Abstract
Alternatives to antibiotics for improving productivity and maintaining the health of livestock health are urgently needed. The scope of this research was conducted to investigate the effects of two alternatives (Bacillus licheniformis and Saccharomyces cerevisiae) to monensin on growth performance, antioxidant capacity, immunity, ruminal fermentation and microbial diversity of fattening lambs. One hundred and sixty Dorper × Thin-tailed Han sheep (32 ± 3.45 kg BW) were randomly assigned into 5 treatments of n = 32 lambs/group. Lambs in the control group were fed a basal diet (NC) while the other four treatments were fed basal diets supplemented with monensin (PC), Bacillus licheniformis (BL), Saccharomyces cerevisiae (SC), and the combination of Bacillus licheniformis and Saccharomyces cerevisiae with protease (BS), respectively. The experiment lasted for 66 d. Feed intake was recorded every 2 d and lambs were weighed every 20 d. Ten lambs from each group were slaughtered at the end of the trial, and samples of serum and rumen fluid were collected. The results indicated that the dietary regimen did not affect the dry matter intake (DMI). The average daily gain (ADG) of BS treatment was significantly higher than NC group (P < 0.05). Compared with the NC treatment, the other four supplementation treatments increased the concentration of growth hormone (GH), insulin-like growth factor I (IGF-I) and insulin (INS) (P < 0.05). The malondialdehyde (MDA) and total antioxidant capacity (TAOC) showed no significant difference among the 5 treatments while the activity of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) of BS group was significantly increased (P < 0.05). The supplementation regimen decreased the concentration of ammonia Nitrogen (NH3-N) and increased the content of microbial crude proteins (MCP) (P < 0.05). The supplementation of antibiotics and probiotics reduced the concentrations of acetate and increased the concentrations of propionate (P < 0.05). The supplementation treatments increased the relative abundance of Lentisphaerae, Fibrobacteres and Tenericutes at the phylum level, whereas at the genus level, they increased the relative abundance of Fibrobacter (P < 0.05). Overall, this study confirmed the facilitating effect of B. licheniformis, S. cerevisiae and their compounds on growth performance, improve the antioxidant capacity and immune function, and beneficially manipulate ruminal fermentation and microbial diversity of fatting lambs.
Similar content being viewed by others
Introduction
The application of feed additives in livestock ration has an immense importance for improving nutrient utilization and productivity in the animal system. Monensin is a common feed additive which is associated the benefits of improving the efficiency of energy metabolism and rumen fermentation1. However, the use of antibiotics as a livestock feed additive is banned gradually due to the risk of accumulation of residues in animal products and the emergence of antibiotics-resistant bacterial strains2,3,4. Growing interest has focused on probiotics as alternatives of antibiotics which is characteristic of improving animal productive performance, inhibiting the colonization of pathogenic microorganisms and maintaining the balance of microflora of the digestive tract of the host5,6,7.
Numerous studies have described the role of Bacillus which has the probiotic characteristics of improving microbial balance, digestive processes and immunity of hosts. Research conducted on the influence of B. subtilis on ruminants showed that B. subtilis altered the rumen fermentation pattern of calves and increased the milk yield of ewes8. Studies of B. subtilis natto supplementation indicated that it was effective in increasing lactation performance of early lactation dairy cows possibly by altering the rumen fermentation pattern9. Qiao et al. reported that B. licheniformis increased ruminal apparent nutrient digestibility of neutral detergent fibre, acid detergent fibre, and organic matter while B. subtilis had no significant effect on rumen fermentation characteristics, duodenal microbial N flow and ruminal apparent nutrient digestibility8. To our knowledge, research on application of Bacillus in fattening lambs is scarce.
Supplementation of yeast is confirmed having the function of stabilizing rumen pH, increasing volatile fatty acids and decreasing ammonia concentration10. Various researches have been proposed to explain the effects that live yeast supplementation strains may have on rumen fermentation and ruminant production. The most commonly used strain of yeast is Saccharomyces cerevisiae, which can help to increase the digestibility of nutrients, improve the weight gain and feed conversion rate and modulate the composition of microbial ecosystem11. But still the results on the use of S. cerevisiae in ruminants are contradictory that in many cases no influence or opposing results were obtained. Part of the study revealed that S. cerevisiae supplemented diets did not improve growth performances of East-Friesian or Malpura lambs in fattening stage. The number of anaerobic and aerobic rumen bacteria was not affected by S. cerevisiae supplementation12.
Variable response to feeding B. licheniformis or S. cerevisiae in ruminant production emphasizes the need for understanding the actual mechanisms through which probiotics exert their functions. Considering the limited research relating to the combine effects of B. licheniformis and S. cerevisiae on microbiota, more insight into the mode of action of them is needed. The objective of this study was to investigate the effect of B. licheniformis, S. cerevisiae and combination of them on growth performance, antioxidant capacity, immunity, ruminal fermentation and microbial diversity of fattening lamb.
Results
Growth performance
The average daily gain (ADG), dry matter intake (DMI) and feed efficiency (FCR, DMI/ADG) were analyzed to determine the effects of B. licheniformi, S. cerevisiae and their combination on growth performance of lambs (Table 1). The IBW and DMI showed no difference among the five treatments. The ADG of lambs in PC, BL, SC and BS group showed no significant difference, while the ADG of lambs in the BS group was significantly higher than that in NC group (P < 0.05). The FCR of the BL group and BS group were significantly lower than that in NC group (P < 0.05).
Serum profiles
Compared with the NC and PC group, the serum concentration of GH and IGF-1 of lambs in SC and BS group were significantly increased (P < 0.05). Serum INS concentration of the lambs in BL and BS groups was higher than that in the NC group (P < 0.05). The TAOC and MDA showed no significant difference among the five groups, while the supplementation of B. licheniformis and S. cerevisiae increased the activity of SOD and GSH-Px. Lambs supplemented with B. licheniformis, S. cerevisiae and the compounds showed an increased serum concentration of IgA and IgG (P < 0.05) (Table 2).
Rumen fermentation
No difference of pH value was detected among the five groups. The concentration of NH3-N of lambs in NC group was significantly higher than that of the other four groups (P < 0.05). Compared to the NC group, dietary biological agent regimen significantly increased the MCP content of lamb (P < 0.05). No difference of MCP and TVFA were detected among the four supplementary treatments. The lambs supplemented with the monensin, B. licheniformis, S. cerevisiae or compounds had a lower percentage of acetate and higher percentage of propionate than that of the NC group (P < 0.05). The butyrate percentage of lambs in SC group was significantly higher than that in the other four groups while no obvious difference was detected among PC, BL and BS groups (Table 3).
Sequencing depth and index of Microbial Community
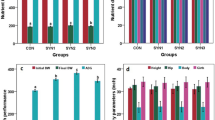
Illumina HiSeq sequencing generated a total of 2,328,167 clean reads with an average of 46,563 ± 20,780 reads per sample after data filtering, quality control, and removal of primers, chimeras, and low-confidence singletons. All reads were classified into 2,052 operational taxonomic units (OTUs) based on ≥97% nucleotide sequence identity between reads. The OTU numbers of NC, PC, BL, SC and BS was 971, 1026, 1034, 1038 and 1018, respectively. The α diversity index analysis was shown in Fig. 1. The community richness estimates and diversity indices showed no significant difference among the five groups. Principal coordinates analysis plots based on unweighted UniFrac distance metrics revealed difference of microbial diversity among the treatments (Fig. S1).
Bacterial composition across different treatments
All sequences were classified from phylum to species based on the SILVA taxonomic database. At the phylum level, 21 phyla were detected in the samples from the rumen fluid of lamb (Table 4, Fig. 2). The major sequences obtained from the five groups belonged to Bacteroidetes, Firmicutes, Lentisphaerae and Proteobacteria. However, the relative abundance of these predominant phyla varied considerably among the different calf groups. Within the CON treatment, Bacteroidetes (67.09%), Firmicutes (19.87%) and Proteobacteria (3.83%) were the dominant phyla while the other treatments were dominated by Bacteroidetes, Firmicutes and Lentisphaerae. The supplementation of B. licheniformis, and S. cerevisiae increased the percentage of Lentisphaerae and Tenericutes (P < 0.10). Compared to the other four treatments, the percentage of Fibrobacteres of SC treatment showed an increased tendency (P < 0.10).
At the genus level, 123 genera belonging to the 21 phyla were detected in the samples. In general, 29 most abundant shared genera with a relative abundance ≥1% were present in all samples across different treatments, and the relative abundance levels were markedly different among the five treatments (Table 5, Fig. 3). The major sequences obtained from the five treatments belonged to Rikenellaceae_RC9_gut_group, the taxa derived from Prevotellaceae (family), Erysipelotrichaceae_UCG-004 and Ruminococcaceae_UCG-010. Compared to the other four treatments, the supplementation of S. cerevisiae significantly increased the abundance of Fibrobacter (P < 0.05). We evaluated the correlation between the ruminal fermentation parameters and bacteria at genus level by performing spearman correlation analysis. Almost 27 genera belonging to 6 phyla were exhibited significant correlation with TVFA, acetic, butyrate, and propionic (P < 0.05) (Fig. 4).
Discussion
Studies of antibiotics in animal husbandry proved the benefits in terms of efficiency in the gain of body weight and reduction in the occurrence of subclinical diseases13,14. As an ionophore antibiotic, monensin has been added to high grain rations as feed additives for approximately four decades based on its ability to alter fermentation pattern, to promote growth, and to decrease the occurrence of subacute ruminal acidosis15,16. Antibiotic alternatives such as B. licheniformis, S. cerevisiae and plant extract have captured the interest of researchers because of the rise in antimicrobial resistance. In the current study, the supplementation of monensin, B. licheniformis, S. cerevisiae and the combination of B. licheniformis and S. cerevisiae with protease improved the growth performance and feed conversion ratio without affecting the DMI of lambs, but no significant differences were detected between the supplementation treatments. Our findings are in agreement with those of previous reports that probiotics supplementation and monensin were equally effective to increase the weight gain of lambs and no significant difference of DMI was detected1,17,18.
Growth Hormone (GH) is a peptide hormone of crucial importance in stimulating the production of IGF-1 and growth of organisms. Many researches have been carried out focused on the effect of supplementation of probiotics on the hormone level and growth performance of aquatic animals. The research on the sea bass juveniles and Oreochromis niloticus showed that the addition of probiotics or the combination of probiotics and prebiotics elevated the serum level of GH, IGF-I and INS, and increased the gene expression of them19,20. In the current study, the supplementation of S. cerevisiae and the compound of B. licheniformis and S. cerevisiae significantly increased the serum level of GH and IGF1.
Numerous studies have described the role of probiotics and bioactive compounds in enhancing the immunity and antioxidation of mammals21. Sohail et al. indicated that dietary supplementation of probiotic mixture reduced the some of the detrimental effects of oxidative damage of broilers22. Study of necrotizing enterocolitis caused by oxidant showed that the activity of SOD and GSH-Px were up-regulated with supplementation probiotics, prebiotics and synbiotics23. In this research, we also found that the compound of B. licheniformis and S. cerevisiae supplementation significantly increased of the serum content of SOD and GSH-Px. Meanwhile, the compound supplementation increased the serum concentrations of IgG and IgM, which are the indicators of nonspecific humoral immune competences in ruminants. Similar results were reported by Mendieta et al. and Andrea et al. that the addition of S. cerevisiae yeast cell walls and B. subtilis to diets increased serum IgG and IgM concentrations in broilers, respectively24,25.
Probiotics have been reported to elevate efficient of rumen function by stabilizing rumen pH and increasing volatile fatty acids production. However, the effect of probiotic supplementation on rumen pH of small ruminants has not been clearly defined. El-Ghani et al. recorded an increase in rumen pH while Tripathi et al. found no effect in rumen pH26,27. What is interesting is that some researchers reported a reduction in rumen pH after supplementation with Saccharomyces or an equal mixture of Saccharomyces to growing lambs. In this research, monensin or probiotics supplementation had no effect on rumen pH but decreased the concentration of ammoniacal nitrogen and increased the content of microbial proteins. The result indicated that probiotics could improve the efficiency of rumen fermentation, which is in accordance with the research of B. subtilis natto applied in dairy cows28. The change of ruminal NH3-N and MCP level that reflects the nitrogen utilization of the rumen microbials might attribute to the higher average daily gain of lambs.
Volatile fatty acids are the primary products of rumen fermentation which contribute to rumen epithelium development of fattening sheep. However, previous studies showed inconsistent idea of the effect of probiotics on ruminal VFAs. Sadiek et al. and Abd El-Ghani et al. reported that feeding of pronifer or S. cerevisiae increased the VFA production of sheep and goats, respectively27,29. On the contrary, studies of growing lambs or adult goats fed on probiotic supplemented diets indicated a significant reduction in ruminal VFA formation. In the present study, the concentration of total volatile fatty acids was not influenced by either a single administration of monensin, B. licheniformis, S. cerevisiae or the blended administration of B. licheniformis and S. cerevisiae, which is consistent with the studies of lambs and goats27,29. However, probiotic or monensin supplemented diets decreased the concentration of acetate while the concentration of propionate and butyrate was increased. There are many reports of this change in the A:P ratio17,30,31. The VFA profile is associated with effects on end-product composition and energy balance in ruminants. Propionate is the main source of glucose and a substrate for gluconeogenesis for the ruminant, while acetate and butyrate are precursors for long-chain fatty acid synthesis. It is in agreement with previous reports that high glucogenic to nonglucogenic VFA ratio is beneficial for growth of finishing cattle.
Ruminants and ruminal microorganisms have evolved together for millions of years and the addition of some probiotics to the diet has been reported to improve feed utilization and growth performance of the lamb by enhancing of rumen microbial ecosystem32. In this research, HiSeq sequencing of 16S rRNA was used to evaluate the changes in the ruminal bacterial community of lambs under different biological agents in diets. The relative abundance of Bacteroidetes and Firmicutes accounted for over 84% among the five treatments, which is in agreement with previous reports that most of the bacterial community of mammals is affiliated with the phyla of Bacteroidetes and Firmicutes33. The richness of phylum Lentisphaerae, Fibrobacteres and Tenericutes was varied among the treatments, which indicated that the supplementation of B. licheniformis or S. cerevisiae affected the distribution of bacteria in the rumen. Several studies have shown that S. cerevisiae is able to stimulate lactate utilizing bacteria thus stabilizing ruminal pH and favouring fibre degradation. In this research, S. cerevisiae supplementation was increased the genus level of Fibrobacter but had no effect on ruminal pH among the five treatments which was consistent with the previous studies32. However, the other genera with a relative abundance above 1% showed no significant difference among the five treatments. Previous studies reported the potential of probiotics in manipulation of gastrointestinal microbiota; however, their efficacies often vary and are inconsistent. Numerous factors, such as the type and level of probiotics, ingredient composition of the experimental diet, and management constraints, have been shown to markedly affect the structure and activities of microbial communities in livestock animals.
Conclusion
This study demonstrated that dietary of B. licheniformis, S. cerevisiae and their compounds improved the growth performance of fattening sheep and could be the alternatives of monensin. It also highlights that supplementation of S. cerevisiae and the compound of B. licheniformis and S. cerevisiae significantly increased the serum level of GH and IGF1, improved the antioxidant capacity and immune function of lambs. It was discovered that B. licheniformis, S. cerevisiae and their compounds increased the nitrogen utilization of the rumen microbials and changed the fermentation pattern which was beneficial for growth of fattening sheep. Based on 16 S rRNA gene sequencing method, this study indicated that supplementation of S. cerevisiae increased the genus level of Fibrobacter. Overall, this study confirmed the effect of B. licheniformis, S. cerevisiae and their compounds on growth performance, antioxidant capacity, immune function, ruminal fermentation and microbial diversity of fattening lambs which are of great importance for the targeted improvement of nutrient levels in ruminants.
Materials and Methods
Ethics statement
The animal experiments were conducted at the Fuchuan Feed Technology Co., Ltd. Modern sheep breeding base, Inner Mongolia, China. The experimental protocol was approved by the Chinese Academy of Agricultural Sciences Animal Ethics Committee, and humane animal care and handling procedures were followed throughout the experiment.
Animals and management conditions
One hundred and sixty lambs (Dorper × Thin-tailed Han) with an initial bodyweight of 32 ± 3.45 kg were randomly assigned into 5 treatments with 4 replicates of each treatment and 8 lambs per replicate. The treatments were as follows: 1) negative control (NC): fed a basal diet; 2) positive control (PC): fed a basal diet supplemented with monensin (21 mg/kg); 3) B. licheniformis treatment (BL): fed a basal diet supplemented with B. licheniformis (4 × 109 CFU); 4) S. cerevisiae treatment (SC): fed a basal diet supplemented with S. cerevisiae (3.2 × 109 CFU); 5) combined B. licheniformis and S. cerevisiae treatment (BS): fed a basal diet supplemented with both B. licheniformis (6 × 109 CFU) and S. cerevisiae (4 × 109 CFU) with protease, respectively.
The basal diet was formulated according to NRC and our previous research and the ingredients and chemical composition of the diet are present in Table 634,35. The diets were provided in the form of pellet feed and lambs at the bodyweight of 30~40 kg were supplemented with Leymus chinensis about 20% of total feeding. The feed and water were available ad libitum during the 66d feeding period. Feed intake was recorded every 2 d and lambs were weighed every 20 d to calculate average daily gain (ADG), average daily feed intake (ADFI) and feed efficiency (G:F ratio). Ten lambs from each treatment were slaughtered at the end of the trial.
Blood profiles
Blood samples were collected before morning feeding by jugular veinpuncture into one 10 ml vacutainer tubes. The samples were centrifuged (5000 g, 20 min, 4 °C) to obtain the serum, which was frozen at −20 °C until further analysis. The activity of TAOC, SOD, GSH-Px and the content of MDA were analyzed with commercial kits from Nanjing Jiancheng Bioengineering Institute (Jiangsu, China) according to the manufacturer’s protocols. Serum concentrations of IGF-1, immunoglobulin (Ig) A, M and G were detected with a microplate reader (Thermo Scientific, Multiskan GO, Waltham, MA, USA). The serum level of growth hormone and insulin was detected with radioimnunoassay kit against its specifications.
Chemical analyses
The composition and nutrient levels of milk replacer and starters were analyzed according the official methods of analysis (Association of Official Analytical Chemists: Washington, DC)36. Samples of the diet were analyzed for dry matter (DM) by drying samples in an airforced oven at 135 °C for 2 h (method 930.15; AOAC 1990). Nitrogen was determined by the Kjeldahl method and crude protein (CP) was calculated as 6.25 × N. Ether extract (EE) was measured by the weight loss of the dry matter upon extraction with diethyl ether in a Soxhlet extraction apparatus for 8 h (method 920.85; AOAC 1990). Ca was analyzed using an atomic absorption spectrophotometer (M9W-700, Perkin-Elmer Corp., Norwalk, CT, USA) (method 968.08; AOAC 1990). The total P was analyzed by the molybdovanadate colorimetric method (method 965.17; AOAC 1990) using a spectrophotometer (UV-6100, Mapada Instruments Co., Ltd., Shanghai, China).
Ruminal fermentation parameters
Samples of ruminal digesta were collected after slaughtered and the pH of the digesta was measured immediately using a pH metre (Model 144 PB-10, Sartorius Co., Germany). A 10-ml sample of the strained fluid was collected, acidified with 2 ml 25% (w/v) metaphosphoric acid, and stored at −20 °C for analysis of volatile fatty acids (VFA) and NH3-N. Volatile fatty acids in ruminal fluid were determined by gas chromatography (GC) using methyl valerate as the internal standard in an Agilent 6890 series GC equipped with a capillary column (HP-FFAP19095F-123, 30 m, 0.53 mm diameter and 1 lm thickness)37. Ammonia-N was assessed by the colorimetric method described by Ma et al.38. The microbial protein was measured by the trichloroacetic acid precipitation method39.
DNA Extraction, amplification and Hiseq sequencing
Rumen samples were thoroughly homogenized and one millilitre of rumen fluid was centrifuged for 10 min at room temperature at 13,200 rpm. The supernatant was discarded and the remaining pellet was resuspended in 400 ml of nuclease-free water. Microbial DNA was extracted using a commercial DNA Kit (Omega Bio-tek, Norcross, GA, U.S.) according to manufacturer’s protocols. After the assessment of integrity and purity of DNA, amplification and sequencing were performed as described by Mao et al.40. The V3-V4 region of the bacteria 16S ribosomal RNA genes were amplified using primers of 338 F (5′-ACTCCTACGGGAGGCAGCAG-3′) and 806 R (5′-GGACTACHVGGGTWTCTAAT-3′) with the barcode attached. The PCR products were purified using the High Pure PCR Cleanup Micro Kit (Roche Diagnostics Ltd, Burgess Hill, West Sussex, UK) and quantified using QuantiFluor™ -ST (Promega, U.S.). Purified amplicons were pooled in equimolar and paired-end sequenced (2 × 250) on an Illumina HiSeq platform (Illumina Inc., San Diego, CA) that finished by RealBio Technology Co., Ltd.
Data processing
The effective tags was obtained after demultiplexed, quality filtered, and analyzed using the Quantitative Insights into Microbial Ecology (QIIME, v.1.8.0) and the assembled sequences were assigned to operational taxonomic units (OTUs) at a 97% identity threshold using UPARSE41,42. The α diversity index, including Chao1, ACE, Shannon and Simpson were performed using QIIME43,44,45. The phylogenetic affiliation of each 16S rRNA gene sequence was analyzed by Ribosomal Database Project (RDP) Classifier (http://rdp.cme.msu.edu/) against the SILVA 16S rRNA database using confidence threshold of 70%. Principal coordinates analysis (PCoA) was used to compare treatments of samples based on unweighted Uni-Frac distance metrics46. Spearman correlation analysis between bacterial and ruminal fermentation parameters was performed using R corrplot.
Statistical analysis
The growth performance, serum profiles and rumen fermentation of lambs, the alpha diversity indices, and the quantification of total bacteria were analyzed using a completely randomized design with one-way analysis of variance (ANOVA) (version 9.2, SAS Institute Inc., Cary, NC, USA). Duncan’s method for multiple comparisons was used for variables where the treatment effect was significant (P < 0.05), and 0.05 ≤ P < 0.10 was designated as a tendency. The sequencing data of this research was submitted to the Sequence Read Archive (SRA) with an accession number of SRP 162913.
References
Mirzaei-Alamouti, H. et al. Both monensin and plant extract alter ruminal fermentation in sheep but only monensin affects the expression of genes involved in acid-base transport of the ruminal epithelium. Anim Feed Sci Tech. 219, 132–143 (2016).
Rong, B., Carmen, A. & Ulrich, S. Field evaluation of the effect of a probiotic-containing Bacillus licheniformis and Bacillus subtilis spores on the health status, performance, and carcass quality of grower and finisher pigs. Journal of Veterinary Medicine A Physiology Pathology Clinical Medicine. 51, 306–312 (2010).
Hayes, D. J., Jensen, H. H. & Fabiosa, J. Technology choice and the economic effects of a ban on the use of antimicrobial feed additives in swine rations. Food Control. 13, 97–101 (2002).
Casewell, M., Friis, C., Marco, E., McMullin, P. & Phillips, I. The European ban on growth-promoting antibiotics and emerging consequences for human and animal health. J Antimicrob Chemoth. 52, 159–161 (2003).
Perdigon, G., Galdeano, C. M., Valdez, J. C. & Medici, M. Interaction of lactic acid bacteria with the gut immune system. Eur J Clin Nutr. 56, S21 (2002).
Fujiwara, K. et al. Effect of Bacillus subtilis var. natto fermented soybean on growth performance, microbial activity in the caeca and cytokine gene expression of domestic meat type chickens. J Poult Sci. 46, 116–122 (2009).
Kritas, S. K. & Morrison, R. B. Evaluation of probiotics as a substitute for antibiotics in a large pig nursery. Vet Rec. 156, 447 (2005).
Qiao GH, Shan AS, Ma N, Ma QQ & Sun ZW. Effect of supplemental Bacillus cultures on rumen fermentation and milk yield in Chinese Holstein cows. J anim physiol An N. (2009).
Peng, H. et al. Effect of feeding Bacillus subtilis natto fermentation product on milk production and composition, blood metabolites and rumen fermentation in early lactation dairy cows. J Anim Physiol An N. 96, 506–512 (2012).
Wang, Z. et al. Comparison of two live Bacillus species as feed additives for improving in vitro fermentation of cereal straws. Anim Sci J. 87, 27–36 (2016).
Stella, A. V. et al. Effect of administration of live Saccharomyces cerevisiae on milk production, milk composition, blood metabolites, and faecal flora in early lactating dairy goats. Small Ruminant Res. 67, 7–13 (2007).
Mikulec, Z., Mašek, T., Habrun, B. & Valpotic, H. Influence of live yeast cells (Saccharomyces cerevisiae) supplementation to the diet of fattening lambs on growth performance and rumen bacterial number. Vet Arhiv. 80, 695–703 (2010).
Cromwell, G. L. Why and how antibiotics are used in swine production. Anim Biotechnol. 13, 7–27 (2002).
Chattopadhyay MK. Use of antibiotics as feed additives: a burning question. FRONT MICROBIOL. 5 (2014).
Nagaraja, T. G., Avery, T. B., Bartley, E. E., Roof, S. K. & Dayton, A. D. Effect of lasalocid, monensin or thiopeptin on lactic acidosis in cattle. J Anim Sci. 54, 649–658 (1982).
Duffield, T. F., Merrill, J. K. & Bagg, R. N. Meta-analysis of the effects of monensin in beef cattle on feed efficiency, body weight gain, and dry matter intake. J Anim Sci. 90, 4583 (2012).
Hernández, R. et al. Effect of a Yeast Culture on Nitrogen Balance and Digestion in Lambs Fed Early and Mature Orchard Grass. J Appl Anim Res. 35, 53–56 (2009).
Antunovic, Z. et al. Effect of probiotic supplementation before and after weaning on fattening performance, biochemical parameters, and acid-base balance of lambs. Tierärztliche Umschau. 66, 106–111 (2011).
Carnevali, O. et al. Growth improvement by probiotic in European sea bass juveniles (Dicentrarchus labrax, L.), with particular attention to IGF-1, myostatin and cortisol gene expression. Aquaculture. 258, 430–438 (2006).
Mustafa, M. The Influence Of Synbiotic On Growth And Expression Of GH. GHR1 And IGF-I Genes In Oreochromis niloticus L Fingerlings. 6, 176–182 (2015).
Rajput, I. R., Li, W. F., Li, Y. L., Jian, L. & Wang, M. Q. Application of probiotic (Bacillus subtilis) to enhance immunity, antioxidation, digestive enzymes activity and hematological profile of Shaoxing duck. Pak Vet J. 33, 69–72 (2013).
Sohail, M. U. et al. Single or combined effects of mannan-oligosaccharides and probiotic supplements on the total oxidants, total antioxidants, enzymatic antioxidants, liver enzymes, and serum trace minerals in cyclic heat-stressed broilers. Poultry Sci. 90, 2573–2577 (2011).
D’Souza, A. et al. Effects of probiotics, prebiotics, and synbiotics on messenger RNA expression of caveolin-1, NOS, and genes regulating oxidative stress in the terminal ileum of formula-fed neonatal rats. Pediatr Res. 67, 526–531 (2010).
Mendieta, C. R. et al Effect of the Addition of Saccharomyces Cerevisiae Yeast Cell Walls to Diets with Mycotoxins on the Performance and Immune Responses of Broilers. J Poult Sci. (2017).
Andrea, K. M. et al. Effect of Bacillus subtilis supplementation in diets on growth performance, carcass quality, gut microflora and immune response of broiler chickens. Magy Allatorvosok. 133, 132–140 (2011).
Tripathi, M. K., Karim, S. A., Chaturvedi, O. H. & Verma, D. L. Effect of different liquid cultures of live yeast strains on performance, ruminal fermentation and microbial protein synthesis in lambs. Journal of Animal Physiology & Animal Nutrition. 92, 631–639 (2010).
Abd El-Ghani, A. A. Influence of diet supplementation with yeast culture (Saccharomyces cerevisiae) on performance of Zaraibi goats. Small Ruminant Res. 52, 223–229 (2004).
Sun, P., Li, J., Bu, D., Nan, X. & Du, H. Effects of Bacillus subtilis natto and Different Components in Culture on Rumen Fermentation and Rumen Functional Bacteria In Vitro. Curr Microbiol. 72, 589–595 (2016).
Sadiek, A. & Böhm, J. Influence of Pronifer® as a probiotic on the rumen fluid and blood parameters of sheep fed different roughage concentrate based diets. Wien Tierarztl Monat. 88, 4–10 (2001).
Erasmus, L. J., Botha, P. M. & Kistner, A. Effect of yeast culture supplement on production, rumen fermentation, and duodenal nitrogen flow in dairy cows. J Dairy Sci. 75, 3056–3065 (1992).
GA, H., RW, H., KA, D., RJ, H. & KB, B. Influence of addition of yeast culture supplement to diets of lactating cows on ruminal fermentation and microbial populations. J Dairy Sci. 71, 2967–2975 (1988).
McCann, J. C., Elolimy, A. A. & Loor, J. J. RumenMicrobiome, Probiotics, and Fermentation Additives. . Veterinary Clinics of North America: Food Animal Practice. 33, 539–553 (2017).
Ley, R. E., Lozupone, C. A., Hamady, M., Knight, R. & Gordon, J. I. Worlds within worlds: evolution of the vertebrate gut microbiota. Nat Rev Microbiol. 6, 776–788 (2008).
Ma, T. et al. Effect of feed intake on metabolizable protein supply in Dorper × thin-tailed Han crossbred lambs. Small Ruminant Res. 132, 133–136 (2015).
Xu, G. S. et al. Energy requirements for maintenance and growth of early-weaned Dorper crossbred male lambs. Livest Sci. 177, 71–78 (2015).
AOAC. Official methods of analysis (15th ed). Association of Official Analytical Chemists, Washington, D.C (1990).
Cao, Y. C. & Yang, H. J. Ruminal digestibility and fermentation characteristics in vitro of fenugreek and alfalfa hay combination with or without the inoculation of Neocallimastix sp. YAK11. Anim Feed Sci Tech. 169, 53–60 (2011).
Ma, T., Tu, Y., Zhang, N. F., Deng, K. D. & Diao, Q. Y. Effect of the Ratio of Non-fibrous Carbohydrates to Neutral Detergent Fiber and Protein Structure on Intake, Digestibility, Rumen Fermentation, and Nitrogen Metabolism in Lambs. Asian-Australas J Anim Sci. 28, 1419–1426 (2015).
Nandakumar, M. P., Shen, J., Babu Raman, A. & Marten, M. R. Solubilization of Trichloroacetic Acid (TCA) Precipitated Microbial Proteins via NaOH for Two-Dimensional Electrophoresis. J Proteome Res. 2, 89–93 (2003).
Mao, S., Zhang, R., Wang, D. & Zhu, W. The diversity of the fecal bacterial community and its relationship with the concentration of volatile fatty acids in the feces during subacute rumen acidosis in dairy cows. Bmc Vet Res. 8, 237 (2012).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 7, 335–336 (2010).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods. 10, 996–998 (2013).
Soren, N. M., Tripathi, M. K., Bhatt, R. S. & Karim, S. A. Effect of yeast supplementation on the growth performance of Malpura lambs. Trop Anim Health Pro. 45, 547–554 (2013).
Plata, F. P., DMM, G., Bárcena-Gama, J. R. & GM, S. Effect of a yeast culture (Saccharomyces cerevisiae) on neutral detergent fiber digestion in steers fed oat straw based diets. Animal Feed Science & Technology. 49, 203–210 (1994).
Amato, K. R. et al. Habitat degradation impacts black howler monkey (Alouatta pigra) gastrointestinal microbiomes. Isme J. 7, 1344–1353 (2013).
Lozupone, C. & Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 71, 8228–8235 (2005).
Acknowledgements
This study was supported by the Inner Mongolia Science and Technology Key Project (201407011) and Earmarked Fund for China Agriculture Research System (CAAS-39).
Author information
Authors and Affiliations
Contributions
The authors’ contributions were as follows: Q.Y.D. and W.Y.W. contributed to the study conception and design; P.J., T.M. and F.W. performed the experiments; K.C. and P.J. analyzed the data and wrote the draft manuscript; D.Y. and Y.F.W. maintained the animals; and B.L.G. and L.F.Z. provided scientific direction and interpreted the results.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jia, P., Cui, K., Ma, T. et al. Influence of dietary supplementation with Bacillus licheniformis and Saccharomyces cerevisiae as alternatives to monensin on growth performance, antioxidant, immunity, ruminal fermentation and microbial diversity of fattening lambs. Sci Rep 8, 16712 (2018). https://doi.org/10.1038/s41598-018-35081-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-35081-4
Keywords
This article is cited by
-
Targeted Screening of Fiber Degrading Bacteria with Probiotic Function in Herbivore Feces
Probiotics and Antimicrobial Proteins (2024)
-
Potential of camel rumen derived Bacillus subtilis and Bacillus velezensis strains for application in plant biomass hydrolysis
Molecular Genetics and Genomics (2023)
-
Characterization and Assessment of Sheep-Origin Probiotic Bacillus licheniformis B63 Strain for Potential Use in Intestinal Health and Disease
Probiotics and Antimicrobial Proteins (2023)
-
Identification and characterization of antimicrobial peptide produced by indigenously isolated Bacillus paralicheniformis UBBLi30 strain
3 Biotech (2020)
-
Dietary Probiotic Bacillus licheniformis H2 Enhanced Growth Performance, Morphology of Small Intestine and Liver, and Antioxidant Capacity of Broiler Chickens Against Clostridium perfringens–Induced Subclinical Necrotic Enteritis
Probiotics and Antimicrobial Proteins (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.