Abstract
The Chikungunya (CHIK) virus is epidemic in Brazil, with 170,000 cases in the first half of 2016. More than 60% of patients present relapsing and remitting chronic arthralgia with debilitating pain lasting years. There are no specific therapeutic agents to treat and rehabilitee infected persons with CHIK. Persistent pain can lead to incapacitation, requiring long-term pharmacological treatment. Advances in non-pharmacological treatments are necessary to promote pain relief without side effects and to restore functionality. Clinical trials indicate transcranial direct current stimulation (tDCS) can treat a broad range of chronic pain disorders, including diffuse neuromuscular pain and arthralgia. Here, we demonstrate that the tDCS across the primary motor cortex significantly reduces pain in the chronic phase of CHIK. High-resolution computational model was created to analyze the cortical electric field generated during tDCS and a diffuse and clustered brain current flow including M1 ipsilateral and contralateral, left DLPFC, nucleus accumbens, and cingulate was found. Our findings suggest tDCS could be an effective, inexpensive and deployable therapy to areas lacking resources with a significant number of patients with chronic CHIK persistent pain.
Similar content being viewed by others
Introduction
The Chikungunya (CHIK) virus is a mosquito-transmitted alphavirus, first isolated in 1953 during an epidemic in Tanzania1. CHIK transmission occurs through the bite of an infected mosquito, Aedes aegypti or Aedes albopictu, from mother to fetus in the intrauterine perinatal period, through blood transfusion and sexual intercourse2. Mainly transmitted in urban areas, CHIK constitutes a significant public health problem in many countries, including in the Brazilian Northeast3. The burden of CHIK in Latin America is especially severe in Brazil with 170,000 cases in the first half of 2016, accounting for 94% of confirmed CHIK cases in the Americas4.
Patients infected with CHIK virus will develop persistent rheumatologic and general disabling symptoms such as joint pain, fever, asthenia, headache, retro-orbital pain, photosensitivity, muscular pains, back pain and tenosynovitis1. CHIK virus induced disease shares many similarities with illnesses caused by other arthritogenic alphaviruses1. Most patients develop severe and often debilitating polyarthralgia that is usually bilateral and symmetric, most commonly in ankles, wrists, and phalanges1. Although complete recovery of these symptoms occurs in some cases within four weeks, several individuals evolve into chronic disability pain for up to 6 years5,6. According to Silva, a large number of patients experience chronic musculoskeletal disorders and usually respond to some extent to analgesics, anti-inflammatory treatments, and physiotherapy1. In an attempt to reduce the physical symptoms caused by CHIK virus, several medications have been tested, such as chloroquine and meloxicam with limited effectiveness7.
The acute or chronic painful process generated by CHIK virus appears to be associated with the expression of specific metabolites in the human body influencing its severity after infection8,9. However, approximately 5% of patients meet the criteria for chronic inflammatory rheumatism, which may be destructive and deforming with severe pain symptoms and disability1. The current challenge for rehabilitation medicine is to provide an optimal treatment for CHIK rheumatic disorders and effectively disrupt the development course of CHIK symptoms.
Transcranial Direct Current Stimulation (tDCS) is a battery-powered non-invasive neuromodulation technique in which low amplitude direct current is conducted to the cortex10. Clinical trials indicate tDCS can treat a broad range of chronic pain disorders, including diffuse neuromuscular pain11,12,13. Studies with anodal stimulation of the primary motor cortex (M1) for the treatment of chronic pain syndromes including chronic back pain, fibromyalgia, trigeminal neuralgia, arthrosis and polyneuropathy demonstrated the effectiveness of tDCS on pain and mood symptoms14,15. Chronic pain may reflect not only local inflammation and peripheral sensitization, by having origins in the central nervous system reflecting maladaptive changes in brain excitability16. Active stimulation was found to be effective for reducing chronic rheumatic symptoms, with no adverse events and good tolerability16.
Anodal M1 and cathodal contra lateral supraorbital right (M1-SO) montages of tDCS may treat chronic pain by influencing pain matrix includes motor cortex and deep brain regions producing long-lasting changes in cortical connectivity or excitability17,18. tDCS thus offers promise as a novel neuromodulation approach for pain-related networks to alleviate CHIK rheumatic chronic symptoms, such as muscular pain and arthralgia. Moreover, tDCS is a low cost, safe, and mobile intervention that can be deployed on a large scale across locations such as health posts, clinics and in home-visit rehabilitation19.
The present study aim to investigate the hypothesis that tDCS would improve pain and functionality in subjects with chronicle CHIK arthralgia.
Materials and Methods
The study was conducted in the Faculty of Health Science of Trairí, Federal University of Rio Grande do Norte. It was approved by the local institutional Ethical Committee (approval number 1.563.690) and conducted according to Declaration of Helsinki (1964), resolution No. 466/12 of the National Health Council, and the CONSORT recommendations20. The study was registered in clinicaltrials.gov on December 5, 2016 with the identifier NCT02993952. All participants provided written informed consent.
Participants
The sample size was estimated based on previous studies of tDCS effect on rheumatic diseases14,21,22, and with significance of 0.05 and power of 0.80. Fregni suggested the mean reduction of 3 points in Visual Analogue Scale (VAS) for the experimental group was expected in contrast to no improvement in the sham group in rheumatic disease such as fibromialgia22. Thus, the sample size resulted in two groups of 10 participants.
All participants were included with previous serologic confirmation of Chikungunya (CHIK) virus infection based on CHIK virus IgG and IgM detected by direct ELISA/IgM/Euroimmun, according to the Central Laboratory (LACEN, Brazil). Paticipants from local communities of the Northeast of Brazil were recruited through advertisements in the electronic media and by health professionals from the communities. The inclusion criteria were positive laboratory tests for the CHIK virus for at least 6 months (chronic phase), preserved intellectual capacity determined by the Mini Mental State Examination (MMSE), physical capacity to do physical evaluation, and between 18 and 65 years old. The exclusion criteria were pain clearly related to any other etiology, such as dengue, zika, rheumatoid arthritis, gout, lupus, neurologic and muscular diseases, psychiatric illness, and history of drug abuse, signs or history of dizziness or epileptic disease, pregnancy, signs of severity and/or indication of hospitalization and metal implants in the head. Participants were not taking analgesic or other pain relieve medication during this study. All participants were women and only one men was included in Sham-tDCS.
Experimental design
The flowchart illustrating the process of the study is shown in Fig. 1. This is a parallel, sham, randomized, double-blind trial with 1-week follow-up. Data were collected from December 2016 to March 2017. A total of 283 individuals were initially identified with laboratory tests for CHIK fever, but after applying the inclusion and exclusion criteria, 53 candidates with positive markers for CHIK fever were considered eligible for the study. Moreover, 33 individuals declined research participation due to the study protocol or the available time. Thus, 20 individuals were selected and randomized into 2 groups: active-tDCS group (n = 10) or sham-tDCS group (n = 10). One participant from the experimental group declined to continue because the improvement was not as expected, so intention-to-treat analysis was done in this group.
Flowchart summarizing the study. Twenty participants were randomized into two groups receiving active-tDCS or sham-tDCS on M1. Only one participant of active-tDCS group withdrew during the stimulation phase. Missing data were treated by intention-to-treat analysis. None of the participants received medication throughout the trial. Patients tolerated the tDCS treatments well, and no adverse effects occurred.
Participants were evaluated 1 week before the intervention (baseline), 1 day after the first intervention (day 1), after the last day of intervention (day 5), and 1 week after the last day of intervention. A random numerical sequence was generated (www.randomization.com) to assign each participant to either the active-tDCS group or the sham-tDCS group by an independent researcher not involved with either stimulation or assessments. Participants and researchers were blind to group allocation throughout the trial.
tDCS procedures
Participants were seated in a comfortable chair with back and arm support. tDCS was carried out with anode electrode on the left primary motor cortex (C3) and the cathode electrode on the right supraorbital region (Fp2) according to the international 10–20 EEG system (“M1-SO” assembly). Electrodes were encased in electrode sponges (7 × 5 cm) soaked in saline solution (150 mMols of NaCl diluted in water Milli-Q), and supported by a rubber band. Active tDCS was performed on 5 consecutive days of constant current of 2 mA for 20 min. A gradual current ramp-up and ramp-down of 30 s was used. Sham-tDCS was performed on 5 consecutive days with electrodes placed on the same position, and a constant current of 2 mA was delivered only for 30 s (10-s ramp-up) of the 20 min. Electrodes (anode and cathode) were connected to a battery (9 V) powered constant current stimulator with current verified by a precision digital multimeter (DT832, WeiHua Electronic Co., Ltd, China).
High-resolution computational model
Finite element models were created to analyze the cortical electric field generated during tDCS. High-resolution MRIs were segmented into seven tissue/material masks of varying conductivities through a combination of automated and manual tools. Computer generated models of electrodes, gel, and/or sponge pads were incorporated into the segmentation. Volume meshes were generated, boundary conditions were applied, and the Laplace equation \((\nabla \cdot (\sigma \nabla V)=0)\) was solved. The resulting cortical electric field was interpreted as a correlate for stimulation and modulation. Our results corroborate previous modeling studies, which show diffuse and clustered brain current flow that includes left motor regions and other regions implicated in the treatment of pain and other regions of interest. (in V/m: M1 ipsilateral 0.23, contralateral 0.15, left DLPFC 0.23, Nucleus accumbens 0.22, and cingulate 0.25).
Instruments and assessments
Socio-demographic data were collected, such as age, disease duration, pain intensity, pain location, marital status, and education. The measures of the outcomes were collected one week before the first simulation (baseline), after the first stimulation (day 1), after the last stimulation (day 5) and one week after the last stimulation (follow-up). Pain levels were evaluated with the self-reported Visual Analogue Scale (VAS), McGill Pain Questionnaire and Brief Pain Inventory (BPI) (short form).
Primary outcomes
VAS consisted of a gradient color-coded scale ranging from 0 (green; corresponding to complete absence of pain) to 10 (red; corresponding to worst imaginable pain) in intervals of 1 cm23. VAS was asked by a researcher after the first interview (one week before the first simulation - baseline), after day 1 and day 5 of tDCS and one week follow-up. In addition, participants recorded their daily pain intensity using VAS every night before sleeping. This pain diary completed 21 days of measurement. To analyze the homogeneity between groups, initial VAS pain levels were calculated for each patient by using the average of 1 week of pain diary reports.
Secondary outcomes
McGill Pain Questionnaire was used to characterize participants’ pain. It presents 20 items 10 sensorial, 5 affective, 1 evaluative and 4 miscellaneous. Each of these items displays from 2 to 5 options, and only one or none must be chosen23. The BPI (short form) was used to assess pain severity and impact in daily living activities. It presents 15 items, including 2 multi-item scales to measure pain and its impact on functionality and well-being24.
Participants performed a hand grip test (hydraulic hand dynamometer Saehan® model SH5001) seated in a chair with armrests, shoulders at 0° of adduction, abduction, flexion, extension, internal and external rotation; elbow at 90° of flexion and wrist between 0° and 30° of extension. They were instructed to squeeze three times as hard as possible for 3 seconds and rest for 30 seconds between repetitions to have the arithmetic mean25. Strength of the lower limbs was evaluated by the 30-second chair stand test. A chair with 43 cm high, with backrest, without armrest and a stopwatch was used. Participants were instructed to keep their arms crossed on their chest, to stand and to sit with their backs resting on the backrest as fast as possible in 30 seconds. The score corresponds to the number of times performed to repeat the movement in 30 seconds26.
Upper limb flexion strength (dominant or more painful) was evaluated by the 30-second arm curl test. The test consisted of requiring the participants to hold a 2 kg weight and to perform full flexions and extensions as quickly as possible within 30 seconds. Participants could discontinue the test at any time, if necessary27.
To assess the flexibility of the lower limbs (posterior thigh muscles), a chair sit and reach test was performed. A chair with 43 cm high and 50 cm of backrest was used. Participants were instructed to sit on the edge of the chair with flat feet on the floor, knees and ankles at 90° flexion; then the dominant or painful leg was stretched (hip and knee) with the calcaneus supported on the floor and with the ankle flexed at 90°. With overlapping hands and with tips of the middle fingers even, participants tried to reach their toes and hold the reach for two seconds in three repetitions to get the arithmetic mean. A negative score was recorded if the middle fingers did not reach the toes, and a score was positive if the middle fingers were beyond the toes27.
Flexibility of the upper limbs was performed using the scratch flexibility test. The participants were instructed to pass one hand (dominant or more painful) over the shoulder to assess flexibility of the shoulder in flexion, abduction and external rotation and try to reach the other hand to assess extension, adduction and internal rotation on the center of the back. Three repetitions were made to obtain the arithmetic mean of the results. The scores were considered negative if there was any distance between the middle fingers, and positive if the middle fingers overlapped27.
A short form health survey (SF-36) was used28 to evaluate quality of life in the last four weeks. It consists of a 36 item questionnaire divided into 8 domains: functional capacity, limitation by physical aspects, pain, general health, vitality, social and emotional aspects and mental health. These domains have between 2 and 6 response options.
Data analyses
Analyses were performed using the SPSS software (V.19.0, Chicago, USA) and Graph Pad Prism 5. Quantitative variables were expressed as means and standard deviations (SD). The Shapiro-Wilk and Levene’s test were applied to assess the normality of the distribution and homogeneity of variance of the data, respectively. Mauchly’s test of sphericity was used to validate the correlation of the repeated measures, and if the assumption of sphericity was violated, the Greenhouse-Geisser correction was applied. To compare age, time with CHIKV and VAS baseline between groups, an unpaired t-test or a Mann-Whitney test were used. The effects of stimulation on VAS were calculated using a mixed ANOVA model, in which the dependent variable was the level of pain, and the independent fixed variables were the treatment (baseline, day 1, day 5 and follow-up), the group of stimulation (active and sham) and the interaction term group vs. time. To determine the difference between groups at each category of time and vice versa it was performed three separate between-subjects ANOVAs. When appropriate, post-hoc comparisons were carried out using Bonferroni correction for multiple comparisons.
For other outcome variables (diary of pain, McGill questionnaire, Pain Inventory, physical tests and SF36), a non-parametric Friedman test was used. Missing data were treated by intention-to-treat analysis, taking into account the method of the last observation carried forward. Partial η2 were calculated as measures of effect size in the ANOVA results (main effects and interaction effects). Partial η2 was used to calculate the effect size, where η2 = 0.01 was considered small, η2 = 0.06 moderate and η2 = 0.14 large effect. Statistical significance was set at p ≤ 0.05.
Results
Twenty patients in the chronic phase of CHIK were randomized into two groups receiving active-tDCS or sham-tDCS with a “M1-SO” assembly. Nineteen individuals completed the intervention period and there was one withdrawal in active-tDCS group. No differences in age (p = 0.969), time with CHIKV (p = 0.806) and VAS baseline (p = 0.257) were found between groups. There were no socio-demographic differences between groups (Table 1).
Active-tDCS simulation of brain current flow (M1-SO) is predicted to produce electric fields across the brain pain neuromatrix regions, including motor cortex, DLPFC, nucleus accumbens and cingulate (Fig. 2B).
(A) High-resolution computational model simulation of brain current flow during tDCS. The “M1-SO” assembly was simulated by sponge with anode positioned vertically over 10–20, location C3, and cathode positioned horizontally on the contralateral-supraorbital, approximately over 10–20, location Fp2. (B) Current flow patterns on a slice (A/m2). The model predicted electric field maps generated in outer cortical regions. Our results show diffuse and clustered brain current flow that includes left motor regions, as well as other regions implicated in treatment of pain and other regions of interest. (in V/m: M1 ipsilateral 0.23, contralateral 0.15, left DLPFC 0.23, nucleus accumbens 0.22, and cingulate 0.25). (C) Consecutive coronal slices and brain current flow.
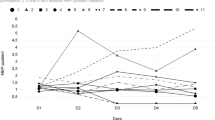
Pain was assessed using VAS at four intervals (baseline, day 1, day 5 and 1 week follow-up). A mixed ANOVA model indicated significant interaction between the group and time on pain evaluated using VAS, F(3.54) = 3.815, p = 0.01, partial η2 = 0.175. Similarly, there were significant main effects of time, F(3.54) = 17.712, p = 0.001, partial η2 = 0.956. It was performed the simple main effect for group and time for testing differences in VAS between groups at each category of the within-subjects factor using three separate one-way ANOVAs. There was no statistically significant simple main effect difference in VAS between groups at the baseline, F(1,18) = 1.894, p = 0.18, partial η2 = 0.09; day 1 F(1,18) = 3.200, p = 0.48, partial η2 = 0.02; day 5 F(1,18) = 1.250, p = 0.65, partial η2 = 0.01 and follow-up F(1,18) = 12.800, p = 0.21, partial η2 = 0.08. There was a statistically significant simple main effect of time on VAS for active-group F(3,27) = 7.300, p = 0.008, partial η2 = 0.44. No significant effect of time was showed for sham-group F(3,27) = 2.019, p = 0.135, partial η2 = 0.18 (Fig. 3A).
(A) Mean visual analog scale (VAS) before treatment (baseline), on days 1 and 5 (during treatment) and follow-up. Error bars denote SEM. (B) Mean McGill score before treatment (baseline), on days 1 and 5 (during treatment) and follow-up. *p < 0.05. (C) Mean daily VAS pain recording. tDCS: describes the five days of treatment. After: denotes the follow-up period. A statistically significant decrease in pain was found only in Active-tDCS (p < 0.05, Friedman).
McGill’s pain questionnaire showed significant effect of Active-tDCS during the intervention, χ2(4) = 18.113, p = 0.001 (Fig. 3B), while there was no effect of Sham-tDCS, χ2(4) = 4.514, p = 0.341. Post hoc analysis in the Active-tDCS group indicated significant differences between baseline vs day 5 (p = 0.001) and follow-up (p = 0.01). Participants recorded daily VAS pain sensation during 21 days (7 days before intervention, during the intervention and 7 days after). Friedman test showed a significant difference only for the Active-tDCS group (p < 0.0001) (Sham-tDCS group p = 0.417) (Fig. 3C). Brief pain inventory showed significant results in intragroup analysis for Active-tDCS in the variables: worst pain of the last 24hs, average of pain, pain at the moment, general activity and normal work, while for sham-tDCS there was no significant difference (Table 2).
Hand grip strength test showed a trend to improve in Active-tDCS group (χ2(3) = 6.704, p = 0.08; Sham-tDCS: χ2(3) = 2.526, p = 0.47). Chair stand, Arm-curl, Chair-sit and reach showed no difference between time and intervention: Active-tDCS (χ2(3) = 4.225, p = 0.23); Sham-tDCS (χ2(3) = 6.148, p = 0.10); Active-tDCS (χ2(3) = 4.014, p = 0.26); Sham-tDCS (χ2(3) = 1.479, p = 0.68); Active-tDCS: (χ2(3) = 6.750, p = 0.08); Sham-tDCS: (χ2(3) = 0.273, p = 0.96), respectively. Back scratch test showed significant difference in the Active-tDCS group (χ2(3) = 15.289, p = 0.002). Baseline back scratch indicated significant difference with day 5 (p = 0.01) and follow-up (p = 0.001). However, Sham-tDCS was also significant (χ2(3) = 11.762, p = 0.008). SF-36 questionnaire showed non-significant difference in both Active-tDCS groups (χ2(3) = 5.055, p = 0.16) and Sham-tDCS (χ2(3) = 3.935, p = 0.26). These tests aimed to evaluate the functionality as function of deficits in resistance, power, mobility and flexibility. These physical fitness tests are simple, quick and economical evaluation, especially in primary care settings and rheumatologic diseases29.
Discussion
Here we showed significant benefits of tDCS on pain sensation following five consecutively days of M1-SO tDCS at 2 mA for 20 min. Our results suggest tDCS could be a critical cost-effective approach for a non-pharmacological strategy to pain relief in a large number of people afflicted by Brazilian CHIK virus epidemic. tDCS has not been previously evaluated to treat CHIK, and the ineffectiveness of the rehabilitation methods to treat the chronic phase of CHIK inspired us to conduct the first clinical trial with tDCS in this population. This preliminary study indicates that several aspects of pain were already ameliorated when M1-SO tDCS was applied for only 5 consecutive days. These results encourage further clinical trials, including long term tDCS treatment (e.g. 10 or 20 sessions) to enhance efficacy. Indeed, CHIK very often results in severe chronic arthralgia and/or arthritis, which can last months to years following the initial infection30,31.
Several studies suggest that chronic pain conditions, such as primary dysmenorrhea, irritable bowel syndrome, interstitial cystitis, fibromyalgia and low back pain, may develop an adaptive neuroplasticity and functional reorganization in pain-related areas32,33. Thus, these areas in the pain neuromatrix (medial prefrontal cortex, posterior cingulate cortex and insula) may exhibit abnormal functional and structural changes32,33. Therefore tDCS can modulate the neuromatrix and provide a significant reduction of pain in patients with chronic pain conditions14. One physiological explanation would be that the tDCS may change maladaptive plasticity both in originally impacted and secondary brain regions in chronic pain syndromes12,15,34. Consistent with this hypothesis, the pain treatment related to CHIK virus in this trial was performed during the chronic phase (after 6 months) and significant improvements were shown for pain and its impacts on daily activities according to BPI. Nonetheless, tDCS effect must be investigated during acute stage, where the maladaptive plasticity might be more susceptible to change, and to increase the benefit. We hypothesize that the association between chronic pain in the CHIK virus and brain reorganization could also exist in others chronic pain syndromes, and tDCS may show similar beneficial results. Mechanism of tDCS action should prevent or revert the ongoing maladaptive plasticity within the pain matrix35.
Moreover, repetitive transcranial magnetic stimulation (rTMS) on M1 was associated with improvement in quality of life and pain in fibromyalgia, chronic pelvic pain and neuropathic pain36,37,38. These evidences support the use of neuromodulation for analgesic effects in chikungunya. While, the M1/SO is the most common tDCS electrode montage for pain11,14, Several meta-analysis suggest an evidence for the effectiveness of anodal M1 stimulation over pain relief34, and others tDCS montage such as dorsolateral prefrontal cortex (DLPFC/SO), primary sensory cortex (S1/SO) or cerebellar (cerebellum/Shoulder) could be used for pain relief34,39.
VAS is the most common method of pain evaluation in clinical trials with tDCS and TMS14, and several adult population, including rheumatic diseases40. Our results showed a reduction over a 30% in VAS, which can be clinically relevant according to Klein at el41. The absence of differences in physical outcomes between the active and sham group could be related to: (1) fitness tests to discriminate specific physical function for CHIK; (2) number of participants (clinical trial power); (3) need to further optimized dose including more sessions. M1-SO tDCS for fibromyalgia showed reduction on pain scores, anxiety, depression and improvement in sleep quality and cognitive performance, but no difference in physical performance14. Therefore, there remains the need of specific physical performance test for CHIK in chronic phase, especially in arthritic symptoms.
According to SF-36 no significant improvement of quality of life was found, and similar studies using the same intervention, but for chronic abdominal pain also showed the same results42. This may reflect the limited number of session and/or the period for participants’ evaluation was insufficient to induce changes in normal routine. In addition, quality of life aspects in CHIK are multifocal and include not only joint pain, but also mental health, emotional role and social functioning5. It can be suggested that future intervention studies with neuromodulation and CHIK should observe not only rheumatic symptoms, but also the social and emotional context of the participants. BPI could be used to evaluate intensity, location and pain effect on the quality of life in patients with CHIK according to Andrade et al.40. Moreover, BPI showed significant improvement in normal work and general activities, suggesting that tDCS could improve the functional status of daily activities.
Marimoutou et al. showed a significant self-reported over morbidity and impaired quality of life persisting in CHIK patients for 6 years after infection5, while in our study the average was 10 months of CHIK infection. Furthermore, chronic CHIK arthralgia has a large psychological impact, frequent depressive moods and social disabilities5. These clinical findings influence quality of life and functionality. The M1-SO tDCS used in this trial is note the best assemble to treat depression and others montages should be tested.
In countries that have already suffered large CHIK outbreaks, attention is focused on the long-term burden of persistent arthralgia5. Understanding new rehabilitation approaches will greatly contribute to future decision-making on appropriate allocation of resources for public health care and research.
Our results provide evidence that tDCS reduces the pain levels with clinically significant changes for patients with the CHIK virus. Future investigations involving additional numbers of sessions of tDCS in CHIK patients could prove more effective not only in pain relief, but also in cognitive function, physical activity assessment, quality of life and general functionality. The tDCS treatment could be useful, inexpensive and attractive to areas lacking resources and with a high number of patients with CHIK symptoms. This study brings a novel approach to the non-pharmacological treatment for CHIK fever with tDCS.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Silva, L. A. & Dermody, T. S. Chikungunya virus: Epidemiology, replication, disease mechanisms, and prospective intervention strategies. J Clin Invest 127, 737–749, https://doi.org/10.1172/JCI84417 (2017).
Morrison, T. E. Reemergence of Chikungunya Virus. J Virol 88, 11644–47, https://doi.org/10.1128/JVI.01432-14 (2014).
Madariaga, M., Ticona, E. & Resurrecion, C. Chikungunya: Bending over the Americas and the rest of the world. Brazilian J Infect Dis 20, 91–98, https://doi.org/10.1016/j.bjid.2015.10.004 (2016).
PAHO. Panamerican Health Organization. http://www.paho.org/usa/.
Marimoutou, C., Ferraro, J., Javelle, E., Deparis, X. & Simon, F. Chikungunya infection: self-reported rheumatic morbidity and impaired quality of life persist 6 years later. Clin Microbiol Infect 21, 688–693, https://doi.org/10.1016/j.cmi.2015.02.024 (2015).
Sam, I. C. et al. Updates on Chikungunya Epidemiology, Clinical Disease, and Diagnostics. Vector-Borne Zoonotic Dis 15, 223–230, https://doi.org/10.1089/vbz.2014.1680 (2015).
Chopra, A., Saluja, M. & Venugopalan, A. Effectiveness of chloroquine and inflammatory cytokine response in patients with early persistent musculoskeletal pain and arthritis following chikungunya virus infection. Arthritis Rheumatol 66, 319–326, https://doi.org/10.1002/art.38221 (2014).
Kelvin, A. et al. Inflammatory cytokine expression is associated with Chikungunya virus resolution and symptom severity. PLoS Negl Trop Dis, 5, https://doi.org/10.1371/journal.pntd.0001279 (2011).
Hashmi, J. A. et al. Shape shifting pain: Chronification of back pain shifts brain representation from nociceptive to emotional circuits. Brain 136, 2751–2768, https://doi.org/10.1093/brain/awt211 (2013).
Boggio, P. S., Zaghi, S. & Fregni, F. Modulation of emotions associated with images of human pain using anodal transcranial direct current stimulation (tDCS). Neuropsychologia 47, 212–217, https://doi.org/10.1016/j.neuropsychologia.2008.07.022 (2009).
Luedtke, K. et al. Transcranial direct current stimulation for the reduction of clinical and experimentally induced pain: a systematic review and meta-analysis. Clin J Pain 28, 452–461, https://doi.org/10.1097/AJP.0b013e31823853e3 (2012).
Naro, A. et al. Non-invasive Brain Stimulation, a Tool to Revert Maladaptive Plasticity in Neuropathic Pain. Front Hum Neurosci 10, 1–9, https://doi.org/10.3389/fnhum.2016.00376 (2016).
Jensen, M. P., Day, M. A. & Miró, J. Neuromodulatory treatments for chronic pain: Efficacy and mechanisms. Nat Rev Neurol 10, 167–178, https://doi.org/10.1038/nrneurol.2014.12 (2014).
Lefaucheur, J. P. A comprehensive database of published tDCS clinical trials (2005—2016). Neurophysiol Clin 46, 319–398, https://doi.org/10.1016/j.neucli.2016.10.002 (2016).
Lefaucheur, J. P. et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol 128, 56–92, https://doi.org/10.1016/j.clinph.2016.10.087 (2017).
Yi, M. & Zhang, H. Nociceptive Memory in the Brain: Cortical Mechanisms of Chronic Pain. J Neurosci 31, 13343–13345, https://doi.org/10.1523/JNEUROSCI.3279-11.2011 (2011).
Chang-E, Z. et al. Effectiveness and safety of transcranial direct current stimulation. J Rehabili Med 49, 2–9, https://doi.org/10.2340/16501977-2179 (2017).
Ahn, H. et al. Efficacy of transcranial direct current stimulation over primary motor cortex (anode) and contralateral supraorbital area (cathode) on clinical pain severity and mobility performance in persons with knee osteoarthritis: An experimenter- and participant-bl. Brain Stimul 10, 902–909, https://doi.org/10.1016/j.brs.2017.05.007 (2017).
Bikson, M. et al. Safety of transcranial Direct Current Stimulation: Evidence Based Update 2016. Brain Stimul 9, 641–661, https://doi.org/10.1016/j.brs.2016.06.004.Safety (2017).
Schulz, K. F., Altman, D. G. & Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. J Clin Epidemiol 63, 834–840, https://doi.org/10.1016/j.jclinepi.2010.02.005 (2010).
Antal, A., Terney, D., Kühnl, S. & Paulus, W. Anodal Transcranial Direct Current Stimulation of the Motor Cortex Ameliorates Chronic Pain and Reduces Short Intracortical Inhibition. J Pain Symptom Manage 39, 890–903, https://doi.org/10.1016/j.jpainsymman.2009.09.023 (2010).
Fregni, F. et al. A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum 54, 3988–3998, https://doi.org/10.1002/art.22195 (2006).
Hawker, G. A., Mian, S., Kendzerska, T. & French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale SF. Arthritis Care Res 63, 240–252, https://doi.org/10.1002/acr.20543 (2011).
Ferreira, K. A., Teixeira, M. J., Mendonza, T. R. & Cleeland, C. S. Validation of brief pain inventory to Brazilian patients with pain. Support Care Cancer 19, 505–511, https://doi.org/10.1007/s00520-010-0844-7 (2011).
Bohannon, R. W. Muscle strength: Clinical and prognostic value of hand-grip dynamometry. Curr Opin Clin Nutr Metab Care 18, 465–470, https://doi.org/10.1097/MCO.0000000000000202 (2015).
Segal, N. A., Boyer, E. R., Wallace, R., Torner, J. C. & Yack, H. J. Association between chair stand strategy and mobility limitations in older adults with symptomatic knee osteoarthritis. Arch Phys Med Rehabil 94, 375–383, https://doi.org/10.1016/j.apmr.2012.09.026 (2013).
Rikli, R. E. & Jones, C. J. Development and validation of criterion-referenced clinically relevant fitness standards for maintaining physical independence in later years. Gerontologist 53, 255–267, https://doi.org/10.1093/geront/gns071 (2013).
Campolina, A. G., Bortoluzzo, A. B., Ferraz, M. B. & Ciconelli, R. M. Validation of the Brazilian version of the generic six-dimensional short form quality of life questionnaire (SF-6D Brazil). Cien Saude Colet 16, 3103–3110, https://doi.org/10.1590/S1413-81232011000800010 (2011).
Aparicio, V. A. et al. Fitness testing as a discriminative tool for the diagnosis and monitoring of fibromyalgia. Scand J Med Sci Sport 23, 415–423, https://doi.org/10.1111/j.1600-0838.2011.01401.x (2013).
Goupil, B. A. & Mores, C. N. A Review of Chikungunya Virus-induced Arthralgia: Clinical Manifestations, Therapeutics, and Pathogenesis. Open Rheumatol J 10, 129–140, https://doi.org/10.2174/1874312901610010129 (2016).
Hawman, D. W. et al. Chronic Joint Disease Caused by Persistent Chikungunya Virus Infection Is Controlled by the Adaptive Immune Response. J Virol 87, 13878–13888, https://doi.org/10.1128/JVI.02666-13 (2013).
Wu, T. H. et al. Dynamic Changes of Functional Pain Connectome in Women with Primary Dysmenorrhea. Sci Rep 6, 24543, https://doi.org/10.1038/srep24543 (2016).
Kregel, J. et al. Structural and functional brain abnormalities in chronic low back pain: A systematic review. Semin Arthritis Rheum 45, 229–237, https://doi.org/10.1016/j.semarthrit.2015.05.002 (2015).
Vaseghi., B., Zoghi, M. & Jaberzadeh, S. Does anodal transcranial direct current stimulation modulate sensory perception and pain? A meta-analysis study. Clin Neurophysiol 125, 1847–1858, https://doi.org/10.1016/j.clinph.2014.01.020 (2014).
Brietzke, A. P. et al. Neuroplastic effects of transcranial direct current stimulation on painful symptoms reduction in chronic hepatitis C: A phase II randomized, double blind, sham controlled trial. Front Neurosci 9, 1–9, https://doi.org/10.3389/fnins.2015.00498 (2016).
Hsu, J. H., Daskalakis, Z. J. & Blumberger, D. M. An Update on Repetitive Transcranial Magnetic Stimulation for the Treatment of Co-morbid Pain and Depressive Symptoms. Curr Pain Headache Rep 22, 51, https://doi.org/10.1007/s11916-018-0703-7 (2018).
Cervigni, M. et al. Repetitive transcranial magnetic stimulation for chronic neuropathic pain in patients with bladder pain syndrome/interstitial cystitis. Neurourol Urodyn 24, https://doi.org/10.1002/nau.23718 (2018).
Knijnik, L. M. et al. Repetitive Transcranial Magnetic Stimulation for Fibromyalgia: Systematic Review and Meta-Analysis. Pain Pract 16, 294–304, https://doi.org/10.1111/papr.12276 (2016).
Bocci, T. et al. Cerebellar direct current stimulation modulates pain perception in humans. Restor Neurol Neurosci 33, 597–609, https://doi.org/10.3233/RNN-140453 (2015).
de Andrade, D. C., Jean, S., Clavelou, P., Dallel, R. & Bouhassira, D. Chronic pain associated with the Chikungunya Fever: long lasting burden of an acute illness. BMC Infect Dis 10, 31, https://doi.org/10.1186/1471-2334-10-31 (2010).
Klein, M. M. et al. Transcranial magnetic stimulation of the brain: Guidelines for pain treatment research. Pain 156, 1601–1614, https://doi.org/10.1097/j.pain.0000000000000210 (2015).
Volz, M. S., Farmer, A. & Siegmund, B. Reduction of chronic abdominal pain in patients with inflammatory bowel disease through transcranial direct current stimulation: a randomized controlled trial. Pain 157, 429–437, https://doi.org/10.1097/j.pain.0000000000000386 (2016).
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) Finance Code 001.
Author information
Authors and Affiliations
Contributions
J.A. and E.C. Conception and design, recruited the patients and evaluated before and after stimulation. ESilva-Filho: Conception and design, recruited the patients, performed the tDCS, wrote the manuscript and submitted the project to clinicaltrials.gov. A.H.O. and E.M. analysis and interpretation of the data, and revise it critically for important intellectual content. G.U. and M.B. cooperation on analysis and the interpretation of the data, revise it critically, prepare the high-resolution computational model analyses. R.P. conception and design of the work, submitted the project to the local institutional Ethical Committee, data analysis, interpretation and draft the work. Final approval of the article: Edson Silva-Filho, Alexandre H. Okano, Edgard Morya, Jessica Albuquerque, E. Cacho, Gozde Unal, Marom Bikson, Rodrigo Pegado.
Corresponding author
Ethics declarations
Competing Interests
The City University of New York has patent on brain stimulation with MB as the inventor. MB has equity in Soterix Medical Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Silva-Filho, E., Okano, A.H., Morya, E. et al. Neuromodulation treats Chikungunya arthralgia: a randomized controlled trial. Sci Rep 8, 16010 (2018). https://doi.org/10.1038/s41598-018-34514-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34514-4
Keywords
This article is cited by
-
Sedentary behavior, increasing age, and overweight/obesity increase the presence and intensity of the chronic joint pain in individuals affected by Chikungunya fever
Clinical Rheumatology (2024)
-
Contributions of musculoskeletal rehabilitation in patients after chikungunya fever: a systematic review
BMC Musculoskeletal Disorders (2023)
-
Chikungunya fever
Nature Reviews Disease Primers (2023)
-
Transcranial direct current stimulation improves quality of life and physical fitness in diabetic polyneuropathy: a pilot double blind randomized controlled trial
Journal of Diabetes & Metabolic Disorders (2020)
-
Beyond the target area: an integrative view of tDCS-induced motor cortex modulation in patients and athletes
Journal of NeuroEngineering and Rehabilitation (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.