Abstract
Parkinson’s disease (PD) and multiple system atrophy (MSA) are neurodegenerative diseases characterized by inclusions mainly composed of α-synuclein (α-syn) aggregates. The objective of this study was to investigate if β-synuclein (β-syn) overexpression could have beneficial effects by inhibiting the aggregation of α-syn. The M83 transgenic mouse is a model of synucleinopathy, which develops severe motor symptoms associated with aggregation of α-syn. M83 neonate or adult mice were injected with adeno-associated virus vectors carrying the human β-syn gene (AAVβ-syn) or green fluorescent protein gene (AAVGFP) using different injection sites. The M83 disease was - or not - accelerated using extracts of M83 brains injected with brain extract from mouse (M83) or human (MSA) origins. AAV vectors expression was confirmed using Western blot and ELISA technics. AAV mediated β-syn overexpression did not delay the disease onset or reduce the α-syn phosphorylated at serine 129 levels detected by ELISA, regardless of the AAV injection route and the inoculation of brain extracts. Instead, a proteinase-K resistant β-syn staining was detected by immunohistochemistry, specifically in sick M83 mice overexpressing β-syn after inoculation of AAVβ-syn. This study indicated for the first time that viral vector-mediated β-syn overexpression could form aggregates in a model of synucleinopathy.
Similar content being viewed by others
Introduction
Parkinson’s disease (PD), dementia with Lewy bodies (DLB) and multiple system atrophy (MSA) are synucleinopathies, characterized by inclusions mainly composed of an aggregated form of α-synuclein (α-syn) in the central nervous system (CNS). As for prions, aggregated forms of α-syn propagate within the CNS during the associated neuro-degenerative diseases. This characteristic was initially suggested in humans by Braak’s description of PD stages, and was confirmed later in experimental models of synucleinopathies, in particular in the M83 transgenic mouse model1. M83 mice express the human A53T mutated α-syn found in some familial PD forms, under the control of the mouse prion promoter1. These mice spontaneously develop severe motor impairment at 8–16 months of age at the homozygous state. The symptomatology is associated with the accumulation in the CNS, of a pathological form of α-syn (α-synP), heavily phosphorylated at serine 129 residue. We previously showed that, in M83 mice, disease onset can be accelerated by the intracerebral inoculation of brain homogenates from sick M83 mice2. This model was further characterized by development of an original ELISA that specifically detects, and allows to easily quantify, the pathological α-syn in sick M83 mice3,4,5. This in vivo model may be useful for testing novel therapeutic strategies, particularly targeting progression of the α-syn aggregation.
β-synuclein (β-syn) is another member of the synuclein family, lacking a part of the non-amyloid component, a specific region suggested to be amyloidogenic in α-syn. According to in vitro studies, unlike α-syn, β-syn alone is not able to form aggregates, but could instead interact with α-syn and reduce its capacity to aggregate6. These anti-aggregative features have been investigated in vivo, and some of these studies indeed reported a neuroprotective effect and reduction of α-syn inclusions after overexpression of the human β-syn mediated by DNA microinjection or lentiviral vectors in the transgenic D mouse model, overexpressing human wild-type α-syn7,8. Another study also suggested that crossing mice overexpressing human β-syn with M83 mice delayed the M83 disease onset and reduced the α-syn aggregation9. However, two recent studies by Taschenberger et al. and Landeck et al. which analyzed the impact of human β-syn overexpression mediated by adeno-associated viral vectors (AAV) on nigral dopaminergic neurons in rats, described a neurodegeneration with β-syn aggregates10,11.
Here, in order to assess the effects of β-syn overexpression in the context of these recent and unexpected data suggesting that β-syn produced using AAV may be able to aggregate, we investigated the impact of human β-syn overexpression mediated by AAV on the onset of the synucleinopathy of M83 mice. Two strategies of inoculation of the AAV vector were sequentially tested: (i) an intracerebroventricular (ICV) inoculation at birth, in order to generate a widespread overexpression of the vector in the central nervous system12, and (ii) an inoculation of the AAV vector into the ventral tegmental area (VTA)13, a region of the mesencephalon in which prominent α-syn aggregation occurs in sick M83 mice1. Our results indicate that, regardless of the AAV inoculation strategy, continuous AAV-mediated β-syn overexpression in neurons did not delay the onset of the disease and did not modify the α-synP levels measured by ELISA, strongly suggesting that β-syn may not protect against α-syn aggregation and propagation.
Results
Human β-syn is widely expressed after intracerebroventricular injection of AAVβ-syn in neonates
To overexpress β-syn in the M83 CNS, we used self-complementary (sc)AAV9 vectors expressing human β-syn (AAVβ-syn) gene or, as a control, the green fluorescent protein (AAVGFP) gene under the control of the human synapsin1 promoter and with a post-transcriptional WPRE regulatory sequence (Supplementary Fig. 1A).
To functionally evaluate the AAV vectors, we first inoculated AAVβ-syn in the right and left cerebral ventricles of wild-type B6C3H newborn mice (genetic background of M83 transgenic mice) to obtain a widespread expression of the transgene in the CNS12. One month later, using viral specific primers, vector mRNA was detected in all regions of the brain and in the spinal cord (Supplementary Fig. 1B), as described14, with higher levels in rostral regions of the brain (olfactory bulbs, cortex, hippocampus, striatum) and mesencephalon.
Intracerebroventricular injection of AAVβ-syn did not modify the disease of M83 mice
AAV vectors were first tested in M83 mice challenged by inoculation of brain extracts from sick M83 mice, an experimental design which is characterized by a quicker onset of the disease with a lower intragroup variability as compared to unchallenged M83 mice2. AAV vectors were ICV inoculated at birth and two months later challenged with different brain extracts of sick M83 mice, one derived from a second passage of a sick M83 brain extract in M83 mice (M83/M83 inoculum), and the other from a second passage of a human brain extract from a MSA patient in M83 mice (MSA/M83 inoculum) (Fig. 1A, Supplementary Table 1)5. M83 mice were euthanized after the detection of the first symptoms of disease, i.e balance disorders or hind limb paralysis, identified by two independent observers.
Impact of the intracerebroventricular inoculation of AAV on the accelerated disease of M83 mice. (A) M83 mice were injected at birth (Week 0) with AAVβ-syn or AAVGFP vectors per lateral ventricle and challenged two months later (Week 8) by the injection of M83/M83 (B–D) or MSA/M83 (E–G) inoculum in the striatum to accelerate the disease. After the appearance of the first specific symptoms of M83 disease (from 3 months to 8 months after challenge), mice were sacrificed and dissected in order to perform biochemical analysis. (B) M83 disease-associated survival after the inoculation of the M83/M83 brain extract is represented (significant difference according to log rank test, p < 0.05). (C,D) Quantification of total β-syn (mouse β-syn and human β-syn produced by the vector) (C) or of pathological Ser129-phosphorylated form of α-syn (α-synP) (D) by ELISA in CNS regions of the same sick M83 mice groups (n = 6 for the treated and the control group). (E) M83 disease-associated survival after the inoculation of the MSA/M83 brain extract (no significant difference according to log-rank test). (F,G) Quantification of total β-syn (F) or α-synP (G) by ELISA in CNS regions of the same sick M83 mice groups (n = 6 for the treated group and n = 5 for the control group). Hi: hippocampus, Mes: mesencephalon, BS: brain stem, CSC: cervical spinal cord, TSC: thoracic spinal cord, LSC: lumbar spinal cord. Data are shown as mean ± sd. *p < 0,05; **p < 0,01; according to Wilcoxon test.
In both experiments, no delay in the onset of the disease was observed in AAVβ-syn injected mice as compared to control AAVGFP mice, regardless of the type of inoculum (M83/M83 or MSA/M83). Instead, AAVβ-syn injection significantly accelerated the onset of the disease in animals challenged with the M83/M83 extract as compared to control mice (log rank test p < 0,05) (Fig. 1B). In contrast, no difference was observed in animals challenged with the MSA/M83 extract (Fig. 1E). In each experiment, we did not detect any difference concerning the clinical symptoms of sick mice after the injection of AAVβ-syn or AAVGFP. After dissection of the brains, biochemical analyses were realized. As the vector expression was higher in the rostral regions of the brain than in other CNS regions after ICV injections, we first checked the vector expression by detecting total β-syn in the hippocampus. An ELISA test adapted from Krassnig et al. allowed to detect a significant overexpression of β-syn (both murine β-syn and human β-syn produced by the vector) in the hippocampus, confirming the continuous expression of the AAVβ-syn until the disease onset (Fig. 1C,F). We next focused on the mesencephalon, the brainstem and the spinal cord because these CNS regions are known to be strongly positive in α-synP in sick M83 mice3,4,5. A significant overexpression of β-syn was only detected in the mesencephalon, the brain stem and in the spinal cord of sick M83 mice injected with AAVβ-syn challenged with M83/M83 inoculum, but not in mice challenged with the MSA/M83 inoculum (Fig. 1C,F). Since the M83 disease is associated with moderate or high α-synP levels into different brain regions and in the spinal cord5, we quantified α-synP using ELISA in order to detect a potential effect of β-syn on this biochemical marker of the disease. Our analyses did not show any significant difference in the α-synP levels in any of the examined CNS regions after AAVβ-syn inoculation, regardless of the nature of the inoculum used to accelerate the disease (Fig. 1D,G).
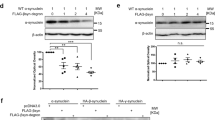
To further study the levels of β-syn protein after AAV injections in M83 mice throughout the CNS, we used Western blot and ELISA which allowed to detect total β-syn in specific brain regions. Comparing total β-syn levels of 5 to 7 months old sick M83 mice inoculated at birth with AAVβ-syn or AAV GFP then challenged with the MSA/M83 inoculum (same mice as in Fig. 1E), an overexpression of β-syn protein was detected by ELISA in the olfactory bulbs, cortex, striatum, hippocampus, but not in the other regions (Fig. 2A), consistent with the results obtained by qRT-PCR at one month after AAV inoculation (Supplementary Fig. 1B). Western blot analyses confirmed an overexpression of β-syn protein in the hippocampus of the same mice after AAVβ-syn inoculation (Fig. 2B,C). Most of the animals inoculated with AAVβ-syn showed a β-syn overexpression detectable until the disease onset (Fig. 1C,F). Only a few animals did not show significant overexpression of β-syn detected by ELISA or by immunohistochemistry (5/48 animals, in the entire study); these animals were excluded for the study analyzing the onset of disease.
Characterization of the AAV vectors expression after their intracerebroventricular inoculation in M83 mice. M83 mice were injected with AAV vectors in the ventricles at birth and challenged with MSA/M83 inoculum (same mice than in Fig. 1E–G). (A) Detection of total β-syn synthesis by ELISA in cerebral regions and spinal cord of sick M83 mice 5 to 7 months after ICV injection of AAV. (B) Detection of total β-syn or GFP protein in the hippocampus of the same M83 mice by Western blot. Recombinant β-syn was loaded as a positive control. Numbers refer to each mouse (n = 6 in the treated group and 5 in the control group). Full blots are shown in Supplementary Fig. 10. (C) Quantification of total β-syn levels detected by WB. OB: olfactory bulbs, Cx: cerebral cortex, Str: striatum, Hi: hippocampus, Mes: mesencephalon, BS: brain stem, CSC: cervical spinal cord, TSC: thoracic spinal cord, LSC: lumbar spinal cord. Data are shown as means ± sd. *p < 0,05; **p < 0,01 according to Wilcoxon test.
We next examined the possible effects of AAVβ-syn ICV injection on the spontaneous development of the M83 disease during aging, without any challenge to accelerate the disease (Fig. 3A). Here again, AAVβ-syn injection neither delayed the disease onset of M83 mice nor modified the α-synP levels (Fig. 3B,D), despite a sustained expression of the transgene, at least in the hippocampus, up to the disease onset (Fig. 3C) (ELISA data correspond to 2 mice for the treated group and 3 mice for the control group; consequently, no statistical analysis was done here).
Impact of the intracerebroventricular inoculation of AAVβ-syn or AAVGFP on the spontaneous M83 disease. (A) M83 mice were injected at birth (Week 0) with AAVβ-syn or AAVGFP vectors per lateral ventricle. After the appearance of the first specific symptoms of M83 disease (from 11 months to 20 months of age), mice were sacrificed and dissected in order to perform biochemical analysis. (B) M83 disease-associated survival after the intracerebroventricular (ICV) inoculation of AAV vectors at birth (no significant difference according to log-rank test). (C,D) Quantification of total β-syn (B) or α-synP (C) by ELISA in CNS regions of the same sick M83 mice. No statistical analysis was done for ELISA results because the number of mice analyzed was too small (2 mice for the treated group and 3 mice for the control group). Hi: hippocampus, Mes: mesencephalon, BS: brain stem, CSC: cervical spinal cord, TSC: thoracic spinal cord, LSC: lumbar spinal cord. Data are shown as means ± sd (n = 2 and 3).
Altogether, these results indicated that AAVβ-syn injection in the ventricles resulted in a widespread expression of the transgene in the CNS, but at a moderate level, especially in the structures that are the most heavily affected by the synucleinopathy lesions. We thus considered another strategy by inoculating the AAV vectors in the ventral tegmental area (VTA), which is located into the mesencephalon, where major brain lesions and accumulation of α-synP are detected in sick M83 mice and which is also connected to multiple brain areas13.
β-syn is overexpressed mostly in the mesencephalon after injection of AAVβ-syn in the VTA of adult mice
In order to validate this injection protocol of AAV vectors into the VTA, two months old wild-type mice were first inoculated with AAVβ-syn in the VTA and sacrificed one month later. After dissection of the brains, vector mRNA was mostly detected in the mesencephalon, but also in the cortex, striatum, hippocampus and brainstem; only traces of vector mRNA were identified in the other regions of the brain (except the olfactory bulbs) and in the spinal cord, as compared to mesencephalon (Supplementary Fig. 1C).
Injection of AAVβ-syn in the VTA did not modify the disease of M83 mice
The same protocol was then applied to M83 mice. After the inoculation of AAV in the VTA in two months old M83 mice, the disease was accelerated by intracerebrally inoculating the mice one month later with the M83/M83 or the MSA/M83 inoculum (Fig. 4A). No significant difference in the survival was observed between the AAVβ-syn and AAVGFP injected mice, between 3 and 8 months (Fig. 4B,E). As before, we did not detect any difference concerning the clinical symptoms of sick mice after the injection of AAVβ-syn or AAVGFP. Importantly, analyses by ELISA confirmed the strong overexpression of β-syn in the mesencephalon although no difference was detected in the levels of pathological α-synP at the disease stage (Fig. 4C–G).
Impact of the inoculation of AAV in the ventral tegmental area on M83 disease. (A) Two months old M83 mice (Week 8) were injected with AAVβ-syn or AAVGFP vectors in the ventral tegmental area (VTA) and challenged one month later (Week 12) by the injection of M83/M83 (B–D) or MSA/M83 (E–G) inoculum in the striatum to accelerate the disease. As before, after the appearance of the first specific symptoms of M83 disease (from 3 months to 8 months after challenge), mice were sacrificed and the CNS was dissected in order to realize biochemical analysis and immunohistochemistry /immunofluorescence studies (Fig. 4H, Supplementary Figs 2–4). (B) M83 disease-associated survival after the inoculation of the brain extract (no significant difference according to log-rank test). (C,D) Quantification of total β-syn (C) or pathological Ser129-phosphorylated form of α-syn (α-synP) (D) by ELISA in CNS regions of the same sick M83 mice groups (n = 5 for the treated group, n = 5 for the control group). (E) The M83 disease-associated survival after the inoculation of the MSA/M83 inoculum (no significant difference according to log-rank test). (F, G) Quantification of total β-syn (F) or α-synP (G) by ELISA in CNS regions of the same mice groups (n = 5 for the treated group, n = 5 for the control group). (H) Immunohistochemistry pictures showing total β-syn staining (mouse β-syn and human β-syn produced by the vector) in the mesencephalon of 7 months old sick M83 mice inoculated with AAV β-syn or AAV GFP and challenged with MSA/M83 inoculum. Mes: mesencephalon, BS: brain stem, CSC: cervical spinal cord, TSC: thoracic spinal cord, LSC: lumbar spinal cord. Data are shown as means ± sd. *p < 0,05; **p < 0,01 according to Wilcoxon test. Scale bar 100 µm.
By immunohistochemistry, using an antibody targeting total β-syn (human and murine β-syn), a specific β-syn staining punctate pattern was detected in the inoculated mesencephalon, as well as in the striatum and brain stem which represent connected regions13, of all the sick M83 mice inoculated with AAVβ-syn (5/5), but not in sick M83 mice inoculated with AAVGFP (0/2) (Fig. 4H). Further analysis indicated that these β-syn immunoreactive dots, which are specifically detected in mice inoculated with AAVβ-syn, mostly co-localize with the presynaptic protein synaptophysin (Supplementary Fig. 2A). Co-localization of GFP with neuronal marker β-tubulin type 3 in sick M83 mice inoculated with AAVGFP also confirmed that these vectors allowed a neuronal specific expression of the transgene (Supplementary Fig. 2B). Interestingly, the morphology of the inclusions containing α-syn phosphorylated at serine 129 detected by immunohistochemistry was not modified by β-syn overexpression (Supplementary Fig. 3A). Furthermore, these inclusions only rarely co-localized with AAV-specific β-syn staining by immunofluorescence (Supplementary Fig. 3B). We further biochemically characterized vectors expression after injection in the VTA in M83 mice euthanized 4 months later. Total β-syn levels were quantified by ELISA and showed a significant increase of the protein in the mesencephalon of mice injected with AAVβ-syn as well as in the hippocampus and brain stem compared to control mice injected with the AAVGFP (Fig. 5A). β-syn protein overexpression was also readily detected by Western blot in the mesencephalon of most of these mice (Fig. 5B).
Characterization and impact of inoculation of AAV in the ventral tegmental area of M83 mice. (A,B) Two months old M83 mice were injected with AAVβ-syn or AAVGFP vectors in the VTA and challenged one month later by the injection of M83/M83 inoculum in the striatum to accelerate the disease. In this experiment, all mice were euthanized 3 months after the challenge for biochemical analysis. (A) Detection of the total β-syn by ELISA in the brain and the spinal cord of M83 mice after inoculation of AAVβ-syn or AAVGFP in the VTA (n = 4 and 6, respectively). (B) Detection of β-syn and GFP proteins by Western blot in the mesencephalon of the same sick M83 mice. Recombinant β-syn was loaded as a positive control. Numbers refer to mice. Full blots are shown in Supplementary Fig. 11. (C) Total β-syn protein quantification by ELISA test using 0,2 µg (instead of 2 µg) of mesencephalon homogenates extracted from sick M83 mice inoculated with 1,5* 109 vg (high dose, 4-times more than the low dose) or 3,75* 108 vg (low dose) of AAVβ-syn or AAVGFP. These results were obtained with the same samples used in Figs 4C and 5D, but 10 times diluted. (D–F) Two months old M83 mice (Week 8) were injected with high dose of AAV vectors (4-times higher than in Fig. 4 for each vector) in the VTA and challenged one month later (Week 12) by injecting M83/M83 inoculum in the striatum. (D,F) Quantification of total β-syn (D) or α-synP (F) by ELISA in CNS regions of the same sick M83 mice groups (n = 3 and 7 for the treated and control group). (E) M83 disease-associated survival after the inoculation of the brain extract (significant difference according to log-rank test, p < 0,001). Mes: mesencephalon, BS: brain stem, CSC: cervical spinal cord, TSC: thoracic spinal cord, LSC: lumbar spinal cord. Data are shown as means ± sd. *p < 0,05, **p < 0,01, according to Wilcoxon test. Scale bar 100 µm.
The absence of protective effect was not due to an insufficient dose of AAV vector injected in the VTA
We finally asked whether the absence of any significant effect of AAVβ-syn could be due to an insufficient dose of AAV vector. We thus injected 4-times more vector in the VTA one month before the challenge by intra-cerebral inoculation of the M83/M83 inoculum. This AAV dose allowed to significantly increase the overexpression of β-syn at the disease stage (p < 0,05) (Fig. 5C,D). However, the M83 disease was still not delayed by β-syn overexpression, but instead, as previously observed after ICV injection of AAV (Fig. 1B), the disease appeared significantly earlier in the AAVβ-syn injected mice as compared to control AAVGFP animals (p < 0,001) (Fig. 5E). As in previous experiments, no impact was found on the α-synP levels even if β-syn was significantly increased at the stage of clinical signs in all the CNS regions (Fig. 5D,F).
By immunohistochemistry, as with the low dose of AAV, a specific β-syn staining punctate pattern was detected in the inoculated mesencephalon, as well as in the striatum and brain stem in two sick M83 mice inoculated with AAVβ-syn, but not in one mouse inoculated with AAVGFP (data not shown).
AAV-mediated overexpression of β-syn might have formed aggregates of β-syn
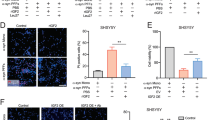
Proteinase K (PK)-resistant β-syn aggregates have been previously described after inoculation of AAV vectors expressing human β-syn in the substantia nigra of rats10,11. We thus analyzed by immunohistochemistry the brain of several sick M83 mice after inoculation of AAVs in the VTA (Fig. 6). After PK digestion, total β-syn antibody revealed a punctate pattern specifically detected after the inoculation of AAVβ-syn, in the mesencephalon of 7/7 sick M83 mice (5 mice injected with low dose of AAV, of which 2 were challenged with M83/M83 inoculum and 3 with MSA/M83 inoculum, as well as 2 mice injected with high dose of AAV and challenged with M83/M83 inoculum). This punctate pattern was also detected in the striatum and the brain stem where the AAV is also expressed, but not in the cerebellum where the AAV is less expressed (Supplementary Fig. 4). A few cell bodies were also stained with total β-syn antibody after PK digestion, particularly in the mesencephalon of these mice (data not shown). Even if diffuse, PK resistant staining was also detected in the hippocampal region of two sick M83 mice injected with AAVβ-syn and not challenged with brain extract inoculation, but not in one sick M83 control mouse (data not shown). These results confirm that overexpression of β-syn resulted in PK-resistant β-syn staining which could be detected independently of the vector dose, the inoculation site or the age of inoculation. Interestingly, Western blot detection of β-syn in the brain homogenates from sick M83 mice injected with high dose of AAVβ-syn showed the appearance of an additional band, consistent with a dimer, that is not visible in the homogenates from sick M83 mice injected or not with AAVGFP (even after long exposure of the blot) (Supplementary Fig. 5).
Proteinase K (PK)-resistant β-syn staining in sick M83 mice inoculated with AAV β-syn. (A–D) Immunoreactivity to total β-syn antibody in mesencephalon sections of two sick M83 mice inoculated with low dose of AAVβ-syn or AAVGFP in the VTA and challenged by MSA/M83 inoculum (mice from the study Fig. 4E–G) is shown without PK digestion (A,B) or after PK digestion (C,D). After PK digestion, these β-syn immunoreactive dots were detected in all mice analyzed by immunohistochemistry inoculated with the AAVβ-syn in the VTA (7/7 mice, comprising 2 mice challenged with M83/M83 inoculum, 3 mice challenged with MSA/M83 inoculum, and also in 2 mice inoculated with high dose of AAV and challenged with M83/M83 inoculum), but not in sick M83 mice inoculated with AAVGFP (3/3, comprising 2 mice challenged with MSA/M83 inoculum and 1 mouse challenged with M83/M83 inoculum). Scale bars 100 µm (low magnification) and 25 µm (high magnification).
Discussion
β-syn was previously described as a neuroprotective protein, able to inhibit the aggregation process of α-syn in vitro and having benefic effects on synucleinopathies in vivo6,7,8,9,15. Here, we tried to slow down a synucleinopathy in a transgenic mouse model with dramatic clinical symptoms linked to pathological α-syn aggregation. In particular, we analyzed the effects of the overexpression of β-syn mediated by an AAV vector on α-syn aggregation. We show here that the delivery of the AAVβ-syn vector via two different injection routes (intracerebroventricular or in the ventral tegmental area) in transgenic M83 mice, neither delayed the disease onset nor reduced the levels of pathological α-syn at the disease stage, despite a sustained overexpression of β-syn. A limitation of our study remains the low number of animals that were followed in each of the experimental groups, given the multiple strategies that were studied (different routes and ages of AAV inoculations, different AAV doses, AAV effects during normal aging or after intracerebral experimental challenges of two M83 sources (M83/M83 or MSA/M83) as summarized in Supplementary Table 1. This represents a statistical limitation of our study, although it is noteworthy that in the only two experimental groups in which a statistically significant difference was found between the survival of mice injected with AAVβ-syn and AAVGFP vectors, this rather suggested a deleterious effect of β-syn overexpression. Studies on larger cohorts will thus be required to confirm our results. A previous study indeed suggested that constitutive overexpression of β-syn in transgenic M83 mice by generating double transgenic mice delays the M83 disease onset by several months9. In our study, interestingly, M83 mice overexpressing β-syn showed a higher spontaneous activity in experiments in which open field was performed and a tendency to have more balance trouble in beam walking test (Supplementary Fig. 6). These results suggest that β-syn expression might have an impact on mice behavior without affecting the ultimate development of the disease. Importantly, in this study of Fan and colleagues, human β-syn was expressed from the pan-neuronal mouse prion promoter, which is the same promoter used to express human mutated α-syn in the M83 model. It is possible that the strategies used in our study did not provide sufficient expression of β-syn, in the CNS regions targeted by the disease. Indeed, as expected, after the injection of AAV in the VTA, we obtained a very high overexpression of β-syn in the mesencephalon in which the aggregation of α-syn is high at the disease stage, but this region may not be the region where the aggregation process of α-syn begins16. It should be emphasized that the major signs of the clinical disease of M83 mice are in relation with some - still poorly explained- disorders in the spinal cord17. In an additional series of experiments, we were unable to identify a degeneration of motor neurons in the lumbar spinal cord after the injection of preformed fibrils of human α-syn in M83 mice, as previously reported after intra-muscular injections17, further interrogating the cause of the appearance of symptoms (Supplementary Fig. 7). The ICV injection of AAV9 vector in SMNΔ7 mice, a severe model of spinal muscular atrophy, allowed to express a codon-optimized human SMN1 coding sequence in the spinal cord, which as a result, has improved significantly their survival18. After the ICV inoculation of AAV β-syn, the overexpression of β-syn was diffuse and lower in caudal regions of the brain and spinal cord, as described14, but it may have been insufficient to slow down the synucleinopathy process. It must however be noted that an in vitro study using AAV vectors suggests that β-syn overexpression is benefic on α-syn linked toxicity only at a low ratio of β-syn/α-syn expression10. Even if it was not evaluated here, another study has described transgene expression in the neuromuscular junction after AAV injection into the ventricles at birth; this may be important since the degeneration of the neuromuscular junction was also described as a possible explanation of the appearance of symptoms in M83 mice14,17. At the cellular level, human β-syn seems mostly localized in the presynaptic button and axons in sick M83 mice, which seems appropriate because the aggregation of α-syn was suggested to start at the presynaptic level in synucleinopathies19,20. Another study suggested that the benefic effect of β-syn is the result of the down regulation of the expression of α-syn, but we did not detect any decrease of α-syn expression, even in the brain regions overexpressing β-syn (data not shown). In another transgenic mouse model (D line) overexpressing human wild type α-syn, two studies reported the reduction of the synucleinopathy by either generating bigenic mice that also overexpress β-syn, or vehiculating the human β-syn transgene, under the control of cytomegalovirus (CMV) promoter, in a lentiviral vector7,8. As some reports indicate a downregulation of the CMV promoter in the CNS over time21, we chose here to use the human synapsin 1 promoter to obtain a long term expression of β-syn, which was confirmed in the present study, even in old mice. Importantly, as previously described22, the synapsin 1 promoter also allowed to express human β-syn specifically in neurons, where α-syn aggregates are found in the M83 model1. According to the study by Hashimoto et al., the neuroprotective effect of β-syn was associated to the activation of Akt, a neuroprotective protein which could be indirectly down-regulated by α-syn during the synucleinopathy process23. However, we did not detect any significant increase of Akt activation by Western blot in 6 months-old M83 mice inoculated with AAVβ-syn in the VTA at 2 months, and challenged one month later with the M83/M83 inoculum (Supplementary Fig. 8). It should be noticed that such Akt changes were not consistently observed, as shown in a study with a lentiviral vector used for β-syn overexpression24.
Surprisingly, in the present study, two experiments in mice challenged with a M83/M83 inoculum rather showed a significant acceleration of the disease after AAV β-syn injection (Figs 1B and 5E). Interestingly, several studies reported that β-syn could be implicated in the pathological process of synucleinopathies. Indeed, an axonal pathology with β-syn accumulation in axons was reported in patients with Parkinson’s disease, DLB or Hallervorden-Spatz syndrome25,26. In addition, two mutations of β-syn, identified in DLB cases, were suggested to be responsible for α-syn aggregation and DLB27. Generating transgenic mice co-expressing P123H mutated β-syn with human α-syn resulted in an enhanced pathology, suggesting that mutated β-syn could potentiate α-syn aggregation28. Variable results were also observed after overexpression of β-syn using a lentiviral vector after inoculation into the hippocampus of transgenic mice overexpressing a mutated form of the human β-amyloid precursor protein (APP) involved in Alzheimer’s disease, as assessed by examining plaque load, memory deficits, and anxiety24. However, it should be noted that the expression level of β-syn was relatively low in this latter study (not detectable at the protein level). Also, it is interesting to notice that a truncated form of human recombinant α-syn lacking the 71–82 residues not able to form aggregates in vitro, like β-syn29, accelerated the disease of a few M83 mice, after injection in the muscle or in the peritoneal cavity17,30. These results raise the possibility that β-syn might be able to accelerate the aggregation of α-syn in M83 mice, although this has not been reported so far.
Further analyzing the effects of β-syn expression in sick M83 mice inoculated with the AAV vector in the VTA allowed detecting a punctate pattern of β-syn staining in the targeted brain region and some connected regions, only in mice inoculated with AAVβ-syn. This staining was more resistant to PK digestion than that endogenous β-syn in control mice. This result suggests that overexpressed β-syn formed aggregates which could explain why β-syn did not act as expected, i.e as an α-syn aggregation inhibitor. Landeck et al. have described the same staining of β-syn resistant to PK digestion referred as “dark punctae”, two months after the inoculation of AAV5 carrying human β-syn under the control of chicken beta-actin promoter including a CMV enhancer element (CBA) in the substantia nigra of rats11. Before this study, Taschenberger et al. have also described the detection of PK-resistant β-syn aggregates in vivo, as early as two weeks following the inoculation of AAV2 carrying human β-syn gene in the substantia nigra of rats10. This time, the vector design was closer to ours, using the same promoter (human synapsin) to express human β-syn, with a WPRE sequence to enhance transgene expression. It is interesting to notice that, even if the AAV serotype used as well as the vector design can differ between studies, all these studies that used AAV to carry β-syn also reported the detection of β-syn aggregates. Furthermore, our observations in immunohistochemistry are supported biochemically in the present study, by the detection of a specific β-syn pattern in Western blot that could be a specific signature of β-syn aggregation, in mice showing overexpression of β-syn after injection of high dose of AAVβ-syn (Supplementary Fig. 5). All these observations suggest that the neuroprotective effect of β-syn may vary according to the strategy followed to overexpress it and the animal model. For example, it would be interesting to inject AAV vectors used in this study in transgenic D mice in order to see if we obtain the same β-syn aggregates. The D line is indeed an example of transgenic mice expressing the normal human α-syn, which may thus be more representative of the common situation in humans. The interest of the M83 mouse line, which is well illustrated in our study, is the possibility to easily quantify both survival of mice, ultimately showing a major clinical disease, and levels of pathological α-syn, using an ELISA test. The A53T mutation in the human protein is however associated with a high propensity to aggregation31 and our observations with AAV-mediated β-syn overexpression may not be representative of the situation of humans with non genetic synucleinopathies. Also, it is important to point out that even if most of neurons express the transgene in the cerebral region injected of mice in our study, there is an important variability of the expression of the transgene in transduced cells after AAV vectors injection (Supplementary Fig. 9). It might explain at least partly the difficulty to detect a benefic effect of β-syn overexpression as it was suggested to depend on a specific α-syn/β-syn ratio in cell culture10.
The present study suggest for the first time that β-syn overexpression could not be benefic in a synucleinopathy model. This may be due at least partly to the formation of a PK-resistant β-syn species after AAVβ-syn inoculation. According to a very recent study, a mildly acidic pH environment, found in several organelles, could induce β-syn aggregation, highlighting the complexity of β-syn fibrillation mechanisms occurring in vivo32. Further studies are needed to better understand how and to what extent β-syn could play a role in synucleinopathies.
However, even though we could not detect any protective effects of β-syn, our study confirms that an AAV vector is suitable for long-term overexpression of proteins into the CNS. Notably, after ICV inoculations of AAV in neonates, we confirmed a widespread expression of the protein, as this was also recently described using an α-syn AAV vector33. The injection of AAV in the VTA also allowed to express human β-syn in various connected regions, as suggested in a previous study using serotype 9 AAV vector carrying a lysosomal enzyme gene13. Our data also illustrate the robustness of the experimental model of M83 mice intracerebrally challenged by inocula containing aggregated α-syn, showing rather short and relatively uniform survival periods before the appearance of characteristics clinical signs, which can be used for the assessment of future therapeutic strategies.
Methods
Animals
M83 transgenic mice were used in this study (B6;C3H-Tg[SNCA]83Vle/J, RRID:MGI:3603036, The Jackson Laboratory, Bar Harbor, ME, USA). These mice express A53T mutated human α-syn protein and spontaneously develop severe motor impairment leading to early death1. Homozygous M83 mice develop characteristic motor symptoms between 8 and 16 months of life, beginning with reduced ambulation, balance disorders, partial paralysis of a hind leg and then progressing to prostration, difficulty in feeding, weight loss, hunched back and general paralysis1. The animals were housed per group in enriched cages in a temperature-controlled room on a 12 h light/dark cycle, and received water and food ad libitum, in our approved facilities (No. C69 387 0801) for breeding and experimental studies, in accordance with EEC Directive 86/609/EEC and French decree No. 2013–118. The experimental studies described in this article were performed in containment level 3 facilities and authorized by the ≪Comité d’éthique≫ CE2A – 16, ComEth ANSES/ENVA/UPEC and by the ≪Ministère de l’enseignement supérieur, de la recherche et de l’innovation≫ (ref. 16-006). All experiments were performed in accordance with relevant guidelines and regulations.
AAV vectors: Recombinant self-complementary AAV9 vectors (scAAV9) encoding human β-syn or enhanced GFP (eGFP) were produced by calcium phosphate transfection of HEK-293 cells34. Three plasmids were transfected simultaneously: (i) a vector plasmid containing the human gene of β-syn or eGFP under control of the neuron-specific synapsin 1 gene promoter (Supplementary Fig. 1A) (ii) a helper plasmid pXX635 and (iii) a plasmid carrying rep2 and cap9 genes36. Vector particles were extracted and purified on an iodixanol step gradient. Titers were determined by quantitative polymerase chain reaction (qPCR) and expressed as viral genomes per milliliter (vg/mL).
Inoculations
AAV vectors were injected at birth or at adulthood (two months of age), at different sites of inoculation. Neonates were inoculated using 5 µL Hamilton syringe, with 9.38* 108 vg of AAVβ-syn or 2.73* 108 vg of AAVGFP vectors per lateral ventricle of the viral solution with 0.05% Trypan blue12. Adults mice were inoculated with 3.75* 108 vg of AAVβ-syn or 1.09* 108 vg of AAVGFP (or with high dose: 1.5* 109 vg of AAVβ-syn or 4.36* 108 vg of AAVGFP) of viral solution in the ventral tegmental area located in the mesencephalon, using stereotaxic coordinates (anteroposterior: −3.16 mediolateral: +0.25 dorsoventral: −4.50). Two months after the inoculation of the viral solution in neonates or one month after the inoculation of the viral solution in adults, M83 mice were inoculated with brain homogenates from a second passage of a sick M83 brain sample in M83 mice (M83/M83 inoculum) or a second passage of a patient with multiple system atrophy (MSA) brain sample in M83 mice (MSA/M83 inoculum) (same sample used in a previous publication5). These homogenates were injected in the left striatum, using stereotaxic coordinates (AP: +0.14 ML: +2 DV: −2.75). Before each stereotaxic surgery (AAV or brain extract inoculation), mice have been anesthetized with a xylazine (10 mg/kg) and ketamine (100 mg/kg) mixture.
qRT-PCR
B6C3H brains were dissected and RNA was extracted from brain regions using RNeasy lipid tissue Minikit (ref. 74804, Qiagen, Courtaboeuf, France). RNA samples were tittered using Nanodrop (Nanodrop 1000, Thermo Fisher scientific, Villebon sur Yvette, France). 500 ng of RNA were reverse-transcripted with Quanta qScript cDNA Super Mix (ref. 95048-100, VWR international, Fontenay-sous-Bois, France) on a Biorad iCycler. After a 1:10 dilution, complementary DNA samples were analyzed by qPCR using primers specific for the WPRE region of viral mRNA, and primers specific for GAPDH mRNA. We used the LC 480 SYBR Green 1 Master kit (ref. 001,352,04887, Roche, Boulogne-Billancourt, France) on a Roche Light Cycler 480 to perform the qPCR and analyzed the results on the LC 480 software.
ELISA
Brains were dissected as described4 and proteins were extracted in high salt buffer (50 mM Tris-HCl, pH 7.5, 750 mM NaCl, 5 mM EDTA, 1 mM DTT, 1% phosphatase and protease inhibitor cocktails), using a mechanical homogenizer (grinding balls, Precellys 24, Bertin Technologies, Montigny-le-Bretonneux, France) to obtain a 10% homogenate (w/v). Each sample was titered using the DCTM protein assay kit (ref. 5000111, Biorad, Marnes-la-Coquette, France).
As already described for α-synP detection3,5, plates were saturated with Superblock T20 (Thermo Scientific, Rockford, IL, USA) for 1 h at 25 °C, under agitation (150 rpm). After 5 washes in PBST, 10 µg of protein (for α-synP detection) diluted in PBST were incubated for 2 h at 25 °C, under agitation (150 rpm). α-synP was detected with a rabbit polyclonal antibody against PSer129 α-syn (ref. ab59264, Cambridge, UK) diluted to 1:3,000 in PBST with 1% bovine serum albumin (BSA); plates were incubated for 1 h at 25 °C under agitation. After 5 washes, anti-rabbit IgG HRP conjugate (ref. 4010-05, SouthernBiotech, Birmingham, AL, USA) was added at 1:2,000 (for α-synP detection). After washing, 100 μL of 3,3′,5,5′-tetramethylbenzidine solution (ref. T0440, Sigma, Saint-Quentin-Fallavier, France) were added to each well and plates were incubated for 15 min with shaking. The reaction was stopped with 100 μL of 1 N HCl, and the absorbance was measured at 450 nm with the microplate reader Model 680 (Clariostar, BMG Labtech, Champigny sur Marne, France).
ELISA allowing total β-syn quantification was adapted based on an ELISA already published24. Briefly, 2 µg or 0.2 µg (for Fig. 5) of brain homogenate was diluted in carbonate-bicarbonate buffer 50 mM (pH 9.6) and incubated at 4 °C overnight. After 5 washes in PBST, plates were saturated with Superblock T20 for 1 h at 25 °C, under agitation (150 rpm). After 5 washes in PBST, plates were incubated with 1:2,000 of an anti-β-syn antibody ab76111 (ref. EP1537Y, Abcam, Cambridge, UK). After 5 washes in PBST, plates were incubated with an anti-rabbit IgG HRP conjugate (ref. 4010-05, SouthernBiotech, Birmingham, AL, USA) at 1:4,000. After washing, immunoreactivity was revealed with the same protocol as for α-syn ELISA.
Western blot
50 µg (for phosphorylated Akt detection) or 10 µg of proteins (for total Akt detection) or 2 µg of proteins (for GFP and β-syn detection) were separated in 12% SDS-polyacrylamide gels and electroblotted onto polyvinylidene fluoride (PvF) membranes 0,45 µm (Bio-Rad). The membranes were washed 3 times in Tris-Buffered Saline (TBS) for 5 min at room temperature (RT) under agitation and were saturated 1 h with 5% BSA in TBS 0.1% Tween20 (TBST). Membranes were incubated with rabbit antibody against phosphorylated Akt at Ser473 (ref. 9271S, Ozyme, Montigny-le-Bretonneux, France) at 1:1,000 or with rabbit antibody against total Akt (ref. 9272, Ozyme, Montigny-le-Bretonneux, France) at 1:1,000 or with anti-β-syn antibody ab76111 (ref. EP1537Y, Abcam, Cambridge, UK) at 1:5,000 or with anti-GFP antibody (ref. ab290, Abcam, Cambridge, UK) at 1:1,000 or with anti-β-actin antibody (ref. ab8226, Abcam, Cambridge, UK) at 1:2000 overnight at 4 °C. After 3 washes, the membranes were incubated for 1 h at RT with anti-rabbit HRP-linked antibody (ref. 7074P2, Ozyme, Montigny-le-Bretonneux, France) for detection of total Akt and phosphorylated Akt at 1:2,000, or with anti-rabbit HRP-linked antibody (ref. 4010-05, SouthernBiotech, Birmingham, AL, USA). The immunocomplexes were revealed with chemiluminescent reagents (Supersignal WestDura, ref. 34076, Pierce, Interchim, MontLuçon, France), and analyzed using the ChemiDoc system (Bio-Rad) and Image Lab software (Bio-Rad) which allowed to quantify the intensity of the bands.
Immunohistochemistry/Immunofluorescence
After dissection, brain samples were fixed in 4% paraformaldehyde and paraffin embedded to be cut into serial 6 µm sections. After deparaffinization, endogeneous peroxidase activity was directly blocked with oxygenated water 3% during 5 minutes at RT. Brain sections were pretreated with a citrate solution (ref. C9999, Sigma, Saint-Quentin-Fallavier, France) with heat antigen retrieval. For the detection of α-syn phosphorylated at serine 129 by immunohistochemistry, we did an additional antigen retrieval step in which sections were treated with a 4 M solution of guanidinium thyocyanate during 20 min. After washing, sections were saturated using a blocking reagent (ref. 11096176001, Roche), 1 h at RT and incubated with antibody against β-syn ab76111 in PBST (ref. EP1537Y, Abcam, Cambridge, UK) diluted at 1:500, anti-synaptophysin antibody SY38 (ref. ab8049, Abcam, Cambridge, UK) diluted at 1:10 or anti-GFP antibody (ref. ab290, Abcam, Cambridge, UK) diluted at 1:500 or anti-tubulin β-3 antibody (ref. MMS-435P, Biolegend, San diego, USA) diluted at 1:100) at 4 °C overnight. For the detection phosphorylated at S129 α-syn we used a rabbit antibody (ref: ab51253, Abcam, Cambridge, GB) diluted at 1:300 in TBST for immunohistochemistry, and a mouse antibody (ref: pSyn#64, FUJIFILM Wako Pure Chemical Corporation, China) diluted at 1:1000 in TBST for immunofluorescence analysis (with the rabbit anti-β-syn antibody, Supplementary Fig. 3), at 4 °C overnight. After another blocking step 30 minutes at RT, for immunohistochemistry, sections were incubated 1 h at RT with anti-rabbit IgG HRP conjugate (ref. 4010-05, SouthernBiotech, Birmingham, AL, USA) diluted at 1:250 in PBST or TBST. Antibody binding was detected using DAB peroxidase substrate (ref. SK-4100, Vector Laboratories, Burlingame, CA USA) intensified with nickel chloride for β-syn or ImmPACT DAB peroxidase (HRP) substrate (ref. SK-4105, Vector Laboratories, Burlingame, CA USA) for phosphorylated α-syn detection. For immunofluorescence, sections were incubated 1 h at RT with anti-rabbit IgG AlexaFluor 488 or 555 (ref. A-11008 and ref. A21428, respectively, Thermofischer scientific, Villebon sur Yvette, France) or with anti-mouse IgG AlexaFluor 488 or 555 (ref. A11001 and A21127 respectively, Thermofischer scientific, Villebon sur Yvette, France) diluted at 1:1000. After washings in PBST then in PBS, sections were treated with an autofluorescence eliminator reagent (ref. 2160, Millipore, Temecula, USA), before mounting.
PK digestion
After being deparaffinized, sections were incubated with a 10 µg/mL solution of proteinase K (ref EU0090-B, Euromedex, Souffelweyersheim, France) diluted in PBS, 10 minutes at RT (protocol adapted from10). Sections were washed three times in water before blocking the endogenous peroxidase activity and pursuing the immunohistochemistry staining protocol.
Statistical analysis
Survival time was defined as the time from birth until death (Fig. 3A, exclusively) or as the time from the inoculation of the brain extract until the appearance of the first specific M83 symptoms and euthanasia of the mouse. The detection of these specific symptoms was done blindly with regard to the initial AAV (β-syn or GFP) treatment. We right-censored mice found dead without M83 disease identification. Survival times were compared using log-rank test. Concerning statistical analysis of ELISA tests results, means were compared using Wilcoxon test. The difference was significant when *p < 0,05, **p < 0,01, ***p < 0,001.
References
Giasson, B. I. et al. Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein. Neuron 34, 521–533, https://doi.org/10.1016/S0896-6273(02)00682-7 (2002).
Mougenot, A. L. et al. Prion-like acceleration of a synucleinopathy in a transgenic mouse model. Neurobiology of aging 33, 2225–2228, https://doi.org/10.1016/j.neurobiolaging.2011.06.022 (2012).
Betemps, D. et al. Alpha-synuclein spreading in M83 mice brain revealed by detection of pathological alpha-synuclein by enhanced ELISA. Acta Neuropathologica Communications 2, 29, https://doi.org/10.1186/2051-5960-2-29 (2014).
Betemps, D. et al. Detection of Disease-associated alpha-synuclein by Enhanced ELISA in the Brain of Transgenic Mice Overexpressing Human A53T Mutated alpha-synuclein. Journal of visualized experiments: JoVE, e52752, https://doi.org/10.3791/52752 (2015).
Sargent, D. et al. ‘Prion-like’ propagation of the synucleinopathy of M83 transgenic mice depends on the mouse genotype and type of inoculum. Journal of neurochemistry 143, 126–135, https://doi.org/10.1111/jnc.14139 (2017).
Janowska, M. K., Wu, K. P. & Baum, J. Unveiling transient protein-protein interactions that modulate inhibition of alpha-synuclein aggregation by beta-synuclein, a pre-synaptic protein that co-localizes with alpha-synuclein. Scientific Reports 5, 15164, https://doi.org/10.1038/srep15164 (2015).
Hashimoto, M. et al. An antiaggregation gene therapy strategy for Lewy body disease utilizing beta-synuclein lentivirus in a transgenic model. Gene therapy 11, 1713–1723, https://doi.org/10.1038/sj.gt.3302349 (2004).
Hashimoto, M., Rockenstein, E., Mante, M., Mallory, M. & Masliah, E. beta-Synuclein inhibits alpha-synuclein aggregation: a possible role as an anti-parkinsonian factor. Neuron 32, 213–223 (2001).
Fan, Y. et al. Beta-synuclein modulates alpha-synuclein neurotoxicity by reducing alpha-synuclein protein expression. Human molecular genetics 15, 3002–3011, https://doi.org/10.1093/hmg/ddl242 (2006).
Taschenberger, G. et al. beta-synuclein aggregates and induces neurodegeneration in dopaminergic neurons. Annals of neurology 74, 109–118, https://doi.org/10.1002/ana.23905 (2013).
Landeck, N., Buck, K. & Kirik, D. Toxic effects of human and rodent variants of alpha-synuclein in vivo. The European journal of neuroscience 45, 536–547, https://doi.org/10.1111/ejn.13493 (2017).
Kim, J. Y., Grunke, S. D., Levites, Y., Golde, T. E. & Jankowsky, J. L. Intracerebroventricular viral injection of the neonatal mouse brain for persistent and widespread neuronal transduction. Journal of visualized experiments: JoVE, 51863, https://doi.org/10.3791/51863 (2014).
Cearley, C. N. & Wolfe, J. H. A single injection of an adeno-associated virus vector into nuclei with divergent connections results in widespread vector distribution in the brain and global correction of a neurogenetic disease. The Journal of neuroscience: the official journal of the Society for Neuroscience 27, 9928–9940, https://doi.org/10.1523/jneurosci.2185-07.2007 (2007).
McLean, J. R. et al. Widespread neuron-specific transgene expression in brain and spinal cord following synapsin promoter-driven AAV9 neonatal intracerebroventricular injection. Neuroscience letters 576, 73–78, https://doi.org/10.1016/j.neulet.2014.05.044 (2014).
Hashimoto, M. et al. Beta-synuclein regulates Akt activity in neuronal cells. A possible mechanism for neuroprotection in Parkinson’s disease. The Journal of biological chemistry 279, 23622–23629, https://doi.org/10.1074/jbc.M313784200 (2004).
Emmer, K. L., Waxman, E. A., Covy, J. P. & Giasson, B. I. E46K human alpha-synuclein transgenic mice develop Lewy-like and tau pathology associated with age-dependent, detrimental motor impairment. The Journal of biological chemistry 286, https://doi.org/10.1074/jbc.M111.247965 (2011).
Sacino, A. N. et al. Intramuscular injection of alpha-synuclein induces CNS alpha-synuclein pathology and a rapid-onset motor phenotype in transgenic mice. Proceedings of the National Academy of Sciences of the United States of America 111, 10732–10737, https://doi.org/10.1073/pnas.1321785111 (2014).
Armbruster, N. et al. Efficacy and biodistribution analysis of intracerebroventricular administration of an optimized scAAV9-SMN1 vector in a mouse model of spinal muscular atrophy. Molecular Therapy. Methods & Clinical. Development 3, 16060, https://doi.org/10.1038/mtm.2016.60 (2016).
Schulz-Schaeffer, W. J. The synaptic pathology of alpha-synuclein aggregation in dementia with Lewy bodies, Parkinson’s disease and Parkinson’s disease dementia. Acta neuropathologica 120, 131–143, https://doi.org/10.1007/s00401-010-0711-0 (2010).
Spinelli, K. J. et al. Presynaptic alpha-synuclein aggregation in a mouse model of Parkinson’s disease. The Journal of neuroscience: the official journal of the Society for Neuroscience 34, 2037–2050, https://doi.org/10.1523/jneurosci.2581-13.2014 (2014).
Gray, S. J. et al. Optimizing promoters for recombinant adeno-associated virus-mediated gene expression in the peripheral and central nervous system using self-complementary vectors. Human gene therapy 22, 1143–1153, https://doi.org/10.1089/hum.2010.245 (2011).
Glover, C. P., Bienemann, A. S., Heywood, D. J., Cosgrave, A. S. & Uney, J. B. Adenoviral-mediated, high-level, cell-specific transgene expression: a SYN1-WPRE cassette mediates increased transgene expression with no loss of neuron specificity. Molecular therapy: the journal of the American Society of Gene Therapy 5, 509–516, https://doi.org/10.1006/mthe.2002.0588 (2002).
Chung, J. Y. et al. Direct interaction of alpha-synuclein and AKT regulates IGF-1 signaling: implication of Parkinson disease. Neurosignals 19, 86–96, https://doi.org/10.1159/000325028 (2011).
Krassnig, S. et al. Influence of Lentiviral beta-Synuclein Overexpression in the Hippocampus of a Transgenic Mouse Model of Alzheimer’s Disease on Amyloid Precursor Protein Metabolism and Pathology. Neuro-degenerative diseases 15, 243–257, https://doi.org/10.1159/000430952 (2015).
Galvin, J. E., Giasson, B., Hurtig, H. I., Lee, V. M. & Trojanowski, J. Q. Neurodegeneration with brain iron accumulation, type 1 is characterized by alpha-, beta-, and gamma-synuclein neuropathology. The American journal of pathology 157, 361–368 (2000).
Galvin, J. E., Uryu, K., Lee, V. M. & Trojanowski, J. Q. Axon pathology in Parkinson’s disease and Lewy body dementia hippocampus contains alpha-, beta-, and gamma-synuclein. Proceedings of the National Academy of Sciences of the United States of America 96, 13450–13455 (1999).
Ohtake, H. et al. Beta-synuclein gene alterations in dementia with Lewy bodies. Neurology 63, 805–811 (2004).
Fujita, M. et al. A beta-synuclein mutation linked to dementia produces neurodegeneration when expressed in mouse brain. Nature communications 1, 110, https://doi.org/10.1038/ncomms1101 (2010).
Giasson, B. I., Murray, I. V., Trojanowski, J. Q. & Lee, V. M. A hydrophobic stretch of 12 amino acid residues in the middle of alpha-synuclein is essential for filament assembly. The Journal of biological chemistry 276, 2380–2386, https://doi.org/10.1074/jbc.M008919200 (2001).
Ayers, J. I. et al. Robust Central Nervous System Pathology in Transgenic Mice following Peripheral Injection of alpha-Synuclein Fibrils. Journal of virology 91, e02095–02016, https://doi.org/10.1128/jvi.02095-16 (2017).
Flagmeier, P. et al. Mutations associated with familial Parkinson’s disease alter the initiation and amplification steps of α-synuclein aggregation. Proceedings of the National Academy of Sciences of the United States of America 113, 10328–10333, https://doi.org/10.1073/pnas.1604645113 (2016).
Moriarty, G. M. et al. A pH-dependent switch promotes beta-synuclein fibril formation via glutamate residues. The Journal of biological chemistry 292, 16368–16379, https://doi.org/10.1074/jbc.M117.780528 (2017).
Delenclos, M. et al. Neonatal AAV delivery of alpha-synuclein induces pathology in the adult mouse brain. Acta Neuropathologica Communications 5, 51, https://doi.org/10.1186/s40478-017-0455-3 (2017).
Gao, G. P. et al. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proceedings of the National Academy of Sciences of the United States of America 99, 11854–11859, https://doi.org/10.1073/pnas.182412299 (2002).
Xiao, X., Li, J. & Samulski, R. J. Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus. Journal of virology 72, 2224–2232 (1998).
Gao, G. et al. Clades of Adeno-associated viruses are widely disseminated in human tissues. Journal of virology 78, 6381–6388, https://doi.org/10.1128/jvi.78.12.6381-6388.2004 (2004).
Acknowledgements
We are grateful to Eric Morignat and Habiba Tlili for their help and advices on statistical and immunofluorescence assays, respectively. We also are grateful for Olivier Biondi for its help setting up behavioral test and the method for quantifying motor neurons in the spinal cord. We thank Ronald Melki and its team for providing us preformed fibrils of recombinant α-syn. This research was partly funded by the Fondation France Parkinson. D.S was supported by funds from the Région Auvergne-Rhône-Alpes – ARC1 Santé.
Author information
Authors and Affiliations
Contributions
D.S., D.B., M.D., D.G., performed experiments. J.V., J.N.A. contributed to biochemical and immunohistochemistry assays respectively. L.L. supervised animal experiments in containment level 3 facilities. A.S. supervised all the construction and the production of viral vectors. T.B., D.B. designed experiments and supervised the study. D.S., T.B., D.B., A.S. wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sargent, D., Bétemps, D., Drouyer, M. et al. Investigating the neuroprotective effect of AAV-mediated β-synuclein overexpression in a transgenic model of synucleinopathy. Sci Rep 8, 17563 (2018). https://doi.org/10.1038/s41598-018-35825-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35825-2
Keywords
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.