Abstract
Evidence suggests that differences in motor function are an early feature of autism spectrum disorder (ASD). One aspect of motor ability that develops during childhood is postural control, reflected in the ability to maintain a steady head and body position without excessive sway. Observational studies have documented differences in postural control in older children with ASD. The present study used computer vision analysis to assess midline head postural control, as reflected in the rate of spontaneous head movements during states of active attention, in 104 toddlers between 16–31 months of age (Mean = 22 months), 22 of whom were diagnosed with ASD. Time-series data revealed robust group differences in the rate of head movements while the toddlers watched movies depicting social and nonsocial stimuli. Toddlers with ASD exhibited a significantly higher rate of head movement as compared to non-ASD toddlers, suggesting difficulties in maintaining midline position of the head while engaging attentional systems. The use of digital phenotyping approaches, such as computer vision analysis, to quantify variation in early motor behaviors will allow for more precise, objective, and quantitative characterization of early motor signatures and potentially provide new automated methods for early autism risk identification.
Similar content being viewed by others
Introduction
Although the core symptoms of autism spectrum disorder (ASD) are defined by atypical patterns of social interaction and the presence of stereotyped and repetitive behaviors and interests, evidence suggests that differences in motor function are also an important early feature of autism. Motor delays could contribute to early hallmark autism symptoms, including difficulties in orienting to name involving the eyes and head turns, coordinating head and limb movements involved in gaze following and other joint attention behavior, such as pointing. Teitelbaum et al.1 found that atypical movements (e.g. shape of mouth, patterns of lying, righting, sitting) were present by 4–6 months of age in infants later diagnosed with ASD. Another study of videotapes taken of infants 12–21 weeks of age detected lower levels of positional symmetry among infants later diagnosed with ASD2 suggesting atypical development of cerebellar pathways that control balance and symmetry. Six-month-old infants who later were diagnosed with ASD tend to exhibit head lag when pulled to sit, reflecting early differences in motor development3. A study of home videos taken between birth and six months of age found that some infants who were later diagnosed with ASD showed postural stiffness, slumped posture, and/or head lag4. Other motor symptoms observed in infants later diagnosed with ASD include fluctuating muscle tone5 and oral-motor abnormalities, such as insufficient opening of the mouth in anticipation of the approaching spoon during feeding6. Longitudinal research with very low birth weight infants revealed that infants who are later diagnosed with ASD had poorer ability in maintaining midline position of the head at 9–20 weeks of age7. The authors used visual inspection to classify head position in each video frame to yield a measure of midline head position and number of changes in position.
The development of postural control is an index of neuromuscular reactions to the motion of body mass in order to retain stability. Previous studies have documented the developmental progression of the ability to maintain an upright posture that is accompanied by decreases in postural sway8. Several studies with older children with ASD have documented deficiencies in postural control, reflected in the presence of postural sway, which is accentuated when children with ASD are viewing arousing stimuli, including complex multi-sensory and social stimuli9,10,11. Less is known about the presence of postural sway in young children with ASD.
Studies of motor and other behaviors in young children have typically relied on subjective and labor-intensive human coding to rate and measure behavior. The recent use of digital phenotyping approaches, such as computer vision analysis (CVA) of videotaped recordings of behavior, has allowed for automated, precise and quantitative measurement of subtle, dynamic differences in motor behavior. We reported previously on a result using CVA to more precisely measure toddlers’ orienting response to a name call, noting that, compared to toddlers without ASD, toddlers with ASD oriented less frequently; when they did orient, their head turn was a full second slower, on average12. Such differences in motor speed would likely not be detected with the naked eye during a typical clinical evaluation. Anzulewicz et al.13 used smart tablet computers with touch-sensitive screens and embedded inertial movement sensors to record movement kinematics and gesture forces in 3–6-year-old children with and without ASD. Children with ASD used greater force and faster and larger gesture kinematics. Machine learning analysis of the children’s motor patterns classified the children with ASD with a high level of accuracy. In another study using automated methods, differences in head movement dynamics were found between 2.5–6.5-year-old children with and without ASD while they watched movies of social and nonsocial stimuli. Children with ASD showed more frequent head turning, especially while watching social stimuli14. The authors suggested that the children with ASD might be using head movement to modulate their arousal while watching social stimuli. Wu et al.15 used electromagnetic sensors to analyze continuous movements at millisecond time scale in older children with ASD versus typical development. They applied a triangular smoothing algorithm to the 3D positional raw movement data that preserved the local speed fluctuations. They found that individuals with ASD exhibited significantly more “sensorimotor noise” when compared to individuals with typical development.
The present study used CVA to characterize head movements that didn’t involve spontaneous or volitional orienting or turning away from the stimuli. Rather, we were interested in subtler midline head movements that are more likely related to postural stability. The study compared the behaviors of toddlers with ASD versus those without ASD while the children watched a series of dynamic movies involving different types of stimuli, including stimuli of both a social and nonsocial nature. While the children watched the movies, their head movements were automatically detected and tracked using landmarks on the participant’s face. The goal of this analysis was to quantify the rate of spontaneous head movements and to determine whether there were differences in this motor feature between young children with and without ASD.
Methods
Participants
Participants were 104 children between 16–31 months of age (Mean = 22 months). Exclusionary criteria included known vision or hearing deficits, lack of exposure to English at home and/or caregivers who did not speak and read English sufficiently for informed consent. Twenty-two of the children had autism spectrum disorder. The non-ASD comparison group was comprised of 96 typically developing children and 8 children with language delay or developmental delay of clinical significance sufficient to qualify for speech or developmental therapy. Participants in the comparison group had a mean age of 21.91 months (SD = 3.78) and those in the ASD group had a mean age of 26.19 months (SD = 4.07). Ethnic/racial composition of the ASD and comparison groups, respectively, was 59% and 45% white, 13% and 14% African American, 6% and 5% Asian, and 22% and 36% multi-racial/other. Percent males was 77% in the ASD group and 59% in the comparison group.
Participants were recruited from primary care pediatric clinics by a research assistant, referral from their physician, and by community advertisement. All caregivers/legal guardians of participants gave written, informed consent, and the study protocol was approved by the Duke University Health System Institutional Review Board. Methods were carried out in accordance with institutional, state, and federal guidelines and regulation.
Diagnostic Assessments
Diagnostic evaluations to confirm ASD were based on the Autism Diagnostic Observation Scale-Toddler (ADOS-T), which were conducted by a licensed psychologist or trained research-reliable examiner overseen by a licensed psychologist16. The mean ADOS-T score was 18.81 (SD = 4.20). The mean IQ based on the Mullen Scales of Early Learning Composite Score for the ASD group was 63.58 (SD = 25.95). Developmental and/or language delay was determined based on the Mullen Scales (>1 SD below the mean in overall learning composite or receptive/expressive language).
Stimuli
A series of stimuli, comprised of brief movies, were shown on a smart tablet while the child sat on a caregiver’s lap. The tablet was placed on a stand approximately 3 feet away from the child to prevent the child from touching the screen. The stimuli consisted of a series of brief developmentally-appropriate movies designed to elicit positive affect and engage the child’s attention. The movies consisted of cascading bubbles, a mechanical bunny, animal puppets interacting with each other, and a split screen showing on one side a woman singing nursery rhymes and on the other side dynamic, noise-making toys. The lengths of the movies were 30 seconds (Bubbles), 60 seconds (Rhyme), and ∼70 seconds (Bunny and Puppets). Each movie was shown once except for Bubbles which was shown at the beginning and end of the series. The entire series of movies lasted 5 minutes. Examples of the stimuli and experimental setup are presented in Fig. 1 and described in two previous publications17. Examples of clips from the movies are provided in the Supplementary Material. During three of the movies, the examiner, standing behind the child, called the child’s name. A failure to orient to name is an early symptom of autism, and results of our analysis of the orienting results have previously been published12. However, all segments when children looked away from the movie, including to orient to name, as well as all 5 second segments post the name-call stimulus, were automatically removed from the present analyses. Specifically, in order to remove any influence on head movement due to the child orienting when his or her name was called, we removed the time window starting at cue for the name call prompt (a subtle icon used to prompt the examiner to call the name) through the point where 75% of the audible name calls actually occurred, plus 150 frames (5 seconds). Since previous studies have shown that orienting tends to occur within a few seconds after a name call, this eliminated segments influenced by the name call.
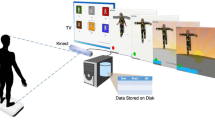
iPad movie task and facial landmark detection: (A) Two examples of facial landmark points detected by CVA and estimated head pose (indicated by the three arrows). The landmarks colored in red are the inner left, inner right, and central nose landmarks that are used for head movement computation. The left example depicts landmarks and head pose of a participant engaged in the movie stimuli; while in the right example, the participant is looking away. Both states are automatically detected. (B) Example frames from movie stimuli. Each row displays a frame from corresponding movie stimuli show in the columns (going from left-to-right): Bubbles (30 seconds, two repetitions), Bunny (66 seconds), Rhymes (60 seconds), and Puppet show (68 seconds).
Parents were asked to attempt to keep the child seated in their lap, but to allow the child to get off their lap if the child became too distressed to stay seated. Researchers stopped the task for 1 child due to crying. Researchers restarted the task for three participants due to noncompliance.
Computer Vision Analysis
The frontal camera in the tablet recorded video of the child’s face throughout the experiment at 1280 × 720 spatial resolution and 30 frames per second. The fully automatic CVA algorithm detects and tracks 49 facial landmarks on the child’s face (see Fig. 1)18 and estimates head pose angles relative to the camera by computing the optimal rotation parameters between the detected landmarks and a 3D canonical face model19. For each video frame the algorithm outputted 2D positional coordinates of the facial landmarks and 3 head pose angles: yaw (left-right), pitch (up-down), and roll (tilting left-right). The yaw head pose angle was used to determine the frames when the child was engaged with the movie stimuli, where frames exhibiting a yaw pose with a magnitude less than 20° were considered as the child being engaged.
Following the work of17, to quantify head movement when the child is engaged (less than 20° yaw), per-frame pixel-wise displacements of 3 central facial landmarks were computed and normalized with respect to the child’s eye width, thus head movement was measured as a (normalized) proportion of the child’s eye width per frame. The pixel-wise displacements of the central facial landmarks are dependent on the child’s distance to the camera in the tablet. Although the tablet was placed approximately 3 feet away from the child at the start of the experiment, the child is free to move throughout the experiment, thus affecting the magnitude of landmark displacements (when the child is near to the camera the pixel displacements are larger than if the child did the same movement but farther away from the camera). Normalizing the displacements with respect to the eye-width diminishes this distance to camera dependency. More formally, the head movement between frame n and n-1 is defined as the average Euclidean displacements of the central nose, left inner eye, and right inner eye landmarks (see Fig. 1) normalized by a ± second windowed-average, centered around frame n, of the Euclidean distances between the inner left and right eye landmarks,
where \(\overline{{d}_{n-1,n}}\) is the average landmark displacement of the three central landmarks between frame n and n-1, and \(\overline{{w}_{n-15,n+15}}\) is the average Euclidean distance between the left and right eye landmarks when the child is engaged between a half-second (15 frames) before and after frame n.
Results evaluating the validity of the CVA methods, which rely on landmark identification and tracking on the face, have been previously published. One study demonstrated high reliability between the automatic methods and the expert human rater of head movements, with agreement between the computer and expert clinical rater occurring 92.5% of the time with interrater reliability based on Cohen’s kappa = 0.7520. A second study compared the automatic classification based on landmarks to human coders for head movement, demonstrated inter-rater reliability based on intraclass correlation coefficient (ICC) = 0.8917. Other papers report high reliability between CVA and human coding for head turning in response to name (ICC = 0.84)12 and positive affective expression (happy; ICC = 0.90 and 0.89 for ASD and non-ASD toddlers)21.
The original dataset consisted of frame-by-frame measurements of head movement, with observations for each 1/30th of a second. Groups interested in direct use of the data can do so via collaboration with the authors due to privacy and consent considerations as well as backend designs, and the data will be stored in a separate partition at Duke University. In order to prepare the data for statistical analysis, we first aggregated the movement measurements by calculating the head movement rate, defined as the moving sum of the cumulative frame-by-frame movement measurements for each 10 frame period (representing 1/3rd of a second). If any individual frames within a 10-frame set were set to missing, such as when the facial landmarks were not visible or during the name-call period, the moving sum was also set to missing. Outliers were addressed by Winsorizing to the 95th percentile prior to aggregation.
All statistical analyses were performed separately for each of the movie stimuli. To visualize the time series, we calculated and plotted the median head movement rate as well as the 1st and 3rd quartiles at each 1/3 second time interval for both ASD and non-ASD children.
Unadjusted and adjusted rate ratios for the association between ASD diagnosis and the rate of head movement in each 1/3 second time interval were estimated using a generalized linear mixed log-gamma regression model. Adjusted estimates controlled for ethnicity/race (white; other), age (in months), and sex (male; female). To account for potential within-subject correlations due to repeated measurement, we included a random intercept for each participant.
Results
The time series data depicting the rate of head movement, defined as the distance traveled per 1/3 second (10 videoframes), for the ASD and non-ASD groups are shown in Fig. 2.
Time series of head movement rate, measured as the distance traveled per 1/3 seconds (10 video frames), by ASD diagnosis. Solid lines are the median values at each time point. Bands represent the first and third quartiles at each time point. Blank sections represent name calls, which were removed from this analysis.
Based on a generalized linear mixed regression model with a log link and gamma distribution (adjusting for ethnicity/race, age, and sex), significant associations between diagnostic group (ASD versus non-ASD) and rate of head movement were found during all movies except for the Bubbles 2, the last movie. For Bubbles 2, the shorter duration might have affected power to detect a result as there was nevertheless a trend toward a group difference in the same direction as all other movies. Results of the analysis are shown in Table 1.
Robust group differences in the rate of head movement were evident during 4 out of 5 of the movies. For example, the rate of head movement among participants with ASD was 2.22 times that of non-ASD participants during the Bunny movie, after adjusting for age, ethnicity/race, and sex (95% Confidence Interval 1.60, 3.07). The rate ratio was higher for all movies that had animated and more complex stimuli (Bunny, Puppets, Rhymes and Toys), as compared to the less complex Bubbles videos.
Although the LD/DD group was too small to conduct independent analyses of that group, as a sensitivity analysis, the 8 patients with LD/DD were from the main regression model and re-estimated the associations, as shown in Table 2. Overall, the results are consistent with those reported in the main analysis; in fact, the associations are slightly stronger when the LD/DD group is removed from the non-ASD group.
Discussion
The present study adds to a large and growing body of literature indicating that differences in early motor development are an important feature of ASD. We found highly significant differences in postural control, reflected in differences in the rate of spontaneous movement of the head between toddlers with ASD versus those without ASD. Using an automated, objective approach, we analyzed data comprised of video-frame-level measurements of head movements with observation for each 1/30th of a second and created 10-frame moving sums to capture movement. Time-series data revealed group differences in the rate of head movement across all movies representing a wide range of stimuli, such as bubbles, a hopping bunny, and a woman singing a nursery rhyme paired with dynamic toys. An increase in the rate of head movement observed in young children with ASD during states of engaged attention might indicate underlying differences in the ability to maintain midline postural control and/or atypical engagement of attentional systems in young toddlers with ASD. These movements were not defined by spontaneous looking away from the stimulus, as was reported by Martin et al.14. Rather, they were characterized by a failure to keep the head in a still midline position while viewing the movie. This is distinct from the feature studied by Martin et al., which was characterized by greater yaw angular displacement and greater yaw and roll angular velocity, which was primarily present during the presentation of social stimuli and might reflect sensory modulation. The movements we describe in this paper may be similar to those described in previous studies of postural sway in older children with ASD, as well as school aged children with attention deficit hyperactivity disorder (ADHD) by Heiser et al.22. Heiser et al. used infrared motion analysis to record head movements during a continuous performance task and found that boys with ADHD moved their head 2.3 times as far as typically-developing boys performing the same task. In a study of siblings of children with ASD, Reiersen and colleagues23 found that siblings who have impaired motor coordination, features of attention deficit hyperactivity disorder (ADHD), or both are much more likely to have ASD than are other siblings. They suggest that identification of nonspecific traits that can amplify risk for ASD, such as attention and motor differences, could allow for earlier identification and targeted therapy that modify these traits and potentially reduce later risk for ASD.
Delays and differences in sensorimotor development have been noted across the lifespan in individuals with ASD from early infancy through adulthood24. For example, Lim et al. showed that postural sway and attention demands of postural control were larger in adults with ASD than in typically developed adults25. Morris et al. found that adults with ASD did not use visual information to control standing posture, in contrast to adults without ASD26. Brain imaging studies suggest that atypical motor function in autism may be related to increased sensitivity to proprioceptive error and a decreased sensitivity to visual error, aspects of motor learning dependent on the cerebellum27. Atypical presentation of motor functions of the cerebellum has been noted in children with ASD as young as 14 months of age. Esposito et al. identified significant differences in gait pattern, reflected in postural asymmetry, in toddlers with ASD as compared to those without ASD28.
The sample of toddlers with ASD was recruited from primary pediatric care where children suspected of having autism were then evaluated using gold-standard diagnostic methods. Although this method of recruitment increases the likelihood of obtaining a more representative population-based sample, it also results in a comparison group of toddlers without ASD that is much larger than the ASD sample. Because the sample of ASD toddlers in this study was relatively small, it will be important to replicate these findings with a larger group of children. A larger sample would also provide the statistical power to examine whether differences in postural control exist based on individual characteristics of children with ASD, such as age, sex, and co-morbid intellectual disability and/or ADHD.
Previous analyses of motor differences associated with ASD often have required labor-intensive coding of patterns of behavior that are recognizable by the naked eye. Moreover, such studies typically use a “top down” approach in which specific behaviors of interest are defined and then rated by more than one person (for reliability assessments). The use of digital phenotyping offers multiple advantages over previous methods that rely on human coding, namely, the ability to automatically and objectively measure dynamic features of behavior on a spatiotemporal scale that is not easily perceptible to the naked eye. Because digital approaches are scalable, they also allow for collection of larger data sets that can be analyzed using machine learning. We anticipate that the use of digital phenotyping will reveal a number of objective biomarkers, such as the head movements described in this report, which can be used as early risk indices and targets for intervention. By combining multiple features that reflect different aspects of sensorimotor function, including patterns of facial expressiomn, orienting, midline head movements, reaching behavior, and others, it might be possible to create a reliable, objective and automated risk profile for ASD and other neurodevelopmental disorders.
Change history
14 January 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Teitelbaum, P., Teitelbaum, O., Nye, J., Fryman, J. & Maurer, R. G. Movement analysis in infancy may be useful for early diagnosis of autism. Proc Natl Acad Sci USA 95, 13982–13987 (1998).
Esposito, G., Venuti, P., Maestro, S. & Muratori, F. An exploration of symmetry in early autism spectrum disorders: analysis of lying. Brain & development 31, 131–138, https://doi.org/10.1016/j.braindev.2008.04.005 (2009).
Flanagan, J. E., Landa, R., Bhat, A. & Bauman, M. Head lag in infants at risk for autism: a preliminary study. The American journal of occupational therapy: official publication of the American Occupational Therapy Association 66, 577–585, https://doi.org/10.5014/ajot.2012.004192 (2012).
Zappella, M. et al. What do home videos tell us about early motor and socio-communicative behaviours in children with autistic features during the second year of life–An exploratory study. Early human development 91, 569–575, https://doi.org/10.1016/j.earlhumdev.2015.07.006 (2015).
Dawson, G., Osterling, J., Meltzoff, A. N. & Kuhl, P. Case Study of the Development of an Infant with Autism from Birth to Two Years of Age. Journal of applied developmental psychology 21, 299–313, https://doi.org/10.1016/s0193-3973(99)00042-8 (2000).
Brisson, J., Warreyn, P., Serres, J., Foussier, S. & Adrien-Louis, J. Motor anticipation failure in infants with autism: a retrospective analysis of feeding situations. Autism: the international journal of research and practice 16, 420–429, https://doi.org/10.1177/1362361311423385 (2012).
Gima, H. et al. Early motor signs of autism spectrum disorder in spontaneous position and movement of the head. Experimental brain research 236, 1139–1148, https://doi.org/10.1007/s00221-018-5202-x (2018).
Hytonen, M., Pyykko, I., Aalto, H. & Starck, J. Postural control and age. Acta oto-laryngologica 113, 119–122 (1993).
Ghanouni, P., Memari, A. H., Gharibzadeh, S., Eghlidi, J. & Moshayedi, P. Effect of Social Stimuli on Postural Responses in Individuals with Autism Spectrum Disorder. J Autism Dev Disord 47, 1305–1313, https://doi.org/10.1007/s10803-017-3032-5 (2017).
Minshew, N. J., Sung, K., Jones, B. L. & Furman, J. M. Underdevelopment of the postural control system in autism. Neurology 63, 2056–2061 (2004).
Gouleme, N. et al. Postural Control and Emotion in Children with Autism Spectrum Disorders. Translational neuroscience 8, 158–166, https://doi.org/10.1515/tnsci-2017-0022 (2017).
Campbell, K. et al. Computer vision analysis captures atypical attention in toddlers with autism. Autism, 1362361318766247, https://doi.org/10.1177/1362361318766247 (2018).
Anzulewicz, A., Sobota, K. & Delafield-Butt, J. T. Toward the Autism MotorSignature: Gesture patterns during smart tablet gameplay identify children with autism. Scientific reports 6, 31107, https://doi.org/10.1038/srep31107 (2016).
Martin, K. B. et al. Objective measurement of head movement differences in children with and without autism spectrum disorder. Molecular autism 9, 14, https://doi.org/10.1186/s13229-018-0198-4 (2018).
Wu, D., Jose, J. V., Nurnberger, J. I. & Torres, E. B. A Biomarker Characterizing Neurodevelopment with applications inAutism. Scientific reports 8, 614, https://doi.org/10.1038/s41598-017-18902-w (2018).
Gotham, K., Risi, S., Pickles, A. & Lord, C. The Autism Diagnostic Observation Schedule: revised algorithms for improved diagnostic validity. J Autism Dev Disord 37, 613–627, https://doi.org/10.1007/s10803-006-0280-1 (2007).
Hashemi, J. et al. In Proceedings of the EAI International Conference on Wireless Mobile Communication and Healthcare. MobiHealth (2015).
De La Torre, F. IntraFace. Proceedings of the IEEE International Conference on Automatic Face and Gesture Recognition Workshops (2015).
Dementhon, D. D. L.D. Model-based object pose in 25 lines of code. International Journal of Computer Vision 15, 123–141 (1995).
Hashemi, J. et al. Computer vision tools for low-cost and noninvasive measurement of autism-related behaviors in infants. Autism Res Treat. 2014, 935686, https://doi.org/10.1155/2014/935686 (2014).
Hashemi, J. et al Computer vision analysis for quantification of autism risk behaviors. IEEE Transactions on Affective Computing, 1–1 (2018).
Heiser, P. et al. Objective measurement of hyperactivity, impulsivity, and inattention in children with hyperkinetic disorders before and after treatment with methylphenidate. European child & adolescent psychiatry 13, 100–104, https://doi.org/10.1007/s00787-004-0365-3 (2004).
Reiersen, A. M., Constantino, J. N. & Todd, R. D. Co-occurrence of motor problems and autistic symptoms in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 47, 662–672, https://doi.org/10.1097/CHI.0b013e31816bff88 (2008).
Cook, J. L., Blakemore, S. J. & Press, C. Atypical basic movement kinematics in autism spectrum conditions. Brain: a journal of neurology 136, 2816–2824, https://doi.org/10.1093/brain/awt208 (2013).
Lim, Y. H. et al. Effect of Visual Information on Postural Control in Adults with Autism Spectrum Disorder. J Autism Dev Disord, https://doi.org/10.1007/s10803-018-3634-6 (2018).
Morris, S. L. et al. Differences in the use of vision and proprioception for postural control in autism spectrum disorder. Neuroscience 307, 273–280, https://doi.org/10.1016/j.neuroscience.2015.08.040 (2015).
Marko, M. K. et al. Behavioural and neural basis of anomalous motor learning in children with autism. Brain 138, 784–797, https://doi.org/10.1093/brain/awu394 (2015).
Esposito, G., Venuti, P., Apicella, F. & Muratori, F. Analysis of unsupported gait in toddlers with autism. Brain & development 33, 367–373, https://doi.org/10.1016/j.braindev.2010.07.006 (2011).
Acknowledgements
Funding for this work was provided by NICHD 1P50HD093074, Duke Department of Psychiatry and Behavioral Sciences PRIDe award, Duke Education and Human Development Initiative, Duke-Coulter Translational Partnership Grant Program, National Science Foundation, and the Department of Defense. Some of the stimuli used for the movies were created by Geraldine Dawson, Michael Murias, and Sara Webb at the University of Washington. We gratefully acknowledge the editorial assistance of Elizabeth Sturdivant and the participation of the children and families in this study.
Author information
Authors and Affiliations
Contributions
G.D. and G.S. were responsible for conceptualizing and drafting the manuscript. All other authors reviewed and contributed to the manuscript. G.S. and J.H. were responsible for carrying-out the computer vision analyses. S.L. and V.S. were responsible for the statistical analyses. G.D., K.C., K.C. and S.V. were responsible for collection of the data and diagnostic confirmations. All authors were responsible for the design of the study and/or data analyses. K.C. and H.E. performed this work while employed at Duke University,
Corresponding author
Ethics declarations
Competing Interests
Geraldine Dawson is on the Scientific Advisory Boards of Janssen Research and Development, Akili, Inc., LabCorps, and Roche Pharmaceutical Company, has received grant funding from Janssen Research and Development, L.L.C. and PerkinElmer, speaker fees from ViaCord, and receives royalties from Guilford Press and Oxford University Press. Geraldine Dawson and Guillermo Sapiro are affiliated with DASIO, LLC.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dawson, G., Campbell, K., Hashemi, J. et al. Atypical postural control can be detected via computer vision analysis in toddlers with autism spectrum disorder. Sci Rep 8, 17008 (2018). https://doi.org/10.1038/s41598-018-35215-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35215-8
Keywords
This article is cited by
-
A hybrid framework for detection of autism using ConvNeXt-T and embedding clusters
The Journal of Supercomputing (2024)
-
Computer Vision Analysis of Caregiver–Child Interactions in Children with Neurodevelopmental Disorders: A Preliminary Report
Journal of Autism and Developmental Disorders (2024)
-
Blink rate and facial orientation reveal distinctive patterns of attentional engagement in autistic toddlers: a digital phenotyping approach
Scientific Reports (2023)
-
A tablet-based game for the assessment of visual motor skills in autistic children
npj Digital Medicine (2023)
-
Prediction of autistic tendencies at 18 months of age via markerless video analysis of spontaneous body movements in 4-month-old infants
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.