Abstract
Environmental fluctuations are ubiquitous and thus essential for the study of adaptation. Despite this, genome evolution in response to environmental fluctuations —and more specifically to the degree of environmental predictability– is still unknown. Saline lakes in the Mediterranean region are remarkably diverse in their ecological conditions, which can lead to divergent local adaptation patterns in the inhabiting aquatic organisms. The facultatively sexual rotifer Brachionus plicatilis shows diverging local adaptation in its life-history traits in relation to estimated environmental predictability in its habitats. Here, we used an integrative approach —combining environmental, phenotypic and genomic data for the same populations– to understand the genomic basis of this diverging adaptation. Firstly, a novel draft genome for B. plicatilis was assembled. Then, genome-wide polymorphisms were studied using genotyping by sequencing on 270 clones from nine populations in eastern Spain. As a result, 4,543 high-quality SNPs were identified and genotyped. More than 90 SNPs were found to be putatively under selection with signatures of diversifying and balancing selection. Over 140 SNPs were correlated with environmental or phenotypic variables revealing signatures of local adaptation, including environmental predictability. Putative functions were associated to most of these SNPs, since they were located within annotated genes. Our results reveal associations between genomic variation and the degree of environmental predictability, providing genomic evidence of adaptation to local conditions in natural rotifer populations.
Similar content being viewed by others
Introduction
Environmental fluctuations —either predictable or unpredictable– occur to a varying extent in any locality, so that adaptation to the degree and type of environmental fluctuation is a critical need for an organism to persist in a locality. Interestingly, climate change, over-exploitation, and habitat alteration often cause not just an environmental novelty in the average conditions, but an increase of fluctuations, with an important unpredictable component1,2,3. Different evolutionary responses are expected to result from adaptation to environmental fluctuations and their degree of predictability4. One of these, phenotypic plasticity, occurs when the individuals modify their phenotype accordingly to environmental conditions, without genetic change5. Another well-studied response is adaptive tracking, in which environmental variation results in correlated variation in mean population traits, since natural selection may favour different phenotypes over evolutionary time by acting on the heritable variation in the population6,7,8. When organisms have dormant stages such as seed banks or diapausing egg banks9,10 adaptive tracking may result in the maintenance of high levels of genetic and ecologically relevant diversity in populations11, which could provide strong signatures of balancing selection in genomes. A third response, bet hedging, occurs when a single genotype produces different phenotypes (i.e. diversified bet hedging, “don’t put all the eggs in the same basket”) or a “low-risk” phenotype (i.e. conservative bet hedging, “a bird in the hand is worth two in the bush”), so that it lowers the variance in its reproductive success but maximizes the reproductive success in the long-term12. It has been suggested that the evolution of bet hedging and phenotypic plasticity could involve regulatory genes in the genome8,13,14. However, to date, few molecular mechanisms underlying some of these evolutionary strategies have been identified, and these were mainly in relation to phenotypic plasticity (e.g. in Daphnia sp.15,16). With the advent of cost-effective genomic technologies, it is now possible to screen hundreds or thousands of genes and assess which ones are diverging in their allele frequencies, in order to assess how they affect evolutionary fitness in the wild17. The ultimate objective is to map multilocus genotypes to phenotypes to environments18,19. Therefore, from a fundamental and applied perspective, it is important to know how genomes respond to environmental fluctuations —and more specifically to unpredictable environments20,21,22–, a topic still scarcely investigated.
The Mediterranean region typically exhibits very different environmental regimes in habitats even within relatively small geographical scales23. In particular, Mediterranean saline lakes are strongly seasonal and temporally unpredictable environments at several time scales24. In addition, these habitats are also affected by global processes, such as climate change or the North Atlantic Oscillation25. Given the lack of detailed long-term, in situ records of most of these habitats, Franch-Gras et al.26 analysed long-term trends in water-surface area using a 27 years long series of remote sensing data on water-surface area to characterize the relative importance of environmental fluctuations in a set of saline lakes in eastern Spain. The indices available to estimate the degree of predictability from time-series data, basically, decompose the time series based on periodic and stochastic variation27 and associate these with predictable and unpredictable fluctuations, respectively. Franch-Gras et al.26 estimated the water-surface area of each lake in each satellite image and calculated the predictable and unpredictable components of variation of that data by using different models that reflected the effect of fluctuations on different types of organisms. Thus, predictability is defined as the organism’s ability to anticipate and adjust to future environmental conditions26. Franch-Gras et al.28 concluded that the predictability metrics based on Colwell’s approach29,30 and on the presence/absence of water were the most appropriate for small aquatic invertebrates28 (see methods for more details). Remarkably, the saline lakes studied showed a wide range of predictability, when predictability was assessed taking into account the point of view of small aquatic invertebrates26.
Cyclically parthenogenetic rotifers are common small invertebrates in ponds and lakes. These passively-dispersed aquatic organisms have been used successfully to study the evolutionary responses to environmental fluctuations described above31,32,33. These rotifers combine clonal proliferation with occasional bouts of sexual reproduction which results in the production of diapausing eggs. These eggs are long-lived, and are able to persist through adverse environmental conditions, including drought. In this temperate region, rotifer populations are temporary —not being active all year round– and colonize the water column during the so-called growing season. Thus, sex and the concomitant diapausing-egg production are critical for population persistence. In the case of organisms established as clones hatching after a sexual reproduction phase (i.e. cyclical parthenogenesis), such as aphids, rotifers and cladocerans, genetic slippage due to a phase of clonal selection followed by sexual reproduction can result in phenotypic traits changing in a direction even opposite to selection34. In addition, rotifer diapausing egg banks maintain high levels of standing genetic variance for life history traits35,36,37, which indicates that they should have a high potential to adapt to environmental fluctuations using adaptive tracking8. Moreover, theory predicts low-risk strategies to occur in rotifer populations inhabiting unpredictable habitats that would consist on (1) early production of diapausing eggs38 and/or (2) intermediate hatching fraction of diapausing eggs39. Indeed, rotifer populations show signatures of local adaptation to salinity and temperature in their life history traits35,36,37. Recently, Franch-Gras et al.28 found high within-population genetic variation in two life-history traits associated to diapause in nine populations of the rotifer Brachionus plicatilis inhabiting saline lakes with varying degrees of environmental predictability26. These traits were: (1) the propensity for sexual reproduction35,40 as a proxy of the timing of diapausing egg production41; and (2) the diapausing egg hatching fraction (the lower, the longer the diapause duration). The propensity for sex differed among populations and was inversely correlated with lake predictability, suggesting a conservative bet-hedging strategy that would protect against unexpectedly short growing seasons.
The study of the genetic basis of adaptation in non-model organisms has recently been fuelled by the rapid development of genomic technologies, especially next generation sequencing42,43,44. One of these technologies, genotyping by sequencing (GBS)45 is a cost-effective technique to screen genome-wide patterns of diversity by using restriction enzymes to reduce the genome complexity and has been used successfully to identify genomic regions under selection in several non-model animals46,47,48. It allows the genotyping of a high number of individuals and the discovery of typically large numbers of single nucleotide polymorphisms (SNPs).
Here, we take advantage of such a well-characterized study system of populations to study the genomic basis of local adaptation to environmental features. We studied the genome-wide patterns of genetic variability and differentiation in response to environmental predictability using GBS in the same set of B. plicatilis clones studied in Franch-Gras et al.28 in populations characterised for their environmental predictability by Franch-Gras26. Since no B. plicatilis genome was available, whole genome sequencing data were obtained in order to assemble the first draft genome for this species with a structural and functional annotation. This genome was used as a reference to facilitate (1) GBS analysis of population structure and (2) discovery of putative loci under selection. We investigated the correlation of loci under selection to life-history traits and environmental variables and identified genes associated to these loci. In this study we firstly, gather evidence of genomic association to local adaptation patterns by considering several factors; and then we focus on the degree of environmental predictability as a factor having a wide range in the studied lakes26 and to which the inhabiting populations have been shown to present patterns of local adaptation28.
Results
Draft genome
A total of 11.7 Gb of raw genomic sequence data were obtained. Based on k-mer statistics (k-mer length = 21; Supplementary Fig. S1), B. plicatilis HYR-1 genome size was estimated to be 115.77 Mb with an heterozygosity of 0.65%. Out of the four assemblers used, the Platanus assembly showed the best features, with the highest N50, no evidence of contamination and high proportion of complete and partial gene sequences in CEGMA completeness report (Table 1). Therefore downstream analyses were performed using this assembly as reference. Genome completeness analysis by CEGMA found 88.7 and 96.4% of complete and partial sequences respectively of the set of core eukaryotic genes included in the analysis (Table 1). The assembly included 108.5 Mb of genomic sequence scaffolds with 50% of all bases in scaffolds longer than 20.4 Kb (N50) and a maximum scaffold length of 169.9 Kb (Table 2). Finally, Blobtools did not find evidence of contamination based on both G + C content and contig coverage (Table 1; Supplementary Fig. S2).
The structural gene annotation yielded a predicted set of 54,725 gene models, some 30% of which could be functionally annotated.
GBS raw data, SNP calling and filtering
A total of 52 Gb of raw sequence data were obtained for the 270 clones from the nine populations. Out of the 630,460,573 raw reads, 379,536,357 quality-filtered barcoded reads were obtained. The SNP calling pipeline yielded 12,856 SNPs, resulting in 4,543 SNPs after filtering. Hardy-Weinberg proportions for each SNP and population showed that most SNPs do not differ from Hardy-Weinberg expectations (99.9% SNP with p-value > 0.05 after Bonferroni correction).
Genome-wide population differentiation
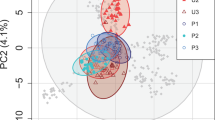
The overall expected heterozygosity (averaged over loci) was 0.17 and the within population expected heterozygosity ranged from 0.15 to 0.23 (Table 3). Heterozygosity correlated significantly to the log-transformed lake size (R2 = 0.40; t = 2.15, df = 7, p-value = 0.034). The overall FST (averaged over loci) was 0.18. Pairwise FST averaged across loci (Table 4) ranged from 0.07 (HYB and HYC) to 0.18 (SAL or HTU and HMT). The two major axes in PCA explained a joined variance of 13.1%. In the ordination space of these axes (Fig. 1), clones of the same populations were clustered, with some overlap between populations. There was not a significant pattern of isolation by distance (r = 0.31, p-value = 0.07, Mantel’s test).
Principal component analysis based on 4,543 genome-wide SNPs for 30 clones (dots) from each of the nine Brachionus plicatilis populations. Ellipsoids are the 95% confidence interval for each population. Percentage of variance explained by each principal component is shown between parentheses in the axis label.
Putative SNPs under selection
Using the BayeScan approach, 93 SNPs within 66 genomic regions (i.e. scaffolds) were identified as candidates to be under selection. 12 of these SNPs had significantly higher FST than the background of the genome (i.e. they are putatively under diversifying selection) and 81 have significantly lower FST than the background of the genome (i.e. they are putatively under balancing/purifying selection; Fig. 2). The latter group of SNPs were found to have a similar coverage than the rest of the SNPs in the genome (Supplementary Fig. S3), ruling out an artifact of merging duplicated loci. In addition, we identified further signatures of balancing rather than purifying selection as of these 81 SNPs, 64 (79.0%) have higher minor allele frequencies than the mean of the rest of the SNPs in the genome (i.e. MAF > 0.145) and 68 (83.9%) have positive Tajima D values.
Using the Bayenv approach, significant correlation with environmental and phenotypic variables was found for a number of SNPs ranging from 34 to 39 (Fig. 3). The highest Bayes factors were found for salinity, propensity for sex and hatching fraction. The highest number of SNPs correlated with two variables in our analysis (Fig. 4) were 10 SNPs located in ten genes (between hatching fraction and salinity) and 10 SNPs located in 9 genes (between propensity for sex and the degree of environmental predictability). Three (33.33%) of the SNPs detected by BayeScan as being under diversifying selection were also found strongly correlated with one of the variables tested in Bayenv (two to hatching fraction, one to propensity for sex and one to salinity; one of them was correlated with both salinity and hatching fraction see Supplementary Table S1). None of the SNPs putatively under balancing/purifying selection was also found to be correlated to any variable tested in Bayenv.
Bayesian factors (BF) and mean rank for the correlation between SNPs (dots) and three environmental (upper row panels) and two phenotypic (lower row panels) variables. Results are based on twenty replicate runs of Bayenv. Vertical red line shows the mean rank threshold (mean rank > 0.99) to consider a SNP to be outlier (red dots).
The GWAS analysis using GenABEL yielded a single SNP significantly associated to the propensity for sex, this SNP was not detected by Bayenv nor by BayeScan. No SNP was significantly associated to hatching fraction.
Genes associated to SNPs putatively under selection
Most of the SNPs detected with BayeScan or Bayenv were located within genes (Supplementary Table S2). Out of the 12 SNPs that BayeScan detected under diversifying selection, 11 (91.7%) were located within 12 genes; the one that was not found within any gene was located at 1,783 bp from the closest gene. Regarding Bayenv, the number of genes ranged from 27 to 35. Finally, the only SNP detected by GenABEL (associated to propensity for sex) was within the coding region of a gene annotated as “Exosome complex exonuclease RRP44”.
The 164 putative genes under selection by either BayeScan, Bayenv or GenABEL (Supplementary Table S2) had a broad range of gene ontology (GO) annotations. According to the enrichment analysis, the genes associated to propensity for sex and environmental predictability were enriched for genes involved in binding, those associated to salinity were enriched in genes implicated in regulation of cellular processes; and those genes associated to hatching fraction were enriched in signal transducer activity (see details in Supplementary Table S3) For those genes associated to hydroperiod length, no significant gene ontology enrichment was found. The gene found to be related to propensity for sex in GenABEL was found to be associated to the following functions: rRNA catabolic process (GO:0006401), regulation of RNA metabolic process (GO:0051252), rRNA processing (GO:0006364), and mRNA catabolic process (GO:0006402).
Discussion
We used a powerful, integrated approach —combining environmental, phenotypic and genomic data for the same set of populations– to identify genomic regions putatively under selection to local conditions, focusing on environmental predictability in rotifer populations. Moreover, we report a new draft genome assembly for B. plicatilis, in a taxonomic group (monogonont rotifers) in which the available genetic resources are still scarce (but see Brachionus calyciflorus genome)49. We identified candidate regions for diversifying selection, some of them are correlated with environmental predictability and some of these are correlated with life-history traits and other ecological factors. Moreover, we found a large set of genes showing signatures of balancing selection in rotifer populations.
Draft genome of Brachionus plicatilis
We have described and annotated the first draft genome assembly of B. plicatilis. The estimated (haploid) genome size is 115.77 Mb, similar to the assembly size obtained (108.5 Mb) and the previous estimates for different B. plicatilis strains using flow-cytometry (between 111.7–128.8 Mb)50. The k-mer analysis revealed that the heterozygosity of the B. plicatilis genome is relatively high (0.65%). This is consistent with the finding that Platanus assembler yielded better results than other assemblers, since it is known to have a good performance when assembling highly heterozygous genomes51. The genome was highly compact, with a large number of predicted gene models (54,725). This compactness produced a high number of genes physically associated to the SNPs putatively under selection. Therefore the analysis was restricted to those genes where these SNPs were in their coding regions. Although this approach might potentially produce a loss of information, since introns and inter-genic space changes can also affect gene expression, the fast linkage decay with physical distance found after a linkage disequilibrium analysis (Supplementary Fig. S4) supports the use of such a restricted area of influence of the SNPs detected in our study species. Although Brachionus plicatilis is regarded as a model species for evolutionary biology studies52,53, before this contribution the available genetic resources for B. plicatilis were limited to EST libraries (NCBI database) and transcriptomes of congeneric species (Supplementary Table S4). Furthermore, there are only two rotifer genomes available: (1) the distantly related bdelloid Adineta vaga, belonging to the strictly asexual class Bdelloidea54 and (2) the recently published Brachionus calyciflorus genome49. It would be desirable both to improve the quality of the genome assembly, for instance, by the inclusion of both more sequencing data (such as mate-pairs or PacBio platform sequencing) and gene mapping information and transcriptome data.
Genes and SNPs putatively under selection
We identified 12 candidate genes under diversifying selection in the nine rotifer populations studied using Bayescan. These results indicate that rotifer genomes show signatures of adaptation to local conditions (not necessarily to predictability). Previous work has shown local adaptation in life history traits at intermediate (29,000 km2; Campillo et al.)37 and small (240 km2; Franch-Gras et al.)28 spatial scales, in the Mediterranean region. Brachionus plicatilis populations in Spain harbour a high degree of ecologically relevant genetic variation frequently involving sexual reproduction and diapause strategies35,55,56. This variation has been found to be correlated with environmental features such as salinity55 and environmental predictability28. However, the genetic basis of local adaptation in this populations remained unknown. There are very few studies considering the genetic basis of local adaptation in aquatic invertebrates. Orsini et al.57 related several genes of Daphnia magna to a set of natural anthropogenic stressors using an experimental evolution approach, and identified repeatable patterns of local adaptation. Roulin et al.16 identified one gene in the same species associated to a photoreceptor inducing a plastic response of diapause that is involved in local adaptation patterns.
We are aware that FST methods such as Bayescan have low power when populations are high differentiated for neutral markers58,59, which is the case for B. plicatilis (see below), however we used a powerful correlation analysis (Bayenv) to identify candidate SNPs strongly associated with environmental predictability, hydroperiod length, salinity and two life history traits related to diapause. This correlation-based analysis uses information of important environmental and phenotypic variables for each lake/population, which are not considered in the FST method (BayeScan), resulting in the ability to detect both adaptive responses to each variable or genes that might determine diapausing-related traits. Three out of the 12 SNPs found to be putatively under diversifying selection by BayeScan were also highlighted as strongly correlated by Bayenv, what highlights the importance of the studied variables in driving local rotifer adaptation. Therefore these three SNPs are very strong candidates to be under diversifying selection in response to hatching fraction, propensity for sex and salinity. In addition, the GWAS analysis using GenABEL yielded one SNP significantly associated to the propensity for sex.
From those SNPs that Bayenv related to the variables, 10 (located in ninegenes) were correlated simultaneously to both the propensity for sex and the degree of environmental predictability (Fig. 4). As a result, 29.4% of the SNPs highlighted by Bayenv as correlated with the degree of predictability were also correlated with the propensity for sex. This provides a genetic basis to the tight relationship between these two variables, according to the findings in Franch-Gras et al.28. In addition, the weak relationship between the degree of predictability and the hatching fraction observed by Franch-Gras et al.28 is consistent with our results at the genomic level. Only four SNPs detected by Bayenv (located in two genes) were correlated with both variables simultaneously. This implies that only 11.7% of the SNPs correlated with the degree of predictability were also correlated with the hatching fraction. The 37 SNPs found to be correlated with propensity for sex in Bayenv plus the one obtained by GWAS and the 38 SNPs found to be correlated with the hatching fraction are strong candidates to be underlying the genomic basis of each of these traits.
Though our main interest was on unravelling the genomic responses to unpredictable environments, we also explored the effects of lake area and geographic distance on population genetic parameters, and adaptive responses to other environmental parameters such as hydroperiod length and salinity, as they are likely additional factors to explain adaptive genetic divergence among populations or can act as confounding factors. Our results agree with the findings by Franch-Gras28 regarding the low influence of hydroperiod length on the propensity for sex once the effect of environmental predictability is discounted.
A large number of SNPs that are correlated with the studied environmental variables (i.e. hydroperiod length, environmental predictability or salinity) do not correlate with life-history traits (i.e. propensity for sex or hatching fraction), suggesting that the former acted as selective pressures on traits not considered here. Conversely, a high number of SNPs correlated with the life-history traits studied were not correlated with the environmental variables. Obviously, not all the genetic variability associated to these fitness components is responding to selection by these particular environmental variables. Other selective pressures not considered here —such as lake trophic status or temperature– could be acting. Nonetheless, through adaptive tracking, genes causing divergence in life-history traits might be responding to short-term fluctuations that are uncoupled across localities. Genetic differentiation in Daphnia populations caused by this selective scenario has been reported11. Interestingly, fluctuations of water-surface area —an important feature and a proxy for other features such as variations in the physicochemical conditions are poorly correlated among the lakes studied26. Most of the SNPs correlated with any of the variables studied here are located within genes, which showed a range of gene ontologies, so that reliable inferences are difficult. However, a specific insight on gene functionality was achieved for the genes associated to the propensity for sex and environmental predictability, because the set showed an enrichment of genes that have been putatively associated to binding functions. Interestingly, both phenotypic plasticity and bet hedging have been proposed to be regulated by epigenetic mechanisms13,14. In addition, genes associated to hatching were found to be enriched in signal transducer activity and genes associated to salinity to regulation of cell cycle or of biological processes. After examining individual gene functions, some interesting putative functions of genes associated to specific variables are discussed below. Indeed, a gene associated to hatching fraction is described as “coronin-7 isoform X1” and is involved in the actin filament polymerization, which is upregulated in diapausing mosquitos60. A gene associated to hydroperiod, described as “estrogen receptor beta”, which increases reproduction when being activated by a ligand in the rotifer Brachionus manjavacas61. Moreover, a gene associated to predictability is described as “POU class transcription factor 1-like”, a transcription factor which is more expressed in non-diapause than in diapause pupae in the moth Helicoverpa armigera62. A gene associated to propensity for sex in this study described as “speedy A” has a “male meiotic nuclear division” GO term, as is involved in reproductive meiosis in mice63 and pigs64. Finally, a gene associated to salinity is described as “G2 mitotic-specific cyclin partial” and has a “regulation of cell cycle” GO term, the arrest of the cell cycle in diapause has been associated to the G2 stage in Bombyx mori65. Although tantalizing, these are tentative gene function descriptions and caution should be taken when considering these results. Undoubtedly, additional information on rotifer genome and transcriptomics would refine the functionality of genes that are candidates to be targets of selection by the degree of environmental predictability.
We identified a high proportion of putative SNPs under selection with signatures of balancing or purifying selection, 54 out of the 66 genomic regions (i.e. scaffolds) under selection. We found that most of these SNPs (74.1%) had both minor allele frequencies higher than the genome average and positive Tajima D values, suggesting that they are under balancing rather than purifying selection or being false positives66. In addition, we confirmed that this result is not due to artifactually over-merging duplicated genes (Supplementary Fig. S3). Although finding evidence of genomic regions under balancing selection is not unexpected67,68,69, it is unusual to find such a large number of genomic regions under balancing selection within a small geographical area. Several mechanisms have been proposed to underlie balancing selection, including heterozygote advantage70, negative frequency-dependent selection71, temporal habitat heterogeneity72,73, genetic slippage34 or conflicting optimal fitness between life-cycle stages74. The fact that the clones used in this study were established from diapausing egg banks may also be relevant, as such egg banks can accumulate diversity relevant to different selection pressures in different time periods9,75 despite not showing differences in neutral markers75. Although further research is needed to determine the mechanisms underlying the observed pattern of balancing selection, temporal habitat heterogeneity (including fluctuating selection patterns) or loci with conflicting fitness effects in different life-cycle stages35 could be important in the studied B. plicatilis populations. Indeed, these results are consistent with the high polymorphism of B. plicatilis genomes despite prolonged clonal phases of proliferation of their populations35, with the large intrapopulation genetic diversity for ecologically relevant life history traits28,33,37. The GWAS analysis allowed this inter-clonal variability to be analysed, and yielded one SNP associated to propensity for sex.
Given that we used a genome representation technique (Genotyping by sequencing, GBS), which does not screen all available SNPs in the genome, our analysis resulted in an average distance between SNPs is 25.73 kb (115.77 Mb/4500), which is over the distance at which the linkage disequilibrium drops (see Supplementary Fig. S4). Thereby, it is likely that this study does not present a comprehensive list of loci involved in local adaptation. Further explorations of the SNPs of the genome of B. plicatilis are needed in order to find out further SNPs involved in adaptation to local conditions, especially when the whole genome sequencing techniques become affordable enough to apply them to a large set of individuals in population genetics studies.
Population structure
Genome-wide patterns of genetic variation did not reveal a geographically based population structure, which was not unexpected given the proximity of the studied populations. In previous studies, isolation-by-distance has been reported in this species at a larger regional scale55,76, but —consistently with what was found in the same geographical area using microsatellites and two mitochondrial genes by Montero-Pau et al.77– no evidence was observed here. The overall population differentiation (FST = 0.18) was slightly lower than the estimate obtained by Montero-Pau et al.77 with microsatellite markers (FST = 0.25) for the same geographical area. Consistently to what Montero-Pau et al.77 observed using mitochondrial data, we found a significant relation between genetic diversity at the genomic scale (measured as estimated heterozygosity) and lake size, suggesting that lake size is a reliable proxy for effective population size in B. plicatilis populations.
Conclusions
We have used a powerful approach in a set of natural rotifer populations integrating genomic data with data on environmental features of their habitats26 and their phenotypic diversity26. Our genomic analysis identifies and screens a large number of high quality SNPs (4,543) in a small —but dense in gene regions– genome, assembled and annotated here. Some of these SNPs were putatively under selection, with strong signatures of both diversifying and balancing selection. Moreover, a large number of SNPs were also correlated with environmental predictability and other environmental and phenotypic variables. Remarkably, most of these SNPs were located within functionally annotated genes on the genome. Our results provide evidence of genomic adaptive responses to local conditions in natural rotifer populations, focusing on the degree of environmental predictability.
Material and Methods
Samples and study sites
Brachionus plicatilis clones from nine Spanish salt lakes differing in predictability conditions (Fig. 1, Table 5) were obtained from diapausing egg banks in a previous study28. A total of 30 clones from each field population were established and maintained under standard laboratory conditions (Supplementary Method S1). Data on the degree of environmental predictability, hydroperiod length and size of the lakes are from Franch-Gras et al.26, data on the life-history traits from the B. plicatilis clones are from Franch-Gras et al.28. Briefly, the predictability metric was calculated in Franch-Gras et al.26 by constructing a contingency table (with two rows: presence/absence of water; and 12 columns: the calendar months) with the complete time series of each lake. With this contingency matrix, as described in Colwell29 and Stearns30, predictability was calculated as the summation of two values: constancy and contingency. The time-series for water-surface area and the resulting predictability values associated to each pond are shown in Fig. 5. Salinity is an important parameter shaping rotifer life-history traits55,78 and the Mediterranean region salt lakes have a wide range of salinity24,79, therefore salinity was also measured during the sampling of the lake sediments (May-September 2013). Note that this salinity measure is based on a single visit, thus although it is a value that may be indicative of the salinity of each of the ponds it does not integrate its temporal variability.
In the center, location of the nine lakes studied here. For each pond the water-surface area time series and its predictability (P) and hydroperiod values (H) are shown. Data obtained from Franch-Gras et al.26.
Brachionus plicatilis genome
DNA extraction and DNA sequencing library construction
A monoclonal culture from one individual stem female hatched from a diapausing egg isolated from Hoya Rasa lake (HYR1 clone)28 was allowed to grow as described in Supplementary Method S2 to obtain enough biomass for DNA extraction. Genomic DNA was extracted with the DNeasy Blood and Tissue kit (Qiagen, Valencia, CA) following the manufacturer’s protocol. DNA quantification was performed using a dsDNA assay (Invitrogen) on a Qubit 2.0 fluorometer (Life Technologies) and quality was assessed by electrophoresis in a 1% agarose gel. For library construction, DNA was sheared through sonication down to ca. 550 bp by using a Diagenode Bioruptor. PCR-free DNA libraries were prepared with the sonicated DNA using the NEBNext Ultra DNA library Prep kit for Illumina combined with NEBNext Singleplex Adaptors. The library was quantified using a Qubit fluorometer and assessed by qPCR using the Illumina Library Quantification Kit (NEBNext Library Quant Kit) and a StepONE plus Real time PCR system (Applied Biosystems). The template DNA was denatured according to the protocol described in the Illumina system guide and loaded at 20 pM concentration. To improve sequencing quality, 1% PhiX control was spiked-in. The library was sequenced on a single Illumina MiSeq sequencer run with v3 chemistry (2 × 250 bp paired-end read lengths). Library preparation and sequencing was carried out at the EvoHull Genomics Lab at the University of Hull (Hull, United Kingdom).
Genome assembly and annotation
The sequenced reads were trimmed using Trimmomatic v.0.3280 to remove (1) Illumina adapters, (2) low quality leading or trailing regions (quality measured as Phred score; Phred score > 30) or unidentified nucleotides (i.e. Ns), (3) reads with average Phred score < 100, and (4) regions (5-base sliding window) with average Phred score < 20. Thereafter, reads were corrected using a Bloom filter-based error correction tool (BLESS v0.16)81. The paired reads were then merged using Usearch v. 882. Pre-assembly assessment of k-mer distributions were obtained by KMC83 and profiling by GenomeScope84.
De novo genome assemblies were constructed with several assemblers using default parameters (Celera, SPAdes, DISCOVAR, and Platanus; Table 1) and discarding contigs shorter than 500 bp in the final assembly. Assembly quality was evaluated using (1) Blobtools85,86 to assess contamination, (2) CEGMA v. 2.5 (Core Eukaryotic Genes Mapping Approach)87, a tool that assesses genome completeness based on the presence of a set of core eukaryotic genes, and (3) Assemblathon scripts88 to obtain length-based statistics, such as N50. Platanus assembly was chosen as the reference assembly (see Results) and downstream analyses were performed on this assembly.
In order to perform the structural annotation of the assembly, GeneMark v. 4.3289 was initially used as an ab initio gene prediction software. After that, GeneMark predictions were provided to run MAKER2 v. 2.31.890 an annotation pipeline that identifies repetitive elements, aligns expressed sequence tags (ESTs), and uses protein homology evidence to generate further gene predictions. MAKER2 was run twice. The first MAKER2 round was performed with permissive settings (Supplementary Data S1) using SNAP91 and RepeatMasker v.3.092. In order to create evidence-based gene model predictions, MAKER2 was provided in this first run with: (1) the core eukaryotic gene models obtained from CEGMA, (2) ESTs from B. plicatilis NCBI database, (accession: 10-07-2016), (3) available transcriptomes from congeneric species (Supplementary Table S4), (4) proteins from Adineta vaga (a high-quality bdelloid rotifer genome)54, and (5) the Swiss-Prot database. The annotation output from MAKER2 first run was converted into a training set to run Augustus. Finally, the Augustus and MAKER2 (1st pass) outputs were provided to perform a second MAKER2 pass with more stringent settings (Supplementary Data S2) in order to obtain the final set of gene predictions.
The functional annotation of the final set of predicted genes was performed by identifying protein domains with InterProScan v.593 and by performing Blastp searches (e-value < 10−5)94 against a subset (metazoa only) of the NCBI’s non-redundant (nr) protein database. Finally, Blast2GO v.4.1.995 was run with default settings to integrate results from BLAST and InterProScan and to retrieve Gene Ontology (GO) terms and annotation.
Genotyping by sequencing of B. plicatilis clones
DNA extraction and DNA sequencing library construction
Each of the 270 clones of B. plicatilis was allowed to grow as described in Supplementary Method S2 to obtain enough biomass for DNA extraction. DNA extraction was performed with the JETFLEX Genomic DNA purification kit (GENOMED, Löhne, Germany) following manufacturer’s protocol. After quality assessment and quantification (see above), DNA samples were submitted to Cornell University Genomic Diversity Facility at the Institute of Biotechnology (IGD, Ithaca, NY, US), where libraries were constructed and sequenced. This was performed on an Illumina HiSeq. 2000/2500 (100 bp, single-end) according to Elshire et al.45, using the restriction enzyme ApeKI (GC[A-T]GC) for digestion. A library with unique barcodes for each clone, plus blank samples, was created.
SNP discovery and filtering
SNPs were called from the raw DNA sequences using the GBS pipeline as implemented in TASSEL-GBS v2 (TASSEL 5)96, in which all reads were trimmed to the same length (64 bp) and identical reads were collapsed into tags. These tags were then aligned against the reference genome using the Burrows-Wheeler alignment tool (BWA)97 and SNPs were called from aligned tags. Pipeline default parameters were used except for two cases, in which more conservative values than default were used: (1) at the Minimum length of aligned base pair to store the Sequence Alignment/Map (SAM) entry (SAMToGBSdbPlugin plugin, option aLen being 30 instead of default 0); and (2) at the Minimum locus coverage (i.e. proportion of Taxa with a genotype; DiscoverySNPCallerPluginV2 plugin, option mnLCov being 0.8 instead of default 0.1).
The set of SNPs were quality filtered using custom developed scripts (Supplementary Data S3) and VCFtools98. In order to obtain confidently assigned genotypes, those genotypes at each sample supported by less than six reads were excluded (considered as not genotyped). A set of filters were applied to retain those SNPs highly informative, of high quality and not representing over-merged repetitive regions. Firstly, SNPs required at least 50% of the clones genotyped in each population. Second, minor allele frequency (MAF) had to be higher than 1%. Third, only two alleles were present. Fourth, the average read depth among clones had to be lower than 150 reads48. Fifth, less than 60% of clones had to be heterozygotes in each SNP.
Population genomic analyses
To estimate genome-wide genetic variation and differentiation, principal component analysis (PCA), a model-free multivariate ordination method, was performed as implemented in the “adegenet” package99 in R v.3.3.3100. Using the package “hierfstat”101, genetic divergence between populations was estimated by the fixation index (FST) for each pairwise comparison, and within-population genetic diversity was measured as the average among loci expected heterozygosity (He), observed heterozygosity (Ho) and inbreeding coefficient (FIS). Mantel tests were used to test for isolation-by-distance patterns in IBDWS v.3.23102 using the geographic distance between lakes calculated using Google Earth (2008) and pairwise FST as genetic distance. The relationship between heterozygosity (He) and log-transformed lake area (Table 5) was tested using correlation analysis. Tajima D-values and Hardy-Weinberg tests were obtained using Plink v. 1.90103. File format conversions were performed using PGDSpider v. 2.1.0.3104.
Putative SNPs under selection and associated genes
To identify SNPs likely to be differentiated as a result of selection, three different approaches were used. Firstly, BayeScan 2.1105 was used to estimate the posterior probability that a given SNP is affected by selection based on the allele frequencies in each population. It identifies those SNPs with significantly higher —i.e. diversifying selection– or lower —i.e. balancing/purifying selection– differentiation among populations than expected under a model of neutral evolution18. Briefly, prior odds of 10 were used to identify the top candidate SNPs under selection with a false discovery rate of 0.05. Secondly, we used Bayenv2106 to identify loci correlated with particular environmental and phenotypic variables. Bayenv uses a Bayesian approach that takes into account population structure -from covariance matrices- when testing for correlations between environmental and population allelic frequencies106. To do so, the full set of SNPs were used for the construction of 10 covariance matrices using different priors (following Hahn et al.)107. The association with genetic variation was tested for three environmental variables known for the lakes under study (environmental predictability, hydroperiod length and salinity)26, and two phenotypic variables previously estimated for the genotyped populations (life-history traits: propensity for sex and hatching fraction, Table 5)28. For the latter, we used the average of the clones genotyped here for the corresponding population. Each environmental or phenotypic variables was standardized by subtracting the mean and dividing by the standard deviation of the variable. Twenty independent runs with different random seeds were run, obtaining for each SNP the Bayesian Factors (a parameter usually used in Bayesian statistics for model choice, hereafter BF). Following Hancock et al.108 we used the BF of all of the SNPs to calculate a rank statistic (each SNP scaled between 0 and 1; corresponding to the SNP with the lowest and highest BF per run, respectively). The rank statistic was averaged between runs to obtain the mean rank of each SNP. SNPs were considered as potentially under selection when their mean rank was above 0.99. Thirdly, a genome-wide association study (GWAS) was performed for both life-history traits in order to explore genotype-phenotype association by using GenABEL R package109 with EIGENSTRAT methodology110. In this approach the inter-clonal variability is analysed by considering individual phenotypic values and genotypes, which was hidden behind the averages used in the Bayenv approach. This methodology uses PCA to correct for population stratification. For each SNP and phenotypic variable association analyzed, it provides the Bonferroni adjusted p-value and the R2. SNPs with p-value lower than 0.05 are considered to be putatively associated to the phenotype.
In order to identify the genes associated to putative SNPs under selection —as identified by BayeScan, Bayenv or GenABEL–, BEDtools v.2.19.1111 was used to find genes in a flanking region of 0, 2.5 or 5 Kb upstream and downstream from the focal SNPs. As expected from the compact genome of B. plicatilis (Table 2), a large number of putative genes were found closely linked to SNPs under selection (Supplementary Table S2). A GO enrichment analysis (Fisher’s exact test, two- tailed, false discovery rate < 0.05) was conducted to test if certain gene ontologies were over- or under- represented in the lists of genes putatively under selection with respect to the genome using Blast2GO v. 4.1.995.
The sequences used in this study were deposited in GenBank (BIOproject ID: PRJNA422189). GBS raw sequences were deposited in the ENA (accession number SRP151997). The Whole Genome Shotgun project has been deposited at DDBJ/ENA/GenBank under the accession REGN00000000. The version described in this paper is version REGN01000000.
References
IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (Cambridge University Press, 2013).
Pimm, S. L., Russell, G. J., Gittleman, J. L. & Brooks, T. M. The Future of Biodiversity. Science 269, 347–350 (1995).
Root, T. L. et al. Fingerprints of global warming on wild animals and plants. Nature 421, 57–60 (2003).
Simons, A. M. Modes of response to environmental change and the elusive empirical evidence for bet hedging. Proc. R. Soc. B. 278, 1601–1609 (2011).
Reed, T. E., Waples, R. S., Schindler, D. E., Hard, J. J. & Kinnison, M. T. Phenotypic plasticity and population viability: the importance of environmental predictability. Proc. R. Soc. B. 277, 3391–400 (2010).
Botero, C. A., Weissing, F. J., Wright, J. & Rubenstein, D. R. Evolutionary tipping points in the capacity to adapt to environmental change. Proceedings of the National Academy of Sciences 112, 184–189 (2015).
Lynch, M. & Lande, R. Evolution and extinction in response to environmental change in Biotic Interactions and Global Change (eds Karieva, P. M., Kingsolver, J. G. & Huey, R. B.) 234–250. (Sinauer Associates, 1993).
Tufto, J. Genetic evolution, plasticity, and bet-hedging as adaptive responses to temporally autocorrelated fluctuating selection: A quantitative genetic model. Evolution 69, 2034–2049 (2015).
Brendonck, L. & De Meester, L. Egg banks in freshwater zooplankton: Evolutionary and ecological archives in the sediment. Hydrobiologia 491, 65–84 (2003).
Lundemo, S., Falahati-Anbaran, M. & Stenøien, H. K. Seed banks cause elevated generation times and effective population sizes of Arabidopsis thaliana in northern Europe. Mol. Ecol. 18, 2798–2811 (2009).
Lynch, M. The consequences of fluctuating selection for isozyme polymorphisms in Daphnia. Genetics 115(4), 657–669 (1987).
Ripa, J., Olofsson, H. & Jonzén, N. What is bet-hedging, really? Proc. R. Soc. B. 277(1685), 1153–1154 (2010).
Duncan, E. J., Gluckman, P. D. & Dearden, P. K. Epigenetics, plasticity, and evolution: How do we link epigenetic change to phenotype? J. Exp. Zool. B: Molecular and Developmental Evolution 322, 208–220 (2014).
O’Dea, R., Noble, D. & Johnson, S. The role of non-genetic inheritance in evolutionary rescue: epigenetic buffering, heritable bet hedging and epigenetic traps. Environ. Epigenet. 2, dvv014 (2016).
Yampolsky, L. Y. et al. Functional genomics of acclimation and adaptation in response to thermal stress in Daphnia. BMC Genomics 15, 859 (2014).
Roulin, A. C., Bourgeois, Y., Stiefel, U., Walser, J.-C. & Ebert, D. A. Photoreceptor contributes to the natural variation of diapause induction in Daphnia magna. Mol. Biol. Evol. 33, 3194–3204 (2016).
Feder, M. & Mitchell-Olds, T. Evolutionary and ecological functional genomics. Nat. Rev. Genet. 4, 651–657 (2003).
Storz, J. F. Using genome scans of DNA polymorphism to infer adaptive population divergence. Mol. Ecol. 14, 671–688 (2005).
Stinchcombe, J. R. & Hoekstra, H. E. Combining population genomics and quantitative genetics: finding the genes underlying ecologically important traits. Heredity 100, 158–170 (2008).
Chevin, L.-M., Lande, R. & Mace, G. M. Adaptation, plasticity, and extinction in a changing environment: towards a predictive theory. PLoS Biol. 8, e1000357 (2010).
Chown, S. et al. Adapting to climate change: a perspective from evolutionary physiology. Climate Res. 43, 3–15 (2010).
Crozier, L. G. et al. Potential responses to climate change in organisms with complex life histories: evolution and plasticity in Pacific salmon. Evol. Appl. 1, 252–270 (2008).
Blondel, J. & Aronson, J. Biology and wildlife of the Mediterranean region. Oxford University Press, New York (1999).
Comín, F., Rodó, X. & Comín, P. Lake Gallocanta (Aragón, NE Spain): a paradigm of fluctuations at different scales of time. Limnetica 8, 79–86 (1992).
Rodó, X., Baert, E. & Comín, F. Variations in seasonal rainfall in Southern Europe during the present century: relationships with the North Atlantic Oscillation and the El Niño-Southern Oscillation. Clim. Dyn. 13(4), 275–284 (1997).
Franch-Gras, L., García-Roger, E. M., Carmona, M. J. & Serra, M. Quantifying unpredictability: a multiple model approach for Mediterranean ponds by using satellite imagery. PLoS ONE 12(11), e0187958 (2017a).
Sabo, J. L. & Post, D. M. Quantifying periodic, stochastic, and catastrophic environmental variation. Ecological Monographs 78, 19–40 (2008).
Franch-Gras, L., García-Roger, E. M., Serra, M. & Carmona, M. J. Adaptation in response to environmental unpredictability. Proc. R. Soc. B. 284, 20170427 (2017b).
Colwell, R. K. Predictability, constancy, and contingency of periodic phenomena. Ecology 55, 1148–1153 (1974).
Stearns, S. C. On measuring fluctuating environments: predictability, constancy, and contingency. Ecology 62, 185–199 (1981).
Hairston, N. G. Jr. & Munns, W. R. The timing of copepod diapause as an evolutionarily stable strategy. Am. Nat. 123, 733–751 (1984).
De Meester, L. Local genetic differentiation and adaptation in freshwater zooplankton populations: Patterns and processes. Ecoscience 3, 385–399 (1996).
Tarazona, E., García-Roger, E. M. & Carmona, M. J. Experimental evolution of bet hedging in rotifer diapause traits as a response to environmental unpredictability. Oikos 126(8), 1162–1172 (2017).
Lynch, M. & Deng, H.-W. Genetic slippage in response to sex. Am. Nat. 144, 242–261 (1994).
Carmona, M. J., Dimas-Flores, N., GarcÍa-Roger, E. M. & Serra, M. Selection of low investment in sex in a cyclically parthenogenetic rotifer. J. Evol. Biol. 22, 1975–1983 (2009).
Campillo, S., GarcÍa-Roger, E. M., Carmona, M. J., Gómez, A. & Serra, M. Selection on life-history traits and genetic population divergence in rotifers. J. Evol. Biol. 22, 2542–2553 (2009).
Campillo, S., García-Roger, E. M., Carmona, M. J. & Serra, M. Local adaptation in rotifer populations. Evol. Ecol. 25, 933–947 (2010).
Serra, M. & King, C. E. Optimal rates of bisexual reproduction in cyclical parthenogens with density-dependent growth. J. Evol. Biol. 12, 263–271 (1999).
García-Roger, E. M., Serra, M. & Carmona, M. J. Bet-hedging in diapausing egg hatching of temporary rotifer populations - A review of models and new insights. Int. Rev. Hydrobiologia 99, 96–106 (2014).
Aparici, E., Carmona, M. J. & Serra, M. Variability for mixis initiation in Brachionus plicatilis. Hydrobiologia 446/447, 45–50 (2001).
García-Roger, E. M., Carmona, M. J. & Serra, M. Modes, mechanisms and evidence of bet hedging in rotifer diapause traits. Hydrobiologia 796, 223–233 (2016).
Van Straalen, N. M. & Roelofs, D. An introduction to ecological genomics. (Oxford University Press, 2011).
Roff, D. A. Contributions of genomics to life-history theory. Nat. Rev. Genet. 8, 116–125 (2007).
Stapley, J. et al. Adaptation genomics: the next generation. Trends Ecol. Evol. 25, 705–712 (2010).
Elshire, R. J. et al. A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PloS ONE 6, e19379 (2011).
Monzón, J. D., Atkinson, E. G., Henn, B. & Benach, J. Population and evolutionary genomics of Amblyomma americanum, an expanding arthropod disease vector. Genome Biol. Evol. 8(5), 1351–1360 (2016).
Muñoz, J., Chaturvedi, A., De Meester, L. & Weider, L. J. Characterization of genome-wide SNPs for the water flea Daphnia pulicaria generated by genotyping-by-sequencing (GBS). Sci. Rep. 6, 28569 (2016).
Johnson, J. et al. Genotyping-by-sequencing (GBS) detects genetic structure and confirms behavioral QTL in tame and aggressive foxes (Vulpes vulpes). PLoS ONE 10(6), e0127013 (2015).
Kim, H. S. et al. The genome of the freshwater monogonont rotifer Brachionus calyciflorus. Molecular ecology resources 18, 646–655 (2018).
Stelzer, C.-P., Riss, S. & Stadler, P. Genome size evolution at the speciation level: the cryptic species complex Brachionus plicatilis (Rotifera). BMC Evol. Biol. 11, 90 (2011).
Kajitani, R. et al. Efficient de novo assembly of highly heterozygous genomes from whole-genome shotgun short reads. Genome Res. 24, 1384–1395 (2014).
Kostopoulou, V., Carmona, M. J. & Divanach, P. The rotifer Brachionus plicatilis: an emerging bio-tool for numerous applications. J. Biol. Res-Thessalon 17, 97–112 (2012).
Declerck, S. & Papakostas, S. Monogonont rotifers as model systems for the study of micro-evolutionary adaptation and its eco-evolutionary implications. Hydrobiologia 796, 131–144 (2017).
Flot, J.-F. et al. Genomic evidence for ameiotic evolution in the bdelloid rotifer Adineta vaga. Nature 500, 453–457 (2013).
Campillo, S., Serra, M., Carmona, M. J. & Gómez, A. Widespread secondary contact and new glacial refugia in the halophilic rotifer Brachionus plicatilis in the Iberian Peninsula. PLoS ONE 6(6), e20986 (2011).
Gabaldón, C. & Carmona, M. J. Allocation patterns in modes of reproduction in two facultatively sexual cryptic rotifer species. J. Plankton Res. 37, 429–440 (2015).
Orsini, L., Spanier, K. I. & De Meester, L. Genomic signature of natural and anthropogenic stress in wild populations of the waterflea Daphnia magna: validation in space, time and experimental evolution. Mol. Ecol. 21, 2160–2175 (2012).
De Kovel, C. G. F. The power of allele frequency comparisons to detect the footprint of selection in natural and experimental situations. Genet. Sel. Evol. 38, 3–23 (2006).
Butlin, R. K. Population genomics and speciation. Genetica 138, 409–418 (2010).
Kim, M., Robich, R. M., Rinehart, J. P. & Denlinger, D. L. Upregulation of two actin genes and redistribution of actin during diapause and cold stress in the northern house mosquito, Culex pipiens. Journal of insect physiology 52, 1226–1233 (2006).
Jones, B. L. et al. Conservation of estrogen receptor function in invertebrate reproduction. BMC evolutionary biology 17, 65 (2017).
Zhang, T. Y. & Xu, W. H. Identification and characterization of a POU transcription factor in the cotton bollworm, Helicoverpa armigera. BMC molecular biology 10, 25 (2009).
Tu, Z. et al. Speedy A–Cdk2 binding mediates initial telomere–nuclear envelope attachment during meiotic prophase I independent of Cdk2 activation. Proceedings of the National Academy of Sciences 114, 592–597 (2017).
Kume, S., Endo, T., Nishimura, Y., Kano, K. & Naito, K. Porcine SPDYA2 (RINGO A2) stimulates CDC2 activity and accelerates meiotic maturation of porcine oocytes. Biology of reproduction 76, 440–447 (2007).
Nakagaki, M., Takei, R., Nagashima, E. & Yaginuma, T. Cell cycles in embryos of the silkworm, Bombyx mori: G 2-arrest at diapause stage. Roux’s archives of developmental biology 200, 223–229 (1991).
Pasternak, J. J. An Introduction to Human Molecular Genetics: Mechanisms of Inherited Diseases. (John Wiley & Sons, Inc Second Edition, 2005).
Mäkinen, H. S., Cano, J. M. & Merilä, J. Identifying footprints of directional and balancing selection in marine and freshwater three-spined stickleback (Gasterosteus aculeatus) populations. Mol. Ecol. 17(15), 3565–3582 (2008).
Guo, B., Li, Z. & Merilä, J. Population genomic evidence for adaptive differentiation in the Baltic Sea herring. Mol. Ecol. 25, 2833–2852 (2016).
Vera, M., Diez-del-Molino, D. & García-Marín, J. Genomic survey provides insights into the evolutionary changes that occurred during European expansion of the invasive mosquitofish (Gambusia holbrooki). Mol. Ecol. 25(5), 1089–1105 (2016).
Fisher, R. A. The Genetical Theory of Natural Selection. (Clarendon Press, 1930).
Clarke, B. C. The evolution of genetic diversity. Proc. R. Soc. B. 205, 453–474 (1979).
Haldane, J. B. S. & Jayakar, S. D. Polymorphism due to selection depending on the composition of a population. Journal of Genetics 58, 318–323 (1963).
Vrijenhoek, R. C. Genetic diversity and fitness in small populations in Conservation genetics (eds Loeschcke V., Jain S. K. & Tomiuk J.) 37–53 (Springer Basel A.G. 1992).
Patten, M. M. & Haig, D. Maintenance or loss of genetic variation under sexual and parental antagonism at a sex-linked locus. Evolution 63, 2888–2895 (2009).
Cousyn, C. et al. Rapid, local adaptation of zooplankton behavior to changes in predation pressure in the absence of neutral genetic changes. Proc. Natl. Acad. Sci. USA 98, 6256–60 (2001).
Gómez, A., Adcock, G. J., Lunt, D. H. & Carvalho, G. R. The interplay between colonization history and gene flow in passively dispersing zooplankton: microsatellite analysis of rotifer resting egg banks. J. Evol. Biol. 15, 158–171 (2002).
Montero-Pau, J., Serra, M. & Gómez, A. Diapausing egg banks, lake size, and genetic diversity in the rotifer Brachionus plicatilis Müller (Rotifera, Monogononta). Hydrobiologia 796, 77–91 (2016).
Montero-Pau, J. & Serra, M. Life-cycle switching and coexistence of species with no niche differentiation. PLoS ONE 6(6), e20314 (2011).
Rodriguez-Puebla, C., Encinas, A. H., Nieto, S. & Garmendia, J. Spatial and temporal patterns of annual precipitation variability over the Iberian peninsula. Int. J. Climatol. 18, 299–316 (1998).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30(15), 2114–2120 (2014).
Heo, Y., Wu, X., Chen, D., Ma, J. & Hwu, W. M. BLESS: bloom filter-based error correction solution for high-throughput sequencing reads. Bioinformatics 30(10), 1354–1362 (2014).
Edgar, R. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26(19), 2460–2461 (2010).
Deorowicz, S., Kokot, M., Grabowski, S. & Debudaj-Grabysz, A. KMC 2: fast and resource-frugal k-mer counting. Bioinformatics 31(10), 1569–1576 (2015).
Vurture, G. W. et al. GenomeScope: fast reference-free genome profiling from short reads. Bioinformatics 33(14), 2202–2204 (2017).
Kumar, S., Jones, M., Koutsovoulos, G., Clarke, M. & Blaxter, M. Blobology: exploring raw genome data for contaminants, symbionts and parasites using taxon-annotated GC-coverage plots. Front. Genet. 4, 237 (2013).
Laetsch, D. R. & Blaxter, M. L. BlobTools: Interrogation of genome assemblies. F1000 Res. 6, 1287 (2017).
Parra, G., Bradnam, K., Ning, Z., Keane, T. & Korf, I. Assessing the gene space in draft genomes. Nucleic Acids Res. 37, 289–297 (2009).
Bradnam, K. R. et al. Assemblathon 2: evaluating de novo methods of genome assembly in three vertebrate species. GigaScience 2, 10 (2013).
Lomsadze, A., Ter-Hovhannisyan, V., Chernoff, Y. O. & Borodovsky, M. Gene identification in novel eukaryotic genomes by self-training algorithm. Nucleic Acids Res. 33, 6494–6506 (2005).
Cantarel, B. L. et al. MAKER: An easy-to-use annotation pipeline designed for emerging model organism genomes. Genome Res. 18, 188–19 (2008).
Korf, I. Gene finding in novel genomes. BMC Bioinformatics 5, 59 (2004).
Smit, A., Hubley, R. & Green, P. RepeatMasker Open-3.0 http://www.repeatmasker.org (1996–2010).
Jones, P. et al. InterProScan 5: Genome-scale protein function classification. Bioinformatics 30, 1236–1240 (2014).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Conesa, A. et al. Blast2GO: A universal annotation and visualization tool in functional genomics research. Bioinformatics 21, 3674–3676 (2005).
Glaubitz, J. C. et al. TASSEL-GBS: A high capacity genotyping by sequencing analysis pipeline. PLoS ONE 9, e90346 (2014).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2010).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Jombart, T. Adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24, 1403–1405 (2008).
R Development Core Team, R. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna (2009).
Goudet, J. HIERFSTAT, a package for R to compute and test hierarchical F-statistics. Mol. Ecol. Notes 5, 184–186 (2005).
Jensen, J. L., Bohonak, A. J. & Kelley, S. T. Isolation by distance, web service. BMC Genet. 6, 13 (2005).
Purcell, S. et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Lischer, H. E. L. & Excoffier, L. PGDSpider: An automated data conversion tool for connecting population genetics and genomics programs. Bioinformatics 28, 298–299 (2012).
Foll, M. & Gaggiotti, O. A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: A Bayesian perspective. Genetics 180, 977–993 (2008).
Coop, G., Witonsky, D., Di Rienzo, A. & Pritchard, J. K. Using environmental correlations to identify loci underlying local adaptation. Genetics 185, 1411–1423 (2010).
Hahn, C., Genner, M. J., Turner, G. F. & Joyce, D. A. The genomic basis of cichlid fish adaptation within the deepwater “twilight zone” of Lake Malawi. Evol. Lett. 1(4), 184–198 (2017).
Hancock, A. M. et al. Adaptation to climate across the Arabidopsis thaliana genome. Science 334(6052), 83–86 (2011).
Aulchenko, Y. S., de Koning, D. J. & Haley, C. Genomewide rapid association using mixed model and regression: a fast and simple method for genomewide pedigree-based quantitative trait loci association analysis. Genetics 177, 577–585 (2007).
Price, A. L. et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat. Genet. 38(8), 904–909 (2006).
Quinlan, A. R. & Hall, I. M. BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Acknowledgements
We thank Luisa Orsini and Francesc Mesquita for their valuable comments about this research article. We also thank Ana Hidalgo for her technical assistance. We thank Robert Hammond for providing bioinformatic support. We thank James Kitson for library preparation and sequencing of the rotifer genome. We acknowledge the Viper High Performance Computing facility of the University of Hull and its support team. This study was supported by the Spanish Plan Nacional de Investigación Científica, Desarrollo e Innovación Tecnológica (I + D + I) from the Spanish Ministry of Economy and Competitiveness grant CGL2012-30779 to MJC (co-financed by FEDER funds, European Union). L.F. was supported by a predoctoral contract (PREDOC13-110502) and two fellowships (EPREDOC15-275447 and EPREDOC16-384721) from the Universitat de Valencia. A.G. was supported through a HEIF (Higher Education Innovation Fund) for research investment by the University of Hull.
Author information
Authors and Affiliations
Contributions
The DNA extractions were carried out by L.F.-G. The bioinformatic data analysis was performed by L.F.-G. with the support of C.H. and A.G. The first draft of the manuscript was written by A.G. and L.F.-G. All authors discussed the results, contributed to subsequent manuscript drafts and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Franch-Gras, L., Hahn, C., García-Roger, E.M. et al. Genomic signatures of local adaptation to the degree of environmental predictability in rotifers. Sci Rep 8, 16051 (2018). https://doi.org/10.1038/s41598-018-34188-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34188-y
Keywords
This article is cited by
-
Bdelloid rotifers deploy horizontally acquired biosynthetic genes against a fungal pathogen
Nature Communications (2024)
-
Development of reproductive barriers in sympatry
Hydrobiologia (2024)
-
Nuclear genome annotation of wheel animals and thorny-headed worms: inferences about the last common ancestor of Syndermata (Rotifera s.l.)
Hydrobiologia (2024)
-
Efficient RNA interference method by feeding in Brachionus plicatilis (Rotifera)
Biotechnology Letters (2024)
-
Transgenerational expression profiles of a sex related and an epigenetic control gene in the rotifer Brachionus plicatilis in relation to environmental predictability
Hydrobiologia (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.