Abstract
γ-aminobutyric acid (GABA) is a small signaling molecule that accumulates rapidly in plants exposed to various stresses; however, it has not been applied in regulating waterlogging tolerance in maize seedlings. Here, the effect of exogenous application of GABA in the determined optimal concentration was performed on seedlings of two maize cultivars under waterlogging treatments initiated at 3-leaf (V3) and 5-leaf stages (V5) in repeated experiments in 2016 and 2017. Chloroplast ultrastructure, photosynthesis, antioxidant capacity, and reactive oxygen species (ROS) production in the leaves were examined and compared with the corresponding values under normal soil water treatment (CK) and waterlogging treatment (WL). Compared with WL treatment, application of GABA significantly increased aboveground and root dry matter by 19.0% and 61.0%, promoted photosynthetic rate and chlorophyll content by 19.8% and 36.0%, increased the number of grana per chloroplast by 36.0%, fortified antioxidants (SOD, POD, CAT, GR, APX, VC) activities by 14.7–42.7%, and reduced the content of MDA, H2O2, and O2− by 30.5%, 32.5%, and 21.8%, respectively (p < 0.05). Collectively, GABA application was shown to promote the growth of maize seedlings under waterlogging, by down regulating ROIs-producing enzymes, activating antioxidant defense systems, and improving chloroplast ultrastructure and photosynthetic traits.
Similar content being viewed by others
Introduction
Globally, maize (Zea mays L.) is one of the most important cereal crops of tropical and subtropical environments. Like other crops, maize is more sensitive to waterlogging, particularly at the seedling stage, which is considered one of the major threats to the production of this crop1,2. In the tropical and subtropical region of China, most rainfall occurs in the maize growing season and excessive rainfall and flooding has caused heavy loss of crop growth and yield3,4. Global climate change contributes to an increasingly unpredictable rain pattern and is causing flooding that is expected to increase crop yield losses5. Globally, it is estimated that 12% of cultivated areas are affected by waterlogging, resulting in a 20% decrease in crop production6,7 and increasing annual losses to billions of dollars8. Waterlogging depletes the oxygen in soil because of slow diffusion and high consumption by plant roots9,10. When maize crop is subjected to waterlogging, stomatal closure and declines in photosynthetic rates are common responses11,12. Excessive soil moisture limits plant growth by altering morphological, physiological, and anatomical mechanisms13,14,15. Waterlogging decreases superoxide dismutase (SOD), peroxidase (POD), ascorbate peroxidase (APX) and catalase (CAT) enzyme activities, causing damage to the protective enzyme system. Waterlogging also regulates the malondialdehyde (MDA) content, resulting in membrane deterioration, leaf chlorosis, and increased leaf senescence16,17,18.
Chloroplasts are the major sites for generating reactive oxygen species (ROS) under environmental stress conditions19. Accumulation of excessive ROS could lead to oxidation of biomolecules, and such processes trigger oxidative damages to proteins, nucleic acids, and lipids and peroxidation of thylakoid membrane lipids20. Previous researchers showed that waterlogging stress changed the shape and internal structure of chloroplasts and their volume, and also damage the structure and integrity of cell membrane and mitochondria1. The physiological reaction to waterlogging results in reduced leaf area, chlorophyll content, chlorophyll fluorescence, and destruction of chloroplast ultrastructure in maize1. The structure of mesophyll cell and chloroplast morphology is an essential component of photosynthesis and plays a key role in determining photosynthetic assimilation capacity21. The morphology and ultrastructure of chloroplasts directly affects photosynthesis and significantly reduces the dry matter accumulation and yield of maize crop1,2,6,7,8,13,15,16,17,19,20,21,22.
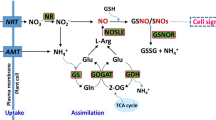
Plant growth regulators play an important role in modulating plant physiological responses to adapt to an unfavorable environment23, and their exogenous application is an effective way to improve crop tolerance to waterlogging stress. Aminobutyric acid (GABA) is an important four- carbon, non-protein amino acid present in animals, plants, and other organisms, and is involved in certain physio-biochemical functions for the regulation of plant growth and stress tolerance, such as signaling, regulation of redox status, sustenance of cytosolic pH, osmotic pressure, C and N metabolism, and C-N fluxes24,25,26. Current studies indicate that GABA responds rapidly to abiotic stresses such as waterlogging, salt stress, heat shock, low temperature, mechanical stimulation, and plant hormones27,28,29,30,31. Moreover, exogenous application of GABA regulates the gene expressions of NO3− uptake and NO3− transport (BnNrt2) in the Brassica napus32 and 14-3-3 in Arabidopsis thaliana33. It has also been reported that gene regulation in GABA application is associated with H2O2 and ethylene production in the roots of caragana34. GABA plays a critical role in alleviating oxidative damage through activation of the antioxidant enzymes, which constitute a defense system against ROS in wheat plants under waterlogging stress35. Furthermore, application of exogenous GABA imparts partial protection from heat stress to rice seedlings by improving leaf turgor and up-regulating the expression of osmoprotectants and antioxidants36. Several other studies have indicated that the exogenous application of GABA promotes photochemical efficiency, chlorophyll biosynthesis, photosynthetic activities, enzymatic and non-enzymatic responses, and membrane stabilization in different crops31,37,38. Although endogenous GABA levels in plants are very low, it is produced rapidly by plants under stressful conditions39.
However, the adaptation of plants under abiotic stress depends on comprehensive responses based on physiological and biochemical changes, and the palliative effects of GABA on waterlogging stress still has not been fully elucidated. Therefore, in this study the exogenous application of GABA was initiated under waterlogging conditions at the third leaf stage (V3) and fifth leaf stage (V5), respectively, for two popular hybrid maize varieties in China: Zhengdan-958 (ZD-958) and Xing Ken-6 (XK-6). Plant growth responses and physiological adaptive mechanisms were evaluated; specifically, leaf gas exchange parameters, chlorophyll content, enzymatic and non-enzymatic systems, lipid peroxides and ROIs- accumulation, and chloroplast ultrastructure were examined. Our findings here will improve our understanding of the adaptation, survival, and tolerance of maize under waterlogging stress and will help to improve the growth status for higher yields under natural conditions.
Results
GABA application increased the GABA content in leaves under waterlogging stress
As shown in Fig. 1, in comparison with CK treatment, GABA content in the leaves at V5 stage under the WL treatment was significantly increased, by 18.6% in XK-6 cultivar and 31.3% in ZD-958 cultivars in 2016 (p ≤ 0.05); however, slight increases in GABA content in the leaves at V3 stages under WL treatment were observed. Similar changes in GABA content were obtained between WL and CK treatments in 2017. Remarkably, exogenous application of GABA significantly increased GABA content in the leaves of maize seedlings under waterlogging stress at V3 and V5 stages (Fig. 1A,B). Compared with the WL and CK treatments, the GABA treatment significantly increased GABA content in the leaves at V3 stage, by 19.6% and 31.2% in XK-6, and by 13.7% and 29.3% in ZD-958, respectively, averaged across two experimental years. Moreover, improvement of GABA content was greater at V5 stages than V3 stages under GABA treatment in both maize varieties and years (Fig. 1A,B).
The GABA contents in leaves at 14 d after treatments initiated at third leaf (V3) and fifth leaf (V5) stage of maize under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA) in 2016 and 2017. Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
GABA application enhances seedling growth under waterlogging stress
Results show that waterlogging for 14 d significantly (p ≤ 0.05) decreased plant height (PH), green leaf area (GLA), and shoot and root dry matter (DM) of two varieties, when compared with control plants (CK) in both years (Table 1). Noticeably, exogenous GABA application improved the performance in these growth parameters under waterlogging conditions compared with the WL plants (p ≤ 0.05), though they still showed weakness when compared with the CK plants. Among these growth parameters, root DM was increased by the largest extent for GABA application under waterlogging conditions, with increases of 57.6% at V3 stage and 74.4% at V5 stage, respectively, averaged across both varieties and years, when compared with the WL plants (Table 1). GABA treatment also showed a positive effect on aboveground DM of waterlogged plants for each variety at each treated growth stage, with an average increase of 19.0%. Green leaf area and plant height of maize seedlings under the GABA treatment was increased by 21.1% and 18.4%, relative to the WL treatment, averaged across both varieties, stages, and years, respectively, though no significant changes were observed at V3 stages in 2017.
GABA application improves photosynthesis under waterlogging stress
Leaf gas exchange parameters of maize seedlings were significantly decreased by waterlogging stress, when compared with the control plants at V3 and V5 stages (Fig. 2). Under waterlogging, average net photosynthetic rate (Pn), stomatal conductance (Gs), intercellular CO2 (Ci), and transpiration rate (Tr) of maize seedlings were decreased by 49%, 48%, 46%, and 56% in XK-6 and 42%, 38%, 35%, and 51% in ZD-958, respectively, when compared with that under CK (Fig. 2). The plants treated with exogenous GABA application improved Pn, Gs, Ci, and Tr by 19.8%, 19.4%, 21.5%, 26.2%, respectively, compared with WL treatments averaged across two varieties, stages, and years. However, there was no significant increase in GABA application on photosynthetic rate (Pn) and transpiration rate (Tr) were observed in 2016 years compared with WL treatment. Minimal differences were observed in leaf gas exchange parameters between two varieties under the same treatment.
Effect of exogenous GABA application on net photosynthetic rate (Pn), (A,B) stomatal conductance (Gs), (C,D) intercellular CO2 concentration (Ci), (E,F) and transpiration rate (Tr), (G and H) in leaves at 14 d after treatment initiated at third leaf (V3) and fifth leaf (V5) stage of maize in 2016 and 2017. Samples were analyzed under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA). Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
GABA application decreases lipid peroxidation and ROIs-producing enzymes under waterlogging stress
Waterlogging stress triggered the lipid peroxidation and production of ROIs; thus, accumulation of Malondialdehyde (MDA), hydrogen peroxide (H2O2), ⋅O2−, and ⋅OH− were remarkably increased in waterlogged maize seedlings (Fig. 3). The values of MDA, H2O2, ⋅O2−, and ⋅OH− content in maize leaves were obviously alleviated under GABA treatment by 30.5%, 32.5%, 21.8%, and 21.0%, averaged across different varieties, stages, and years, respectively, compared with the WL treatment (Fig. 3). It appears that GABA application had a more positive effect on reducing MDA and H2O2 accumulation than on ⋅O2− and ⋅OH−.
Determination of changes in malondialdehyde (MDA) content (A,B), hydrogen peroxide (H2O2) content (C,D), superoxide anion radical production (O2˙−) content (E,F) and hydroxyl ion (OH−) content (G,H) in leaves at 14 d after treatment initiated at third leaf (V3) and fifth leaf (V5) stage of maize in 2016 and 2017. Samples were analyzed under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA). Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
In contrast, the activities of ROIs-producing enzymes, monoamine oxidase and xanthine oxidase (MAO and XOD), in the leaves were significantly higher under WL treatment and significantly reduced in GABA application treatments in most cases (Fig. 4). In plants treated with exogenous GABA, the activity of MAO and XOD were restrained by 18.0% and 21.6% in XK-6 and 27.6% and 39.1% in ZD-958, respectively, compared with that under the WL stressed plants averaged across two stages and years (Fig. 4). GABA application had a more profound effect on MAO than on XOD, and on ZD-958 than on XK-6. However, no significant changes in MAO at the V5 stage in 2016 and in XOD at the V3 stage in 2017 were found between XK- 6 plants under GABA and WL treatments (Fig. 4A,C).
Activities of monoamine oxidase (A,B) and Xanthine Oxidase (C,D) in leaves at 14 d after treatment initiated at third leaf (V3) and fifth leaf (V5) stage of maize in 2016 and 2017. Samples were analyzed under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA). Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
GABA application fortifies antioxidants in cells under waterlogging stress
Although antioxidant enzyme activities in leaves had apparent variations across different maize growth stages and experimental years, their values were significantly increased under GABA application and WL treatment (p ≤ 0.05) compared with the control under the same stage in the same year (Fig. 5). Moreover, the activities of superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), ascorbate peroxidase (APX) and glutathione reductase (GR), were significantly (p ≤ 0.05) increased under GABA application, when compared with the same maize variety under WL treatment at both V3 and V5 stages. The increase in CAT under GABA application was particularly high, with an increase of 42.7% averaged across varieties, stages, and years (Fig. 5C). Exogenous GABA application enhanced SOD and GR activities, with increases of 32.2% and 35.5%, respectively, averaged across varieties, stages, and years. A relatively small increase in APX was found under GABA treatment (14.7%); meanwhile, increases in APX were similar among different varieties and growth stages in both years (Fig. 5E). As shown in Fig. 4A–D, the ZD-958 variety under GABA treatment showed a higher increase in SOD, POD, CAT, and GR at the V3 stage (40.0%, 38.8%, 58.1%, and 45.0%, respectively), while the XK-6 variety under GABA treatment only showed increases of 25.0%, 20.7%, 26.8%, and 25.8% at the same stage, respectively. At the V5 stage, increases in SOD, POD, and GR were similar between varieties under GABA application; however, increases in CAT activity in ZD-958 leaves under GABA treatment was higher than that in XK-6 leaves compared with the same variety under WL treatment (Fig. 5C).
The activities of superoxide dismutase (SOD) (A), peroxidase (POD), (B), catalase (CAT), (C), ascorbate peroxidase (APX) (D) and glutathione reductase (GR) (E) in leaves at 14 d after treatment initiated at third leaf (V3) and fifth leaf (V5) stage of maize in 2016 and 2017. Samples were analyzed under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA). Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
The GSH content did not show consistently significant increases in the WL treated maize seedlings, when compared with the CK plants (Fig. 6A,B). At V3 and V5 stages, the GSH content in plants treated with exogenous GABA was slightly increased, by 2.8% and 2.4% in XK-6 and by 4.5% and 3.9% in ZD-958, respectively, when compared with waterlogging-stressed plants. Nevertheless, the VC content were significantly enhanced in both growth stages of two varieties under waterlogging conditions with GABA application, when compared with plants under CK or WL treatments (Fig. 6C,D). After spraying exogenous GABA, the VC content increased by 29% and 31% in XK-6 and ZD-958 at the V3 stage and 19% and 26% at V5 stage, respectively, compared with the WL treatment.
Effects of spraying GABA after waterlogging on non-enzymatic antioxidants in leaves at 14 d after treatment initiated at third leaf (V3) and fifth leaf (V5) stage of maize in 2016 and 2017. GSH (A,B) and VC (C,D) Samples were analyzed under normal soil water conditions (CK), waterlogging treatment (WL) and aminobutyric acid treatment for waterlogged plants (GABA). Error bars represent the standard error (S.E.) of mean (n = 3). Different letters indicate significant differences at P < 0.05 based on the Least Significant Difference test.
GABA application increases chlorophyll content and chloroplasts under waterlogging stress
Chlorophyll content of maize leaves was significantly affected (p ≤ 0.05) at V3 and V5 stages by waterlogging stress compared with CK plants (Table 2). WL treatment significantly decreased chlorophyll content, with reductions of 44% and 44% at the V3 stage for XK-6 and ZD-958, respectively, and 41% and 52% at V5 stage for XK-6 and ZD-958, respectively, averaged across years, when compared with CK. By contrast, chlorophyll content was significantly increased in the GABA treatments, by 32% and 43%, at the V5 stage and 32% and 37% at the V3 stage (p ≤ 0.05) for XK-6 and ZD-958, respectively, when compared with the WL treatment (Table 2). Chloroplast numbers per mesophyll cell in leaves of two varieties were significantly reduced (p ≤ 0.05) under WL conditions at two stages in two years (Table 2). At the V5 stage, the GABA and WL treatments exhibited reduced number of chloroplasts, by 26% and 42% in XK-6 and by 31% and 52% in ZD-958, respectively, averaged across years. However, the plants treated with exogenous GABA application under waterlogging stress exhibited obvious increases number of chloroplast at both stages of two varieties in 2016 in compared with WL treatments, though these increases were not observed in 2017 year.
GABA application improved chloroplast ultrastructure in mesophyll cells and mitochondrion structure
In control plants, the chloroplasts had complete external envelopes and clear boundaries, the thylakoid were well-developed with good shape, the lamella structure pile folds were in order, and both grana lamella and stroma lamellae were arranged compactly and clearly in both maize cultivars (Figs 7D–14D). However, chloroplast configuration became irregular under WL conditions. Chloroplasts in maize seedlings under waterlogging stress were characterized by compressed grana lamellae, disrupted stroma lamellae, and distorted thylakoid. Additionally, the number of grana and grana lamellae were reduced significantly to varying degrees (Figs 7G–10G and 12G–14G). At the V3 stage, the number of grana was reduced by 49.8% and 43.7% in XK-6 and ZD-958 plants under WL treatment, respectively, compared with CK (Table 2, Figs 7 and 8). However, the plants treated with GABA application had a consistently positive effect on number of grana of maize varieties under waterlogging stress in both years, and significantly increased the number of grana per chloroplast by an average of 41% and 31% in XK-6 and ZH-958, respectively, relative to WL treatment (Table 2). The size of chloroplasts in maize seedlings waterlogged under WL treatment and GABA treatment were obviously reduced compared with those in CK plants (Table 2, Figs 7, 8 and 14). In comparison with CK plants, the length and width of chloroplasts under WL treatments were significantly decreased by 33% and 43% in XK-6 and 30% and 48% in ZD-958, averaged across stages and years (Table 2). However, application of GABA to waterlogged maize seedlings did not result in increased chloroplast size in most experimental conditions (Table 2). In contrast with the WL treatment, significant increases in the length of chloroplasts by GABA treatment were only found in XK-6 plants at the V3 stage in 2016 and in ZD-958 plants at the V5 stage in 2017 (Table 2). Additionally, only maize seedlings at the V3 stage of XK-6 in 2017 increased in chloroplast width under GABA treatment (Table 2). Nevertheless, exogenous GABA alleviated the ultrastructure changes of chloroplasts in mesophyll cells induced by waterlogging (Figs 7–9, 12 and 14). The plants treated with GABA maintained a well-protected internal lamellar system in the chloroplasts of waterlog-stressed leaves in both maize seedlings (Figs 7A–9A, 12A and 14A).
Chloroplast ultrastructure in leaves of XK-6 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedlings (GABA) at 14 d after treatment initiated at third leaf (V3) in 2016. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of ZD-958 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedlings (GABA) at 14 d after treatment initiated at third leaf (V3) in 2016. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of XK-6 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedlings (GABA) at 14 d after treatment initiated at five leaf (V5) stage in 2016. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of ZD-958 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedlings (GABA) at 14 d after treatment initiated at five leaf (V5) stage in 2016. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of XK-6 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedlings (GABA) at 14 d after treatment initiated at third leaf (V3) in 2017. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of ZD-958 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedling (GABA) at 14 d after treatment initiated at third leaf (V3) in 2017. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of XK-6 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seedling (GABA) at 14 d after treatment initiated at five leaf (V5) stage in 2017. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
Chloroplast ultrastructure in leaves of ZD-958 maize cultivars under different treatments. Chloroplast ultrastructure under GABA application for waterlogging maize seeding’s (GABA) at 14 d after treatment initiated at five leaf (V5) stage in 2017. Ch: chloroplast, GL: grana lamella, P: particles, CM: chloroplast envelope membrane, CW: cell wall, Mi: mitochondria, BS: bundle sheath.
In control leaf cells, mitochondria were arranged randomly in the cytoplasm and were almost elliptical or circular. The double membrane structure was complete and crests were clearly visible (Figs 7E–10E, 12E and 14E). However, waterlogging stress altered the structure of mitochondria to certain extent. Mitochondria in the WL treatment were in clustered arrangements near the chloroplasts and were not clearly visible. (Figs 7H–9H, 12H and 14H). In contrast, there was no effect on mitochondria structure in the GABA treatment when compared with the WL treatment (Figs 11E and 14E). However, GABA alleviated the waterlogging-induced damage of mitochondria structure. An increased number of mitochondria and improved mitochondria structure were observed in both growth stages (Figs 7B–10B,12B and 14B).
Discussion
GABA is the main component of the free ammonia acid library in plant cells. GABA usually accumulates in plants experiencing environmental stress, such as oxygen deficit, chilling, heat stimulus, or mechanical damage39,40. Similarly, increases in GABA content in maize seedling leaves under waterlogging stress were observed in this study (Fig. 1). GABA is a non-protein four-carbon amino acid that can be absorbed directly by plants25,26,27,28. In the present study, exogenous application of GABA significantly enhanced GABA content in maize seedlings under waterlogging conditions at V3 and V5 stages compared with CK and WL treatments (Fig. 1A,B). Such absorption of exogenous GABA by plants has also been reported in previous studies41,42. Moreover, we found that maize seedlings at the V5 stage had a stronger ability to produce GABA and absorb exogenous GABA than the plants at V3 stages under waterlogging stress (Fig. 1A,B); this might be one of the reasons that maize plants at the V5 stage had a greater ability to tolerate waterlogging stress, shown by better growth attributes in these plants (Table 1).
It has been reported that GABA can ameliorate plant growth under different abiotic stress conditions in maize, rice, brassica, and wheat32,36,38,39,40,41,42,43. However, GABA’s role in plants has remained unclear, while its role as a neurotransmitter in animal cells is well recognized. The major roles of GABA are regulating cytosolic pH, anti-oxidative enzymatic systems, buffering C and N metabolism, osmoregulation, armoring against oxidative stress, and signal transduction, and these might lead to improvement in overall plant performance44,45. The effect of GABA application to maize seedlings under waterlogging stress was examined in the present study, and it was found that waterlogging stress had profound adverse effects on maize seedling growth (Table 1), similar to reports in previous studies3,14,46,47. However, exogenous GABA application improved these plant growth attributes when plants were subjected to waterlogging stress (Table 1). Exogenous GABA application on leaves significantly increased the plant height and leaf area of waterlogging-treated Pakchio seedlings31. Also, exogenous GABA was reported to increase the dry weight of maize under hypoxia stress48. In our study, compared with WL treatment, GABA application improved the tolerance of maize seedling to waterlogging stress, showing that GABA application increased the plant height, leaf area, and biomass at two treated growth stages of maize cultivars XK-6 and ZD-958 (Table 1). Previously, studies found that GABA-induced improvement in many plant species could be due to improved photosynthetic activities, relative water content, osmolyte accumulation, leaf turgor, and other related physio-metabolical mechanisms36,49,50. GABA application might have improved the maize seedlings’ growth by inciting cell elongation and division and/or by maintaining metabolic balance within plant tissues. The results of the current study further confirm the beneficial effects of GABA application in maize under waterlogging conditions. Studies examining the physiological mechanisms underpinning the regulation of GABA on waterlogged maize seedlings are further needed.
Previous studies have found that the chloroplasts of plants subjected to abiotic stress are a major source of ROS, such as O2− and H2O2, which are generated by the direct transfer of the excitation energy from chlorophyll or by oxygen reduction in the Mehler reaction51,52. In turn, the overproduction of ROS leads to lipid peroxidation and accumulation of MDA53. Our study indicated that waterlogging stress activated the production of H2O2 content, OH− content, and O2− content in maize seedlings, when compared with CK (Fig. 3A,C,D). The level of enhancements of these ROIs also coincided with a higher lipid peroxidation rate, which is regarded as a biochemical maker for free radicals mediated by the plant’s injury (Fig. 2A). These results are consistent with previous studies that examined the increments of ROIs production and MDA content under waterlogging stress1,17,19,23,24,26,28,29,30,31,34,35,36,37,38,41,42,43,46,48,49,50,51,52,53,54. The results of the present experiment showed that GABA treatment significantly decreased MDA content in both maize cultivars relative to the WL treatment (Fig. 3A,B), indicating that the application GABA effectively ameliorated the damages of waterlogging stress on the cell membrane system. It was also proposed that exogenous GABA was efficient at reducing the oxidative damage to cell membranes in melon seedlings under hypoxic conditions55, and tomato seedlings under chilling stress42. In the present study, we found that GABA might be involved in mitigating lipid peroxidation under waterlogging stress, resulting in a significant decrease in the accumulation of OH−, H2O2, and O2−. Our results showed that oxidative stress induced by waterlogging stress and damage to maize seedlings were effectively alleviated after exogenous GABA application. Previously, the beneficial effects of GABA have been observed under abiotic stress in maize seedlings28,38,39,44,45,46,48,51,54,55,56.
Commonly, photosynthetic apparatus, mitochondrial respiration, and photorespiration are considered as the main sources for generating ROIs under control conditions. Nevertheless, some other sources, such as XOD and MAO, also contribute to an active production of ROIs under abiotic stress57. The MAO, a flavoprotein localized on the outer membrane of mitochondria, catalyzes the oxidative deamination of aromatic amines and produces a large quantity of H2O2, which ultimately contributes to an increase in the steady state concentrations of ROIs within the plant cell58. Likewise, XOD can generate toxic ⋅O2− as well as H2O2 in the plant cell and sometimes high production of ⋅O2− from a XOD system can cause epidermal cell death that cannot be prevented by plant defense system59. In the current study, the activities of ROIs-producing enzymes of MAO and XOD were significantly higher under waterlogging stress treatments but remained lower under control conditions at different growth stages (Fig. 4). These results are in line with previous research, which found that the abiotic stress increased the activities of MAO and XOD in rice seedlings60. The activities of these two enzymes were concomitant with the level of ROIs in maize cultivars (Figs 3 and 4). For instance, MAO and XOD activities were significantly higher under waterlogging treatment, which led to higher levels of ROIs production. However, MAO and XOD activities were significantly lowered in plant leaves treated with the GABA application (Fig. 4), which might have caused the down-regulation of ROIs accumulation and oxidative stress in maize seedlings (Figs 3 and 4).
Previous studies reported that the SOD, APX, POD and GR enzymes of the plant antioxidative defense system were triggered under water stress61,62. The results of the present experiment showed that the activities of SOD, POD, CAT, APX, and GR activities significantly increased under waterlogging stress relative to that under CK in both stages (Fig. 5). It also has been reported that waterlogging significantly increased the activity of enzymatic antioxidants in maize leaves under waterlogging stress62,63. SOD is considered a main antioxidant enzyme that provides the first line of defense against ROIs because it catalyzes the dismutation process of ⋅O2− to H2O264, while CAT, POD, and APX help to remove H2O2 by converting it to oxygen and water65,66. Interestingly, exogenous GABA application significantly improved the activities of these five antioxidative enzymes at both stages of two varieties in two years compared with CK and WL treatments (Fig. 5). Increased activity of antioxidant enzymes in maize seedlings under GABA application is in agreement with previous studies on waterlogging stress in wheat35. These findings indicate that exogenous GABA application protected maize seedlings from oxidative stress caused by excessive water through the inhibition of lipid peroxidation, decreasing ROI levels, and activating antioxidant enzyme defense systems (Figs 3 and 5). Several studies have documented that exogenous GABA stimulates activities of antioxidant enzymes and reduces oxidative stress under different environmental stressors30,35,36. However, among these antioxidant enzymes, CAT, GR, and SOD were significantly activated by GABA application, with increases of 42.7%, 35.5%, and 32.2%, respectively, compared with those under WL treatment (Fig. 5C,D,A). Additionally, generation of ROIs often caused membrane damage and disintegration of various cellular structures and organelles, ultimately causing cell death66,67,68,69. However, SOD and POD acted against ROIs in GABA-treated maize seedlings to protect membrane damage, showing that GABA can potentially maintain cell integrity. GABA-induced maintenance of higher antioxidant activity is crucial to improve a plant’s ability to tolerate oxidative stress. Further research needs to be conducted to understand the molecular mechanism by which GABA regulates these antioxidant enzymes.
Similar to plant growth attributes, photosynthesis is one of the primary processes affected by waterlogging stress. Leaf gas exchange parameters of maize seedlings at V3 and V5 stages of two varieties in two years were significantly hindered by waterlogging stress (Fig. 2). These results are in agreement with findings from previous studies1,46. It was reported that the decline of Pn induced by stress was triggered by stomatal closure, which leads to inhibition of ambient CO2 diffusion to the mesophyll69. A similar phenomenon was observed in this study, where declines in Gs and Ci occurred and led to reduction in Pn. Meanwhile, our current results (Table 2) are in accordance with recent studies that show waterlogging stress causes reductions in the chlorophyll content in maize seedlings1,46. Nevertheless, application of GABA alleviated waterlogging stress on gas exchanges parameters in most experimental conditions in this study, when compared with WL treatment in both maize cultivars (Fig. 2). Our results showed that exogenous application of GABA on maize leaves after waterlogging could potentially retard declines in leaf chlorophyll content caused by waterlogging stress, resulting in the enhancement of photosynthetic characteristics (Table 2). Enhancements of photosynthetic characteristics in response to GABA application on leaves have also been observed in Pakchio seedlings under waterlogging stress31, and pepper seedlings under low light stress29. Exogenous GABA application was further reported to enhance the photochemical efficiency of PSII in muskmelon seedlings under hypoxia stress37. It was also reported that GABA can be transformed into succinic acid via the catalysis of γ-aminobutyrate transaminase and succinate semialdehyde dehydrogenase45. Succinic acid is then involved in the tricarboxylic acid cycle to maintain the carbon–nitrogen cycle in plants, which is known as the GABA metabolic bypass70. GABA application resulted in improvements in net photosynthesis in maize seedlings, possibly due to maintenance of cell turgor, which promoted chlorophyll biosynthesis (Table 2) and reduced oxidative damage by regulating various physio-biochemical processes35,36,37.
Decreases in chlorophyll content and photosynthetic efficiency were mostly caused by the disturbance of chloroplast morphology and ultrastructure of functional leaves20,22. Chloroplasts are major sites for generating reactive oxygen species (ROS) under environmental stress conditions18. However, present studies showed that waterlogging damaged leaf chloroplast ultrastructure, the number of chloroplast and grana number per chloroplast were reduced in response to the disintegration in a portion of the granum and reorganization of the chloroplast, the integrity of ultrastructure of chloroplast was demolished, their membranes and thylakoids were deliquescent in both maize seedling (Figs 7G–10G and 12G–14G and Table 2). Leading to reduced chlorophyll content and photosynthetic assimilation capacity22,71. At the same time, the chloroplast ultrastructure might be affected or damaged by waterlogging stress resulting from ROIs accumulation, because chloroplasts are a major source of active oxygen in plant tissue18,72. However, GABA application obviously increased the number of chloroplasts and grana compared with the waterlogging treatment (Table 2). In GABA treated plants, the chloroplast structure became a normal oval shape and grana were clear and visible (Figs 7A–9A,12A and 14A). However, the plant treated with exogenous GABA application increased the size of chloroplast and number of grana per chloroplast as compared with waterlogging treatment in both maize verities (Table 2). These results show that GABA application enhanced photosynthetic assimilation capacity, possibly because of an alleviation of chlorophyll degradation and preservation of ultrastructure. It was previously observed that exogenous 6-BA application enhanced chlorophyll content and alleviated the degeneration of photosynthetic performance caused by waterlogging stress in maize seedlings42. However, GABA application has not been previously shown to have an effect on the ultrastructure of chloroplasts, and further studies are needed to clearly understand the mechanisms at the transcriptional molecular and gene level.
In summary, results of our study suggest that exogenous GABA application could enhance the growth of maize seedlings under waterlogging stress through the activation of antioxidative enzymes, alleviation of ROIs impacts, mitigation in damage on leaf chloroplast ultrastructure, and enhanced photosynthesis. Findings of this study provide insights into the physiological mechanisms underlying GABA-induced acclimation of maize to waterlogging stress and provides a possible solution to manage waterlogged maize at an early stage in the field.
Materials and Methods
Plant culture and experimental design
The experiment was conducted in 2016 and 2017 in a greenhouse to avoid the influence of rainfall on soil water treatment at the Huazhong Agricultural University in Wuhan, China (30°47′N, 114°35′E). Two maize hybrids commonly found in China were used in this study: Zhengdan-958 (ZD-958) and Xing Ken-6 (XK-6). The experiments were laid out in a randomized design with three replications. Before sowing, the healthy seeds of both maize varieties were sterilized by soaking in 1% (v/v) sodium hypochlorite for 30 min and then kept in the incubator for germination at 28 °C in darkness for about 3 d. Uniformly germinated seeds were selected and sown in soil in pots that had been prepared 10 d prior to the experiment and had already reached the appropriate soil water content for maize emergence. Six germinated seeds were sown in plastic pots (32 cm lower inside diameter, 34 cm upper inner diameter, 30 cm height); these pots were filled with 20 kg of sieved dry soil amended with 0.14 g urea, 0.14 g diammonium phosphate, and 0.18 g potassium chloride per kg soil and seedlings were thinned to three plants per pot at the one-leaf (V1) stage. Each treatment included 12 pots for each growth stage and each variety with three replicates. All measures against diseases and insect infestation of the maize seedlings were deployed at the appropriate time during the experimental period.
To examine the effects of GABA application on the growth of maize seedlings, waterlogging was initiated at third leaf stage (V3) and fifth leaf (V5) stages in both years. The pots intended for the waterlogging treatments were filled with water to 1–2 cm above the soil surface for 14 d. Different concentrations of GABA (0.25 mmol L−1, 1.00 mmol L−1, and 1.75 mmol L−1) were sprayed on plant leaves during waterlogging in 2016. Based on plant growth, chlorophyll content, and photosynthesis data in 2016 (Table S1), one optimal concentration of GABA (1 mmol L−1) was chosen and used in the validating experiments in 2016 and 2017, with same regime as described above. The waterlogged plants were sprayed three times with GABA (GABA treatment) 1 d, 3 d, and 6 d after waterlogging implementation. To prevent contamination to other treated plants, non-targeted pots were fully isolated with plastic film every time the plants were sprayed. A randomized complete block design was applied with three replications. Meanwhile, two control treatments were carried out each year, including normal soil moisture control treatment (CK) and waterlogging control treatment without spraying GABA (WL). Representative maize seedlings under different treatments at the V5 stage are shown in Figure S1.
Plant sampling and measurements
Twelve representative plants of each treatment were carefully removed from 4 selective pots for each variety at V3 and V5 stages at the end of the waterlogging period (14 d) and then separated into root parts and shoot parts. An upmost fully expanded young leaf was selected from three plants per replicate and leaves were quickly stored at −80 °C for physiological indicator analysis. Six plants from each treatment were rapidly transferred to ovens, dried at 105 °C for 30 min, and then dried at 80 °C to a constant mass and weight for dry matter determination. Growth was determined in terms of plant height, stem diameter, green leaf area, and dry weight. Green leaf area (GLA) was calculated according a method published previously73.
Measurement of Endogenous GABA Content
The endogenous GABA content was determined by Berthelot reaction with some modifications74. Leaves (0.1 g) were ground with methanol at room temperature. The homogenate was centrifuged at 5,000 g for 15 min and discarded the supernatant (2–3 times). The sediment was re-dissolved in 1.5 mL distilled water. Subsequently, the samples were heated in water bath at 50 °C for 2 h, and then centrifuged at 7,000 g for 15 min. One milliliter supernatant was added 0.1 mL 2 mol/L AlCl3 and oscillated. The mixture was cooled to room temperature and then centrifuged at 12,000 g for 10 min. The supernatant (0.5 mL) was shaken for 5 min with 0.3 mL KOH and centrifuged at 12,000 g for 5 min. The resulting supernatant was used to measure the content of GABA based on the following procedure: 0.3 mL supernatant was added to the reaction solutions [including 0.5 mL 0.1 mol/L sodium tetraborate (pH = 10.0), 0.4 mL 6% phenol and 0.6 mL 5% sodium hypochlorite]. The mixture was put into a boiling water for 10 min and rapidly placed in ice bath for 5 min. Finally, the solution was shaken with 2 mL 60% ethyl alcohol and measured the absorbance in 645 nm. The endogenous GABA content was calculated using a standard curve.
Gas exchange parameters and chlorophyll contents
Gas exchange parameters such as the net photosynthetic rate (Pn), stomatal conductance (Gs), intercellular CO2 (Ci), and transpiration rate (Tr) were measured in both years between 10:00 and 12:00 h, by using a LI-6400 portable photosynthesis system (LI-COR Inc., Lincoln, NE, USA) during three (V3) and fifth leaf (V5) stage of both maize varieties. Chlorophyll (Chl) contents were quantified by using the method75. Chlorophyll contents were extracted from 0.1 g leaf discs with 8 mL acetone (80%) and kept in dark conditions for 24 h. The absorbance of the supernatant was measured at 646 and 663 nm using spectrophotometer (UV2102; Unico, Shanghai, China).
Sample preparation and observation by Transmission Electron Microscope (TEM)
For leaf tissue preparation and electron microscopy, small leaf discs (4.0 mm × 1.2 mm) from within the gas exchange chamber were removed and infiltrated in a syringe with the fixative 2.5% glutaric aldehyde in 0.1 m phosphate buffer (pH = 7.6) at 4 °C, and post-fixed in 2% buffered osmium tetroxide at 20 °C for 2 h. The samples were embedded in Spurr’s epoxy resin (Sigma-Aldrich, St. Louis, USA). For light microscopy, semi thin leaf cross sections were stained with toluidine blue, and observed at 200× magnification with an Olympus IX71 light microscope (Olympus Optical, Tokyo, Japan). Ultrathin leaf cross sections were stained with 4% (w/v) uranyl acetate followed by 2% (w/v) lead citrate. Transmission electron microscope (H-7650; Hitachi – Science &Technology, Tokyo, Japan) and Soft Imaging System software (H-7650; Hitachi –Science & Technology, Tokyo, Japan) were used for observation and photography. The width and length of chloroplast was measured by software Image J, a free, Java-based image-processing package.
Determination of membrane lipid peroxidation, contents of H2O2, superoxide (O2) and contents of hydroxyl ion (OH−)
The biochemical analyses were carried out in the fresh leaves samples. MDA determination kit (A003-1), (Nanjing Jiancheng Bioengineering Institute, NJBI) according to the manufacturer’s instructions. Briefly, the leaf tissue homogenate was centrifuged at 12000 g and 4 °C for 15 min, and the supernatants were collected. MDA and thiobarbituric acid (TBA) mixture was produced during the reaction of MDA in samples with TBA, and then this mixture was measured at 535 nm and final result of MDA was expressed as mg−1 protein. H2O2 was extracted and its content was measured by monitoring the absorbance of the titanium-peroxide complex at 405 nm according to the method of Nanjing Jiancheng Bioengineering Institute (NJBI) determination kit. The contents of H2O2 were demonstrated as unit mg−1 protein. The contents of hydroxyl ion (⋅OH−) and superoxide anion radical (⋅O2−) in the leaves of maize seedlings were determined using the commercial OH− assay kit (A018) and O⋅−2 assay kit (A052), respectively, obtained from Nanjing Jiancheng Bioengineering Institute, China. The ⋅OH− was expressed as units mg−1 protein, and one unit was the amount required to reduce 1 M of H2O2 in the reaction mixture per minute at 37 °C. The ⋅O2− were demonstrated as units g−1 protein, and one unit was equivalent of the value required to inhibit superoxide anion by 1 mg of VC for 40 min at 37 °C.
Assay of Monoamine oxidase and Xanthine oxidase contents
The activities of reactive oxygen species -producing enzymes viz., XOD and MAO were measured by using the commercial kits in accordance with manufacturer’s instruction (Nanjing Jiancheng Bioengineering Institute, NJBI). The XOD contents was assayed by determination kit A034 (NJBI). The XOD was defined as 1 g of protein required transform as 1 µM of hypoxanthine to xanthine formed per minute at 37 °C. The activities of XOD enzymes were expressed as 1 unit mg−1 protein. The MAO contents was assayed by determination kit A002 (NJBI). The MAO was defined as the amount of enzyme that increased the absorbance by 0.01 at 37 °C in 1 hour; 1 unit g−1 protein. The activities of MAO enzymes were demonstrated as 1 unit mg−1 protein.
Assay of antioxidant enzymes
The antioxidants enzymatic were analyses by using the commercial kits in accordance with the manufacturer’s guidelines. The kits for antioxidants enzymatic were purchased from Nanjing Jiancheng Bioengineering Institute (NJBI). The SOD activity was assayed by determination kit A001 (NJBI). One unit of SOD activity was defined as the amount of enzyme required for 1 mg tissue protein in 1 ml of reaction mixture SOD inhibition rate to 50% as monitored at 550 nm. The activities of SOD were demonstrated with unit mg-1 protein. The determination of POD activity was determined with the assays kit A084-3 (NJBI). One unit of POD activity was defined as the amount of enzyme necessary for the decomposition of 1 μg substrate in 1 minute at 37 °C. The POD activities were expressed with unit mg−1 proteins. CAT activity was assayed by determination kit A007-2 (NJBI). One unit of CAT activity was defined as 1 mg tissue protein consumed 1 µmol H2O2 at 405 nm for 1 sec. The activities of CAT were demonstrated with units mg−1 protein. The determination of GR activity was determined with the assays kit A062 (Nanjing Jiancheng Bioengineering Institute). One unit of GR activity was defined as 1 g tissue protein consumed 1 mmol NADPH at 340 nm for 1 min. The activities of GR were demonstrated with units mg−1 protein. The determination of APX activity was determined with the assays kit A123 (NJBI). One unit of APX activity was defined as 1 mg tissue proteins catalysised 1μmol ascorbate at 290 nm for 1 min. The APX activity was demonstrated as units g−1 protein.
Statistical Analysis
Analysis of variance (ANOVA) were performed using the software Statistix 10.0 (Analytical Software, Tallahassee, FL, USA) Statistically significant differences between control and GABA treatment samples were tested by Fisher’s least significant difference (LSD) test at a probability of P < 0.05.
References
Ren, B., Zhang, J., Dong, S., Liu, P. & Zhao, B. Effects of waterlogging on leaf mesophyll cell ultrastructure and photosynthetic characteristics of summer maize. Plos One. 11, e0161424 (2016).
Arora, K. et al. Rnaseq revealed the important gene pathways controlling adaptive mechanisms under waterlogged stress in maize. Sci Reports. 7, 1–10950 (2017).
Ren, B. et al. Effects of waterlogging on the yield and growth of summer maize under field conditions. Canadian Journal of plant science. 94, 23–31 (2014).
Xie, X. L. Wang, W. Tian, W. W. & Xie, K. J. Waterlogging accelerates the loss of soil organic carbon from abandoned paddy fields in the hilly terrain in subtropical china. Scientific Reports. 7–1 (2017).
Cairns, J. E., Sonder, K., Zaidi, P. H. & Verhulst, N. Maize Production in a Changing Climate-Chapter one: Impacts Adaptation and Mitigation Strategies. Advance in Agronomy. 114, 1–58 (2012).
Jackson, M. B. & Colmer, T. D. Response and adaptation by plants to flooding stress. Annals of botany 96, 501–5 (2005).
Sergey, S. Physiological and cellular aspects of phyto-toxicity tolerance in plants: the role of membrane transporters and implications for crop breeding for waterlogging tolerance. New Phytologist. 190, 289–298 (2011).
Voesenek, L. A. & Baileyserres, J. Flood adaptive traits and processes: an overview. New Physiology. 206, 57–73 (2015).
Drew, M. C., He, C. J. & Morgan, P. W. Programmed cell death and aerenchyma formation in roots. Trends Plant Science. 5, 123–127 (2000).
He, L., Li, B. & Lu, X. The effect of exogenous calcium on mitochondria, respiratory metabolism enzymes and ion transport in cucumber roots under hypoxia. Scientific Reports. 5, 11391 (2014).
Zaidi, P. H., Rafique, S. & Singh, N. N. Response of maize (Zea mays L.) genotypes to excess soil moisture stress: Morpho-physiological effects and basis of tolerance. European Journal of Agronomy. 19, 383–399 (2003).
Folzer, H., Dat, J. F. & Capelli, N. Response of sessile oak seedlings (Quercus petraea) to flooding: an integrated study. Tree Physiology. 26, 759–766 (2006).
Cortezi, D. G. & Colli, S. Effect of flooding and application of plant growth regulators on sprouting of guazuma ulmifolia (malvaceae) and sesbania virgata (fabaceae). Brazilian Journal of Botany. 34, 423–430 (2011).
Abiko, T., Kotula, L., Shiono, K., Malik, A. I. & Colmer, T. D. Enhanced formation of aerenchyma and induction of a barrier to radial oxygen loss in adventitious roots of zea nicaraguensis contribute to its waterlogging tolerance as compared with maize (zea mays ssp. mays). Plant Cell and Environments. 35, 1618–1630 (2012).
Luan, H. et al. Elucidating the hypoxic stress response in barley (hordeum vulgare l.) during waterlogging: a proteomics approach. Scientific Reports. 8, 1 (2018).
Irfan, M., Hayat, S., Hayat, Q., Afroz, S. & Ahmad, A. Physiological and biochemical changes in plants under waterlogging. Protoplasma. 241, 3–17 (2010).
Puyang, X., An, M., Xu, L., Han, L. & Zhang, X. Antioxidant responses to waterlogging stress and subsequent recovery in two kentucky bluegrass (poa pratensis, l.) cultivars. Acta Physiologiae Plantarum. 37, 10–197 (2015).
Sun, W., Montagu, M. V. & Verbruggen, N. Small heat shock proteins and stress tolerance in plants. Biochim Biophys Acta. 1577, 1–9 (2002).
Partelli, F. L. & Batista, S. Characterization of the main lipid components of chloroplast membranes and cold induced changes in Coffea spp. Environmental and Experimental Botony. 74, 194–204 (2001).
Weston, E. Thorogood, K, Vinti, G. & López-Juez, E. Light quantity controls leaf-cell and chloroplast development in Arabidopsis thaliana wild type and blue-light-perception mutants. Planta. 211, 807–815 (20002).
Araki, H., Hanada, A., Hossain, M. A. & Takahashi, T. Waterlogging at jointing and or after anthesis in wheat induces early leaf senescence and impairs grain filling. Field Crops Research. 137, 27–36 (2012).
Shao, Q., Wang, H. & Guo, H. Effects of shade treatments on photosynthetic characteristics, chloroplast ultrastructure, and physiology of anoectochilus roxburghii. Plos One 9, e85996 (2014).
Gadallah, M. A. Effects of proline and glycinebetaine on vicia faba responses to salt stress. Biologia Plantarum. 42, 249–257 (1999).
Shelp, B. J., Bown, A. W. & Mclean, M. D. Metabolism and functions of gamma-aminobutyric acid. Trends in Plant Sciences. 4, 446–452 (1999).
Michaeli, S. & Fromm, H. Closing the loop on the GABA shunt in plants: are GABA metabolism and signaling entwined. Fronter and Plant Science. 6, 419 (2015).
Gilliham, M. & Tyerman, S. D. Linking metabolism to membrane signaling: the GABA malate connection. Trends in Plant Science. 21, 295–301 (2016).
Hugues, R. et al. The Arabidopsis pop2-1 mutant reveals the involvement of GABA transaminase in salt stress tolerance. BMC Plant Biology. 10, 1–20 (2006).
Shaw, A. K. Β-aminobutyric acid mediated drought stress alleviation in maize (Zea mays, L.). Environmental Science and Pollution Research. 23, 2437–2453 (2016).
Li, Y., Fan, Y., Ma, Y., Zhang, Z. & Yue, H. Effects of exogenous γ-aminobutyric acid (gaba) on photosynthesis and antioxidant system in pepper (capsicum annuum, l.) seedlings under low light stress. Journal of Plant Growth Regulation. 36, 1–14 (2017).
Li, Z., Yu, J., Peng, Y. & Huang, B. Metabolic pathways regulated by γ-aminobutyric acid (GABA) contributing to heat tolerance in creeping bentgrass (Agrostis stolonifera). Scientific Reports. 6, 30338 (2016).
Liu, J. P. Xiao, F. L. Zhu, H. F. Zhu, Y. & Hou, X. L. Effects of γ-aminobutyric acid on the growth and photosynthesis of pakchoi under waterlogging stress. Acta Agriculture Shanghai. (2016).
Beuve, N. et al. Putative role of aminobutyric acid (GABA) as a long-distance signal in up-regulation of nitrate uptake in Brassica napus L. Plant Cell and Environment. 27, 1035–1046 (2004).
Lancien, M. & Roberts, M. R. Regulation of Arabidopsis thaliana gene expression by -aminobutyric acid. Plant Cell and Environment. 29, 1430–1436 (2006).
Shi, S. Q. et al. Effects of exogenous GABA on gene expression of Caragana intermedia roots under NaCl stress: regulatory roles for H2O2 and ethylene production. Plant Cell and Environment. 33, 149–162 (2010).
Zheng, S. W. Qi, X. U. & Zou, H. W. Effect of GABA on yield of wheat under waterlogging condition. Hubei Agricultural Sciences. (2016).
Nayyar, H., Kaur, R., Kaur, S. & Singh, R. γ-aminobutyric acid (GABA) imparts partial protection from heat stress injury to rice seedlings by improving leaf turgor and up regulating osmoprotectants and antioxidants. Journal of Plant Growth Regulation. 33, 408–419 (2014).
Xia, Q. P., Gao, H. B. & Li, J. R. Effects of gamma-aminobutyric acid on the photosynthesis and chlorophyll fluorescence parameters of muskmelon seedlings under hypoxia stress. The journal of applied ecology. 22, 4–999 (2011).
Wang, Y., Gu, W., Meng, Y. & Xie, T. γ-aminobutyric acid imparts partial protection from salt stress injury to maize seedlings by improving photosynthesis and up regulating osmoprotectants and antioxidants. Scientific Reports 7, 43609 (2017).
Kinnersley, A. M. & Turano, F. J. Gamma aminobutyric (GABA) and plant responses to stress. Critical Reviews in Plant Sciences. 19, 479–509 (2000).
Perez, M., Invers, O., Ruiz, J. M., Frederiksen, M. S. & Holmer, M. Physiological responses of the seagrass Posidonia oceanica to elevated organic matter content in sediments: an experimental assessment. J Exp Mar Biol Ecol. 344, 149–16 (2007).
Xing, S. G., Jun, Y. B., Hau, Z. W. & Liang, L. Y. Higher accumulation of gamma-aminobutyric acid induced by salt stress through stimulating the activity of diamine oxidases in Glycine max (L.) Merr. Roots. Plant Physiol. Biochem. 45, 560–566 (2007).
Malekzadeh, P., Khara, J. & Heydari, R. Alleviating effects of exogenous gamma-aminobutiric acid on tomato seedling under chilling stress. Physiol. Mol. Biol. Plants 20, 133–137 (2014).
Wang, X. D., Xie, B. T., Jian, L. & Duan, L. S. Effects of exogenous GABA on waterlogged tolerance in wheat seedlings. Acta Agriculturae Boreali Sinica. 25, 155–160 (2010).
Kinnersley, A. M. & Fang, L. Receptor modifiers indicate that 4-aminobutyric acid (gaba) is a potential modulator of ion transport in plants. Plant Growth Regulation. 32(1), 65–76 (2000).
Bouché, N. & Fromm, H. GABA in plants: just a metabolite? Trends in Plant Science. 9, 110–115 (2004).
Ren, B., Zhang, J., Dong, S., Liu, P. & Zhao, B. 2017. Regulations of 6-benzyladenine (6-ba) on leaf ultrastructure and photosynthetic characteristics of waterlogged summer maize. Journal of Plant Growth Regulation. 36, 1–12 (2010).
Li, X. Y., Liu, Z. Y., Li & An, T. X. Impact test study of the flood disasters on summer corn’s characters and yield. Scientia Meteorologica Sinica. 31, 79–82 (2011).
Yin, Y. Q. & Jin, X. Studies on changes of physiology, biochemistry and γ-aminobutyric acid content during germination of maize (Zea mays l.) under hypoxia stress. Food Science (2014).
Shang, H. T., Cao, S. F., Yang, Z. F., Cai, Y. T. & Zheng, Y. H. Effect of exogenous γ-aminobutyric acid treatment on proline accumulation and chilling injury in peach fruit after long-term cold storage. J Agric Food Chem. 59, 1264–1268 (2011).
Yang, A. P., Cao, S. F., Yang, Z. F., Cai, Y. T. & Zheng, Y. H. γ-Aminobutyric acid treatment reduces chilling injury and activates the defence response of peach fruit. Food Chem. 129, 1619–1622 509 (2011).
Gill, S. S. & Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiology Biochemistry. 48, 909–930 (2010).
Apel, K. & Hirt, H. Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol. 55, 373–399 (2004).
Perveen, S., Anis, M. & Aref, I. M. Lipid peroxidation, H2O2 content, and antioxidants during acclimatization of Abrus precatorius to ex vitro conditions. Biol Plant. 57, 417–424 (2013).
Candan, N. & Tarhan, L. Tolerance or sensitivity responses of mentha pulegium to osmotic and waterlogging stress in terms of antioxidant defense systems and membrane lipid peroxidation. Environmental and Experimental Botany. 75, 83–88 (2012).
Wang, C. Y. et al. Polyamine biosynthesis and degradation are modulated by exogenous gamma-aminobutyric acid in root-zone hypoxia-stressed melon roots. Plant Physiol Biochem. 82, 17–26 (2014).
Zhou, X., Wu, X., Li, Y. & Zhang, S. Accumulations and correlations of ABA and GABA in maize seedling under salt stress. Chinese Journal of Applied and Environmental Biology. 11, 412–415 (2005).
Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Science. 7, 405–410 (2002).
Cadenas, E. & Davies, J. A. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic Biol Med. 29, 3222–230 (2002).
Schrader, M. & Fahimi, H. D. Peroxisomes and oxidative stress. BBA - Molecular Cell Research. 1763, 1755–1766 (2006).
Hussain, S., Khan, F., Cao, W., Wu, L. & Geng, M. Seed priming alters the production and detoxification of reactive oxygen intermediates in rice seedlings grown under sub-optimal temperature and nutrient supply. Frontiers in Plant Science. 7, 116 (2016).
Hongbo, S., Zongsuo, L. & Mingan, S. Changes of anti-oxidative enzymes and MDA content under soil water deficits among 10 wheat (Triticum aestivum L.) genotypes at maturation stage. Colloids and Surfaces B Biointerfaces. 45, 7–13 (2005).
Tang, B., Xu, S. Z., Zou, X. L., Zheng, Y. L. & Qiu, F. Z. Changes of antioxidative enzymes and lipid peroxidation in leaves and roots of waterlogging-tolerant and waterlogging-sensitive maize genotypes at seedling stage. Agriculture Science China. 9, 651–661 (2010).
Bansal, R. & Srivastava, J. P. Antioxidative responses to short term waterlogging stress in pigeon pea. Indian Journal of Plant Physiology. 20, 182–185 (2015).
Gill., M., B. Zeng, F. & Shabala, L. The ability to regulate voltage-gated K+-permeable channels in the mature root epidermis is essential for waterlogging tolerance in barley. Journal of Experimental Botany 69, 667–680 (2017).
Miller, G., Shulaev, V. & Mittler, R. Reactive oxygen signaling and abiotic stress. Physiologia Plantarum. 133, 481–489 (2010).
Anjum, S. A., Tanveer. Hussain, S., Bao, M., Wang, L. & Khan, I. Cadmium toxicity in Maize (Zea mays L.) consequences on antioxidative systems, reactive oxygen species and cadmium accumulation. Environmental Science and Pollution Research. 22, 17022–17030 (2015).
Mittler, R. et al. Reactive oxygen gene network of plants. Trends in Plant Science. 9, 490–498 (2004).
Ashraf, U. et al. Lead toxicity in rice effects mechanisms and mitigation strategies - a mini review. Environ. Sci. Pollut. Res. 22, 18318–18332 (2015).
Flexas, J., Espejo, A., Galmés, J. & Medrano, H. Mesophyll conductance toco2: current knowledge and future prospects. Plant Cell & Environment. 31, 602–621 (2010).
Mclean, M. D. Metabolism and functions of gamma-aminobutyric acid. Trends in Plant Science. 11, 446–452 (1999).
Xu, S., Li, J., Zhang, X., Wei, H. & Cui, L. Effects of heat acclimation pretreatment on changes of membrane lipid peroxidation, antioxidant metabolites, and ultrastructure of chloroplasts in two cool-season turfgrass species under heat stress. Environmental and Experimental Botany. 56, 274–285 (2006).
Asada, K. E., Mano, T. J. & Miyake, C. 3–molecular mechanism for relaxation of and protection from light stress. Stress Responses of Photosynthetic Organisms. 106, 37–52 (1998).
Qi, W. Z. et al. Morphological and physiological characteristics of corn (Zea mays L.) roots from cultivars with different yield potentials. European Journal of agronomy. 38, 54–63 (2012).
Guijin, Z. & Bown, A. W. The rapid determination of g-aminobutyric acid. Phytochemistry. 44, 1007–1009 (1997).
Agrawal, S. B. & Rathore, D. Changes in oxidative stress defense in wheat (Triticum aestivum L.) and mung bean (Vigna radiata L.) cultivars grown with and without mineral nutrients and irradiated by supplemental ultraviolet. Environmental and Experimental Botany. 59, 21–33 (2007).
Acknowledgements
The authors would like to thank Professor Guosheng Xie, Huazhong Agricultural University, for checking and polishing the manuscript. This work was supported by the National Key R&D Program of China (2016YFD0300308), the Fundamental Research Funds for the Central Universities of China (2662015PY067).
Author information
Authors and Affiliations
Contributions
Akram Salah performed the experiment, analyzed data and wrote the paper. Ming Zhan designed research and wrote the paper. Cougui Cao assisted research design and supervise. Zhihui Liu, Lin Ling, Yuling Han and Miao Ye helped in conducting experiment and analyzing the data. Yang Jiang and Ping Li helped in the revision and improved the quality of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salah, A., Zhan, M., Cao, C. et al. γ-Aminobutyric Acid Promotes Chloroplast Ultrastructure, Antioxidant Capacity, and Growth of Waterlogged Maize Seedlings. Sci Rep 9, 484 (2019). https://doi.org/10.1038/s41598-018-36334-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-36334-y
This article is cited by
-
Transcriptomic analysis reveals the regulatory mechanisms of messenger RNA (mRNA) and long non-coding RNA (lncRNA) in response to waterlogging stress in rye (Secale cereale L.)
BMC Plant Biology (2024)
-
The contribution of Ca and Mg to the accumulation of amino acids in maize: from the response of physiological and biochemical processes
BMC Plant Biology (2024)
-
Predicting and optimizing reactive oxygen species metabolism in Punica granatum L. through machine learning: role of exogenous GABA on antioxidant enzyme activity under drought and salinity stress
BMC Plant Biology (2024)
-
Selenium treatment alters the accumulation of osmolytes in arsenic-stressed rice (Oryza sativa L.)
Environmental Science and Pollution Research (2024)
-
Gamma-aminobutyric acid interactions with phytohormones and its role in modulating abiotic and biotic stress in plants
Stress Biology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.