Abstract
Inherited retinal diseases (IRD) are a heterogeneous group of diseases that mainly affect the retina; more than 250 genes have been linked to the disease and more than 20 different clinical phenotypes have been described. This heterogeneity both at the clinical and genetic levels complicates the identification of causative mutations. Therefore, a detailed genetic characterization is important for genetic counselling and decisions regarding treatment. In this study, we developed a method consisting on pooled targeted next generation sequencing (NGS) that we applied to 316 eye disease related genes, followed by High Resolution Melting and copy number variation analysis. DNA from 115 unrelated test samples was pooled and samples with known mutations were used as positive controls to assess the sensitivity of our approach. Causal mutations for IRDs were found in 36 patients achieving a detection rate of 31.3%. Overall, 49 likely causative mutations were identified in characterized patients, 14 of which were first described in this study (28.6%). Our study shows that this new approach is a cost-effective tool for detection of causative mutations in patients with inherited retinopathies.
Similar content being viewed by others
Introduction
Inherited retinal dystrophies (IRDs) are a group of heterogeneous diseases responsible for different clinically distinctive phenotypes. The most common IRD is Retinitis Pigmentosa (RP) with a prevalence of 1 in 3500 people. RP starts with night blindness and is followed by progressive loss of peripheral vision, leading to loss of central vision and blindness in most advanced cases. Although RP is clinically distinct from other IRDs, advanced stage of RP can be difficult to distinguish from other IRDs, including cone-rod or macular dystrophies1. Moreover, in some cases, clinical manifestations can differ among members of the same family. IRDs can be inherited in different traits including autosomal dominant (adRP), autosomal recessive (arRP) or X-linked (XlRP). The rate of inheritance has varied across populations studied. To date, over 250 genes have been related to various IRDs and some of them are responsible for the different phenotypes observed2 (https://sph.uth.edu/retnet/sum-dis.htm, 3 July 2017).
Since the publication of the first draft of the human genome in 20013,4, we have seen an unprecedented flourishing of sequencing technologies that provide genomic information in an accurate, fast and cost-efficient way. Methods of massive parallel sequencing such as targeted Next Generation Sequencing technologies (NGS) and Whole Exome Sequencing (WES) are the most widely used methods for the diagnosis of IRD. These methods have contributed to an exponential reduction in time and costs for the execution of the sequencing5,6. Nevertheless, the use of whole genome sequencing for diagnostic purposes is limited, mainly by the amount of data generated, which demands high degree of expertise in terms of big data handling and interpretation of the results, and these factors complicate its transfer to the clinicians and to the patients. Comprehensive sequencing of the coding regions of all genes (Whole Exome Sequencing or WES) is more affordable, but still has high technical requirements that are an obstacle to its use as a diagnostic method in routine clinical practice. A more practical approach for clinical diagnosis may consist of an initial genetic screening of a subset of genes associated with a phenotype using targeted NGS, followed by a second more extensive genome analysis, such as WES6, and the analysis of the copy number variations (CNVs)1, for challenging cases for which the first strategy fails to indicate any genetic explanation.
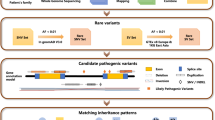
In this study, we sequenced 316 genes associated with IRDs including several syndromic retinopathies. Targeted NGS typically involves a DNA-barcode labelling of each of the individuals to be sequenced for genotyping purposes, this processing being a bottleneck process in terms of consumables, equipment and human resources. In order to simplify the sequencing process and to reduce the costs associated with individual labelling of DNA samples, we have developed a mutation detection approach based on targeted NGS in combination with high resolution melting (HRM) analysis. NGS was performed using pools of 16 DNA samples per pool, and identification of the sample/s carrying the mutation/s was performed using HRM analysis in individual samples, which allowed us to link mutations found in the pooled DNA samples to the DNA from individual patients. We sequenced samples from a total of 115 unrelated patients and 13 controls, 5 of which corresponded to samples from patients with IRD characterized by a third party laboratory. Information regarding mutations in these five controls was not revealed to us until completion of our analysis, to further test the sensitivity of our method in an objective way.
For those samples with negative results after the sequencing process, we used multiplex ligation-dependent probe amplification (MLPA) method for CNV analysis. After combining our sequencing strategy with MLPA, we were able to conclusively identify mutations in 36 patients, meaning that a genetic diagnosis rate was obtained in 31.3% of cases.
Results
Targeted Sequencing
A total of 316 genes (Supplementary Table S1) divided into 7222 amplicons were analysed. A total of 2864 and 3350 genetic variants were found in the 4 and 8 sample pools, respectively, while 3997 +/− 58 variants found in the 7 pools with 16 samples. Mean and median read depth obtained per sample were 196X and 193X, respectively. Less than 3.4% of targeted regions were covered less than 30X per pool, which we established as the cut off.
Sensitivity
In order to assess the sensitivity of our method we performed two independent experiments. In the first experiment, we included a set of 3 pools all containing an increasing number of control samples prepared from DNA from 16 patients (see methodology section and Supplementary Fig. S1 for a more detailed description). Each control sample carried at least one mutation that had been previously validated by Sanger sequencing (see methodology section). As a result, previously characterized mutations from all control samples were identified in the first set of samples, regardless of the size of the pool.
Following our method, one would expect a relative level of coverage of 1/32 in heterozygous variants and 2/32 in one homozygous or in two heterozygous variants. However, we found that the number did not fit exactly to these values when analysing variants among solved patients (see variants in Table 1). Thus, in heterozygous variants the relative coverage ranged between 0.56 to 1.54/32 with 5 outliers with relative coverage of 1.75/32, 1.88/32, 1.99/32, 1.93/32 and 2/32, with values more suggestive of mutations present in two alleles rather than in one.
With respect to variants expected to be in two alleles (in homozygosis in one patient or in heterozygosis in two patients), the relative coverage ranged between 1.5–2.3/32. In this case we found 4 outliers with relative levels of coverage as low as 1.25/32 (2 cases), or as high as 2.98/32 and 3.13/32. In all cases with a higher relative coverage, in relation with the number of alleles found, all the pool was Sanger sequenced individually, in order to test for the presence of another allele with that variant and we found that there were no more alleles with the mutation among the pool.
Moreover, we tested 9 SNPs with higher MAFs in order to assess if the relative level of coverage was the same in the case of having more alleles with a specific SNP within the pool. All 16 samples from the pool in which the SNP was found, were directly Sanger sequenced. Similarly to what we observed in the candidate variants, we found some variability between expected vs. sequenced SNPs, with a slight mismatch of the variants present according to expected values (Supplementary Table S2).
Variant Identification
Once we established 16 as the most cost-effective sample size, we sequenced 7 pools of 16 samples/each, including a set of 19 different controls carrying a total of 21 previously detected rare (MAF < 0.003), non-causative variants (control variants). All variants selected had a MAF < 0.003 for genes mainly associated with a recessive inheritance pattern and were absent from the databases in the case of genes associated with a dominant inheritance pattern (Supplementary Table S3). As a result, all 21 control variants were also redetected. In both sets of experiments our methodology yielded 100% sensitivity.
Furthermore, we included five samples from patients with IRD provided by a third party laboratory. As information about mutations within these samples was not initially disclosed to us, we were able to use these samples as an additional way to test the sensitivity of our method. We succeeded in identifying causal mutations in all of the samples. These were: a homozygous mutation c.1645G>T (p.Glu549Ter) in the BBS1 gene; c.1040C>A (p.Pro347Gln) mutation in the RHO gene; c.1703TA (p.Leu568Ter) mutation in the CHM gene; c.2888_2888del (p.Gly963fs) and c.3386G>T (p.Arg1129Leu) mutations in the ABCA4 gene and a homozygous mutation, c.397C>T (p.His133Tyr) in MYO7A gene.
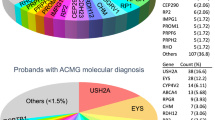
With regard to the 115 unrelated patients analysed, disease causing mutations were found in at least one allele in 61 patients. Nevertheless, since in some patients, mutations were found only in one allele in recessive genes, causal mutations were found in 36 patients, reaching a detection rate of 31.3% (Tables 1, 2 and Supplementary Fig. S2). Most of the pathogenic mutations were found in the USH2A gene, although in many cases only in one allele without a second mutation, and therefore in these recessive cases, we could not determine the causal mutation. Among all mutations found in characterized patients, 15 were novel, 2 missense and 13 loss-of-function (LOF) mutations. Novel missense and splicing variant mutations were potentially pathogenic, this being inferred from the score obtained from different in-silico tools and the fact that they co-segregated with the disease (Supplementary Table S4).
Regarding the distribution of mutations among our cohort of patients, most findings were found among the following five genes:
USH2A
Mutations within this gene were responsible for most cases of arRP in our cohort. Most of the patients were carriers of biallelic mutations. Compound heterozygous mutations are frequently reported in this gene7,8. Four of the mutations found in USH2A were novel: c.11241C>G, in patient RP15, c.3669del in patient RP91, c.1570G>A in patient RP109 and c.14565del in patient RP180. Except for patient RP180, homozygote carrier of the mutation, the rest of the patients were carriers of mutations in compound heterozygosis with the previously reported pathogenic mutations c.12093del, c.11754G>A and c.2276G>T respectively (Table 1).
CERKL
This was the second most commonly mutated gene in our cohort. We characterized 5 patients with the same mutation c.847C>T in this gene. In 4 of the cases it was in homozygosis and in one case it was in compound heterozygosis with c.356G>A mutation. This nonsense mutation is relatively common in Spanish cohorts9,10.
EYS
This was the third most commonly mutated gene in our cohort. Three out of four patients shared mutations, such as RP1 and RP117 with c.9405T>A11 and RP49 and RP117 with c.4045T>A12, probably indicating the sharing of a common ancestor. This finding is consistent with previous studies involving Spanish cohorts, in which EYS was one of the most commonly mutated genes in recessive retinitis pigmentosa13,14. In addition, we found three novel mutations in this gene: two frameshift mutations in compound heterozygosis c.1830del in patient RP1 and c.888del in patient RP106; and a nonsense mutation also in compound heterozygosis c.14C>A, in patient RP106.
RPGR
We were able to detect a novel mutation c.2232_2235del in patient RP27 in the ORF15 region of this gene. Mutations in this region are challenging to amplify due to a large segment of highly repetitive purine-rich sequences15. Nevertheless, the high coverage of this region we obtained using our pooled-based approach, allowed us to detect this variant (Supplementary Fig. S3).
Variants of Uncertain Significance (VUS)
For the family RP92, two heterozygous variants were observed in PCDH15 and CDH23. Despite the fact that this digenic inheritance pattern has previously been found to be causative of Usher Syndrome16, and that the variants segregated correctly within our family, there is some controversy with the pathogenicity of this digenism and, as far as we know, the CDH23 and PCDH15 digenism has been only reported in one study16. Despite cochlear degeneration specific to hair cells was observed in this type of mice, USH mutant mice do not display visual defects. Based on ultrastructural analyses, it has been shown that the USH1 proteins localize at the level of microvilli-like structures, called calyceal processes, which form a collar around the base of photoreceptor outer segments. These structures have only been found in primate and other large mammals, but not in mouse photoreceptor cells17. This has led to propose that the absence of these structures in the mouse retina is responsible for the lack of a visual phenotype in mouse models of Usher syndrome. Regardless of this structural difference, we cannot confirm that this digenism is the causative mutation.
In the case of family RP148, a novel missense mutation c.6835T>G was found in PRPF8 gene. The mutation was predicted to be damaging by at least 5 in silico predictors. Nevertheless, given the lack of a complete segregation analysis due to the unavailability of many of the samples required, we were unable to conclude that c.6835T>G is the causal adRP mutation in this family. Similarly, in family RP181, we found a novel nonsense mutation, c.1165C>T, in PRPF31 gene. However we were not able to validate this finding in a segregation analysis due to a lack of samples available. In fact, the only family sample we were able to study was a non-affected sister who was also a mutation carrier.
Multiplex Ligation-dependent Probe Amplification (MLPA)
Among the 32 families analysed by this method, we detected a large deletion in the PRPF31 gene expanding from exon 9 to 13 in family RP40, previously unreported. The deletion was also detected in an affected grandmother and the asymptomatic mother. Confirmation of the deletion region was performed sequencing the deleted DNA fragment (Fig. 1A).
Novel deletion in PRPF31. (A) Electropherogram showing a reduced dosage of exons 9–13 (arrows) in patient RP40. (B) Schematic representation of PRPF31 deletions described in the literature, and the deletion of exons 9–13 we found in this study, represented by the red bar. Abbreviations: P: control probes; Ex: Exon.
Discussion
In the present work, we have developed a cost-effective method for the diagnosis of IRDs based on pooled genomic DNA targeted NGS, in combination with HRM as a highly sensitive, versatile and affordable genotyping method. Following our methodology, we were able to find the causal mutation in 36 of our patients (31.3%) (Table 1).
Several studies have validated the feasibility of DNA sequencing pools to identify and quantify the genetic variants or single nucleotide polymorphisms (SNPs) in small genomes or small genomic regions of prokaryotes18; and single human genes19,20. Previous studies tested experimentally the accuracy in re-sequencing pools of strains of highly isogenic D. melanogaster, whose genome had been previously sequenced individually. They showed that the sequenced pool provides a correct estimate of the population allele frequency, enabling the discovery of new SNPs with a low rate of false positives21.
Regarding clinical applications22 evaluated the use of pooled DNA sequencing to accurately assess allele frequencies on transmitted and non-transmitted chromosomes in a set of families in an allelic association study23 combined DNA samples from 1,111 individuals and sequenced 4 genes to identify rare germline variants. The main bottleneck in the use of a pooling strategy for genetic studies is related to the challenges of detecting rare and low-frequency variants reliably, allowing an accurate estimation of MAFs24. Moreover, pooled DNA sequencing was applied for the analysis of 3 genes of Gitelman’s syndrome using semiconductor NGS in pooled DNA samples from 20 patients25. In a more recent study, 72 genes were analysed in pools consisting of samples from 12 individuals26. With respect to RP, pooled DNA NGS was used to search for mutations in the SNRNP200 gene in a cohort of 96 unrelated patients from North America27. Pooled DNA sequencing has recently been used for population genetics studies (GWAS), in several different pathologies28.
Compared to previous studies that limited to the sequencing of a restricted number of genes, this represents the first study based on the pooled sequencing of more than 300 genes. To estimate the reduction in costs derived from the use of our methodology we compared the costs per patient of our pooled method with an individual sequencing approach. The main source of cost savings was related to expenses involved in the preparation of DNA libraries. Specifically, there was a 10.6-fold reduction in sequencing costs with our methodology. Once we added costs associated with the HRM analysis-based genotyping method, the overall reduction in mutation detection/patient was 6.25-fold.
The choice of 16-sample pools was based, not only on terms of sensitivity, but also on the optimal number of samples for further analysis by HRM, which we found to be around 16 in a previous study29. One of the main advantages over previous pooled-NGS-based strategies for mutations detection is the genotyping method we used. HRM analysis is significantly more affordable than other methods including TaqMan probes (Thermo Fisher Scientific) especially if used for a large cohort of patients and/or for a large number of genes30; or DNA arrays Sequenom IPLEX (CD Genomics), which requires specific equipment, making the applicability of the methodology highly dependent on the equipment available in each laboratory30.
In order to test the sensitivity of our method we included a set of positive controls. Five of these positive controls were samples from IRD patients previously diagnosed elsewhere, for whom we only had access to their clinical data, but not to information on the causative mutations. Given that we obtained a sensitivity of 100%, the fact that our detection rate is not as high as in previous studies, ranging from 51 to 66%31,32,33,34, might be explained, at least in part, by the nature of the cohort of patients included in our study, since over half of our cohort of patients (69/115) were analysed in previous studies with no results, using a repertoire of different approaches9,29,35.
Therefore, we believe that the great number of samples analysed in previous studies is the main factor for the relative low yield obtained. A similar observation was recently reported, where they found that the patients who were screened for the first time had a higher pathogenic variant detection rate than the overall rate, suggesting that their cohort was enriched for intractable cases giving a lower detection rate36.
Another possibility is that the detection rate varies depending on the ethnicity of the individuals analysed36. In this regard, they reported a lower rate of homozygous variants detected in individuals of European origin, comparing with other populations, in recessive transmitted diseases36. Similarly, we found heterozygous mutations in recessive genes in 25 patients, which therefore cannot be regarded as the causal mutation on their own. One possibility is that a fraction of our patients might be bearing large DNA re-arrangements, or mutations in deep intronic regions not covered by our approach, which would act in compound heterozygosis.
One limitation of the approach used in this work was that the relative level of coverage expected in validated variants (1/32 in heterozygous variants and 2/32 in one homozygous or in two heterozygous variants) did not fit exactly to expected values in some cases (see Results section and supplementary Table S2). This could be due to the fact that there is a pre-amplification step for library preparation. Despite great care was taken for preparing the pools using equimolar amounts of each DNA sample, we cannot discard the possibility of having some samples over or under-represented, offering higher or lower relative values, respectively. This might be reflecting an unequal sample bias, or that all DNAs of each pool were not amplified in all regions, which might be one of the potential explanations for the relative low diagnostic yield. However, we consider this possibility unlikely, considering that we were able to detect all control variants introduced in each pool.
Another limitation of pooled sequencing method is related to the lack of use of multiplex barcodes, which complicates CNV detection using NGS technology37.
There is increasing evidence of genomic rearrangements resulting in CNVs responsible for IRDs in several genes including PRPF3138; EYS39; USH2A40 and X-linked RPGR and CHM41,42. Several recent studies have emphasized the importance of CNV analysis in IRD cases. For instance, Bujakowska et al.43 found mutations in 5 out of 28 IRD cases in SNRNP200, PRPF31, EYS and OPN1LW genes. Khateb et al.44; identified rearrangements in 6 IRD patients out of 60 involving EYS, MYO7A, NPHP4, RPGR and CHM. This last case CHM was deleted in conjunction with other 6 genes. Van Cauwenbergh et al., 20161 identified CNV in 3 patients out of 57 analysed, with mutations in USH2A, HGSNAT and RCBTB1 genes. Interestingly, a recent paper has established a ranking of IRD genes according to genomic features and CNV occurrence. These authors recommend performing routinely a targeted CNV screening in the most prevalent 30 top-ranked IRD genes according to their genomic length45.
Despite some authors have described the use of read depth methods for pooled multiple sequencing46, we decided to select a group of 9 genes, most of which known to be prone to CNV formation45 using MLPA. We analysed several patients with negative results after the sequencing of the 316 IRD genes, and we included some of the genes reported as the main contributors to CNV in different studies, such as USH2A, EYS, CHM, PRPF31 and RPGR1,38,43,44,47,48.
Using this approach, we were able to diagnose a patient with a deletion expanding from exon 9 to 13 in PRPF31. Rearrangements in this gene have been described to account for around 2.5% in autosomal dominant cases38. Although different mutated regions have been described in PRPF31, the deletion of exons 9 to 13 has not been described before (Fig. 1B).
The pattern of inheritance in family 40 is suggestive of an autosomal dominant pattern with incomplete penetrance. Segregation analysis was conducted in two family members, revealing the presence of an obligate carrier. Mutations in PRPF31 have been mostly associated with cases of incomplete penetrance49,50,51.
A limitation inherent to the technique employed, which is shared by WES, is the impossibility of finding mutations in deep intronic regions, not covered by the primer design. In this regard, in an attempt to find the second mutant allele, we analysed two commonly reported deep intronic mutations: c.2991+1655A>G in CEP29052 and c.7595−2144A>G in USH2A genes53,54, in patients with heterozygous mutations in those genes. We did not however, find the mutations that were likely causative of the disease within these regions.
Despite limitations inherent to NGS sequencing regarding its performance in repetitive or CG-rich regions of the genome, we were able to detect the mutation c.2232_2235del in ORF15 of the RPGR gene, a region regarded as challenging, with a poor sequencing performance, both in panel based NGS and Whole exome sequencing15. Using our methodology we were able to detect this mutation among one of the 16 samples of the pool, which further support the validity of our method in terms of sequencing capacity, genotyping and filtering methods (Supplementary Fig. S3).
Regarding the mutations found, USH2A represents the most commonly mutated gene within our cohort of patients, with eleven different mutations found in this gene in seven patients characterized. Among USH2 genes, USH2A is the most commonly mutated gene and it is responsible for approximately 74–90% of USH2 cases8,55,56. Mutations in USH2A, are responsible for Usher syndrome type 2 and non-syndromic RP57. CERKL and EYS are the next most commonly mutated genes in our cohort, which is also in accordance with previous studies58,59. In case of mutations in EYS genes, high prevalence has also been observed among Spanish population14, Americans with European origin13 and among Japanese populations60.
For those patients for whom we failed to identify putative disease-causing mutations, the use of alternative approaches will hopefully succeed in characterizing their disease, at the molecular level. For instance, WES aimed at the identification of mutations in genes not currently linked to IRDs; aCGH arrays for the analysis of CNVa in other genes or regions not covered by our MLPA analysis; or whole genome sequencing to extend the analysis to the 99% of non-coding DNA. Despite being highly dependent on technical support, the use of whole genome sequencing is gaining momentum in clinical practice, and it seems plausible that it will become feasible in a near future, once a robust translational genomics workflow becomes an affordable option both in economic and technical terms, to allow feedback of potentially diagnostic findings to clinicians and research participants61.
Materials and Methods
Study subjects
IRD patients were clinically diagnosed by the Ophthalmology Service at Donostia University Hospital, San Sebastian, Spain. Most patients studied had been given a diagnosis of retinitis pigmentosa, though a few patients with an undetermined inherited retinal dystrophy (IRD) were also included, based on pedigrees and clinical criteria. The inclusion criteria used were night blindness, peripheral visual field loss, pigmentary deposits resembling bone spicules, retinal vessels attenuation, optic disc pallor and reduced rod and cone response amplitudes and a delay in their timing in the electroretinogram (Hartong, 2006). A total of 115 probands were selected. In addition, samples from 13 patients were included as characterized control patients. This control group was composed of 8/13 samples selected from our cohort of IRD patients with mutations identified in previous studies9,29,35 and a further 5 control samples from IRD patients characterized by a third party laboratory, (those for which we were blinded to information regarding mutations until we had completed our analysis). Family pedigrees were generated from information obtained from probands. All procedures performed in studies involving human participants received approval from the ethical standards of the Clinical Research Ethics Committee of the Basque Country, Spain (CEIC-E) and were in accordance with the 2013 Helsinki declaration or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Human sample collection
High molecular weight DNA was extracted from blood samples from RP patients and their available family members. Total DNA from samples was extracted and isolated with the AutoGenFlex Star instrument (AutoGen, Holliston, MA, USA) using the FlexiGene DNA Kit (Qiagen, Hilden, Germany) following the manufacturer´s instructions. DNA concentrations were measured on the Qubit fluorometer using Quant-iT PicoGreen reagent (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA). Equimolar amounts of DNA samples were pooled (100 ng/ul per sample). For a detailed description of the procedure see29.
Pooled sequencing
In order to assess the sensitivity and cost-effectiveness of our method we performed a first experiment to compare the yield obtained after sequencing pools with increasing number of DNA samples and we estimated the differences in costs involved in individual vs. pooled sequencing. All pools were made up from samples from carriers of low-frequency variants, which corresponded to either causal, variants of uncertain significance (VUS) or non-pathogenic variants identified in previous studies9,29,35. A total of 13 control samples were used in 3 sets of pools, with 4, 8 and 16 control samples in each. Of these control samples, 9 carried pathogenic variants (one provided by a third party laboratory), while 7 carried low frequency variants with a minor allele frequency (MAF) <0.003, and therefore we used these 7 samples both as controls and as test samples. Samples were prepared as follows: An initial pool of 4 samples was generated. This pool was used to generate the 3 pools, adding 0, 4 or 8 more samples to generate the pools with 4, 8 and 16 samples, respectively (Supplementary Fig. S1A and Supplementary Table S3A).
In order to further test the sensitivity of our method and to detect possible differences in the sequencing yield, inherent to each sequencing run, we conducted a complementary experiment. For this, we used a different set of controls, all from carriers of low-frequency, non-disease causing variants or individuals with recessive phenotypes with disease causing mutations present in only one allele. In this case, out of 115 patients analysed, a total of 108 test samples were interrogated: 16/108 corresponded to carriers of a total of 21 previously detected non disease causing variants with low MAF (<0.003) and were, therefore, used as both control and test samples (Supplementary Table S3B). 53/108 samples corresponded to patients that had been interrogated previously with negative results, and 39/108 corresponded to new samples interrogated in this study for the first time. As additional controls we used four samples from carriers of disease causing mutations provided by a third party laboratory (for which we were blinded to mutation-related information until after our analysis). For this experiment, patients were divided into 7 pools with 16 samples each. Control samples were distributed among each pool such as that each pool contained at least 2 control samples, and 4/7 pools had also control from a third party laboratory (Supplementary Fig. S1B).
Amplicon Library preparation
Ion AmpliSeq Library Preparation Kit v2.0 (Thermo Fisher Scientific) was used to construct an amplicon library from genomic target regions with a maximum read length of approximately 200 base pairs (average length, 142 bp) for shotgun sequencing on an Ion Proton system (Thermo Fisher Scientific). Briefly, target genomic regions were amplified by simple PCR using Ion Ampliseq primer pools and 10 ng of each DNA samples.
Sequencing Analysis
Ion Proton Sequencing
NGS was carried out on the Ion Proton system (Thermo Fisher Scientific). Briefly, enriched ion sphere particles (ISPs) were annealed with the sequencing primer and mixed with the sequencing polymerase from the Ion PGM_200 Sequencing Kit (Thermo Fisher Scientific). Then, the polymerase-bound and primer-activated ISPs were loaded into the previously checked and washed Ion PI Chips (Life Technologies) and having planned the run on the Ion Proton System software, chips were subjected to 500 cycles of sequencing with the standard nucleotide flow order. Signal processing and base calling of data generated from the Ion Proton runs were performed with the Ion Torrent platform-specific analysis software (Torrent Suite version 4.0).
Variant calling
Using the Ion Reporter software we performed the variant calling. First of all GRCh37/hg19 was used as reference genome and alignment was performed against a bed file containing all regions corresponding to 316 genes sequenced. A key aspect in our mutation detection pipeline was to take into consideration the dilution effect of each variant due to our pooled sequencing approach. Therefore we used the pipeline provided by the ion reporter program for the detection of somatic mutations with minor modifications. We used a somatic mutation detection approach, since this is the most suited for the detection of variants represented in very low frequency (1 in 32 alleles, in the lowest case). The only modification to the default parameters provided by the ion reporter program (5.0 version) consisted on the switch of 10 parameters within the Variant Filtering section in Parameters tab. All parameters are described in detail in Supplementary Table S5. Finally, a Variant Caller File (VCF) was generated.
Genotyping by high resolution melting (HRM) analysis
Likely disease causing variants from each pool of 16 samples were selected from the VCF. Specific primers were designed to perform a HRM analysis generating amplicons ranging between 250 to 330 bp in length, in order to cover the mutation position. HRM analysis was used to identify which sample/s among 16 in the pool carried the mutation. We followed the methodology described in29, with minor modification. Briefly, PCR amplification and HRM were performed in a single run on a 7900HT Fast Real-Time PCR System in 384-well plates (Applied Biosystems), each plate contained individual samples (in triplicates) from the 16 probands of the pool in which the variant was detected. We analysed up to 7 different variants in parallel in a single run. After HRM run, the analysis of post amplification fluorescent melting curves was performed using the HRM V2.0.1 software (ThermoFisher Scientific). Melting curves were normalized and difference plots were generated to compare the samples. Only samples showing a different melting curve (Fig. 2) were Sanger sequenced.
HRM analysis of TULP1 gene. Difference plot shows c.1495+1G>C mutation in TULP1 gene, with 2 out of 16 samples that clearly differ from the non-carrier samples (grey lines). Sanger sequencing confirmed the presence of the mutation c.1495+1G>C in two patients, one in heterozygosis (blue lines) and the other one in homozygosis (red lines). Note that samples are in triplicates.
Sanger sequencing
Sanger sequencing was used to confirm those mutations detected by NGS and for co-segregation analysis using a 16-capillary ABI 3130xl platform (Applied Biosystems, Foster City, CA, USA) according to manufacturer´s protocol. Sequences were analysed and compared with wild-type samples and a reference sequences using BioEdit software (Ibis Biosciences, Carlsbad, CA, USA) and Ensembl and NCBI databases.
Relevant variant priorization and pathogenicity score
In order to determine genomic variants of relevance, we selected the potential disease causing variants according to the following pre-established criteria:
-
(1)
Variants previously reported as pathogenic.
-
(2)
Variants with a MAF <0.001 for dominant genes or MAF <0.003 for recessive genes obtained from genome aggregation database (gnomAD).
-
(3)
Novel Splicing variants and loss-of-function variants such as nonsense mutations, frameshift deletions or insertions.
-
(4)
Previously reported missense variants with pathogenicity scores assessed by in silico predictive software.
-
(5)
Novel missense variants predicted to be damaging by in-silico predictive software (as mentioned below).
Presence for all candidate variants was checked using the Spanish Variant Server Database (CSVS), (http://csvs.babelomics.org/)62. For dominant variants, only those absent from this database were considered further. With regard to recessive variants, only those variants with a MAF lower than 0.003 and only present in heterozygosis were considered further.
Multiplex Ligation-dependent Probe Amplification assay (MLPA)
MLPA was used to search for genomic copy number variations in 32 patients without causative mutations found after sequencing of 316 IRD genes. We selected 9 genes with high prevalence of reported rearrangements38,39,40.
Patients with a dominant inheritance pattern were analysed using MLPA Retinitis Probemix (P235). This probemix contains PRPF31, RHO, RP1 and IMPDH1 genes.
Patients with heterozygotic mutations in USH2A genes or EYS were also analysed for CNVs, in search of the second mutated allele within these genes (Salsa Mixes P361/2 and P328, respectively).
In addition, patients with an X-linked inheritance pattern, clinically diagnosed with choroideremia or families with only males affected, were analysed for RP2, RPGR and CHM genes (Salsa probemix P366).
MLPA reactions were run according to the manufacturer’s general recommendations (MRC-Holland, Amsterdam, Holland) as previously described63. The MLPA reaction products were separated by capillary electrophoresis on Abi Prism 3130XL Analyzer (Applied Biosystems) and the results obtained were analysed by GeneMapper software (Thermo Fisher Scientific).
Pathogenicity predictive software
SIFT (http://www.sift.bii.a-star.edu.sg).
Polyphen2 (http://www.genetics.bwh.harvard.edu/pph2/).
PROVEAN (http://provean.jcvi.org/seq_submit.php)64.
GVGD (agvgd.iarc.fr/agvgd_input_php)65.
MutationTaster (www.mutationtaster.org)66.
Web sources
Ensembl, http://www.ensembl.org/.
NCBI, http://www.ncbi.nlm.nih.gov/.
Polyphen-2, http://www.genetics.bwh.harvard.edu/pph2/.
RetNet, http://www.sph.uth.tmc.edu/Retnet/.
SIFT, http://www.sift.bii.a-star.edu.sg/.
SNPnexus, http://www.snp-nexus.org/.
The Human Genome Variation Society (HGVS), http://www.hgvs.org/.
1000 Genomes, http://www.1000genomes.org/_ENREF_48.
NHLBI Exome Sequencing Project (ESP), http://evs.gs.washington.edu/EVS/.
Babelomics, http://csvs.babelomics.org.
ExacBrowse, http://exac.broadinstitute.org/.
GnomAD browser, http://gnomad.broadinstitute.org/.
Change history
28 May 2019
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Van Cauwenbergh, C. et al. arrEYE: a customized platform for high-resolution copy number analysis of coding and noncoding regions of known and candidate retinal dystrophy genes and retinal noncoding RNAs. Genetics in medicine: official journal of the American College of Medical Genetics 19, 457–466, https://doi.org/10.1038/gim.2016.119 (2017).
Sohocki, M. M. et al. Prevalence of AIPL1 mutations in inherited retinal degenerative disease. Mol Genet Metab 70, 142–150, https://doi.org/10.1006/mgme.2000.3001S1096-7192(00)93001-4 (2000).
Lander, E. S. et al. Initial sequencing and analysis of the human genome. Nature 409, 860–921, https://doi.org/10.1038/35057062 (2001).
Venter, J. C. et al. The sequence of the human genome. Science 291, 1304–1351, https://doi.org/10.1126/science.1058040 (2001).
Ferrari, S. et al. Retinitis pigmentosa: genes and disease mechanisms. Curr Genomics 12, 238–249, https://doi.org/10.2174/138920211795860107CG-12-238 (2011).
Lazaridis, K. N. et al. Outcome of Whole Exome Sequencing for Diagnostic Odyssey Cases of an Individualized Medicine Clinic: The Mayo Clinic Experience. Mayo Clinic proceedings 91, 297–307, https://doi.org/10.1016/j.mayocp.2015.12.018 (2016).
Bonnet, C. et al. An innovative strategy for the molecular diagnosis of Usher syndrome identifies causal biallelic mutations in 93% of European patients. European journal of human genetics: EJHG 24, 1730–1738, https://doi.org/10.1038/ejhg.2016.99 (2016).
Garcia-Garcia, G. et al. Mutational screening of the USH2A gene in Spanish USH patients reveals 23 novel pathogenic mutations. Orphanet journal of rare diseases 6, 65, https://doi.org/10.1186/1750-1172-6-65 (2011).
Barandika, O. et al. A Cost-Effective Mutation Screening Strategy for Inherited Retinal Dystrophies. Ophthalmic research 56, 123–131, https://doi.org/10.1159/000445690 (2016).
Tuson, M., Marfany, G. & Gonzalez-Duarte, R. Mutation of CERKL, a novel human ceramide kinase gene, causes autosomal recessive retinitis pigmentosa (RP26). American journal of human genetics 74, 128–138, https://doi.org/10.1086/381055 (2004).
Collin, R. W. et al. Identification of a 2 Mb human ortholog of Drosophila eyes shut/spacemaker that is mutated in patients with retinitis pigmentosa. American journal of human genetics 83, 594–603, https://doi.org/10.1016/j.ajhg.2008.10.014 (2008).
Eisenberger, T. et al. Increasing the yield in targeted next-generation sequencing by implicating CNV analysis, non-coding exons and the overall variant load: the example of retinal dystrophies. PloS one 8, e78496, https://doi.org/10.1371/journal.pone.0078496 (2013).
Daiger, S. P., Bowne, S. J. & Sullivan, L. S. Perspective on genes and mutations causing retinitis pigmentosa. Archives of ophthalmology 125, 151–158, https://doi.org/10.1001/archopht.125.2.151 (2007).
Barragan, I. et al. Mutation spectrum of EYS in Spanish patients with autosomal recessive retinitis pigmentosa. Human mutation 31, E1772–1800 (2010).
Li, J. et al. Improved Diagnosis of Inherited Retinal Dystrophies by High-Fidelity PCR of ORF15 followed by Next-Generation Sequencing. The Journal of molecular diagnostics: JMD 18, 817–824, https://doi.org/10.1016/j.jmoldx.2016.06.007 (2016).
Zheng, Q. Y. et al. Digenic inheritance of deafness caused by mutations in genes encoding cadherin 23 and protocadherin 15 in mice and humans. Human molecular genetics 14, 103–111, https://doi.org/10.1093/hmg/ddi010 (2005).
Sahly, I. et al. Localization of Usher 1 proteins to the photoreceptor calyceal processes, which are absent from mice. The Journal of cell biology 199, 381–399, https://doi.org/10.1083/jcb.201202012 (2012).
Van Tassell, C. P. et al. SNP discovery and allele frequency estimation by deep sequencing of reduced representation libraries. Nature methods 5, 247–252, https://doi.org/10.1038/nmeth.1185 (2008).
Margraf, R. L. et al. Variant identification in multi-sample pools by illumina genome analyzer sequencing. Journal of biomolecular techniques: JBT 22, 74–84 (2011).
Out, A. A. et al. Deep sequencing to reveal new variants in pooled DNA samples. Human mutation 30, 1703–1712, https://doi.org/10.1002/humu.21122 (2009).
Zhu, Y., Bergland, A. O., Gonzalez, J. & Petrov, D. A. Empirical validation of pooled whole genome population re-sequencing in Drosophila melanogaster. PloS one 7, e41901, https://doi.org/10.1371/journal.pone.0041901 (2012).
Shaw, S. H., Carrasquillo, M. M., Kashuk, C., Puffenberger, E. G. & Chakravarti, A. Allele frequency distributions in pooled DNA samples: applications to mapping complex disease genes. Genome research 8, 111–123 (1998).
Druley, T. E. et al. Quantification of rare allelic variants from pooled genomic DNA. Nature methods 6, 263–265, https://doi.org/10.1038/nmeth.1307 (2009).
Wang, J. et al. Investigation of rare and low-frequency variants using high-throughput sequencing with pooled DNA samples. Scientific reports 6, 33256, https://doi.org/10.1038/srep33256 (2016).
Tavira, B. et al. A labor- and cost-effective non-optical semiconductor (Ion Torrent) next-generation sequencing of the SLC12A3 and CLCNKA/B genes in Gitelman’s syndrome patients. Journal of human genetics 59, 376–380, https://doi.org/10.1038/jhg.2014.37 (2014).
Anand, S. et al. Next Generation Sequencing of Pooled Samples: Guideline for Variants’ Filtering. Scientific reports 6, 33735, https://doi.org/10.1038/srep33735 (2016).
Benaglio, P. et al. Next generation sequencing of pooled samples reveals new SNRNP200 mutations associated with retinitis pigmentosa. Human mutation 32, E2246–2258, https://doi.org/10.1002/humu.21485 (2011).
Jin, S. C., Benitez, B. A., Deming, Y. & Cruchaga, C. Pooled-DNA Sequencing for Elucidating New Genomic Risk Factors, Rare Variants Underlying Alzheimer’s Disease. Methods in molecular biology 1303, 299–314, https://doi.org/10.1007/978-1-4939-2627-5_18 (2016).
Anasagasti, A. et al. Genetic high throughput screening in Retinitis Pigmentosa based on high resolution melting (HRM) analysis. Experimental eye research 116, 386–394 (2013).
Jin, S. C. et al. Pooled-DNA sequencing identifies novel causative variants in PSEN1, GRN and MAPT in a clinical early-onset and familial Alzheimer’s disease Ibero-American cohort. Alzheimer’s research & therapy 4, 34, https://doi.org/10.1186/alzrt137 (2012).
Consugar, M. B. et al. Panel-based genetic diagnostic testing for inherited eye diseases is highly accurate and reproducible, and more sensitive for variant detection, than exome sequencing. Genetics in medicine: official journal of the American College of Medical Genetics 17, 253–261, https://doi.org/10.1038/gim.2014.172 (2015).
Zhang, Q. et al. Next-generation sequencing-based molecular diagnosis of 35 Hispanic retinitis pigmentosa probands. Scientific reports 6, 32792, https://doi.org/10.1038/srep32792 (2016).
Carrigan, M. et al. Panel-Based Population Next-Generation Sequencing for Inherited Retinal Degenerations. Scientific reports 6, 33248, https://doi.org/10.1038/srep33248 (2016).
Tiwari, A. et al. Next generation sequencing based identification of disease-associated mutations in Swiss patients with retinal dystrophies. Scientific reports 6, 28755, https://doi.org/10.1038/srep28755 (2016).
Ezquerra-Inchausti, M. et al. High prevalence of mutations affecting the splicing process in a Spanish cohort with autosomal dominant retinitis pigmentosa. Scientific reports 7, 39652, https://doi.org/10.1038/srep39652 (2017).
Carss, K. J. et al. Comprehensive Rare Variant Analysis via Whole-Genome Sequencing to Determine the Molecular Pathology of Inherited Retinal Disease. American journal of human genetics 100, 75–90, https://doi.org/10.1016/j.ajhg.2016.12.003 (2017).
Marelli, C. et al. Mini-Exome Coupled to Read-Depth Based Copy Number Variation Analysis in Patients with Inherited Ataxias. Human mutation 37, 1340–1353, https://doi.org/10.1002/humu.23063 (2016).
Sullivan, L. S. et al. Genomic rearrangements of the PRPF31 gene account for 2.5% of autosomal dominant retinitis pigmentosa. Investigative ophthalmology & visual science 47, 4579–4588, https://doi.org/10.1167/iovs.06-0440 (2006).
Pieras, J. I. et al. Copy-number variations in EYS: a significant event in the appearance of arRP. Investigative ophthalmology & visual science 52, 5625–5631, https://doi.org/10.1167/iovs.11-7292 (2011).
Steele-Stallard, H. B. et al. Screening for duplications, deletions and a common intronic mutation detects 35% of second mutations in patients with USH2A monoallelic mutations on Sanger sequencing. Orphanet journal of rare diseases 8, 122, https://doi.org/10.1186/1750-1172-8-122 (2013).
Sanchez-Alcudia, R. et al. A Comprehensive Analysis of Choroideremia: From Genetic Characterization to Clinical Practice. PloS one 11, e0151943, https://doi.org/10.1371/journal.pone.0151943 (2016).
Simunovic, M. P. et al. The Spectrum of CHM Gene Mutations in Choroideremia and Their Relationship to Clinical Phenotype. Investigative ophthalmology & visual science 57, 6033–6039, https://doi.org/10.1167/iovs.16-20230 (2016).
Bujakowska, K. M. et al. Copy-number variation is an important contributorto the genetic causality of inherited retinal degenerations. Genetics in medicine: official journal of the American College of Medical Genetics 19, 643–651, https://doi.org/10.1038/gim.2016.158 (2017).
Khateb, S. et al. Identification of genomic deletions causing inherited retinal degenerations by coverage analysis of whole exome sequencing data. Journal of medical genetics 53, 600–607, https://doi.org/10.1136/jmedgenet-2016-103825 (2016).
Van Schil, K. et al. Mapping the genomic landscape of inherited retinal disease genes prioritizes genes prone to coding and noncoding copy-number variations. Genetics in medicine: official journal of the American College of Medical Genetics 20, 202–213, https://doi.org/10.1038/gim.2017.97 (2018).
Ellingford, J. M. et al. Assessment of the incorporation of CNV surveillance into gene panel next-generation sequencing testing for inherited retinal diseases. Journal of medical genetics 55, 114–121, https://doi.org/10.1136/jmedgenet-2017-104791 (2018).
Martin-Merida, I. et al. Analysis of the PRPF31 Gene in Spanish Autosomal Dominant Retinitis Pigmentosa Patients: A Novel Genomic Rearrangement. Investigative ophthalmology & visual science 58, 1045–1053, https://doi.org/10.1167/iovs.16-20515 (2017).
Abu-Safieh, L. et al. A large deletion in the adRP gene PRPF31: evidence that haploinsufficiency is the cause of disease. Molecular vision 12, 384–388 (2006).
Saini, S., Robinson, P. N., Singh, J. R. & Vanita, V. A novel 7 bp deletion in PRPF31 associated with autosomal dominant retinitis pigmentosa with incomplete penetrance in an Indian family. Experimental eye research 104, 82–88, https://doi.org/10.1016/j.exer.2012.09.010 (2012).
Rose, A. M. & Bhattacharya, S. S. Variant haploinsufficiency and phenotypic non-penetrance in PRPF31-associated retinitis pigmentosa. Clinical genetics 90, 118–126, https://doi.org/10.1111/cge.12758 (2016).
Rose, A. M. et al. Transcriptional regulation of PRPF31 gene expression by MSR1 repeat elements causes incomplete penetrance in retinitis pigmentosa. Scientific reports 6, 19450, https://doi.org/10.1038/srep19450 (2016).
den Hollander, A. I. et al. Mutations in the CEP290 (NPHP6) gene are a frequent cause of Leber congenital amaurosis. American journal of human genetics 79, 556–561, https://doi.org/10.1086/507318 (2006).
Vache, C. et al. Usher syndrome type 2 caused by activation of an USH2A pseudoexon: implications for diagnosis and therapy. Human mutation 33, 104–108, https://doi.org/10.1002/humu.21634 (2012).
Slijkerman, R. W. et al. Antisense Oligonucleotide-based Splice Correction for USH2A-associated Retinal Degeneration Caused by a Frequent Deep-intronic Mutation. Molecular therapy. Nucleic acids 5, e381, https://doi.org/10.1038/mtna.2016.89 (2016).
Espinos, C., Millan, J. M., Beneyto, M. & Najera, C. Epidemiology of Usher syndrome in Valencia and Spain. Community genetics 1, 223–228, https://doi.org/10.1159/000016167 (1998).
Rivolta, C., Sweklo, E. A., Berson, E. L. & Dryja, T. P. Missense mutation in the USH2A gene: association with recessive retinitis pigmentosa without hearing loss. American journal of human genetics 66, 1975–1978, https://doi.org/10.1086/302926 (2000).
Pierrache, L. H. et al. Visual Prognosis in USH2A-Associated Retinitis Pigmentosa Is Worse for Patients with Usher Syndrome Type IIa Than for Those with Nonsyndromic Retinitis Pigmentosa. Ophthalmology 123, 1151–1160, https://doi.org/10.1016/j.ophtha.2016.01.021 (2016).
Bocquet, B. et al. Relative frequencies of inherited retinal dystrophies and optic neuropathies in Southern France: assessment of 21-year data management. Ophthalmic epidemiology 20, 13–25, https://doi.org/10.3109/09286586.2012.737890 (2013).
Yan, D. et al. Spectrum of DNA variants for non-syndromic deafness in a large cohort from multiple continents. Human genetics 135, 953–961, https://doi.org/10.1007/s00439-016-1697-z (2016).
Iwanami, M., Oshikawa, M., Nishida, T., Nakadomari, S. & Kato, S. High prevalence of mutations in the EYS gene in Japanese patients with autosomal recessive retinitis pigmentosa. Investigative ophthalmology & visual science 53, 1033–1040, https://doi.org/10.1167/iovs.11-9048 (2012).
Wright, C. F. et al. Genetic diagnosis of developmental disorders in the DDD study: a scalable analysis of genome-wide research data. Lancet 385, 1305–1314, https://doi.org/10.1016/S0140-6736(14)61705-0 (2015).
Alonso, R. et al. Babelomics 5.0: functional interpretation for new generations of genomic data. Nucleic acids research 43, W117–121, https://doi.org/10.1093/nar/gkv384 (2015).
Kozlowski, P. et al. Identification of 54 large deletions/duplications in TSC1 and TSC2 using MLPA, and genotype-phenotype correlations. Human genetics 121, 389–400, https://doi.org/10.1007/s00439-006-0308-9 (2007).
Choi, Y., Sims, G. E., Murphy, S., Miller, J. R. & Chan, A. P. Predicting the functional effect of amino acid substitutions and indels. PloS one 7, e46688, https://doi.org/10.1371/journal.pone.0046688 (2012).
Mathe, E. et al. Computational approaches for predicting the biological effect of p53 missense mutations: a comparison of three sequence analysis based methods. Nucleic acids research 34, 1317–1325, https://doi.org/10.1093/nar/gkj518 (2006).
Schwarz, J. M., Rodelsperger, C., Schuelke, M. & Seelow, D. MutationTaster evaluates disease-causing potential of sequence alterations. Nature methods 7, 575–576, https://doi.org/10.1038/nmeth0810-575 (2010).
Corton, M. et al. Exome sequencing of index patients with retinal dystrophies as a tool for molecular diagnosis. PloS one 8, e65574, https://doi.org/10.1371/journal.pone.0065574 (2013).
Banerjee, P. et al. TULP1 mutation in two extended Dominican kindreds with autosomal recessive retinitis pigmentosa. Nature genetics 18, 177–179, https://doi.org/10.1038/ng0298-177 (1998).
Roux, A. F. et al. Four-year follow-up of diagnostic service in USH1 patients. Investigative ophthalmology & visual science 52, 4063–4071, https://doi.org/10.1167/iovs.10-6869 (2011).
Paloma, E. et al. Novel homozygous mutation in the alpha subunit of the rod cGMP gated channel (CNGA1) in two Spanish sibs affected with autosomal recessive retinitis pigmentosa. Journal of medical genetics 39, E66 (2002).
Jaijo, T. et al. MYO7A mutation screening in Usher syndrome type I patients from diverse origins. Journal of medical genetics 44, e71, https://doi.org/10.1136/jmg.2006.045377 (2007).
Baux, D. et al. Enrichment of LOVD-USHbases with 152 USH2A genotypes defines an extensive mutational spectrum and highlights missense hotspots. Human mutation 35, 1179–1186, https://doi.org/10.1002/humu.22608 (2014).
Fujinami, K. et al. ABCA4 gene screening by next-generation sequencing in a British cohort. Investigative ophthalmology & visual science 54, 6662–6674, https://doi.org/10.1167/iovs.13-12570 (2013).
Aller, E. et al. Genetic analysis of 2299delG and C759F mutations (USH2A) in patients with visual and/or auditory impairments. European journal of human genetics: EJHG 12, 407–410, https://doi.org/10.1038/sj.ejhg.5201138 (2004).
Gerber, S. et al. The photoreceptor cell-specific nuclear receptor gene (PNR) accounts for retinitis pigmentosa in the Crypto-Jews from Portugal (Marranos), survivors from the Spanish Inquisition. Human genetics 107, 276–284 (2000).
Morimura, H., Saindelle-Ribeaudeau, F., Berson, E. L. & Dryja, T. P. Mutations in RGR, encoding a light-sensitive opsin homologue, in patients with retinitis pigmentosa. Nature genetics 23, 393–394, https://doi.org/10.1038/70496 (1999).
Peng, C., Rich, E. D. & Varnum, M. D. Achromatopsia-associated mutation in the human cone photoreceptor cyclic nucleotide-gated channel CNGB3 subunit alters the ligand sensitivity and pore properties of heteromeric channels. The Journal of biological chemistry 278, 34533–34540, https://doi.org/10.1074/jbc.M305102200 (2003).
Perez-Carro, R. et al. Panel-based NGS Reveals Novel Pathogenic Mutations in Autosomal Recessive Retinitis Pigmentosa. Scientific reports 6, 19531, https://doi.org/10.1038/srep19531 (2016).
Dryja, T. P., Rucinski, D. E., Chen, S. H. & Berson, E. L. Frequency of mutations in the gene encoding the alpha subunit of rod cGMP-phosphodiesterase in autosomal recessive retinitis pigmentosa. Investigative ophthalmology & visual science 40, 1859–1865 (1999).
Kohl, S. et al. Total colourblindness is caused by mutations in the gene encoding the alpha-subunit of the cone photoreceptor cGMP-gated cation channel. Nature genetics 19, 257–259, https://doi.org/10.1038/935 (1998).
Wissinger, B. et al. CNGA3 mutations in hereditary cone photoreceptor disorders. American journal of human genetics 69, 722–737, https://doi.org/10.1086/323613 (2001).
Mykytyn, K. et al. Identification of the gene (BBS1) most commonly involved in Bardet-Biedl syndrome, a complex human obesity syndrome. Nature genetics 31, 435–438, https://doi.org/10.1038/ng935 (2002).
Weisschuh, N. et al. Mutation Detection in Patients with Retinal Dystrophies Using Targeted Next Generation Sequencing. PloS one 11, e0145951, https://doi.org/10.1371/journal.pone.0145951 (2016).
Avila-Fernandez, A. et al. Identification of an RP1 prevalent founder mutation and related phenotype in Spanish patients with early-onset autosomal recessive retinitis. Ophthalmology 119, 2616–2621, https://doi.org/10.1016/j.ophtha.2012.06.033 (2012).
Lewis, R. A. et al. Genotype/Phenotype analysis of a photoreceptor-specific ATP-binding cassette transporter gene, ABCR, in Stargardt disease. American journal of human genetics 64, 422–434, https://doi.org/10.1086/302251 (1999).
Riveiro-Alvarez, R. et al. Frequency of ABCA4 mutations in 278 Spanish controls: an insight into the prevalence of autosomal recessive Stargardt disease. The British journal of ophthalmology 93, 1359–1364, https://doi.org/10.1136/bjo.2008.148155 (2009).
Vallespin, E. et al. Gene symbol: CRB1. Disease: Leber congenital amaurosis. Accession #Hm0540. Human genetics 118, 778 (2006).
Corton, M. et al. High frequency of CRB1 mutations as cause of Early-Onset Retinal Dystrophies in the Spanish population. Orphanet journal of rare diseases 8, 20, https://doi.org/10.1186/1750-1172-8-20 (2013).
Ben-Yosef, T. et al. A mutation of PCDH15 among Ashkenazi Jews with the type 1 Usher syndrome. The New England journal of medicine 348, 1664–1670, https://doi.org/10.1056/NEJMoa021502 (2003).
Acknowledgements
This work was supported by grants from the National Institute of Health Carlos III and the European Regional Development Fund (Institute of Health Carlos III/ISCIII) (CP10/00572, PI13/02621 and RD16/0008/0027 to JRE, PI17/01413 to CI, and a Research Intensification Contract to ALdM); the Basque Government’s Industry Department (SAIOTEK: SAIO11-PE11BN002; and SAIO12-PC12BN001 to JRE), a grant from the Mutua Madrileña Foundation and support from the Retinitis Pigmentosa Patients of Gipuzkoa Foundation (BEGISARE). JR-E is a Miguel Servet II Fellow, National Institute of Health Carlos III (ISCIII). MEI was supported by grants from the Basque Government’s Department of Education (DEDUC14/309). OB is supported by funding from the Retinitis Pigmentosa Patients of Gipuzkoa Foundation (BEGISARE) and a grant from the Mutua Madrileña Foundation. AA was supported by grants from the Fundación Jesús de Gangoiti Barrera and from the Basque Government’s Departments of Industry and Education (SAIOTEK-11BN002/PC12BN001/DEPLC13/002). CI is partially supported by a Research Intensification Contract (INTBIO15/001). The authors are grateful to Xabier Elcoroaristizabal and Marta Fernández-Mercado for their helpful advice on developing the base-calling setup. Maribel Gómez; Naiara Telletxea and Nahikari Pastoriza at the Basque Biobank for isolating DNA samples; and Dr. Carmen Ayuso for kindly providing control samples. We also give special thanks to all patients with IRD and their families involved in the study.
Author information
Authors and Affiliations
Contributions
M.E.-I. analysed most data and interpreted the results, generated all figures and drafted the manuscript. A.A. analysed part of the data and interpreted the results O.B. analysed part of the data and interpreted the results. G.G.A. selected a group of patients and collected the clinical data. M.G. selected a group of patients and collected the clinical data. A.L.d.M. interpreted the results and revised the manuscript. C.I. selected most patients collected the clinical data and obtained funding. J.R.-E. planned the experiments, interpreted the results, drafted the manuscript and obtained funding. All authors revised and approved the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ezquerra-Inchausti, M., Anasagasti, A., Barandika, O. et al. A new approach based on targeted pooled DNA sequencing identifies novel mutations in patients with Inherited Retinal Dystrophies. Sci Rep 8, 15457 (2018). https://doi.org/10.1038/s41598-018-33810-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33810-3
Keywords
This article is cited by
-
Retinitis Pigmentosa Due to Rp1 Biallelic Variants
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.