Abstract
In High Arctic tundra ecosystems, seabird colonies create nitrogen cycling hotspots because of bird-derived labile organic matter. However, knowledge about the nitrogen cycle in such ornithocoprophilous tundra is limited. Here, we determined denitrification potentials and in-situ nitrous oxide (N2O) emissions of surface soils on plant-covered taluses under piscivorous seabird cliffs at two sites (BL and ST) near Ny-Ålesund, Svalbard, in the European High Arctic. Talus soils at both locations had very high denitrification potentials at 10 °C (2.62–4.88 mg N kg−1 dry soil h−1), near the mean daily maximum air temperature in July in Ny-Ålesund, with positive temperature responses at 20 °C (Q10 values, 1.6–2.3). The talus soils contained abundant denitrification genes, suggesting that they are denitrification hotspots. However, high in-situ N2O emissions, indicating the presence of both active aerobic nitrification and anaerobic denitrification, were observed only at BL (max. 16.6 µg N m−2 h−1). Rapid nitrogen turnover at BL was supported by lower carbon-to-nitrogen ratios, higher nitrate content, and higher δ15N values in the soils at BL compared with those at ST. These are attributed to the 30-fold larger seabird density at BL than at ST, providing the larger organic matter input.
Similar content being viewed by others
Introduction
Arctic tundra ecosystems are experiencing rapid warming due to climate change1,2,3,4. The soil in tundra ecosystems contains large stocks of organic matter, carbon (C), and nitrogen (N) because the cold and wet climate limits organic matter turnover1. Tundra ecosystems are therefore under limitation of inorganic N such as ammonium and nitrate, like many other terrestrial ecosystems5,6,7,8. However, mineralisation in mesic meadow tundra produces a large amount of ammonium9, suggesting that N turnover in tundra ecosystems can vary depending on the local conditions. The rapid warming due to climate change most likely further perturbs organic matter and N turnover in Arctic tundra ecosystems.
Nitrogen turnover is controlled by various processes comprising N cycle. For example, organic N is decomposed to ammonium by mineralisation, part of which is then oxidised to nitrate, via nitrite, by nitrification; both are regulated by microbial processes10,11,12,13. Ammonium and nitrate are also input from the atmosphere to terrestrial ecosystems by deposition14,15. Denitrification is the series of microbial processes that reduce nitrate to dinitrogen (N2) via nitrous oxide (N2O) in an anaerobic condition; these processes require nitrate and organic matter as substrates10,11,12,13. Denitrification returns N from the terrestrial ecosystem to the atmosphere through the emission of N2 and N2O.
N2O is an environmentally important compound because of its strong positive radiative forcing and ability to deplete stratospheric ozone16,17. In anaerobic soils, N2O is produced mainly by denitrification10,12,13, as well as by nitrification in aerobic soils and other processes11. Nitrification provides an important supply of nitrate as the substrate for denitrification18,19. N2O in soil is reduced to N2 under a strong anaerobic condition (complete denitrification). Under a partial anaerobic condition, N2O production outweighs the consumption and a part of the excess N2O in soil is released to the atmosphere11,20. Soils under natural vegetation are the world’s largest source of atmospheric N2O, accounting for 37% of global emissions (6.6–17.9 Tg N yr−1)16.
Although tundra soils are traditionally considered as poor N2O emitters16,21,22,23, recent studies in the Arctic have provided evidence to the contrary. For example, an increased N input has been shown to induce the emission of N2O from peat due to the decline of oligotrophic mosses24; specific areas of thermokarst (permafrost collapse due to ground ice melt) can be active N2O emitters, particularly in the part of thermo-erosion gullies25; and peat circles (bare peat surfaces) emit N2O at high rates during the growing season (1.2–20 mg N m−2 d−1)19. Thus, further research to elucidate how Arctic tundra soils contribute to N2O emissions to the atmosphere is needed.
Bird colonies accelerate soil N turnover via continuous inputs of nutrient-rich labile organic matter in the form of bird-derived faeces, feathers, egg shells, and carcasses. Consequently, bird-affected soils are high emitters of nitrogenous gases such as ammonia26,27,28,29 and N2O30,31,32,33. In the High Arctic tundra, taluses (steep slopes) at the bottom of cliffs inhabited by seabirds are known to be the area, which receives intensive organic matter inputs34,35,36. Such taluses are covered by vegetation consisting of mosses and compact vascular plants34. Taluses are formed from debris that has been fallen from the cliff and are therefore well drained topographically; however, because mosses are the parent material of the surface soil on bird-affected taluses, this soil has a high water retentivity37. Thus, bird-affected taluses can become denitrification and N2O emission hotspots because an anaerobic condition is developed in the moist surface soils and the continuous supply of labile organic matter and nitrate provided by mineralisation and nitrification facilitates denitrification. Furthermore, the mixture of drained and moist conditions produces a partial anaerobic environment that promotes the excess N2O production.
In the present study, we hypothesised that organic matter inputs from seabird cliffs in the High Arctic accelerate N turnover in talus soil, which produces denitrification hotspots with possible N2O emissions (Fig. 1). Specifically, we hypothesised that (1) the surface soil on seabird-affected taluses has a high denitrification potential, (2) seabird-affected taluses emit N2O to the atmosphere, and (3) these denitrification potentials and N2O emissions decrease with increasing distance from the bottom of the cliff in association with the decrease in organic matter input.
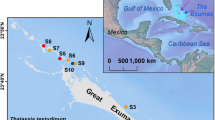
Denitrification potential corresponds to the maximum N2O production rate under a strong anaerobic condition in the presence of abundant substrates and at a constant temperature. To determine denitrification potentials, in-situ N2O emissions, and other relevant properties of seabird-affected talus soil, we conducted field surveys at two sites, one in Blomstrandhalvøya (BL) and the other in Stuphallet (ST), near Ny-Ålesund, Svalbard, in the European High Arctic (Fig. 2; Supplementary Information). Each site comprises a talus at the base of a cliff inhabited by breeding piscivorous seabirds and a control area not affected by seabirds. The field survey was conducted in July during the nightless midsummer when ca. 400 pairs of black-legged kittiwakes (Rissa tridactyla) and a few northern fulmars (Fulmarus glacialis) inhabited the cliff at the BL site and ca. 10 pairs of Atlantic puffins (Fratercula arctica) and ca. 5 pairs of northern fulmars inhabited on the cliff at the ST site.
Results
Denitrification potentials
The talus soils (0–5 cm depth) at both BL and ST had high denitrification potentials at all locations (upper, middle, and lower parts of the taluses; Fig. 2; Supplementary Information) compared with those at control plots (Fig. 3). Denitrification potentials were measured at 10 and 20 °C, with the former temperature approximating the mean daily maximum air temperature in July in Ny-Ålesund and the latter being used to examine how the soils responded to higher-than-usual temperatures. The mean denitrification potentials for the soils from the three parts of the taluses at 10 °C were 2.62–4.88 and 3.38–4.20 mg N kg−1 dry soil h−1 at BL and ST, respectively, and those at 20 °C were 6.06–11.1 and 5.59–6.99 mg N kg−1 dry soil h−1 at BL and ST, respectively; significant positive temperature responses (P < 0.001) were observed with Q10 values of 1.9–2.3 at BL and 1.6–1.8 at ST. The mean denitrification potentials of the soils in the control areas were a few hundredths of the values recorded for the talus soils (Fig. 3). At the two taluses, the effect of distance from the cliff bottom (distance) on denitrification potential and the interaction between site and incubation temperature were not significant (Fig. 3).
Denitrification potentials of the soils (0–5 cm depth) at 10 and 20 °C. Bars denote standard deviation (n = 3). A normal distribution assumption was accepted for the talus soil data. BL, study site in Blomstrandhalvøya; ST, study site in Stuphallet; T-U, talus (upper part); T-M, talus (middle part); T-L, talus (lower part); C, control area not directly affected by seabirds; n.s., not significant.
In-situ N2O emissions
In-situ N2O emissions from undisturbed soil surfaces were recorded at the talus at BL, but were mostly below the detection limit at the talus at ST and the two control areas (Fig. 4). The difference in N2O emissions between the two taluses was significant (P = 0.016). The mean N2O emissions at the three parts of the talus at BL were in the range of 2.1–7.2 µg N m−2 h−1, with the highest emission (16.6 µg N m−2 h−1) recorded at a plot on the middle part of the talus (Fig. 4). The effect of distance on N2O emission was not significant (P = 0.072) (Fig. 4).
In-situ N2O emission flux. Bars denote standard deviation (n = 3). A log-normal distribution assumption with Yeo–Johnson transformation was accepted for the talus soil data. BL, study site in Blomstrandhalvøya; ST, study site in Stuphallet; T-U, talus (upper part); T-M, talus (middle part); T-L, talus (lower part); C, control area not directly affected by seabirds; n.s., not significant.
Soil properties related to N turnover
The talus soils at both sites were organic soils originating from plant litter, mostly mosses, with the following characteristics favourable for denitrification compared with those of the soils in control areas: a high water content (58.5–82.0% w/w), which promotes the development of anaerobic conditions, and high total C content (20.5–38.8%) and total N content (1.79–2.77%), which suggests a high input of organic matter (Table 1). In addition, the talus soils had a low bulk density (127–513 kg m−3), near-neutral pH (7.1–7.4), and a very low nitrite content (data not shown). The two talus soils also had a high denitrification gene content (nirS in cluster I, nirK in clusters I and II [nir: nitrite reductase], and nosZ in clades I and II [nos: N2O reductase]; Fig. 5), suggesting that active denitrification was on-going in the talus soils. In contrast, the soils at the two control areas were mineral soils with lower water, total C, total N, and inorganic N content, higher bulk density (Table 1); and less denitrification gene content (Fig. 5) compared with the talus soils.
Copy numbers of denitrifier genes encoding nitrite reductase (nirS cluster I, nirK cluster I, and nirK cluster II) and nitrous oxide reductase (nosZ clade I and nosZ clade II) in soils. Bars denote standard deviation (n = 3). Different letters denote a significant difference (P < 0.05) under a log-normal distribution assumption. BL, study site in Blomstrandhalvøya; ST, study site in Stuphallet; U, talus (upper part); M, talus (middle part); L, talus (lower part); C, control area not directly affected by seabirds.
However, the two talus soils were not identical. The carbon-to-nitrogen (CN) ratios of the soils on the middle and lower parts of the talus at ST (15.4–16.2) were significantly higher than those of the soils collected at BL (9.9–11.4; P < 0.05; Table 1). The nitrate content of the soils on the upper and middle part of the talus at BL (2270–2430 mg N kg−1 dry soil), which accounted for 8–9% of the total N content, were significantly higher than those at ST (21.2–165 mg N kg−1 dry soil; P < 0.05; Table 1). The δ15N values (stable N isotope ratio [15N/14N] expressed in δ notation) of the soils on the middle and lower parts of the talus at BL (19.4–21.3‰) were significantly higher than those at ST (5.9–10.7‰; P < 0.05; Table 1).
At the two taluses, the effect of distance was not significant for CN ratio but was significant for ammonium content (P < 0.001), nitrate content (P < 0.001), and δ15N (P = 0.016) (Fig. 6). The effect of site was significant for CN ratio (P = 0.028) and nitrate content (P = 0.003) and not significant for ammonium content (P = 0.084) and δ15N (P = 0.057) (Fig. 6). The interaction between distance and site was significant for CN ratio (P = 0.026), which was attributed to the sharper increase in CN ratio with increasing distance from the cliff bottom at ST than at BL, and nitrate content (P = 0.005), which was attributed to the sharper decrease in nitrate content with increasing distance from the cliff bottom at BL than at ST, not significant for δ15N (P = 0.052) and ammonium content (Fig. 6).
(a) Carbon-to-nitrogen (CN) ratio, (b) ammonium content, (c) nitrate content, and (d) δ15N of soils (0–5 cm depth). Bars denote standard deviation (n = 3). A normal distribution assumption was accepted for CN ratio and ammonium content, and a log-normal distribution assumption was accepted for nitrate content and δ15N. BL, study site in Blomstrandhalvøya; ST, study site in Stuphallet; n.s., not significant. The grey bar shows the δ15N range for lipid or liver tissue of three seabird species (black-legged kittiwake, northern fulmar, and Atlantic puffin).
Discussion
High denitrification potentials of talus soils
The talus soils had high dentification potentials at both sites (Fig. 3); this result supported our first hypothesis. However, the effect of distance from the cliff bottom on denitrification potential was not significant (Fig. 3); this part of results was contrary to third hypothesis. The denitrification potentials of the talus soils at BL and ST (2.62–4.88 [10 °C] and 5.59–11.1 [20 °C] mg N kg−1 dry soil h−1) were very high compared with the following literature values: 0.1 (meadow snowbed) to 1.6 (mesic heath) mg N kg−1 dry soil h−1 at room temperature for tundra soils collected in northern Sweden9, 0.78 (cryoturbated soil) and 0.50 (unturbated soil) mg N kg−1 dry soil h−1 at 20 °C collected in a Russian discontinuous permafrost zone38, 0.51 (surface fen soil) mg N kg−1 dry soil h−1 at 20 °C collected in northeast Finland39, and 0.0056–1.74 mg N kg−1 h−1 at 25 °C for 26 topsoil (0–20 cm) samples collected at farmlands in Shanxi province and Inner Mongolia, China40. Thus, the high denitrification potentials (Fig. 3) and abundance of denitrification genes (Fig. 5) in the present talus soils indicate that the bird-affected taluses at the study sites are denitrification hotspots within the High Arctic. We did not observe a significant change in denitrification potential with increasing distance from the cliff bottom (Fig. 3); we attributed this finding to the high water and total C contents (Table 1), which are properties advantageous to denitrification, of the soils throughout the length of the taluses. Note that the denitrification potentials were determined in the presence of abundant nitrate and that real-world denitrification activities depend on local nitrate availability.
The positive temperature responses of the denitrification potentials of the talus soils (Fig. 3) denote that warming might accelerate in-situ denitrification of the talus soils. The Q10 values of the talus soils, 1.9–2.3 at BL and 1.6–1.8 at ST, are comparable with that reported from Swedish lake sediment (1.69)41. Of the two temperatures examined, the lower temperature (10 °C) was close to the mean maximum daily air temperature in Ny-Ålesund in July (warmest month in the year) for the decade from 2008 to 2017 (8.1 °C)42; however, the higher temperature (20 °C) was higher than the maximum air temperature recorded in the same period (14.8 °C)42. Thus, the positive temperature response indicates that future climate warming might promote denitrification in bird-affected talus soils in the High Arctic.
Marked N2O emissions only at BL
The talus at BL emitted N2O and the talus at ST was a poor N2O emitter (Fig. 4). These results partly supported our second hypothesis. The effect of distance from the cliff bottom on N2O emission was not significant contrary to third hypothesis (Fig. 4). The in-situ N2O emissions recorded at the talus at BL (0.85–16.6 µg N m−2 h−1; mean ± SD, 5.3 ± 5.3 µg N m−2 h−1; n = 9 plots) challenge the conventional understanding that Arctic soils are poor N2O emitters16,21,22,23. The N2O emissions from the talus at BL were smaller than the highest reported N2O emitters in the Arctic: 50–820 µg N m−2 h−1 from peat circles in subarctic eastern European tundra throughout the growing season from June to October19 and 20.9 ± 3.9 µg N m−2 h−1 from a bird-affected, flat, semi-wet tundra with high bird activity (a bird sanctuary inhabited mainly by Arctic terns [Sterna paradisaea] and barnacle geese [Branta leucopsis]) in Ny-Ålesund from late July to early August30. However, they are comparable with those reported for medium- and low-bird activity areas (4.8 ± 2.5 and 4.5 ± 2.4 µg N m−2 h−1, respectively) in the flat tundra in Ny-Ålesund30.
In tundra soil, the main pathway of N2O production is denitrification under intermediate or high moist soil conditions18,19,38,39, although sufficient nitrate must also be present18,19. Furthermore, for N2O to be emitted, there must be surplus of N2O produced. Soil moisture has a major effect on the availability of oxygen to soil microbes, and there is an optimum wetness to facilitate surplus N2O production11, which is intermediately moist conditions to promote both aerobic nitrification and anaerobic denitrification19,25. Excessively moist conditions promote complete denitrification where N2O is reduced to N2 and therefore there is no surplus N2O to emit11. There is a contradictory finding that nitrification is the main pathway of N2O production in the Canadian Arctic tundra43, which is a result of competition between denitrifiers and fungi for nitrate that increases the relative contribution of nitrification to N2O emissions44. Since the talus soils at BL contained high amounts of nitrate (Table 1), we conclude that denitrification is the main pathway of N2O production in the talus soils owing to the abundance of substrate; however, nitrification supplying the abundant nitrate might also contribute to N2O production. Further research is needed to elucidate contributions of nitrification and denitrification to N2O emitted from bird-affected taluses to the atmosphere.
The marked in-situ N2O emissions observed at the talus at BL (Fig. 4) indicate the presence of a mixture of aerobic and anaerobic conditions. This interpretation is supported by the active denitrification, active nitrification, and rapid N turnover in the talus soils at BL. Active denitrification in the talus soils at BL was confirmed by the high denitrification potentials (Fig. 3) and the abundance of denitrification genes (Fig. 5) in the soils; however, the talus soils at ST were also characterised by active denitrification. Denitrification potential does not predict in-situ N2O emission, as shown by the lack of correlation between the two variables (Fig. 7a). Active nitrification in the talus soils at BL was confirmed by the high nitrate content in the soils (Table 1), particularly at the upper and middle parts of the talus (2270–2430 mg N kg−1 dry soil). These values are higher than reported values (ca. 480 mg N kg−1) for soils in southwest Svalbard beneath a colony of ca. 10,000 breeding pairs of Brünnich’s guillemots (Uria lomvia) and black-legged kittiwakes36, and is comparable with values (ca. 1452 mg N kg−1) for soils collected from a breeding colony of black-tailed gulls (Larus crassirostris) in Japan with high N2O emissions45 and from a year-round colony of great cormorants (Phalacrocorax carbo) in Japan (ca. 3530 mg N kg−1) with extremely high N2O emissions33. The correlation between in-situ N2O emission and nitrate content was significant and positive (P = 0.013; Fig. 7c). Rapid N turnover in the talus soils at BL was confirmed by the low CN ratios and high δ15N values for the soils (Table 1). High nutrient input accelerates organic matter turnover in tundra soil, which decreases the soil CN ratio6. The CN ratios of the talus soils at BL (9.9–11.4) were lower than those at ST (12.3–16.2), which indicates that there is faster organic matter turnover at BL than at ST (Table 1). N2O emissions are negatively correlated with soil CN ratio in European forest soils11. Similarly, the present data showed a significant negative correlation between in-situ N2O emission and CN ratio (P = 0.009; Fig. 7b). In German and Finnish histosols, annual N2O emissions are negligible at a CN ratio higher than 25, but below this threshold N2O emissions increase exponentially with decreasing CN ratio18. Such a negative exponential relationship was also found in the present data (R2 = 0.63; Fig. 7b), where N2O emissions were below the detection limit (0.6 µg N m−2 h−1; Fig. 4) at CN ratios greater than 12. The δ15N value of bulk soil increases with inorganic N loss from the soil via nitrate leaching and atmospheric emissions of ammonia, N2, and N2O46. The δ15N values of soils from the middle and lower parts of the talus at BL were higher than those of the seabird species that inhabited the cliffs (Fig. 6d). Such enrichment of 15N indicates active inorganic N loss, that is, there is rapid inorganic N turnover in the talus soils at BL. The present data showed a significant positive correlation between in-situ N2O emission and the δ15N value of bulk soil (P = 0.024; Fig. 7d).
Relationship between in-situ N2O emission flux and (a) denitrification potential, (b) carbon-to-nitrogen (CN) ratio, (c) nitrate content, and (d) δ15N of bulk soil. Bars denote standard deviation (n = 3). *P < 0.05; **P < 0.01. BL, study site in Blomstrandhalvøya; ST, study site in Stuphallet; T-U, talus (upper part); T-M, talus (middle part); T-L, talus (lower part); C, control area not directly affected by seabirds.
We conducted our sampling in July because we wanted to examine the conditions at the time during the year when N2O emissions are greatest, and the maximum N2O emissions from peat circles19 and bird-affected flat tundra30 were recorded in July. The air temperature during our field surveys were 8–9 °C at BL and 7–8 °C at ST, and these temperatures were comparable with the mean daily maximum air temperature in July in Ny-Ålesund (8.1 °C)42. The present data were obtained close to the average daily maximum air temperature. However, our snapshot measurements in the present study do not support the conclusion that the talus at BL always emits N2O and the talus at ST never emits N2O.
Effects of seabirds on soil N turnover
The differences in soil properties between the two taluses, particularly with respect to the lower CN ratio, much higher nitrate content, and higher δ15N at BL (Table 1), are attributable to the approximately 30-fold larger seabird density at BL compared with that at ST. It is natural that there is a quantitative difference in organic matter input due to the difference in bird density.
Piscivorous and planktivorous seabirds promote the growth of different plant species near their colonies and the total N content and δ15N values of plants are higher near piscivorous colonies than near planktivorous colonies47. This implies that piscivorous seabirds accelerate N turnover of the local soil more than planktivorous seabirds do. The three seabird species inhabiting our study sites are all piscivorous; however, these birds also feed on zooplankton, such as amphipods and krill, depending on prey availability48,49. The δ15N of liver tissue of black-legged kittiwake, northern fulmar, and Atlantic puffin in Iceland has been reported as 13.2–14.2‰, 13.5–14.0‰, and 12.2–13.0‰, respectively48, and that of lipids of black-legged kittiwake and northern fulmar in northwest Svalbard as 12.1‰ and 13.6‰, respectively49. In contrast, the faeces of little auk (Alle alle), a planktivorous seabird, in southwest Svalbard has a lower δ15N of 8.0‰34. Seabirds might alter their diet to adapt to environmental changes. In northwest Svalbard, the diet of black-legged kittiwakes is shifting from Arctic to Atlantic species due to recent climate warming50. Population and colonisation of High Arctic seabirds might be also changing due to climate warming and environmental changes. Seabirds have an important role as a nutrient carrier from marine to terrestrial ecosystems; therefore, further research is needed to elucidate how changes in seabird activities impact seabird-affected talus soils as denitrification hotspots and possible N2O emitters in the High Arctic.
Methods
Study sites
We selected two study sites near Ny-Ålesund, Svalbard, in the European High Arctic (Fig. 2). Detailed information of the two study sites is shown in Supplementary Information.
Field survey
Three lines on the upper, middle, and lower parts of each talus were set in relation to the distance from the cliff bottom. A control area unaffected by the seabirds was also set at each site for comparison with the characteristics of the talus soils. At each location (three lines on each talus and control areas), three plots were set 10 to 15 m apart. Gas and soil samples were collected from three points within each plot (Supplementary Information).
The field survey was conducted on 21 July 2015 at the talus and control area at BL, on 12 July 2017 at the upper and middle parts of the talus at ST, and on 15 July 2017 at the lower part of the talus and control area at ST. Gas and soil samples were collected from the same points to enable direct comparison between in-situ N2O emissions and soil-related variables including denitrification potential. We used a closed-chamber technique51, downsized and simplified, to first collect gas samples and then to collect soil samples. The chamber was made by connecting two stainless-steel soil cores (volume, 100 mL each; DIK-1801, Daiki, Saitama, Japan) using polyvinylchloride adhesive tape, and the upper and lower cores were used for gas and soil sampling, respectively. This simple method was effective for conducting multipoint sampling on the steep, uneven surfaces of the taluses, and the small chambers reduced environmental disturbance due to the destructive nature of the soil sampling. The sampling procedure is explained in detail in the Supplementary Information.
Measurement of denitrification potential
The denitrification potential of the soil samples was determined by using an acetylene inhibition technique9,38,39,40,41,52,53,54. Briefly, 8 g of fresh soil and 12 mL of substrate solution were added to a glass vial (volume, 140 mL). The substrate solution contained 1 mM nitrate as the denitrification substrate and 0.1 mM chloramphenicol as an inhibitor of cell growth to eliminate apparent increases in the N2O production rate due to cell growth52. The vial was purged with N2 to produce a strong anaerobic condition and then 10% of the vial air volume was substituted with purified acetylene to inhibit the final stage of denitrification from N2O to N2. No additional organic matter as an electron donor for denitrification was added because the talus soils were rich in organic matter. The vials were incubated at 10 or 20 °C for 6 h, and gas sampling was conducted at 2, 4, and 6 h from the start of incubation. The N2O mixing ratio in the gas samples was determined by using a gas chromatograph equipped with an electron capture detector (GC-14A, Shimadzu, Kyoto, Japan). N2O production rates between 2–4 h and 4–6 h were averaged to obtain the denitrification potential for the sample.
Our use of the acetylene inhibition technique allows comparisons to be made between our results and those of other studies that use the same technique9,38,39,40. In this technique, N2O is originated only by denitrification because the anaerobic conditions and acetylene inhibit nitrification53. The acetylene inhibition technique is a common means of determining denitrification potential41,52,55; however, possible underestimation has been reported due to incomplete inhibition of N2O reduction40,54 arising from incomplete diffusion of the acetylene into soil microsites, microbial consumption of the acetylene, and differences in acetylene sensitivity among the denitrifiers in the soil40. We addressed the issue of incomplete diffusion of acetylene into soil microsites by using vigorous shaking during incubation, and we consider microbial consumption of acetylene to have been negligible during the short-term (6 h) anaerobic incubation40. Although the current knowledge regarding the relationship between denitrifying flora and denitrification potential of Arctic soil is incomplete, experiments showed that the bias of denitrification potentials determined by the acetylene inhibition technique is lower in high-fertility soils40. The talus soils in the present study were rich in total C and total N (Table 1) and were therefore of high fertility, suggesting that the determined denitrification potentials had low biases; we therefore concluded that the present data sufficiently approximated the true denitrification potential.
Determination of in-situ N2O emissions
The N2O mixing ratio in the collected gas samples was determined by gas chromatography, as described in the previous section. In-situ N2O emissions were calculated by using the rate of change in N2O mixing ratio inside the chamber, atmospheric pressure, air temperature, chamber volume, and soil surface area51. The detection limit of N2O flux was 0.0 ± 0.6 μg m−2 h−1, which corresponded to the 10-fold standard deviation (two-sided) of the mixing ratio for a standard N2O gas (300 ppb).
Soil analyses
Fresh soil samples were used to determine soil water content (w/w), bulk density, and soil pH (H2O). Soil water content was determined using a dry oven. Bulk density was calculated from the initial soil weight and the soil water content. Soil pH was measured by using a pH meter (B-712, Horiba, Tokyo, Japan). Air-dried soil samples were used to determine total C, total N, ammonium, nitrite, and nitrate content and δ15N. Total C and total N content were measured by using an NC analyser (Sumigraph NC-22F, Sumika Chemical Analysis Service, Tokyo, Japan). Inorganic N content was determined by using a flow injection analyser (AQLA-1000, Aqualab, Tokyo, Japan) for which 5 g of fresh soil was extracted with 40 mL of 10% KCl solution. δ15N was determined with an elemental analyser/isotope ratio mass spectrometer (Flash EA 1112 coupled to a Delta V Advantage via ConFlo III interface, Thermo Fisher Scientific, Bremen, Germany). Soil samples containing ca. 45 µg N were placed in tin capsules, loaded into a Costech Zero-Blank Autosampler (Costech, Valencia, CA, USA), and then introduced to the reactor furnace of the elemental analyser. δ15N was determined as (15N/14N)sample/(15N/14N)standard−1, where ‘standard’ refers to international standards of known 15N/14N ratios calibrated against N2 in air. The analytical reproducibility of this method was 0.2‰.
To examine the abundance of denitrification genes in soil samples, DNA was extracted from 0.4 g of soil sample by using a FastDNA SPIN Kit for Soil (MP Biomedical, Santa Ana, CA, USA) in accordance with the methods of Hayashi et al.56. Then, the polymerase chain reaction (PCR) was used to determine the existence of gene sequences encoding target denitrification enzymes in the DNA extracted from the soil samples by using primers targeting the nitrite reductase-encoding genes nirK (clusters I, II, III, and IV) and nirS (clusters I, II, and III) and the nitrous oxide reductase-encoding gene nosZ (clades I and II)57 in accordance with the methods of Wang et al.58. The detected genes were subjected to quantitative PCR analysis using SYBR Premix Ex Taq (Takara Bio, Kyoto, Japan) and a StepOnePlus Real-Time PCR system (Thermo Fisher Scientific) in accordance with the methods of Wang et al.58. A 1-μL sample of 10-fold diluted soil DNA was used as the template in a reaction volume of 20 μL.
Statistical analysis
Tests of statistical significance for the denitrification potential of the talus soils were performed by using a mixed model (‘proc mixed’ ver. 9.4) in the SAS software (SAS Institute Inc., Cary, NC, USA) to assess the effects of distance, site, incubation temperature, and their interactions, for which α = 0.05 was used as the limit of significance. Distance was treated as a covariate. Location and sampling plot, nested within each location (Supplementary Information), were treated as random effects. Variance components were estimated by using the restricted maximum likelihood method. Effects of distance and site, and their interaction, on in-situ N2O emission from talus were tested using the SAS mixed model but with the additional random effect of sampling point (replication) nested within each sampling plot (Supplementary Information), for which Yeo–Johnson transformation of the data using the ‘powerTransform’ function in the ‘car’ package of R59 was performed because some negative values showed a highly skewed distribution and violated the homoscedasticity assumption. The effects of distance and site, and their interactions, on CN ratio, ammonium and nitrate content, and δ15N of talus soil were similarly analysed using the SAS mixed model. For all soil properties and the denitrification gene copy number of talus and control soils, one-way ANOVA was conducted to test the effect of location by using ‘proc glm’ in SAS. Tukey’s multiple comparison was then conducted (α = 0.05) to identify significant differences among locations; logarithmic transformation was applied to the data of bulk density, nitrate content, and δ15N because the data violated homoscedasticity assumption. Pearson’s correlation coefficients were evaluated for the relationship between in-situ N2O emission and the variables denitrification potential, CN ratio, nitrate content, and δ15N. For CN ratio, a decaying exponential curve was also fitted.
Data Availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Callaghan, T. V. et al. Arctic tundra and polar desert ecosystems [Symon, C., Arris, L. & Heal, B. (eds)] Arctic Climate Impact Assessment. 243–352 (Cambridge University Press, Cambridge, 2005).
Bekryaev, R. V., Polyakov, I. V. & Alexeev, V. A. Role of polar amplification in long-term surface air temperature variations and modern arctic warming. J. Climate 23, 3888–3906 (2010).
Hill, G. B. & Henry, G. H. R. Responses of High Arctic wet sedge tundra to climate warming since 1980. Global Change Biol. 17, 276–287 (2011).
Descamps, S. et al. Climate change impacts on wildlife in a High Arctic archipelago – Svalbard, Norway. Global Change Biol. 23, 490–502 (2017).
Shaver, G. R. et al. Global change and the carbon balance of arctic ecosystems. Bioscience 42, 433–441 (1992).
Mack, M. C., Schuur, E. A., Bret-Harte, M. S., Shaver, G. R. & Chapin, F. S. III Ecosystem carbon storage in arctic tundra reduced by long-term nutrient fertilization. Nature 431, 440–443 (2004).
LeBauer, D. S. & Treseder, K. K. Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89, 371–379 (2008).
Oulehle, F., Rowe, E., Myška, O., Chuman, T. & Evans, C. Plant functional type affects nitrogen use efficiency in high-Arctic tundra. Soil Biol. Biochem. 94, 19–28 (2016).
Björk, R. G. et al. Linkages between N turnover and plant community structure in a tundra landscape. Plant Soil 294, 247–261 (2007).
Hayatsu, M., Tago, K. & Saito, M. Various players in the nitrogen cycle: diversity and functions of the microorganisms involved in nitrification and denitrification. Soil Sci. Plant Nutr. 54, 33–45 (2008).
Butterbach-Bahl, K., Baggs, E. M., Dannenmann, M., Kiese, R. & Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos. T. Roy. Soc. B 368, 20130122 (2013).
Stein, L. Y. & Klotz, M. G. The nitrogen cycle. Curr. Biol. 26, R94–R98 (2016).
Kuypers, M. M. M., Marchant, H. K. & Kartal, B. The microbial nitrogen-cycling network. Nat. Rev. Microbiol. 16, 263–276 (2018).
Forsius, M. et al. Assessing the impacts of long-range sulfur and nitrogen deposition on arctic and sub-arctic ecosystems. Ambio 39, 136–147 (2010).
Hodson, A., Roberts, T. J., Engvall, A. C., Holmen, K. & Mumford, P. Glacier ecosystem response to episodic nitrogen enrichment in Svalbard, European High Arctic. Biogeochemistry 98, 171–184 (2010).
Ciais, P. et al. Carbon and Other Biogeochemical Cycles. [Stocker, T. F. et al. (eds)] Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the FifthAssessment Report of the Intergovernmental Panel on Climate Change. 465–570 (Cambridge University Press, Cambridge, 2013).
Ravishankara, A. R., Daniel, J. S. & Portmann, R. W. Nitrous oxide (N2O): The dominant ozone-depleting substance emitted in the 21st century. Science 326, 123–125 (2009).
Klemedtsson, L., von Arnold, K., Weslien, P. & Gundersen, P. Soil CN ratio as a scalar parameter to predict nitrous oxide emissions. Global Change Biol. 11, 1142–1147 (2005).
Repo, M. E. et al. Large N2O emissions from cryoturbated peat soil in tundra. Nat. Geosci. 2, 189–192 (2009).
Hayashi, K., Tokida, T., Kajiura, M., Yanai, Y. & Yano, M. Cropland soil–plant systems control production and consumption of methane and nitrous oxide and their emissions to the atmosphere. Soil Sci. Plant Nutr. 61, 2–33 (2015).
Ludwig, B., Teepe, R., de Gerenyu, V. L. & Flessa, H. CO2 and N2O emissions from gleyic soils in the Russian tundra and a German forest during freeze-thaw periods – a microcosm study. Soil Biol. Biochem. 38, 3516–3519 (2006).
de Gerenyu, V. L., Kurganova, I., Teepe, R., Loftfild, N. & Flessa, H. Effect of contrasting changes in hydrothermic conditions on the N2O emission from forest and tundra soils. Eurasian Soil Sci. 40, 795–799 (2007).
Bouwman, A. F. et al. Global trends and uncertainties in terrestrial denitrification and N2O emissions. Philos. Trans. R. Soc. Ser. B 368, 20130112 (2013).
Francez, A.-J., Pinay, G., Josselin, N. & Williams, B. L. Denitrification triggered by nitrogen addition in Sphagnum magellanicum peat. Biogeochemistry 106, 435–441 (2011).
Abbott, B. W. & Jones, J. B. Permafrost collapse alters soil carbon stocks, respiration, CH4, and N2O in upland tundra. Global Change Biol. 21, 4570–457 (2015).
Riddick, S. N. et al. The global distribution of ammonia emissions from seabird colonies. Atmos. Environ. 55, 319–327 (2012).
Riddick, S. N. et al. Measurement of ammonia emissions from temperate and sub-polar seabird colonies. Atmos. Environ. 134, 40–50 (2016).
Riddick, S. N. et al. High temporal resolution modelling of environmentally-dependent seabird ammonia emissions: Description and testing the GUANO model. Atmos. Environ. 161, 48–60 (2017).
Croft, B. et al. Contribution of Arctic seabird-colony ammonia to atmospheric particles and cloud-albedo radiative effect. Nat. Commun. 7, 13444 (2016).
Zhu, R. B., Chen, Q. Q., Ding, W. & Xu, H. Impact of seabird activity on nitrous oxide and methane fluxes from High Arctic tundra in Svalbard, Norway. J. Geophys. Res.: Biogeosciences 117, G04015 (2012).
Sun, L. G., Zhu, R. B., Xie, Z. Q. & Xing, G. X. Emissions of nitrous oxide and methane from Antarctic tundra: role of penguin dropping deposition. Atmos. Environ. 36, 4977–4982 (2002).
Zhu, R. B. et al. Nitrous oxide emissions from sea animal colonies in the maritime Antarctic. Geophys. Res. Lett. 35, L09807 (2008).
Mizota, C., Noborio, K. & Mori, Y. The great cormorant (Phalacrocorax carbo) colony as a “hot spot” of nitrous oxide (N2O) emission in central Japan. Atmos. Environ. 57, 29–34 (2012).
Skrzypek, G. et al. Diversification of nitrogen sources in various tundra vegetation types in the High Arctic. Plos One 10, e0136536 (2015).
Zwolicki., A., Zmudczynska-Skarbek, K., Richard, P. & Stempniewicz, L. Importance of marine-derived nutrients supplied by planktivorous seabirds to high Arctic tundra plant communities. Plos One 11, e0154950 (2016).
Zwolicki, A., Zmudczynska-Skarbek, K. M., Iliszko, L. & Stempniewicz, L. Guano deposition and nutrient enrichment in the vicinity of planktivorous and piscivorous seabird colonies in Spitsbergen. Polar Biol. 36, 363–372 (2013).
Gornall, J. L., Jonsdottir, I. S., Woodin, S. J. & van der Wal, R. Arctic mosses govern below-ground environment and ecosystem processes. Oecologia 153, 931–941 (2007).
Palmer, K., Biasi, C. & Horn, M. A. Contrasting denitrifier communities relate to contrasting N2O emission patterns from acidic peat soils in arctic tundra. ISME J. 6, 1058 (2012).
Palmer, K. & Horn, M. A. Denitrification activity of a remarkably diverse fen denitrifier community in Finnish Lapland is N-oxide limited. Plos One 10, e0123123 (2015).
Qin, S. P. et al. Relationship between soil properties and the bias of N2O reduction by acetylene inhibition technique for analyzing soil denitrification potential. Soil Biol. Biochem. 66, 182–187 (2013).
Myrstener, M., Jonsson, A. & Bergström, A.-K. The effects of temperature and resource availability on denitrification and relative N2O production in boreal lake sediments. J. Environ. Sci. 47, 82–90 (2016).
Norwegian Meteorological Institute. eKlima (weather and climate data), https://www.met.no/en (2018).
Ma, W. K. et al. Assessing the potential of ammonia oxidizing bacteria to produce nitrous oxide in soils of a high arctic lowland ecosystem on Devon Island, Canada. Soil Biol. Biochem. 39, 2001–2013 (2007).
Siciliano, S. D., Ma, W. K., Ferguson, S. & Farrell, R. E. Nitrifier dominance of Arctic soil nitrous oxide emissions arises due to fungal competition with denitrifiers for nitrate. Soil Biol. Biochem. 41, 1104–1110 (2009).
Mizota, C. Temporal variations in the concentration and isotopic signature of ammonium- and nitrate-nitrogen in soils under a breeding colony of Black-tailed gulls (Larus crassirostris) on Kabushima Island, northeastern Japan. Appl. Geochem. 24, 328–332 (2009).
Hobbie, J. E. et al. Mycorrhizal fungi supply nitrogen to host plants in Arctic tundra and boreal forests: 15N is the key signal. Can. J. Microbiol. 55, 84–94 (2009).
Zwolicki, A., Zmudczynska-Skarbek, K., Matula, J., Wojtun, B. & Stempniewicz, L. Differential responses of Arctic vegetation to nutrient enrichment by plankton- and fish-eating colonial seabirds in Spitsbergen. Front. Plant Sci. 7, 1959 (2016).
Thompson, D. R. et al. Trophic relationships among six species of Icelandic seabirds as determined through stable isotope analysis. Condor 101, 898–903 (1999).
Dahl, T. M. et al. Lipids and stable isotopes in common eider, black-legged kittiwake and northern fulmar: a trophic study from an Arctic fjord. Mar. Ecol. Prog. Ser. 256, 257–269 (2003).
Vihtakari, M. et al. Black-legged kittiwakes as messengers of Atlantification in the Arctic. Sci. Rep. 8, 1178 (2018).
Hayashi, K. et al. Potential role of Svalbard reindeer winter droppings in atmosphere-land exchanges of methane and nitrous oxide during summer. Polar Sci. 8, 196–206 (2014).
Tiedje, J. M., Simkins, S. & Groffman, P. M. Perspectives on measurement of denitrification in the field including recommended protocols for acetylene based methods. Plant Soil 115, 261–284 (1989).
Groffman, P. M. et al. Methods for measuring denitrification: Diverse approaches to a difficult problem. Ecol. Appl. 16, 2091–2122 (2006).
Qin, S. P., Hu, C. S. & Oenema, O. Quantifying the underestimation of soil denitrification potential as determined by the acetylene inhibition method. Soil Biol. Biochem. 47, 14–17 (2012).
Bernot, M. J. et al. Comparing denitrification estimates for a Texas estuary by using acetylene inhibition and membrane inlet mass spectrometry. Appl. Environ. Microbiol. 69, 5950–5956 (2003).
Hayashi, K. et al. Characteristics of ammonia oxidation potentials and ammonia oxidizers in mineral soil under Salix polaris–moss vegetation in Ny-Ålesund, Svalbard. Polar Biol. 39, 725–741 (2016).
Wei, W. et al. Higher diversity and abundance of denitrifying microorganisms in environments than considered previously. ISME J. 9, 1954–1965 (2015).
Wang, Y., Uchida, Y., Shimomura, Y., Akiyama, H. & Hayatsu, M. Investigation of denitrifier communities in a Fluvisol and their responses to a short-term waterlogging. Sci. Rep. 7, 803 (2017).
Fox. J. & Weisberg. S. An R companion to applied regression. Sage, Thousand Oaks, CA, USA (2011).
Acknowledgements
We thank Dr. Midori Yano for her support in obtaining stable isotope measurements at the Advanced Analysis Center, National Agricultural and Food Research Organization, Japan. SAS was run on the supercomputer of the Agriculture, Forestry and Fisheries Research Information Technology Center, Ministry of Agriculture, Forestry and Fisheries, Japan. This study was supported by Grants-in-Aid for Scientific Research (No. 26304018) provided by the Japan Society for the Promotion of Science. This study was also partly supported by the National Institute of Polar Research, Japan, through Project Research No. KP309.
Author information
Authors and Affiliations
Contributions
K.H. designed the study, conducted the field survey and analyses, and wrote the manuscript. Y.T., K.O., M.A. and H.F. contributed to the field survey. M.J.J.E.L. contributed to the field survey and provided bird-related information. T.T. contributed to the statistical analyses. M.U. coordinated the field survey. M.H. contributed to the microbial analyses. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hayashi, K., Tanabe, Y., Ono, K. et al. Seabird-affected taluses are denitrification hotspots and potential N2O emitters in the High Arctic. Sci Rep 8, 17261 (2018). https://doi.org/10.1038/s41598-018-35669-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35669-w
Keywords
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.