Abstract
The conversion of monoculture rubber (Hevea brasiliensis) plantations into rubber-based agroforestry systems has become a common trend in forestry management in the past few decades. Rubber–Flemingia macrophylla (a leguminous shrub) systems are popular in southwestern China’s Xishuangbanna region. The biogeochemical cycles of soil carbon and nitrogen in forests are mainly affected by their fractions. This study investigated the effect of introducing Flemingia macrophylla to rubber plantations of different ages on soil carbon and nitrogen fractions. The experimental treatments included R1 (young rubber plantation), RF1 (young rubber–Flemingia macrophylla system), R2 (mature rubber plantation) and RF2 (mature rubber–Flemingia macrophylla system). The results showed that the introduction of Flemingia macrophylla to rubber plantations of different ages significantly changed soil carbon and nitrogen fractions, improved soil labile organic carbon and nitrogen contents, and ameliorated soil environments. The average soil microbial biomass organic carbon, nitrogen and nitrate-nitrogen in the 0–10 cm soil layer during the experimental period was 38.9%, 55.5%, and 214.7% higher in RF1 than R1, respectively, and 22.1%, 22.2%, and 652.2% higher in RF2 than R2, respectively. Therefore, Flemingia macrophylla can be used as an alternative interplanted tree species within rubber plantations in similar environments of southeastern Asia.
Similar content being viewed by others
Introduction
Monoculture rubber (Hevea brasiliensis) plantations have rapidly expanded in the last few decades in southeastern Asia1,2. Approximately 90% of global natural rubber production is derived from plantations in this region (http://www.rubberstudy.com), accounted for an estimated 84% of the total global rubber plantation area in 20121,3. Rapid growth in the Chinese economy has increased demand for natural rubber. In response to this demand, the natural tropical forests of southwestern China’s Xishuangbanna region were deforested and replaced with more than 470,000 ha of rubber plantations, which equates to more than 24% of the total land area of the region4. The expansion of these rubber plantations has led to water loss and soil erosion5,6, environmental degradation3,7, and threatened environmental biodiversity8. At present, rubber-based agroforestry systems are considered the best way to resolve the environmental problems associated with rubber monoculture. In recent years, the local government of Xishuangbanna proposed the development of environmentally friendly rubber plantations to reduce the water and soil losses and increase environmental biodiversity9,10.
Legume plants could greatly enhance ecosystem services. Lucerne (Medicago sativa) and erect milkvetch (Astragalus adsurgens) have the potential to phytoextract rhenium from coal fly ash-amended alkaline soils11. Alfalfa (Medicago sativa L.) can improve its phosphorus acquisition by increasing specific root length and exuding gcarboxylates into the rhizosphere in phosphorus-deficient environments12. A combination of legumes grass species can enhance soil C and N storage, productivity, and diversity in semi-arid grasslands13. Flemingia macrophylla is used in traditional medicine for various therapeutic uses and is widely planted in the Xishuangbanna area of China. Flemingia macrophylla is a perennial leguminous leafy shrub with strong biological nitrogen fixation and high biomass14. As a result, rubber–Flemingia macrophylla intercropped systems have become popular in the Xishuangbanna area to improve soil carbon and nitrogen storage.
Precise and accurate estimations of carbon and nitrogen levels in forest soil are important for understanding biogeochemical cycles15,16,17. Some studies have indicated that soil organic carbon (SOC) has been depleted in rubber plantations18,19. Rubber plantations have 15% lower annual surface soil CO2 fluxes than natural forests, because they have lower soil respiration during the dry season20. The conversion of tropical rainforests into rubber plantations has increased N2O emissions, which may potentially enhance local climate warming trends21. Rubber plantations have lower mean CH4 uptake rates than secondary and tropical forests22. These transformations and biogeochemical cycles of soil carbon and nitrogen are mainly affected by their fractions23,24,25,26,27 that play essential roles in the turnover of nutrients in soil, including water-soluble organic carbon (WSOC), light fraction of organic C (LFOC), microbial biomass organic C (MBC), ammonium N (AN), nitrate N (NN), light fraction of organic N (LFON), and microbial biomass organic N (MBN). These fractions are often used to study the impacts of land management and ecological succession26,28,29. Rubber-based agroforestry systems have higher SOC and nitrogen levels and lower carbon and nitrogen losses than rubber plantations due to improved soil macroaggregates30. However, little is known about the effect of these systems on soil carbon and nitrogen fractions.
The objectives of this study were to examine: (1) soil carbon and nitrogen fractions, and (2) the relationships between soil carbon and nitrogen fractionsin rubber and rubber–Flemingia macrophylla plantations of different ages.
Results
Soil carbon fractions
The SOC content in the 0–10 or 10–30 cm soil layers did not differ between the plantation treatments (R1, R2, RF1, RF2) for the duration of the study (April 2014 to January 2017) (Table 1). However, SOC content in the 0–10 cm soil layers was significantly higher than the 10–30 cm soil layers in each plantation type. In the 0–10 cm soil layer, RF1 had consistently higher WSOC content than R1 from June 2015 to January 2017, which differed significantly in August 2016 and January 2017. The WSOC content between R2 and RF2 did not differ significantly for the duration of the experiment (Table 2). In the 10–30 cm soil layer, the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on WSOC content.
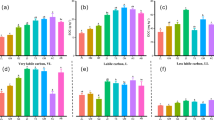
The 0–10 cm soil layer had significantly higher LFOC content than the 10–30 cm soil layer in each plantation type. In the 0–10 cm soil layer, RF1 had consistently higher LFOC content than R1 from June 2015 to January 2017, which differed significantly in August 2016 and January 2017. In the same layer, RF2 had consistently lower LFOC content than R2, with significant differences observed in January 2016 and 2017 (Table 3). In the 10–30 cm soil layer, the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on LFOC content but increased MBC content and the ratios of MBC/SOC in the 0–10 and 10–30 cm soil layers (Table 4 and Fig. 1). In the 0–10 cm soil layer, RF1 had consistently higher ratios of LFOC/SOC than R1 from January 2016 to January 2017, but no significant differences were observed between the RF2 and R2 for the duration of the study (Fig. 2). In the 10–30 cm soil layer, the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on the ratios of LFOC/SOC.
Ratio of soil microbial biomass carbon (MBC) to soil organic carbon (SOC) (MBC/SOC); and ratio of soil microbial biomass nitrogen (MBN) to soil total nitrogen (TN) (MBN/TN) in the rubber and rubber–Flemingia macrophylla plantations from June 2015 to January 2017. R1: rubber plantations established in 2006; R2: rubber plantations established in 1994; RF1: Flemingia macrophylla introduced to R1 in 2010; RF2: Flemingia macrophylla introduced to R2 in 2010. Vertical bars are LSD at P ≤ 0.05.
Ratio of soil light fraction carbon (LFOC) to soil organic carbon (SOC) (LFOC/SOC); and ratio of soil light fraction nitrogen (LFON) to soil total nitrogen (TN) (LFON/TN) in the rubber and rubber–Flemingia macrophylla plantations from June 2015 to January 2017. R1: rubber plantations established in 2006; R2: rubber plantations established in 1994; RF1: Flemingia macrophylla introduced to R1 in 2010; RF2: Flemingia macrophylla introduced to R2 in 2010. Vertical bars are LSD at P ≤ 0.05.
Soil nitrogen fractions
The TN content in the 0–10 or 10–30 cm soil layers did not differ between the plantation treatments (R1, R2, RF1, RF2) for the duration of the study (April 2014 to January 2017) (Table 5). However, the 0–10 cm soil layer had significantly higher TN contents than the 10–30 cm soil layer in each plantation type. The introduction of Flemingia macrophylla to the rubber plantations generally decreased AN content and increased NN content in the 0–10 and 10–30 cm soil layers (Fig. 3). In the 0–10 cm soil layer, RF1 had consistently higher LFON content than R1 from June 2015 to January 2017, with significant differences observed from January 2016 to January 2017. In the same layer, no significant differences in LFON content were observed between R2 and RF2, except for January 2017 when the LFON content was significantly higher in R2 than RF2 (Table 6). In the 10–30 cm soil layer, the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on LFON content. The 0–10 cm soil layer had significantly higher LFON contents than the 10–30 cm soil layer in each plantation type.
Soil ammonium N (AN) and nitrate N (NN) content levels in the rubber and rubber–Flemingia macrophylla plantations from June 2015 to January 2017. R1: rubber plantations established in 2006; R2: rubber plantations established in 1994; RF1: Flemingia macrophylla introduced to R1 in 2010; RF2: Flemingia macrophylla introduced to R2 in 2010. Vertical bars are LSD at P ≤ 0.05.
The introduction of Flemingia macrophylla to the rubber plantations increased MBN content and the ratios of MBN/TN in the 0–10 and 10–30 cm soil layers (Table 7 and Fig. 1). In the 0–10 cm soil layer, RF1 had consistently higher ratios of LFON/TN than R1 from January 2016 to January 2017, but no significant differences were observed between the RF2 and R2 for the duration of the study (Fig. 2). In the 10–30 cm soil layer, the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on the ratios of LFON/TN. The introduction of Flemingia macrophylla to the rubber plantations decreased the ratios of LFOC/LFON in the 0–10 and 10–30 cm soil layers (Fig. 4).
Ratio of organic C to total N in the soil light fractions (LFOC/LFON) in the rubber and rubber–Flemingia macrophylla plantations from June 2015 to January 2017. R1: rubber plantations established in 2006; R2: rubber plantations established in 1994; RF1: Flemingia macrophylla introduced to R1 in 2010; RF2: Flemingia macrophylla introduced to R2 in 2010. Vertical bars are LSD at P ≤ 0.05.
Relationships between soil carbon and nitrogen fractions
MBC and MBN had significant positive correlations with LFOC, LFON, and the ratios of MBC/SOC, LFOC/SOC, MBN/TN and LFON/TN (Table 8). MBC and MBN also had negative correlations with the ratio of LFOC/LFON. AN and NN had significant positive correlations with LFOC and LFON, and the ratios of LFOC/SOC and LFON/TN. Furthermore, AN and NN had significant negative correlations with LFOC/LFON.
Discussion
Soil carbon and nitrogen fractions in the rubber and rubber–Flemingia macrophylla plantation systems
Plantations with suitable native, broad-leaved species (for example, Alnus subcordata C. A. Mey.) along with planned forestation management could potentially rehabilitate the degraded natural forests of northern Iran31. Mo and Sha32 reported that adding carbon-sink plants into rubber plantations increased the soil organic carbon storage. The presence of legumes in semi-arid grasslands increased soil C and N storage by increasing the above- and below-ground biomass, litter biomass, plant species richness, and diversity13. In 2014 in present study, biomass of Flemingia macrophylla accumulated in the young and mature rubber plantations at 23.02 and 0.55 t ha−1, respectively14 but had no significant effect on SOC or TN contents. There are four possible explanations for this response: (1) the rubber–Flemingia macrophylla system had only been established for seven years, and it is unlikely that soil C and N storage would have changed significantly in such a short time; (2) the high-temperature and high-humidity environments in the rainy season would promote the decomposition of plant residues and nutrient leaching, which is not conducive to C and N storage; (3) the introduction of Flemingia macrophylla to the rubber plantations reduced plant species richness and diversity (Fig. 5) and the input of litter biomass of other species; (4) most rubber plantations in this area have been planted on sloping land20. In our study, the rubber and rubber–Flemingia macrophylla plantations were planted on sloping land between 47 and 58%, and the nutrients of the litter decomposition and some un-decomposed litter of Flemingia macrophylla would have been lost due to runoff.
While the introduction of Flemingia macrophylla to the rubber plantations had no significant effect on the C and N contents in the soil, the fractions of C and N changed significantly. The introduction of Flemingia macrophylla in the young rubber plantations was propitious for improving the labile organic C (WSOC and LFOC) and LFON contentin the 0–10 cm soil layersdue to the input of large amounts of litter from Flemingia macrophyllaeach year (Fig. 5C). The light fraction of the soil mainly consisted of plant residues, small animals, and microorganisms in various stages of decomposition. It served as a readily decomposable substrate for soil microorganisms and a short-term reservoir for plant nutrients. It potentially served as an early indicator of the effects of the management practices33,34. The WSOC also played a dominant role in several soil processes and was more sensitive for land use than the total SOC pool28,35. The WSOC indicated that the introduction of Flemingia macrophylla to the young rubber plantations could change the soil C fractions in the 0–10 cm soil layer within a short time. However, the introduction of Flemingia macrophylla into the mature rubber plantations had no significant effect on WSOC, LFOC, or LFON content in the short term, due to the lower input levels of Flemingia macrophylla litter each year.
In this study, the introduction of Flemingia macrophylla to the rubber plantations decreased AN content and increased NN content, which may have been due to the increased rate of nitrification from improved microbial activity in the rubber–Flemingia macrophylla systems. The NN content in soil is important for the growth of rubber trees, as well as rubber production36. In this region, soil NN content in rubber plantations is generally <1 mg kg−1 37. In our study, soil NN content in the rubber plantations ranged from 0.02–0.93 mg kg−1. These findings suggest that Flemingia macrophylla is important for improving NN content in the soil of the rubber plantations37.
Relationships between soil carbon and nitrogen fractions
The microbial biomass of soil controls organic matter sequestration and decomposition and is generally used as an ecological sensitivity indicator for changes in soil nutrients due to land use practices38,39. For example, high MBC/SOC and MBN/TN ratios indicate that the organic matter decomposed quickly40. In the current study, the introduction of Flemingia macrophylla to the rubber plantations increased MBC and MBN, along with the MBC/SOC and MBN/TN ratios in the 0–10 and 10–30 cm soil layers. These results agree with those of Wen et al.41, who suggested that the conversions of monocultures into mixed forests had a strong positive effect on soil microbial biomass, and increased the efficiency of microbes in soil carbon decomposition along the profile by improving the ratios of the MBC/SOC and MBN/TN. Interestingly, the introduction of Flemingia macrophylla to the rubber plantations in our study decreased the ratio of LFOC/LFON. It was observed that large amounts of leaves from the Flemingia macrophylla were incorporated into the soil each year, which tended to decrease the LFOC/LFON ratio in the soil due to the low C/N ratio in the leaves of Flemingia macrophylla. Generally speaking, the C/N ratio of Flemingia macrophylla is approximately 15:114. Chen42 and Huang43 reported that low C/N ratios in soil could increase microbial biomass, and accelerate the decomposition of soil organic matter. We also observed a significant negative correlation between the ratio of LFOC/LFON, and MBC and MBN (P ≤ 0.01). These results suggest that the decreased ratios of LFOC/LFON in the rubber–Flemingia macrophylla plantations enhanced microbial activity.
Conclusions
While the introduction of Flemingia macrophylla to the rubber plantations did not significantly increase total soil carbon or nitrogen levels over a short period, it changed the carbon and nitrogen fractions, improved labile organic carbon and nitrogen contents, and ameliorated the soil environment. We recommend that local governments and farmers in southeastern Asia use Flemingia macrophylla as alternative interplanted tree species within rubber plantations.
Materials and Methods
Description of the study site
This study was conducted in the Xishuangbanna region (21°33′N, 101°28′E; 880 to 900 m asl) of Yunnan Province in southwestern China. This region has a typical tropical monsoon climate, with an annual mean temperature of 21.8 °C. The area receives mean annual precipitation of ~1,500 mm, 80% of which occurs in the rainy season (May to October)44. Furthermore, Xishuangbanna contains the largest area of tropical rainforests in China. Its biodiversity is rich as it is part of the Indo-Burma world biodiversity hotspot45. The soil has been classified as laterite (Oxisol), which developed from arenaceous shale sediment44,46.
In 1991 and 2003, the tropical forests with slopes ranging from 47–58% were deforested. Sugarcane (Saccharum officinarum L.) was then planted annually. Rubber trees were plantedon these sites in May 1994 and 2006 at a density of 450 rubber trees ha−1, with 8 m spacing between adjacent rows. In accordance with the local practices for rubber trees less than three years of age, fertilizers were applied between the rubber trees at depths of 20 cm using spades at rates of 27.0 kg ha−1 N, 5.9 kg ha−1 P, and 11.2 kg ha−1 K, which were split into two applications per year (May and October). Once the rubber trees were more than three years of age, the fertilizer application rates changed to 54.0 kg ha−1 N, 11.8 kg ha−1 P, and 22.4 kg ha−1 K. The rubber plantation farmers generally sprayed sulfur powder at 30–60 kg ha−1 yr−1 to control powdery mildew on the rubber trees. Weeds in the plantations were cut using a sickle twice per year (April/May and November/December) and left on the ground. In July 2010, Flemingia macrophylla was introduced into the differently aged rubber plantations (4 and 16 years of age) at a density of 10,830 plants ha−1. From 2011 onwards, the Flemingia macrophylla in the different rubber plantations was cut using a sickle in December each year and left as ground cover. From 2012 onwards, given the strong biological nitrogen fixation of Flemingia macrophylla, no additional N was applied in the rubber–Flemingia macrophylla plantations. The inputs of P, K, and S in the rubber–Flemingia macrophylla plantations remained the same as those in the adjacent rubber plantations.
Experimental design, sampling, and measurements
In this study, three replicate sites were selected within each rubber and rubber–Flemingia macrophylla plantation (Fig. 5). Each replication site consisted of 20 × 25 m2 survey plots (four rows of rubber trees, and three 8-m wide hedgerows) containing nine sampling subplots (8 × 6 m2), with three located at each slope position (upper, middle, and lower slope).
For each of the nine subplots, soil samples were collected using a soil auger, avoiding the fertilization holes, at two depths (0–10 cm and 10–30 cm) after carefully removing the litter-fall and/or grass layer. For each replicate site in the different rubber and rubber–Flemingia macrophylla plantations, soil core samples were collected in April 2014, June 2015, January and August 2016, and January 2017. The nine soil cores were combined into a composite sample, which were air-dried, ground, and sieved (at <2 mm) for analysis of ammonium nitrogen (AN) and nitrate-nitrogen (NN), and the light fraction organic carbon (LFOC) and nitrogen (LFON). The sieved samples were sieved again (at <0.25 mm) for determination of soil organic carbon (SOC) and total nitrogen (TN). For soil water-soluble organic carbon (WSOC), microbial biomass carbon (MBC) and nitrogen (MBN) measurements, the samples were taken to the laboratory and stored at 4 °C for subsequent analyses.
The SOC and TN of the bulk soil were determined using a Vario MAX CN-Analyzer (Elementar Analysensysteme GmbH, Germany). A density fractionation scheme for light was used following the method described by Gregorich and Ellert47. During fractionation, 25 g of air-dried soil was shaken with 50 mL of NaI solution (sp.Gr. = 1.70) for 60 min. After centrifugation, the supernatant was passed through a Millipore filter (0.45 μm) and the light fraction collected. The soil residue in the centrifuge was extracted again with NaI, and the additional light fraction collected. The light fraction was oven-dried at 60 °C of 72 h. The concentration of organic carbon and nitrogen was determined by dry combustion using a Vario MAX CN-Analyzer (Elementar Analysensysteme GmbH, Germany). Dried samples, each weighing 5 g, were added to 50 mL of 2 M KCl, shaken for one hour, and analyzed with an Auto Analyzer 3 (SEAL Analytical GmbH, Germany) to determine AN and NN contents34,48. Microbial biomass carbon (MBC) and nitrogen (MBN) in the soil were estimated using a fumigation-extractionmethod49 that included a purified CHCl3 treatment, followed by a 0.5 M K2SO4 extraction of fumigated and unfumigated soil50. After which, soil samples (equivalent to 25 g of dry soil weight) were fumigated for 24 h at 25 °C with CHCl3 (ethanol-free). Following fumigant removal, the soil was extracted with 100 mL of 0.5 M K2SO4 by shaking for 1 h at 200 rpm, followed by filtering. The non-fumigated portions were extractedat similar time intervals. Following the extraction, MBC and MBN contents were measured by determining the C and N masses in the filtrate using a Vario TOC cube-Analyzer (Elementar Analysensysteme GmbH, Germany). Microbial biomass carbon was calculated as follows: MBC = (Corg(fum) − Corg(non))/0.3851. Microbial biomass nitrogen wascalculated as follows: MBN = (TN(fum) − TN(non))/0.4551. The ratios of MBC to total carbon (MBC/SOC) and MBN to total nitrogen (MBN/TN) were then calculated. Water-soluble organic carbon (WSOC) was extracted from field-moist samples within 24 h of sampling by shaking 15 g soil with 30 mL distilled water for 2 h at 25 °C, followed by centrifugation at 5000 r min−1 at 4 °C for 15 min. The supernatant was filtered through a 0.45 μm carbon-free membrane. The filtrates were stored at 4 °C and analyzed within 24 h using a Vario TOC cube-Analyzer (Elementar Analysensysteme GmbH, Germany)35.
Statistical analysis
The data were subjected to analysis of variance (ANOVA) using SAS statistical analysis software version 8.0. One-factor ANOVA was deployed to compare treatment effects. The least significant difference (LSD at 0.05 level of probability) test was applied to assess the differences between means. Pearson’s coefficient analysis was used for correlation.
References
Warren-Thomas, E., Dolman, P. M. & Edwards, D. P. Increasing demand for natural rubber necessitates a robust sustainability initiative to mitigate impacts on tropical biodiversity. Conservation Letters 8, 230–241 (2015).
Tangena, J. A. A., Thammavong, P., Wilson, A. L., Brey, P. T. & Lindsay, S. W. Risk and control of mosquito-borne diseases in Southeast Asian rubber plantations. Trends in Parasitology 32(5), 402–415 (2016).
Ahrends, A. et al. Current trends of rubber plantation expansion may threaten biodiversity and livelihoods. Global Environmental Change 34, 48–58 (2015).
Mei, C. C. The spatial-temporal evolution of rubber plantation and the distribution patterns of aboveground biomass carbon storage in Xishuangbanna. M.Sc degree thesis, University of Chinese Academic of Sciences (2015).
Tan, Z. H. et al. Rubber plantations act as water pumps in tropical China. Geophysical Research Letters 38, L24406 (2011).
Guillaume, T., Damris, M. & Kuzyakov, Y. Losses of soil carbon by converting tropical forest to plantations: erosion and decomposition estimated byδ13C. Global Change Biology 21(9), 3548–3560 (2015).
Zheng, G., Li, S. & Yang, X. Spider diversity in canopies of Xishuangbanna rainforest (China) indicates an alarming juggernaut effect of rubber plantations. Forest Ecology and Management 338, 200–207 (2015).
Yi, Z. F., Cannon, C. H., Chen, J., Ye, C. X. & Swetnam, R. D. Developing indicators of economic value and biodiversity loss for rubber plantations in Xishuangbanna, southwest China: A case study from Menglun township. Ecological Indicators 36, 788–797 (2014).
Bai, J. K. Developing environmental-friendly rubber plantations in Yunnan province for promoting the sustainability of ecological rubber. China State Farms 5, 20–22 (2015).
Liu, W., Zhu, C., Wu, J. & Chen, C. Are rubber-based agroforestry systems effective in controlling rain splash erosion? Catena 147, 16–24 (2016).
He, H. et al. Phytoextraction of rhenium by lucerne (Medicago sativa) and erect milkvetch (Astragalus adsurgens) from alkaline soils amended with coal fly ash. Science of the Total Environment 630, 570–577 (2018).
He, H. et al. Growth, morphological and physiological responses of alfalfa (Medicago sativa) to phosphorus supply in two alkaline soils. Plant and Soil 416, 565–584 (2017).
Wu, G. L., Liu, Y., Tian, F. P. & Shi, Z. H. Legumes functional group promotes soil organic carbon and nitrogen storage by increasing plant diversity. Land Degradation & Development 28, 1336–1344 (2017).
Wang, F. J. The content of nitrogen fixed by Flemingia macrophylla and its effects on the growth of rubber and concentration of soil total nitrogen in rubber–Flemingia macrophylla intercropped systems in different stand ages. M.Sc degree thesis, University of Chinese Academic of Sciences (2015).
Janssens, I. A. et al. Reduction of forest soil respiration in response to nitrogen deposition. Nature Geoscience 3, 315–322 (2010).
Li, D., Niu, S. & Luo, Y. Global patterns of the dynamics of soil carbon and nitrogen stocks following afforestation: a meta-analysis. New Phytologist 195(1), 172–181 (2012).
Tashi, S., Singh, B., Keitel, C. & Adams, M. Soil carbon and nitrogen stocks in forests along an altitudinal gradient in the eastern Himalayas and a meta-analysis of global data. Global Change Biology 22(6), 2255–2268 (2016).
Zhang, M., Fu, X. H., Feng, W. T. & Zou, X. M. Soil organic carbon in pure rubber and tea-rubber plantations in southwestern China. Tropical Ecology 48, 201–207 (2007).
de Blécourt, M., Hänsel, V. M., Brumme, R., Corre, M. D. & Veldkamp, E. Soil redistribution by terracing alleviates soil organic carbon losses caused by forest conversion to rubber plantation. Forest Ecology and Management 313, 26–33 (2014).
Goldberg, S. D. et al. Soil respiration in sloping rubber plantations and tropical natural forests in Xishuangbanna, China. Agriculture, Ecosystem & Environment 249, 237–246 (2017).
Zhou, W. J. et al. The effects of nitrogen fertilization on N2O emissions from a rubber plantation. Scientific Reports 6, 28230 (2016).
Werner, C. et al. N2O, CH4 and CO2 emissions from seasonal tropical rainforests and a rubber plantation in Southwest China. Plant and Soil 289(1–2), 335–353 (2006).
Shi, Y., Chen, X. & Shen, H. M. Light fraction carbon and water-stable aggregates in black soils. Pedosphere 17(1), 97–100 (2007).
Demisie, W., Liu, Z. & Zhang, M. Effect of biochar on carbon fractions and enzyme activity of red soil. Catena 121, 214–221 (2014).
Shahid, M. et al. Carbon and nitrogen fractions and stocks under 41 years of chemical and organic fertilization in a sub-humid tropical rice soil. Soil & Tillage Research 170, 136–146 (2017).
de Moraes Sá, J. C. et al. Soil carbon fractions and biological activity based indices can be used to study the impact of land management and ecological successions. Ecological Indicators 84, 96–105 (2018).
Ren, C. et al. Differential responses of soil microbial biomass, diversity, and compositions to altitudinal gradients depend on plant and soil characteristics. Science of the Total Environment 610–611, 750–758 (2018).
Tang, X., Zhang, X., Wang, Z. & Ci, Z. Water-soluble organic carbon (WSOC) and its temperature-resolved carbon fractions in atmospheric aerosols in Beijing. Atmospheric Research 181, 200–210 (2016).
Liu, C. A., Siddique, K. H. M., Hua, S. & Rao, X. The trade-off in the establishment of artificial plantations by evaluating soil properties at the margins of oases. Catena 157, 363–371 (2017).
Chen, C., Liu, W., Jiang, X. & Wu, J. Effects of rubber-based agroforestry systems on soil aggregation and associated soil organic carbon: Implications for land use. Geoderma 299, 13–24 (2017).
Moghimian, N., Hossein, S. M., Kooch, Y. & Darki, B. Z. Impacts of changes in land use/cover on soil microbial and enzyme activities. Catena 157, 407–414 (2017).
Mo, H. Z. & Sha, L. Q. The carbon stock and carbon sequestration potential of rubber plantations under different agro-forestry systems in Xishuangbanna, SW China. Mountain Research 34(8), 707–715 (2016).
Liu, C. A. et al. Effects of water management with plastic film in a semi-arid agricultural system on available soil carbon fractions. European of Journal Soil Biology 57, 9–12 (2013).
Zhong, Y., Yan, W. & Shangguan, Z. Soil carbon and nitrogen fractions in the soil profile and their response to long-term nitrogen fertilization in a wheat field. Catena 135, 38–46 (2015).
Wei, Z. Q., Wu, S. H., Zhou, S. L., Li, J. T. & Zhao, Q. G. Soil organic carbon transformation and related properties in urban soil under impervious surfaces. Pedosphere 24(1), 56–64 (2014).
Li, Y. Accumulated impacts of sulfur spraying on soil nutrient availability and microbial biomass in Rubber Plantations. CLEAN-Soil, Air, Water 44(8), 1001–1010 (2016).
Zhang, M. & Zou, X. M. Comparison of soil C and N in rubber plantation and seasonal rainforest. Chinese Journal of Applied Ecology 20(5), 1013–1019 (2009).
Spohn, M., Klaus, K., Wanek, W. & Richter, A. Microbial carbon use efficiency and biomass turnover times depending on soil depth-Implications for carbon cycling. Soil Biology and Biochemistry 96, 74–78 (2016).
Buyer, J. S., Baligar, V. C., He, Z. & Arévalo-Gardini, E. Soil microbial communities under cacao agroforestry and cover crop systems in Peru. Applied Soil Ecology 120, 273–280 (2017).
Jiang, H. M., Li, F. M. & Jiang, J. P. Soil carbon pool and effects of soil fertility in seeded alfalfa fields on the semi-arid Loess Plateau in China. Soil Biology and Biochemistry 38(8), 2350–2358 (2006).
Wen, L., Lei, P., Xiang, W., Yan, W. & Liu, S. Soil microbial biomass carbon and nitrogen in pure and mixed stands of Pinus massoniana and Cinnamomum camphora differing in stand age. Forest Ecology and Management 328, 150–158 (2014).
Chen, W. X. Soil Science and Environmental Microbiology. BeijingAgriculture University Press, Beijing (1990).
Huang, C. Y. Soil Science. China Agriculture Press, Beijing (2000).
Li, H., Ma, Y., Liu, W. & Liu, W. Soil changes induced by rubber and tea plantation establishment: comparison with tropical rain forestsoil in Xishuangbanna, SW China. Environmental Management 50(5), 837–848 (2012).
Myers, N., Mittermeier, R. A., Mittermeier, C. G., da Fonseca, G. A. B. & Kent, J. Biodiversity hotspots for conservation priorities. Nature 403, 853–858 (2000).
Wang, W. F., Qiu, D. Y., Wu, J. C. & Ye, H. M. The Soils of Yunnan. Yunnan Science and Technology Press, Kunming, China (1996).
Gregorich, E. G. & Ellert, B. H. Light fraction and macroorganic matter in mineral soils, in: M.R. Carer (Ed.), Soil Sampling and Methods of Analysis, Can Soc Soil Sci, Lewis Publishers, Division of CRC Press, Boca Raton, FL, pp. 397–405 (1993).
Zhang, Y. & Zhao, W. Vegetation and soil property response of short-time fencing in temperate desert of the Hexi Corridor, northwestern China. Catena 133, 43–51 (2015).
Brookes, P. C., Landman, A., Pruden, G. & Jenkinson, D. S. Chloroform fumigation and release of soil nitrogen: rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biology and Biochemistry 17(6), 837–842 (1985).
Singh, H. & Singh, K. P. Effect of residue placement and chemical fertilizer on soil microbial biomass under tropical dryland cultivation. Biology and Fertility of Soils 16(4), 275–281 (1993).
Ocio, J. A. & Brooks, P. C. An evaluation of methods for measuring the microbial biomass in soils following recent addition of wheat straw, and the characterization of the biomass that develops. Soil Biology and Biochemistry 22(5), 262–685 (1990).
Acknowledgements
We would like to acknowledge the Public Technology Service Center of XTBG, CAS, for their support with field measurements and soil analyses. This study was funded by the National Natural Science Foundation of China (31470639), the Natural Science Foundation of Yunnan Province (2017FB059 and 2016FA047), the Key Program of CAS (KFZD-SW-312) and the Project of Xishuangbanna Science and Technology Bureau (200915, 201116).
Author information
Authors and Affiliations
Contributions
C.A.L. conducted the experiments and wrote the manuscript; Y.N. analyzed the data assisted. J.W.T., K.H.M.S. and Y.M.Z. assisted with writing the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, CA., Nie, Y., Zhang, YM. et al. Introduction of a leguminous shrub to a rubber plantation changed the soil carbon and nitrogen fractions and ameliorated soil environments. Sci Rep 8, 17324 (2018). https://doi.org/10.1038/s41598-018-35762-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35762-0
Keywords
This article is cited by
-
Improved soil moisture, nutrients, and economic benefits using plastic mulchs in balsa-based agroforestry systems
Environmental Science and Pollution Research (2024)
-
Understory vegetation management regulates soil carbon and nitrogen storage in rubber plantations
Nutrient Cycling in Agroecosystems (2023)
-
Improving soil pH, nutrient concentrations, and enzyme activities by green manure returning in young and mature rubber plantation on Hainan Island, China
Plant and Soil (2023)
-
Temporal changes of topsoil nematode communities in rubber plantations in Ivory Coast in response to logging residue management and legumes introduction
Plant and Soil (2023)
-
Can larch-Aralia elata agroforestry systems improve the soil chemical and microbial properties of larch plantations?
Agroforestry Systems (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.