Abstract
Legumes rely on soil mineral nitrogen (N) and biological N fixation (BNF). The interplay between these two sources is biologically interesting and agronomically relevant as the crop can accommodate the cost of BNF by five non-mutually exclusive mechanisms, whereby BNF: reduces shoot growth and seed yield, or maintains shoot growth and seed yield by enhanced photosynthesis, or reduced root:shoot ratio, or maintains shoot growth but reduces seed yield by reducing the fraction of shoot biomass allocated to seed (harvest index), or reducing concentration of oil and protein in seed. We explore the impact of N application on the seasonal dynamics of BNF, and its consequences for seed yield with emphasis on growth and shoot allocation mechanisms. Trials were established in 23 locations across the US Midwest under four N conditions. Fertilizer reduced the peak of BNF up to 16% in applications at the full flowering stage. Seed yield declined 13 kg ha−1 per % increase in RAUR6. Harvest index accounted for the decline in seed yield with increasing BNF. This indicates the cost of BNF was met by a relative change in dry matter allocation against the energetically rich seed, and in favor of energetically cheaper vegetative tissue.
Similar content being viewed by others
Introduction
Globally, soybean [Glycine max (L.) Merr.] is a major source of protein and oil. In the US, soybean is grown in a range of latitudes and environments representing 29% of the national crop acreage1. Significant breeding effort during the last century sought to improve seed yield and maintain seed protein2. One of the challenges to further improve soybean seed yield is the high demand of nitrogen (N) in comparison to cereals and oilseed crops3,4. Legumes rely on two sources of N: mineral N from soil and biological fixation (BNF); the proportion from each source varies with environmental and soil conditions including temperature5, soil moisture6,7, soil pH8, mineral soil N9, strain10 and crop genotype7,11. In soybean, BNF is greater in genotypes with longer reproductive periods reflected in maturity group (MG)12.
Biological N fixation requires plant’s reduced carbon (C) and energy, as reviewed by Kaschuk et al.13. For soybean, BNF requires 6-7 g C g−1 N in comparison to 4 g C g−1 N for assimilation of mineral N; integrated over the growing season the difference in cost is substantial, with potential implications for seed yield and seed protein or oil concentrations. The cost of BNF can be partially compensated by increase in photosynthesis of plants associated with rhizobia13 or shifts in allocation of biomass. For instance, nodulated roots accumulated less biomass compared with plants growing with high soil N supply14 and lower biomass partitioning to seeds associated with increasing BNF7. Thus, the crop can accommodate the cost of BNF by five non-mutually exclusive mechanisms, whereby N fixation: (a) reduces shoot growth and seed yield, or maintains shoot growth and seed yield by (b) enhanced photosynthesis13, or (c) reduced root:shoot ratio15, or maintains shoot growth but reduces seed yield by (d) reducing seed oil and protein concentration in seed, or (e) the fraction of shoot biomass allocated to seed (i.e., harvest index; HI).
Further, there is an agronomic interest on the role of mineral N to support high seed yield16,17 and avoid protein dilution18,19. A recent review of Mourtzinis et al.20 concluded that N fertilization has a small and inconsistent effect on soybean seed yield. This conclusion is, however, largely based on generic trials where coarse fertilization regimes were established to shift the contribution of mineral N and BNF. In contrast, a full-N treatment devised with a careful experimental protocol to ensure an ample N supply during the entire crop season increased soybean seed yield by 11% in relation to unfertilized controls, with a range from no effect for stressful environments (ca. 2500 kg ha−1) but increases of 900 kg ha−1 in high potential environments (ca. 6000 kg ha−1)16.
The goal of this study was to investigate the effect of fertilizer N application on BNF and its implications for soybean seed yield and seed protein concentration. We tested the hypothesis that the cost of N fixation is mediated by reduced biomass, reduced allocation to seed captured in the HI21, and reduced concentration of protein and oil in seed. Quantification of these effects will provide insights of BNF impact on crop C and N economy, and will contribute to explain the apparent inconsistency in soybean seed yield responses to N fertilization.
Results
Effect of N fertilizer on N fixation
Data for this study were collected from 23 different locations across the US Midwest during the 2016 growing season (Table 1; Fig. 1). Table 2 shows the relative abundance of ureides in R6 (full seed stage; RAUR6) for each location and treatment, Fig. 2 illustrates the seasonal dynamics of the relative abundance of ureides (RAU) for crops grouped in high, medium and low BNF, and Table 3 summarizes the parameters of the curves. The fitted model (equation 2) returned R2 between 0.62 and 0.87, with P < 0.001 in all cases.
Seasonal changes in the proportion of the relative abundance of ureides (RAU) and RAU rate for the high (a,d), medium (b,e), and low (c,f) BNF groups for the control (black full line and black circles), N at sowing (broken line and empty squares), N at V4 (dotted line and empty triangles), and N at R2 (red full line and empty inverted triangles). Each point in V4 (fourth-leaf), R2 (full flowering), R6 (full seed), and R8 (full maturity) represents the average from the locations of each group.
The RAUR6 ranged between 42 to 93% (Table 2) and responded to all three sources of variation: treatment, location and their interaction (P < 0.001). Fertilizer reduced RAUmax in all BNF groups, but not where BNF was already low in the control treatment (Table 3). In the high BNF group, RAUmax dropped from 90% in controls to 75% in their fertilized counterparts (averaging all N treatments; Table 3); the reduction in RAU was larger when the application of N was delayed from vegetative to reproductive stages. In the medium group, RAUmax declined from 84% in controls to 68% in the V4 (fourth-leaf) application and 74% in both sowing and R2 (full flowering) applications, with no clear effect of N application timing.
The reduction in RAUmax can be a consequence of a shorter time to peak RAU, a reduced rate or a combination of both effects. Combination of both traits contributed to reduced N fixation in the medium BNF group, as peak RAU and maximum rate were attained earlier in N-fertilized crops. In the low BNF group, N fertilizer reduced RAU rate but time of peak RAU was not affected. Reduction of the area under the curve (AUC) and the time when RAU reached 50% was observed from high to low BNF groups; however, treatments did not affect AUC (Table 3).
Fertilization treatments affected the dynamics of RAU rate. Both the timing of peak rate (tm), and the timing when rate became negative (tmax) were delayed from low to high BNF groups (Fig. 2d–f). For the high and medium groups, reproductive N treatment was the most effective reducing both tm and tmax hence contributing to an overall reduction of RAUmax.
Effect of N fixation on seed yield, biomass, harvest index, protein and oil concentration
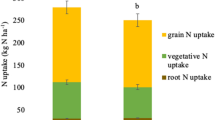
Seed yield ranged from 3151 to 7175 kg ha−1, seed protein concentration from 31.9 to 41.8 g 100 g–1, and seed oil concentration from 16.7 to 23.9 g 100 g−1 (Table 2). For these traits, ANOVA showed location was a significant source of variation (P < 0.001), with no effect of treatment and its interaction with location (P > 0.05). Total biomass ranged from 6093 to 11376 kg ha−1 showing differences only among locations (P < 0.001). Harvest index ranged from 0.37 to 0.56, and was affected by both treatment (P < 0.05) and location (P < 0.001).
In this experiment, MGs were allocated to locations for agronomic relevance (Table 1; Fig. 1). The dominant effect of location on crop traits is therefore confounded with crop phenology. For instance, the range of thermal time to R6 was 909 to 1733 °Cd. We thus fitted bilinear models to account for the effect of phenology on crop traits (Fig. 3a,c,d), and regressed the residuals against RAUR6 (Fig. 3b,d,f; see section on materials and method). Analysis of residuals showed seed yield declined at 13 kg ha−1 per % of RAUR6 for the whole data set and 10 kg ha−1 per % of RAUR6 (p < 0.05, Fig. 3b) for top yielding crops (0.99 quartile). Under stressful conditions leading to low HI, the rate of decline in HI with RAUR6 computed as the slope of the 0.01 quartile, was 2.5 times larger than the rate for the pooled data (Fig. 3d). Oil seed concentration was negatively associated with RAUR6 (p < 0.05; Fig. 3f) and crops with higher oil concentration (0.99 quartile) were more responsive to RAUR6. After removing the effects of phenology, biomass and seed protein concentration (not shown) did not relate to RAUR6 (both p > 0.05).
Relationship between seed yield (a), harvest index (c), seed oil concentration (e) and thermal time to R6. In (a–c), solid lines are bilinear models with fitted parameters a, b, c, TTo (equation 5). Relationship between residuals from relationship in a (c), c (b), and e (f) with RAU at R6. Solid lines are least square regressions, and dashed lines are regressions for the 0.99 (b,f) and 0.01 (d) quantiles. Asterisks indicate significance of the coefficient: three asterisks, P < 0.001; two asterisks, P < 0.01; one asterisk, P < 0.05.
The association between BNF traits, soil attributes, seed yield, biomass, HI, seed protein and oil concentrations, adjusted by the effects of phenology, were explored using principal component analysis (PCA). Results from the ordination analysis were presented in a bi-plot showing the two first principal components (Fig. 4) where angles between variable vectors denote the level of association among them (i.e., acute angles denote positive associations and obtuse angles negative associations between variables). Pearson correlation analysis complements the associations depicted on the PCA analysis (Supplementary Table S1). Data points from the same MG grouping together showed similarities for the variables of the PCA, reinforcing the dominant influence of phenology previously observed (Fig. 3a,c,e). The MGs, from the shortest to the longest, grouped in the biplot along the first principal component. For instance, data from groups 4.4 and 3.2 were associated with higher AUC when compared to 0.3 MG.
Principal component analysis of crop traits for each combination of treatment and soybean maturity group. Concentration ellipses includes points each maturity group. Traits are maximum rate (MaxRate), residuals from seed protein concentration (Res_Prot), seed yield (Res_Yield), total biomass (Res_Biom), harvest index (Res_HI), oil concentration (Res_Oil), maximum RAU during the growing season (RAUmax), RAU at R6 (RAUR6), relative AUC (AUC), thermal time for RAUmax (tmax), soil organic matter (OM), soil pH, clay, sand, and cation exchange capacity (CEC) from each location.
Soil attributes were positively correlated in the first principal component, discriminating the locations with shorter maturity groups and higher values in soil attributes. Interestingly, AUC was negatively correlated with OM, pH, clay and sand percentage, and the RAUR6 and rate were positively correlated with the OM and sand (Supplementary Table S1). Seed yield correlated positively with both HI and biomass and negatively with RAUmax, RAUR6, and protein concentration. The AUC correlated positively with RAUmax and tmax and negatively with the maximum RAU rate.
Discussion
Biological N fixation in soybean has been quantified at different scales, from field to country22,23,24. The range of RAU in unfertilized controls in our study ranged from 48 to 93%. This compares with an average of 60% of N derived from BNF for the US23. In Argentina, BNF in 86 location-years averaged 60% and ranged from 12 to 90%24. In Brazil, measurements in 6 environments returned an average of 81% and a range from 69 to 94%22. All these studies reflected a similar BNF ceiling around 90%, comparable to the maximum recently reported by Ciampitti and Salvagiotti25.
Nitrogen fertilization reduced BNF and increased seed yield by enhancing C allocation to seed
Phenology was the main source of variation in seed yield and traits associated with BNF clustered with maturity group (Figs 2 and 4). Soybean maturity group influences not only phenology but also growth, and allocation of biomass and nitrogen26. Variation in BNF with MG relates to both the duration of the reproductive period when BNF and biomass growth rate peak, and the delay in the exponential phase of BNF27,28,29. In our study, application of N reduced peak and altered dynamics of RAU during the season (Fig. 2), but phenology masked the association between N fixation and seed yield. After removing the dominant effect of phenology, crop yield declined with increasing BNF (Figs 3 and 4). This effect of maturity group has not been considered in previous comprehensive studies20,30.
Reduced HI was the primary driver of the reduction in seed yield with increasing RAUR6, with an additional weak but significant reduction in oil seed concentration. Low yielding environments showed a steeper decline of HI with increasing RAUR6 (Fig. 3d), highlighting the interaction with overall environmental conditions affecting dry matter allocation; stress during reproduction often reduces HI31,32.
Changes in plant C allocation in association with BNF have been reported at different levels of organization and time scales. Reduction in BNF associated with phosphorus deficiency altered short-term allocation of C in lupin (Lupinus luteus), reducing photosynthesis:respiration ratio, and increasing the ratio between growth respiration and maintenance respiration33. Likewise, low magnesium supply altered carbohydrate allocation in soybean, increasing sucrose and starch allocation to leaves that later limited nodule growth34. Decreases in biomass allocation in seeds for chickpea (Cicer arietinum L.) and pea (Pisum sativum L.) were reported with increasing BNF7,35. Re-analyzing the data of Sadras et al.7, where 20 chickpea varieties were grown in 8 environments, HI declined witn BNF at a rate of 0.0022 units per %BNF (Supplementary Fig. S1), in comparison with 0.0011 units per %RAUR6 for soybeans in our study; similar to our trial, the decline in HI with BNF was larger for stressed chickpea crops.
Generically, plants require 1 g of glucose to produce either 0.33 g of lipid, 0.40 g of protein, or 0.83 g of carbohydrates36. Reducing oil concentration in seed is therefore an energetically effective way to meet the cost of BNF, as found in this study (Fig. 3e). This is in contrast to previous studies where seed protein concentration was reduced and oil concentration did not change in response to BNF37,38.
Our findings are in contrast to other studies where N fertilizer reduced BNF but did not increase soybean seed yield39,40. For example, Santachiara et al.39 found no seed yield response in heavily fertilized crops (600 kg N ha−1 spread over the season) that reduced BNF to 16% in comparison to 69% in unfertilised controls. However, Santachiara et al.39 neither report seed yield in equivalent glucose nor changes in protein and oil concentrations. Results from these experiments can be influenced from soil variables influencing BNF activity. In our study, long-term stable soil attributes were included in the analysis. Organic matter was positively correlated with RAUR6 and the maximum RAU rate but negatively with the AUC (Supplementary Table S1). The negative association between AUC and soil organic matter might be attributed to the soil N mineralized from N organic fraction during the season41. Collino et al.24 compared average BNF in soybean production systems of Argentina and Brazil, and attributed the lower BNF in the former to better soil fertility.
Of the five putative mechanisms to account for the metabolic cost of BNF, enhanced sink-driven photosynthesis13 and reduced root:shoot ratio are the remaining hypotheses to explain the lack of seed yield response with reduced BNF in Santachiara et al.39. Implicit in the photosynthesis hypothesis is that soybean seed yield is sink-limited; although unlikely, it requires further research. Enhancing photosynthesis by increasing atmospheric carbon dioxide, Ryle et al.42 reported increases in nodule activity for shadowed white clover (Trifolium repens L.) plants but not in their non-stressed counterparts. It is likely that effects of photosynthetic rates on nodule activity depend on reserve carbohydrates43, suggesting a link with the differential trends in HI between favourable and poor environments observed in this study. An alternative, less explored mechanism for the maintenance of seed yield in crops relying on N fixation relative to fertilized crops is the reduction in root:shoot ratio; reduced root:shoot ratio is a generic response of plants to high availability of soil N15.
Agronomic and breeding implications
Soybean plays a relevant role in crop rotations44 and is a major source of oil and protein worldwide. Improving BNF can be achieved by breeding and selection targeting the plant, the N-fixing bacteria, and better matching plant and bacteria45,46,47. Selection for maintenance of BNF in dry soil has been proposed to improve seed yield of soybean under drought48,49. Sinclair et al.49,50 combined ureide concentration in petioles and acetylene reduction activity to test this proposition. Selected lines were compared with high-yielding commercial cultivars under broad environmental conditions. Two lines were identified that outperformed commercial checks under water deficit, but trade-offs were apparent under high yielding conditions. In this context, the trade-off between BNF and seed yield mediated by HI needs attention. Solving this trade-off needs quantification of the costs (seed yield reduction), agronomic and environmental benefit of BNF. Selection for high biomass partitioning to seed in genotypes growing under low concentration of soil nitrate represents a possible breeding strategy as higher rates of BNF are expressed. In both soybean51 and common bean (Phaseolus vulgaris L.)52, sensitivity of N fixation to soil mineral N varies with genotype. In alfalfa (Medicago sativa L.), selection for BNF improved plant growth53.
Conclusion
Seasonal characterizations over a wide range of agronomic and environmental conditions revealed that N application reduced maximum RAU at R6, particularly for late applications. At the crop level, soybean met the cost of BNF by a reduction in seed yield mediated by lower HI, particularly in stressful environments, and a secondary contribution of reduced seed oil concentration. The lower-level mechanisms underlying shifts in HI associated with BNF warrant further attention.
Methods
Experimental sites and treatments
During the 2016 growing season, soybean N fertilization studies were replicated in 23 sites across the US Midwest in a latitude range from 34°16′ N to 48°14′ N and from 90°29′ W 98°53′ W (Fig. 1, Table 1). Due to the range of latitudes between locations, the length of the growing season differs among sites. Thus, sowing dates and MG were considered following local management practices and recommendations which ranged from 0 to IV due to the large range of latitude in the locations54. The seeding rate of 300,000 seeds per hectare, targeted maximum seed yield. Crops were rainfed and received supplementary irrigation to avoid severe water stress (Table 1). On-site weather stations recorded daily temperature, precipitation, and relative humidity; the vapor pressure deficit (VPD, kPa) was estimated using the maximum daily temperature and relative humidity55. Soil parameters from every location are presented in Table 1. Data for percentages of clay and sand, organic matter (OM), soil pH and cation exchange capacity (CEC) was extracted from the California Soil Resource Lab (http://casoilresource.lawr.ucdavis.edu, accessed 11 June 2018) using latitude and longitude coordinates from each experiment.
Four treatments were established: an unfertilized control, and 112 kg N ha−1 as urea (46-0-0 N-P-K,) at one of three stages: at sowing, at V4 (fourth-leaf), and between R2 (full flowering) to R3 (beginning of pod formation)56. The experimental design at each location was a randomized complete block with three replicates. Plot size was 8.4 m long by eight rows at 0.76 m row spacing. The supply of other nutrients was done with N-free fertilizer.
Phenology, biomass, seed yield, harvest index, seed protein and oil concentration
Plant development stages in relation with calendar time is usually referred as phenology. Phenological stages were recorded during the season following Fehr and Caviness56. Shoot biomass samples were collected at the R8 stage (full maturity) from 1.6 linear m and fractioned into stem, leaves, and seeds. The relative proportion of seeds to the total shoot biomass was quantified as the harvest index (HI)57. Variations on this ratio can be associated with the influence of the environmental effects on seed yield and biomass production. Seed yield was collected from two-central rows at maturity and adjusted to 13.5 g 100 g−1 seed moisture content. Seed samples were collected from harvest for oil and protein determination by near infrared (NIR) spectroscopy using a completely automated Fourier Transform-IR imaging Microscope (Hyperion 3000, Bruker Optics, Ettlingen, Germany) and a sample of >50 seeds. Seed protein and oil concentrations are reported on dry weight basis (Table 2).
Seasonal dynamics of BNF
Biological N fixation was measured four times, at V4 (fourth leaf), R2 (full flowering)-R3 (beginning of pod formation), R6 (full seed), and R8 (full maturity) using main stem samples. Stems were dried at 65 °C until constant weight and ground to pass through a 2-mm sieve. The BNF percentage was calculated as the relative abundance of ureide-N (RAU) in the main stems using the procedure of Hungria and Araujo58. The RAU was calculated as a function of ureides nitrate-N (NO3-N) concentration59.
Time units used to measure the progress of RAU during the season was thermal units (degree-days; °Cd) to account for thermal differences in growing conditions and be independent from the temperature in which different developmental stages occurs. A degree-day is the result from every degree on the daily mean temperature above the base temperature60. Thus, cumulative thermal time was calculated as:
where Tmax and Tmin is the maximum and minimum daily air temperature (°C), and Tb is the base temperature (8 °C)61.
Changes in RAU during the growing season has been described as a sigmoidal pattern with a slow increase early in the season and a maximum attainable between R5 (beginning of seed filling) and R6 (full seed)62. However, owing to the large variation in genotypes and growing conditions, RAU at R6 varied widely. To account for this variation, we used RAU at R6 in unfertilized controls to split data into three groups: below the 25th quartile (low BNF), between the 25th–75th quartiles (medium BNF), and above the 75th quartile (high BNF). The low BNF comprised five sites with RAU below 72%; the medium BNF included twelve sites with RAU from 72 to 88%, and the high BNF group comprised of six sites with RAU above 88%. For the data combined for each group, the seasonal RAU evolution was described with the beta growth function63 with three parameters:
where t is the thermal time from V2 (vegetative leaf), RAUmax is the maximum RAU at thermal time tmax, and tm is the thermal time for maximum RAU growth rate. Biological meaning on parameters allowed us to make inferences on the magnitude of the N treatments on the RAU dynamics by statistical comparisons. Differences between parameters of equation (2) were tested using the Akaike’s Information Criteria (AIC) by performing pairwaise comparisons of individual curves against a global fit. Maximum rate, t0.5, and the AUC where compared using the 95% confidence interval. Both RAU and thermal time were estimated through least squares mean analysis by fitting a mixed model with PROC MIXED procedure64 (lsmeans statement) to adjust corrected means to the factors of the model. For this analysis, treatment and locations were considered as fixed factors, and block was nested within location as a random factor. The goodness of fit of the model was assessed with the coefficient of determination (R2) and the standard deviation of residuals (Syx)65.
Using equation (3), we derived three related traits: the AUC to integrate seasonal N fixation66 normalized to the maximum of the data set; t0.5 the thermal time when RAU is 50% of RAUmax; and the maximum rate of RAU expressed in changes of % units of RAU per unit of thermal time (°Cd)63:
The first derivative of equation (3) with respect to thermal time can be solved to calculate the RAU rate changes across the growing season63:
Same approach has been utilized to describe other biological process such as N uptake rate in corn67 or grain growth rate68.
Analysis of treatment effects and associations between traits
Analysis of variance (ANOVA) was used to investigate effects of treatments on crop traits (seed yield, biomass, HI, RAUR6, seed protein and oil concentration). Sources of variation in ANOVA included N treatment, location, and their interaction as fixed factors, and block as a random effect nested within location; this analysis was implemented by using the R software (version 3.4.0, lme4 package, lmer function)69,70.
The effects of BNF on seed yield, biomass, HI, seed protein and oil concentration were analyzed in two steps. First, due to the geographical distribution of the experiments, responses on crop traits are confounded with the different duration of the developmental stages. Thus, effects of phenology were captured with non-linear models:
where Y is the trait, X is the thermal time to R6 (°Cd), and a, b, TTo, and d, are parameters. Next, linear regressions and quantile regressions were fitted between residuals of these models and RAUR6. This simple approach on the use of residuals allows to netting out71 the effect of the phenology on the traits observed when are regressed against RAUR6. There are relationships on other parts of the distribution of the response variable that can provide more complete view of the processes studied besides of the mean effect observed. Slopes from quantile regression analysis estimate the changes at the maximum and minimum response that can be missed when other regression methods are used72. Thus, regressions for 0.99 and 0.01 quantiles capture the boundaries of the relationships, and were fit in R (quantreg package73). The rest of linear and non-linear regression analyses, computation of AUC, and estimation and comparison of parameters from equations (3) and (5) were performed using GraphPad Prism74.
Principal component analysis (PCA) was used to analyze general associations between traits allowing the identification of any grouping association within the data set when environmental and crop attributes are analyzed together75. Data were classified according to MG, which in turn had a geographical correlation (Table 1). Traits included RAUmax, tmax, maximum rate of RAU evolution, seed protein and oil concentration, AUC, residuals from seed yield, biomass, and HI vs thermal time to R6 relationship, and soil attributes (clay, sand, organic matter, pH, and CEC). Principal component analysis was fit using the “FactoMineR” package in R76. Pearson correlation coefficients were calculated to complement associations found in the PCA.
References
USDA. USDA National Agricultural Statistics Service. USDA–NASS, Washington, DC (2017).
Jin, J. et al. Agronomic and physiological contributions to the yield improvement of soybean cultivars released from 1950 to 2006 in Northeast China. F. Crop. Res. 115, 116–123 (2010).
Balboa, G. R., Sadras, V. O. & Ciampitti, I. A. Shifts in Soybean Yield, Nutrient Uptake, and Nutrient Stoichiometry: A Historical Synthesis-Analysis. Crop Sci. 58, 43 (2018).
Tamagno, S. et al. Nutrient partitioning and stoichiometry in soybean: A synthesis-analysis. F. Crop. Res. 200, 18–27 (2017).
Trang, K. M. & Giddens, J. Shading and Temperature as Environmental-Factors Affecting Growth, Nodulation, and Symbiotic-N2 Fixation by Soybeans. Agron. J. 72, 305–308 (1980).
de Silva, M., King, A., Purcell, L. C. & Andy, C. Soybean Petiole Ureide Response to Water Deficits and Decreased Transpiration. Crop Sci. 36, 611–616 (1996).
Sadras, V. O., Lake, L., Li, Y., Farquharson, E. A. & Sutton, T. Phenotypic plasticity and its genetic regulation for yield, nitrogen fixation and δ13C in chickpea crops under varying water regimes. J. Exp. Bot. 67, 4339–4351 (2016).
Ferguson, B., Lin, M.-H. & Gresshoff, P. M. Regulation of legume nodulation by acidic growth conditions. Plant Signal. Behav. 8, e23426 (2013).
Eaglesham, A. R. J., Ayanaba, A., Rangarao, U. & Eskew, D. L. Mineral N effects on cowpea and soybean crop in a Nigerian soil. II. Amounts of N fixed and accrual to the soil. Plant Soil 68, 183–192 (1982).
Danso, S. K. A., Hera, C. & Douka, C. Nitrogen fixation in soybean as influenced by cultivar and Rhizobium strain. Plant Soil 99, 163–174 (1986).
Purcell, L. C., De Silva, M., King, C. A. & Kim, W. H. Biomass accumulation and allocation in soybean associated with genotypic differences in tolerance of nitrogen fixation to water deficits. Plant Soil 196, 101–113 (1997).
Patterson, T. G. & LaRue, T. A. N2 Fixation (C2H2) and Ureide Content of Soybeans: Environmental Effects and Source-Sink Manipulations. Crop Sci. 23, 819–824 (1983).
Kaschuk, G., Kuyper, T. W., Leffelaar, P. A., Hungria, M. & Giller, K. E. Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biol. Biochem. 41, 1233–1244 (2009).
Salon, C. et al. Grain legume seed filling in relation to nitrogen acquisition: A review and prospects with particular reference to pea. Agronomie 21, 539–552 (2001).
Lambers, H. ‘The functional equilibrium’, nibbling on the edges of a paradigm. Netherl. J. Agric. Sci. 31, 305–311 (1983).
Cafaro La Menza, N., Monzon, J. P., Specht, J. E. & Grassini, P. Is soybean yield limited by nitrogen supply. F. Crop. Res. 213, 204–212 (2017).
Tamagno, S. & Ciampitti, I. A. Seed yield and biological N fixation for historical soybean genotypes. Kansas Field Research Report (2017).
Rotundo, J. L., Naeve, S. L. & Miller-Garvin, J. E. Regional and Temporal Variation in Soybean Seed Protein and Oil across the United States. Crop Sci. 56, 797 (2016).
Medic, J., Atkinson, C. & Hurburgh, C. R. Current knowledge in soybean composition. JAOCS, J. Am. Oil Chem. Soc. 91, 363–384 (2014).
Mourtzinis, S. et al. Soybean response to nitrogen application across the United States: A synthesis-analysis. F. Crop. Res. 215, 74–82 (2018).
Hay, R. K. M. Harvest index: a review of its use in plant breeding and crop physiology. Ann. Appl. Biol. 126, 197–216 (1995).
Hungria, M., Franchini, J. C., Campo, R. J. & Graham, P. H. The importance of nitrogen fixation to soybean cropping in South America. In Nitrogen fixation in agriculture, forestry, ecology, and the environment 25–42 (Springer, 2005).
Herridge, D. F., Peoples, M. B. & Boddey, R. M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 311, 1–18 (2008).
Collino, D. J. et al. Biological nitrogen fixation in soybean in Argentina: relationships with crop, soil, and meteorological factors. Plant Soil 392, 239–252 (2015).
Ciampitti, I. A. & Salvagiotti, F. New insights into soybean biological nitrogen fixation. Agron. J. In press, 1–42 (2018).
Santachiara, G., Borrás, L. & Rotundo, J. L. Physiological Processes Leading to Similar Yield in Contrasting Soybean Maturity Groups. Agron. J. 109, 158 (2017).
Keyser, H. H. & Li, F. Potential for increasing biological nitrogen fixation in soybean. Plant Soil 141, 119–135 (1992).
Patterson, T. G. & LaRue, T. A. Nitrogen Fixation by Soybeans: Seasonal and Cultivar Effects, and Comparison of Estimates. Crop Sci. 23, 488 (1983).
Havelka, U. D., Boyle, M. G. & Hardy, R. W. F. Biological Nitrogen Fixation. In Nitrogen in agricultural soils 365–422 (American Society of Agronomy, Crop Science Society of America, Soil Science Society of America, 1982).
Salvagiotti, F. et al. Nitrogen uptake, fixation and response to fertilizer N in soybeans: A review. F. Crop. Res. 108, 1–13 (2008).
Sinclair, T. R. Historical Changes in Harvest Index and Crop Nitrogen Accumulation. Crop Sci. 38, 638–643 (1998).
Shiraiwa, T., Sakashita, M., Yagi, Y. & Horie, T. Nitrogen Fixation and Seed Yield in Soybean under Moderate High-Temperature Stress. Plant Prod. Sci. 9, 165–167 (2006).
Kleinert, A., Venter, M., Kossmann, J. & Valentine, A. The reallocation of carbon in P deficient lupins affects biological nitrogen fixation. J. Plant Physiol. 171, 1619–1624 (2014).
Peng, W. T. et al. Magnesium promotes root nodulation through facilitation of carbohydrate allocation in soybean. Physiol. Plant. 0–1, https://doi.org/10.1111/ppl.12730 (2018).
Voisin, A. S., Salon, C., Munier-Jolain, N. G. & Ney, B. Effect of mineral nitrogen on nitrogen nutrition and biomass partitioning between the shoot and roots of pea (Pisum sativum L.). Plant Soil 242, 251–262 (2002).
De Vries, F. W. T. P., Brunsting, A. H. M. & Van Laar, H. H. Products, requirements and efficiency of biosynthesis a quantitative approach. J. Theor. Biol. 45, 339–377 (1974).
Purcell, L. C., Serraj, R., Sinclair, T. R. & De, A. Soybean N2 fixation estimates, ureide concentration, and yield responses to drought. Crop Sci. 44, 484–492 (2004).
Ray, J. D., Fritschi, F. B. & Heatherly, L. G. Large applications of fertilizer N at planting affects seed protein and oil concentration and yield in the Early Soybean Production System. F. Crop. Res. 99, 67–74 (2006).
Santachiara, G., Salvagiotti, F., Gerde, J. A. & Rotundo, J. L. Does biological nitrogen fixation modify soybean nitrogen dilution curves. F. Crop. Res. 223, 171–178 (2018).
Kaschuk, G., Nogueira, M. A., de Luca, M. J. & Hungria, M. Response of determinate and indeterminate soybean cultivars to basal and topdressing N fertilization compared to sole inoculation with Bradyrhizobium. F. Crop. Res. 195, 21–27 (2016).
Thies, J. E., Singleton, P. W. & Bohlool, B. B. Modeling symbiotic performance of introduced rhizobia in the field by use of indices of indigenous population size and nitrogen status of the soil. Appl. Environ. Microbiol. 57, 29–37 (1991).
Ryle, G. J. A., Powell, C. E. & Gordon, A. J. Responses of N2-fixation linked respiration to host-plant energystatus in white clover acclimated to a controlled environment. 39, 879–887 (1988).
Walsh, K. B., Thorpe, M. R. & Minchin, P. E. H. Photoassimilate partitioning in nodulated soybean II. The effect of changes in photoassimilate availability shows that nodule permeability to gases is not linked to the supply of solutes or water. J. Exp. Bot. 49, 1827–1834 (1998).
Sisworo, W. H., Mitrosuhardjo, M. M., Rasjid, H. & Myers, R. J. K. The relative roles of N fixation, fertilizer, crop residues and soil in supplying N in multiple cropping systems in a humid, tropical upland cropping system. Plant Soil 121, 73–82 (1990).
Emerich, D. W. & Krishnan, H. Nitrogen Fixation in Crop Production. (American Society of Agronomy, Crop Science Society of America, Soil Science Society of America, https://doi.org/10.2134/agronmonogr52 (2009).
Denison, R. F. Evolutionary tradeoffs as opportunities to improve yield potential. F. Crop. Res. 182, 3–8 (2015).
Kiers, E. T., Ratcliff, W. C. & Denison, R. F. Single-strain inoculation may create spurious correlations between legume fitness and rhizobial fitness. New Phytol. 198, 4–6 (2013).
Patterson, R. P. & Hudak, C. M. Drought-avoidant soybean germplasm maintains nitrogen-fixation capacity under water stress. Plant Soil 186, 39–43 (1996).
Sinclair, T. R. et al. Drought tolerance and yield increase of soybean resulting from improved symbiotic N2 fixation. F. Crop. Res. 101, 68–71 (2007).
Sinclair, T. R. Challenges in breeding for yield increase for drought. Trends in Plant Science 16, 289–293 (2011).
Santachiara, G., Borrás, L., Salvagiotti, F., Gerde, J. A. & Rotundo, J. L. Relative importance of biological nitrogen fixation and mineral uptake in high yielding soybean cultivars. Plant Soil, https://doi.org/10.1007/s11104-017-3279-9 (2017).
Hardarson, G. Methods for enhancing symbiotic nitrogen fixation. Plant Soil 152, 1–17 (1993).
Barnes, D. K., Sheaffer, C. C., Heichel, G. H., Smith, D. M. & Peaden, R. N. Registration of ‘Nitro’alfalfa. Crop Sci. 28, 718 (1988).
Mourtzinis, S. & Conley, S. P. Delineating soybean maturity groups across the United States. Agron. J. 109, 1397–1403 (2017).
Howell, T. & Dusek, D. Comparison of vapor-pressure-deficit calculation methods – Southern High Plains. J. Irrig. Drain. Eng. 121, 191–198 (1996).
Fehr, W. R. & Caviness, C. E. Stages of soybean development. (Cooperative Extension Service; Agriculture and Home Economics Experiment Station, Iowa State University of Science and Technology, 1977).
Donald, C. M. & Hamblin, J. The biological yield and harvest index of cereals as agronomic and plant breeding criteria. Adv. Agron. 28, 361–405 (1976).
Hungria, M. & Araujo, R. S. Manual de métodos empregados em estudos de microbiologia agrícola. (Embrapa-Serviço de Produção e Informação, 1994).
Schweiger, P., Hofer, M., Hartl, W., Wanek, W. & Vollmann, J. N2 fixation by organically grown soybean in Central Europe: Method of quantification and agronomic effects. Eur. J. Agron. 41, 11–17 (2012).
Monteith, J. L. Climate. in Ecophysiology ofTropical Crops (eds Alvim, P. T. & Kozlowski, T. T.) 1–27 (Academic Press, https://doi.org/10.1016/B978-0-12-055650-2.50006-X (1977).
Wu, T. et al. Analysis of the independent- and interactive-photo-thermal effects on soybean flowering. J. Integr. Agric. 14, 622–632 (2015).
Herridge, D. F. Use of the ureide technique to describe the nitrogen economy of field-grown soybeans. Plant Physiol. 70, 7–11 (1982).
Yin, X., Goudriaan, J., Lantinga, E. A., Vos, J. & Spiertz, H. J. A flexible sigmoid function of determinate growth. Ann. Bot. 91, 361–371 (2003).
SAS Institute. SAS Software Release 9.2. SAS Institute Cary, NC (SAS Institute Cary, NC, 2012).
Archontoulis, S. V. & Miguez, F. E. Nonlinear regression models and applications in agricultural research. Agron. J. 107, 786–798 (2015).
Gagnon, R. C. & Peterson, J. J. Estimation of confidence intervals for area under the curve from destructively obtained pharmacokinetic data. J. Pharmacokinet. Biopharm. 26, 87–102 (1998).
Osterholz, W. R., Rinot, O., Liebman, M. & Castellano, M. J. Can mineralization of soil organic nitrogen meet maize nitrogen demand? Plant Soil 415, 73–84 (2017).
Meade, K. A., Cooper, M. & Beavis, W. D. Modeling biomass accumulation in maize kernels. F. Crop. Res. 151, 92–100 (2013).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/ (2017).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Software; Vol 1, Issue 1 (2015).
Greene, W. H. Econometric Analysis. Upper Saddle River, New Jersey, USA. (Prentice Hall, 2003).
Cade, B. S. & Noon, B. R. A gentle introduction to quantile regression. Front. Ecol. Environ. 1, 412–420 (2003).
Koenker, R. quantreg: Quantile Regression. R package version 5.35. https://CRAN.R-project.org/package=quantreg (2017).
Motulsky, H. & Christopoulos, A. Fitting models to biological data using linear and nonlinear regression. A practical guide to curve fitting. Graph Pad Software Inc., San Diego CA, www.graphpad.com (2003).
Ju, J., Banfelder, J. & Skrabanek, L. Quantitative Understanding in Biology Principal Component Analysis. 1–12 Available at, http://physiology.med.cornell.edu/people/banfelder/qbio/lecture_notes/3.4_Principal_component_analysis.pdf (Accessed: 21st October 2018) (2017).
Lê, S., Josse, J. & Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. Software, Vol 1, Issue 1, https://doi.org/10.18637/jss.v025.i01 (2008).
FAO (Land and Water Division). Guidelines for Soil Description (2006).
Acknowledgements
Mr. Luiz HM Rosso and Ms. Anelise Lencina are acknowledged for their technical and organizational assistance in the laboratory. We thank Dr. Rotundo for useful discussions. We thank WinField United and the International Plant Nutrition Institute (IPNI, Project GBL 62) for supporting this research study. This is Contribution no. 18-502-J from the Kansas Agricultural Experiment Station. Mention of a proprietary product or trade name does not constitute a recommendation or endorsement by the US Department of Agriculture. The USDA is an equal opportunity provider and employer. Publication of this article was funded in part by the Kansas State University Open Access Publishing Fund.
Author information
Authors and Affiliations
Contributions
Study design: J.W.H., I.A.C. and S.T. Data collection: S.T., J.W.H. and P.R.A. Data analysis: S.T., I.A.C. and V.O.S. Results interpretation: S.T., I.A.C. and V.O.S. Manuscript preparation: S.T., V.O.S., I.A.C. and P.R.A.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tamagno, S., Sadras, V.O., Haegele, J.W. et al. Interplay between nitrogen fertilizer and biological nitrogen fixation in soybean: implications on seed yield and biomass allocation. Sci Rep 8, 17502 (2018). https://doi.org/10.1038/s41598-018-35672-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35672-1
Keywords
This article is cited by
-
Influence of nitrogen fertilization, seed inoculation and the synergistic effect of these treatments on soybean yields under conditions in south-western Poland
Scientific Reports (2024)
-
Nitrogen Acquisition and Allocation Traits in Peanuts with Different Nodulation Efficiency Revealed by the 15N Tracer Analysis
Journal of Soil Science and Plant Nutrition (2024)
-
Environmental variables controlling biological nitrogen fixation in soybean
Symbiosis (2024)
-
Identification of geographical origin and adulteration of Northeast China soybeans by mid-infrared spectroscopy and spectra augmentation
Journal of Consumer Protection and Food Safety (2024)
-
Optimization of evaluation method for low nitrogen tolerance in soybean germplasm seedlings
Plant Growth Regulation (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.