Abstract
This study investigates the replacement of vegetable oil (VO) in aquaculture feed for Arctic char (Salvelinus alpinus) with oil produced by the oleaginous yeast Lipomyces starkeyi grown in lignocellulose (wheat straw) hydrolysate. VO is extensively used to partially replace fish oil in aquaculture feed, which can be seen as non-sustainable. VO itself is becoming a limited resource. Plant oils are used in many different applications, including food, feed and biodiesel. Its replacement in non-food applications is desirable. For this purpose, yeast cells containing 43% lipids per g dry weight were mechanically disrupted and incorporated into the fish feed. There were no significant differences in this pilot study, regarding weight and length gain, feed conversion ratio, specific growth rate, condition factor and hepatosomatic index between the control and the yeast oil fed group. Fatty and amino acid composition of diet from both groups was comparable. Our results in fish demonstrate that it is possible to replace VO by yeast oil produced from lignocellulose, which may broaden the range of raw materials for food production and add value to residual products of agriculture and forestry.
Similar content being viewed by others
Introduction
Fish is one of the most traded food commodities and has great potential to contribute to food security for a growing population1. Fish is already the major source of protein in many cultures2. Moreover, it is also the major resource of n-3 long chain polyunsaturated fatty acids (LCPUFA)3. Hence, aquaculture is a rapidly growing industry and is an important source of animal-based foods. This growth generates an increased demand for feed for farmed fish. Currently, fish meal and fish oil are still the primary resources to meet the demand for protein and lipids of farmed fish2. Aquaculture consumes about 70% of the globally produced fish oil (FO), and 90% of this oil is derived from reduction fisheries3. Thus, a sustainable further expansion of the aquaculture industry can only happen when alternative resources/replacements for FO can be found. Those alternatives can be both vegetable oil (VO) and terrestrial animal oil3,4,5,6. Although both VOs and animal fat do not provide a good supply of n-3 long chain polyunsaturated FA (LCPUFA), they are metabolised by the fish in beta-oxidation, to provide energy. It has been shown that FO can be replaced by VO or animal fat without negatively impacting fish health or growth4,5,6,7,8. In Europe, VO is the most common partial substitute for fish oil, whereas in other parts of the world, terrestrial animal fats are also incorporated into aquafeeds. Nevertheless, VOs have a broad range of applications, including direct food production and biodiesel production. Especially with a view towards the latter, discussions about the sustainability of VO production have been raised. Finding alternatives to VO may lessen the push towards monocultures, with risk for land use changes and rainforest cutting, and in general, reduce the food carbon print of aquaculture9,10,11,12,13.
Microbial oils or single cell oils have been regarded as a potential replacement for VO in biodiesel production, and in some cases even for food purposes. Oleaginous yeasts, i.e. yeasts that can accumulate 20% and more of their biomass as lipids, can form single cell oils from a variety of low value substrates, including lignocellulose hydrolysate14,15,16.
While there are a number of reports on utilising yeasts as a protein source in fish feed (e.g.17,18), only little is known about utilising yeast-derived oil in fish feed. Several oleaginous yeast species can utilise lignocellulosic hydrolysates and convert them to lipids. We have recently demonstrated that the oleaginous yeast Lipomyces starkeyi can efficiently synthesise lipids from the hemicellulose fraction of birch wood and the cellulose fraction of wheat straw19,20, and other studies have also used lignocellulose hydrolysate as a substrate for oil production with this yeast21,22,23,24. The lipid composition of L. starkeyi was shown to be similar to that of saturated fatty acids (SFA) rich VO, for example olive oil or palm oil19,20,25. In this pilot study, the aim was to test whether it is possible to replace VO in the feed for Arctic char (Salvelinus alpinus) with single cell oils derived from L. starkeyi grown on lignocellulose hydrolysate from wheat straw, i.e. a non-edible, residual material.
Results
Hydrolysate analysis
The cellulosic hydrolysate from wheat straw (i.e. the enzymatically hydrolysed solid phase after steam explosion (see method part)) contained glucose 87.3 g/l, xylose 22.2 g/l and acetic acid 3.8 g/l. Due to the high acetic acid concentration we started the fermentation with 50% hydrolysate and then pumped in 100% hydrolysate in the feeding phase of the cultivation.
Yeast cultivation
At harvest, L. starkeyi cells had consumed all carbon sources, and the total yeast dry weight of 575 g (cells from four fermentors) was produced from a total amount of 23.2 l hydrolysate, i.e. 2628.6 g carbon sources (glucose, xylose, and acetic acid). The final intracellular lipid content of the yeast was determined to be 43 ± 0.8%, thus the total amount of yeast lipids produced was 247.25 g, corresponding to a lipid yield of 0.09. Yeast growth and carbon source consumption are illustrated in Supplementary Fig. S1. Cells were disrupted by French press, as described in Methods; successful disruption was confirmed by microscopic inspection of the cell lysate. No further oil extraction was performed, to avoid contamination with toxic solvents and to retain the yeast proteins and polysaccharides in the hydrolysate.
Fish performance
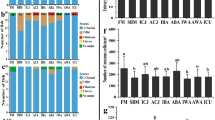
Fish were fed with a standard experimental diet26 (see Methods) containing standard ingredients and either VO and casein (control diet) or disrupted L. starkeyi cells instead of VO and casein. The fatty acid composition of the feeds is shown in Table 1. In both feeds, the main source of amino acids was fish meal. Accordingly, the amino acid profiles of the control- and yeast-based feeds did not differ significantly (Table 2). Initial and final weight and length, liver weight and the calculated performance factors are presented in Table 3. Initial weight of fishes was 148.2 g (control) and 149.8 g (yeast oil feeding), and the final weight was 265 g in both cases. There were no significant differences between the control and yeast fed fish regarding feed conversion rate (FCR), specific growth rate (SGR), condition factor (CF) and hepatosomatic index (HSI), indicating that both feeds were metabolised in a similar way and the addition of yeast in the feed did not negatively impact growth.
There was a large standard deviation of the individual fish weight, both in the yeast-fed treatment and the control towards the end of the experiment. This effect was most likely due to the small number of fish, which enabled a few dominant individuals to consume a major proportion of the provided feed, at the costs of other, minor individuals, which hardly showed any growth. The number of fishes was adjusted to the size of the 3 tanks and 2 months feeding to ensure appropriate water parameters such as NH4+ and oxygen tension when fish biomass increases. However, the total mass of the fish did not significantly differ, in spite of the dominant individuals. Consequently, sampling was carried out on fishes representing all sizes from all units. This growth effect does not hinder the evaluation of the fatty acid composition of the tissue samples, as the individual fish reflected the feed fatty acid profile, which is a common result in experiments performed on salmonids.
Lipid content and fatty acid composition
The total weight (whole fish), fat content and fatty acid profile of the muscle tissue of six yeast fed fishes and six control fed fishes are shown in Table 4 (two from each tank).
Overall, no significant differences between the two different feeds were observed, except for linoleic acid (C18:2 n-6) where the fish fed with control feed had slightly higher levels compared to yeast fed fish.
Discussion
In this study, we investigated whether it is possible to replace VO with oil produced by an oleaginous yeast, L. starkeyi, grown on lignocellulose (wheat straw) hydrolysate. Inclusion of yeast oil into fish feed has been tested previously but in the context of replacing fish oil in the feed, using oil from genetically engineered Yarrowia lipolytica cultivated on first generation substrate (glucose)27.
Our study shows that it is possible to convert second generation substrate (lignocellulose) to a feed component, enabling the replacement of feed oil (mostly VO) as an energy source in aquaculture. VOs are listed among the products causing the largest environmental impacts. They are also regarded as the fastest growing food commodities worldwide28. Some vegetable oils have a high greenhouse gas potential associated with their production: for instance palm- and soybean oil are estimated to emit more than 2000 kg CO2 equivalents per ton produced, and considerable areas of arable land are used for producing vegetable oils29. Since biodiesel is also produced from vegetable oils, their consumption in the EU greatly exceeds local production, and thus, a major proportion of the utilised plant oil has to be imported30. There are reports of rainforest clearing due to palm- and soya oil production and there are moves in the EU to reduce the use of imported vegetable oils, especially palm oil (http://www.europarl.europa.eu/sides/getDoc.do?pubRef=-//EP//TEXT+REPORT+A8-2017-0066+0+DOC+XML+V0//EN).
The yeast cells contain, apart from oil, also proteins and other components that can be utilised by the fish. The first implication of this is that the yeast cells contributed to protein biomass in the feed; this was adjusted by removing the casein from the yeast feed, whereas it was the standard protein additive in the control feed, as commonly used in other fish feeding trials26. The second implication is that it was not necessary to extract the oil from the mechanically disrupted yeast cells. This is advantageous compared to for instance microbial biodiesel production, where extraction is regarded as one of the most crucial steps in obtaining a sustainable process31. Analyses of growth parameters and composition of the final fish demonstrated that there was no negative impact of replacing VO and casein by L. starkeyi-biomass. The amino acid profile of the yeast-based feed did not change compared to the control. There was a slight but significant decrease in the total amount of n-6 fatty acids in the yeast fed fish. A low n-6/n-3 ratio is advantageous, as in most modern diets this ratio is too high, leading to a variety of diseases32. Our experiment demonstrates that it is possible to replace terrestrial plant- and animal based lipid and protein sources by yeast biomass. The fatty acid composition of yeast strains varies with both strain and cultivation conditions19,20,33. Selecting appropriate yeast strains and culture conditions may thus represent a possibility to positively influence the fatty acid composition and thereby the n-6/n-3 ratio.
From the lignocellulose substrate, 0.09 g lipids were produced per g consumed carbon source. This is within the range of values previously reported in similar cultivations19,20,34. In this study, yeast cultivation was performed to generate biomass for the fish trial; optimisation of the yeast fermentation conditions was not within the scope of the study. Nevertheless, rapid and efficient lipid production from the substrate can greatly improve the overall energy output and greenhouse gas impacts of any single cell oil production process31,35, and therefore, optimisation of fermentation conditions and strains for lipid production is one of the major topics of our ongoing research.
This pilot study, to the best of our knowledge, investigates for the first time the utilisation of lignocellulose-derived yeast oil in fish feed. The results demonstrate that it is possible to completely replace VO and partially replace protein (casein in the control feed, in the present study) with the yeast biomass, without any significant effects on fish growth and final quality. Previous studies have shown that there is a limit to including yeast-based protein into fish diets17,27. On the other hand, there are also studies indicating a positive effect of yeast cell wall β-glucan on the immune system of fish36 and a barrier function of yeasts against prions37. Moreover, utilising different yeast strains and different lignocellulose substrates may have some impact on the final quality of the fish. All these possible effects require further investigation and will be the subject of future studies.
Methods
Strains and media
L. starkeyi CBS 1807 (Centraalbureau vor Schimmelcultures, Utrecht, The Netherlands) was maintained on YM-agar plates (glucose 10 g/l, yeast extract 3 g/l, peptone 5 g/l, malt extract 3 g/l, Agar 16 g/l). The pre-culture medium was YPD (glucose 20 g/l, yeast extract 10 g/l, peptone 20 g/l).
Preparation of hydrolysate
The steam explosion and enzymatic hydrolysis was performed at the Department of Chemical Engineering, Lund University, Sweden. Wheat straw was soaked with 1% acetic acid over night, and fluid removed by pressing. The acid soaked biomass was then steam exploded at 190 °C for 10 min in a 10 L steam pretreatment reactor. The liquid fraction (mainly hemicellulose) was separated from the solid fraction and the latter was enzymatically hydrolysed. The hydrolysis was performed at 45 °C and pH 4.8. Cellic CTec3 enzyme cocktail (Novozyme A/S, Bagsværd, Denmark) was added at 10 FPU/g substrate. After hydrolysis, the suspension was centrifuged to separate the solid residues (mainly lignin) and repeatedly filtered, using filters with decreasing pore size in each step. The last filtration step was performed with a 0.45 μm sterile filter. The sugar and acetic acid concentration was determined by HPLC as described previously19.
Pre cultures
Before inoculation in fermentors, L. starkeyi was cultivated in two steps with increasing medium volumes. For the first pre-culture, a loopful of L. starkeyi cells was inoculated from a YM-agar plate into 100 ml YPD-medium in 500 ml baffled shake flasks and incubated at 25 °C and 150 rpm in a rotatory shaker. After 48 h, the 100 ml culture was transferred to 400 ml YPD medium in a 3 l shake flask and incubated at 25 °C and 150 rpm for 72 h. The cells were harvested by centrifugation (4000 g, 10 min) and washed twice with saline solution (NaCl, 9 g/l). After washing, the pellet was resuspended in 50 ml saline and inoculated into the fermentor.
Fed-batch cultivation in fermentors
L. starkeyi was cultivated in four Dolly fermentors (Belach Bioteknik, Stockholm, Sweden, working volume 8 l) at 25 °C. A volume of 1.5 l of sterile filtered cellulose hydrolysate was added to each fermentor containing 1.5 l sterile deionised water, representing a starting volume of 3 l comprised of 50% cellulose hydrolysate. The pH was set at 5 and automatically controlled by addition of NaOH (25% w/w) or 3 M H3PO4. The aeration was initially 1 l/min; during the experiment it was continuously increased up to 5 l/min. The dissolved oxygen tension (pO2) was controlled by a DO-electrode, set to 20% and maintained by changing the stirring speed. One ml of polypropylene glycol 2000 (Alfa Aesar, Karlsruhe, Germany) was added to prevent foaming. L. starkeyi was first cultivated in a batch phase for 48 h, then the fed-batch phase started with pumping cellulosic hydrolysate at a speed of approx 24 ml/h in 7.5 days, i.e. 4.3 l of hydrolysate was added to each fermenter during the feeding phase; the total amount of hydrolysate was thus 5.8 l per fermenter.
L. starkeyi harvesting
Cells were centrifuged at 5400 g for 10 min, washed with deionised water and then disrupted in a French press (Constant systems LTD, Daventry, UK) at 40 psi. Dry weight of the disrupted cells was determined by drying a portion of the cell-lysate in a Precisa xm 60 oven (Precisa Instruments LTD, Dietikon, Switzerland) and the disrupted cells were stored at −20 °C until incorporation into the fish feed18.
Fish feed preparation
The composition of the fish feed is shown in Table 5 26. The ingredients were mixed by hand to a homogeneous consistency and pressed through a kitchen meat grinder. The feed was dried at room temperature for 48 h before vacuum packing into air tight plastic bags. Total amino acids were quantified in the prepared feeds (Eurofins Food & Feed Testing Sweden AB, Lidköping, Sweden, Method: SS-EN ISO 13903:2005).
Feeding trial
The experiment was carried out in accordance with EU legislation (i.e., Directive 2010/63/EU), and received the approval of the Ethical Committee for Animal Experiments in Umeå, Sweden.
Arctic char was kept in flow through system with natural photo period at Kälarne Aquaculture North, Sweden. The water temperature was ambient, approx 12 °C and the water system was always fully aerated from the inlet. Inlet water quality was always assured. The tanks were 1 × 1 m and water depth 20 cm. Tanks were randomly assigned to the two diets with randomly selected fish (n = 8/tank). Prior to the trial all fish were fed a commercial diet suitable for Arctic char juveniles. The feed was distributed by band feeders 4 times a day26. Fish was anaestesised before handling38.
The Arctic char were measured for weight and length and then divided into six tanks (n = 8): three were fed with the control feed and three with yeast feed. Feeding ratio was 2% of the actual biomass in the tanks.
After 2 months, the fish were weighed and measured again after a 24 h starvation period and liver weight was registered. After filleting, the muscle tissue was frozen on dry ice and then stored at −80 °C until lipid extraction.
Fish performance
Based on the measurements and the consumed feed, feed conversion ratio (FCR), specific growth rate (SGR), condition factor (CF) and hepatosomatic index (HSI) were calculated as follows:
where Wt = final weight of fish in g; W0 = initial weight of fish in g; F = amount of dry feed fed in g; t = time (days); TL = total length in cm, Wl = weight of liver in g; Tw = total weight of fish without liver in g.
Lipid extraction from yeast, feed and fish
The lipid content of yeast cells was determined as previously described19,20. For the fish and feed, total lipid analysis was performed according to Pettersson, et al.26. Lipids were extracted from six muscle samples from each treatment (sourced from two fish from each replicate) and from the feeds. A subsample of 1 g of fish feed or muscle (light and dark) of individual Arctic char was used for lipid extraction (in duplicate). The sample was homogenised in hexane:isopropanol (HIP; 3:2, v-v) with an Ultra-Turrax (Janke and Kunkel, IKA Werke, Staufen, Germany). For lipid and non-lipid phase separation, 6.67% of Na2SO4 was added to the homogenate and it was centrifuged. After gravimetrical identification of the total lipid content from dried samples, the lipids were stored in hexane at −80 °C for further analysis. All chemicals and solvents (reagent grade) were purchased from Merck (Darmstadt, Germany) except chloroform (Sigma Chemicals Co. St. Louis, MO, USA). The solvents were used without further purification.
Determination of fatty acid profiles
Fatty acid methyl esters (FAME) from total lipids in muscle and feeds were prepared with BF3 methanol according to the method described by Appelqvist39. FAME were stored in hexane at −80 °C for further analysis.
FAME were analysed by GC using a CP 3800 instrument (Varian AB, Stockholm, Sweden) equipped with a flame ionization detector and a split injector, and separated on a 50 m fused silica capillary column BPX 70 (SGE, Austin, Tex) (0.22 mm i.d × 0.25 µm film thickness)40. The injector temperature was 230 °C and the detector temperature 250 °C. Helium was the carrier gas, at a flow rate of 0.8 mL/min, and nitrogen was used as make-up gas. Peaks were identified by comparing their retention times with those of the standard mixture GLC 68A (Nu-check Prep, Elysian, USA) and quantified using an internal standard (methyl-15-methylheptadecanoate; Larodan Fine Chemicals AB, Malmö, Sweden). Peak areas were integrated using Galaxie chromatography data system software version 1.9 (Varian AB, Stockholm, Sweden).
Statistics and calculations
Mean values, standard deviations and FA percentages were calculated in Excel and statistical analyses were performed using the Statistica CZ 12 software package. One-way analysis of variance (ANOVA) and Tukey’s HSD test were performed to characterise the differences between control and experimental group. The performance factors data were treated by One-way ANOVA in Excel.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author upon request.
References
FAO. The state of world fisheries and aquaculture, contributing to food security and nutrition for all. (Rome, 2016).
Tacon, A. G. J., Hasan, M. R. & Metian, M. Vol. No. 564 (FAO Fisheries and Aquaculture Technical paper No 564, Rome, 2011).
Tocher, D. R. Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 449, 94–107, https://doi.org/10.1016/j.aquaculture.2015.01.010 (2015).
Emery, J. A., Smullen, R. P. & Turchini, G. M. Tallow in Atlantic salmon feed. Aquaculture 422–423, 98–108, https://doi.org/10.1016/j.aquaculture.2013.12.004 (2014).
Hatlen, B. et al. Growth, feed utilization and endocrine responses in Atlantic salmon (Salmo salar) fed diets added poultry by-product meal and blood meal in combination with poultry oil. Aquaculture Nutrition 21, 714–725, https://doi.org/10.1111/anu.12194 (2015).
Salini, M. et al. Marginal efficiencies of long chain-polyunsaturated fatty acid use by barramundi (Lates calcarifer) when fed diets with varying blends of fish oil and poultry fat. Aquaculture 449, 48–57, https://doi.org/10.1016/j.aquaculture.2015.02.027 (2015).
Bell, J. G., Henderson, R. J., Tocher, D. R. & Sargent, J. R. Replacement of dietary fish oil with increasing levels of linseed oil: Modification of flesh fatty acid compositions in Atlantic salmon (Salmo salar) using a fish oil finishing diet. Lipids 39, 223–232, https://doi.org/10.1007/s11745-004-1223-5 (2004).
Sprague, M., Dick, J. R. & Tocher, D. R. Impact of sustainable feeds on omega-3 long-chain fatty acid levels in farmed Atlantic salmon, 2006–2015. Scientific Reports 6, 21892, https://doi.org/10.1038/srep21892 https://www.nature.com/articles/srep21892#supplementary-information (2016).
Azócar, L., Ciudad, G., Heipieper, H. J. & Navia, R. Biotechnological processes for biodiesel production using alternative oils. Appl Microbiol Biotechnol 88, 621–636, https://doi.org/10.1007/s00253-010-2804-z (2010).
Escobar, J. C. et al. Biofuels: Environment, technology and food security. Renewable and Sustainable Energy Reviews 13, 1275–1287, https://doi.org/10.1016/j.rser.2008.08.014 (2009).
Pinzi, S., Leiva, D., López-García, I., Redel-Macías, M. D. & Dorado, M. P. Latest trends in feedstocks for biodiesel production. Biofuels, Bioproducts and Biorefining 8, 126–143, https://doi.org/10.1002/bbb.1435 (2014).
Xu, Z., Sun, D.-W., Zeng, X.-A., Liu, D. & Pu, H. Research developments in methods to reduce the carbon footprint of the food system: A review. Critical Reviews in Food Science and Nutrition 55, 1270–1286, https://doi.org/10.1080/10408398.2013.821593 (2015).
Margono, B. A., Potapov, P. V., Turubanova, S., Stolle, F. & Hansen, M. C. Primary forest cover loss in Indonesia over 2000-2012. Nat Climate Change 4, 730–735, https://doi.org/10.1038/nclimate2277 (2014).
Ochsenreither, K., Glück, C., Stressler, T., Fischer, L. & Syldatk, C. Production strategies and applications of microbial single cell oils. Frontiers in Microbiology 7, 1539, https://doi.org/10.3389/fmicb.2016.01539 (2016).
Passoth, V. In Biotechnology of Yeasts and Filamentous Fungi (ed. Andriy A. Sibirny) 149–204 (Springer International Publishing, 2017).
Sitepu, I. R. et al. Oleaginous yeasts for biodiesel: Current and future trends in biology and production. Biotechnology Advances 32, 1336–1360, https://doi.org/10.1016/j.biotechadv.2014.08.003 (2014).
Huyben, D. et al. Effects of dietary inclusion of the yeasts Saccharomyces cerevisiae and Wickerhamomyces anomalus on gut microbiota of rainbow trout. Aquaculture 473, 528–537, https://doi.org/10.1016/j.aquaculture.2017.03.024 (2017).
Nasseri, A. T., Rasoul-Amini, S., Morowvat, M. H. & Ghasemi, Y. Single cell protein: Production and process. American Journal of Food Technology 6, 103–116, https://doi.org/10.3923/ajft.2011.103.116 (2011).
Brandenburg, J. et al. Lipid production from hemicellulose with Lipomyces starkeyi in a pH regulated fed-batch cultivation. Yeast 33, 451–462, https://doi.org/10.1002/yea.3160 (2016).
Brandenburg, J. et al. Bioethanol and lipid production from the enzymatic hydrolysate of wheat straw after furfural extraction. Appl Microbiol Biotechnol 102, 6269–6277, https://doi.org/10.1007/s00253-018-9081-7 (2018).
Angerbauer, C., Siebenhofer, M., Mittelbach, M. & Guebitz, G. M. Conversion of sewage sludge into lipids by Lipomyces starkeyi for biodiesel production. Bioresource Technology 99, 3051–3056, https://doi.org/10.1016/j.biortech.2007.06.045 (2008).
Calvey, C. H., Su, Y.-K., Willis, L. B., McGee, M. & Jeffries, T. W. Nitrogen limitation, oxygen limitation, and lipid accumulation in Lipomyces starkeyi. Bioresource Technology 200, 780–788, https://doi.org/10.1016/j.biortech.2015.10.104 (2016).
Huang, C. et al. Bioconversion of corncob acid hydrolysate into microbial oil by the oleaginous yeast Lipomyces starkeyi. Applied biochemistry and biotechnology 172, 2197–2204, https://doi.org/10.1007/s12010-013-0651-y (2014).
Yu, X., Zheng, Y., Dorgan, K. M. & Chen, S. Oil production by oleaginous yeasts using the hydrolysate from pretreatment of wheat straw with dilute sulfuric acid. Bioresour Technol 102, 6134–6140, https://doi.org/10.1016/j.biortech.2011.02.081 (2011).
Dubois, V., Breton, S., Linder, M., Fanni, J. & Parmentier, M. Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. European Journal of Lipid Science and Technology 109, 710–732, https://doi.org/10.1002/ejlt.200700040 (2007).
Pettersson, A., Johnsson, L., Brännäs, E. & Pickova, J. Effects of rapeseed oil replacement in fish feed on lipid composition and self-selection by rainbow trout (Oncorhynchus mykiss). Aquaculture Nutrition 15, 577–586, https://doi.org/10.1111/j.1365-2095.2008.00625.x (2009).
Hatlen, B., Berge, G. M., Odom, J. M., Mundheim, H. & Ruyter, B. Growth performance, feed utilisation and fatty acid deposition in Atlantic salmon, Salmo salar L., fed graded levels of high-lipid/high-EPA Yarrowia lipolytica biomass. Aquaculture 364, 39–47, https://doi.org/10.1016/j.aquaculture.2012.07.005 (2012).
Khatri, P. & Jain, S. Environmental life cycle assessment of edible oils: A review of current knowledge and future research challenges. J Cleaner Prod 152, 63–76 (2017).
Schmidt, J. H. Life cycle assessment of five vegetable oils. J Cleaner Prod 87, 130–138 (2015).
Harnesk, D., Brogaard, S. & Peck, P. Regulating a global value chain with the European Union’s sustainability criteria- experiences from the Swedish liquid transport biofuel sector. J Cleaner Prod 153, 580–591 (2017).
Karlsson, H. et al. A systems analysis of biodiesel production from wheat straw using oleaginous yeast: process design, mass and energy balances. Biotechnol Biofuels 9, 229, https://doi.org/10.1186/s13068-016-0640-9 (2016).
Simopoulos, A. P. Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: nutritional implications for chronic diseases. Biomed Pharmacother 60, 502–507, doi:S0753-3322(06)00243-5 (2006).
Olstorpe, M., Pickova, J., Kiessling, A. & Passoth, V. Strain- and temperature-dependent changes of fatty acid composition in Wickerhamomyces anomalus and Blastobotrys adeninivorans. Biotechnol Appl Biochem 61, 45–50, https://doi.org/10.1002/bab.1130 (2014).
Slininger, P. J. et al. Comparative lipid production by oleaginous yeasts in hydrolyzates of lignocellulosic biomass and process strategy for high titers. Biotechnol Bioeng 113, 1676–1690, https://doi.org/10.1002/bit.25928 (2016).
Karlsson, H. et al. Greenhouse gas performance of biochemical biodiesel production from straw: soil organic carbon changes and time-dependent climate impact. Biotechnol Biofuels 10, 217, https://doi.org/10.1186/s13068-017-0907-9 (2017).
Meena, D. K. et al. Beta-glucan: an ideal immunostimulant in aquaculture (a review). Fish Physiology and Biochemistry 39, 431–457, https://doi.org/10.1007/s10695-012-9710-5 (2013).
Huyben, D. et al. Screening of intact yeasts and cell extracts to reduce Scrapie prions during biotransformation of food waste. Acta Vet Scandinavia 60, 9 (2018).
Cheng, K. et al. Metabolomics approach to evaluate a baltic sea sourced diet for cultured Arctic Char (Salvelinus alpinus L.). Journal of Agricultural and Food Chemistry 65, 5083–5090, https://doi.org/10.1021/acs.jafc.7b00994 (2017).
Appelqvist, L. Å. Rapid methods of lipid extraction and fatty acid methyl ester preparation for seed and leaf tissue with special remarks on preventing accumulation of lipid contaminants. Arkiv for Kemi 28, 551–570 (1968).
Fredriksson Eriksson, S. & Pickova, J. Fatty acids and tocopherol levels in M. Longissimus dorsi of beef cattle in Sweden - A comparison between seasonal diets. Meat Sci 76, 746–754, https://doi.org/10.1016/j.meatsci.2007.02.021 (2007).
Acknowledgements
We thank Dr. Su-Lin Leong for linguistic advice. This study was supported by a strategic grant of The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas), grant number 213-2013-80.
Author information
Authors and Affiliations
Contributions
J.Bl. was involved in study design, and performed a major part of laboratory work and writing the manuscript. J.P. provided a major contribution for study design, data analysis and final manuscript writing. S.K.T. performed parts of the laboratory work and data analysis. S.S. performed chemical analysis, data evaluation and was involved in manuscript writing. N.M. and J.Br. performed major parts of laboratory work, M.S. and V.P. coordinated the project and provided major contributions to study design, data analysis and final manuscript writing. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Blomqvist, J., Pickova, J., Tilami, S.K. et al. Oleaginous yeast as a component in fish feed. Sci Rep 8, 15945 (2018). https://doi.org/10.1038/s41598-018-34232-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34232-x
Keywords
This article is cited by
-
Production of short-chain fatty acids (SCFAs) as chemicals or substrates for microbes to obtain biochemicals
Biotechnology for Biofuels and Bioproducts (2023)
-
Enhanced glycerol assimilation and lipid production in Rhodotorula toruloides CBS14 upon addition of hemicellulose primarily correlates with early transcription of energy-metabolism-related genes
Biotechnology for Biofuels and Bioproducts (2023)
-
From straw to salmon: a technical design and energy balance for production of yeast oil for fish feed from wheat straw
Biotechnology for Biofuels and Bioproducts (2023)
-
Effects of Red Yeast (Sporidiobolus pararoseus) on Growth, Innate Immunity, Expression of Immune-related Genes and Disease Resistance of Nile Tilapia (Oreochromis niloticus)
Probiotics and Antimicrobial Proteins (2023)
-
Effects of urbanization on fungal communities and their functions in the sediment of the Haihe River
Journal of Soils and Sediments (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.