heuer, R. M. & Grosell, M. Physiological impacts of elevated carbon dioxide and ocean acidification on fish. Am. J. Physiol. Regul. Integr. Comp. Physiol. 307, R1061–R1084 (2014).
Nilsson, G. E. et al. Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nat. Clim. Chang. 2, 201–204 (2012).
Heuer, R. M., Welch, M. J., Rummer, J. L., Munday, P. L. & Grosell, M. Altered brain ion gradients following compensation for elevated CO2 are linked to behavioural alterations in a coral reef fish. Sci. Rep. 6, 33216 (2016).
Schulte, P. M. The effects of temperature on aerobic metabolism: towards a mechanistic understanding of the responses of ectotherms to a changing environment. J. Exp. Biol. 218, 1856–1866 (2015).
Rombough, P. The effects of temperature on embryonic and larval development. In Global warming: implications for freshwater and marine fish (eds. Wood, C.M. & McDonald, C.G.) 177-223 (Cambridge University Press, 1997).
Melzner, F. et al. Physiological basis for high CO2 tolerance in marine ectothermic animals: pre-adaptation through lifestyle and ontogeny? Biogeosci. 2313–2331 (2009).
Almany, G. R. & Webster, M. S. The predation gauntlet: early post-settlement mortality in reef fishes. Coral Reefs 25, 19–22 (2006).
Burton, T., Killen, S. S., Armstrong, J. D. & Metcalfe, N. B. What causes intraspecific variation in resting metabolic rate and what are its ecological consequences? Proc. R. Soc. B Biol. Sci. 278, 3465–3473 (2011).
Sih, A. et al. Animal personality and state-behaviour feedbacks: a review and guide for empiricists. Trends Ecol. Evol. 30, 50–60 (2015).
Jacobson, L. Hypothalamic-pituitary-adrenocortical axis regulation. Endocrinol. Metab. Clin. N. Am. 34, 271–292 (2005).
Kelly, M. W. & Hofmann, G. E. Adaptation and the physiology of ocean acidification. Funct. Ecol. 27, 980–990 (2013).
Kavanagh, K. D. Larval brooding in the marine damselfish Acanthochromis polyacanthus (Pomacentridae) is correlated with highly divergent morphology, ontogeny and life-history traits. Bull. Mar. Sci. 66, 321–337 (2000).
Dickson, A. G., Sabine, C. L. & Christian, J. R. Guide to best practices for ocean CO2 measurements. PICES Spec. Publ. 3, 191 (2007).
Mehrbach, C., Culberson, C. H., Hawley, J. E. & Pytkowitz, R. M. Measurement of the aparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol. Oceanogr. 18, 897–907 (1973).
Dickson, A. G. & Millero, F. J. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep Sea Res. Part A, Oceanogr. Res. Pap. 34, 1733–1743 (1987).
Dickson, A. G. Standard potential of the reaction AgCl(s) + 1/2H2 (g) = Ag(s) + HCl(aq) and the standard acidity constant of the ion HSO4 − in synthetic sea water from 273.15 K to 318.15 K. J. Chem. Thermodyn. 22, 113–127 (1990).
Larson, J. K. & McCormick, M. I. The role of chemical alarm signals in facilitating learned recognition of novel chemical cues in a coral reef fish. Anim. Behav. 69, 51–57 (2005).
Mitchell, M. D., McCormick, M. I., Ferrari, M. C. O. & Chivers, D. P. Friend or foe? The role of latent inhibition in predator and non-predator labelling by coral reef fishes. Anim. Cogn. 14, 707–714 (2011).
Chivers, D. P., McCormick, M. I., Allan, B. J. M. & Ferrari, M. C. O. Risk assessment and predator learning in a changing world: understanding the impacts of coral reef degradation. Sci. Rep. 6, 32542 (2016).
Ferrari, M. C. O. et al. Effects of ocean acidification on learning in coral reef fishes. PLoS One 7, e31478 (2012).
Chivers, D. P., Dixson, D. L., White, J. R., McCormick, M. I. & Ferrari, M. C. O. Degradation of chemical alarm cues and assessment of risk throughout the day. Ecol. Evol. 3, 3925–3934 (2013).
Rummer, J. L., Binning, S. A., Roche, D. G. & Johansen, J. L. Methods matter: considering locomotory mode and respirometry technique when estimating metabolic rates of fishes. Conserv. Physiol. 4, cow008 (2016).
Roche, D. G., Binning, S. A., Bosiger, Y., Johansen, J. L. & Rummer, J. L. Finding the best estimates of metabolic rates in a coral reef fish. J. Exp. Biol. 216, 2103–2110 (2013).
Niimi, A. J. & Beamish, W. H. Bioenergetics and growth of largemouth bass (Micropterus salmoides) in relation to body weight and temperature. Can. J. Zool. 52, 447–456 (1974).
Clark, T. D., Sandblom, E. & Jutfelt, F. Aerobic scope measurements of fishes in an era of climate change: respirometry, relevance and recommendations. J. Exp. Biol. 216, 2771–2782 (2013).
Laubenstein, T. et al. Correlated effects of ocean acidification and warming on behavioral and metabolic traits of a large pelagic fish. Diversity 10, 35 (2018).
Hess, S. et al. Species-specific impacts of suspended sediments on gill structure and function in coral reef fishes. Proc. R. Soc. B Biol. Sci. 284, 20171279 (2017).
McLeod, I. M. et al. Climate change and the performance of larval coral reef fishes: the interaction between temperature and food availability. Conserv. Physiol. 1, cot024 (2013).
Killen, S. S. et al. Aerobic scope predicts dominance during early life in a tropical damselfish. Funct. Ecol. 28, 1367–1376 (2014).
Ferrari, M. C. O. et al. Interactive effects of ocean acidification and rising sea temperatures alter predation rate and predator selectivity in reef fish communities. Glob. Chang. Biol. 21, 1848–1855 (2015).
R Core Team. R: A language and environment for statistical computing. R Found. Stat. Comput. Vienna, Austria (2014).
Acknowledgements
We thank S. McMahon, G. Rodgers, B. Spady, D. Warren, and S. Watson for assisting in the collection of the fish used in this study and A. Downie, C. Gervais, S. McMahon, M. Morin, T. Nay, L. Prescott, and L. Wenk for assistance in the laboratory. Staff at James Cook University Marine and Aquaculture Research Facility provided outstanding logistical support throughout the project. We thank R. Jones for statistical support. This research was supported by funding from the Australian Research Council (ARC) to P.L.M. and the ARC Centre of Excellence for Coral Reef Studies to P.L.M. and J.L.R.
Author information
Authors and Affiliations
Contributions
P.L.M., T.D.L., J.L.R., and M.I.M. conceived and designed the experiments. T.D.L. performed the experiments and analysed the data. T.D.L. drafted the first version of the manuscript with input from P.L.M, M.I.M., and J.L.R.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laubenstein, T.D., Rummer, J.L., McCormick, M.I. et al. A negative correlation between behavioural and physiological performance under ocean acidification and warming. Sci Rep 9, 4265 (2019). https://doi.org/10.1038/s41598-018-36747-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-36747-9
This article is cited by
-
Neuromolecular responses in disrupted mutualistic cleaning interactions under future environmental conditions
BMC Biology (2023)
-
Links between behaviour and metabolic physiology in fishes in the Anthropocene
Reviews in Fish Biology and Fisheries (2022)
-
Methods matter in repeating ocean acidification studies
Nature (2020)
-
Elevated temperature, but not decreased pH, impairs reproduction in a temperate fish
Scientific Reports (2020)
-
Swimming performance of marine fish larvae: review of a universal trait under ecological and environmental pressure
Reviews in Fish Biology and Fisheries (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.