Abstract
The obesity paradox has been described in several observational cohorts and meta-analysis. However, evidence of the intentionality of weight loss in all-cause deaths and major cardiovascular events (MACE) in prospective cohorts is unclear. We analysed whether involuntary weight loss is associated with increased cardiovascular events and mortality. In a systematic review, we searched multiple electronic databases for observational studies published up to October 2016. Studies reporting risk estimates for unintentional weight loss compared with stable weight in MACE and mortality were included. Fifteen studies met the selection criteria, with a total of 178,644 participants. For unintentional weight loss, we found adjusted risk ratios (RRs) with confidence intervals (CIs) of 1.38 (95% CI: 1.23, 1.53) and 1.17 (95% CI: 0.98, 1.37) for all-cause mortality and MACE, respectively. Participants with comorbidities, overweight and obese populations, and older adults yielded RRs (95% CI) of 1.49 (1.30, 1.68), 1.11 (1.04, 1.18), and 1.81 (1.59, 2.03), respectively. Unintentional weight loss had a significant impact on all-cause mortality. We found no protective effect of being overweight or obese for unintentional weight loss and MACE.
Similar content being viewed by others
Introduction
The prevalence of obesity is increasing in most countries1, and has led to a global impact as it has been associated with all-cause mortality2, onset of serious diseases such as diabetes3 and cancer4,5, and increased cardiovascular risk6. There is continued interest in ideal weight ranges among epidemiological studies. The “U-” or “J-shaped” curves found in analyses of large cohorts indicate extremes of body mass index (BMI), underweight and obesity, associated with increased mortality in adult populations, regardless of sex or race7,8,9,10.
Randomized controlled trials have shown that intentional weight loss decreases mortality11,12,13; conversely, observational studies report that being overweight might be protective for some outcomes14. A higher BMI seems to be beneficial, especially in certain circumstances or populations, such as among older adults15,16 and patients with heart failure17, chronic obstructive pulmonary disease18, and chronic kidney disease19,20. Similarly, although the benefits of weight loss for decreasing some cardiovascular risk factors (blood pressure, glucose, and lipid levels) have already been established21,22,23,24, several observational studies have shown that weight reduction is associated with increased mortality from all causes and from cardiovascular disease25,26,27,28.
However, it has been noted that the variable “intentionality of weight loss” has not been adequately explored in most cohorts, especially with respect to methodological aspects26,29,30,31,32,33,34, which can possibly lead to a false positive association of weight loss and these adverse consequences. Unintentional weight loss is the involuntary decline in total body weight over time and it is mostly caused by malignant diseases, chronic organ diseases, drug-induced weight-loss or psychological disorders but up to one quarter of all cases have no identifiable cause, despite extensive investigation since its pathophysiology is poorly understood35.
Based on this gap in the analytical evidence regarding the intentionality of weight loss and its association with MACE and mortality, we conducted a systematic evaluation of current observational studies to estimate the increased risk of MACE or death linked with unintentional weight loss in general populations.
Methods
Search strategy
We performed a systematic electronic search in the Medline/PubMed, Web of Science, SciELO, and LILACS databases of studies published up to October 2016 to identify articles that studied the effect of unintentional weight loss on MACE and all-cause mortality. To perform a thorough search of the databases, the following Medical Subject Heading (MeSH) terms were used: prospective study, observational study, cohort study, follow-up study, body mass index, body weight change, stroke, myocardial infarction, acute coronary syndrome, coronary disease, incidence, and death. Full search strategies and keywords are summarized in the Supplementary Method. We used a pre-defined protocol, in accordance with the standards of quality for reporting Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines36.
Eligibility criteria
The inclusion criteria were: English language cohort studies; studies conducted with adult populations; studies that reported BMI and weight loss as numeric variables or percentages; studies that distinguished intentional from unintentional weight loss; studies that reported risk estimates for MACE (stroke, myocardial infarction, acute coronary syndrome, and cardiovascular death) or/and all-cause mortality. The intentionality of weight loss was described in the studies; unintentional weight loss was defined as any weight loss in the absence of self-reported action to try to lose weight, which may include diet, physical activity, use of medications, or by medical recommendation.
The exclusion criteria were letters, abstracts, conference proceedings, clinical trials, cluster trials, and randomized controlled trials studies; studies involving children and animals; studies conducted among bariatric surgery populations; cohorts that included only populations with poor health status at baseline (cancer, heart failure, diabetes, chronic kidney disease, or other diseases). We excluded randomized controlled trials with weight loss interventions because we assumed that all participants included in those trials intended to lose weight.
After removal of duplicates, two authors independently reviewed each title and abstract to determine whether the study met the inclusion and exclusion criteria. Disagreements about any selected items were resolved through discussions and by a third author if needed. After the initial screening, articles were chosen based on their complete text. Reference lists of the selected articles were searched manually for additional publications. The authors were contacted directly for any additional information and/or unpublished studies. If results from a single study were reported more than once, the study with the more complete set of data was included in the analysis.
Quality assessment
To evaluate the quality of studies, we customized the Newcastle-Ottawa Scale for cohort studies37. For “Selection” criteria, one star was given for each of the following items: good representativeness of the exposed cohort (general population, whole population of an area or a representative sample, non-hospitalized subjects), good representativeness of the non-exposed cohort (people with stable weight within the same cohort), at least two measures of BMI during follow-up, and participants without comorbidities at baseline. For “Comparability”, one star was given for studies that adjusted for smoking (or if this population was excluded) and another star for those that used a threshold for significant weight loss of up to a 3% or 4-kilogram change38. For “Outcome”, one star was given for each of the following: studies with at least three years of valid follow-up, studies with no more than 10% of participant baseline information missing, and studies in which outcomes were registered in national records. A total of nine stars could be achieved for each study. Two reviewers independently assessed the quality of the manuscripts. Quality was assigned as suboptimal for studies with less than four stars (Supplementary Table S1).
Data extraction and analysis
Two authors independently performed extraction of data from the articles through use of a data collection form, which was designed prior to the database searches. The main study and participant characteristics recorded were first author’s last name; year of publication; name of the cohort; geographic origin; population size; participants’ sex, age and comorbidities (diabetes, cardiovascular disease, cancer, hypertension); smoking status; alcohol intake; BMI; weight loss amount (in pounds, kilograms, or percentage); whether change in weight was reported or measured; time of follow-up; reference group; hazard ratio (HR) or relative risk (RR) and corresponding 95% confidence interval (CI) from the most fully adjusted multi-variable model; adjustment factors as potential confounders; and quality score.
Statistical analysis
Study HRs or RRs were pooled and a random effects model was used to summarize the results, as a conservative approach. Heterogeneity among studies was measured using the I2 statistic. We focused on two possible outcomes: all-cause mortality and MACE, which included cardiovascular mortality or cardiovascular events (stroke, myocardial infarction, and acute coronary syndrome). Subgroup analysis was performed to compare studies by age, sex, initial BMI, and health status at baseline. Results are shown in the form of forest plots. To evaluate the effects of time of follow-up, initial BMI, and all-cause mortality risk, we conducted meta-regression analysis. A funnel plot evaluated publication bias (Supplementary Figure S1). All analyses were carried out using Stata version 14.0 (StataCorp LP, College Station, TX, USA).
Results
Study selection
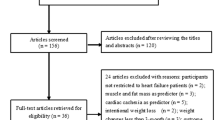
A total of 22 747 references were identified via electronic searches. After de-duplication and review of 11 more articles from a manual search of bibliographic ref.28, studies were selected for full-text reading, and 15 were included in the systematic review. Our inter-rater agreement between reviewers for inclusion criteria was k = 0,69. The reasons for excluding studies are outlined in Fig. 1.
Study characteristics
Table 1 shows a summary of characteristics of the studies and populations eligible for inclusion in the systematic review. Nine studies were conducted in the United States15,39,40,41,42,43,44,45,46, one in Australia47, one in Finland48, one in Israel25, one in the Netherlands49, one in Norway50, and one in the United Kingdom51. The sample comprised 178 644 participants with reported intentionality of weight loss, 50.3% women, and mean age ranging from 42.2 to 75.3 years. Unintentional weight loss was reported in 24 995 participants. Follow-up time varied from two to 20 years. Five studies exclusively assessed men25,39,41,43,51, two exclusively assessed women40,45, and the remainder included individuals of both sexes. Regarding health status, most cohorts included some participants with comorbidities (diabetes, cardiovascular disease, cancer and hypertension), but four articles reported a complete absence of these diseases at baseline39,40,46,48.
Fourteen studies reported all-cause mortality outcomes15,25,39,40,41,42,43,44,45,47,48,49,50,51 and five studies reported MACE outcomes39,40,45,46,51. Five studies excluded participants who died within the first two years of follow-up25,39,40,44,46. Most studies assessed weight change retrospectively15,25,39,40,42,43,44,45,46,47,49,50; a few studies prospectively assessed weight change41,48,51 and some measured weight more than once rather than using self-reported measures25,41,43,46. There were no disagreement between reviewers in quality assessment scale and two studies did not attain a minimum of four points and were excluded from the meta-analysis41,47.
Four studies analysed 10 307 participants over 65 years old, which we considered in subgroup analysis of older adults15,42,43,51, with a total 3429 participants who had unintentional weight loss. Six studies39,40,44,45,48,51 analysed 113 503 participants with BMI ≥ 25 kg/m2 (obese/overweight subgroup); of these, 7757 participants had unintentional weight loss.
Baseline BMI and amount of weight loss differed among studies, and we compared the largest weight loss group with the reference group. One study used BMI gain of 1 kg/m2 with intention to lose weight as the reference set (RR 1.0)50. All the others used “weight stable, with no intention to lose weight” as the reference category for comparison, although some levels of weight change considered non-significant by study authors could be found among some “weight stable” populations.
Unintentional weight loss and MACE
Unintentional weight loss was not associated with increased risk of MACE, with RR = 1.17 (95% CI: 0.98, 1.37); I2 = 68%; P = 0.005 (Fig. 2). There was heterogeneity across these studies. Subgroup analysis by sex, presence of comorbidities, and overweight/obese population showed no risk (see the Supplementary Figs S2–S4). The small number of studies was insufficient to investigate associations in the older adult subgroup with MACE.
Unintentional weight loss and all-cause mortality
Unintentional weight loss was associated with significant risk of death from any cause, RR = 1.38 (95% CI: 1.23, 1.53); I2 = 71.7%; P < 0.001 (Fig. 3), and this risk was even greater among older participants with RR = 1.81 (95% CI: 1.59, 2.03); I2 = 0.0%; P = 0.69 (Fig. 4). Subgroup analysis by sex is shown in the Supplementary Fig. S5. When we looked only at overweight and obese populations at baseline, we also found the presence of mortality risk with RR = 1.11 (95% CI: 1.04, 1.18); I2 = 0.0%; P = 0.61 (Fig. 5). For participants with no comorbidities, there was no association between unintentional weight loss and mortality, RR = 1.08 (95% CI: 0.95, 1.20); I2 = 0.0%; P = 0.58 (Fig. 6).
Meta-regression for time of follow-up showed that the longer the follow-up, the lower the risk of death due to unintentional weight loss; these data were statistically significant (P = 0.001) (Fig. 7). Meta-regression for initial BMI showed that the greater the initial BMI, the lower the risk of death if unintentional weight loss occurred (P = 0.018) (Fig. 8).
Dose–response meta-analysis was not feasible because studies reported weight loss differently, as percentage, change in BMI, or change in weight. Even after contacting the authors, no variations in participants height used to calculate BMI were reported, nor were estimates of the different categories of baseline BMI, weight loss amount or reference groups.
Discussion
This systematic review and meta-analysis showed that unintentional weight loss was significantly associated with mortality risk in observational studies, also in overweight/obese population and especially in older adults. No protective association of unintentional weight loss was found for MACE in any of the groups studied.
Commonly, unintentional weight loss in observational studies is concomitant with smoking, which is an important risk factor for cardiovascular diseases52; however, confusion bias may be present in such studies53. Most studies controlled for smoking status to limit confounders, and this could lower the risk association between unintentional weight loss and both outcomes. The general association of unintentional weight loss with MACE remains unclear. Most previous studies that demonstrated this association were among older populations and within the context of frailty and disability54,55,56,57; we had insufficient data of older adults with unintentional weight loss and MACE outcomes in our review. We can suppose that people with unintentional weight loss died of non-cardiovascular causes, even in the subgroups of overweight and obese. One should use caution in interpreting the association of unintentional weight loss with MACE because of competing risk events, such as cancer mortality. Studies with MACE outcomes that used Cox proportional hazards analysis could bias the estimated rate ratios. This means that in long-term studies with seven to 20 years of follow-up, MACE could be masked by non-cardiovascular deaths that occurred before this outcome of interest58,59.
The association of unintentional weight loss and all-cause mortality has been published in several other studies most of them involving older subjects and populations with chronic diseases like cancer, respiratory diseases, renal dysfunction and heart failure60,61,62,63,64. However, evidence exploring the association of unintentional weight loss and all-cause mortality in obese population is lacking.
Concerning methodology, it has not been well established whether the effects of weight change depend on initial BMI65,66,67, final BMI (i.e., BMI after weight reduction)68, or on the magnitude of weight loss or gain69. Misclassification of weight loss category can occur if one adjusts for initial or final BMI70. Furthermore, according to recent research, if maximum BMI had been established in a weight history taken by recall rather than using baseline BMI to calculate weight loss, we may have found more obvious results in the overweight and obese subgroup, because participants in this population who had unintentional weight loss may have been misclassified into the normal weight group71.
It was also difficult to establish the actual short- and long-term effects of weight loss72,73 because follow-up times varied, and weight loss occurred during mid-life in some studies and later in life in others. In fact, long-term analysis can also be impaired by weight cycling74. Survival bias may be present in some cohorts included in this review, as suggested by sensitivity analysis according to time of follow-up. It is known that in observational studies, the method used to detect weight change is not always described; weight is not always measured, sometimes only reported; and weight changes before and after the follow-up period are sometimes masked75,76,77,78. Questionnaires used to identify weight loss attempts are sometimes subjective and differ between cohort studies. We found that in some studies, participants were asked about their intention to lose weight before weight loss occurred and in other studies, intention was queried only after weight had changed, and changes in lifestyle, diet, physical activity, or medication use were often not quantified. Defining unintentional weight loss is difficult and in studies where questions about diet and physical activity are absent, it is mostly deduced using self-reported surveys. Gregg44 found that participants who intended to lose weight had 24% lower mortality, regardless of whether they lost weight. That author generated the hypothesis that healthy life habits are protective, even when they do not result in reduced BMI. In addition, large intentional weight loss could mask concomitant unintentional losses in some conditions like diabetes. According to some authors, the best study design for evaluating the real impact of intentionality of weight loss is a randomized controlled trial11,12, especially because interventions to lose weight are not specified in observational studies and do not allow causal inference for estimating effects79. However, long-term clinical trials that access mortality and cardiovascular events, with large samples and groups adhering to prescribed dietary or exercise regimens, have time-bound challenges, and such trials are scarce. In addition, recruitment for intervention and control groups already implies intentional weight loss, leaving open the possibility for a lack of intentionality; this can be better analysed in observational studies.
Our review has some limitations that merit consideration. Most studies assessed weight change retrospectively, which could bring some information bias to their estimates. It is also known that measures of weight change do not distinguish between changes in lean or fat mass80. Declining muscle mass is associated with higher levels of cytokines and inflammatory markers81, and such endogenous inflammation also predicts a higher risk of mortality82. Considering other measures of adiposity in addition to BMI may help in the assessment of body compositional disorders83. The different amounts of body fat between men and women can also impair the combined analysis of these groups. Our inability to categorize different weight loss amounts for comparison may also weaken the overall results, owing to a lack of dose–response analysis, because there is no consensus on how much weight loss is clinically relevant in cohorts.
The strengths of our study include our very strict criteria for distinguishing healthy and unhealthy cohorts. A previous meta-analysis by Harrington84 accessed risk estimates of unintentional weight loss and all-cause mortality among both healthy and unhealthy populations up to 2008. They found higher overall mortality (RR = 1.27, 95% CI: 1.09, 1.47) for unintentional weight loss in the healthy group and no difference in mortality (RR = 1.16, 95% CI: 0.97, 1.38) in the unhealthy group, which is paradoxical. We chose to classify healthy cohorts as only when comorbidities were excluded; we also optimized a quality scale in order to exclude studies with possible information bias. It is known that participants who enter cohort studies with an underlying disease may have higher early mortality85,86. Although improvements in hypertension, dyslipidemia, and diabetes can be mediators by which weight loss affects vascular mortality87,88,89, we tried to clear our results of these possible confounders by providing stratified analysis of subgroups without comorbidities and by extracting HRs from the most fully adjusted models with possible intermediaries in the causal pathway. It could be hypothesized that our study results in over-adjusted analysis once we used RRs from the most fully adjusted models. However, we believe that the absence of risk for unintentional weight loss and overall mortality that we have showed in a healthy population, which is also controlled for smoking, yields more reliable data. We can speculate that the 11%, 49% and 81% higher risk-association of unintentional weight loss and all-cause mortality found in overweight and obese participants, those with comorbidities, and older adults, respectively, are not only a marker of higher morbidity but provide strong evidence for clinicians to take into consideration.
In conclusion, we found that participants with unintentional weight loss had no protective risk for MACE and significant increased risk for overall mortality. A lack of information on the intentionality of weight loss could explain part of the disagreements found among studies of weight loss, obesity, and mortality. Careful attention should be given to individuals with suspected unintended weight loss, particularly in overweight and obese, older adult, or unhealthy populations. Observational studies on weight loss with mortality or cardiovascular event outcomes should consider the intentionality factor in order to avoid important bias in the weight effect estimates of these major clinical events.
Data Availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
World Health Organization. Obesity: preventing and managing the global epidemic: report of a WHO consultation. World Health Organization Technical Report Series. 894, 1–253 (2000).
Di Angelantonio, E. et al. Global BMI Mortality Collaboration. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet. 388(10046), 776–86 (2016).
Wannamethee, S. G. & Shaper, A. G. Weight change and duration of overweight and obesity in the incidence of type 2 diabetes. Diabetes Care. 22(8), 1266–72 (1999).
Polednak, A. P. Estimating the number of US incident cancers attributable to obesity and the impact on temporal trends in incidence rates for obesity-related cancers. Cancer Detect Prev. 32(3), 190–99 (2008).
Hubert, H. B., Feinleib, M., McNamara, P. M. & Castelli, W. P. Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation. 67(5), 968–77 (1983).
Guh, D. P. et al. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 9, 88 (2009).
Bogers, R. P. et al. Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: a meta-analysis of 21 cohort studies including more than 300 000 persons. Arch Intern Med. 167(16), 1720–28 (2007).
Whitlock, G. et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet. 373(9669), 1083–96 (2009).
Flegal, K. M., Graubard, B. I., Williamson, D. F. & Gail, M. H. Excess deaths associated with underweight, overweight, and obesity. JAMA. 293(15), 1861–67 (2005).
Cooper, R. S. Which factors confound or modify the relationship between body weight and mortality? Int J Obes. 32, 47–51 (2008).
Kritchevsky, S. B. et al. Intentional weight loss and all-cause mortality: a meta-analysis of randomized clinical trials. PLOS ONE. 10(3), e0121993 (2015).
Shea, M. K. et al. The effect of randomization to weight loss on total mortality in older overweight and obese adults: the ADAPT Study. J Gerontol A Biol Sci Med Sci. 65(5), 519–25 (2010).
Sjostrom, L. Bariatric surgery and reduction in morbidity and mortality: experiences from the SOS study. Int J Obes. 32(Suppl 7), S93–97 (2008).
Flegal, K. M., Kit, B. K., Orpana, H. & Graubard, B. I. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. JAMA. 309(1), 71–82 (2013).
Diehr, P. et al. Body mass index and mortality in nonsmoking older adults: the Cardiovascular Health Study. Am J Public Health. 88(4), 623–29 (1998).
Breeze, E., Clarke, R., Shipley, M. J., Marmot, M. G. & Fletcher, A. E. Cause-specific mortality in old age in relation to body mass index in middle age and in old age: follow-up of the Whitehall cohort of male civil servants. Int J Epidemiol. 35(1), 169–78 (2006).
Clark, A. L., Chyu, J. & Horwich, T. B. The obesity paradox in men versus women with systolic heart failure. Am J Cardiol. 110(1), 77–82 (2012).
Blum, A., Simsolo, C., Sirchan, R. & Haiek, S. “Obesity paradox” in chronic obstructive pulmonary disease. Isr Med Assoc J. 13(11), 672–75 (2011).
Vashistha, T. et al. Effect of age and dialysis vintage on obesity paradox in long-term hemodialysis patients. Am J Kidney Dis. 63(4), 612–22 (2014).
Park, J. et al. Obesity paradox in end-stage kidney disease patients. Prog Cardiovasc Dis. 56(4), 415–25 (2014).
Dattilo, A. M. & Kris-Etherton, P. M. Effects of weight reduction on blood lipids and lipoproteins: a meta-analysis. Am J Clin Nutr. 56(2), 320–28 (1992).
Sjostrom, L. et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 351(26), 2683–93 (2004).
Tuomilehto, J. et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 344(18), 1343–50 (2001).
Will, J. C., Williamson, D. F., Ford, E. S., Calle, E. E. & Thun, M. J. Intentional weight loss and 13-year diabetes incidence in overweight adults. Am J Public Health. 92(8), 1245–48 (2002).
Yaari, S. & Goldbourt, U. Voluntary and involuntary weight loss: associations with long term mortality in 9,228 middle-aged and elderly men. Am J Epidemiol. 148(6), 546–55 (1998).
Lee, I. M. & Paffenbarger, R. S. Jr. Change in body weight and longevity. JAMA. 268(15), 2045–49 (1992).
Astrup, A. Weight loss and increased mortality: epidemiologists blinded by observations? Obesity reviews. 4(1), 1–2 (2003).
Andres., R., Muller, D. C. & Sorkin, J. D. Long-term effects of change in body weight on all-cause mortality. A review. Ann Intern Med. 119(7 Pt 2), 737–43 (1993).
Elliott, A. M., Aucott, L. S., Hannaford, P. C. & Smith, W. C. Weight change in adult life and health outcomes. Obes Res. 13(10), 1784–92 (2005).
Chou, W. T. et al. Impact of Weight Change Since Age 20 and Cardiovascular Disease Mortality Risk - The Ohsaki Cohort Study. Circ J. 77(3), 679–86 (2013).
Bowman, T. S., Kurth, T., Sesso, H. D., Manson, J. E. & Gaziano, J. M. Eight-year change in body mass index and subsequent risk of cardiovascular disease among healthy non-smoking men. Prev Med. 45(6), 436–41 (2007).
Amador, L. F., Al Snih, S., Markides, K. S. & Goodwin, J. S. Weight change and mortality among older Mexican Americans. Aging Clin Exp Res. 18(3), 196–204 (2006).
Karahalios, A. et al. Change in Body Size and Mortality: Results from the Melbourne Collaborative Cohort Study. PLOS ONE. 9(7), e99672 (2014).
Higgins, M., D’Agostino, R., Kannel, W., Cobb, J. & Pinsky, J. Benefits and adverse effects of weight loss. Observations from the Framingham Study. Ann Intern Med. 119(7 Pt 2), 758–63 (1993).
Alibhai, S. M. H., Greenwood, C. & Payette, H. An Approach to the Management of Unintentional Weight Loss In Elderly People. CMAJ. 172(6), 773–780 (2005).
Stroup, D. F. et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 283(15), 2008–12 (2000).
Wells, G.A. et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa, Canada: Department of Epidemiology and Community Medicine, University of Ottawa (2009).
Stevens, J., Truesdale, K. P., McClain, J. E. & Cai, J. The definition of weight maintenance. Int J Obes. 30(3), 391–99 (2006).
Williamson, D. F. et al. Prospective study of intentional weight loss and mortality in overweight white men aged 40–64 years. Am J Epidemiol. 149(6), 491–503 (1999).
Williamson, D. F. et al. Prospective study of intentional weight loss and mortality in never-smoking overweight US white women aged 40–64 years. Am J Epidemiol. 141(12), 1128–41 (1995).
Wallace, J. I., Schwartz, R. S., LaCroix, A. Z., Uhlmann, R. F. & Pearlman, R. A. Involuntary weight loss in older outpatients: incidence and clinical significance. J Am Geriatr Soc. 43(4), 329–37 (1995).
Locher, J. L. et al. Body mass index, weight loss, and mortality in community-dwelling older adults. J Gerontol A Biol Sci Med Sci. 62(12), 1389–92 (2007).
Lee, C. G. et al. Mortality risk in older men associated with changes in weight, lean mass, and fat mass. J Am Geriatr Soc. 59(2), 233–40 (2011).
Gregg, E. W., Gerzoff, R. B., Thompson, T. J. & Williamson, D. F. Intentional weight loss and death in overweight and obese U.S. adults 35 years of age and older. Ann Intern Med. 138(5), 383–89 (2003).
French, S. A., Folsom, A. R., Jeffery, R. W. & Williamson, D. F. Prospective study of intentionality of weight loss and mortality in older women: the Iowa Women’s Health Study. Am J Epidemiol. 149(6), 504–14 (1999).
Stevens, J., Erber, E., Truesdale, K. P., Wang, C. H. & Cai, J. Long- and short-term weight change and incident coronary heart disease and ischemic stroke: the Atherosclerosis Risk in Communities Study. Am J Epidemiol. 178(2), 239–48 (2013).
Atlantis, E., Browning, C. & Kendig, H. Body mass index and unintentional weight change associated with all-cause mortality in older Australians: the Melbourne Longitudinal Studies on Healthy Ageing (MELSHA). Age Ageing. 39(5), 643–46 (2010).
Sorensen, T. I., Rissanen, A., Korkeila, M. & Kaprio, J. Intention to lose weight, weight changes, and 18-y mortality in overweight individuals without co-morbidities. PLOS Med. 2(6), e171 (2005).
Wijnhoven, H. A. H., van Zon, S. K. R., Twisk, J. W. R. & Visser, M. Attribution of causes of weight loss and weight gain to 3-year mortality in older adults: results from the Longitudinal Aging Study Amsterdam. J Gerontol A Biol Sci Med Sci. 69(10), 1236–43 (2014).
Wilsgaard, T., Jacobsen, B. K., Mathiesen, E. B. & Njølstad, I. Weight loss and mortality: a gender-specific analysis of the Tromsø study. Gend Med. 6(4), 575–86 (2009).
Wannamethee, S. G., Shaper, A. G. & Lennon, L. Reasons for intentional weight loss, unintentional weight loss, and mortality in older men. Arch Intern Med. 165(9), 1035–40 (2005).
Wannamethee, S. G., Shaper, A. G., Whincup, P. H. & Walker, M. Characteristics of older men who lose weight intentionally or unintentionally. Am J Epidemiol. 151(7), 667–75 (2000).
Lawlor, D. A., Hart, C. L., Hole, D. J. & Smith, G. D. Reverse causality and confounding and the associations of overweight and obesity with mortality. Obesity (Silver Spring). 14(12), 2294–304 (2006).
Nadruz, Jr. et al. Cardiovascular Dysfunction and Frailty Among Older Adults in the Community: The ARIC Study. J Gerontol A Biol Sci Med Sci. 72(7), 958–64 (2017).
Sergi, G. et al. Pre-frailty and risk of cardiovascular disease in elderly men and women: the Pro.V. A. study. J Am Coll Cardiol. 65(10), 976–83 (2015).
Ricci, N. A., Pessoa, G. S., Ferriolli, E., Dias, R. C. & Perracini, M. R. Frailty and cardiovascular risk in community-dwelling elderly: a population-based study. Clin Interv Aging. 9, 1677–85 (2014).
Yannakoulia, M., Ntanasi, E., Anastasiou, C. A. & Scarmeas, N. Frailty and nutrition: From epidemiological and clinical evidence to potential mechanisms. Metabolism. 68, 64–76 (2017).
Satagopan, J. M. et al. A note on competing risks in survival data analysis. Br J Cancer. 91(7), 1229–35 (2004).
Austin, P. C., Lee, D. S. & Fine, J. P. Introduction to the Analysis of Survival Data in the Presence of Competing Risks. Circulation. 133(6), 601–09 (2016).
Marton, K. I., Sox, H. C. Jr. & Krupp, J. R. Involuntary weight loss: diagnostic and prognostic significance. Ann Intern Med 95(5), 568–74 (1981).
Rabinovitz, M., Pitlik, S. D., Leifer, M., Garty, M. & Rosenfeld, J. B. Unintentional weight loss. A retrospective analysis of 154 cases. Arch Intern Med 146(1), 186–7 (1986).
Bilbao-Garay, J. et al. Assessing clinical probability of organic disease in patients with involuntary weight loss: a simple score. Eur J Intern Med. 13(4), 240–5 (2002).
Cabezas-Rodriguez, I. et al. Influence of Body Mass Index on the Association of Weight Changes with Mortality in Hemodialysis Patients. Clin J Am Soc Nephrol. 8(10), 1725–1733 (2013).
Rossignol, P. et al. Loss in Body Weight is an Independent Prognostic Factor for Mortality in Chronic Heart Failure: Insights From The Gissi-Hf And Val-Heft Trials. Eur J Heart Fail. 17(4), 424–33 (2015).
Somes, G. W., Kritchevsky, S. B., Shorr, R. I., Pahor, M. & Applegate, W. B. Body mass index, weight change, and death in older adults: the systolic hypertension in the elderly program. Am J Epidemiol. 156(2), 132–38 (2002).
Reynolds, M. W., Fredman, L., Langenberg, P. & Magaziner, J. Weight, weight change, mortality in a random sample of older community-dwelling women. J Am Geriatr Soc. 47(12), 1409–14 (1999).
Pamuk, E. R. et al. Weight loss and mortality in a national cohort of adults, 1971–1987. Am J Epidemiol. 136(6), 686–97 (1992).
Williamson, D. F. et al. Intentional weight loss and mortality among overweight individuals with diabetes. Diabetes Care. 23(10), 1499–504 (2000).
Goldstein, D. J. Beneficial health effects of modest weight loss. Int J Obes Relat Metab Disord. 16(6), 397–415 (1992).
Flegal, K. M., Keyl, P. M. & Nieto, F. J. Differential misclassification arising from nondifferential errors in exposure measurement. Am J Epidemiol. 134(10), 1233–44 (1991).
Yu, E. et al. Weight History and All-Cause and Cause-Specific Mortality in Three Prospective Cohort Studies. Ann Intern Med. 166(9), 613–20 (2017).
Veronese, N. et al. Body mass index as a predictor of all-cause mortality in nursing home residents during a 5-year follow-up. J American Med Dir Assoc. 14(1), 53–57 (2013).
Poobalan, A. S. et al. Long-term weight loss effects on all-cause mortality in overweight/obese populations. Obes Rev. 8(6), 503–13 (2007).
Stevens, V. L. et al. Weight cycling and mortality in a large prospective US study. Am J Epidemiol. 175(8), 785–92 (2012).
Yang, D., Fontaine, K. R., Wang, C. & Allison, D. B. Weight loss causes increased mortality: cons. Obes Rev. 4(1), 9–16 (2003).
Sorensen, T. I. Weight loss causes increased mortality: pros. Obes Rev. 4(1), 3–7 (2003).
Simonsen, M. K., Hundrup, Y. A., Obel, E. B., Gronbaek, M. & Heitmann, B. L. Intentional weight loss and mortality among initially healthy men and women. Nutr Rev. 66(7), 375–86 (2008).
Nilsson, P. M. Is weight loss beneficial for reduction of morbidity and mortality? What is the controversy about? Diabetes Care. 31(Suppl 2), S278–83 (2008).
Hernan, M. A. & Taubman, S. L. Does obesity shorten life? The importance of well-defined interventions to answer causal questions. Int J Obes. 32(Suppl 3), S8–14 (2008).
Lee, D. H. et al. Predicted lean body mass, fat mass, and all cause and cause specific mortality in men: prospective US cohort study. BMJ 362, k2575 (2018).
Visser, M. et al. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: the Health ABC Study. J Gerontol A Biol Sci Med Sci. 57(5), M326–32 (2002).
Kaptoge, S. et al. Emerging Risk Factors Collaboration, C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet. 375(9709), 132–40 (2010).
Pischon, T. et al. General and abdominal adiposity and risk of death in Europe. N Engl J Med. 359(20), 2105–20 (2008).
Harrington, M., Gibson, S. & Cottrell, R. C. A review and meta-analysis of the effect of weight loss on all-cause mortality risk. Nutr Res Rev. 22(1), 93–108 (2009).
Manson, J. E., Stampfer, M. J., Hennekens, C. H. & Willett, W. C. Body weight and longevity. A reassessment. JAMA. 257(3), 353–58 (1987).
Allison, D. B., Faith, M. S., Heo, M., Townsend-Butterworth, D. & Williamson, D. F. Meta-analysis of the effect of excluding early deaths on the estimated relationship between body mass index and mortality. Obes Res. 7(4), 342–54 (1999).
Noppa, H. Body weight change in relation to incidence of ischemic heart disease and change in risk factors for ischemic heart disease. Am J Epidemiol. 111(6), 693–704 (1980).
Wing, R. R., Jeffery, R. W. & Hellerstedt, W. L. A prospective study of effects of weight cycling on cardiovascular risk factors. Arch Intern Med. 155(13), 1416–22 (1995).
Olefsky, J., Reaven, G. M. & Farquhar, J. W. Effects of weight reduction on obesity. Studies of lipid and carbohydrate metabolism in normal and hyperlipoproteinemic subjects. J Clin Invest. 53(1), 64–76 (1974).
Acknowledgements
Source data for this systematic review were collected and analysed by the independent authors. All cost of the research was funded by the authors.
Author information
Authors and Affiliations
Contributions
Fernanda do Carmo De Stefani, MD MSc contributed to conception, design, literature review, acquisition, analysis and interpretation of data, drafting and reviewing the article. Priscila Saia Pietraroia, MD contributed to conception, design, literature review and acquisition of data, quality analysis and drafting the article. Miguel Morita Fernandes-Silva, PhD contributed to analysis, interpretation of data, review and to approve of the final version. José Faria-Neto, PhD contributed to analysis, interpretation of data, review and to approve of the final version. Cristina Pellegrino Baena, PhD contributed to conception, design, statistical analysis, interpretation of data and reviewing the article to be published.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
De Stefani, F.d.C., Pietraroia, P.S., Fernandes-Silva, M.M. et al. Observational Evidence for Unintentional Weight Loss in All-Cause Mortality and Major Cardiovascular Events: A Systematic Review and Meta-Analysis. Sci Rep 8, 15447 (2018). https://doi.org/10.1038/s41598-018-33563-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33563-z
Keywords
This article is cited by
-
Adulthood weight changes, body mass index in youth, genetic susceptibility and risk of atrial fibrillation: a population-based cohort study
BMC Medicine (2024)
-
Physical activity attenuated the negative effects of obesity on chronic disease-free life expectancy in rural adults
Journal of Public Health (2023)
-
Mortality risks of body mass index and energy intake trajectories in institutionalized elderly people: a retrospective cohort study
BMC Geriatrics (2022)
-
TOMM40 genetic variants associated with healthy aging and longevity: a systematic review
BMC Geriatrics (2022)
-
Relationship between weight loss and regular dental management of older adults residing in long-term care facilities: a 1-year multicenter longitudinal study
European Geriatric Medicine (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.