Abstract
The amphibian chytrid fungus Batrachochytrium dendrobatidis is an emerging infectious pathogen present on every continent except Antarctica. It causes the disease chytridiomycosis in a subset of species but does not always result in disease or death for every host. Ambient temperature influences both amphibian metabolism and chytrid pathogenicity, however the interactive effects on host physiology is not well understood. We investigated the sublethal effect of B. dendrobatidis infection on a susceptible host, Litoria aurea to test (1) whether the infection load, metabolic activity, body fat and gonad size differed in L. aurea at either 24 °C or 12 °C ambient temperatures and (2) whether previous Bd infection caused long-term changes to body fat and gonad size. Litoria aurea in 12 °C treatments had higher infection loads of B. dendrobatidis and lower survivorship. Metabolic rate was higher and fat mass was lower in infected individuals and in animals in 24 °C treatments. Male L. aurea previously infected with B. dendrobatidis had smaller testes 5 months-post clearance of infection, an effect likely to translate to fitness costs in wild populations. These experiments demonstrate a physiological cost to sublethal B. dendrobatidis infection, which suggests a reduction in host fitness mediated by temperature in the host’s environment regardless of whether infection leads to mortality.
Similar content being viewed by others
Introduction
Chronic sublethal infections can cause a range of deleterious impacts in the host1, placing a substantial and often unpredictable stress on host fitness2. Such stress can result in the redistribution of energy away from reproduction, investing instead in high priority resources such as immunity and the nervous system3,4, ultimately affecting host population dynamics5. Persistent and sublethal infections may dramatically alter the behaviour and physiology of host species causing both direct reductions in survival, or affecting fitness through reductions in reproductive potential6. Hosts also exhibit costly defence responses against parasites, including complex immune systems, behavioural and chemosensory driven avoidance, fever responses and terminal investment strategies to increase fitness7. These complex interactions in species susceptible to emerging infectious diseases will be better understood by first understanding the key driving mechanisms in which pathogens directly affect their host.
The amphibian chytrid fungus B. dendrobatidis infects a multitude of amphibian hosts around the world8 and has been detected in over 42% of the 1240 species currently surveyed9. It causes the disease chytridiomycosis in a subset of species susceptible to increases in infection loads beyond a tolerance threshold10,11,12. However, infection does not necessarily result in disease or death in all individuals, including susceptible species13, and in some individuals of such species the epidermal load of B. dendrobatidis can fluctuate without resulting in obvious symptoms or harm10,14. Progression of infection is influenced by the external environment, and ambient temperature can strongly influence the infection load of ectothermic hosts15,16,17. While B. dendrobatidis grows optimally at 17–25 °C18 in culture media, interactions between frog immune systems, the skin microbiome19 and pathogen growth at different temperatures can result in higher infection loads at lower temperatures (below pathogen thermal optima) where host-immunity is inhibited20,21,22, or in slower growth in the optimal pathogen temperature range when frog immune function is greater, even allowing susceptible individuals to sometimes clear infections23,24. Host responses to infection must therefore be considered within the context of the environment, particularly ambient temperature, given its ability to drive pathogen-specific traits as well as ectothermic host responses25.
Whilst B. dendrobatidis has the potential to induce sublethal responses, our understanding of the effects of infection on the physiology of individuals is poor. Previous studies on sublethal impacts of Batrachochytrium dendrobatidis infection have demonstrated lower respiration rate12, reduced body mass26,27 and locomotion metrics28 and retarded growth rates29,30. Reproductive metrics such as calling activity may increase in infected individuals31 while call frequency remains unchanged32. Spermatogenesis and oogenesis can increase in diseased individuals suggesting a terminal investment strategy33, while in other cases there are no spermatogenic differences between infected and uninfected individuals28. While much research has focused on B. dendrobatidis as a direct driver of population declines through increased mortality, understanding such sublethal effects of infection is crucial to understanding how the fitness of populations of susceptible species that persist with the disease and even relatively unaffected reservoir species might be challenged. Given the influence of temperature on host – parasite interactions25 and the energetically costly effects of sublethal infections3,4, it is important to understand the metabolic responses of B. dendrobatidis infection in the context of the ambient environmental temperature of the host.
The objective of this study was to explain the causes of previously demonstrated sublethal effects of B. dendrobatidis infection by focussing on metabolic and temperature interactions of a susceptible species34,35, the green and golden bell frog, Litoria aurea. Specifically, we aimed to test (1) whether the infection load, metabolic activity, and fat storage differed between L. aurea at high (24 °C) and low (12 °C) ambient temperatures following infection with B. dendrobatidis and (2) whether body fat and gonad size differed in frogs previously infected, but subsequently cleared of infection.
Methods
Experiment 1: Sublethal B. dendrobatidis infection of sexually immature L. aurea at winter (12 °C) and summer (24 °C) mean temperatures
Husbandry and experimental setup
Litoria aurea were bred from second generation captive breeding stock (derived from Kooragang Island, New South Wales, Australia, latitude: −32.862911°S, longitude: 151.728685°N) and reared at the University of Newcastle, New South Wales. Offspring were housed in groups of approximately 200 tadpoles in 450 × 300 × 150 mm aquaria as tadpoles under laboratory conditions with a 12 hour light/dark regime and were fed a mixed diet of boiled lettuce and trout pellets ad libitum. All individuals were transferred to 170 × 120 × 75 mm aquaria maintained under laboratory conditions post-metamorphosis. Eighty frogs that were 6–10 weeks post-metamorphosis were selected at random from 8 different clutches and placed individually into separate transparent plastic aquaria (170 × 120 × 75 mm). Aquaria contained autoclaved deionised water and pebbles as the substrate, and were tilted so that water covered 50% of the base in each aquarium. Each individual was allocated randomly to avoid effects of clutch to one of the four experimental groups (n = 20 per treatment) according to a two-way factorial design with temperature (12 °C or 24 °C) and B. dendrobatidis exposure (infected or uninfected) as treatments. The temperature treatments were selected because they are the approximate means of winter and summer in the sourced frogs’ range respectively. Each aquarium was maintained in an illuminated, refrigerated incubator (TRISL – 1175, Thermoline Scientific Equipment, Wetherill Park, NSW) with air-temperature set to 12 °C or 24 °C. The incubator cycled on a 12 hour light/dark regime and the temperature was monitored continuously within each incubator. Each day, all aquaria were randomly reallocated to a new position in the temperature cabinet in order to normalise any differences in temperature and lighting within the cabinet. Aquarium water was changed every 2–3 days and frogs were provided with 4–6 small, calcium powder dusted crickets twice per week. The condition and health of each individual was monitored daily.
Batrachochytrium dendrobatidis cultivation and inoculation
Samples of B. dendrobatidis (strain: Gibbo River-Llesueuri-00-LB-1, passage number 4) were obtained and cultured on TGhL agar plates, which were then flooded with sterilised water to harvest the actively growing zoospores. Exposure treatment individuals (n = 40) received a single inoculate with B. dendrobatidis by aliquoting 2 mL of the zoospore suspension directly into the aquarium water of each treatment individual. A 100 µl sample of the B. dendrobatidis suspension stock solution was used to quantify the concentration of zoospores using a haemocytometer, which was determined to be 9.05 × 106 cells/mL. For sham controls, sterile TGhL agar plates were prepared and maintained under the same conditions as the plates containing active B. dendrobatidis. The sterile plates were flooded with sterilised water and 2 mL of the sham solution was aliquoted into the water bath of each control individual as a sham inoculation. Water changes were postponed for approximately 10 days after addition of B. dendrobatidis or sham inoculations. All individuals were closely monitored after inoculation for signs of rapid onset of disease.
Batrachochytrium dendrobatidis detection and quantification
All individuals were tested for infection prior to inoculation with either the B. dendrobatidis or sham solutions to ensure that individuals were naïve to their corresponding treatments. A standardised swabbing technique was employed over the epidermal surfaces prone to high infection loads, which involved wiping both sides of the ventral skin 8 times, the inner and outer thighs 4 times and the hind and fore feet 2 times each10. DNA extraction and quantification from the swabbed samples was conducted following standard qPCR Taqman assay methods36, using a Rotor Gene 6000 Real Time DNA amplification system. Each swab was assessed for the presence of B. dendrobatidis in triplicate using a 1/10 diluted sample from the extracted DNA, and are given as the mean chytrid DNA molecules per microlitre (molecules/µl) of a standardised extract volume (5 µl) from three replicates of the same swab sample. To generate standard curves, calibration standards were obtained from Pisces Molecular (Boulder, CO, USA) and were serially diluted to prepare four known B. dendrobatidis concentrations (18700, 1870, 187 and 18.7 molecules/microliter). Where replication of B. dendrobatidis DNA occurred in all three replicates, the number of molecules per microliter was summarised as the mean infection load. Where a result occurred with amplification in two of three replicates and the negative template control revealed no contamination, the sample was considered positive and the mean was calculated from all three replicates. Results with amplification of only one replicate were considered negative for B. dendrobatidis DNA. It was necessary to include zero values in the calculations as it was assumed to be the result of a low quantity of DNA in the sample10,37. All values were multiplied by 10 to account for the dilution step. Negative results were recorded for samples if all three replicates did not amplify, providing that the sample was not inhibited. To test for inhibition, internal positive controls were included in one of the replicates for each sample including the negative template control. Following qPCR, a threshold was set midway up the amplification curve, where inhibition was confirmed if the sample crossed the threshold more than five cycles after the negative template control. If the sample was inhibited, a 1/100 dilution with sterile Milli-Q water was performed on the originally extracted sample to dilute any potential inhibiting compounds and the qPCR process was repeated. The result of the diluted sample was multiplied by 100 to factor for the dilution step.
Batrachochytrium dendrobatidis infection loads and survival
Each frog was swabbed and tested for infection loads at 7, 17, 39 and 48 days post-exposure to the B. dendrobatidis or sham inoculations. Survival at 48 days was recorded.
Standard metabolic rate
Eight individuals were randomly selected from each of the four experimental treatment groups forty-one days post-exposure to B. dendrobatidis to measure the standard metabolic rate (SMR), however only the surviving 8 individuals were used for the 12 °C infected group. Individual frogs were fasted for six days before standard metabolic rate measurements to ensure they were in a post-absorptive state38. Each individual was placed in a 100 ml Schott bottle chamber that contained 80 ml of agarose gel, added for the purpose of reducing excess volume of the chamber to improve the accuracy of the reading. The chamber was sterilised between individuals by boiling the agarose gel in a microwave to prevent any cross-contamination of B. dendrobatidis. Each chamber also contained filter paper with 1 ml of deionised water to prevent desiccation of frogs during the procedure. Individual metabolic chambers containing frogs were immersed in a water bath at either 12 °C or 24 °C according to their allocated experimental temperature treatment. The rate of oxygen consumption in each metabolic chamber was measured by attaching the chamber to a computer-controlled, closed-circuit, indirect respirometer (Micro Oxymax, Columbus Instruments, Columbus, OH). The respirometer was manually calibrated each time the experiment was repeated. All metabolic measurements were conducted during daylight hours (09:00–19:00 h) to maintain consistency of readings within the diurnal phase of the diel cycle for each treatment group. The oxygen consumption of each individual was monitored for five hours, but only data from the last four hours were recorded. After the final data point was recorded each individual was weighed and returned to its aquarium. The SMR was calculated by dividing the rate of oxygen consumption (O2 ml/h) by the body mass of the individual (g) raised to the power of 0.7539.
Euthanasia and determination of abdominal fat mass
After 48 d post-exposure to the B. dendrobatidis or sham inoculation, all surviving frogs were euthanized via immersion in tricaine methanesulfonate (MS-222; Sigma-Aldrich). The fat bodies were surgically removed under a dissecting microscope, blot-dried and weighed whole on an analytical balance ( ± 0.0001 g).
Experiment 2: Long-term sublethal effects of a previous B. dendrobatidis infection in L. aurea
Litoria aurea (n = 84) were investigated to examine the chronic effect of a previous B. dendrobatidis infection on gonad mass. Frogs were obtained from the same captive colony described in experiment 1 and also followed the same animal husbandry, B. dendrobatidis cultivation, inoculation, detection and quantification procedures. There was no experimental temperature treatment for experiment 2 and all frogs were held at 20 °C. Batrachochytrium dendrobatidis exposed (n = 48) and sham exposed (n = 48) frogs were maintained for 95 days prior to undergoing a standard heat-treatment protocol to clear them of infection40. All frogs survived in the sham exposed group while 12 individuals died following B. dendrobatidis exposure. These frogs, cleared of infection, were held for five months following confirmation of the absence of infection. All individuals were then euthanized with MS-222, weighed, dissected and the gonad mass was determined. The gonads were blot dried and weighed as indicated above.
All animals were held under a NSW scientific licence (approval number SL100421), and the experiments were conducted with the approval and in accordance of the University of Newcastle ACEC (approval number A-2008-165).
Statistical analyses in Experiments 1 and 2
Infection load within the infected proportion of the population was analysed over time using a generalised linear mixed effects model (GLMM) in SAS (version 9.4) using the PROC MIXED procedure. Temperature, time and the interaction between the two were modelled as fixed effects with animal ID modelled as a random effect. Repeated measures over time were modelled with a residual covariance matrix with compound symmetry covariance structure. The infection load data were log transformed (molecules/μl + 0.1). Adding 0.1 to all infection load data allowed individuals that were negative for infection (i.e. 0 molecules/μl) to be log transformed.
The data for the proportion of individuals surviving across time in experiment 1 were analysed using a Log Rank test in a Kaplan-Meier survival analysis in SPSS (Version 21, IBM, New York). Post-Hoc pairwise comparisons of survival were made between the four treatment groups (+/− B. dendrobatidis exposure; 12 °C and 24 °C) using data for the proportion of individuals alive at 48 days.
The SMR data were analysed using a two-way analysis of variance (two-way ANOVA) in JMP (Version 11, SAS Institute, Cary, North Carolina) with temperature (12 °C or 24 °C), B. dendrobatidis exposure (infected or uninfected) and their interactions as the predictor variables. Individual body mass was taken into account by raising to the power of 0.75 (equation 1).
The fat mass data of frogs from experiment 1 were analysed using a two-way analysis of variance (two-way ANOVA) in JMP (Version 11, SAS Institute, Cary, North Carolina) where temperature (12 °C or 24 °C), B. dendrobatidis exposure (infected or uninfected) and their interactions were the selected predictor variables.
Ovary and testes mass data of frogs from experiment 2 were analysed separately using an analysis of covariance (ANCOVA) in JMP (Version 11, SAS Institute, Cary, North Carolina) where B. dendrobatidis exposure (infected or uninfected) was the predictor variable. The body mass of each individual was included as a covariate in order to account for size differences between frogs. The weight of the individual was regressed against the weight of the ovaries or testes and separated by B. dendrobatidis exposure using a simple linear regression.
Results
Experiment 1: Sublethal B. dendrobatidis infection of sexually immature L. aurea
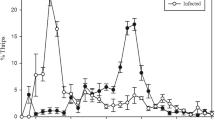
All B. dendrobatidis-exposed frogs at 12 °C were infected (n = 20), while 16 of the 20 individuals held at 24 °C became infected for the duration of the study. These four uninfected frogs were removed from the analyses. All unexposed frogs tested negative for infection. Temperature significantly affected the growth of B. dendrobatidis; infection loads were much higher in infected frogs at 12 °C than 24 °C (F = 22.28, df = 1, P = 0.001; Fig. 1a). Days post exposure also significantly affected the growth rate of B. dendrobatidis with infection loads increasing over time (F = 2.86, df = 3, P = 0.045, Fig. 1a). Temperature also interacted with days post exposure; infection loads were greater and increased faster in frogs held at 12 °C compared to those held at 24 °C (F = 3.12, df = 3, P = 0.034, Fig. 1a). There was no significant difference between infection loads at 7 days compared to 48 days in frogs held at 24 °C (t-value = −0.44, df = 57.3, P = 0.659, Fig. 1a), whereas the infection loads between 7 and 48 days post-exposure significantly differed in frogs held at 12 °C (t-value = −4.97, df = 55.3, P < 0.001, Fig. 1a).
Effect of temperature and Bd infection in L. aurea for experiment 1. (a) Effect of temperature (12 °C or 24 °C) on mean infection load in Bd infected frogs. Values are least squares means ± 1CI (95%); (b) Effect of temperature (12 °C or 24 °C) and Bd infection (infected or uninfected) on survival over 48 days; (c) Mean SMR in infected and uninfected frogs at 12 °C or 24 °C. Values are least squares means ± 1CI (95%); (d) Mean fat mass in infected and uninfected frogs at 12 °C or 24 °C. Values are log transformed least squares means ± 1CI (95%).
After 48 days, infected frogs that were held at 12 °C had significantly lower survivorship (30%), than uninfected frogs from both temperature treatments (95%), and frogs infected with B. dendrobatidis at 24 °C (90%) (χ2 = 31.77, df = 3, P < 0.001; Fig. 1b).
Infected individuals had a mean SMR of 0.16 O2 mL/h/g 0.75 ± 0.01 which was higher than uninfected individuals (0.13 O2 mL/h/g 0.75 ± 0.01) (F = 7.03, df = 1, P = 0.013, Fig. 1c). The SMR of individuals at 24 °C was 0.18 O2 mL/h/g 0.75 ± 0.01, which was significantly higher than those held at 12 °C (0.11 O2 mL/h/g 0.75 ± 0.01, F = 33.81, df = 1, P < 0.001). However, there was no interaction between temperature and infection status on SMR (F = 0.17, df = 1, P = 0.684, Fig. 1c).
Infected frogs had less stored fat (0.002 g ± 0.003) compared to uninfected individuals (0.01 g ± 0.002) (F = 5.19, df = 1, P < 0.03, Fig. 1d). Total fat mass was highly dependent on temperature; frogs maintained at 12 °C had far heavier fat bodies (0.02 g ± 0.003) than those kept at 24 °C (0.001 g ± 0.002) (F = 17.65, df = 1, P < 0.001, Fig. 1d). However, the interaction between temperature and infection status was non-significant (F = 3.32, df = 1, P = 0.078).
Experiment 2: Long-term sublethal effects of a previous B. dendrobatidis infection in L. aurea
Previously infected and non-infected male frogs had similar body weights (F = 0.60, df = 1, P = 0.552). Testes mass was positively correlated with the frog body mass (F = 70.60, df = 1, P < 0.001) and mean testes mass was significantly lower in the previously infected frogs (0.044 g ± 0.005) than uninfected frogs (0.078 g ± 0.005) (F = 14.35, df = 1, P = 0.001). The testes mass of previously infected frogs was much smaller for equivalent body weight than uninfected frogs causing an interaction between body mass and testes mass (F = 20.91, df = 1, P < 0.001; Fig. 2).
Female body weight was similar between the infected and uninfected groups (F = 0.36, df = 1, P = 0.552). Ovary mass positively correlated with the frog body weight (F = 93.57, df = 1, P = 0.001). However, there were no significant differences in ovary mass between infected and uninfected females (see Supplementary Fig. S3 online) (F = 3.71, df, = 1, P = 0.065).
Discussion
Litoria aurea experienced a range of sublethal impacts during and following infection with B. dendrobatidis. Metabolic activity increased and abdominal fat bodies decreased in juvenile frogs during infection whereas previously infected males had smaller testes. These effects are likely to translate to fitness costs in wild populations. Temperature had direct effects on physiological processes (e.g. metabolic rate and fat bodies) but did not interact with infection load to change the magnitude of physiological effects. Together, these experiments demonstrate an energetic cost to sublethal B. dendrobatidis infection, which suggests a physiological trade-off occurs to infected individuals regardless of the outcome of infection.
Infection loads were highest in the coolest temperature treatment, despite the warmer temperature group occurring within the optimum temperature range for B. dendrobatidis growth in culture (24 °C)18. This may be a result of life-history trade-offs in the growth of B. dendrobatidis zoospores, which though encysting and developing into zoosporangium faster at 17–25 °C, produce more zoospores that remain infectious for a longer period at 7–10 °C8. An additional explanation for this counter-intuitive effect may be owing to a decreased immune system function of frogs in response to cold-exposure20,41,42, which could allow B. dendrobatidis to proliferate in the host. The effects of temperature were clear in the response variables we measured, where frogs that are normally metabolically unchallenged at 12 °C were affected by the higher infection loads. In contrast, frogs held at 24 °C have higher temperature-driven metabolic rates, but experienced much lower infection loads. The disproportionate, temperature-dependent growth of B. dendrobatidis highlights the importance of investigating responses of the host in the context of the ambient temperature, especially if the effects are to be extrapolated to wild populations. Furthermore, the increased SMR of infected frogs can have major implications on host fitness if frogs are concurrently infected during the higher Summer temperatures. It is unclear how transferable these findings are for other amphibian species, and individual species responses are likely to involve interactions between the range of thermal tolerance of species and populations and their immune response to B. dendrobatidis infection. For example, alpine species with low tolerances to high temperatures and a low capacity to tolerate infections are likely to respond very differently to L. aurea, which can self-cure infections under specific conditions23,43.
Elevated SMR and reduced abdominal fat in frogs with active B. dendrobatidis infections supported the hypothesis that infection has physiological effects even when the infection has not progressed to chytridiomycosis; this adds to the growing body of literature demonstrating sublethal impacts of B. dendrobatidis. In tadpoles, reduced developmental rates are probably driven by reduced activity and efficiency when foraging44, possibly owing, in part, to physical degradation of mouth parts. In frogs, the increase in standard metabolic rate contributes to an individual’s energy budget and represents a fixed cost of maintaining the body’s processes45 and the increase in infected individuals may reflect the body’s requirement to work harder through damage caused by infection. Initial host invasion of susceptible and tolerant hosts is superficially similar and involves varying levels of disruption to epidermal cells46, resulting in increased skin sloughing47, which may direct resources away from other core functions and thus increase the SMR. The increase in SMR is the likely mechanism behind the reduction in abdominal fat because infected individuals had higher metabolic rates and were therefore using more energy, which is acquired through ingested prey and is removed from accumulated body energy stores in the form of fat bodies. Alternatively, it is possible that infected individuals did not eat as much as uninfected frogs, as we would expect if their foraging efficiency was reduced, but we did not see evidence of this during our controlled experiment. Determining the relationship between high SMR and total fitness in infected individuals would allow us to better understand the consequence of this physiological effect.
Abdominal fat bodies are important for both gonadal development48 and the production of gametes49 and so it is plausible that B. dendrobatidis infection reduced the amount of available energy for reproductive growth and development. Gonads of males previously infected with B. dendrobatidis had smaller testes despite a five month recovery period following clearance of infection. It is possible that the reduced testes size was a result of the energetic impacts that reduced fat bodies and increased SMR in experiment 1. Small testes may have fitness consequences because testis mass correlates with hormone production and reproductive effort50. However, testes size is a crude measure of short-term changes in reproductive effort and it is possible that increased spermatogenesis still occurred33 but did so within smaller testes. Alternatively, increased spermatogenesis may only occur once chytridiomycosis has progressed as is reflected in the condition dependent vocalisation effort of Litoria rhecoloa when infected with B. dendrobatidis31. Our results do not reflect the terminal investment hypothesis suggested in other studies, e.g. increased testes width of infected Rana pipiens28,33,51, but support studies suggesting supressed reproduction occurs through down regulation of sex hormones52. The reduced growth rate of the testes could have major implications for the reproductive output of a host population. Further investigation of this phenomenon in L. aurea is required to determine whether smaller testes are associated with a lower sperm production; this is obviously an important measure of reproductive function to understand in the context of overall male reproductive fitness.
The reduced abdominal fat and testes mass in B. dendrobatidis-infected frogs shows support for an energetic tradeoff, favouring other systems over reproduction. While there is the possibility that these individuals may still have the capacity to reproduce physiologically, the reduced energy supplies may affect their immediate survival53 and the ability to carry out expensive reproductive behaviours such as vocal calling and mate-seeking50. Such a reduction in fitness of individuals could possibly result in reduced breeding and increased mortality from secondary causes (i.e. predation), which may then lead to less stable populations. Host-populations that are then able to persist with low infection loads may still be at risk of decline despite the absence of mortality events attributed directly to B. dendrobatidis infection. Further research is needed to understand whether the fitness consequences of B. dendrobatidis infection on the individual demonstrated in this study are transferrable to wild populations of L. aurea and directly impact fitness at the population level.
The fact that females infected with B. dendrobatidis had similar ovary size than uninfected females, despite the differences observed in the male gonad mass, is interesting. Our results differ from those of infected Litoria verreauxii alpina, which had larger ovaries and oviducts in infected animals suggesting terminal investment occurs in diseased individuals28 in this species. We are unsure why such sex differences occurred in this species, but it is an area worthy of future investigation. There has been little research in this area to date and further studies should aim to resolve mechanisms and effects on female reproductive biology in other species.
In conclusion, our findings reveal further insights into the sublethal effects of B. dendrobatidis infection on both host physiology and reproductive potential. The colonisation of the amphibian chytrid fungus and subsequent battle between host and parasite have clear costs to L. aurea, regardless of infection outcome and there is mounting evidence that frogs do not have to be susceptible to chytridiomycosis to be at least temporarily affected by the fungus28,54,55. This is an important consideration in our understanding of the amphibian chytrid fungus, especially where declines have occurred without an obvious mortality of, or disease risk to, the host. Physiological effects involving increased SMR, reduced abdominal fat and reduced testes mass seem likely to translate to consequences for wild populations in terms of reduced total fitness, but this relationship is often assumed but rarely confirmed. Ecological studies that can incorporate infection dynamics into host fitness outcomes are necessary to provide a comprehensive understanding of the magnitude of these physiological effects.
Data Accessibility
We intend for the data of this study to be archived at the Dryad Digital Repository.
References
Boots, M. & Norman, R. Sublethal infection and the population dynamics of host–microparasite interactions. Journal of Animal Ecology 69, 517–524 (2000).
Kristan, D. M. & Hammond, K. A. Combined effects of cold exposure and sub-lethal intestinal parasites on host morphology and physiology. Journal of Experimental Biology 203, 3495–3504 (2000).
Apanius, V. Stress and immune defense. Advances in the Study of Behavior 27, 133–153 (1998).
Andre, S. E., Parker, J. & Briggs, C. J. Effect of temperature on host response to Batrachochytrium dendrobatidis infection in the mountain yellow-legged frog (Rana muscosa). Journal of Wildlife Diseases 44, 716–720 (2008).
Ives, A. R. & Murray, D. L. Can sublethal parasitism destabilize predator-prey population dynamics? A model of snowshoe hares, predators and parasites. J Anim Ecol, 265–278 (1997).
Bonsall, M., Sait, S. & Hails, R. Invasion and dynamics of covert infection strategies in structured insect–pathogen populations. J Anim Ecol 74, 464–474 (2005).
Shang, Y., Feng, P. & Wang, C. Fungi that infect insects: altering host behavior and beyond. PLoS Pathog. 11, e1005037 (2015).
Whitfield, S. M. et al. Infection and co-infection by the amphibian chytrid fungus and ranavirus in wild Costa Rican frogs. Diseases of aquatic organisms 104, 173–178 (2013).
Kerby, J. L., Schieffer, A., Brown, J. R. & Whitfield, S. Utilization of fast qPCR techniques to detect the amphibian chytrid fungus: a cheaper and more efficient alternative method. Methods in Ecology and Evolution 4, 162–166 (2013).
Stockwell, M. P., Clulow, J. & Mahony, M. J. Host species determines whether infection load increases beyond disease-causing thresholds following exposure to the amphibian chytrid fungus. Anim Conserv 13((Suppl. 1)), 62–71, https://doi.org/10.1111/j.1469-1795.2010.00407.x (2010).
Stockwell, M. P., Garnham, J. I., Bower, D. S., Clulow, J. & Mahony, M. J. Low disease-causing threshold in a frog species susceptible to chytridiomycosis. FEMS Microbiol. Lett. 363, fnw111 (2016).
Carey, C. et al. Experimental exposures of boreal toads (Bufo boreas) to a pathogenic chytrid fungus (Batrachochytrium dendrobatidis). EcoHealth 3, 5–21 (2006).
Gahl, M. K., Longcore, J. E. & Houlahan, J. E. Varying responses of northeastern North American amphibians to the chytrid pathogen Batrachochytrium dendrobatidis. Conserv Biol 26, 135–141 (2012).
Briggs, C. J., Knapp, R. A. & Vredenburg, V. T. Enzootic and epizootic dynamics of the chytrid fungal pathogen of amphibians. Proceedings of the National Academy of Sciences 107, 9695–9700 (2010).
Woodhams, D. C., Alford, R. A., Briggs, C. J., Johnson, M. & Rollins-Smith, L. A. Life-history trade-offs influence disease in changing climates: strategies of an amphibian pathogen. Ecology 89, 1627–1639 (2008).
Klop-Toker, K. et al. We made your bed, why won’t you lie in it? Food availability and disease may affect reproductive output of reintroduced frogs. PLoS One 11, e0159143 (2016).
Kriger, K. M., Hero, J.-M. & Ashton, K. J. Cost efficiency in the detection of chytridiomycosis using PCR assay. Diseases of aquatic organisms 71, 149–154 (2006).
Piotrowski, J. S., Annis, S. L. & Longcore, J. E. Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 96, 9–15 (2004).
Kolby, J. E. et al. First evidence of amphibian chytrid fungus (Batrachochytrium dendrobatidis) and ranavirus in Hong Kong amphibian trade. PLoS One 9, e90750 (2014).
Maniero, G. D. & Carey, C. Changes in selected aspects of immune function in the leopard frog, Rana pipiens, associated with exposure to cold. Journal of Comparative Physiology B 167, 256–263 (1997).
Rowley, J. J., Skerratt, L. F., Alford, R. A. & Campbell, R. Retreat sites of rain forest stream frogs are not a reservoir for Batrachochytrium dendrobatidis in northern Queensland, Australia. Diseases of aquatic organisms 74, 7–12 (2007).
Woodhams, D. C. & Alford, R. A. Ecology of chytridiomycosis in rainforest stream frog assemblages of tropical Queensland. Conservation Biology 19, 1449–1459 (2005).
Abu Bakar, A. et al. Susceptibility to disease varies with ontogeny and immunocompetence in a threatened amphibian. Oecologia, 1–13, https://doi.org/10.1007/s00442-016-3607-4 (2016).
Searle, C. L., Mendelson, J. R. III, Green, L. E. & Duffy, M. A. Daphnia predation on the amphibian chytrid fungus and its impacts on disease risk in tadpoles. Ecology and evolution 3, 4129–4138 (2013).
Blaustein, A. R. et al. Ecophysiology meets conservation: understanding the role of disease in amphibian population declines. Philosophical Transactions of the Royal Society of London B: Biological Sciences 367, 1688–1707 (2012).
Retallick, R. W. & Miera, V. Strain differences in the amphibian chytrid Batrachochytrium dendrobatidis and non-permanent, sub-lethal effects of infection. Diseases of aquatic organisms 75, 201–207 (2007).
Ramsey, J. P., Reinert, L. K., Harper, L. K., Woodhams, D. C. & Rollins-Smith, L. A. Immune defenses against Batrachochytrium dendrobatidis, a fungus linked to global amphibian declines, in the South African clawed frog, Xenopus laevis. Infection and immunity 78, 3981–3992 (2010).
Chatfield, M. W. et al. Fitness consequences of infection by Batrachochytrium dendrobatidis in northern leopard frogs (Lithobates pipiens). EcoHealth 10, 90–98 (2013).
Davidson, C. et al. Effects of chytrid and carbaryl exposure on survival, growth and skin peptide defenses in foothill yellow-legged frogs. Environmental science & technology 41, 1771–1776 (2007).
Garner, T. W. et al. Life history tradeoffs influence mortality associated with the amphibian pathogen Batrachochytrium dendrobatidis. Oikos 118, 783–791 (2009).
Roznik, E. A., Sapsford, S. J., Pike, D. A., Schwarzkopf, L. & Alford, R. A. In Proc. R. Soc. B. 20150694 (The Royal Society).
Greenspan, S. E., Roznik, E. A., Schwarzkopf, L., Alford, R. A. & Pike, D. A. Robust calling performance in frogs infected by a deadly fungal pathogen. Ecol. Evol. 6, 5964–5972 (2016).
Brannelly, L. A., Webb, R., Skerratt, L. F. & Berger, L. Amphibians with infectious disease increase their reproductive effort: evidence for the terminal investment hypothesis. Open Biology 6, 150251 (2016).
Mahony, M. J. et al. Identifying conservation and research priorities in the face of uncertainty: a review of the threatened bell frog complex in eastern Australia. Herpetol Conserv Biol 8, 519–538 (2013).
Stockwell, M., Clulow, S., Clulow, J. & Mahony, M. The impact of the amphibian chytrid fungus Batrachochytrium dendrobatidis on a green and golden bell frog Litoria aurea reintroduction program at the Hunter Wetlands Centre Australia in the Hunter Region of NSW. Australian Zoologist 34, 379–386 (2008).
Boyle, D., Boyle, D., Olsen, V., Morgan, J. & Hyatt, A. Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Diseases of aquatic organisms 60, 141–148 (2004).
Hyatt, A. et al. Diagnostic assays and sampling protocols for the detection of Batrachochytrium dendrobatidis. Dis Aquat Org 73, 175–192 (2007).
Homyack, J. A., Haas, C. A. & Hopkins, W. A. Influence of temperature and body mass on standard metabolic rate of eastern red-backed salamanders (Plethodon cinereus). Journal of Thermal Biology 35, 143–146 (2010).
White, C. R., Phillips, N. F. & Seymour, R. S. The scaling and temperature dependence of vertebrate metabolism. Biology Letters 2, 125–127 (2006).
Johnson, M. L., Berger, L., Philips, L. & Speare, R. Fungicidal effects of chemical disinfectants, UV light, desiccation and heat on the amphibian chytrid Batrachochytrium dendrobatidis. Diseases of Aquatic Organisms 57, 255–260, https://doi.org/10.3354/dao057255 (2003).
Green, N. & Cohen, N. Effect of temperature on serum complement levels in the leopard frog, Rana pipiens. Developmental & Comparative Immunology 1, 59–64 (1977).
Cooper, E. L., Wright, R. K., Klempau, A. E. & Smith, C. T. Hibernation alters the frog’s immune system. Cryobiology 29, 616–631 (1992).
Clulow, S. et al. Elevated salinity blocks pathogen transmission and improves host survival for a globally pandemic disease: implications for amphibian translocations. J Appl Ecol (In Press).
Venesky, M. D., Parris, M. J. & Storfer, A. Impacts of Batrachochytrium dendrobatidis infection on tadpole foraging performance. EcoHealth 6, 565–575 (2009).
Steyermark, A. C., Miamen, A. G., Feghahati, H. S. & Lewno, A. W. Physiological and morphological correlates of among-individual variation in standard metabolic rate in the leopard frog Rana pipiens. Journal of Experimental Biology 208, 1201–1208 (2005).
Greenspan, S. E., Longcore, J. E. & Calhoun, A. J. Host invasion by Batrachochytrium dendrobatidis: fungal and epidermal ultrastructure in model anurans. Dis Aquat Org 100, 201–210 (2012).
Berger, L., Speare, R. & Skerratt, L. F. Distribution of Batrachochytrium dendrobatidis and pathology in the skin of green tree frogs Litoria caerulea with severe chytridiomycosis. Dis Aquat Org 68, 65–70 (2005).
Fitzpatrick, L. C. Life history patterns of storage and utilization of lipids for energy in amphibians. American Zoologist 16, 725–732 (1976).
Scott, D. E., Casey, E. D., Donovan, M. F. & Lynch, T. K. Amphibian lipid levels at metamorphosis correlate to post-metamorphic terrestrial survival. Oecologia 153, 521–532 (2007).
Emerson, S. B. & Hess, D. L. Glucocorticoids, androgens, testis mass, and the energetics of vocalization in breeding male frogs. Horm Behav 39, 59–69 (2001).
An, D. & Waldman, B. Enhanced call effort in Japanese tree frogs infected by amphibian chytrid fungus. Biology letters 12, 20160018 (2016).
Kindermann, C., Narayan, E. J. & Hero, J.-M. Does physiological response to disease incur cost to reproductive ecology in a sexually dichromatic amphibian species? Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 203, 220–226 (2017).
Sorci, G., Clobert, J. & Michalakis, Y. Cost of reproduction and cost of parasitism in the common lizard, Lacerta vivipara. Oikos, 121–130 (1996).
Bielby, J., Fisher, M. C., Clare, F. C., Rosa, G. M. & Garner, T. W. Host species vary in infection probability, sub-lethal effects, and costs of immune response when exposed to an amphibian parasite. Scientific Reports 5 (2015).
Padgett-Flohr, G. E. Pathogenicity of Batrachochytrium dendrobatidis in two threatened California amphibians: Rana draytonii and Ambystoma californiense. Herpetological Conservation and Biology 3, 182–191 (2008).
Acknowledgements
We would like to thank Kim Colyvas for statistical advice and Adam Spring for assistance in the laboratory.
Author information
Authors and Affiliations
Contributions
L.C., M.S. and D.B. conceived the original concept and designed the methodology; L.C. collected the data; L.C. and M.S. analysed the data; S.C., J.C. and M.M. assisted with experimental design, supervision and specialist advice, and reviewed the manuscript; L.C., D.B. and S.C. wrote the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Campbell, L., Bower, D.S., Clulow, S. et al. Interaction between temperature and sublethal infection with the amphibian chytrid fungus impacts a susceptible frog species. Sci Rep 9, 83 (2019). https://doi.org/10.1038/s41598-018-35874-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35874-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.