Abstract
Small non-coding RNAs (sncRNA) are regulators of cell functions and circulating sncRNAs from the majority of RNA classes are potential non-invasive biomarkers. Understanding how common traits influence ncRNA expression is essential for assessing their biomarker potential. In this study, we identify associations between sncRNA expression and common traits (sex, age, self-reported smoking, body mass, self-reported physical activity). We used RNAseq data from 526 serum samples from the Janus Serum Bank and traits from health examination surveys. Ageing showed the strongest association with sncRNA expression, both in terms of statistical significance and number of RNAs, regardless of RNA class. piRNAs were abundant in the serum samples and they were associated to sex. Interestingly, smoking cessation generally restored RNA expression to non-smoking levels, although for some sncRNAs smoking-related expression levels persisted. Pathway analysis suggests that smoking-related sncRNAs target the cholinergic synapses and may therefore potentially play a role in smoking addiction. Our results show that common traits influence circulating sncRNA expression. It is clear that sncRNA biomarker analyses should be adjusted for age and sex. In addition, for specific sncRNAs, analyses should also be adjusted for body mass, smoking, physical activity and technical factors.
Similar content being viewed by others
Introduction
Approximately two thirds of the mammalian genome is transcribed to produce different RNA classes, the majority of which are non-coding RNAs (ncRNA)1,2. The major ncRNA classes are microRNA (miRNA), transfer RNA (tRNA), piRNAs, long non-coding RNA (lncRNA), small nucleolar (snoRNA), small nuclear RNA (snRNA) and miscellaneous RNA (miscRNAs). In addition, fragments and isoforms of RNAs may have important biological roles independent of the canonical, full-length RNAs from which they derive3,4,5. Circulating snc RNAs (sncRNA) are secreted from cells, either bound to RNA binding proteins6, high-density lipoproteins7, within extracellular vesicles or released during cell death8. sncRNAs are protected from degradation, and miRNAs, the most studied sncRNA class, have been identified in all body fluids9,10,11. Aberrant expression of small and long regulatory non-coding RNAs are related to many diseases12,13.
Circulating ncRNAs have considerable potential as minimally invasive cancer biomarkers14,15,16,17,18,19, however, few if any have reached their translational potential. To be reliably used as biomarkers, variation and traits that influence sncRNA expression levels need to be identified in non-diseased individuals. Common traits may include age, sex, smoking, body mass and physical activity. Technical factors, such as sample processing and storage, may also influence RNA levels20,21. Almost all studies to date have focused on miRNAs, and have inadequate sample sizes to assess normal variation and identify the effects of traits on expression.
sncRNAs may be encoded on the sex chromosomes22 and sex-specific miRNA expression patterns have been shown in tissues23. Several steroid sex hormones, such as estradiol, progesterone and testosterone have been found to directly or indirectly regulate miRNA expression24,25,26 or Argonaute, Drosha and Dicer, the major enzymes of miRNA biogenesis27. Some isomiRs have also been shown to be sex-specific28.
Ageing is more strongly associated with circulating miRNA expression than sex. The miRNAs significantly influenced by age included hsa-miR-1284, hsa-miR-93–3p, hsa-miR-1262, hsa-miR-34a-5p, and hsa-miR-145–5p29. This is in agreement with the first observations of altered circulating miRNA levels during ageing showing an increase in miR-34a in the plasma of old mice30. 127 of 150 miRNAs analysed were shown to be affected by age in a study on whole-blood from 5221 individuals. A miRNA age prediction model was developed using this large dataset and the miRNA predicted age correlated with chronological age with an r = 0.61 adjusted for cell type composition31. Transforming growth factor beta signalling has been suggested as one of the main pathways regulated by the differentially expressed circulating miRNAs32. However, cellular senescence, ageing and age-related diseases, have been associated with alterations in miRNA expression that could have multiple physiological effects. Whether the changes have an etiological origin or are a consequence of deleterious age-induced dysfunctions is still unknown33.
A large study (N = 226) showed that smoking alters circulating miRNA expression. There was no significant finding when comparing former to never smokers34. A study of small airway epithelium from 10 smokers, identified differences in miRNA expression after smoking cessation which persisted in 8 out of the 34 (FDR <0.05) smoking-related miRNAs, with the Wnt/β-catenin signalling pathway being the most significant pathway35. A smaller study of 12 never-smokers and 28 smokers, all males, identified 35 differentially expressed miRNAs, and target enrichment analyses identified the immune system and hormone regulation as possible pathways differing between the groups36.
Studies of differential miRNA expression related to body mass have mostly focused on adipose tissue, and a small number of miRNAs were found to be differentially expressed in individuals with obesity and type 2 diabetes mellitus (T2DM)37. These miRNAs influence the expression and secretion of inflammatory proteins. Ameling et al. found 19 of 179 miRNAs to be associated with body mass in 372 population-based samples, 12 miRNAs were age-associated and 7 were sex-associated38.
Physical activity-related miRNAs have mainly been found in intervention studies, identifying 4 to 23 differentially expressed miRNAs39,40,41. Oppose to changes in circulating miRNAs in acute exercise, the changes of circulating miRNAs in chronic exercise remain unclear42. A positive linear correlation between training-induced changes in circulating miR-20a levels and changes in VO2max has been shown, suggesting potential biomarkers of cardiorespiratory fitness trainability43.
With few exceptions, miRNAs are the only ncRNA class that have been studied in relationship to sex, age, body mass and physical activity. In addition, small sample sizes in most of these studies hampers discovery, and the widespread use of disease-related samples may introduce bias. tRNA, piRNAs, lncRNA, snoRNA, snRNA, miscRNAs and their isoforms may be potential biomarkers as long as they are stable, quantifiable, and population variation due to common traits is known.
In this study, we explore the relationship between sex, age, smoking, body mass, physical activity, technical factors and circulating ncRNA expression levels. We use RNAseq to high depth (on average 18 mill. sequences) from a large serum sample set (N = 526) of cancer-free donors from the Janus Serum Bank (JSB)44. This data, combined with high-quality survey information45, provides a unique opportunity to identify and compare trait associations that might influence sncRNA biomarker potential.
Results
Significant trait associations
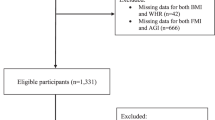
We produced RNAseq expression profiles for sncRNAs 17 to 47 nucleotides long in serum samples from cancer-free JSB donors and analysed associations with age, sex, body mass, smoking, physical activity or technical factors (blood donor group (BDg), see Methods section) (Fig. 1). We analysed 27251 sncRNAs, including 15217 mRNA fragments. 2362 trait associations (categorical and unadjusted) were significant using an adjusted p-value < 0.05 cut-off (Supplementary File 1: Table S1). When applying a stricter cut-off (adjusted p-value < 0.001), 651 of the sncRNAs showed trait associations (Table 1).
Associations between traits and small non-coding RNAs (sncRNA) were investigated in samples from in total 526 Janus Serum Bank donors, including 156 Red Cross Blood Donors and 370 Health Examination Blood Donors. (A) The total number of samples included in each trait analyses after excluding samples with missing data and low sncRNA yielding samples. (B) Number of samples in each category for age, blood donor group (BDg), body mass, self-reported physical activity (phys act), sex and self-reported smoking. (C) Number of sncRNA counts on log scale after less stringent, medium and high stringent filtering.
Age had the highest number of trait associations with 1340 sncRNAs with adjusted p-value < 0.05 and 554 sncRNAs associated with adjusted p-value < 0.001 (Supplementary File 1:Tables S2 and S3). Only three sncRNAs were significantly associated with blood donor group (adjusted p-value < 0.001; Table 1). Using age as a continuous variable, we identify 1311 sncRNAs associations compared to 554 sncRNAs with age as a categorical variable (p-value < 0.001). There is a correlation between age at donation and blood donor group in this cohort (Supplementary File 2: Figure S1). However, the age-associations are similar with and without adjusting for blood donor group (Supplementary File 2: Figure S4). Body mass as a continuous variable (body mass index, BMI) did not reveal additional associated sncRNAs.
We adjusted for age (categorical) in the analyses of sex, body mass, smoking and physical activity. Age-adjustment increased the significant associations from 33 to 439 for sex, 44 to 411 for body mass, 5 to 208 for physical activity and 11 to 182 for smoking (adjusted p-value < 0.001; Table 1 and Supplementary File 1:Table S4). In total, 1240 sncRNAs were associated with sex, body mass, physical activity or smoking (categorical) after age adjustment (p-values < 0.001; Supplementary File 1:Table S5). The top 5 associated miRNAs, piRNAs, lncRNA and tRNAs are shown in Table 2.
Hierarchical clustering of adjusted p-values for all associations were visualized using heatmaps (Fig. 2). The age-associations were more numerous and with higher −log p-values than other traits and are an outgroup in the vertical dendrogram. Associations with sex, body mass, smoking and physical activity with age as a covariate, showed more and stronger associations. Notably, piRNAs were associated with sex, after adjusting for age (Supplementary File 2: Figure S2).
Heatmaps of the hierarchical clustering of -log10 p-values adjusted for multiple testing from the associations between sncRNAs from the classes miRNAs, isomiRs, tRNAs, tRNA fragments, piRNAs, lncRNAs, miscRNAs, snRNAs and mRNA fragments and the attributes blood donor group (BDg), sex, body mass, smoking (current vs never smokers) and physical activity (low vs high activity). sncRNAs are visualized if any of the associations produced p-values < 0.01. Colors are yellow to orange for -log10 p-value 0 to 5 and red for -log10 p-values > 6.
Expression differences
The majority of sncRNAs showed log2fold transformed differences between 1 and −1 (Fig. 3). However, numerous associations with age, and to a lesser extent blood donor groups, showed differences greater than +/−1. Specifically these larger differences were seen for: miRNAs and blood donor group and age; isomiRs and smoking, age and blood donor group; snRNAs and age; mRNA fragments and age. The majority of miRNAs were upregulated with age, while the majority of mRNA fragments were downregulated with age. snRNAs and lncRNAs were also downregulated with age.
Volcano plots showing differential expression in log2fold change on the x-axis and adjusted p-values from the associations in -log10 on the y-axis for miRNAs, isomiRs, tRNAs, tRNA fragments, piRNAs, lncRNAs, miscRNAs, snRNAs and fragments mapping mRNA and the traits; blood donor group (BDg), sex, body mass smoking (current vs never smokers) and physical activity (low vs high activity).
Adjusting for age in the association analyses with sex, body mass, smoking and physical activity, showed larger log2fold differences compared to unadjusted analyses (Supplementary File 2: Figure S3). The changes were striking when compared to the unadjusted analyses (Fig. 4), specifically for sncRNA associations with sex. miRNAs and piRNAs were upregulated and lncRNAs and mRNA fragments downregulated in men. Strong smoking differences between current and never smokers on sncRNA expression were also seen for tRNAs, isomiRs, tRNA fragments, snRNAs, lncRNAs and one piRNA. One sex and two body mass associations with mRNAs fragments have log2fold differences larger than two and −log10 adjusted p-values > 40.
Volcano plots showing differential expression in log2fold change on the x-axis and adjusted p-values from the associations in -log10 on the y-axis for all sncRNAs associated to sex, smoking, body mass and physical activity. Associations without age as co-variable are shown in red and associations adjusted for age are shown in blue.
Co-expression module analyses
Module analyses showed that age and blood donor group are more strongly correlated with co-expression modules than any other trait, followed by physical activity (p-value < 0.01; Supplementary File 2: Figure S5). A set of lncRNAs are strongly correlated with sex (Pearson r = 0.7). A module of 16 tRNA fragments were associated with sex, body mass and physical activity. Smoking is associated with fewer modules than the other traits.
KEGG Pathway analyses
We performed KEGG pathway analyses for mRNA fragments and miRNA targets (see Methods). Pathways were enriched for age-, sex- (age adjusted) and smoking-associated (age adjusted) mRNA fragments and miRNA targets (p-value < 0.05; Table 3). We did not detect any significant pathway enrichments for body mass and physical activity. Four out of the top five age-related pathways are involved in carcinogenesis, while all the top five smoking-related pathways have been associated with smoking.
Smoking and sncRNA associations
To identify sncRNAs associated with smoking cessation, we assessed differential expression in never vs current smokers, relative to never vs former smokers. We identified smoking-related differential expression in isomiRs, piRNA, lncRNA, tRNA and mRNA fragments which persist after smoking cessation. A single piRNA and two tRNAs show persistent expression differences after smoking cessation, while two smoking-associated miRNAs revert to never-smoking levels (Fig. 5).
Differential sncRNA expression in never vs current smokers relative to never vs former smokers suggesting smoking related sncRNA expression that persist after smoking cessation (upper right corner) and sncRNA expression that revert to never smoking levels (lower right corner). The -log10 p-value are shown for smoking associations in current smokers vs never smokers (x-axis) and the former smoker vs never smokers (y-axis). -log10 p-values > 2 in both analyses are marked in red, signifying associations both in current and former smokers. -log10 p-values > 2 in current, but not in former smokers are marked in blue, signifying associations in current smokers and not in former smokers. Associations with expression differences more than +/−0.5 in both analyses are marked with a cross, all other relationships are marked with a dot. The analyses were done for miRNAs, isomiRs, tRNAs, tRNA fragments, piRNAs, lncRNAs, miscRNAs, snRNAs and mRNA fragments.
The top three smoking-associated miRNAs show a slight increase in expression levels between never, former and current smokers (Supplementary File 2: Figure S6A). This effect became more pronounced in heavy smokers, specifically for individuals smoking more than 20 cigarettes per day for miR-3656 and miR-7704 (Supplementary File 2: Figure S6B).
Discussion
Expression levels of circulating sncRNAs vary between healthy individuals46,47. However, not much is known about which traits influence this variation. Relationships between miRNA expression and age, sex, body mass, smoking and physical activity have been reported29,34,35,38,41,42, although most studies have small sample sizes and therefore will be unlikely to detect subtle changes in expression. Furthermore, very little has been reported about these traits in relationship to expression levels of other sncRNA classes and no other study to date that we are aware of has compared association between common traits within the same dataset.
In this paper sncRNA association analyses show that ageing is strongly correlated with all sncRNA classes. The age-association was confirmed by analysing sncRNA co-expression modules. Our study is the largest to date showing a strong age effect for all sncRNA classes. The age effect has been consistently reported in previous studies only for miRNAs. However, the age-associated miRNAs reported in these studies differ presumably due to differences in biological materials, sample processing and sample size.
Age-associated miRNA expression has previously been shown in model organisms30, tissues48 and blood29. In agreement with our results, ageing was reported to be more strongly associated with miRNA expression than sex29. miRNA-320b was found to be age-related in both our study and in a large study on whole blood31. We found that the age-associated pathways are mostly signaling pathways such as Ras, PI3K-Akt, MAPK and AMPK. This may explain the role of aging in oncogenesis. Ageing is also known to affect dopamine receptors which can explain enrichment of dopaminergic synapse pathway for aging.
The relationship between age and circulating sncRNA expression implies that all sncRNA biomarker studies should take age at sampling into consideration when analysing and interpreting results. Sample groups should be age-matched, stratified by age, or age-adjusted. sncRNAs mediate a number of cellular functions, and age-associated expression changes may implicate these in ageing processes. Changes in blood cell counts with age49,50, may explain some of the differential sncRNA expression. The present study provides a valuable data set for studying mechanisms of ageing and age-related diseases such as cancer.
Our data also showed significant associations between sex, body mass, smoking and physical activity and the expression levels of 1240 sncRNAs after adjusting for age. Age is an important effect modifier for these associations since the differences with and without age-adjustment increase significant associations more than 10-fold.
We observed sex-related expression for all sncRNA classes. miRNA expression correlated with sex has previously been shown23,29 and in some cases directly or indirectly linked to hormonal regulation24,25,26. piRNAs were initially thought to be specific to germ cells51,52, however circulating piRNAs have recently been identified at significant levels11,53. Our dataset identified a large number of RNAs mapping to piRNA databases. JSB serum samples were stored at −25 °C for up to 40 years47, indicating that piRNAs are stable. The cellular origin of the piRNAs is unknown. We observe a difference in expression between males and females for a large fraction of piRNAs, indicating that some of the circulating piRNAs might originate from germ cells. Our data also showed sex-specific differences in lncRNAs and mRNA fragments. For example, one lncRNA co-expression module is highly correlated with sex and includes Y chromosome-derived lncRNA fragments. Based on our findings, matching or adjusting for sex in differential expression studies may be crucial.
Our data show that smoking alters expression levels for all classes of sncRNAs, which had previously only been shown for miRNAs34,35. Wang et al.35 indicated that only a portion of the smoking-related miRNAs revert to a never smoker expression level after smoking cessation. In contrast, our analyses indicate that miRNA expression in former smokers is similar to never smokers. Futhermore, the expression levels of isomiRs, lncRNAs and mRNA fragments, as well as two tRNA and one piRNA, are significantly different in former smokers compared to never smokers, indicating smoking-related expression persists after smoking cessation for these sncRNAs. Similar results have been shown for DNA methylation54,55. It is noteworthy that the top three smoking-related miRNAs (hsa-miR-7704, hsa-miR-3655 and hsa-miR-203-3p) showed a clear relationship between smoking-dose and expression levels. However, the status as canonical miRNAs are questioned for hsa-miR-7704, hsa-miR-3655. Interestingly, the top five age-adjusted smoking related pathways have previously been related to smoking. For example, the cholinergic synapse pathway is associated with nicotine addiction56, suggesting that sncRNAs play a role in smoking addiction. The relaxin signaling pathway is also disrupted by smoking57. Our results show that smoking-related pathway RNAs (e.g. mRNAs and miRNA targets) can be identified in serum.
Body size was associated with 411 sncRNAs of which 63% are mRNA fragments. No miRNAs were statistically significant (P < 0.001). 208 sncRNAs were associated with physical activity, of which 70% mapped to mRNA fragments. Notably, the overlap between body mass and physical activity related sncRNAs was observed for three tRNAs and 13 tRNA fragments. No comparable study, to our knowledge, is available showing physical activity and body mass associations with circulating non-miRNA sncRNAs. Our results indicate that differential expression studies in obesity and exercise should consider studying other sncRNAs in addition to miRNAs.
The serum samples were stored long-term at −25 °C. Under these conditions all unstable RNAs have been degraded. We have shown that the total amount of miRNA was affected by the processing of the serum and to a lesser extent by storage time21. Also, the number of other sncRNAs decrease with storage47. The differential expression analyses between the blood donor groups shown here shed further light on which sncRNAs are affected by storage and processing. Since blood donor groups and age are correlated, some of the blood donor group associations identified might be due to age differences. SncRNA studies using JSB data should take blood donor group into consideration.
The primary functions of most RNA classes are known. For example, snRNAs are involved in mRNA splicing, tRNAs decode mRNAs into peptides, snoRNAs carry chemical modifications to mRNA fragments, miRNA regulate post-transcriptional gene expression, piRNAs target and repress the expression of transposable elements and lncRNAs provide epigenetic control of gene expression and promoter-specific gene regulation58,59,60,61. However, secondary functions are largely unknown and therefore pathways and network approaches for functional analyses are not yet feasible. Another challenge in the interpretation of the results are insufficient accuracy and completeness of the annotation databases. Recognized databases such as miRBase62, ENCODE1 and piRbase63, may include degradation products, misclassifications and mapping errors. Curated databases such as miRgenedb64 may improve the interpretability. piRNAs are particularly difficult and there is a highly probable that the available piRNA databases contain RNAs unrelated to the piRNAs produced by germ cells. However, the discovery of sncRNA biomarkers are less affected by poor annotation.
Circulating sncRNAs originate from multiple cell types, and cell type compositional differences might introduce variation or confounding. Still, it is not known if all cell types display age-related miRNA expression65, and only small expression differences of cell type composition were seen in one of the largest studies to date31. In addition, traits such as obesity, low activity and smoking will likely affect RNA expression less than diseases like cancer, therefore, large samples sizes are needed to discover signal over noise.
The main strength of our study is the large sample size. 526 donors included in the study provided sufficient statistical power to detect small differences in expression. Linkage to a complete cancer registry ensures that all donors were free from cancer at least 10 years after sample donation, removing the effects of potential cancer progression on sncRNA expression. Harmonized and quality-assured smoking, body mass data improves the accuracy of the measured traits45. High sequence read-depth (on average 18 mill reads per sample) serum RNAseq data targeting RNAs between 17 and 47 nucleotides in length enables comprehensive assessment of all main RNA classes.
The primary limitation of the study is the long-term storage of the samples and the effect it might have on RNA quality. Although the advantage of long-term storage is long follow-up time for the disease outcome. The expression differences from storage and sample handling may affect the associations, however, the effects found in previous studies were minor21,47. Common with all sncRNA studies, problems with annotation and the lack of functional information makes interpreting the findings challenging. Trusted annotations are essential to correctly identify transcripts1, yet well-known annotation databases are not error free64,66. For example, piRNA annotations contain fragments corresponding to other sncRNAs67. Although the data has unprecedented sample size, the moderate-high physical activity group and individuals less than 40 years old are represented by fewer than 100 individuals. Associations were calculated from variable samples sizes, due to missing data. This might to some extent reduce comparability between trait associations. Due to the historic nature of the questionnaires data, the validity of the physical activity variable has not been fully validated. However, the validity of self-reported leisure time physical activity, and its relationship to serum cholesterol, blood pressure and body mass index has been investigated in data from the health examination surveys68. Aires et al.68 find that that the slopes relating year of birth and serum cholesterol and BMI are parallel for self-reported physical activity, thus they state that the validity of the physical activity variable is confirmed.
In conclusion, our study showed that sncRNA expression levels in serum are strongly age-dependent, and therefore age should be considered in studies of circulating sncRNA expression. sncRNA expression also differed between sexes, and this difference may reflect key biological differences, such as germ cell specificity of piRNAs. Some of the expression signatures are also influenced by body mass, smoking, physical activity and sample processing. The relationships between traits and sncRNA expression levels are of key importance in all sncRNA biomarker research and should be accounted for in the study design and analyses of data.
Methods
Study subjects
The Janus Serum Bank (JSB) is a population-based cancer research biobank containing prediagnostic biospecimens from 318 628 Norwegians44. We identified 550 JSB donors that were cancer free at least 10 years after sample donation using data from the Cancer Registry of Norway. Information on age at donation, processing of samples according to blood donor groups (BDg) and sex were available from all donors, body mass, smoking and physical activity were available for most of the health examination donors (Fig. 1A).
Inclusion criteria for each analysis were a high-quality sncRNA profile (see filtering criteria in the bioinformatics section) and available trait information. 156 samples included were from red cross blood donors (RCBD) and 370 were from donors participating in health examinations (HEBD), in total 526. JSB has prospectively collected serum samples between 1972 and 2004. The collection procedure and serum processing have varied throughout the collection period. The samples were grouped according to sample collection period and processing (Grp1:HEBD from 1972–1978, iodoactetate added, Grp2: HEBD from 1979–1986, Grp3: HEBD from 1987–2004 collected in separating gel tubes, Grp4A: RCBD from 1973–1979, Grp4B: RCBD from 1973–1979, lyophilized, Grp5: RCBD from 1980–1990 and Grp6: RCBD from 1997–2004)21 (Fig. 1B). The number of samples in each blood donor (BD) group from 1 to 6 were 33, 143, 194, 62, 66 and 28, respectively. 171 women and 288 men (Fig. 1B) with a mean age of 50 (standard deviation 11.2) at donation were included. Age at donation was categorized into less than 40, between 40 and 60 and above 60, and used as a continuous variable (Fig. 1B and Supplementary File 2: Figure S1). Data from the health examination studies were available for the HEBD donors, including information on self-reported smoking habits, body mass and self-reported physical activity (Fig. 1B)45. The donors with smoking information were categorized into current (N = 107), former (N = 85) and never (N = 111) smokers. The number of cigarettes per day in current smokers were categorized into 1–9 (N = 36), 10–14 (N = 33), 15–19 (N = 22), 20–24 (N = 19) or > = 25 (N = 5). There were 152 normal weight, 4 underweight, 156 overweight and 39 obese donors using WHO standards. Analyses were done contrasting normal weight vs overweight and obese combined. Questionnaire data on physical activity was reported in four activity groups: sedentary (reading, watching tv, other sedentary activities, N = 37), moderate active (walking, bicycling, ≤4 hours per week, N = 177) or active (light sports, heavy gardening ≥ 4 hours per week, N = 70, and/or hard exercise, competitive sports regularly, N = 9). The sedentary and moderate (N = 214) were compared to active and hard exercise (N = 79). (Fig. 1B).
RNA isolation and sequencing protocols
RNA was extracted from 2 × 200 µl serum using phenol-chloroform separation and the miRNeasy Serum/Plasma kit (Cat. no 1071073, Qiagen) on a QIAcube (Qiagen). Glycogen (Cat. no AM9510, Invitrogen) was used as carrier during the RNA extraction step. The eluate was concentrated using Ampure beads XP (Agencourt). Small RNAseq was performed using NEBNext® Small RNA Library Prep Set for Illumina (Cat. No E7300, New England Biolabs Inc.) with a cut size on the pippin prep (Cat. No CSD3010, Sage Science) covering RNA molecules from 17 to 47 nucleotides. Sequencing libraries were indexed and 12 samples were sequenced per lane on a HiSeq2500 (Illumina) to an average depth of 18 million reads per sample.
Bioinformatics analyses
The RNAseq reads were initially trimmed for adapters using AdapterRemoval (v2.1.7)69. We then mapped the collapsed reads (generated by FASTX v0.14) to the human genome (hg38) using Bowtie2 (10 alignments per read were allowed). All multi-mapped reads with equivalent mapping score were counted. We compiled a comprehensive annotation set from miRBase (v21)62 for miRNAs, pirBAse (v1.0) for piRNAs63, GENCODE (v26)1 for other RNAs and tRNAs. We used SeqBuster (v3.1)70 to get isomiR and miRNA profiles. To count the mapped reads, HTSeq (v0.7.2)71 was used. The candidate tRNA fragments (tRFs) were selected from the reads mapped to tRNA annotations. For biomarker purposes, we excluded RNAs with fewer than 10 reads in more than 20% of the samples (Fig. 1C medium stringent filtering). To show how filtering influenced the number of RNAs we produced tables with stringent and less stringent filtering cut-offs. Stringent filtering excluded RNAs with less than 10 reads in more than 50% of the samples. Less stringent filtering excluded RNAs with less than 1 read in more than 20% of samples (Fig. 1C).
Statistics
Differential gene expression analyses based on the negative binomial distribution and Wald significance tests were performed for each trait using the R package DESeq2 version 1.14.172. All traits were categorical when different traits were compared. Analyses with age and BMI as continuous variables are also presented. The analyses were performed with and without adjustment for age at donation. P-values after adjusting for multiple testing, using DESeq2 default adjustments, were reported72. Heatmaps of trait-associated RNAs were created using the heatmap.2 function in the gplots package. sncRNAs where any of the traits had adjusted p-value < 0.001 for analyses not adjusted for age at donation and p-values < 0.01 for analyses adjusted for age at donation are shown. We performed variance stabilizing transformation (VST) from the fitted dispersion-mean relations and then transformed the normalized count data using the function varianceStabilizingTransformation. Variance stabilized normalized counts were extracted and in-depth analyses of the top 3 strongest associations for smoking, body mass and physical activity were performed. For this, current, former, never smokers and number of cigarettes per day were investigated. Body mass was categorized according to WHO standardized cutoffs and physical activity were analysed according to the levels low and high.
We performed KEGG pathway73 analysis on differentially expressed mRNA fragments and miRNA targets. The analysis was performed using R function kegga from the limma package. The miRNA targets were extracted from miRDB (v5.0) predictions74 (score cut off >60).
Co-expression module analysis
We used the weighted correlation network analysis (WGCNA) R package (v1.61)75 to determine co-expression modules among serum RNAs. A co-expression network shows a set of genes which tends to display a coordinated expression. This helps to identify genes that have common functions and regulators which can be missed by classical differential expression methods76.
The samples that have any missing values among their traits were filtered out and the remaining samples were utilized for co-expression module identification. The identified modules (min. module size is 10) were mapped to the sample traits to find significantly (p-values < 0.01) correlated associations between the modules and traits. The effect sizes were measured using Pearson correlation coefficients.
Ethics approval
This study was approved by the regional committees for medical and health research ethics, Oslo, Norway [2016/1290] and we confirm that all experiments were performed in accordance with the committee’s guidelines and regulations. The donors to the Janus Serum Bank have given their broad consent for their samples to be used for research. Samples collected in 1997 and onwards are based on an explicit informed consent. The Norwegian Data Protection Authority has approved the use of data and serum samples collected in the period 1972–2009, based on a broad consent from the donors.
Data Availability
Sequence data have been deposited at the European Genome-phenome Archive (EGA), which is hosted by the EBI, under accession number EGAS00001002814. Custom scripts are available from the corresponding author upon request.
References
Harrow, J. et al. GENCODE: The Reference Human Genome Annotation for The ENCODE Project. Genome Res 22, 1760–1774 (2012).
Mattick, J. S. Non-coding RNAs: The architects of eukaryotic complexity. EMBO Rep. 2, 986–991 (2001).
Pircher, A., Bakowska-Zywicka, K., Schneider, L., Zywicki, M. & Polacek, N. An mRNA-Derived Noncoding RNA Targets and Regulates the Ribosome. Mol. Cell 54, 147–155 (2014).
Tuck, A. C. & Tollervey, D. RNA in pieces. Trends Genet. 27, 422–432 (2011).
Lee, Y. S. et al. A novel class of small RNAs: tRNA-derived RNA fragments (tRFs). Genes Dev. 23, 2639–2649 (2009).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 108, 5003–8 (2011).
Vickers, K. C., Palmisano, B. T., Shoucri, B. M., Shamburek, R. D. & Remaley, A. T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat Cell Biol 13, 423–433 (2011).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9, 654–659 (2007).
Mitchell, P. S. et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 105, 10513–8 (2008).
Weber, J. A. et al. The microRNA spectrum in 12 body fluids. Clin. Chem. 56, 1733–1741 (2010).
Freedman, J. E. et al. Diverse human extracellular RNAs are widely detected in human plasma. Nat. Commun. 7, 11106 (2016).
Esteller, M. Non-coding RNAs in human disease. Nat Rev Genet 12, 861–874 (2011).
Krol, J., Loedige, I. & Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nat. Rev. Genet. 11, 597–610 (2010).
Wang, K. et al. Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc. Natl. Acad. Sci. USA 106, 4402–7 (2009).
Ai, J. et al. Circulating microRNA-1 as a potential novel biomarker for acute myocardial infarction. Biochem. Biophys. Res. Commun. 391, 73–77 (2010).
Tsujiura, M. et al. Circulating microRNAs in plasma of patients with gastric cancers. Br. J. Cancer 102, 1174–9 (2010).
Zhu, W., Qin, W., Atasoy, U. & Sauter, E. R. Circulating microRNAs in breast cancer and healthy subjects. BMC Res. Notes 2, 89 (2009).
Lawrie, C. H. et al. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br. J. Haematol. 141, 672–675 (2008).
Bianchi, F. et al. A serum circulating miRNA diagnostic test to identify asymptomatic high-risk individuals with early stage lung cancer. EMBO Mol. Med. 3, 495–503 (2011).
Maclellan, S. A., Macaulay, C., Lam, S. & Garnis, C. Pre-profiling factors influencing serum microRNA levels. BMC Clin. Pathol. 14, 1–11 (2014).
Rounge, T. B. et al. MicroRNA biomarker discovery and high-throughput DNA sequencing are possible using long-term archived serum samples. Cancer Epidemiol. Biomarkers Prev. 24, 1381–1387 (2015).
Morgan, C. P. & Bale, T. L. Sex differences in microRNA regulation of gene expression: no smoke, just miRs. Biol. Sex Differ. 3, 22 (2012).
Guo, L., Zhang, Q., Ma, X., Wang, J. & Liang, T. miRNA and mRNA expression analysis reveals potential sex- biased miRNA expression. Nature 7, 39812 (2017).
Sharma, S. & Eghbali, M. Influence of sex differences on microRNA gene regulation in disease. Biol Sex Differ 5, 3 (2014).
Kuokkanen, S. et al. Genomic profiling of microRNAs and messenger RNAs reveals hormonal regulation in microRNA expression in human endometrium. Biol Reprod 82, 791–801 (2010).
Waltering, K. K. et al. Androgen regulation of micro-RNAs in prostate cancer. Prostate 71, 604–614 (2011).
Macias, S., Michlewski, G. & Cáceres, J. F. Hormonal Regulation of MicroRNA Biogenesis. Mol. Cell 36, 172–173 (2009).
Loher, P., Londin, E. R. & Rigoutsos, I. IsomiR expression profiles in human lymphoblastoid cell lines exhibit population and gender dependencies. Oncotarget 5, 8790–8802 (2014).
Meder, B. et al. Influence of the Confounding Factors Age and Sex on MicroRNA Profiles from Peripheral Blood. Clin. Chem. 1208, 1200–1208 (2014).
Li, X., Khanna, A., Li, N. & Wang, E. Circulatory mir-34a as an RNA-based, noninvasive biomarker for brain aging. Aging (Albany. NY). 3, 985–1002 (2011).
Huan, T. et al. Age-associated microRNA expression in human peripheral blood is associated with all-cause mortality and age-related traits. Aging Cell 1–10, https://doi.org/10.1111/acel.12687 (2017).
Olivieri, F. et al. Age-related differences in the expression of circulating microRNAs: miR-21 as a new circulating marker of inflammaging. Mech. Ageing Dev. 133, 675–685 (2012).
Dhahbi, J. M. Circulating small noncoding RNAs as biomarkers of aging. Ageing Res. Rev. 17, 86–98 (2014).
Georgiadis, P. et al. Omics for prediction of environmental health effects: Blood leukocyte-based cross-omic profiling reliably predicts diseases associated with tobacco smoking. Sci. Rep. 6, 20544 (2016).
Wang, G. et al. Persistence of smoking-induced dysregulation of MiRNA expression in the small airway epithelium despite smoking cessation. PLoS One 10, 1–18 (2015).
Shi, B., Gao, H., Zhang, T. & Cui, Q. Analysis of plasma microRNA expression profiles revealed different cancer susceptibility in healthy young adult smokers and middle-aged smokers. Oncotarget 7, 21676–85 (2016).
Arner, P. & Kulyté, A. MicroRNA regulatory networks in human adipose tissue and obesity. Nat. Publ. Gr. 11, 276–28825 (2015).
Ameling, S. et al. Associations of circulating plasma microRNAs with age, body mass index and sex in a population-based study. BMC Med. Genomics 8, 61 (2015).
Chilton, W. L. et al. Acute exercise leads to regulation of telomere-associated genes and microrna expression in immune cells. PLoS One 9 (2014).
Radom-aizik, S. Jr., Zaldivar, F. P., Haddad, F. & Cooper, D. M. Brain, Behavior, and Immunity Impact of brief exercise on circulating monocyte gene and microRNA expression: Implications for atherosclerotic vascular disease. Brain Behav. Immun. 39, 121–129 (2014).
Parr, E. B. et al. Circulating microrna responses between ‘high’ and ‘low’ responders to a 16-Wk diet and exercise weight loss intervention. PLoS One 11, 1–14 (2016).
Xu, T. et al. Circulating microRNAs in response to exercise. Scand. J. Med. Sci. Sports 25, n/a–n/a (2015).
Baggish, A. L. et al. Dynamic regulation of circulating microRNA during acute exhaustive exercise and sustained aerobic exercise training. J Physiol 58916, 3983–3994 (2011).
Langseth, H., Gislefoss, R. E., Martinsen, J., Dillner, J. & Ursin, G. Cohort Profile: The Janus Serum Bank Cohort in Norway. Int. J. Epidemiol. 46, 403–404g (2016).
Hjerkind, K. et al. Cohort Profile Update: The Janus Serum Bank Cohort in Norway. Int. J. Epidemiol. 46, 1101–1102f (2017).
Keller, A. et al. Sources to variability in circulating human miRNA signatures. RNA Biol. 6286, 1791–1798 (2017).
Umu, S. U. et al. A comprehensive profile of circulating RNAs in human serum. RNA Biol. 15, 242–250 (2017).
Hackl, M. et al. miR-17, miR-19b, miR-20a, and miR-106a aredown-regulated in human aging. Aging Cell 9, 291–296 (2010).
Ruggiero, C. et al. White Blood Cell Count and Mortality in the Baltimore Longitudinal Study of Aging. J. Am. Coll. Cardiol. 49, 1841–1850 (2007).
McKay, D. & Jameson, J. Kidney transplantation and the ageing immune system. Nat. Rev. Nephrol. 8, 700–708 (2012).
Grivna, S. T., Beyret, E., Wang, Z. & Lin, H. RESEARCH COMMUNICATION A novel class of small RNAs in mouse spermatogenic cells. Genes Dev. 20, 1709–1714 (2006).
Girard, A., Sachidanandam, R., Hannon, G. J. & Carmell, M. A. A germline-specific class of small RNAs binds mammalian Piwi proteins. Nature 442, 199–202 (2006).
Yuan, T. et al. Plasma extracellular RNA profiles in healthy and cancer patients. Sci. Rep. 6, 19413 (2016).
Guida, F. et al. Dynamics of smoking-induced genome-wide methylation changes with time since smoking cessation. Hum. Mol. Genet. 24, 2349–2359 (2015).
Tsaprouni, L. G. et al. Cigarette smoking reduces DNA methylation levels at multiple genomic loci but the effect is partially reversible upon cessation. Epigenetics 9, 1382–1396 (2014).
Pidoplichko, V. I. et al. Nicotinic Cholinergic Synaptic Mechanisms in the Ventral Tegmental Area Contribute to Nicotine Addiction. Learn Mem 11, 60–69 (2004).
Ma, Y. & Li, M. D. Establishment of a Strong Link Between Smoking and Cancer Pathogenesis through DNA MethylationAnalysis. Sci. Rep. 1–13, https://doi.org/10.1038/s41598-017-01856-4 (2017).
Mao, Y. S., Sunwoo, H., Zhang, B. & Spector, D. L. No TitleDirect visualization of the co-transcriptional assembly of a nuclear body by noncoding RNAs. Nat. Cell Biol. 13, 95–101 (2011).
Martianov, I., Ramadass, A., Serra Barros, A., Chow, N. & Akoulitchev, A. Repression of the human dihydrofolate reductase gene by a non-coding interfering transcript. Nature 445, 666 (2007).
Feng, J. et al. The Evf-2 noncoding RNA is transcribed from the Dlx-5/6 ultraconserved region and functions as a Dlx-2 transcriptional coactivator. Genes Dev. 20, 1470–1484 (2006).
Lee, J. T. & Bartolomei, M. S. X-inactivation, imprinting, and long noncoding RNAs in health and disease. Cell 152, 1308–1323 (2013).
Kozomara, A. & Griffiths-Jones, S. MiRBase: Annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 42, 68–73 (2014).
Zhang, P. et al. PiRBase: A Web resource assisting piRNA functional study. Database 2014, 1–7 (2014).
Fromm, B. et al. A Uniform System for the Annotation of Vertebrate microRNA Genes and the Evolution of the Human microRNAome. Annu. Rev. Genet. 49, 213–42 (2015).
Teteloshvili, N. et al. Age-associated differences in MiRNA signatures are restricted to CD45RO negative T cells and are associated with changes in the cellular composition, activation and cellular ageing. PLoS One 10, 1–13 (2015).
Umu, S. U. et al. A comprehensive profile of circulating RNAs in human serum. RNA Biol. 0, 1–9 (2017).
Tosar, J. P., Rovira, C. & Cayota, A. Non-coding RNA fragments account for the majority of annotated piRNAs expressed in somatic non-gonadal tissues. Commun. Biol. 1, 2 (2018).
Aires, N., Selmer, R. & Thelle, D. The validity of self-reported leisure time physical activity, and its relationship to serum cholesterol, blood pressure and body mass index. A population based study of 332,182 men and women aged 40–42 years. Eur J Epidemiol 18, 479–85 (2003).
Schubert, M., Lindgreen, S. & Orlando, L. AdapterRemovalv2: rapid adapter trimming, identification, and read merging. BMC Res. Notes 9, 88 (2016).
Pantano, L., Estivill, X. & Martí, E. SeqBuster, a bioinformatic tool for the processing and analysis of small RNAs datasets, reveals ubiquitous miRNA modifications in human embryonic cells. Nucleic Acids Res. 38 (2009).
Anders, S., Pyl, P. T. & Huber, W. HTSeq-A Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq. 2. Genome Biol. 15, 550 (2014).
Ogata, H. et al. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 27, 29–34 (1999).
Wong, N. & Wang, X. miRDB: An online resource for microRNA target prediction and functional annotations. Nucleic Acids Res. 43, D146–D152 (2015).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9, 559 (2008).
van Dam, S., Võsa, U., van der Graaf, A., Franke, L. & de Magalhães, J. P. Gene co-expression analysis for functional classification and gene–disease predictions. Brief. Bioinform. 19, bbw139 (2017).
Acknowledgements
This work was supported by The Norwegian Research Council’s Programme ‘Human Biobanks and Health Data [229621/H10, 248791/H10]. We would like to acknowledge Cecilie Bucher-Johannessen, Marianne Lauritzen, Magnus Leithaug for performing lab and coordination tasks and Ronnie Babigumira, Jan Ivar Martinsen and Steinar Auensen for data management and IT support. We acknowledge the Norwegian Institute of Public Health for access to survey data in this study. The sequencing service was provided by the Norwegian Sequencing Centre (www.sequencing.uio.no), a national technology platform hosted by Oslo University Hospital and the University of Oslo supported by the Research Council of Norway and the Southeastern Regional Health Authority.
Author information
Authors and Affiliations
Contributions
T.B.R. and H.L. designed the study, T.B.R. and S.U.U. performed the analyses of the data, T.B.R., R.L. and H.L. wrote the draft of the paper, T.B.R., S.U.U., A.K., E.M., G.U., S.T., R.L. and H.L. discussed the results and contributed to the writing of the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rounge, T.B., Umu, S.U., Keller, A. et al. Circulating small non-coding RNAs associated with age, sex, smoking, body mass and physical activity. Sci Rep 8, 17650 (2018). https://doi.org/10.1038/s41598-018-35974-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35974-4
Keywords
This article is cited by
-
Promising microRNAs in pre-diagnostic serum associated with lung cancer up to eight years before diagnosis: a HUNT study
Journal of Cancer Research and Clinical Oncology (2024)
-
Current challenges and best practices for cell-free long RNA biomarker discovery
Biomarker Research (2022)
-
Faecal miRNA profiles associated with age, sex, BMI, and lifestyle habits in healthy individuals
Scientific Reports (2021)
-
Modulations of obesity-related microRNAs after exercise intervention: a systematic review and bioinformatics analysis
Molecular Biology Reports (2021)
-
Evaluation of commercially available small RNASeq library preparation kits using low input RNA
BMC Genomics (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.